Abstract
Introduction:
Variations in the left atrial appendage (LAA) morphology are associated with different embolic risk in patients with atrial fibrillation (AF). Data are scarce regarding the association between LAA morphology and Embolic stroke of undetermined source (ESUS).
Patients and methods:
Using cardiac computed tomography (CCT) scans, LAA morphology was categorized as either chicken wing (CW), cactus, windsock, or cauliflower. Furthermore, we examined the presence of large secondary lobes arising from the main lobe, considering their existence as indicative of a complex LAA morphology. LAA morphologies were compared between ESUS (n = 134) and AF patients (n = 120); and between ESUS patients with (n = 24) and without (n = 110) subsequent AF diagnosis during long-term follow-up.
Results:
ESUS patients had a significantly higher prevalence of cauliflower morphology compared to AF group (52% vs 34%, respectively, p = 0.01); however, no significant difference was found between the groups when categorizing LAA morphology to either CW or non-CW. ESUS patients had significantly higher prevalence of large secondary lobes compared with AF patients (50% vs 29%, respectively, p = 0.001). When comparing ESUS patients with and without AF diagnosis during follow-up (20–48 months of follow-up, median 31 months), there were no significant differences in the prevalence of the “classical” morphologies, but large secondary lobes were significantly more prevalent among those without subsequent AF diagnosis.
Conclusion:
ESUS patients have a high prevalence of complex LAA morphology, which might be associated with an increased risk for thrombus formation even in the absence of AF.

Keywords
Introduction
The term Embolic Stroke of Undetermined Source (ESUS) was introduced to describe a non-lacunar cerebral infarction occurring in the absence of a specific identifiable stroke mechanism.1,2 Determining the cause of IS in general and of ESUS in particular has significant importance, as it can directly affect subsequent management and secondary prevention strategies, specifically, the choice between antiplatelet or anticoagulation treatment. 3 It was previously speculated that undetected or subclinical atrial fibrillation (AF) is the underlying etiology in most of ESUS cases, but it was shown that although 30% of ESUS patients are eventually diagnosed with AF on a long-term cardiac monitoring, a large proportion of patients remain without identifiable stroke etiology. 4 Moreover, three large clinical trials did not show unequivocal benefit of routine prescription of oral anticoagulation in ESUS patients,5–7 further supporting the heterogeneity of this group of patients. 8 Therefore, other pathophysiologic mechanisms for thromboembolism should be investigated.
The left atrial appendage (LAA) morphology may constitute an additional anatomic marker of thrombo-embolic risk, being the most common site of thrombus formation in patients with AF. 9 Significant variations exist in LAA shape and size; currently, four main LAA morphology variants are recognized: “chicken wing,” “cactus,” “windsock,” and “cauliflower.” 10 Of those, chicken wing (CW) morphology was found to be associated with the least likelihood of embolic stroke in patients with AF,10–13 which led to a more simplified classification to either CW or non-CW morphology in some studies. 14 Although few recent studies have suggested an association between ESUS and non-CW LAA morphology,15,16 data are still scarce in this group of patients.
In the present study we aimed to study the association between LAA morphological complexity and ESUS, using a consecutive group of patients who underwent cardiac computed tomography (CCT) as part of ESUS etiology evaluation. Apart from the “classical” morphological classification, we additionally evaluated the LAA complexity by looking at the presence and size of secondary branches. We compared the LAA properties of ESUS patients with those of a group of patients with AF, who underwent routine pre-procedural CCT before anticipated AF ablation.
Methods
Study population
Acquisition protocolAll scans were performed either with a 256-slice system (Brilliance iCT, Philips Healthcare, Cleveland, Ohio) or with a Dual Source CT scanner SOMATOM Force (Siemens Healthcare, Erlangen, Germany). The scan direction was cranio-caudal and the scan volume ranged from the aortic arch to below the diaphragmatic pace of the heart. Patients were given intravenous metoprolol (up to 4 doses of 5 mg) whenever the heart rate was >65 beats/min.
