Abstract
Background and aim
In acute ischemic stroke, under- or overestimation of body weight can lead to dosing errors of recombinant tissue plasminogen activator with consequent reduced efficacy or increased risk of hemorrhagic complications. Measurement of body weight is more accurate than estimation of body weight but potentially leads to longer door-to-needle times. Our aim was to assess if weight modality (estimation of body weight versus measurement of body weight) is associated with (i) symptomatic intracranial hemorrhage rate, (ii) clinical outcome, and (iii) door-to-needle times.
Methods
Consecutive patients treated with intravenous thrombolysis between 2009 and 2016 from 14 hospitals were included. Baseline characteristics and outcome parameters were retrieved from medical records. We defined symptomatic intracranial hemorrhage according to the European Cooperative Acute Stroke Study (ECASS)-III definition and clinical outcome was assessed with the modified Rankin Scale. The association of weight modality and outcome parameters was estimated with regression analyses.
Results
A total of 4801 patients were included. Five hospitals used measurement of body weight (n = 1753), six hospitals used estimation of body weight (n = 2325), and three hospitals (n = 723) changed from estimation of body weight to measurement of body weight during the study period. In 2048 of the patients (43%), measurement of body weight was used and in 2753 (57%), estimation of body weight. In the measurement of body weight group, an inbuilt weighing bed was used in 1094 patients (53%) and a patient lift scale in 954 patients (47%). In the estimation of body weight group, policy regarding estimation was similar. Estimation of body weight was not associated with increased symptomatic intracranial hemorrhage risk (adjusted odds ratio = 1.16; 95% confidence interval 0.83–1.62) or favorable outcome (adjusted odds ratio = 0.99; 95% confidence interval 0.82–1.21), but it was significantly associated with longer door-to-needle times compared to measurement of body weight using an inbuilt weighing bed (adjusted B = 3.57; 95% confidence interval 1.33–5.80) and shorter door-to-needle times compared to measurement of body weight using a patient lift scale (−3.96; 95% confidence interval −6.38 to −1.53).
Conclusion
We did not find evidence that weight modality (estimation of body weight versus measurement of body weight) to determine recombinant tissue plasminogen activator dose in intravenous thrombolysis eligible patients is associated with symptomatic intracranial hemorrhage or clinical outcome. We did find that estimation of body weight leads to longer door-to-needle times compared to measurement of body weight using an inbuilt weighing bed and to shorter door-to-needle times compared to measurement of body weight using a patient lift scale.
Keywords
Introduction
Acute ischemic stroke patients should receive intravenous thrombolysis (IVT) with recombinant tissue plasminogen activator (rt-PA) as quickly as possible for optimal clinical efficacy.1–5 The most feared complication of IVT is symptomatic intracranial hemorrhage (sICH) occurring in 2.7–5.7% of patients. 6
Dose finding trials for rt-PA indicate that 0.9 mg/kg body weight has an optimal safety and efficacy profile: a lower dose resulted in reduced efficacy and a higher dose in increased sICH risk.7-9 The patient’s weight is therefore essential, but exact measurement can be time consuming leading to increased door-to-needle times (DNTs) with less clinical IVT efficacy. Therefore, estimation of body weight (EBW), rather than exact measurement of body weight (MBW), is often used with potential under- or overestimation.10–14 Indeed, overestimation due to EBW was shown to result in increased rt-PA dose and increased sICH risk.11,12,15,16 In contrast, other studies did not confirm this observation, indicating that EBW is acceptable since dosing errors did not influence outcomes.13,17 However, the sample sizes of these studies so far are small (n = 222; n = 308) and were therefore underpowered to detect differences in sICH rate. Hence, based on available evidence, it is not possible to draw conclusions on the best weight modality.
National and the American Stroke Association guidelines lack recommendations regarding weight modality, thus both EBW and MBW are being used in clinical practice.18,19 We used this disparity to assess if weight modality is associated with (i) sICH rate, (ii) clinical outcome, and (iii) DNT.
Methods
Study design and patient selection
We derived data from prospective IVT registries of 14 centers and included consecutive adult patients with acute ischemic stroke (AIS) treated with IVT between January 2009 and December 2016. Patients were excluded if weight modality was unknown or if no clinical data were available.
