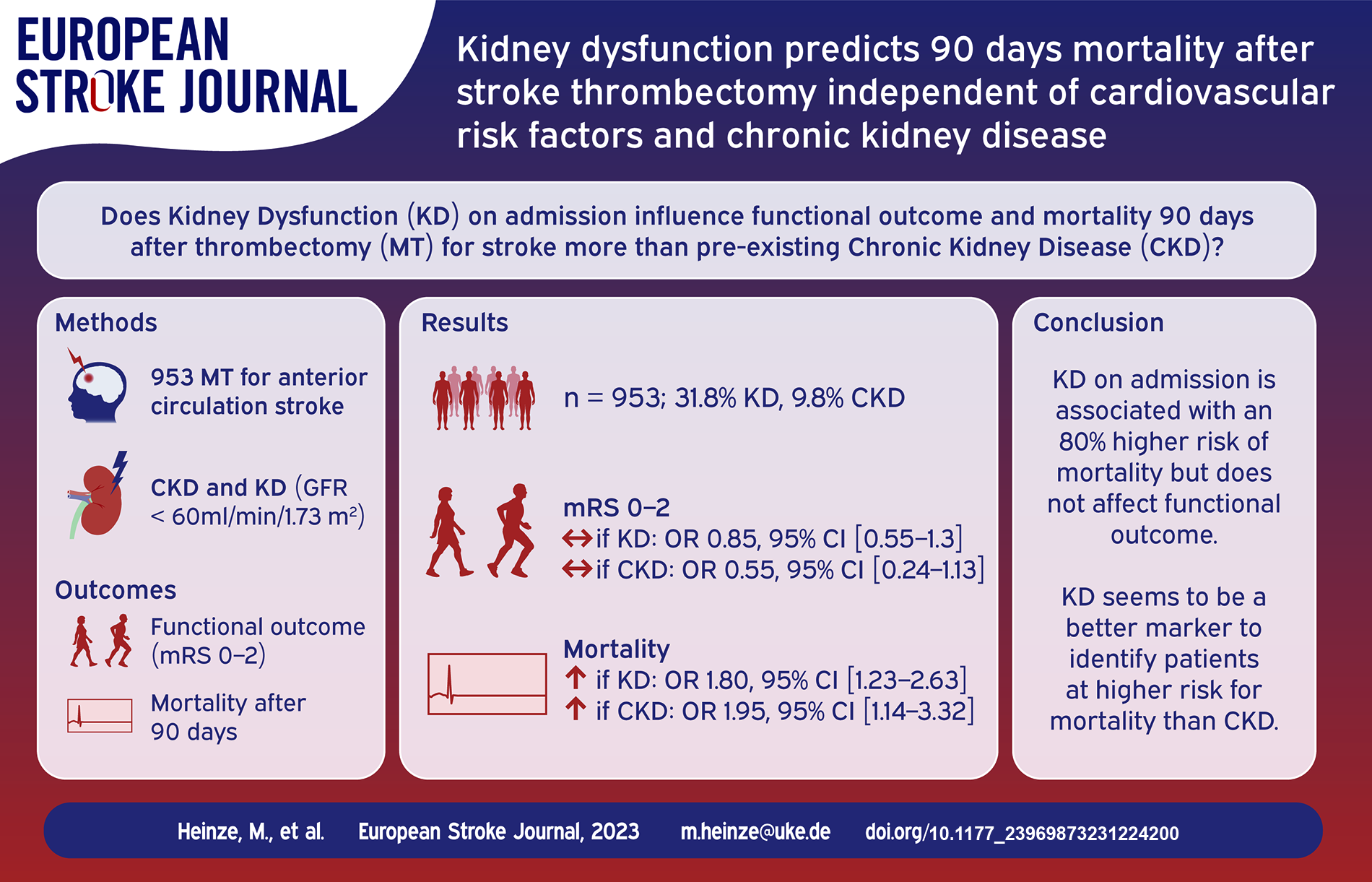
Abstract
Introduction:
Kidney dysfunction (KD) is a risk factor for cerebrovascular events and has been shown to have a detrimental effect on outcome after stroke. We evaluated the influence of KD at admission and pre-existing diagnosis of chronic kidney disease (CKD) before thrombectomy for anterior circulation stroke on functional independence and mortality 90 days after stroke in this cross-sectional study.