Intravenous injection of 55–85 ml of nonionic contrast agent (Iopromide 370; Bayer Schering, Berlin, Germany) was administered at a flow rate of 4–6 ml/s and was followed by a 40 ml saline chase bolus. Data acquisition consisted of two acquisitions. The first acquisition was automatically initiated using a bolus tracking with a region of interest placed in the ascending aorta (Dual source scanner) or in the descending aorta (256-slice system), a threshold level of 180 Hounsfield units was used to initiate the first scan. The second acquisition was automatically initiated 60 s after administration of the contrast agent. Acquisition was performed during an inspiratory breath hold while the electrocardiogram was recorded simultaneously to allow retrospective gating of data.
Image reconstruction and data analysis. The 3-dimensional reconstructed systolic and diastolic dataset of the contrast-enhanced CT scan was transmitted to a dedicated CT workstation (Philips Intellispace Portal, version 11.0) for data analysis.
For assessment of left atrium (LA) and LAA volumes and morphologies, the data set was reconstructed at 5% increments over the cardiac cycle. For assessment of maximal LA volume (LAVmax) the phase with the smallest left ventricular volume, corresponded with peak ventricular systole, was used. For assessment of traditional LAA morphology three- dimensional volume rendering was reconstructed. LAA volume was determined after reduction of LAVmax from the total rendered volume of the LA and LAA structure. LAA side branch morphology was determined using multiplanar reconstruction based upon the criteria which was previously used in prior studies 10 and as detailed in Figure 1. Secondary lobes were defined as side