Abstract
Introduction:
Previous reports and meta-analyses derived from small case series reported a mortality rate of up to 40% in patients with coronavirus disease 2019 associated cerebral venous thrombosis (COVID-CVT). We assessed the clinical characteristics and outcomes in an international cohort of patients with COVID-CVT.
Patients and methods:
This was a registry study of consecutive COVID-CVT patients diagnosed between March 2020 and March 2023. Data collected by the International Cerebral Venous Thrombosis Consortium from patients with CVT diagnosed between 2017 and 2018 served as a comparison. Outcome analyses were adjusted for age and sex.
Results:
We included 70 patients with COVID-CVT from 23 hospitals in 15 countries and 206 controls from 14 hospitals in 13 countries. The proportion of women was smaller in the COVID-CVT group (50% vs 68%,
Conclusion:
In contrast to previous studies, the in-hospital mortality rate and functional outcomes during follow-up did not differ between COVID-CVT patients and the pre-COVID-19 controls.

Introduction
Coronavirus disease 2019 (COVID-19) induces a hypercoagulable state which may cause arterial and venous thrombosis, including cerebral venous thrombosis (CVT).1,2 Information on CVT associated with COVID-19 (COVID-CVT) is currently primarily derived from case reports and small case series, while data from larger cohorts with detailed patient-level data are still lacking.3–19 In individuals infected with COVID-19, the incidence of CVT is estimated at 42.8 per million, 5 the incidence being even higher among hospitalized patients with COVID-19 (231 per million). For comparison, the CVT incidence from the pre-COVID era was estimated to be 13.9–20.2 per million.20,21 COVID-CVT was reported to present with concomitant arterial ischemic stroke, but also with arterial and venous occlusions at unusual sites. 4 The temporal relationship between CVT and COVID-19 was described as heterogeneous, with CVT being the first symptom of COVID-19 in some patients,6–8 while in other patients COVID-19 resolved before the symptoms of CVT started.9–12 The severity of COVID-19 in patients with COVID-CVT also varied, ranging from asymptomatic or mildly ill patients,13–15 to severe or critically ill ones. 16 The in-hospital mortality of COVID-CVT was reported to be around 15%–40%, which is high compared to the in-hospital mortality of CVT unrelated to COVID-19 (around 4.5%–5%),22–25 We aimed to describe baseline characteristics, treatment, clinical course, and outcomes of consecutive COVID-CVT patients and to compare these to CVT cases from the pre-COVID era.
Methods
Patient selection
We conducted an international registry study of consecutive patients with COVID-CVT. We invited investigators to participate in this research initiative through the network of the International Cerebral Venous Thrombosis Consortium (ICVTC) 26 and requested the investigators to further disseminate the study protocol within their network. This study was approved by the Ethics Committee of the Canton of Bern, Switzerland (KEK 2021–02206). Each participating center was responsible for complying with the legal requirements of the local ethics committee and hospital regulations for study participation.
Patients were eligible for inclusion if they met the following inclusion criteria: 1) age of 18 years or older, 2) confirmed CVT diagnosis by neuroimaging or autopsy, 27 and 3) clinically manifest COVID-19 infection or asymptomatic infection confirmed by polymerase chain reaction (PCR) nasopharyngeal swab testing within 90 days prior to or at the time of CVT diagnosis. The 90-day time frame was chosen based on data showing the occurrence of CVT and the continued risk of venous thrombosis several months after a COVID-19 infection.10,28 Data of some included patients were described in previous publications.17,18,29
This study was conducted in accordance with the STROBE guidelines (Strengthening the Reporting of Observational Studies in Epidemiology).
Data collection and definitions
For the COVID-CVT group, data were collected from 1st of March 2022 to 17th April 2023 and included both prospectively and retrospectively enrolled patients diagnosed between 25th March 2020 and 4th March 2023. A standardized electronic case report form was used (REDCap 12.4.12, Vanderbilt University), which included baseline characteristics, risk factors for CVT and COVID-19, clinical symptoms, laboratory data, treatment, complications during hospitalization, in-hospital outcome, and 3- and 6-month follow-up data. Queries were sent to participating centers regarding baseline characteristics and outcomes. For the control group, data on consecutive patients diagnosed between 2017 and 2018 were extracted from the International CVT Consortium registry. 26 COVID-19 severity was classified as asymptomatic or presymptomatic, mild, moderate, severe, and critical according to National Institute of Health (NIH) COVID-19 treatment guidelines (Supplemental Table 1). 30 Overweight was defined as BMI > 25 kg/m2, and obesity as BMI > 30kg/m2. History of cancer was defined as cancer in the past 10 years. 31 Coma was defined as Glasgow Coma Scale (GCS) lower than 9 and altered mental state as a GCS lower than 14 points. For thrombus load, the number of sinuses or veins that were thrombosed were added up. 32 Neurological worsening during hospitalization was defined as a change of baseline National Institutes of Health Stroke Scale (NIHSS) + 4 points or of GCS –2 points. The cut-off of NIHSS + 4 points was previously used in ischemic stroke studies. 33 A drop of GCS of 2 points was chosen to capture relevant neurological worsening; a drop of one point only might be due to minute fluctuations of vigilance or factors that do not necessarily prompt further evaluation for new intracerebral pathology. Clinical outcome at discharge and at 3- and 6-month follow-up was assessed using the modified Rankin Scale (mRS). Functional independency was defined as mRS 0–2 and functional dependency and death as mRS 3–6.
