Abstract
Introduction:
Severe sleep apnea (SA) affects one-third of stroke patients. Sleepiness, one of the cardinal symptoms of SA, negatively impacts functional stroke outcomes. The impact of continuous positive airway pressure (CPAP) on post-stroke sleepiness is poorly described. We aimed to compare through a propensity score matching the trajectories of self-reported sleepiness post-stroke with matched individuals including SA patients adherent or not to CPAP.
Patients and methods:
Sixty five (80.2%) ischemic stroke and 16 (19.8%) TIA patients (median [Q1;Q3] age = 67.0 [58.0;74.0] years, 70.4% male, body mass index [BMI] = 26.1 [24.5;29.8] kg·m−2, admission NIHSS = 3.0 [1.0;5.0]), with polysomnography and an Epworth Sleepiness Scale (ESS) performed within 1 year following stroke and with a follow-up ESS (delay = 236 [147;399] days) were included in the analysis. A 2:1 propensity score matching based on age, gender, BMI, and the apnea-hypopnea index was performed to identify 162 matched individuals referred for SA suspicion, free of stroke or TIA. Multivariable negative binomial regression models were performed to identify the determinants of sleepiness trajectories post-stroke.
Results:
Baseline ESS was comparable between stroke/TIA and matched individuals (median [Q1; Q3] ESS = 7 [4;10] versus 6 [4;10],
Discussion and conclusion:
Sleepiness improvement was unexpectedly higher in stroke patients compared to matched individuals, with no significant influence of comorbid SA and CPAP on its trajectory. Sleepiness may not be primarily indicative of SA in stroke or TIA patients.

Keywords
Introduction
Sleep-wake disturbances are frequent following stroke,1–4 and are acknowledged risk factors for both ischemic stroke and poor cardiovascular and functional outcome after stroke.3,4 Sleep apnea (SA), a sleep-related breathing disorder characterized by recurrent complete (apnea) or incomplete (hypopnea) cessation of airflow during sleep is one of the most common chronic diseases, affecting nearly a billion people worldwide. 5 Following ischemic stroke, severe SA is diagnosed in one-third of patients,1,2 and is associated with poorer functional prognosis and increased risk of cardiovascular morbidity and mortality. 2
Excessive daytime sleepiness (EDS), one of the manifestations of hypersomnolence, is defined as an inability to stay awake during the normal wake period of the day. 6 EDS is a cardinal symptom of obstructive SA, of which severity is related to obstructive SA severity.7,8 Continuous positive airway pressure (CPAP) treatment, the reference treatment for obstructive SA is effective in reducing self-reported daytime sleepiness in a majority of SA patients. 9 Screening for EDS is mostly based on self-administered questionnaires such as the Epworth sleepiness scale (ESS), 10 and EDS is retained for ESS scores>10/24. 11 The prevalence of post-stroke EDS ranges between 6% and 49.5%, depending on the type of stroke and the delay between stroke and ESS assessment [See Ding et al. 12 for review of 18 studies]. Data regarding the temporal evolution of post-stroke EDS are scarce. Denis et al. 13 in a prospective study including 90 patients with supra-tentorial ischemic stroke, found EDS in 16.7% of patients at 15 ± 4 days following stroke, and in 18.7% at 3 months. Duss et al., 14 in a prospective longitudinal study, found the prevalence of EDS ranging between 10% and 14% from 1 month to 2 years post-stroke and transient ischemic attack (TIA), with a significant decrease in mean ESS scores at 2 years compared to 1 month after stroke/TIA. EDS is of clinical and prognosis relevance, as it is associated with an increased stroke risk 15 and as it negatively affects daytime functional performances, quality of life, and cognitive functioning post-stroke. 16 Stroke lesion localizations, and especially subcortical, ponto-mesencephalic lesions, 12 and thalamic stroke,17,18 as well as restless legs syndrome, diabetes, and higher body mass index are factors associated with post-stroke EDS.13,19 Surprisingly, for a given severity of obstructive SA, patients with stroke reported less EDS in comparison with OSA free of stroke. 20
The trajectories of self-reported sleepiness post-stroke and interactions with comorbid SA and CPAP treatment have not been studied. We aimed to compare through a propensity score matching the trajectories of self-reported sleepiness post-stroke with matched individuals including sleep apnea patients adherent or not to CPAP.