branches arising from the main LAA lobe, which have a significantly smaller diameter than the main LAA (i.e. not resulting merely from a band in the main LAA lobe). The complexity of the LAA was evaluated according to the presence of secondary branches with a base orifice diameter ⩾10 mm. In patients with LAA thrombus the delayed scan was used to evaluate the traditional and proposed LAA morphology. Figure 2 shows representative example of LAA characterization according to our suggested classification.
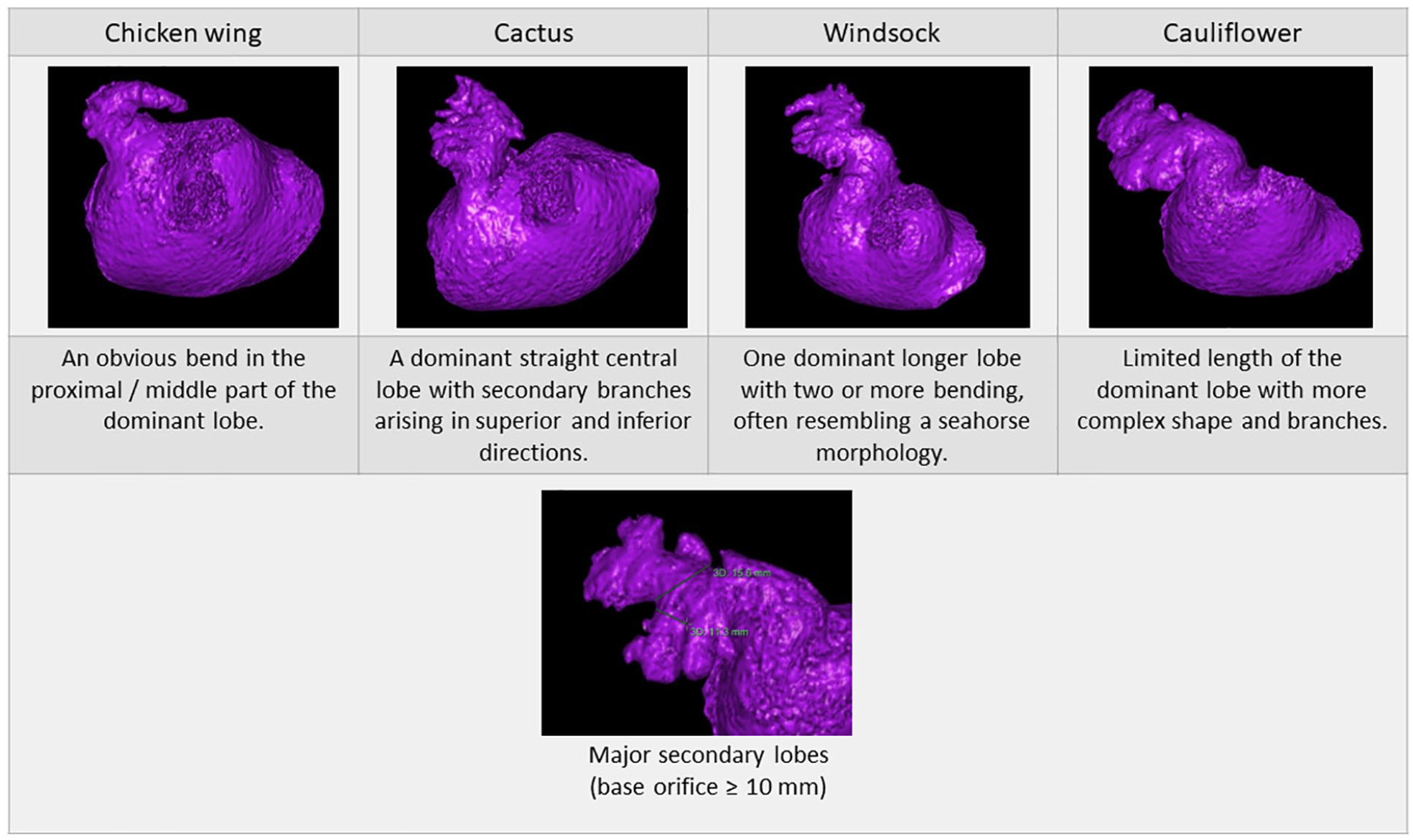
“Classic” and suggested secondary branching classification of LAA.
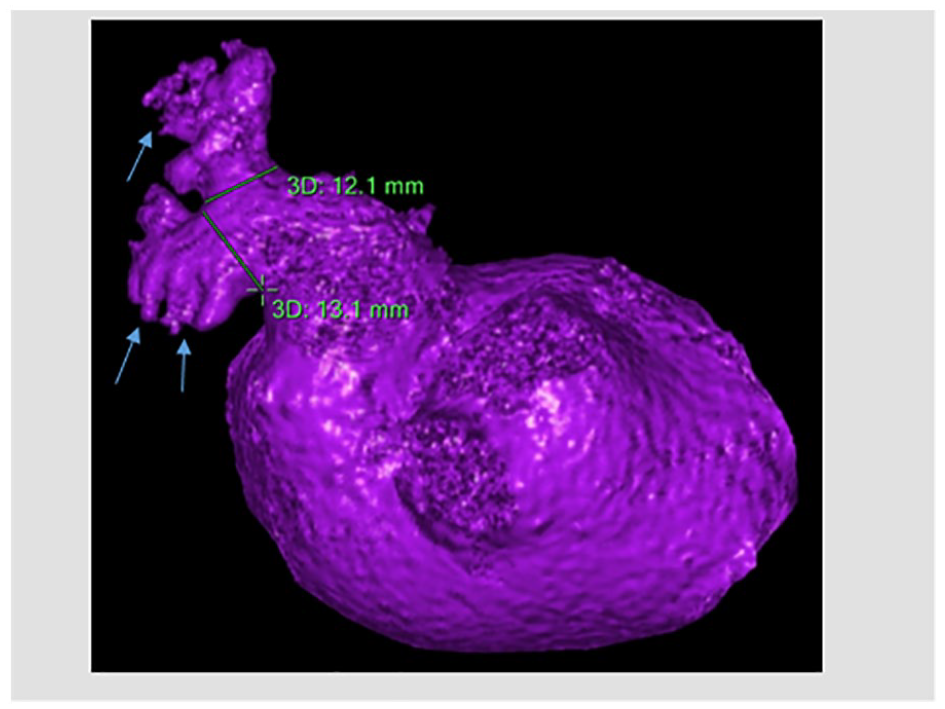
LAA secondary and tertiary branches. Morphology was defined as cauliflower. This LAA has two secondary branches (green lines show their base) and small tertiary branches (blue arrows).
Study analyses
The study was divided into two parts: (1) In the first part, we used CCT to compare LA and LAA volumes and LAA morphologies between the ESUS group and the AF group. (2) In the second part of the study, we conducted a prolonged retrospective follow-up on each of ESUS patients in February 2023, so the longest follow-up period was 48 months for the first patients; and the shortest was 20 months for the last patient in the ESUS registry; the median follow-up time was 31 months. By using inpatient and outpatient medical records and clinical investigations such as Holter monitoring, emergency department/family physician/cardiologist/neurologist visits, etc., we identified all ESUS patients who had consequent diagnosis of AF that occurred during the follow-up period. We then compared LAA morphologies within the ESUS group, between ESUS patients with and without subsequent AF diagnosis.