Statistical analysis
Given the rarity of COVID-CVT, we did not calculate the sample size in advance. Instead, we aimed to assemble a convenience sample of all patients reported in the database by the study investigators. Categorical data are presented as counts and proportions, continuous data as means and standard deviations or medians and interquartile ranges, as appropriate. In case of missing values, valid percentages are shown. No imputation of missing data was performed. Confidence intervals for proportions were calculated using the Wilson formula.
We compared the available variables on baseline characteristics, risk factors, clinical presentation, imaging findings, hospitalization data, and treatment between patients with COVID-CVT and controls. Furthermore, we compared the in-hospital and post-acute outcomes of cases to those of the control group. For the univariable analysis, Chi-square and Fisher’s exact tests were used for comparisons of categorical data, as appropriate, and for continuous variables, Mann-Whitney
We performed a subgroup analysis to compare the clinical presentation, treatment, and outcome in patients with different COVID severities. We dichotomized COVID severity into low COVID severity, comprising asymptomatic, mild, and moderate COVID, and high COVID severity, comprising severe and critical COVID-19. 30 No formal statistical analysis was performed for this subgroup analysis.
Results
Data were collected from 21 hospitals in 13 countries. Of 71 COVID-CVT patients reported in the registry, one was excluded due to duplicate reporting, leaving 70 patients, diagnosed between 25th March 2020 and 4th March 2023, for analysis. In the control group, 206 consecutive patients with CVT from 14 hospitals in 13 countries were included (Supplemental Tables 2 and 3).
Baseline characteristics and risk factors
Median age was 43 (IQR 36–54) and 44 (IQR 31–53) for the COVID-CVT and control patients, respectively. The proportion of women was smaller in the COVID-CVT group (50% vs 68%,
Baseline characteristics of patients with CVT associated with COVID-19 and controls from the pre-COVID era.
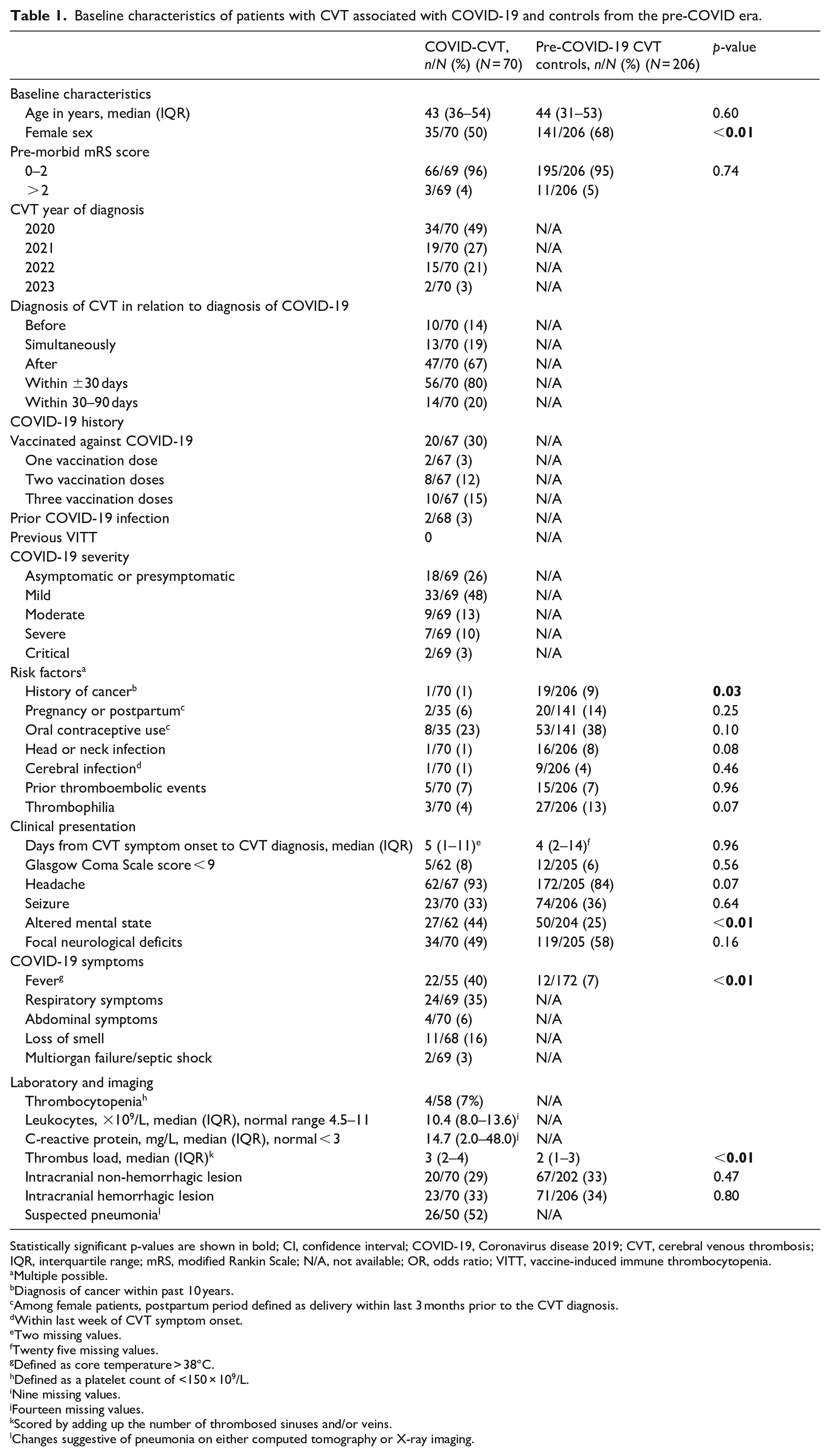
Statistically significant p-values are shown in bold; CI, confidence interval; COVID-19, Coronavirus disease 2019; CVT, cerebral venous thrombosis; IQR, interquartile range; mRS, modified Rankin Scale; N/A, not available; OR, odds ratio; VITT, vaccine-induced immune thrombocytopenia.