Methods
Study design and population
This is a propensity score matching study of patients referred for SA suspicion, who were prospectively included in the
This database is managed in compliance with the European General Data Protection Regulation rules. This study was conducted in accordance with applicable good clinical practice requirements in Europe, French law, the ethical principles of the World Medical Association Declaration of Helsinki, and reported in compliance with STROBE guidelines.
Eighty one first ischemic stroke or TIA patients, aged between 18 and 85 years, with a BMI ⩾ 18.5 and <40 kg·m−2, polysomnography (PSG) performed within 1 year following stroke (Visit 1) and at least two ESS evaluations (one by the time of PSG [Visit 1] and the second within a 1.5 to 18-month follow-up period [Visit 2]) were identified. We decided to include both ischemic stroke and TIA patients since the two conditions share the same pathophysiological mechanisms and to ease comparisons with previous studies conducted on the same topic. 14
A 2:1 nearest neighbor propensity score matching 21 (using a collider of 0.1), an experimental method used to construct an artificial control group by matching each TIA/Stroke patient with two non-TIA/stroke patients of similar characteristics, was performed for age, gender, BMI, and AHI to identify 162 matched individuals in the research database (Figure 1 and Supplemental Figure S1). The burden of comorbidities was similar in the two groups and did not require inclusion in the propensity score matching. The choice of including twice as many controls as stroke or TIA patients aimed at enhancing statistical power and reducing bias through a better adjustment for confounders.
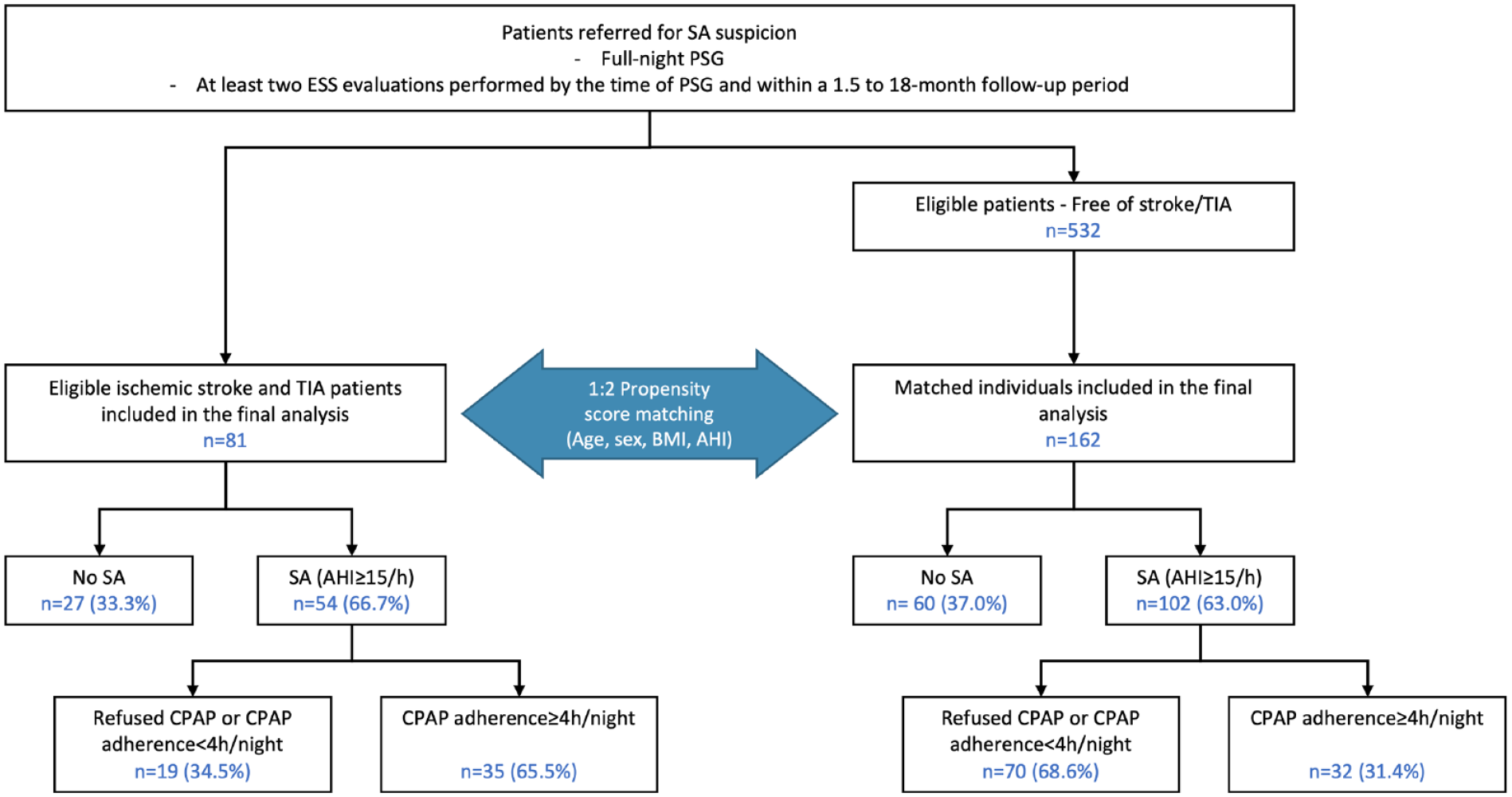
Study flow-chart.
Evaluations
Ischemic stroke/TIA was diagnosed by trained stroke neurologists, by the time of acute stroke care, based on clinical and imaging assessments (magnetic resonance imaging or computed tomography imaging). Acute stroke data were extracted from the patient’s medical records stored in the medical database of Grenoble Alpes University Hospital (Supplementary information can be found in Supplemental Material).