Demographic and clinical data of all patients were extracted from the electronic medical records. Covariates of interest were selected upon previous knowledge regarding their relationship with stroke and cardiac pathologies, such as ischemic heart disease (IHD), congestive heart failure (CHF), peripheral vascular disease (PVD), previous stroke, chronic renal failure (CRF), diabetes mellitus (DM) and hypertension (HTN).
Statistical analysis
A descriptive analysis was performed, and data was presented as median and interquartile range or percentage, whenever appropriate. All data were compared using κ² analysis for categorical variables, Student’s two-sided t-test, or Wilcoxon rank-sum for normal or non-normal distributed continuous variables, whichever appropriate. p-Value of <0.05 was considered as statistically significant.
Inter-observer variability
To assess interobserver agreement for the interpretation of left atrial appendage morphology and assessment of presence and number of secondary lobes, a second observer who was blinded to the results performed the analysis in a randomly selected sample of 50 studies. Cohen’s κ was applied to allow the quantitative measure of agreement between the 2 observes. Cohen κ was applied to allow quantitative measure of agreement by using the following grading: κ grades of 0–0.2 = poor agreement; 0.21–0.40 = fair agreement; 0.41–0.6 = moderate agreement; 0.61–0.80 = substantial agreement; and 0.81–1.0 = excellent agreement. For all κ values, the respective 95% confidence intervals (CIs) are given. 20
This study was approved by the Ethics Committee of Rabin Medical Center and was performed consistently with Helsinki declaration.
Results
ESUS versus AF groups
In the first part of the study, 134 consecutive ESUS patients were compared with 120 AF patients. Compared to AF patients, ESUS patients were significantly older (74.2 ± 10.2 vs 60.6 ± 10.9 years; respectively, p < 0.001), and were more likely to be females (49% vs 30%; respectively, p < 0.001). They had more baseline hypertension and diabetes mellitus compared to the AF group. LA