Multiple possible.
Diagnosis of cancer within past 10 years.
Among female patients, postpartum period defined as delivery within last 3 months prior to the CVT diagnosis.
Within last week of CVT symptom onset.
Two missing values.
Twenty five missing values.
Defined as core temperature > 38°C.
Defined as a platelet count of <150 × 109/L.
Nine missing values.
Fourteen missing values.
Scored by adding up the number of thrombosed sinuses and/or veins.
Changes suggestive of pneumonia on either computed tomography or X-ray imaging.
Clinical presentation and imaging
In 49/69 (71%) of COVID-CVT patients, the primary reason for hospitalization was CVT, in 7/69 (10%) COVID-19 and in 13/69 (19%) both CVT and COVID-19. COVID-19 was diagnosed in 37/68 (54%) patients before CVT symptom onset, in 24/68 (35%) after CVT symptom onset, and in 7/68 (10%) at the same time as the onset of symptoms. COVID-19 was diagnosed a median of 3 days before (IQR [ 18 days prior]–[2 days after]) CVT symptom onset. The COVID-19 severity was asymptomatic or pre-symptomatic in 18/69 (26%), mild in 33/69 (48%), moderate in 9/69 (13%), severe in 7/69 (10%), and critical in 2/69 (3%) patients.
In the COVID-CVT group the median NIHSS was higher than in controls (1 [IQR 0–8] vs 0 [0–4],
In COVID-CVT patients, the median value of d-dimer levels was 1562 ng/ml (IQR 966–6300), which is above the normal threshold (<500 ng/ml). The median values of the international normalized ratio, activated thromboplastin time, thrombin time, and platelet count were within normal range. Details on clinical presentation, imaging, and laboratory values at admission are given in Tables 1 and 2.
Treatment and outcomes of patients with CVT associated with COVID-19 and controls from the pre-COVID era.
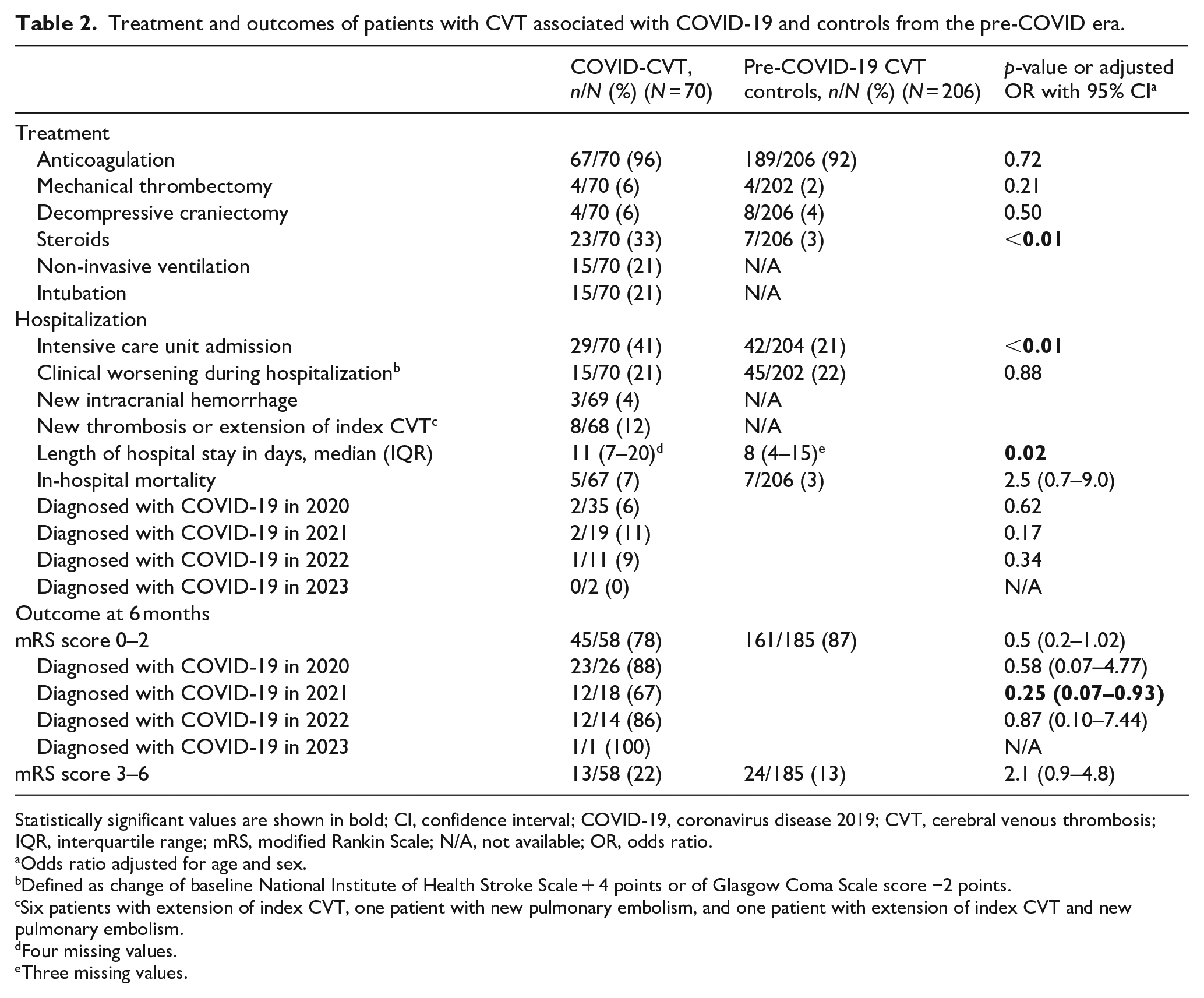
Statistically significant values are shown in bold; CI, confidence interval; COVID-19, coronavirus disease 2019; CVT, cerebral venous thrombosis; IQR, interquartile range; mRS, modified Rankin Scale; N/A, not available; OR, odds ratio.
Odds ratio adjusted for age and sex.
Defined as change of baseline National Institute of Health Stroke Scale + 4 points or of Glasgow Coma Scale score −2 points.