Patients and methods:
We included patients with acute ischemic stroke in the anterior circulation treated with thrombectomy at our hospital between June 2015 and May 2022. We analyzed clinical characteristics, laboratory values and pre-existing diagnosis of CKD. KD at admission was defined as glomerular filtration rate (GFR) <60 ml/min/1.73 m2. Outcomes were defined as a modified Rankin Scale Score of 0-2 for functional independence and mortality at 90 days. We fitted multivariate regression analysis to examine the influence of pre-treatment KD and pre-diagnosed CKD on outcome.
Results:
Nine hundred fifty-three patients were included in this analysis (mean age 73.8 years, 54.2% female). KD was present in 31.8%, and patients with KD were older and more often female, presented more often with comorbidities such as arterial hypertension, diabetes, and atrial fibrillation, and were less often independent before the index stroke. In multivariate analysis adjusted for age, independence before the index stroke, diabetes, hypertension, atrial fibrillation, initial NIHSS, thrombolysis treatment, and recanalization outcome, KD on admission had no significant influence on functional independence 90 days after stroke, but predicted mortality with an odds ratio of 1.80 (95% CI 1.23–2.63, p = 0.003). This influence also persisted when controlling for pre-diagnosed CKD (OR 1.60, 95% CI 1.05–2.43, p = 0.027).
Discussion:
KD might function as a surrogate parameter for comorbidity burden and thus increased risk of mortality in this cohort.
Conclusions:
KD on admission is associated with an 80% higher risk of mortality at 90 days after stroke thrombectomy independent of cardiovascular risk factors and CKD awareness. KD on admission should not exclude patients from thrombectomy but might support prognostic evaluation.
Introduction
Kidney dysfunction (KD) and chronic kidney disease (CKD) are well-known cardiovascular risk factors.1–3 Recent studies have shown a detrimental effect of KD on functional independence4–7 and 90 days mortality4,6,8,9 after mechanical thrombectomy (MT) for stroke. However, as summarized in a recent review article, data on the effect of KD on outcome of endovascular stroke treatment is still limited, with only eleven studies on the topic 10 and one more publication since. 11 Most previous studies use a single time point measurement with a cut-off of glomerular filtration rate (GFR) of 60 ml/min/1.73 m2 as a definition of KD. Reasons for decreased GFR in the acute stroke setting are multiple and often reversible. Thus, the single time point assessment of kidney function does not equal pre-diagnosed CKD.
Aims
We evaluated KD together with pre-diagnosed CKD prior to thrombectomy in a large, single-center study to explore the influence of acute KD and CKD on functional independence and mortality independent from other cardiovascular risk factors. We aim to provide insight on the usefulness of distinguishing these markers of decreased kidney function so as to provide guidance in clinical decision making and identifying patients at higher risk for worse outcome.
Methods
Study design and study population
For this post-hoc analysis, we included patients with acute ischemic stroke (AIS) in the anterior circulation treated with thrombectomy at the University Medical Center Hamburg-Eppendorf from 06/2015 to 05/2022. All patients were prospectively enrolled in the German Stroke Registry – Endovascular Treatment (GSR-ET), 12 and functional outcome was assessed 90 days after stroke by face-to-face visit or telephone contact. Patients without available laboratory values before thrombectomy were excluded from this analysis (Figure 1).