diameter and area were significantly higher in AF patients (4.2 ± 0.6 vs 3.8 ± 0.6 mm and 23.2 ± 5.2 vs 20.1 ± 4.9 mm2, respectively, p < 0.001 for both) and there was no significant difference in left ventricular ejection fraction (LVEF) between the groups. Baseline characteristic of patients in both groups are presented in Table 1. Of note, LAA thrombus was detected by CCTA in 9 ESUS patients.
Baseline characteristic of the study groups.
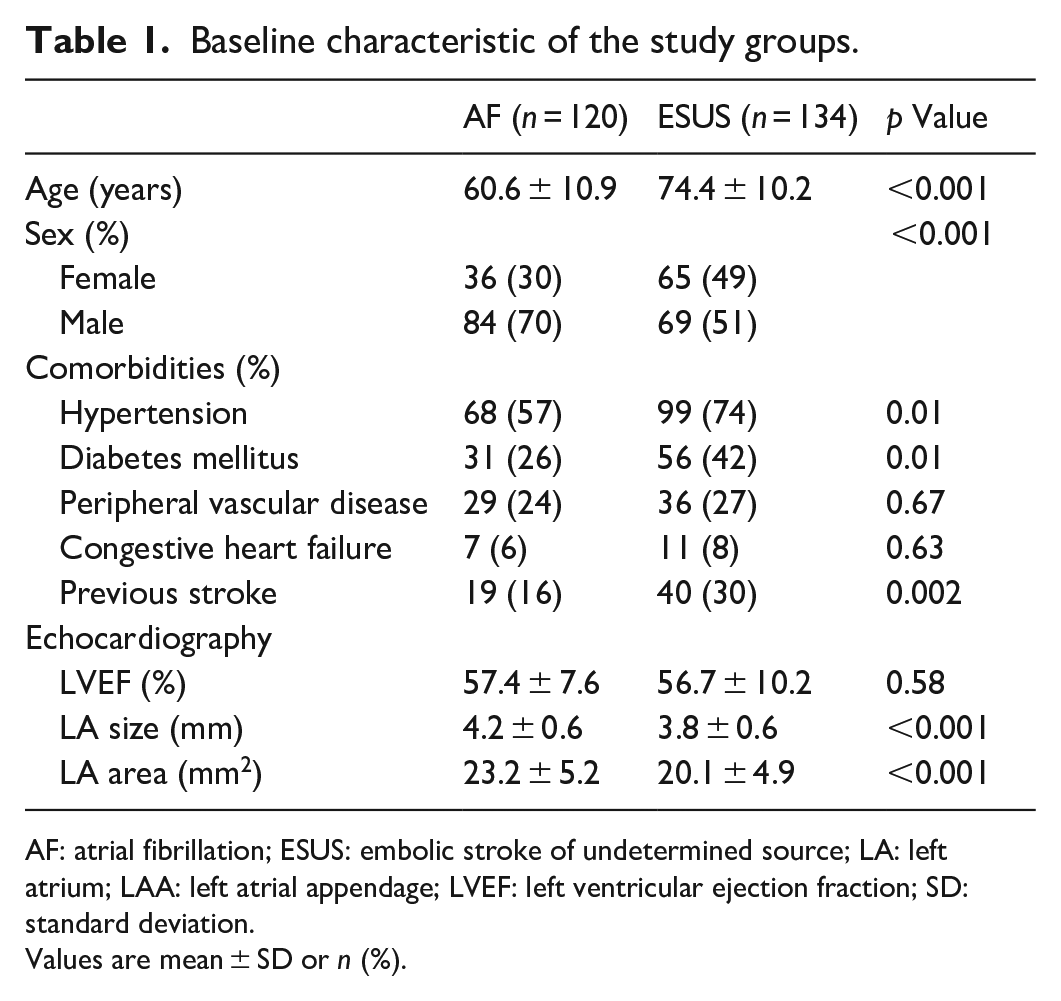
AF: atrial fibrillation; ESUS: embolic stroke of undetermined source; LA: left atrium; LAA: left atrial appendage; LVEF: left ventricular ejection fraction; SD: standard deviation.
Values are mean ± SD or n (%).
Using CCT, ESUS patients had significantly smaller LA and LAA volumes compared to AF patients (102.7 ± 29.3 vs 119.5 ± 40 mm3, p < 0.001 and 5 ± 2 vs 6 ± 3 mm3, p = 0.03; respectively). The prevalence of the four common LAA morphologies differed significantly between ESUS and AF patients (Table 2). Cauliflower morphology was more common in ESUS than in AF group (52% vs 34%, respectively, p = 0.01). However, when considering the simplified CW versus non-CW categorization, there was no significant difference between the groups (p = 0.24). ESUS patients had significantly higher prevalence of large secondary branches (complex LAAs) compared to AF patients (50% vs 29%; respectively, p = 0.001). The classic CW/ non-CW classification did not reliably represent the presence of secondary branches: 21% of CW LAAs had large secondary branches, while 55% of the non-CW LAAs did not have them.
Cardiac computer tomography analysis.
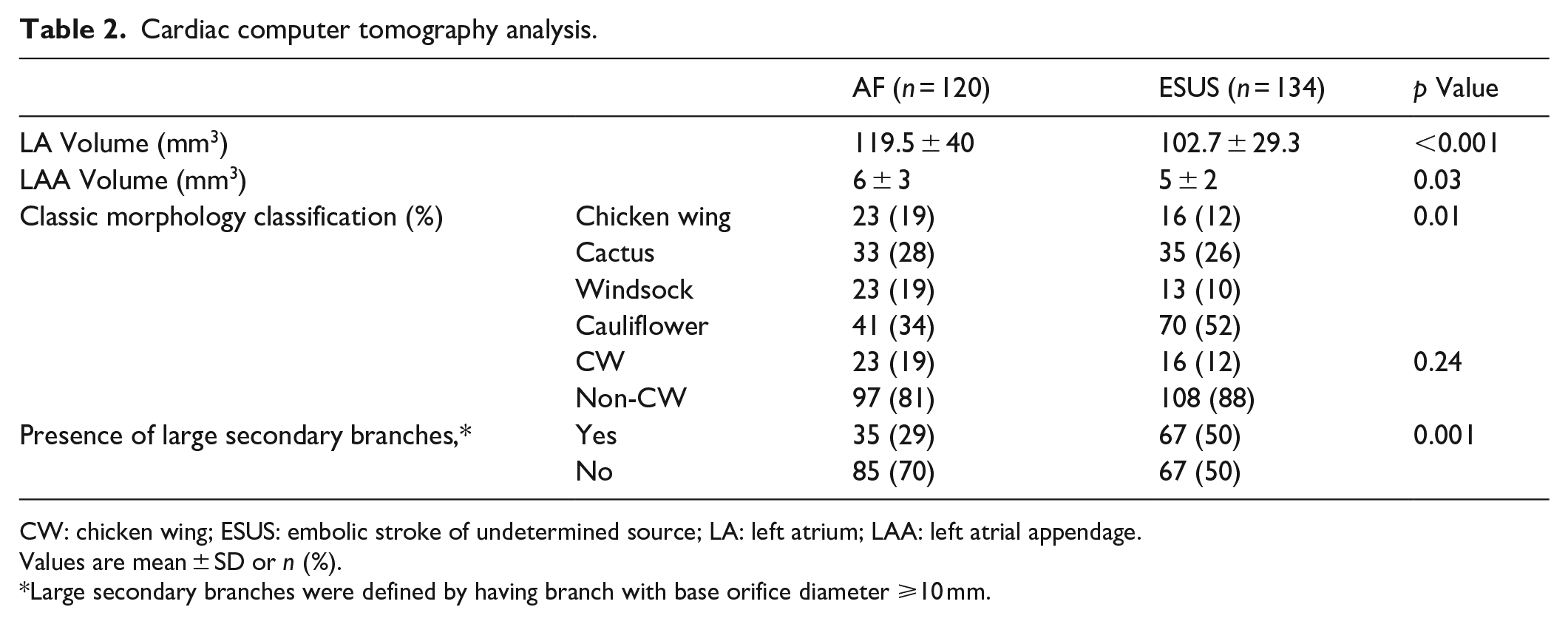
CW: chicken wing; ESUS: embolic stroke of undetermined source; LA: left atrium; LAA: left atrial appendage.
Values are mean ± SD or n (%).
Large secondary branches were defined by having branch with base orifice diameter ⩾10 mm.
ESUS patients with/without subsequent AF diagnosis