Six patients with extension of index CVT, one patient with new pulmonary embolism, and one patient with extension of index CVT and new pulmonary embolism.
Four missing values.
Three missing values.
Treatment and hospitalization
Anticoagulant treatment was started in 67/70 (96%) COVID-CVT patients, which was similar to controls (189/206 [92%]). The proportion of patients who worsened during hospitalization was also similar (15/70 [21%] vs 45/202 [22%]). In 10/70 (14%) of the COVID-CVT cases, the worsening was due to CVT, in 7/70 (10%) due to COVID-19 and in 3/70 (4%) due to other reasons.
A higher proportion of COVID-CVT patients required treatment in an intensive care unit (ICU) compared to controls (29/70 [41%] vs 42/204 [21%],
Outcomes
The in-hospital mortality was similar in the COVID-CVT group (5/67 [7%, 95% CI 3–16]) compared to controls (7/206 [3%, 2–7], aOR 2.6 [95% CI 0.7–9]). The causes of death in the COVID-CVT group were brain herniation in two and cardiac and/or respiratory failure in three patients. Of the COVID-CVT patients, 46/67 (69%, 95% CI 57–78) were functionally independent and 21/67 (31%, 22–43) functionally dependent or dead at discharge. No patient had a recurrent venous cerebral or extracerebral thrombosis at follow-up. Among the surviving patients, at 3-months two additional COVID-CVT patients had died, both due to cardiac and respiratory failure. No patient died between the 3- and 6-month follow-up. At 6-months, compared to controls, a similar proportion of COVID-CVT patients was functionally independent (45/58 [78%, 95% CI 65–86] vs 161/185 [87%, 81–91], adjusted OR 0.5 [95% CI 0.2–1.02]).
After only including patients with CVT diagnosis within 14 days of COVID-19 diagnosis (42/70 [60%]), the adjusted OR for in-hospital mortality (aOR 3.76 [95% 0.95–15]) and functional independence at 6 months (aOR 0.45 [95% CI 0.13–1.58]) did not change. Patients diagnosed with COVID-19 in 2021 were less likely to be functionally independent at 6 months (12/18 [67%, 95% CI 44–84] vs 161/185 [87%, 81–91], aOR 0.25 [0.07–0.93]). The outcomes for the years 2020, 2022, and 2023 did not differ in COVID-CVT patients compared to controls (see Table 2, Figures 1 and 2).
