Abstract
Background:
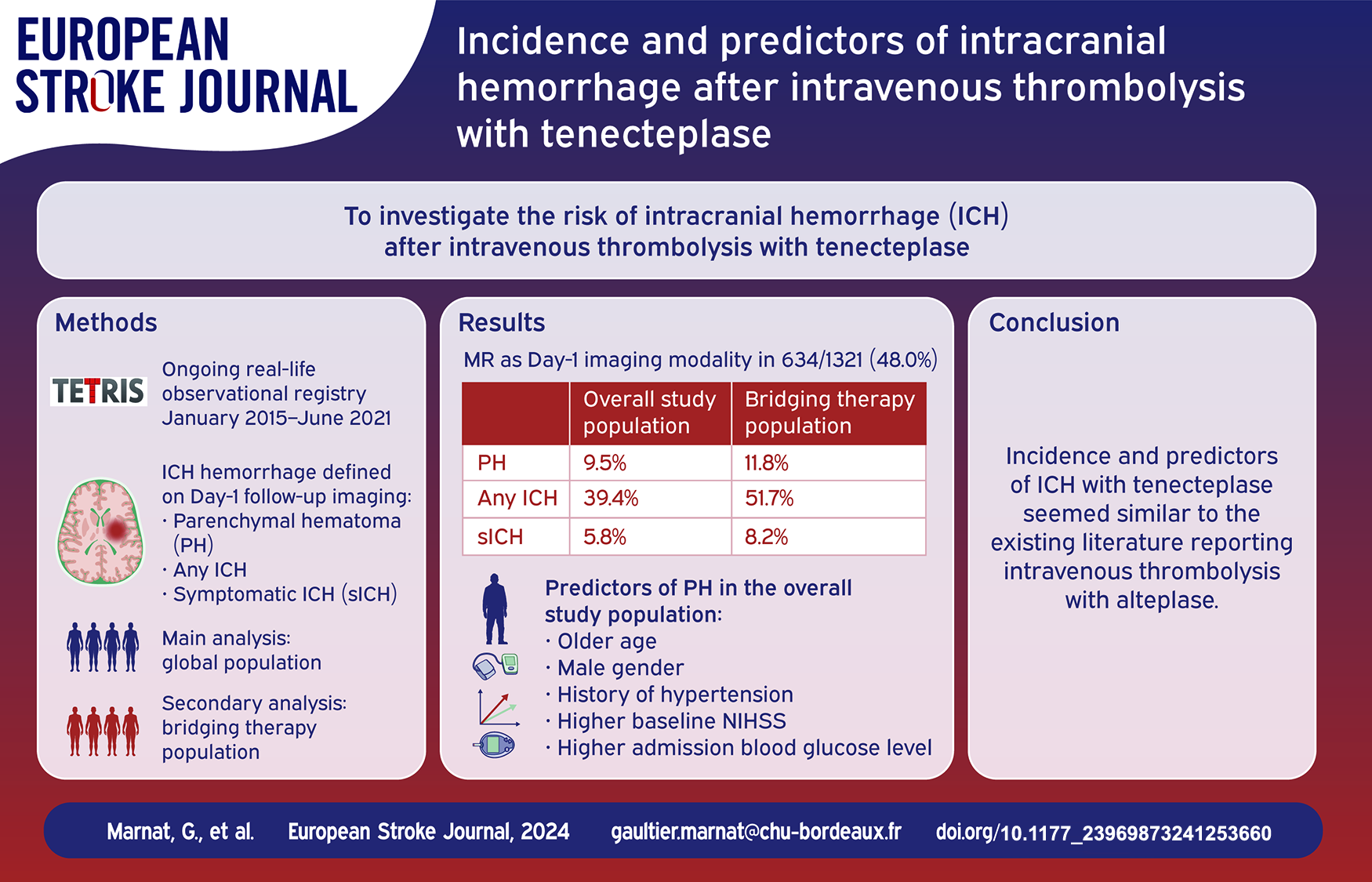
Despite its increasing use, there are limited data on the risk of intracranial hemorrhage (ICH) after intravenous thrombolysis with tenecteplase in the setting of acute ischemic stroke. Our aim was to investigate the incidence and predictors of ICH after tenecteplase administration.
Methods:
We reviewed data from the prospective ongoing multicenter TETRIS (Tenecteplase Treatment in Ischemic Stroke) registry. Patients with available day-1 imaging were included in this study. Clinical, imaging and biological variables were collected. Follow-up imaging performed 24 h after IVT was locally reviewed by senior neuroradiologists and neurologists. The incidence of parenchymal hematoma (PH) and any ICH were investigated. Potential predictors of PH and any ICH were assessed in multivariable logistic regressions. Subgroup analyses focusing on patients intended for endovascular treatment were performed.
Results:
PH and any ICH occurred in 126/1321 (incidence rate: 9.5%, 95% CI 8.1–11.2) and 521/1321 (39.4%, 95% CI 36.8–42.1) patients, respectively. Symptomatic ICH was observed in 77/1321 (5.8%; 95% CI 4.7–7.2). PH occurrence was significantly associated with poorer functional outcomes (p < 0.0001) and death (p < 0.0001) after 3 months. Older age (aOR = 1.03; 95% CI 1.01–1.05), male gender (aOR = 2.07; 95% CI 1.28–3.36), a history of hypertension (aOR = 2.08; 95% CI 1.19–3.62), a higher baseline NIHSS (aOR = 1.07; 95% CI 1.03–1.10) and higher admission blood glucose level (aOR = 1.12; 95% CI 1.05–1.19) were independently associated with PH occurrence. Similar associations were observed in the subgroup of patients intended for endovascular treatment.
Conclusion:
We quantified the incidence of ICH after IVT with tenecteplase in a real-life prospective registry and determined independent predictors of ICH. These findings allow to identify patients at high risk of ICH.