The ethical standards committee of the Leiden University Medical Centre approved the protocol and waived the need for written informed consent from individual patients.
Patient data
The following data were collected: patient characteristics including demographics, vascular risk factors and history, medication use, admission blood pressure, and baseline stroke severity assessed with the National Institute of Health Stroke Scale (NIHSS) score. In case data were missing, these were complemented from the medical records. In case NIHSS score was not noted, this was reconstructed from neurological examination at admission with a validated algorithm as described previously. 20
Weight assessment
Mode of weight assessment during the inclusion period was acquired by asking the stroke neurologist involved and by assessing local protocols of each participating center. In all centers, either estimation or exact MBW was done before the CT scan. None of the EBW centers measured body weight during (infusion of) IVT, so possible discrepancies between reported and estimated weight did not led to alteplase dose adjustments.
In the EBW group, policy was similar in all centers: (i) weight was assessed first by asking the patient; (ii) in case this was not possible (e.g. due to aphasia) by asking a relative; and (iii) if this was not possible estimation was always done by the treating physician, but in case another health care worker had a different estimation, consensus was reached.
In the MBW group, weight was measured: (i) by transferring the patient to a bed with an inbuilt weighing option or a stretcher standing on a ground scale or (ii) by using a patient lift scale, requiring to lift the patient in a sling.
Outcome measures
Our primary outcome measure was the sICH rate. We defined sICH according to the ECASS-III definition, i.e. any apparently extravascular blood in the brain or within the cranium that was associated with clinical deterioration, as defined by an increase of 4 or more points on NIHSS score, or that led to death and that was identified as the predominant cause of the neurological deterioration. 3 In our study, we included all sICH within seven days after stroke onset. Secondary outcome measures included favorable outcome at 90 days (defined as a score of 0–2 on the modified Rankin Scale (mRS)) and DNT (which was defined as the time between patient arrival at the hospital and intravenous rt-PA initiation). 21 In case of missing data on clinical outcome at 90 days, the mRS was derived using available follow-up data before 3 months and ≥1 month after hospital discharge. Both sICH and clinical outcome were retrieved from medical records, including neuro-imaging data by two independent reviewers (TTMN and AEG). Discrepancies were solved by discussion. Time of symptom onset, time of center arrival, and time of IVT initiation were extracted to calculate the DNT.
Statistical analysis
Descriptive statistics were used to compare patient characteristics. Categorical variables were compared with χ 2 test. Continuous variables were compared using the t test or Mann–Whitney U test, and are presented as mean ± standard deviation (SD) or median and interquartile range (IQR).
We used logistic regression to assess the association of separate outcomes (sICH and clinical outcome) in relation to weight modality, expressed as odds ratios (ORs) or adjusted ORs (aOR) with corresponding 95% confidence interval (CI). Linear regression analysis was performed to assess the association between weight modality and DNT, presented as regression coefficient (B) and corresponding 95% CI. In secondary analysis, we adjusted for baseline characteristics associated with outcomes (P < 0.1) except for the analysis related to the outcome DNT where we adjusted for variables known to have an association with the DNT: availability of a CT in the emergency room (ER), blood pressure above the threshold for IVT (>185/110 mmHg), NIHSS score at baseline,22,23 onset-to-door time (defined as the time between stroke onset and patient arrival at the hospital) and for annual IVT-volume divided as follows: low-volume (≤24), medium-volume (25–49), or high-volume (≥50) as described previously, with low-volume as reference category. 24 In subgroup analyses we investigated if differences in methods within the EBW or the MBW group could have affected the association between weight modality and the outcome measures.
Missing data
For missing data we performed multiple imputations with the fully conditional specification method with five sets of imputations. The predictive mean matching model type was used for scale variables. Then, we compared the results of the analysis of the imputed dataset with the nonimputed dataset to assess if this leads to consistent parameter estimates. Additionally, we performed post hoc sensitivity analyses to assess the effect of missing data with regards to mRS score after 90 days, by recalculating the estimates while omitting patients with missing mRS score after 90 days.
Statistical analysis was performed using SPSS software (version 23, IBM, New York, USA).