During a prolonged follow-up period, 24/134 (18%) of ESUS patients were diagnosed with AF: in 13 cases, diagnosis was incidental when an ECG was performed for other reasons; 4 were diagnosed when ECG was performed due to palpitation symptoms; and in 7 cases, diagnosis was done using prolonged patch- using Holter monitoring. When comparing LAA morphology between ESUS patients with and without AF diagnosis (Table 3), we found no significant differences in the prevalence of the “classical” morphologies, but ESUS patients without subsequent AF diagnosis had significantly higher prevalence of LAAs with large secondary branches.
LAA morphology according to long-term AF diagnosis in ESUS patients.

AF: atrial fibrillation; ESUS: embolic stroke of undetermined source; LAA: left atrial appendage.
Large secondary branches were defined by having branch with base orifice diameter ⩾10 mm.
Inter-observer variability
In 50 randomly selected patients from the study population, the interobserver agreement for the evaluation of traditional left atrial appendage morphology was substantial: κ = 0.72 (95% CI 0.0.56–0.88) and for the presence of large secondary branches (with base orifice diameter ⩾10 mm): κ = 0.80 (95% CI 0.64–0.96).
Discussion
In this study, we found a higher prevalence of cauliflower LAA morphology in patients with ESUS compared to patients with AF, but no significant difference between the groups when categorizing the LAA morphology to either CW or non-CW. When analyzing the CCT of ESUS patients, we noticed that many of the LAAs had complex structure, which may affect the thrombotic risk due to turbulent blood flow through the different shape and size of variable number of lobes. This observation led us to evaluate the LAA structure not only by the classic morphology classification but also by the presence of large secondary lobes, which we defined by a base orifice diameter ⩾10 mm. Indeed, ESUS patients had significantly higher prevalence of LAAs with large secondary branches compared to AF patients. Moreover, in a long-term follow-up, LAAs with large secondary branches were significantly more prevalent among ESUS patients in whom no AF was detected, compared to those who were diagnosed with AF during the follow-up period.