Introduction
Intracranial hemorrhage (ICH) is a potential complication after ischemic stroke, increased after intravenous thrombolysis (IVT).1,2 Various ICH subtypes associated with distinct clinical impairments are identified from minor petechial or subarachnoid hemorrhages up to major intra parenchymal hematoma with local mass effect and intracranial hypertension. 3 The negative impact of these cerebral hemorrhagic complications on functional outcome has been widely demonstrated. 4 Even minor petechial ICH have been reported to be associated with worse prognosis.5–7 Mechanisms involved in impairing neurological status include the direct role of ICH through mass effect, local cerebral parenchyma damages, and acute intracranial hypertension. Also, indirect consequences such as delays in the introduction of antithrombotic medication for secondary prevention, indirectly favoring the risk of recurrent stroke, and prolonged hospitalizations must be taken into account. Therefore, precisely appreciating the incidence and identifying factors associated with an increased risk of ICH is essential.
Although the benefits of IVT have been formally established in randomized trials, the decision to perform IVT (with or without additional endovascular treatment) in a given patient should always be weighed against the increased risk of ICH.1,8 ICH occurrence and its predictors have already been investigated in patients undergoing IVT with alteplase.1,9,10 However, over the recent years, tenecteplase has been increasingly used while its safety and efficacy were demonstrated through several trials.11–13 The incidence and predictors of ICH after IVT with tenecteplase remain poorly investigated in the literature. 14 Given the pharmacological differences between these two thrombolytics, investigating ICH risks associated with tenecteplase seems relevant.
Therefore, our aim was to estimate the incidence of ICH and to identify its potential predictors in a large cohort of patients treated with IVT with tenecteplase for acute ischemic stroke.
Methods
Data supporting this study are available from the corresponding author and TETRIS investigators upon reasonable request and after signing a data sharing agreement.
Study design and population
We conducted a retrospective analysis of the large real-life observational multicenter ongoing TETRIS (Tenecteplase Treatment in Ischemic Stroke) registry. Details on TETRIS have been published elsewhere. 15 Briefly, TETRIS is a multicenter registry based on prospective local database in five stroke centers, collecting consecutive patients receiving IVT with tenecteplase for acute ischemic stroke since May 2015. Patients were informed of their participation and offered the possibility to withdraw, in compliance with French law regarding retrospective studies of pseudonymized standard care data.
All consecutive patients receiving IVT with tenecteplase for acute ischemic stroke (with or without additional MT) and with an available day-1 follow-up imaging were included in the present study. Patients without available day-1 imaging were excluded.
Day-1 imaging was performed between 18 and 36 h after IVT administration, with MR or CT.
Treatment
In the participating centers, tenecteplase was routinely administered as first-line thrombolytic according to usual recommendations, in the absence of contraindication.16,17 Patients were given a single 0.25 mg/kg intravenous bolus of tenecteplase immediately after initial imaging. In cases of large vessel occlusions (LVO), an endovascular treatment was considered following standard guidelines for MT. 18 According to the intracranial recanalization status after endovascular treatment and to the proximal arterial occlusion etiology (e.g. tandem occlusion related to cervical atherosclerosis or dissection, underlying intracranial atherosclerosis, refractory occlusions), additional mechanical (angioplasty and/or stenting) and/or pharmacological (aspirin, GpIIbIIIa inhibitor, cangrelor, heparin) treatments could have been administered on a case-by-case basis.
Collected data, definitions, and outcomes
Baseline patient, imaging and timeframe data along with therapeutic and procedural characteristics were collected. Angiographic and imaging data were locally assessed by senior neuroradiologists. Baseline, angiographic and day-1 imaging data were locally assessed by trained neuroradiologists or neurologists with at least 5 years of experience. LVO were defined as proximal occlusions and included intracranial ICA, M1, proximal M2 (defined as the initial M2 segment, originating from the main middle cerebral artery bifurcation until the mid-height of the insula), basilar artery and intracranial vertebral artery occlusions. Medium vessel occlusions (MeVO) included distal M2 (defined as M2 occlusions occurring above the mid-height of the insula), M3, M4, A1, and A2 segments of the anterior cerebral artery and P1 and P2 segments of the posterior cerebral artery.
“Any ICH” was defined as any intracranial bleeding detected on day-1 imaging. ICH subtypes were defined according to the European Cooperative Acute Stroke Study classification including hemorrhagic infarction 1 (HI-1), hemorrhagic infarction 2 (HI-2), parenchymal hematoma 1 (PH-1), and parenchymal hematoma 2 (PH-2). 19 HI-1 was defined as scattered small petechial hemorrhage, without mass effect. HI-2 was defined as confluent petechial hemorrhage within the infarct territory, without mass effect. PH-1 was defined as hematoma in less than 30% of the infarcted area with some slight space occupying effect. PH-2 was defined as a hematoma occupying more than 30% of the infarcted area associated with substantial mass effect. Parenchymal hematoma (PH) aggregated the occurrence of any PH-1 or PH-2. We also considered “remote ICH,” which was defined as any intra intracerebral hemorrhage, either petechial or presenting as a hematoma, occurring outside the infarcted territory. Symptomatic ICH (sICH) was defined as an increase in NIHSS score by greater than or equal to 4 points related to an ICH of any subtype.
Favorable outcome was defined as a modified Rankin Scale (mRS) score of 0–2 after 3 months. Three-month mRS were collected by certified investigators during routinely scheduled visits or by trained research nurses during a standardized telephone interview.
In patients treated with MT, favorable and successful recanalization were respectively defined as a final modified Thrombolysis in Cerebral Infarction (mTICI) ⩾2b and ⩾2c. First Pass Effect was defined as mTICI ⩾2c after the first mechanical thrombectomy pass. Procedural complications occurring during the endovascular procedure were also recorded (arterial perforation, embolus in a new territory, puncture-related complications).