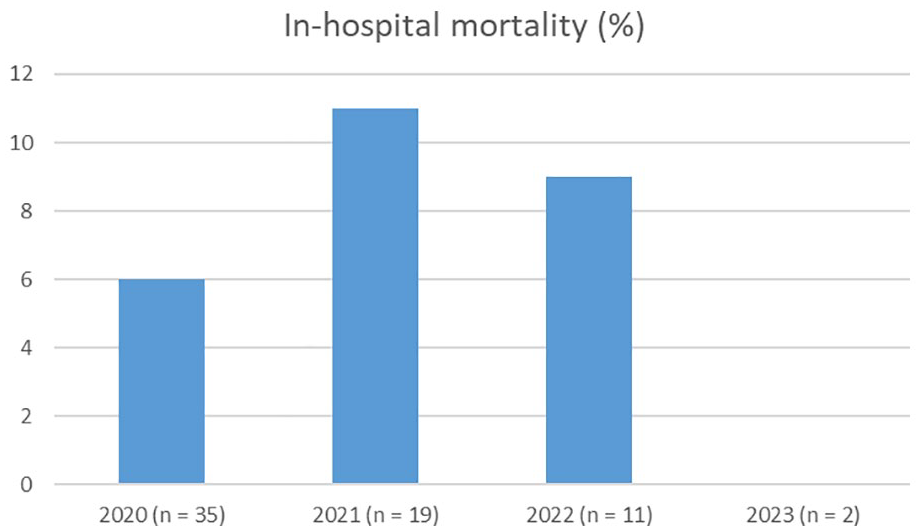
Distribution of in-hospital mortality over the years 2020–2023 in patients with cerebral venous thrombosis associated with COVID-19.
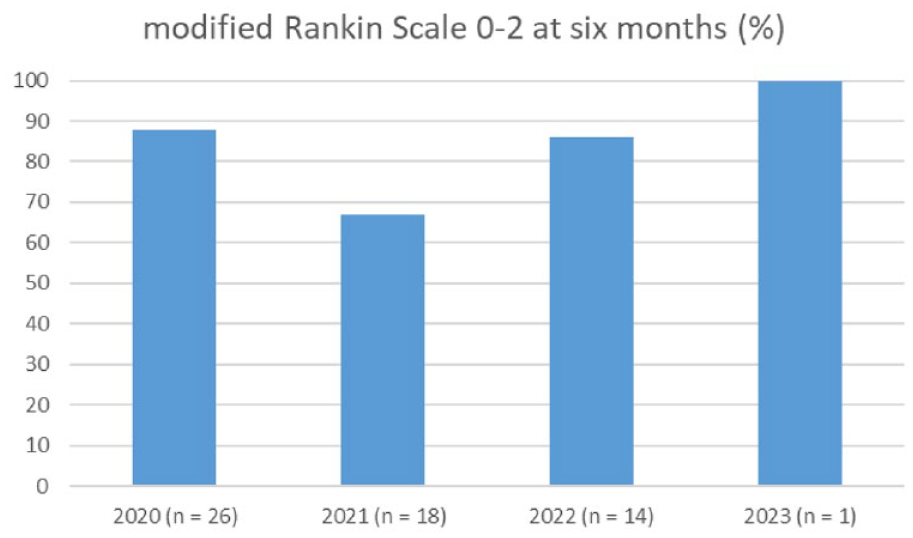
Distribution of modified Rankin Scale 0–2 at 6 months over the years 2020–2023 in patients with cerebral venous thrombosis associated with COVID-19.
Association of COVID severity with clinical presentation, treatment, and outcome
Compared to COVID-19 cases with low severity, patients with high severity COVID-19 were older (59 years [IQR 42–64] vs 41 years [IQR 35–51]) and predominantly male (8/9 [89%] vs 27/61 [44%]) (see Table 3). Furthermore, patients with high COVID-19 severity were more often treated with steroids (8/9 [89%] vs 15/61 [25%]), had more often clinical worsening during hospitalization (5/9 [56%] vs 10/61 [16%]) and were more often functionally dependent at 6 months (3/6 [50%, 95% CI 19–81] vs 3/52 [6%, 2–16]).
Clinical presentation, treatment, and outcome of patients with CVT associated with COVID-19 stratified by COVID-19 severity.