At visit 1, a certified sleep specialist carried out a standardized evaluation for all patients referred to the Sleep Clinic. Past medical history was collected and an exhaustive physical examination was performed including anthropometric measurements with the determination of BMI (kg·m−2). Nocturnal and diurnal symptoms of sleep apnea (SA) were assessed according to the diagnosis criteria of the
After the initial sleep clinic visit, a full-night polysomnography (PSG) was planned in the Sleep Clinic of Grenoble Alpes University Hospital (Visit 1). PSG was performed according to the recommendations of the American Academy of Sleep Medicine (Supplemental Material). 24 Sleep stages, arousals from sleep, and respiratory events were scored manually by a certified sleep specialist according to the American Academy of Sleep Medicine criteria. 25
Patients were classified according to SA severity in no-mild SA (apnea-hypopnea index [AHI] < 15 events/hour of sleep) and moderate-severe SA (AHI ⩾ 15 events/hour of sleep). Moderate-severe SA (⩾15/h) was treated according to French recommendations, mainly by using CPAP as a primary therapy.
Primary CPAP therapy for SA
All patients were followed by the same home care provider with the same standardized procedures. Objective data on daily CPAP usage was recorded by the CPAP device and can be downloaded by the homecare provider’s technician during the routine mandatory home visits or via telemonitoring. The mean CPAP adherence was assessed during follow-up visits, and defined as the mean usage from treatment initiation to follow-up evaluation. Adherence to CPAP was defined as a mean nightly usage of at least 4 h per night.
Statistical analysis
Data are described using counts and percentages for qualitative variables and medians and quartiles (Q1; Q3) for quantitative variables. Comparison tests were performed using the χ2 test for qualitative variables and the non-parametric Mann-Whitney for quantitative variables. A
For this analysis, stroke patients were further stratified into three groups according to SA severity and CPAP adherence: No-mild SA (SA−), moderate-severe SA with no CPAP treatment or CPAP adherence < 4 h/night (SA + CPAP−), and moderate-severe SA with CPAP adherence ⩾ 4 h/night (SA + CPAP+).