Statistical analysis
Qualitative variables were expressed as numbers (percentages) and compared using Chi-Squared test or Fisher’s exact test, as appropriate. Quantitative variables were expressed as mean ± standard deviation or median (interquartile range (IQR)) and compared using Student’s t-test or Mann-Whitney U test, as appropriate. Incidence rates for different definitions of ICH were calculated from raw numbers, and their 95% confidence intervals (95% CI) were estimated using Wilson’s method. Associations between clinical or radiological variables and ICH (dependent variable) were assessed with binary logistic regressions and expressed as crude and adjusted odds ratios (OR) with their 95% CI. Variables associated with ICH at p < 0.20 in univariable analysis were candidates for inclusion in multivariable logistic models, taking into account potential multicollinearity. Only variables which remained associated with ICH at p < 0.05 were retained in the final models (bidirectional stepwise variable selection). A subgroup analysis focusing on patients intended for endovascular treatment was conducted. All statistical tests were two-sided and statistical significance was set at p < 0.05. Statistical analysis was conducted with SAS 9.4 (SAS Institute, Cary, NC).
Results
Cohort description
Between January 2015 and June 2021, 1339 patients were included in the TETRIS registry. Among them, 18 (1.3%) were excluded because of no day-1 imaging was available to assess ICH. A total of 1321 patients were included in the analysis. Baseline and treatment characteristics and functional outcomes of the whole study population and of the subgroup of patients who were intended for endovascular treatment are presented in Supplemental Table 1. Mean age was 71.8 ± 15.9 years and 673 (50.9%) were female. Admission median NIHSS score and baseline ASPECTS were 12 (IQR: 5–18) and 8 (IQR: 7–9), respectively. Three hundred ninety-six (30.0%) patients were already under antiplatelet therapy at the time of ischemic stroke and 102 (7.7%) under anticoagulant treatment. Among patients with a known time of onset (1073 [81.2%]), median time from symptom onset to IVT was 160 min (IQR = 129–195). LVO and MeVO were identified in 903 (68.4%) patients and 191 (14.5%) patients respectively. Endovascular treatment was intended in 856 (64.8%). Among these, median time from onset to arterial puncture was 232 min (IQR = 194–277).
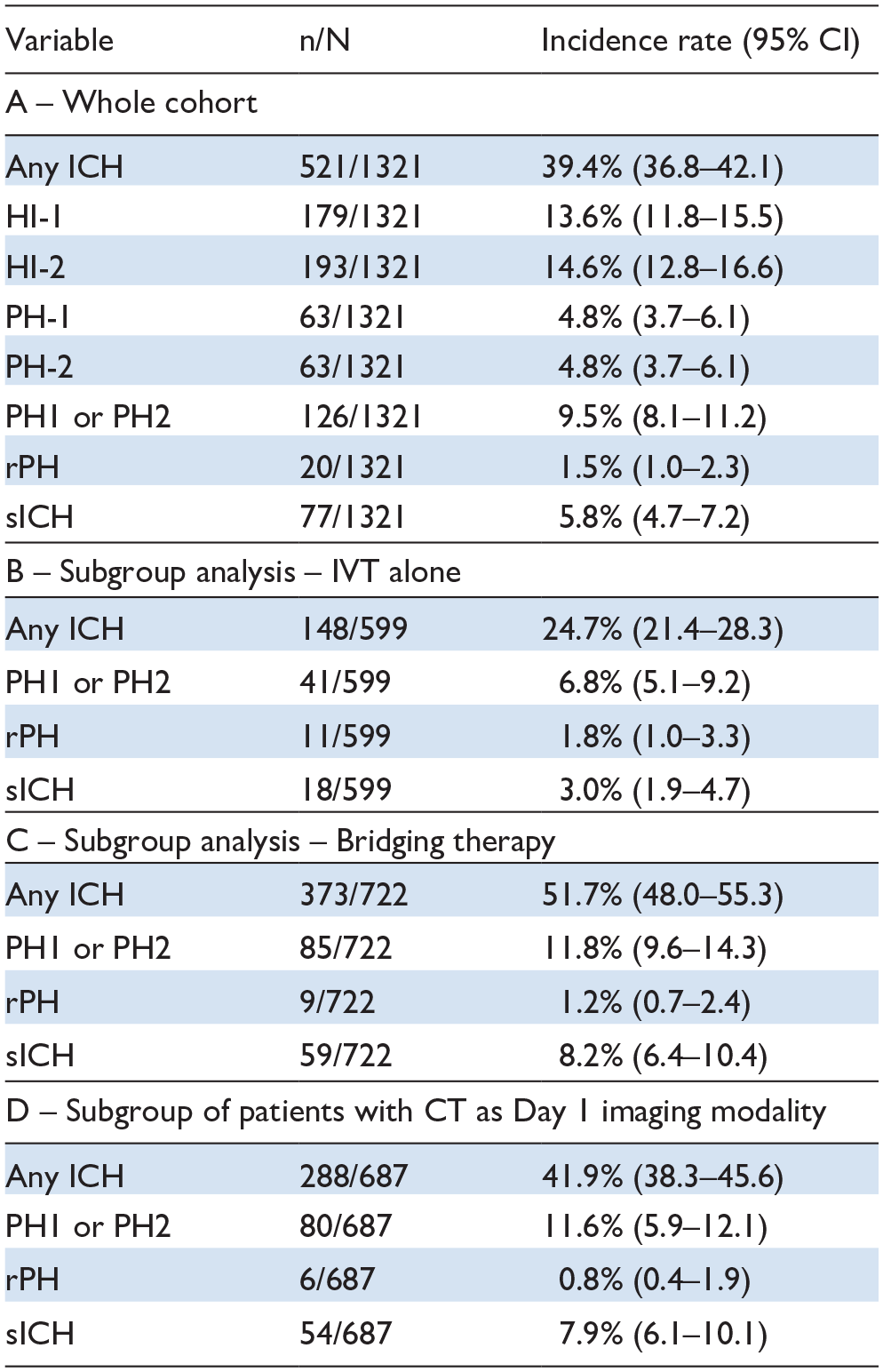
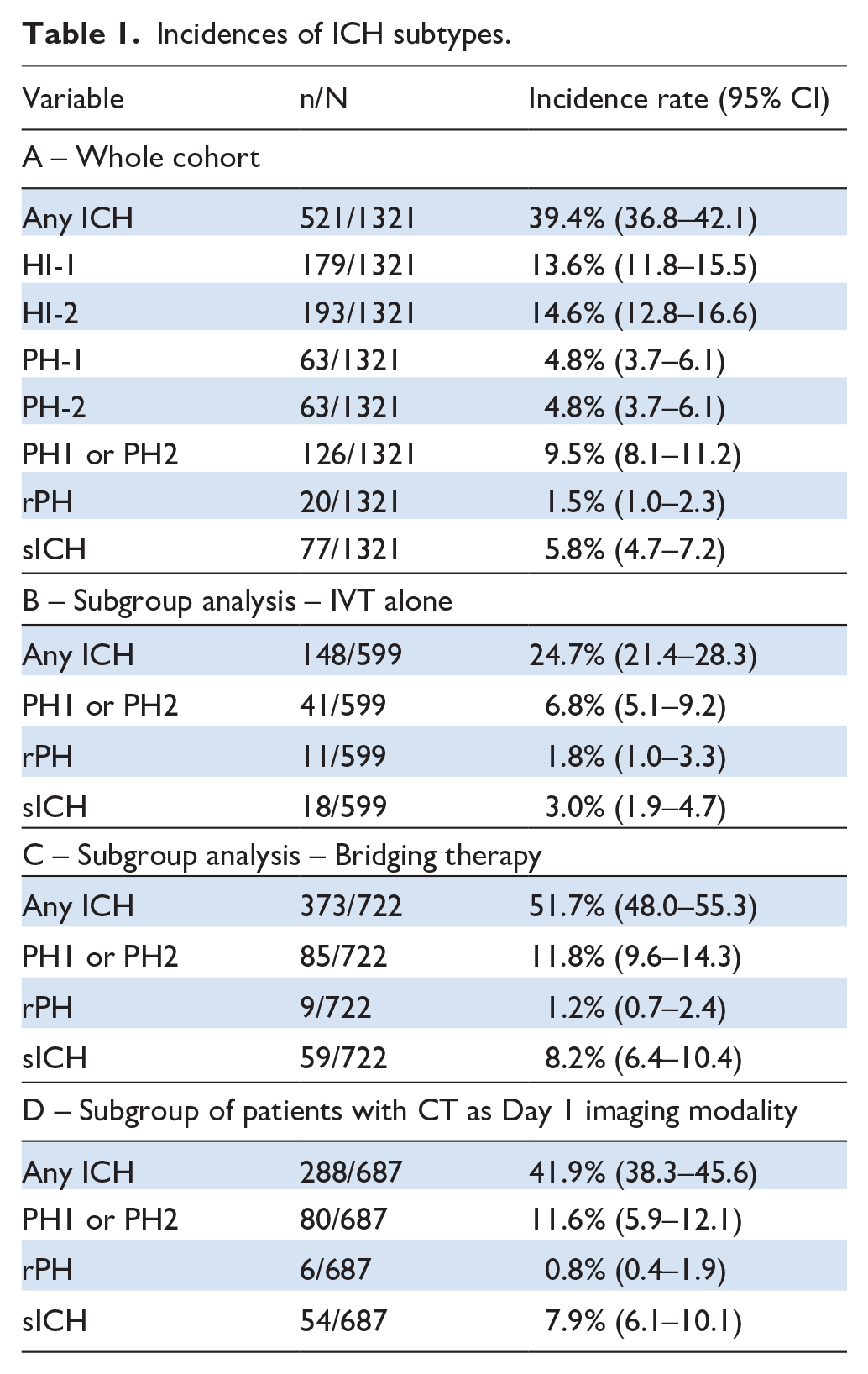
Day-1 imaging modality was MRI in 634 patients (48.0%) and CT in 687 (52.0%). ICH occurrence was as follow: any ICH in 521 (39.4%), PH in 126 (9.5%) and sICH in 77 (5.8%). Among patients with CT as day 1 imaging modality, details of ICH were: any ICH in 288/687 (41.9%), PH in 80/687 (11.6%) and sICH in 54/687 (7.9%).
In the overall study population, 679 (51.4%) patients presented a favorable functional outcome after 3 months. Mortality rate at 3 months was 15.4%. Stroke etiology was cardio-embolism in 638 patients (48.3%).