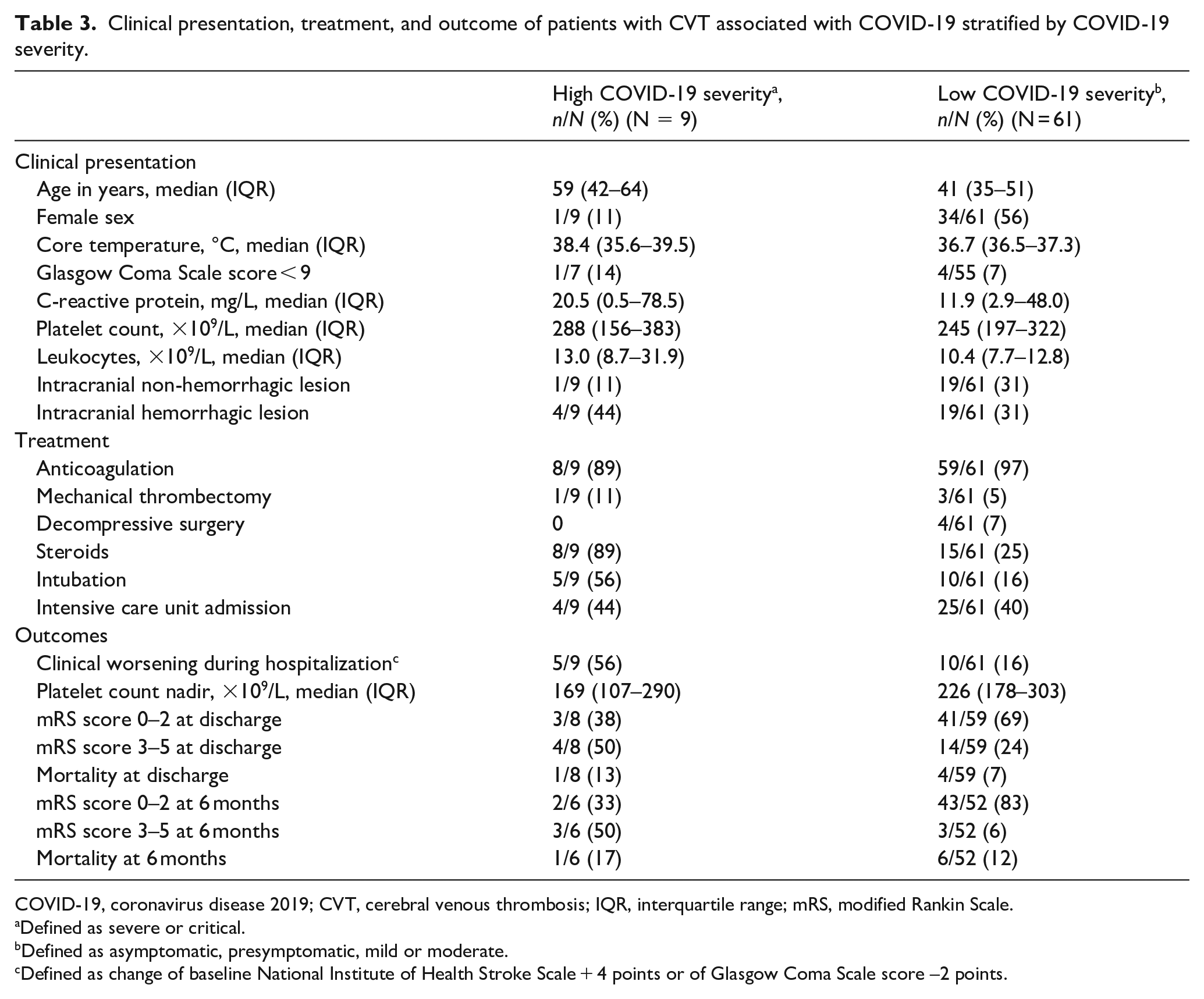
COVID-19, coronavirus disease 2019; CVT, cerebral venous thrombosis; IQR, interquartile range; mRS, modified Rankin Scale.
Defined as severe or critical.
Defined as asymptomatic, presymptomatic, mild or moderate.
Defined as change of baseline National Institute of Health Stroke Scale + 4 points or of Glasgow Coma Scale score –2 points.
Discussion
The main findings of our study are that in-hospital mortality and post-acute outcomes of COVID-CVT patients are similar compared to pre-COVID CVT patients, despite COVID-CVT patients having a higher baseline NIHSS score, a higher incidence of altered mental status, higher median thrombus load at admission, a higher frequency of ICU treatment, and longer hospital stays. Our findings are in stark contrast to previous reports on in-hospital mortality of patients with CVT associated with COVID-19 of up to 40%.2,8,12,17,24,25,34 It is possible that the results of previous studies were driven by reporting of cases with more severe COVID-19. Therefore, our contrasting results suggest that mainly the severity of COVID-19, and not the infection with SARS-CoV2 or CVT alone, likely predicts unfavorable outcomes in CVT patients. 36 Although limited by the small sample sizes, our data suggest that patients with more severe COVID-19 might have a worse long-term clinical outcome. The worse outcome of patients with CVT diagnosed with COVID-19 in 2021 might be explained by the emergence of the delta variant of SARS-CoV-2, which is known to cause more severe COVID-19.35,37
Although non-significant, the mortality rate in COVID-CVT was higher than in controls (7% vs 3%). This finding may have clinical significance, indicating that COVID-19 contributes to the burden of CVT, in-line with recent findings on impact of COVID-19 on CVT and ischemic stroke outcomes.24,38
In our study, the majority of COVID-CVT patients (87%) had asymptomatic, mild or moderate disease. Additionally, the baseline characteristics were largely similar in COVID-CVT and controls, likely explaining the similar outcomes. There were, however, few differences that should be noted.