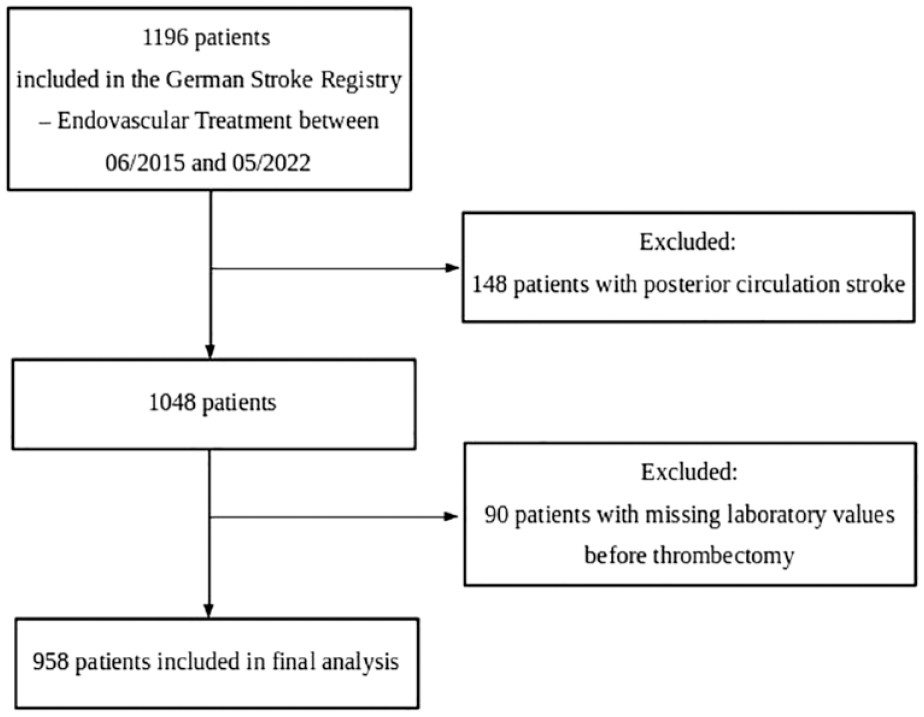
Flow chart of patient inclusion.
Baseline and follow-up data
Clinical data included cardiovascular risk factors, National Institute of Health Stroke Scale Score (NIHSS) on admission and vital parameters. Independence before the index stroke was defined as modified Rankin Scale (mRS) 0-1. Information on imaging and treatment included Alberta Stroke Program Early CT SCORE (ASPECTS), intravenous thrombolysis treatment (IVT), recanalization result [Thrombolysis in Cerebral Infarction (TICI) 13 ] and time from symptom onset to recanalization. Creatinine values were extracted from electronic hospital records. We used the nephro package in R 14 to calculate GFR based on the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) Study equation using serum creatinine without race coefficient. 15 KD at admission was defined as GFR <60 ml/min/1.73 m2. Information on pre-diagnosed CKD was extracted from individual electronic hospital records and discharge letters. TICI results were binarized as a favorable recanalization outcome meaning TICI 2b-3. Outcomes were functional independence at 90 days, defined as mRS 0-2, and mortality at 90 days.
Statistical analysis
Statistical analysis was performed using R version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria). We performed a group comparison of baseline variables between patients with and without KD at admission. To identify predictors of functional independence and mortality at 90 days, we fitted multivariate regression analysis. We chose confounders based on literature research. The model included age, independence, diabetes, hypertension, atrial fibrillation, NIHSS, thrombolysis treatment, ASPECTS, and favorable recanalisation outcome. All tests were carried out with a two-sided alpha level of 5% without correction for multiple comparisons.
Standard protocol approvals, registrations, and patient consents
The study was approved by the ethics committee of the chamber of physicians at Ludwig-Maximilians University LMU, Munich (689-15) as the leading ethics committee. Approval by local ethics committees or institutional review boards was obtained for all participating sites according to local regulations. The GSR-ET is registered at https://www.clinicaltrials.gov; Unique identifier: NCT03356392.
Data availability
Anonymized data not published within this article will be made available by reasonable request from any qualified investigator.
Results
Patient characteristics
Nine hundred fifty-eight patients were included in this analysis, mean age was 73.8 years (SD ± 12.6), 519 (54.2%) were female. Laboratory values before thrombectomy showed KD in 305 (31.8%) patients. Of those, 115 (38.6%) showed reversal of KD (GFR >60 ml/min/1.73 m2) at discharge. Pre-existing diagnosis of CKD was present in 94 (9.8%) patients. Table 1 shows patient characteristics as well as a group comparison between patients with KD at admission and those without KD. Patients with KD were older [80.5 (±9.3) vs 70.7 (±9.3), p < 0.001], more often female [197/305 (64.6%) vs 322/653 (49.3%), p < 0.001], and less often independent before the stroke [212/305 (69.5%) vs 561/653 (85.9%), p < 0.001]. Stroke severity assessed by NIHSS did not differ between the groups (14 [9;18] vs 15 [10;18], p = 0.192), however, ASPECTS in baseline imaging was higher in those with KD (8 [7;9] vs 7 [6;9] p = 0.001). Rates of IVT and the type of anesthesia used for thrombectomy were comparable between groups. Successful recanalization (TICI 2b/3) was less often achieved in patients with KD [215/305 (71.4%) vs 494/653 (77.7%), p = 0.046]. Patients with KD at admission achieved functional independence at 90 day follow up less often [54/305 (19.4%) vs 212/653 (35.6%), p < 0.001] and exhibited higher 90 day mortality [116/305 (41.7%) vs 117/653 (19.6%), p < 0.001]. Figure 2 visualizes the distribution of mRS scores at 90 days between groups.