Incidence and predictors of PH
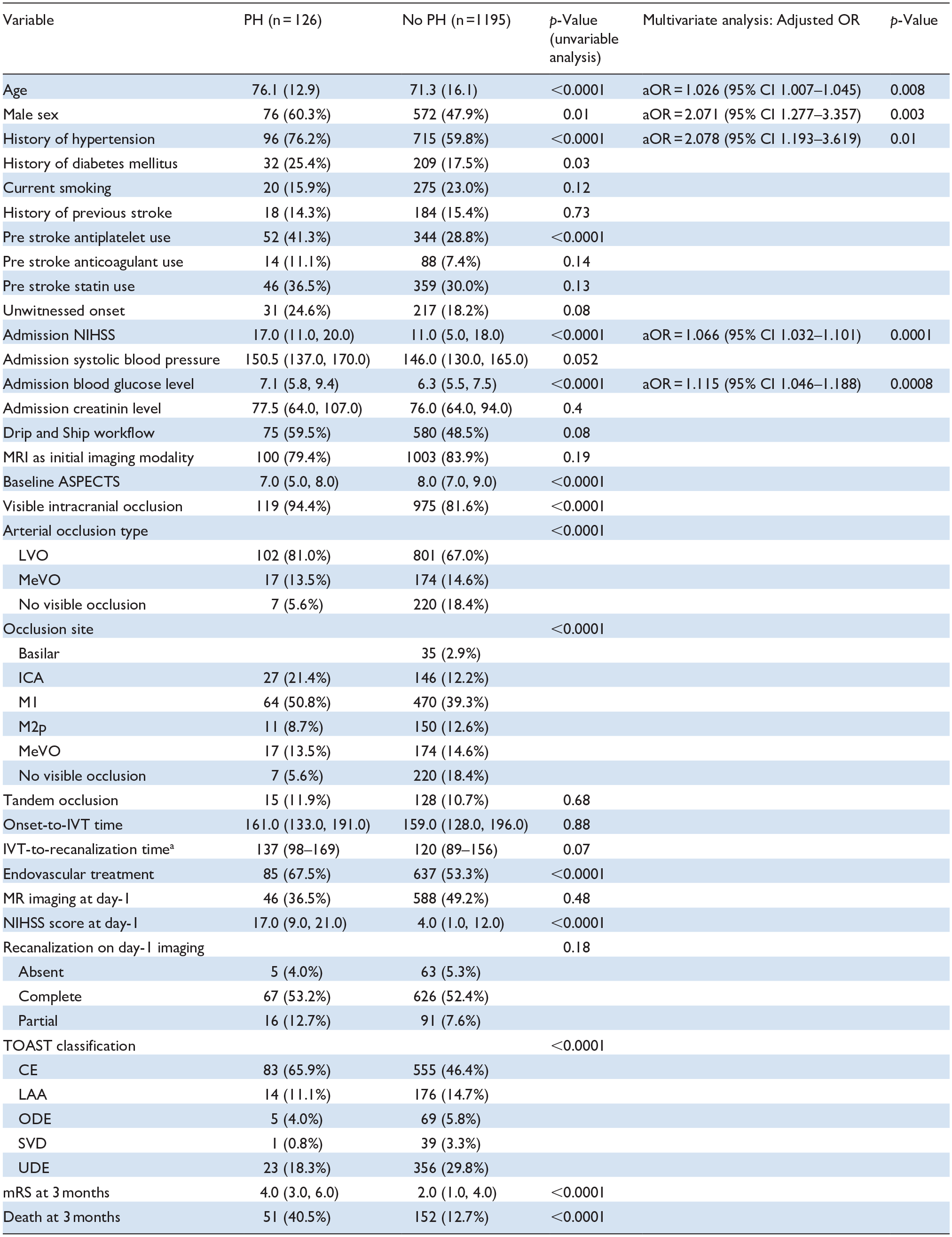
In the whole study population, PH was detected in 126 patients (incidence rate: 9.5%, 95% CI 8.1–11.2) on day-1 imaging (Table 1). PH occurrence was associated with significantly worse prognosis: a higher day-1 NIHSS (17 vs 4, p < 0.0001), a higher median 3-month mRS (4 vs 2, p < 0.0001) and an increased mortality rate (40.5% vs 12.7%, p < 0.0001) (see Table 2 and Supplemental Figures 1 and 2).
Incidences of ICH subtypes.
Predictors of PH in the whole study population.
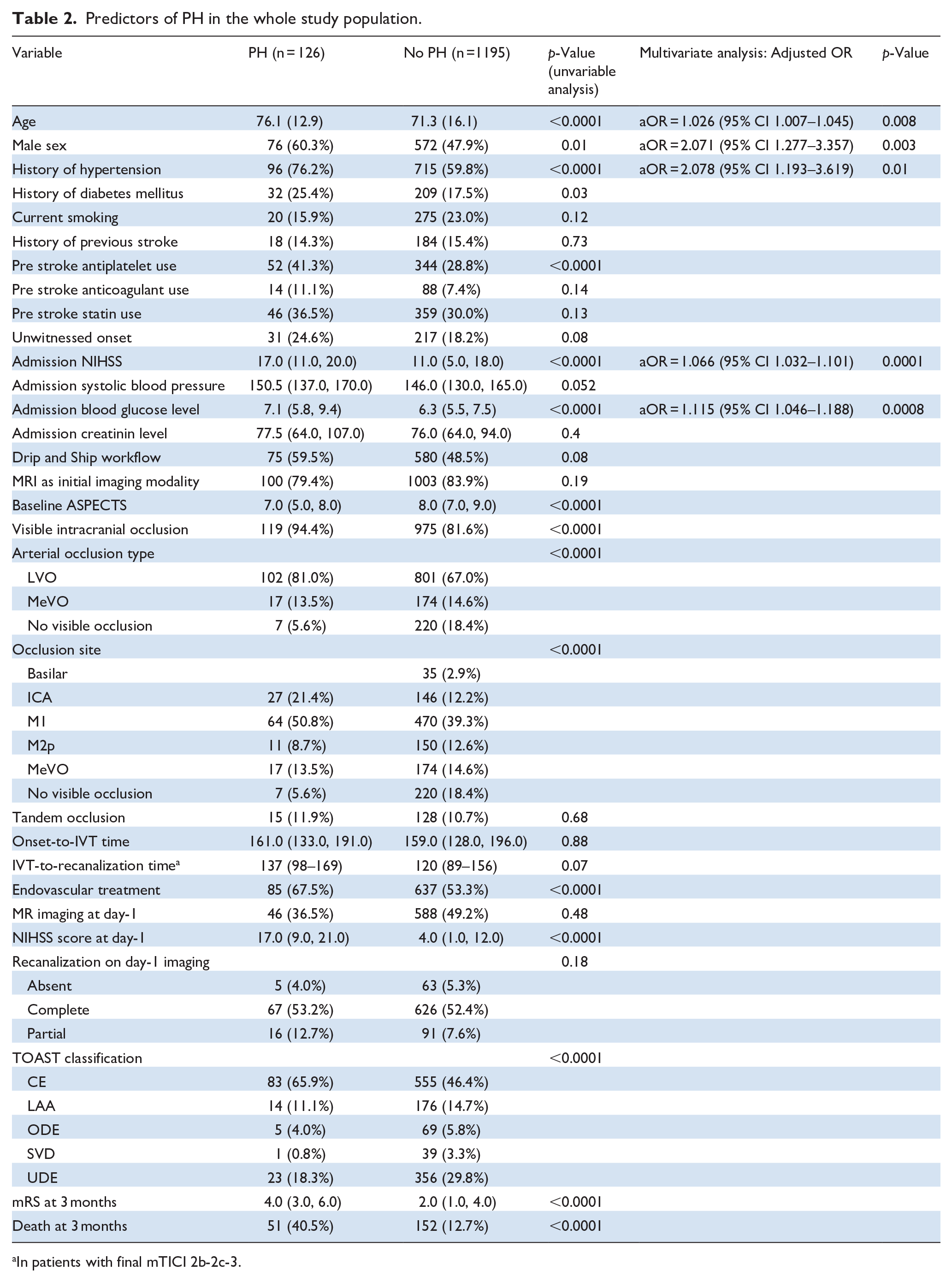
In patients with final mTICI 2b-2c-3.