Results
Baseline characteristics
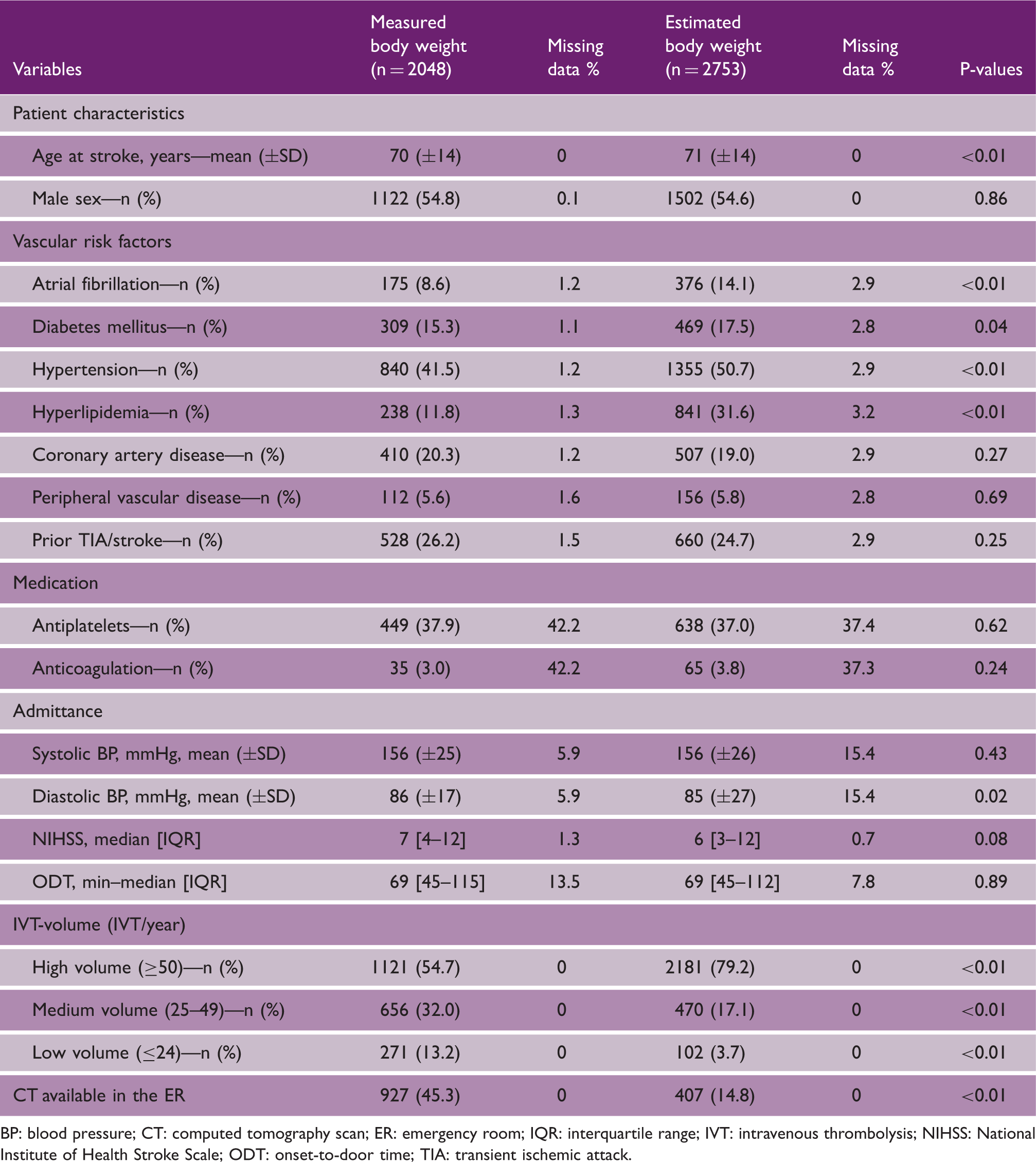
Data from 5066 patients with AIS were collected. A total of 4801 (95%) patients met the inclusion criteria (Figure 1). Five centers used MBW, six centers EBW, and three centers changed from EBW to MBW during our inclusion time window. In 2048 of the patients (43%), MBW was used and in 2753 patients (57%) EBW. EBW-patients were slightly older and they had more cardiovascular risk factors (atrial fibrillation, diabetes mellitus, hypertension, and hyperlipidemia) (Table 1). Other known predictors for sICH (sex, NIHSS score, blood pressure, and onset-to-door time) did not differ between the EBW and the MBW group. More EBW-patients were treated in high-volume centers (n = 2181; 79%) compared to MBW-patients (n = 1121; 55%) and a CT in the ER was present for 407 EBW-patients (15%) and for 927 MBW-patients (45%).