Baseline demographic data and group comparison by KD on admission.
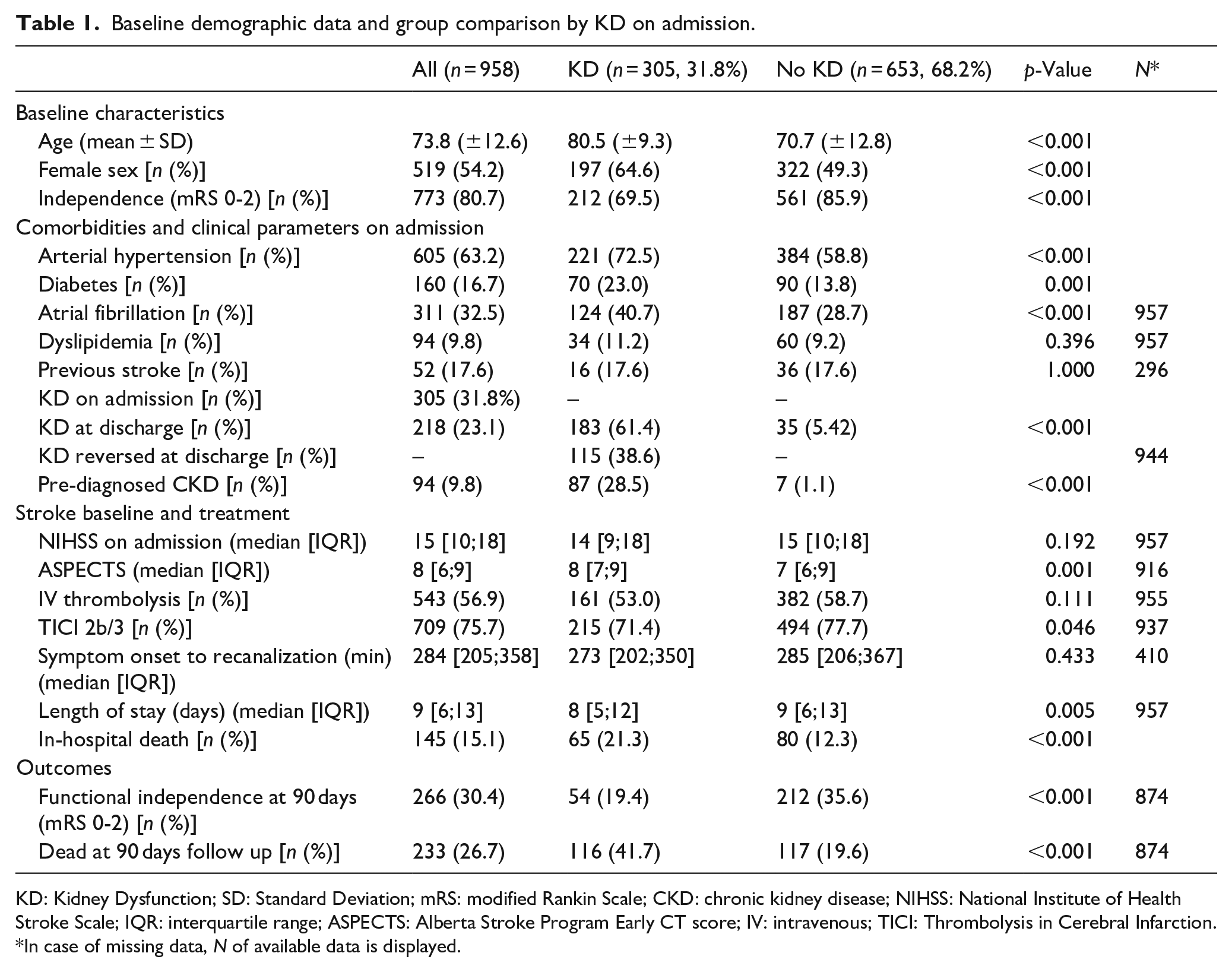
KD: Kidney Dysfunction; SD: Standard Deviation; mRS: modified Rankin Scale; CKD: chronic kidney disease; NIHSS: National Institute of Health Stroke Scale; IQR: interquartile range; ASPECTS: Alberta Stroke Program Early CT score; IV: intravenous; TICI: Thrombolysis in Cerebral Infarction.