First, in line with previous reports, 25 we observed a balanced sex distribution in COVID-CVT patients compared with controls, who were predominantly women. Female-specific risk factors likely play a smaller role in COVID-CVT than in CVT unrelated to COVID-19, as reflected by a lower proportion of women taking oral contraceptives or being in the postpartum period. Other traditional risk factors for CVT were less frequent in the COVID-CVT group compared to controls as well. This is likely due to COVID-19 being the main risk factor for CVT in the COVID-CVT group rather than traditional risk factors. In patients with cancer, the COVID-19 severity and mortality are higher compared to non-cancer patients, possibly further contributing to an underrepresentation of patients with cancer in the COVID-CVT group. 39
Second, although the median NIHSS score was higher in COVID-CVT patients compared to controls, the absolute difference is unlikely to be clinically relevant. Patients with COVID-CVT had a significantly higher frequency of altered mental state at presentation than CVT patients from the pre-COVID era. Given that altered mental state was found in almost 10% of COVID-19 patients even in the absence of CVT, 40 the co-morbidity of COVID-19 and CVT probably explains our findings. Third, the higher median thrombus load in COVID-CVT patients could be explained by the prothrombogenic effects of SARS-CoV-2 through the induction of pro-inflammatory cytokines 40 but also by direct activation of the coagulation cascade by the virus through endothelial activation. 1 However, the clinical significance of this finding is unclear, as the frequency of hemorrhagic and non-hemorrhagic lesions did not differ between the groups. Fourth, compared to controls, twice as many COVID-CVT patients required ICU treatment and the length of hospital stay was longer. The additional morbidity of COVID-19 to CVT, due to possible systemic complications such as acute respiratory distress syndrome at least in part explains these results. Nevertheless, more COVID-CVT patients were treated in the ICU for CVT than for COVID-19 complications (24% vs 6%).
The similar outcomes in COVID-CVT patients and controls might be further explained by increased awareness and improved treatment over time.
To our knowledge, this is the largest cohort of COVID-CVT patients to date, with detailed information on clinical presentation, hospitalization, treatment, and in-hospital and long-term outcomes. Patients were consecutively enrolled in the study from different countries around the world, resulting in a representative depiction of the clinical profile of COVID-CVT. The inclusion of a control group of pre-COVID patients contributes to a better understanding of COVID-CVT in a broader context and excludes any influence of undiagnosed COVID. Our study has several limitations. First, the number of included patients with COVID-CVT is small, which limits adjustment for all potential confounding variables. Second, there was no central adjudication of the data, potentially introducing bias, even though we examined endpoints that are difficult to misinterpret, such as mortality and binary functional outcomes. We did not collect standardized data on, for example, repeated imaging during hospitalization. Third, one-fifth (19%) of the COVID-CVT group were lost to follow-up. Fourth, we did not have data on the SARS-CoV-2 strain, which might be relevant, given that some strains cause more severe COVID-19 than others. Besides less virulent virus variants, evolving treatments and high SARS-CoV-2 vaccination rates might have reduced the COVID-19 severity over time resulting in improved outcomes of COVID-CVT. Fifth, patients in the control group were from an earlier time period compared to the COVID-CVT group, possibly limiting comparability. The pandemic forced the hospitals to select patients, leading to a drop of hospitalizations and a change of in-hospital stroke care, 41 possibly introducing a selection bias for the COVID-CVT group. However, CVT requires urgent care and affected patients are less likely to be deferred. One study did not find a drop in CVT-hospitalizations during the first year of the COVID-19 pandemic. 24 In addition, by including patients with CVT from an earlier period we excluded confounding by undiagnosed COVID-19, COVID-19 vaccination, and past asymptomatic SARS-CoV-2 infections. Lastly, the patients from the control group were from different geographical regions than COVID-CVT cases. For example, there were no Asian countries contributing to the control group. Due to the limited sample size, we did not perform an analysis including only patients from similar countries.
In conclusion, COVID-CVT patients were more often male, slightly less often had traditional risk factors for CVT, and were in a slightly more severe clinical state compared to pre-COVID-19 CVT patients. In contrast to previous studies, the in-hospital mortality rate and functional outcomes during follow-up did not differ between COVID-CVT patients and the pre-COVID-19 controls.