LAA morphology and its association with thromboembolic risk was most extensively studied in the context of AF, and this is the main reason for choosing AF patients as the control group in this study.10–13 Our results show a high prevalence of non-CW morphology (88%) in ESUS patients compared to non-AF patients, in whom the prevalence of non-CW morphologies was reported to be less than 60%. 21 The assumption that the trigger for thrombus formation in the LAA is blood stasis during episodes of AF was challenged by demonstrating no clear relationship between AF episodes and the occurrence of stroke,22,23 nor a clear protective advantage in maintaining rhythm control. 24 Previous studies demonstrated that LAA structural complexity and orifice size have a direct influence on blood flow velocity and blood stasis inside the LAA.25,26 Xu et al. 27 recently reported an association between LAA morphology and flow velocity with local platelet activity, fibrinolysis function and endothelial dysfunction, with cauliflower LAA associated with increased thrombotic factors. Those observations suggest an underlying mechanism for the association between LAA morphology and embolic events, independently of the presence of atrial arrhythmia.10,11,28
In recent years, data on the association between LAA morphology and embolic stroke in patients without AF are emerging. Gwak et al. 15 found the risk of embolic stroke recurrence in ESUS patients to be independently associated with non-CW LAA morphology (hazard ratio 3.17, 95% CI 1.13–8.91; p = 0.029). A recent systematic review showed a higher prevalence of non-CW LAA morphology in ESUS compared to patients with non-cardioembolic stroke. 29 Soni et al. 16 found a complex LAA morphology to be independently associated with ESUS (odd ratio 6.003, 95% CI 1.225–29.417; p = 0.027).
The classical morphologic LAA classification is sometimes difficult to determine, with variable and low interobserver agreement.11,30,31 Figure 3 demonstrates how different perspectives of the same LAA may change the presumed morphology. The classic classification may therefore not represent the thrombotic risk reliably. In our study, two observers blindly classified the LAAs according to the classic morphology and according to the presence of large secondary lobes (regarded as complex LAA). The classification of the LAA according to the presence of large secondary lobes yielded higher intra-observer agreement (k = 0.80 (95% CI 0.64–0.96) compared to the classic morphological LAA classification k = 0.72 (95% CI 0.0.56–0.88). In previous studies, this issue was partially addressed by simply diving LAA morphology into CW or non-CW; however, we observed that more than 50% of LAAs which would be considered thrombogenic according to a non-CW classification, would be rather considered as having a more benign nature according due to absence of large secondary lobes. In the same way, about one-fifth of CW LAAs would be considered complex, which may imply a more thrombogenic structure. We therefore claim that the traditional LAA morphology might be of less clinical importance, but rather LAA complexity and the presence of large secondary lobes, in which reduced velocity of blood flow and blood stasis may promote a pro-thrombotic environment inside the LAA.25–27
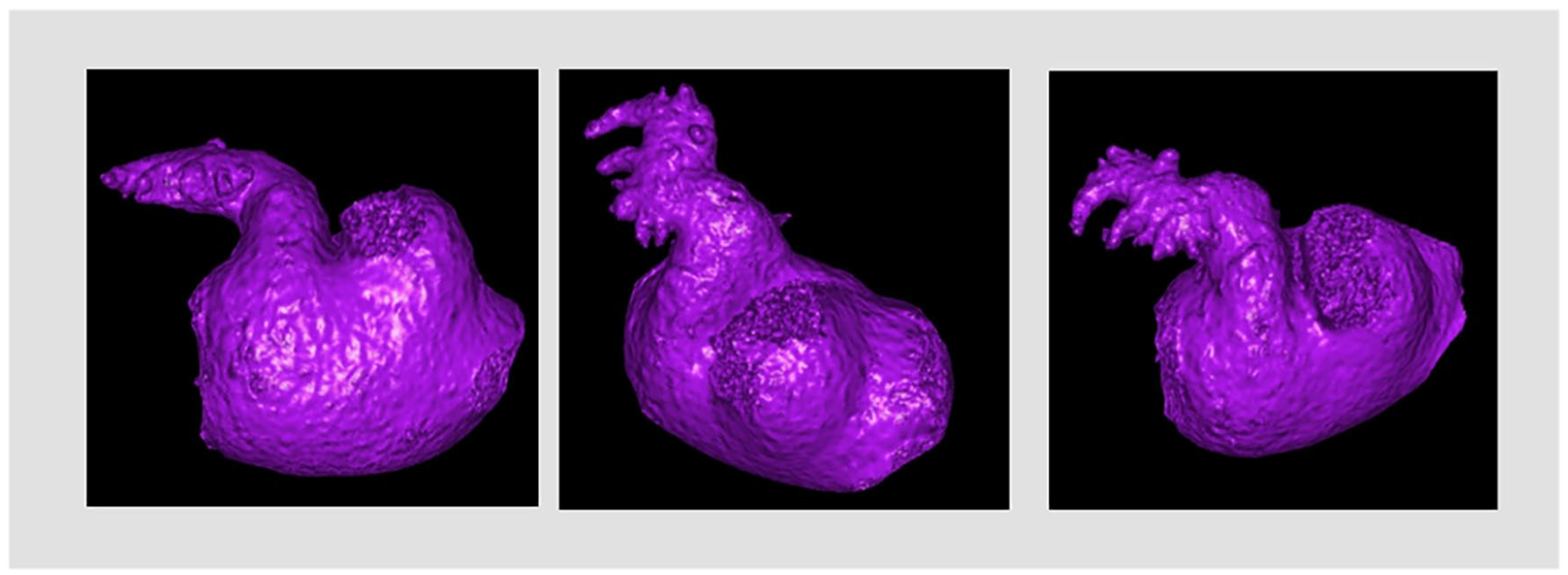
Different views of the same LAA may lead to different morphologic interpretations.