In case of missing data, N of available data is displayed.
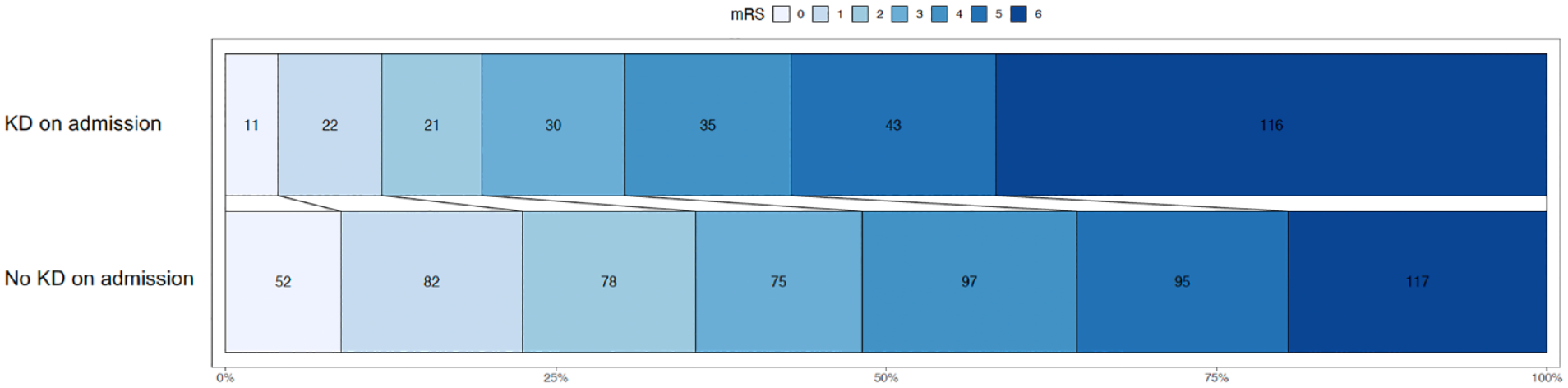
Distribution of mRS at 90 days between groups (KD on admission vs no KD on admission).
Predictors of functional independence and mortality at 90 day follow up
We fitted multivariate regression analysis with functional independence (mRS 0-2) as the outcome for the entire study population (Table 2). KD did not have significant influence on functional independence as outcome, while higher age (OR 0.94, CI 0.92–0.95, p < 0.001), dependency before the index stroke (OR 0.10, CI 0.04–0.21, p < 0.001), a higher NIHSS at baseline (OR 0.92, CI 0.89–0.95, p < 0.001), not having received IVT (OR 0.61, CI 0.41–0.88, p = 0.010), an unfavorable recanalization result (OR 0.17, CI 0.10–0.27, p < 0.001) proved to have a detrimental effect, while higher ASPECTS (OR 1.32, CI 1.19–1.47, p < 0.001) positively influenced functional independence.
Multivariate regression models for KD or pre-diagnosed CKD on outcomes.