Supplemental Material
sj-doc-2-eso-10.1177_23969873241241885 – Supplemental material for Characteristics and outcomes of cerebral venous thrombosis associated with COVID-19
Supplemental material, sj-doc-2-eso-10.1177_23969873241241885 for Characteristics and outcomes of cerebral venous thrombosis associated with COVID-19 by Adrian Scutelnic, Anita van de Munckhof, Angel T. Miraclin, Sanjith Aaron, Sajid Hameed, Mohammad Wasay, Oxana Grosu, Katarzyna Krzywicka, Mayte Sánchez van Kammen, Erik Lindgren, Tiago Moreira, Roberto Acampora, Alberto Negro, Theodoros Karapanayiotides, Shadi Yaghi, Anna Revert, Elisa Cuadrado Godia, Sebastian Garcia-Madrona, Paolino La Spina, Francesco Grillo, Fabrizio Giammello, Thanh N. Nguyen, Mohamad Abdalkader, Alina Buture, Maria Sofia Cotelli, Nicolas Raposo, Georgios Tsivgoulis, Paolo Candelaresi, Antonio Ciacciarelli, Joshua Mbroh, Tatiana Batenkova, Pasquale Scoppettuolo, Marialuisa Zedde, Rosario Pascarella, Kateryna Antonenko, Espen S. Kristoffersen, Johanna A. Kremer Hovinga, Katarina Jood, Diana Aguiar de Sousa, Sven Poli, Turgut Tatlisumak, Jukka Putaala, Jonathan M. Coutinho, José M. Ferro, Marcel Arnold and Mirjam R. Heldner in European Stroke Journal
Supplemental Material
sj-docx-1-eso-10.1177_23969873241241885 – Supplemental material for Characteristics and outcomes of cerebral venous thrombosis associated with COVID-19
Supplemental material, sj-docx-1-eso-10.1177_23969873241241885 for Characteristics and outcomes of cerebral venous thrombosis associated with COVID-19 by Adrian Scutelnic, Anita van de Munckhof, Angel T. Miraclin, Sanjith Aaron, Sajid Hameed, Mohammad Wasay, Oxana Grosu, Katarzyna Krzywicka, Mayte Sánchez van Kammen, Erik Lindgren, Tiago Moreira, Roberto Acampora, Alberto Negro, Theodoros Karapanayiotides, Shadi Yaghi, Anna Revert, Elisa Cuadrado Godia, Sebastian Garcia-Madrona, Paolino La Spina, Francesco Grillo, Fabrizio Giammello, Thanh N. Nguyen, Mohamad Abdalkader, Alina Buture, Maria Sofia Cotelli, Nicolas Raposo, Georgios Tsivgoulis, Paolo Candelaresi, Antonio Ciacciarelli, Joshua Mbroh, Tatiana Batenkova, Pasquale Scoppettuolo, Marialuisa Zedde, Rosario Pascarella, Kateryna Antonenko, Espen S. Kristoffersen, Johanna A. Kremer Hovinga, Katarina Jood, Diana Aguiar de Sousa, Sven Poli, Turgut Tatlisumak, Jukka Putaala, Jonathan M. Coutinho, José M. Ferro, Marcel Arnold and Mirjam R. Heldner in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AS reports a grant from the Swiss Heart Foundation. MRH reports grants from SITEM Research Support Funds and Swiss National Science Foundation, Swiss Heart Foundation, not directly related to this manuscript. MA reports personal fees from AstraZeneca, Bayer, Bristol Myers Squibb, Covidien, Daiichi Sankyo, Medtronic, Novartis, Pfizer, and Amgen. JMC has received grants paid to his institution from Boehringer Ingelheim and Bayer, and payments paid to his institution for data safety monitoring board participation by Bayer. JMF has received personal fees from Boehringer Ingelheim, Bayer, and Daiichi Sankyo as well as grants from Bayer. DAS reports travel support from Boehringer Ingelheim, speaker fees from Bayer, and Advisory Board participation for AstraZeneca. TT has received personal fees from Argenx, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Inventiva, and Portola Pharma. NR received consultant fees from Novartis. KJ has received academic grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF agreement (ALFGBG 965417) for research on CVT. SP received research support from BMS/Pfizer, Boehringer-Ingelheim, Daiichi Sankyo, European Union, German Federal Joint Committee Innovation Fund, and German Federal Ministry of Education and Research, Helena Laboratories and Werfen as well as speakers’ honoraria/consulting fees from Alexion, AstraZeneca, Bayer, Boehringer-Ingelheim, BMS/Pfizer, Daiichi Sankyo, Portola, and Werfen (all outside the submitted work). TNN reports advisory board Idorsia, Brainomix. KA reports a grant from Swiss National Science Foundation and Medtronic advisory board participation in 2022, not related to this manuscript. EL has received academic grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF agreement (ALFGBG 942851), Swedish Neurologic Society, Elsa and Gustav Lindh’s Foundation, Wennerströms’ Foundation, P-O Ahl’s Foundation and Rune and Ulla Amlöv’s Foundation for research on CVT. All other co-authors report no disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of the Canton of Bern, Switzerland (KEK 2021-02206). Each participating center was responsible for complying with the legal requirements of the local ethics committee and hospital regulations for study participation.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article. Each participating center was responsible for complying with the legal requirements of the local ethics committee and hospital regulations for study participation.
Guarantor
MRH.
Contributorship
MA, MRH, JC, JF, SP, TT, JP, and DAS contributed to the concept and design of the study. All authors contributed to the acquisition, analysis or interpretation of data. AM, AS, MA, MRH, JC, JF, DAS, KK, MSK, EL, SP, TT, and JP drafted the manuscript and all authors revised it for intellectual content. All authors approved the manuscript to be published.
ORCID iDs
Data availability
Anonymized original data will be made available by reasonable request from any qualified investigator.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