In univariable analysis, patients with PH on day-1 imaging were older (76.1 vs 71.3, p < 0.0001), more frequently males (60.3% vs 47.9%, p = 0.01), presented more frequently a history of hypertension (76.2% vs 59.8%, p < 0.0001) and of diabetes mellitus (25.4% vs 17.5%, p = 0.03), were more frequently under prior antiplatelet therapy (41.3% vs 28.8%, p < 0.0001), had higher baseline NIHSS (17 vs 11, p < 0.0001), higher admission blood glucose (7.1 vs 6.3 mmol/L, p < 0.0001), lower baseline ASPECTS (7 vs 8, p < 0.0001), more frequently presented LVOs (81.0% vs 67.0%, p < 0.0001) requiring more often an endovascular treatment (67.5% vs 53.3%, p < 0.0001) and presented a higher rate of stroke from cardio-embolic etiology (65.9% vs 46.4%, p < 0.0001) (Table 2).
In multivariable analysis, older age (aOR = 1.03; 95% CI 1.01–1.05, per 1-year increase), male gender (aOR = 2.07; 95% CI 1.28–3.36), a history of hypertension (aOR = 2.08; 95% CI 1.19–3.62), a higher baseline NIHSS (aOR = 1.07; 95% CI 1.03–1.10, per 1-point increase) and higher admission blood glucose level (aOR = 1.12; 95% CI 1.05–1.19, per 1-mmol/L increase) remained independently associated with PH.
In the subgroup of patients intended for endovascular treatment (see Supplemental Table 2), PH occurred in 85/722 (11.8%; 95% CI 9.6–14.3). Similar predictors were identified (older age, male gender, history of high hypertension, higher baseline NIHSS and a higher admission blood glucose level) but an unknown time of onset was also independently associated with PH occurrence (aOR = 1.93; 95% CI 1.07–3.49).
Incidence and predictors of any ICH
Any ICH occurred in 521 patients (39.4%; 95% CI 36.8–42.1) in the whole cohort but in 148 (24.7%; 95% CI 21.4–28.3) of the IVT alone subgroup and in 373 (51.7%; 95% CI 48.0–55.3) of the IVT + Endovascular treatment subgroup (Table 1). ICH occurrence was associated with significantly worse prognosis: a higher day-1 NIHSS (11 vs 3, p < 0.0001), a higher median 3-month mRS (3 vs 2, p < 0.0001) and an increased mortality rate (23.0% vs 10.4%, p < 0.0001) (see Table 3 and Supplemental Figures 1 and 2).
Predictors of any ICH in the whole study population.
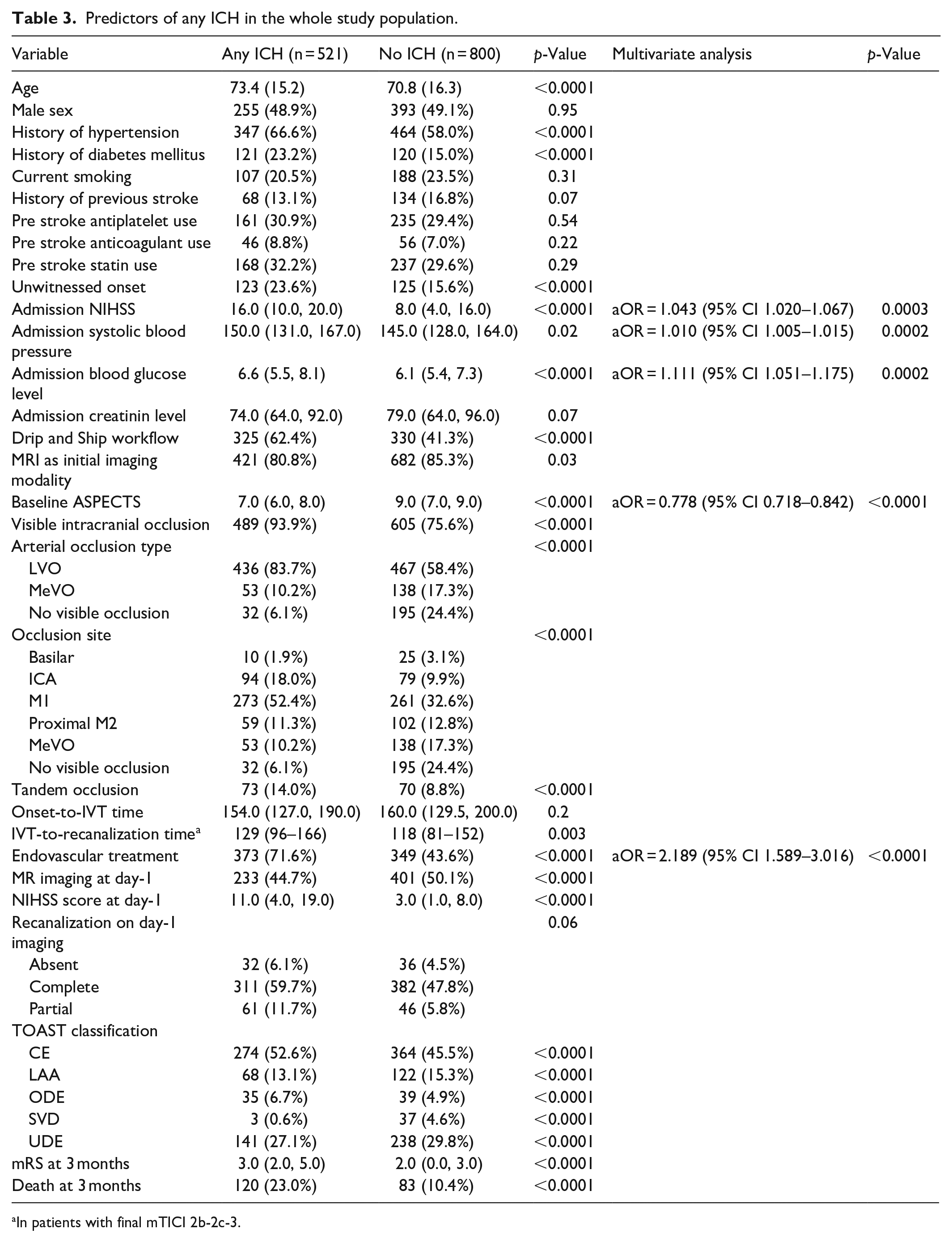
In patients with final mTICI 2b-2c-3.