ESUS constitutes a heterogenic group of patients, as emphasized also by the failure to demonstrate clear benefit of routine use of oral anticoagulation in clinical trials.5–7 Thus, different systemic and anatomic biomarkers can potentially delineate stroke etiology and may provide guidance in choosing best recurrent stroke prevention strategies. If indeed the LAA complexity contributes to embolic stroke risk in the absence of AF as we suggest in this hypothesis-generating study, LAA may become in the future a direct therapeutic target in ESUS prevention – for example, by planning a patient specific anti-platelet/anti-coagulation regimen and/or mechanical closure of the LAA. This may also indicate that future secondary-prevention studies in ESUS may need to use stricter criteria for choosing the most proper patients for each prevention strategy. 32
Limitations
The study has the inherent limitation of an observational study, which is part of a registry that enrolls consecutive patients with ESUS evaluated with CCT. Moreover, we did not have echocardiographic left atrial volume index (LAVI) measurements for the AF patients, a parameter which has been shown to be associated both with cardioembolic stroke and AF detection in ESUS patients 33 ; however, the use of three dimensional and multiplanar CCT data may better define the complex LA and LAA morphology. Another limitation is the lack of flow measurements in the LAAs, which are important to support the theory of slower flow velocities within complex LAA structures. In addition, we don’t have a routine long-term follow-up for cardiac rhythm monitoring of ESUS patients, which implies the possibility that subclinical AF remained undetected in some patients.
Conclusion
ESUS patients have a high prevalence of complex LAA morphology, that is, cauliflower morphology and an increased number of secondary branches. The classic morphologic classification, particularly into CW and non-CW morphologies, may not reliably represent the associated thrombotic risk. Complex LAA morphology might be a congenital risk factor for LAA thrombus formation, independently of other clinical risk factors. Further prospective studies are warranted to confirm the association between LAA morphology and thromboembolic risk in ESUS patients and to guide secondary prevention strategies in ESUS patients with complex LAA anatomy.
List of abbreviations
AF: atrial fibrillation
CCT: cardiac computer tomography
CW: Chicken wing
ESUS: embolic stroke of undermined source
IS: ischemic stroke
LA: left atrium
LAA: left atrial appendage
TTE: transthoracic echocardiography
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Ethics Committee of Rabin Medical Center.
Informed consent
Informed consent was not sought for the present study because of its retrospective observational nature.
Trial registration
Not applicable.
Guarantor
EA
Contributorship
INA conceived the study, did the LAA analysis and wrote the first draft of the manuscript. RB was involved in protocol development and neurological aspects. JN and EA were involved in the neurological aspects. AL was involved in study planning and did the statistical analysis. OM, GS, NS and AS were involved in imaging acquisition and analysis. AH, RK, and EA were involved in protocol development and supervision. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