Multivariable negative binomial regression models were performed to assess the impact of SA and its treatment on the evolution of ESS (∆ESS = ESS follow-up − ESS baseline). The following confounding variables were introduced as fixed effects: stroke/TIA, baseline ESS, delay between ESS baseline and follow-up, Pichot Depression questionnaire score, Pichot Fatigue questionnaire score, and propensity scores. Three distinct models were performed: (1) SA- vs SA+, entire study population (
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Results
Population characteristics
Population characteristics are presented in Tables 1 and 2. Sixty five (80.2%) ischemic stroke and 16 (19.8%) TIA patients were included and the median (Q1; Q3) NIHSS score at admission was 3.0 (1.0; 5.0). The proportion of TIA patients didn’t differ between the three stroke groups, defined according to SA severity and CPAP adherence. Comorbidities didn’t differ between stroke and matched individuals, except for hypertension (54.3% in stroke/TIA patients vs 34.6% of the matched individuals,
Characteristics of the population.
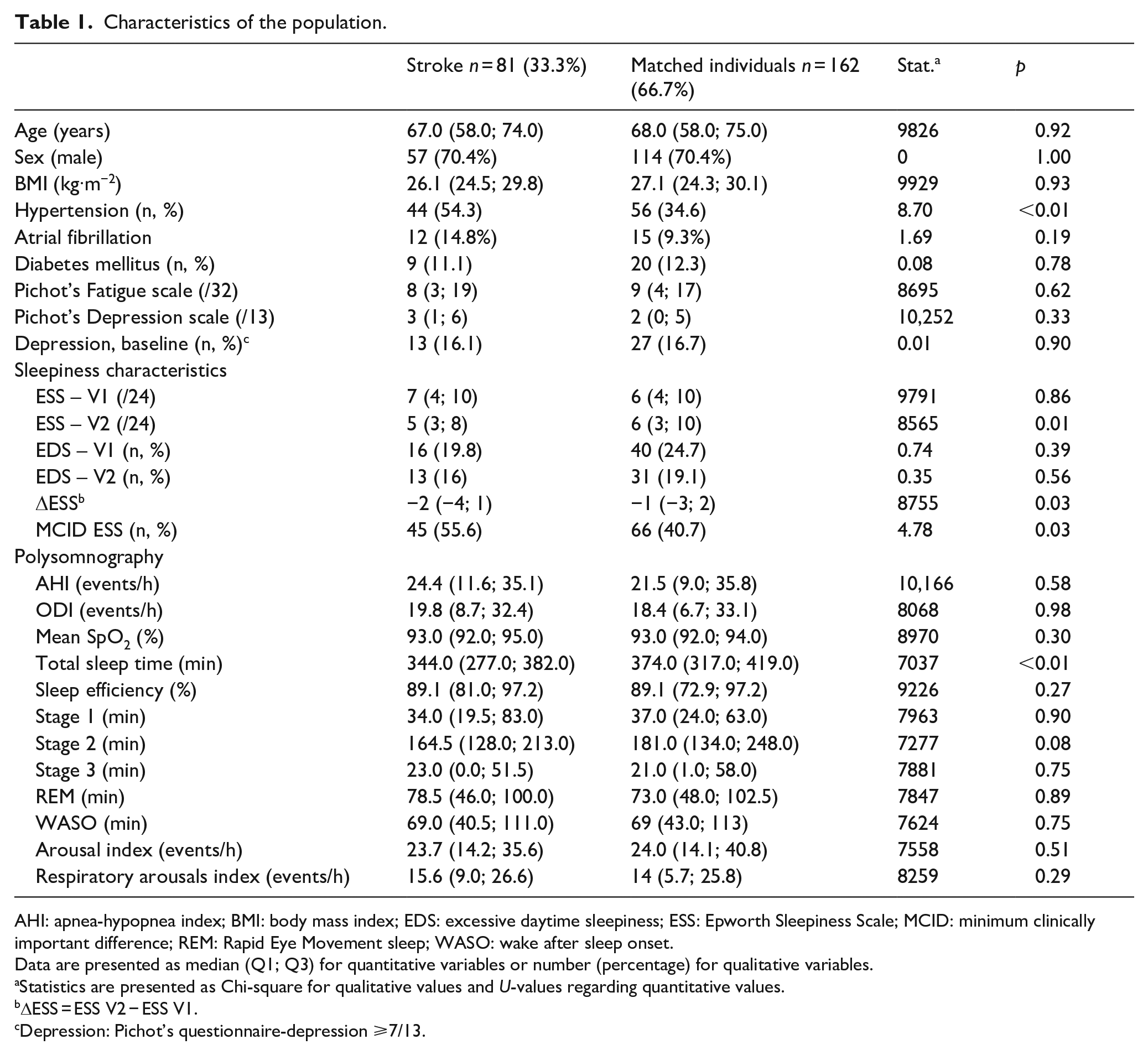
AHI: apnea-hypopnea index; BMI: body mass index; EDS: excessive daytime sleepiness; ESS: Epworth Sleepiness Scale; MCID: minimum clinically important difference; REM: Rapid Eye Movement sleep; WASO: wake after sleep onset.