In univariable analysis in the entire cohort (Table 2), older age (73.4% vs 70.8%; p < 0.0001), history of hypertension (66.6% vs 58.0%; p < 0.0001) and diabetes mellitus (23.2% vs 15.0%; p < 0.0001), unknown time of onset (23.6% vs 15.6%; p < 0.0001), higher admission NIHSS score (16 vs 8; p < 0.0001), higher admission blood glucose level (6.6 vs 6.1 mmol/L; p < 0.0001), higher admission systolic blood pressure (150 vs 145 mmHg; p = 0.02), admission in a primary stroke center (drip and ship workflow; p < 0.0001), MRI as initial imaging modality (p = 0.03), lower baseline ASPECTS (7 vs 9; p < 0.0001), LVO (83.7% vs 58.4%; p < 0.0001), M1 and intracranial ICA occlusions (respectively 52.4% vs 32.6% and 18.0% vs 9.9%; p < 0.0001), tandem occlusions (14.0% vs 8.8%; p < 0.0001) and endovascular treatment (71.6% vs 43.6%; p < 0.0001) were associated with increased risk of any ICH at day-1.
Multivariable analysis revealed a persisting significant association between any ICH occurrence and higher baseline NIHSS score (aOR = 1.04; 95% CI 1.02–1.07, per 1-point increase), higher admission systolic blood pressure (aOR = 1.01; 95% CI 1.01–1.02, per 1-mmH increase), higher admission blood glucose level (aOR = 1.11; 95% CI 1.05–1.18, per 1-mmol/L increase), lower baseline ASPECTS (aOR = 0.78; 95% CI 0.72–0.84, per 1-point increase) and endovascular treatment (aOR = 2.19; 95% CI 1.59–3.02).
In a subgroup analysis restricted to patients intended for endovascular treatment (see Supplemental Table 3), higher baseline NIHSS (aOR = 1.04; 95% CI 1.01–1.07, per 1-point increase), higher admission systolic blood pressure (aOR = 1.01; 95% CI 1.01–1.02), higher admission blood glucose level (aOR = 1.07; 95% CI 1.01–1.014) and lower baseline ASPECTS (aOR = 0.80; 95% CI 0.73–0.87, per 1-point increase) remained independently associated with any ICH occurrence.
Remote ICH
Remote ICH was detected in 20 patients (1.5%; 95% CI 1.0–2.3) in the whole study cohort (see Table 1). Given the rarity of this ICH subtype, no further analysis was conducted.
Discussion
In this analysis of a large real-life observational registry, we observed that PH after IVT with tenecteplase was detected in 9.5% of patients. Older age, male gender, history of hypertension, higher baseline NIHSS and blood glucose level were independent predictors of PH. Any ICH was encountered in 39.4% of the whole study cohort and in 51.7% of patients treated with IVT + MT. Similar independent risk factors were identified (higher baseline NIHSS, elevated admission systolic blood pressure, higher admission blood glucose level, lower baseline ASPECTS and patients treated with MT).
In this study, we decided to focus on imaging subtypes of ICH (PH, any ICH and remote ICH) irrespective of their clinical impact. Indeed, first, the negative impact of various ICH subtypes, either detected on CT or MRI at day-1, on functional outcome has already been demonstrated.5–7,20 Second, any ICH occurrence at the acute phase of an ischemic stroke, either symptomatic or not, might have immediate and meaningful therapeutic consequences (e.g. delay in the introduction of antithrombotic medication, extension of in-hospital stay, etc.).1,21 In addition, the very wide majority of the existing literature in the field focused on sICH, reinforcing the need for additional data on imaging-based definitions of ICH.