Flowchart of the study. aTen patients had incomplete data and an unknown weight modality. AIS: acute ischemic stroke; IVT: intravenous thrombolysis. Patient characteristics BP: blood pressure; CT: computed tomography scan; ER: emergency room; IQR: interquartile range; IVT: intravenous thrombolysis; NIHSS: National Institute of Health Stroke Scale; ODT: onset-to-door time; TIA: transient ischemic attack.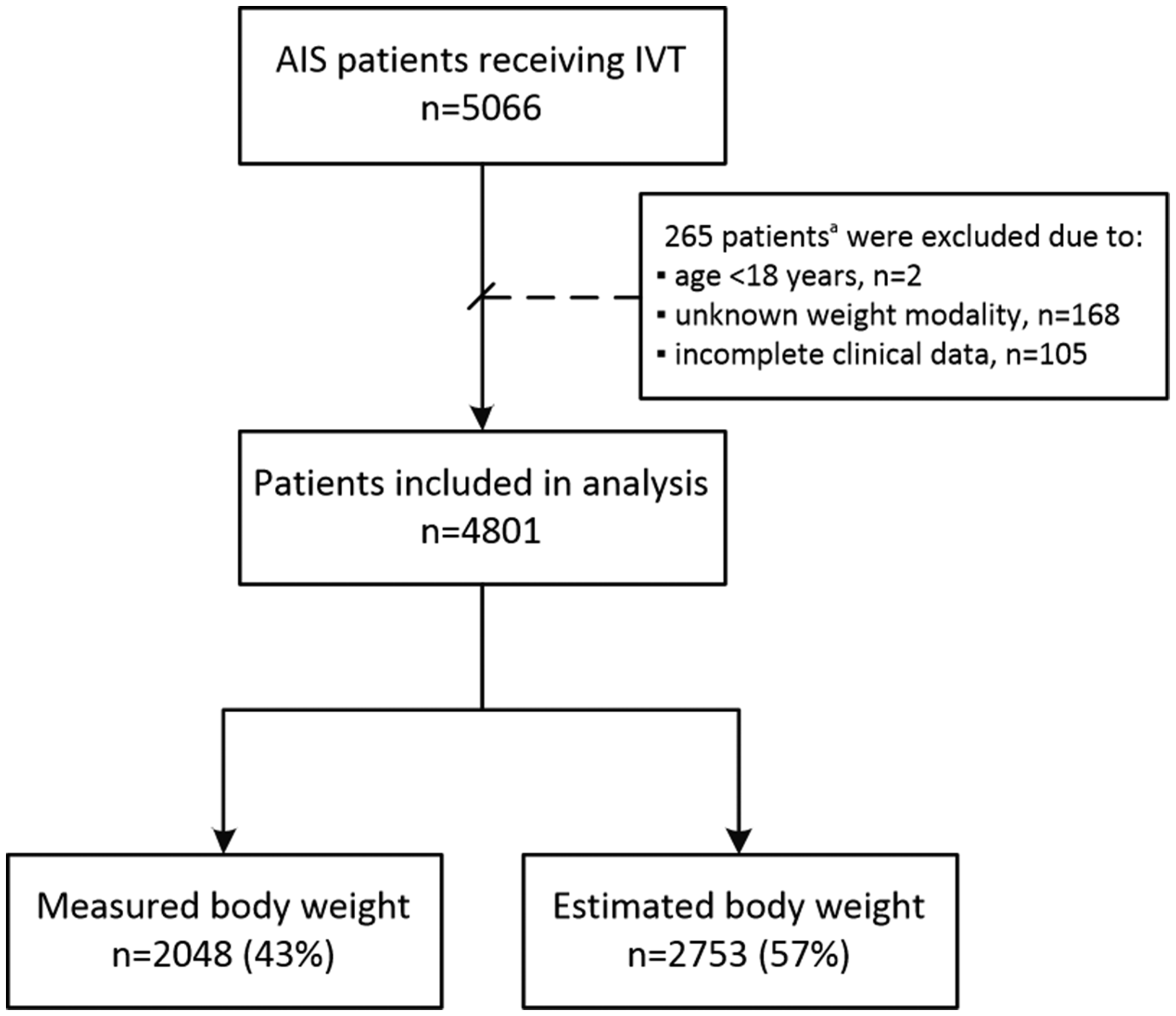
Outcomes
Outcome measures
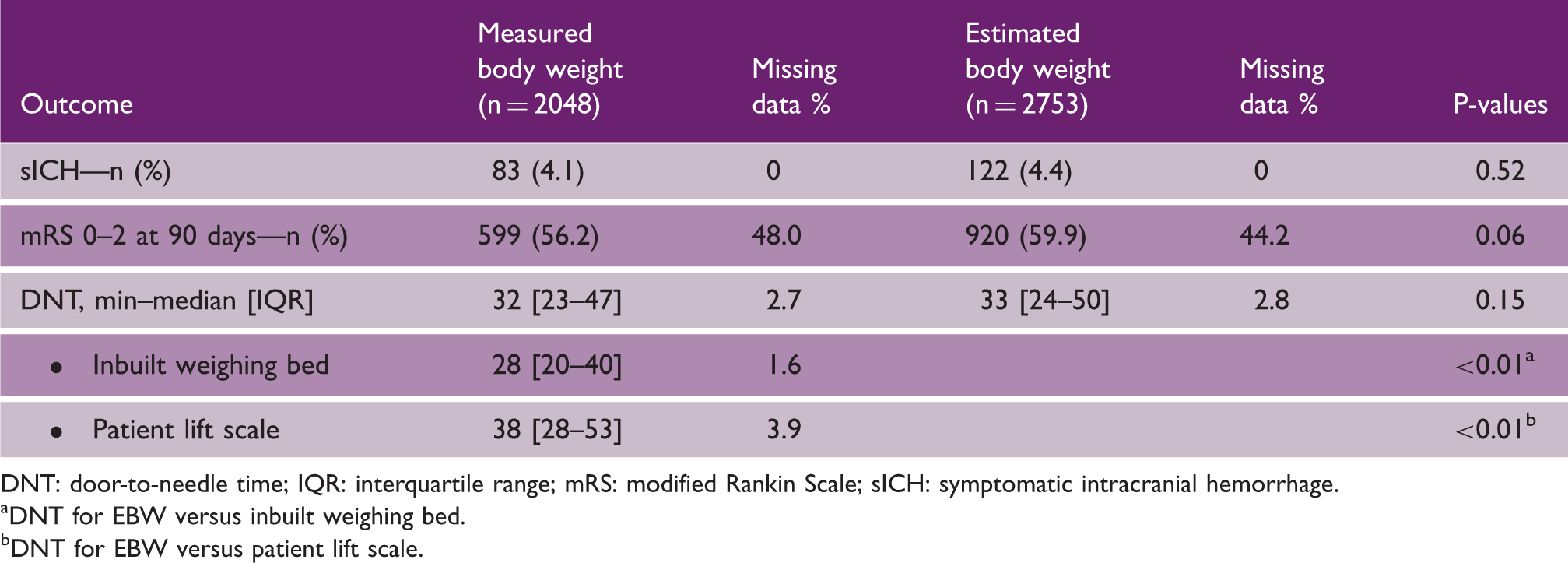
DNT: door-to-needle time; IQR: interquartile range; mRS: modified Rankin Scale; sICH: symptomatic intracranial hemorrhage.
DNT for EBW versus inbuilt weighing bed.
DNT for EBW versus patient lift scale.