Data are presented as median (Q1; Q3) for quantitative variables or number (percentage) for qualitative variables.
Statistics are presented as Chi-square for qualitative values and
ΔESS = ESS V2 − ESS V1.
Depression: Pichot’s questionnaire-depression ⩾7/13.
Characteristics of stroke population according to sleep apnea status and treatment adherence.
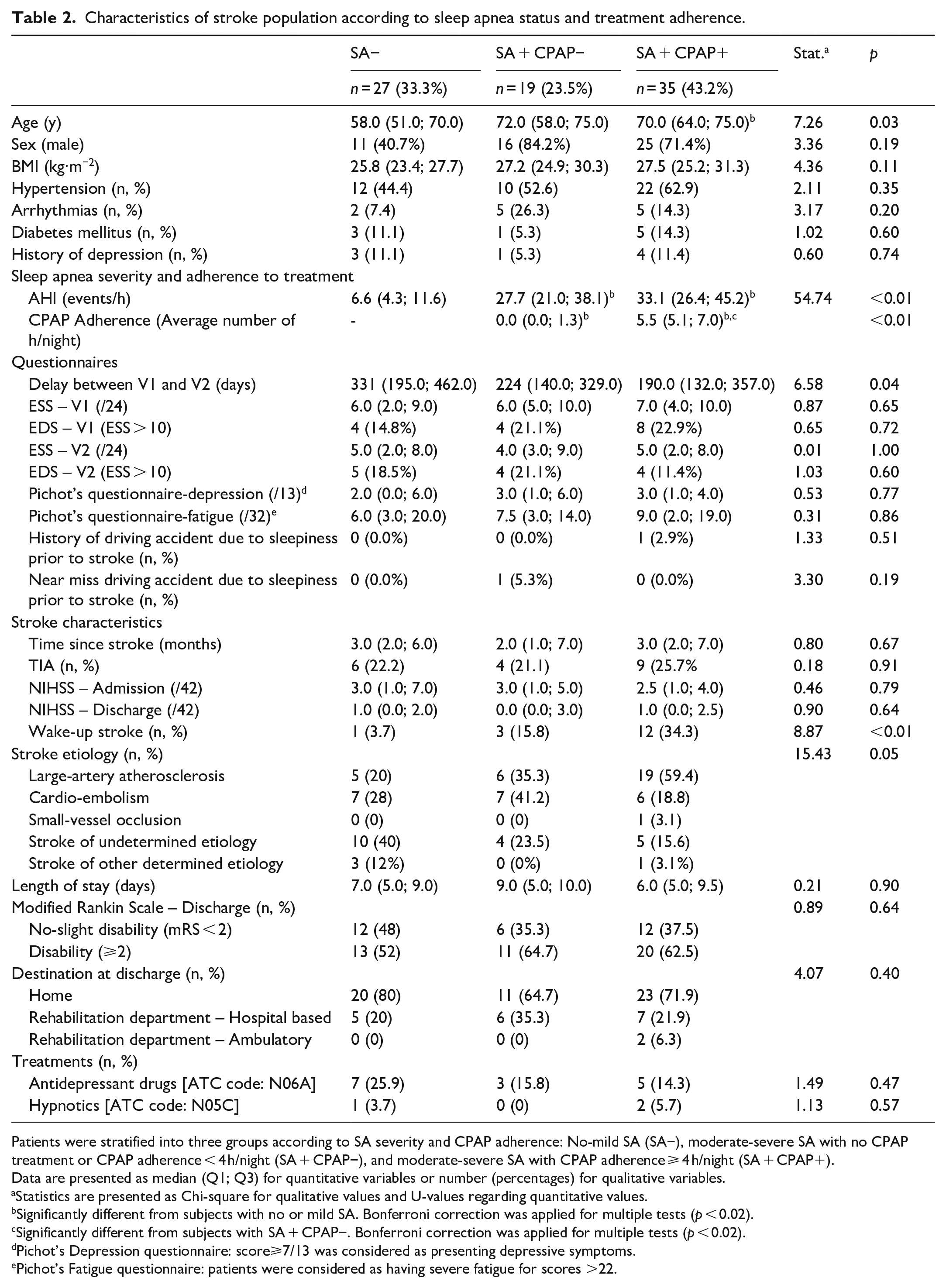
Patients were stratified into three groups according to SA severity and CPAP adherence: No-mild SA (SA−), moderate-severe SA with no CPAP treatment or CPAP adherence < 4 h/night (SA + CPAP−), and moderate-severe SA with CPAP adherence ⩾ 4 h/night (SA + CPAP+).