In our cohort, the incidence of ICH subtypes was slightly higher than in the recent literature. However, available data remain scarce to date. In the ACT-trial, the rate of any ICH with tenecteplase on day-1 imaging was 19.3%, without significant difference with the alteplase group (20.6%). Also, PH-1 and PH-2 were reported in 3.5% and 2.6% respectively of patients in the tenecteplase group, again without significant difference compared with those treated with alteplase (respective rates of PH-1 and PH-2: 2.6% and 2.4%). 12 In the recent TWIST trial, among patients with unknown time of stroke onset, authors reported odds of any ICH and PH-2 of respectively 11% and 2%. 22 Among patients treated with a bridging approach (IVT + MT) included in the EXTEND-TNK trial, 5.9% patients of the tenecteplase group experienced PH. 11 In their recent publication, the CERTAIN investigators focused only on sICH and observed a 1.8% rate after tenecteplase, which was significantly lower than in the alteplase group. 14 Several points might explain these differences. Importantly, in all of the studies mentioned above, day-1 imaging modality was CT. In our cohort, day-1 control imaging were MRI in nearly half of the study population (634/1321; 48.0%). Yet, MR has been associated with an increased likely of ICH detection, in particular in the setting of post ischemic stroke ICH. 23 MR ability to detect even small ICH or to overrate ICH probably accounts, at least partially, for these high rates. 24 Finally, our study population was extracted from a multicenter observational registry including real-life data from non-selected patients. In the participating centers, alteplase had been recently replaced with tenecteplase in the treatment of acute ischemic stroke eligible for IVT. 25 Consequently, patients were not specifically selected to receive tenecteplase. Moreover, it is important to mention that our results remain in line with previous real-life data of alteplase use.26,27
Interestingly, we observed similar predictors of ICH after tenecteplase administration than those previously described among literature after alteplase. Indeed, older age, hyperglycemia, history of hypertension, elevated systolic pressure at admission, higher initial NIHSS score and lower initial ASPECTS have already been demonstrated associated with either sICH or PH occurrence in patients treated with alteplase.10,28,29 As with alteplase, taking into account these factors and controlling them whenever possible may have important safety implications when considering IVT with tenecteplase. Regarding modifiable risk factors, blood pressure control and glycemia normalization are major targets that should be even more carefully controlled in patients with an increased risk of ICH. Potential additional antithrombotic therapies at the acute phase of the ischemic stroke should be carefully considered, according to an individualized benefit-risk ratio and under a close control of predictors of ICH. Remarkably, despite previous observations in patients treated with alteplase and despite a significant association in univariate analysis in our study, we did not found that pre stroke antiplatelet medication was significantly associated with an increased risk of ICH. 10 Despite an association with any ICH and PH occurrence in univariable analyses, we did not observe an independent association between these ICH subtypes and cardio-embolic etiology in multivariable analyses. We also observed that endovascular treatment was significantly associated with any ICH occurrence, which might not be in line with previous publications. Despite a rigorous statistical approach with multivariable analysis, we hypothesize that this association may be confounded by the combination of several features, including proximal occlusion, higher baseline NIHSS and lower baseline ASPECTS.
Several limitations should be acknowledged. First, this was a retrospective study and as such presented usual shortcomings associated with this study design. Then, although all images were locally systematically reviewed by trained neuroradiologists and/or neurologists, these data were not centrally reviewed by a core lab. Despite derived from a large multicenter registry, the statistical power of some analyses might have been limited by reduced patient sample in subgroup or by the rarity of specific events. Unmeasured confounders such as factors related to each participating centers’ protocols might also have influenced the results. The use of additional antithrombotic therapy such as per-thrombectomy antiplatelet therapy (cangrelor, aspirin) or heparin were relatively rare in our study population, limiting our ability to investigate their influence on ICH in the setting of prior IVT with tenecteplase. Subarachnoid hemorrhage occurrence was not reported because these data were not available in our study. Also, we chose to focus mainly on imaging definitions of ICH (PH and any ICH). Consequently, our results should be cautiously compared to the existing literature mostly reporting sICH.
Conclusion
In this large real-life study, we found that rates of PH and any ICH after IVT were 9.5% and 39.4%, respectively. In particular, older age, male gender, history of hypertension, higher baseline NIHSS and higher admission blood glucose levels were significantly associated with an increased risk of PH. These findings can help to identify patients presenting with an increased risk of ICH.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241253660 – Supplemental material for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase
Supplemental material, sj-docx-1-eso-10.1177_23969873241253660 for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase by Gaultier Marnat, Gaspard Gerschenfeld, Stephane Olindo, Igor Sibon, Pierre Seners, Frederic Clarençon, Didier Smadja, Nicolas Chausson, Wagih Ben Hassen, Michel Piotin, Jildaz Caroff, Sonia Alamowitch and Guillaume Turc in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241253660 – Supplemental material for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase
Supplemental material, sj-docx-2-eso-10.1177_23969873241253660 for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase by Gaultier Marnat, Gaspard Gerschenfeld, Stephane Olindo, Igor Sibon, Pierre Seners, Frederic Clarençon, Didier Smadja, Nicolas Chausson, Wagih Ben Hassen, Michel Piotin, Jildaz Caroff, Sonia Alamowitch and Guillaume Turc in European Stroke Journal
Supplemental Material
sj-docx-3-eso-10.1177_23969873241253660 – Supplemental material for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase
Supplemental material, sj-docx-3-eso-10.1177_23969873241253660 for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase by Gaultier Marnat, Gaspard Gerschenfeld, Stephane Olindo, Igor Sibon, Pierre Seners, Frederic Clarençon, Didier Smadja, Nicolas Chausson, Wagih Ben Hassen, Michel Piotin, Jildaz Caroff, Sonia Alamowitch and Guillaume Turc in European Stroke Journal
Supplemental Material
sj-jpg-4-eso-10.1177_23969873241253660 – Supplemental material for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase
Supplemental material, sj-jpg-4-eso-10.1177_23969873241253660 for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase by Gaultier Marnat, Gaspard Gerschenfeld, Stephane Olindo, Igor Sibon, Pierre Seners, Frederic Clarençon, Didier Smadja, Nicolas Chausson, Wagih Ben Hassen, Michel Piotin, Jildaz Caroff, Sonia Alamowitch and Guillaume Turc in European Stroke Journal
Supplemental Material
sj-png-5-eso-10.1177_23969873241253660 – Supplemental material for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase
Supplemental material, sj-png-5-eso-10.1177_23969873241253660 for Incidence and predictors of intracranial hemorrhage after intravenous thrombolysis with tenecteplase by Gaultier Marnat, Gaspard Gerschenfeld, Stephane Olindo, Igor Sibon, Pierre Seners, Frederic Clarençon, Didier Smadja, Nicolas Chausson, Wagih Ben Hassen, Michel Piotin, Jildaz Caroff, Sonia Alamowitch and Guillaume Turc in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Local ethics committee approval was obtained (IDRCB number: 2017-A03457-46).
Informed consent
Patients were informed of their participation and offered the possibility to withdraw, in compliance with French law regarding retrospective studies of anonymized standard care data.
Guarantor
GM is the guarantor for the present study.
Contributorship
GM and GT researched literature, conceived the study. GM, GG, SA and GT were involved in protocol development, gaining ethical approval and patient recruitment. GT and GM were involved in data and statistical analysis. GM wrote the first draft of the manuscript. All authors collected data and reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.