Logistic and linear regression analysis for the association between weight modality (EBW versus MBW) and the outcome measures

aOR: adjusted OR; B: unstandardized regression coefficient; DNT: door-to-needle time; EBW: estimated body weight; MBW: measurement of body weight; OR: odds ratio.
aOR, adjusted for age; atrial fibrillation; diabetes mellitus; hypertension; hyperlipidemia; admission NIHSS, CT in the ER, and IVT-volume.
B, adjusted for blood pressure exceeding threshold for IVT; admission NIHSS, CT in the ER, onset-to-door time, and IVT-volume.
DNT in minutes for EBW versus MBW, inbuilt weighing bed.
DNT in minutes for EBW versus MBW, patient lift scale.
Missing data
Baseline characteristics did not show a relevant difference in patients with or without a known clinical outcome and missing outcome data were also evenly distributed between the groups. Results of the analysis of the imputed dataset were essentially the same as the results of the analysis without imputed data (supplementary data Table 5). Furthermore, post hoc sensitivity analysis excluding patients with an unknown clinical outcome yielded similar robustness of the primary analysis (supplementary Table 6).
Discussion
Our findings did not demonstrate an association between weight modality and sICH rates or clinical outcome. While previous prospective studies have shown that EBW leads to dosing errors, our results showed that this does not translate into a different safety and efficacy profile of intravenous rt-PA in clinical practice. Interestingly, we found that EBW leads to a longer DNT compared to MBW using an inbuilt weighing bed, but to a shorter DNT compared to MBW using a patient lift scale.
Our main results are in line with some previous studies.13,17 However, our study has a much larger study population and unlike the previous studies it concerns a multicenter study. Therefore, it is unlikely that we missed a difference in outcome related to weight modality rendering our results more generalizable to routine clinical practice.
In contrast, two studies showed a difference in clinical outcome related to weight modality. One retrospective mono-center study (n = 164) found that EBW led to rt-PA overdose in 13 (16%) patients. Of those 13 patients, four had an intracranial hemorrhage (however, it remained unclear whether these were symptomatic or not). 12 Another prospective mono-center study (n = 128) found that EBW leads to rt-PA overdose in 52% of the patients with more sICH in the first 24 h. 15 The overall sICH rate for the whole group was 7.8% in the first 24 h which is much higher than one would expect from previous studies with this sICH definition. 25 This may have influenced the results limiting generalizability. A possible explanation for the high sICH rate is the predominantly Asian population in this study as Asian ethnicity is associated with increased risk of sICH.26–28 Furthermore, a follow-up brain CT scan was performed as part of standard clinical care at 24 h. Therefore, researchers could have been more prone to attribute clinical symptoms to a hemorrhage seen on these standard imaging protocols. Finally, in our study weight modality was not associated with DNT even after adjusting for factors such as IVT volume, CT availability on the ER, baseline NIHSS, and blood pressure above IVT threshold.24,29–31 Nevertheless, other unknown factors related to the DNT we could not adjust for could possibly explain this lack of an association.
Somewhat surprisingly, the median DNT was shorter in the MBW group using an inbuilt weighing bed compared to the EBW group. An explanation for this could be that in practice weight estimation can require multiple steps (asking the patient or relative and estimation by the treating physician), whereas an inbuilt weighing bed scale only requires one step (transfer of the patient), which is also done in the EBW group (e.g. from ambulance stretcher to hospital bed).
Of note, this difference in DNT does not affect the finding that weight modality is not associated with an increased risk of sICH or clinical outcome, since we adjusted for the DNT in these analyses.