Data are presented as median (Q1; Q3) for quantitative variables or number (percentages) for qualitative variables.
Statistics are presented as Chi-square for qualitative values and U-values regarding quantitative values.
Significantly different from subjects with no or mild SA. Bonferroni correction was applied for multiple tests (
Significantly different from subjects with SA + CPAP−. Bonferroni correction was applied for multiple tests (
Pichot’s Depression questionnaire: score⩾7/13 was considered as presenting depressive symptoms.
Pichot’s Fatigue questionnaire: patients were considered as having severe fatigue for scores >22.
Moderate-severe SA was present in 54 (66.7%) of stroke/TIA patients, of whom 35 (64.8%) were treated with CPAP with a mean adherence ⩾ 4 h/night (SA + CPAP+). There was no difference in depression scores between stroke and matched individuals, as well as between the three groups of stroke patients.
Sleepiness characteristics and trajectories
Baseline ESS was comparable between stroke/TIA and matched individuals (median (Q1; Q3) ESS = 7 (4; 10) versus 6 (4; 10),
The range of improvement in ESS (∆ESS) was higher in stroke patients compared to matched individuals (Table 1,
Multivariable analysis
In multivariable analysis, stroke/TIA status was associated with greater ΔESS, that is, greater improvement in ESS. On the contrary, higher baseline ESS and fatigue levels were associated with an increase in ∆ESS, that is, an ESS value at the follow-up visit higher compared to the baseline visit (Table 3). SA and CPAP adherence did not influence the evolution of ESS (all
Summary of the multivariable negative binomial regression models to assess parameters associated with delta in ESS between baseline and follow-up.
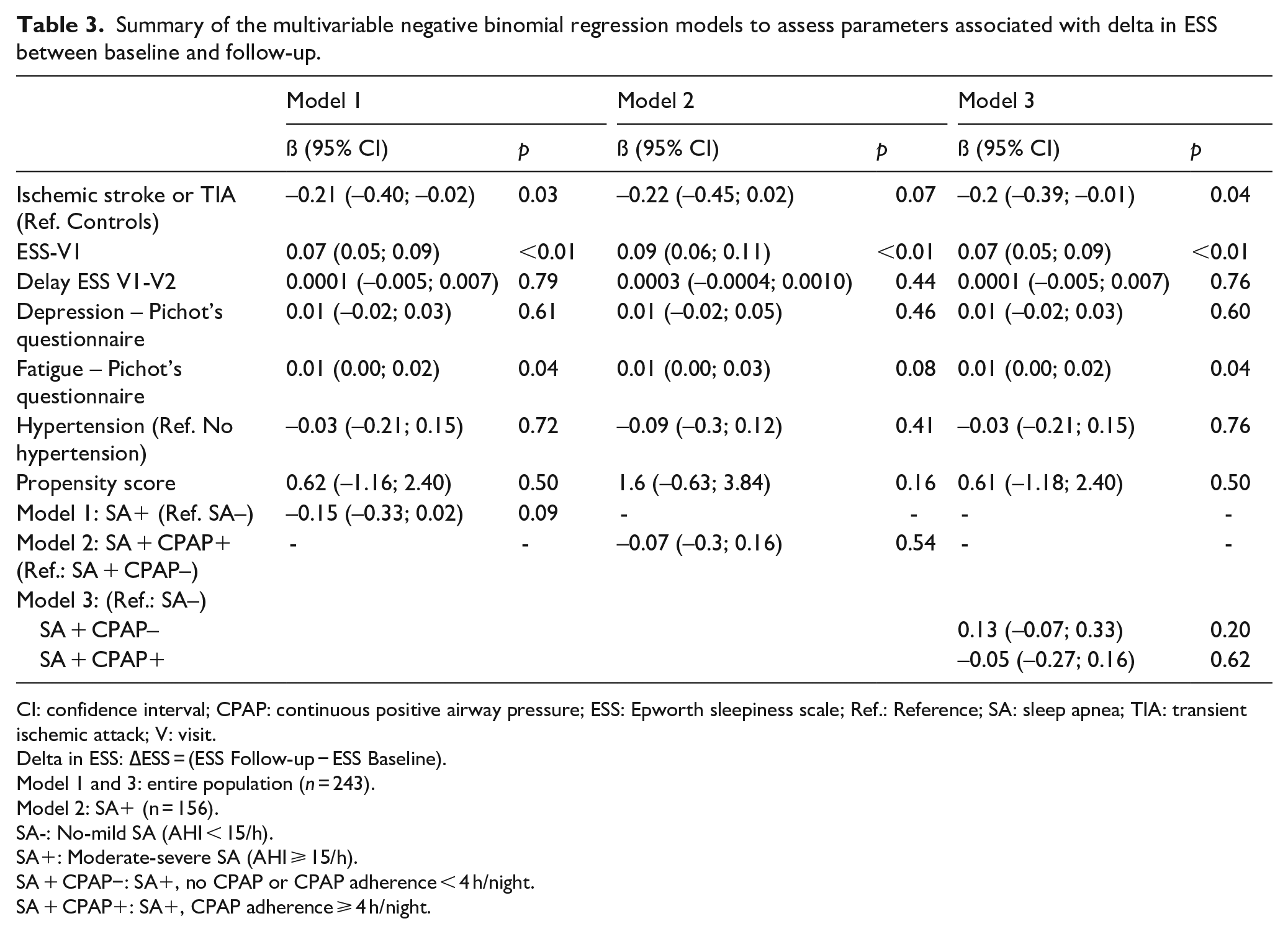
CI: confidence interval; CPAP: continuous positive airway pressure; ESS: Epworth sleepiness scale; Ref.: Reference; SA: sleep apnea; TIA: transient ischemic attack; V: visit.
Delta in ESS: ∆ESS = (ESS Follow-up − ESS Baseline).
Model 1 and 3: entire population (
Model 2: SA+ (n = 156).
SA-: No-mild SA (AHI < 15/h).
SA+: Moderate-severe SA (AHI ⩾ 15/h).
SA + CPAP−: SA+, no CPAP or CPAP adherence < 4 h/night.
SA + CPAP+: SA+, CPAP adherence ⩾ 4 h/night.
Discussion
In this propensity score matched cohort study, we report a significant improvement in self-reported sleepiness scores following a first ischemic stroke or TIA, with no influence of comorbid SA and CPAP adherence on the trajectories of improvement.