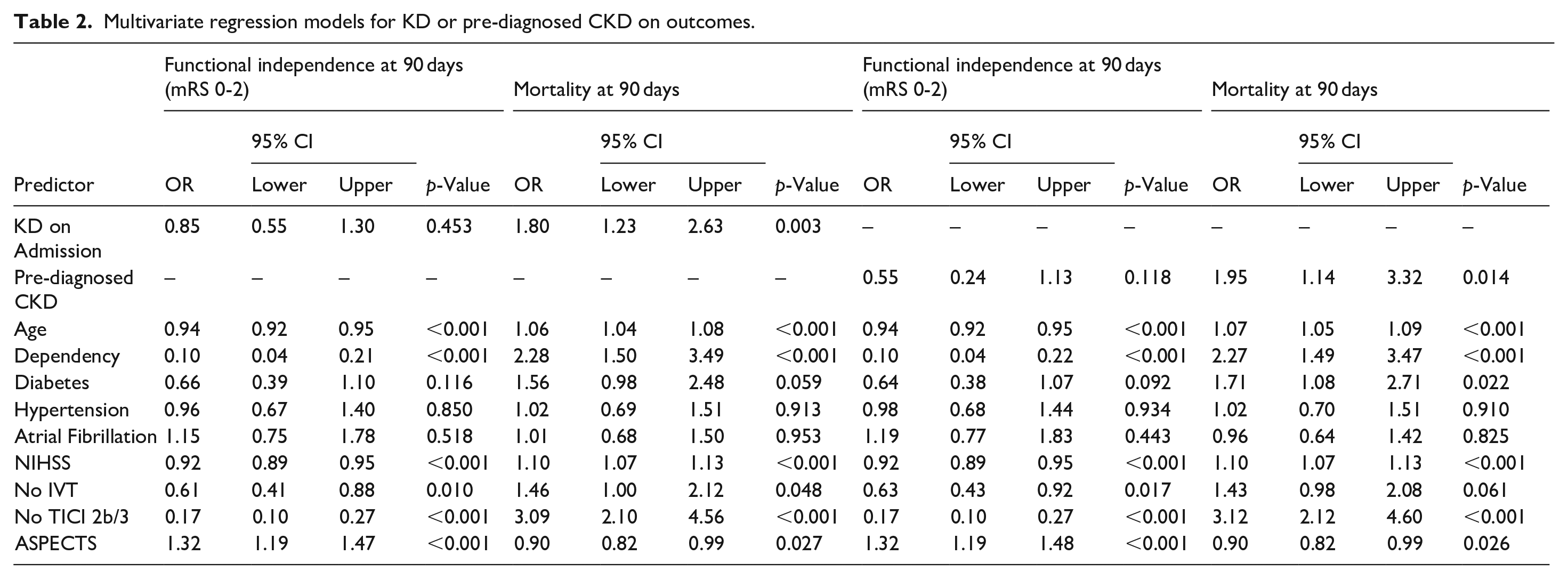
We fitted the same model for mortality at 90 days. In this analysis, KD at admission was associated with higher risk of mortality with an odds ratio of 1.80 (CI 1.23–2.63, p = 0.003). The other factors associated with functional outcome also showed a significant association with mortality but in the opposite direction (higher age, dependency, higher NIHSS, no IVT, unfavorable recanalization result, higher ASPECTS).
When controlling for CKD as a pre-existing condition, the effect of acute KD at admission on mortality remained significant (p = 0.027, Table 3).
Multivariate regression model for KD and CKD on mortality.