Our study has several limitations. First, a cluster-randomized trial would be a more suitable design for our research question, but in practice this does not seem feasible since clinics using MBW are not likely to change this to EBW. Due to the retrospective nature of our design, extraction of (outcome) data could have led to bias. However, assessment of our primary outcome, sICH, was done according to strict definitions by two independent reviewers and sICH rates are similar to previous studies using the same definition criteria.3,6 Second, clinical outcome was missing for a substantial proportion of patients. We investigated the possible influence of missing data on our parameter estimates, by performing different methods of handling missing data in our cohort. Results of the primary analysis remained consistent after imputing missing data (supplementary data Table 5) and after post hoc sensitivity analysis (excluding patients with unknown clinical outcome), indicating that missing data were not of significant influence on our outcome parameters (supplementary data Table 6). Additionally, missing outcome data were evenly distributed between the groups and baseline patient characteristics did not show a relevant difference in patients with or without a known clinical outcome (data not shown). Of note, even when excluding patients with unknown clinical outcome our cohort still remains the largest so far investigating weight modality in IVT treated patients. As for the DNT, this is an obligatory practice parameter in all centers and is therefore not likely to be affected by retrospective assessment. Furthermore, data on actual rt-PA dosage and (measured) body weight were lacking. Although this would have enabled us to determine exactly in which patient body weight was over- or underestimated and whether this resulted in over- or under dosing rt-PA, it apparently does not translate into an increased risk of sICH or a clinically meaningful difference. Our data indicate that EBW was not associated with increased risk of sICH (aOR = 1.16; 95% CI 0.83–1.62), therefore a possible effect of weight modality on sICH would be smaller than 1.62 with 95% certainty, independent of whether there is a difference between estimated or measure bodyweight. Finally, a limitation is that centers, with or without a certain weighing modality, could differ in local policies which could lead to a bias related to outcomes. However, all centers treat IVT patients according to the same national guidelines, including prehospital notification of potential IVT patients and since patients in each group came from at least five centers and the outcomes are evenly distributed in both groups, we consider this risk minimal.
Our study provides the largest multicenter cohort study to date assessing the association between weight modality (EBW or MBW) with sICH, clinical outcome, and DNT. We found that MBW with an inbuilt weighing bed leads to shorter DNTs compared to EBW, whereas the latter strategy leads to shorter DNTs compared to MBW with a patient lift scale. We did not find evidence that weight modality for rt-PA titration in IVT eligible patients leads to clinically relevant dosing errors, since it was not associated with an increased risk of sICH or favorable clinical outcome.
Supplemental Material
Supplemental material for Thrombolysis related symptomatic intracranial hemorrhage in estimated versus measured body weight
Supplemental Material for Thrombolysis related symptomatic intracranial hemorrhage in estimated versus measured body weight by T Truc My Nguyen, Stephanie IW van de Stadt, Adrien E Groot, Marieke JH Wermer, Heleen M den Hertog, Hanneke M Droste, Erik W van Zwet, Sander M van Schaik, Jonathan M Coutinho and Nyika D Kruyt: for the SOCRATES Steering Committee and Investigators in International Journal of Stroke
Footnotes
Authors’ contributions
TTMN: study concept and design, acquisition, analysis, and interpretation of data, statistical analysis, and drafting and revising the manuscript. SIWS: acquisition and analysis of data and revising the manuscript for scientific content. AEG: acquisition of data and revising the manuscript for scientific content. MJHW, HMH, and SMS: critical revision of the manuscript for important intellectual content. HMD: acquisition of data. EWZ: statistic analytical support, revising the manuscript for scientific content. JMC: study concept and design and critical revision of the manuscript for important intellectual content. NDK: study concept and design, interpretation of data, critical revision of the manuscript for important intellectual content, and study supervision.
Acknowledgements
We thank all participating hospitals and physicians for their help: Yvo Roos (Amsterdam University Medical Centre, locatie AMC), Marieke Visser (Amsterdam University Medical Centre, locatie VUmc), Nyika Kruyt (Leiden University Medical Centre), Heleen den Hertog (Isala hospital, Zwolle), Patricia Halkes (Noord West Ziekenhuisgroep, locatie Alkmaar), Lahcen Hani (Noord West Ziekenhuisgroep, locatie Den Helder), Vincent Kwa (Onze Lieve Vrouwen Gasthuis, locatie Oost), Sander van Schaik (Onze Lieve Vrouwen Gasthuis, locatie West), Willem van der Meulen (Rode Kruis Ziekenhuis), Marieke de Graaf (Medical Centre Slotervaart), Frank de Beer (Spaarne Gasthuis), Jelle de Kruijk (Tergooi Ziekenhuis), Caspar Zwetsloot (Waterlandziekenhuis), and Taco van der Ree (West Fries Gasthuis).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