We used the ESS to assess self-reported sleepiness post-stroke, which demonstrated a good construct validity and reliability following stroke. 27 The reported EDS prevalence in the stroke population (16% at V2) is in line with recent results from a prospective cohort study, and the observed improvement of self-reported sleepiness followed the expected evolution of sleepiness post-stroke. 14
The originality of our findings is to demonstrate a higher resolution of self-reported sleepiness in stroke compared to matched individuals and no influence of moderate to severe SA and CPAP treatment on the trajectories of self-reported sleepiness post-stroke. Available evidence from a cross-sectional study indicates that for a given severity of OSA, patients with stroke reported less sleepiness in comparison with stroke-free controls. 20 In our cohort, 65% of stroke/TIA patients were adherent to CPAP, which is comparable to the proportion of patients adherent to CPAP in a classical SA population at 1 year. 28 A CPAP adherence threshold ⩾ 4 h/night is demonstrated as significantly associated with improvements in self-reported sleepiness. 29 Thus, in first ischemic stroke or TIA patients, EDS may not be primarily indicative of SA, and less sensitive to CPAP adherence. 20 These results should not, however, dampen the efficacy of CPAP on stroke functional recovery. 30 Moreover, we observed an improvement in EDS prevalence following stroke in the SA + CPAP+ group, which was not observed in SA + CPAP− group as well as SA− group. This result is in line with available evidence showing an improvement of EDS in patients with SA managed by CPAP after stroke.30,31
Post-stroke depression is considered the most frequent neuropsychiatric consequence of a stroke, negatively affecting stroke outcomes. 32 Fatigue is also highly prevalent post-stroke, negatively affecting patients’ quality of life, independence, and prognosis, and is often intricated with depression. 33 Even if fatigue may accompany EDS and hypersomnia, 6 it is a qualitatively different complaint, with a clear difficulty in distinguishing fatigue from EDS in the clinical post-stroke setting. In our study, depression didn’t influence ESS trajectories. We found that heightened post-stroke fatigue levels as well as high baseline ESS values were associated with a significant dampening in ESS improvement, resulting in greater daytime sleepiness levels at the follow-up visit. Factors that influence the trajectories of ESS improvement appear to be complex and intricated, and post-stroke fatigue may have modulated the influence of CPAP treatment on daytime sleepiness trajectories. Further studies are warranted to improve the reliability of the respective assessment of post-stroke fatigue and sleepiness as well as to better assess their respective roles in the evolution of post-stroke non-motor outcomes.
Our study has limitations that warrant discussion. First, the timing of ESS evaluations was not standardized and may have induced variability in the evolution of sleepiness. However, delay between the two ESS evaluations was introduced in all our regression models limiting the impact. Second, most of the included patients were ambulatory stroke patients with limited disabilities along with TIA patients, restraining the generalizability of our findings. Finally, our study lacks objective assessments of sleepiness and a more precise consideration of CNS treatments.
In conclusion, self-reported sleepiness improvement was unexpectedly high in stroke patients compared to a matched individuals’ population, with no influence of comorbid SA and CPAP treatment. Self-reported sleepiness, assessed by the ESS, may not be primarily indicative of SA in first ischemic stroke or TIA patients, nor an indicator to monitor CPAP efficacy.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241227751 – Supplemental material for Trajectories of self-reported daytime sleepiness post-ischemic stroke and transient ischemic attack: A propensity score matching study versus non-stroke patients
Supplemental material, sj-docx-1-eso-10.1177_23969873241227751 for Trajectories of self-reported daytime sleepiness post-ischemic stroke and transient ischemic attack: A propensity score matching study versus non-stroke patients by Sébastien Baillieul, Renaud Tamisier, Bastien Gévaudan, Sarah Alexandre, Olivier Detante, Yves Dauvilliers, Claudio Bassetti, Jean-Louis Pépin and Sébastien Bailly in European Stroke Journal
Footnotes
Acknowledgements
The authors are grateful to Dr. Isabelle Favre-Wiki, Dr. Katia Garambois, Dr. Pauline Cuisenier (Stroke Unit, Grenoble Alpes University Hospital, Grenoble, France) for their substantial help in data collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JLP, RT, S Baillieul and S Bailly are supported by “My Way to health” through the French National Research Agency in the framework of the ’Investissements d’avenir’ program (ANR-15-IDEX-02) and the ’e-health and integrated care and trajectories medicine and MIAI artificial intelligence’ Chairs of excellence from the Grenoble Alpes University Foundation. This work has been partially supported by MIAI@Grenoble Alpes, (ANR-19-P3IA-0003).
Ethical approval
Data collection and analysis were approved by the French biomedical research data processing agency (C.C.T.I.R.S: No 15.925bis, 23 March 2016) and data privacy agency (CNIL: MR003 No 1 996 650v0, 5 October 2016).
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Guarantor
S. Baillieul.
Contributorship
SBaillieul, RT, JLP, and SBailly conceived the study. SBaillieul, RT, JLP, SA, OD were involved in protocol development and patient recruitment. SBailly and BG performed data analysis. SBaillieul, RT, JLP, SBailly, CLAB, and YD critically reviewed the statistical analysis and interpreted the results. SBaillieul and SBailly wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