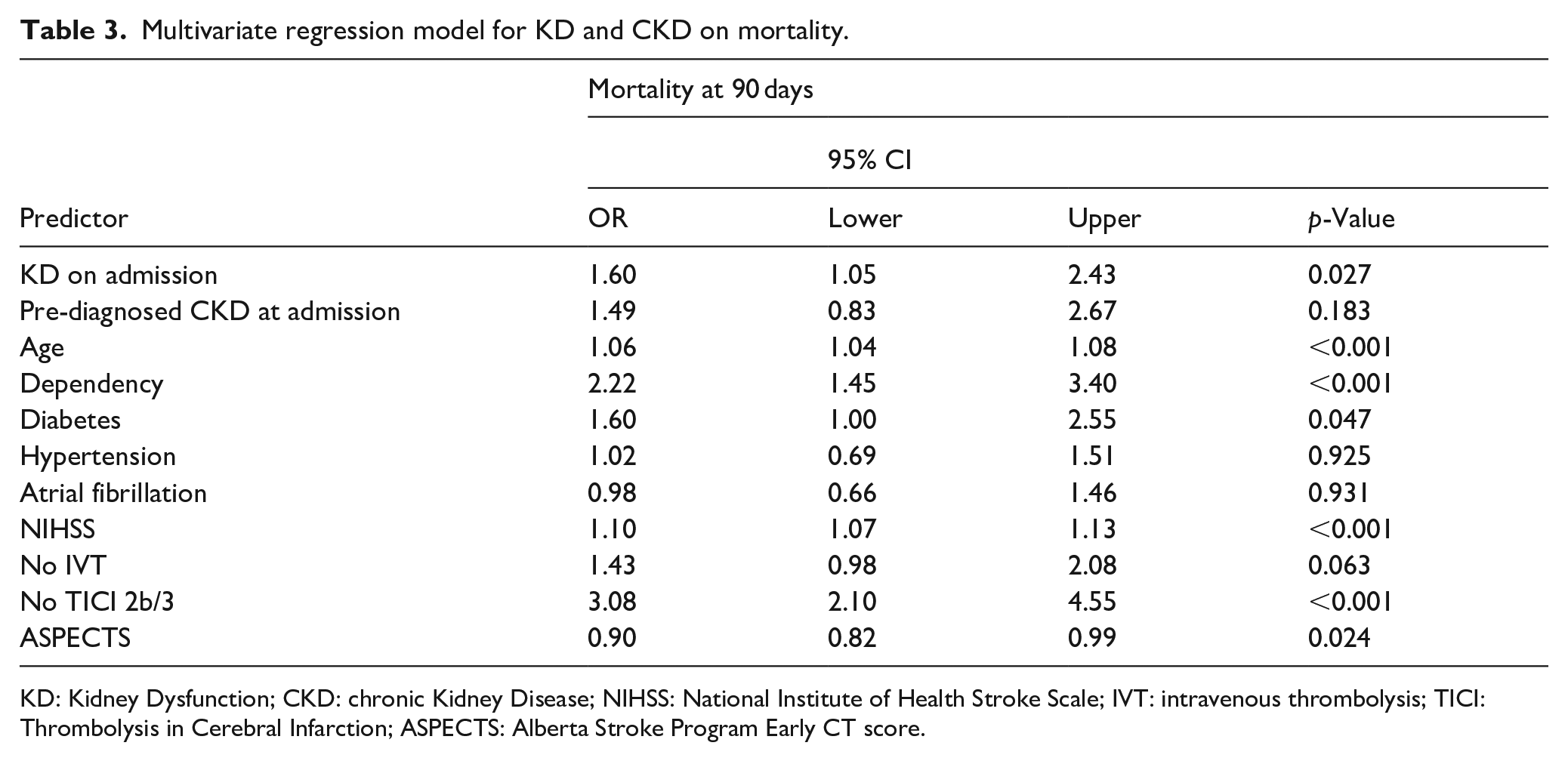
KD: Kidney Dysfunction; CKD: chronic Kidney Disease; NIHSS: National Institute of Health Stroke Scale; IVT: intravenous thrombolysis; TICI: Thrombolysis in Cerebral Infarction; ASPECTS: Alberta Stroke Program Early CT score.
In the primary analysis, we did not include a measurement of time in the model, as symptom onset to recanalization time was only available for 410 patients. In a sensitivity analysis we repeated regression analysis for this subgroup. This analysis confirmed the results from the primary analysis with no significant association of KD at baseline with functional independence, but a detrimental effect of KD on mortality at 90 days (OR 1.91, CI 1.3–2.75, p = 0.004, Supplemental Table S1). When performing the same model within the subpopulation without CKD, a similar OR resulted for mortality, but only as a trend (OR 1.47, CI 0.96–2.25, p = 0.078, Supplemental Table S2). We further evaluated the influence of KD at discharge on the outcome and found a significant detrimental effect of GFR <60 ml/min at discharge on mortality (OR 2.51, CI 1.43–4.39, p = 0.001, Supplemental Table S3). For this analysis, we excluded all patients with in-hospital death to account for those patients with early withdrawal of care who had no follow-up lab values. Among those patients that had KD at admission, reversal of KD during the hospital stay lowered the risk of mortality (OR 0.27, CI 0.14–0.52, p < 0.001), but did not influence good outcome (OR 1.14, CI 0.51–2.51, p = 0.752, Supplemental Table S4).
Discussion
We evaluated the association between KD assessed by pre-interventional GFR and pre-existing diagnosis of CKD with functional independence and 90 days mortality after thrombectomy for AIS in a large-center cohort. Our main finding showed that pre-treatment KD assessed by a reduced GFR predicted mortality at 90 days independent from other cardiovascular risk factors and beyond the effect of known pre-existing CKD.
The effect of kidney function on outcome after MT for AIS has gained interest in the research community within the past few years, yet data on this important topic remain scarce. A 2022 review found eleven studies with case numbers ranging from 110 to 628 (one study with 1169 cases evaluated acute kidney injury during the hospital stay), 10 and we found one more study published since with 1304 patients receiving MT. 11 Comparing baseline characteristics of our cohort to previous studies, the mean age in our study was in the upper range, stroke severity was in the lower range, and prevalence of KD defined as a GFR <60 ml/min/1.73 m2 (31.8% in our study) was in the upper range, ranging from 7 to 32% in other studies4–9,16).
The observed association of both KD and pre-diagnosed CKD at admission with a higher mortality at 90 days (see Table 2) is in line with previous work.4,6,8,9 KD and CKD increase the risk of stroke independent of other cardiovascular risk factors 17 and are associated with pre- and posttreatment complications that limit treatment results and recovery. Pre-treatment morbidity includes cerebrovascular changes, atherosclerosis and hypertension.18–20 Post-treatment complications include a higher risk of hemorrhage and higher all-cause mortality.1,17 The kidney and the brain have been hypothesized to share a common pattern of microvascular impairment resulting from cardiovascular risk factors, due to their shared status as high-flow end organs with autoregulatory mechanisms. Diverse pathways, such as cerebral blood flow inhibition, accelerated atherosclerosis, oxidative stress, chronic inflammation and genetic factors 21 have been suggested to be involved in kidney-brain-interactions.
The persistence of acute KD as a risk factor in this analysis after controlling for known and diagnosed CKD underlines the importance of kidney function measurement at admission as a robust marker for mortality prediction. As creatinine values are usually measured at admission, our findings implicate a practical approach for kidney related risk estimation in the setting of thrombectomy for acute stroke without the need of prior medical record analyses. In contrast, established risk factors such as diabetes, arterial hypertension and atrial fibrillation were not associated with mortality in multivariate analysis, highlighting the important crosstalk between brain and kidney. The KD at admission group was compiled of both patients with known pre-diagnosed CKD and those without. KD in the acute stroke setting can be due to acute or chronic kidney impairment including reversible causes, such as dehydration and medication side effects. However, in acute real-world clinical scenarios such as stroke it is often challenging to distinguish between these two entities. Since KD at admission remained a strong predictor of mortality when controlling for pre-diagnosed CKD, this suggests that a single measurement of decreased kidney function is a practical indicator for renal effects on mortality. In a supplementary analysis excluding those with pre-diagnosed CKD the effect of KD on mortality persisted as a trend. Cause of death was not recorded in the database, so possible explanations for the association of kidney function with mortality have to remain speculative, but might include an earlier withdrawal of care in multimorbid patients. Furthermore, previous works have identified KD as the most important predictor of contrast-associated acute kidney injury, which in turn is associated with increased mortality.10,22 As KD and CKD are also associated with higher all-cause mortality,1,17 KD in this patient cohort might function as a surrogate parameter for comorbidity burden and thus increased risk of mortality. From a clinical standpoint, our findings should urge clinicians to pay close attention to GFR assessed on admission and optimize care for patients with KD at admission, especially when considering that we were able to show that reversal of KD during the hospital stay decreased risk of mortality. Based on our results, we would suggest including baseline GFR in future study designs when evaluating outcome after MT.
In this study, KD patients less often achieved a favorable recanalization result. Explicit reasons for failed recanalization were not recorded in the database, so we can only speculate on reasons for this observation. As with mortality, higher age within the KD group certainly played a role here. A possible underlying mechanism might be the proven association between KD and vascular pathologies including vascular calcification and arterial stiffness, 23 which might come along with technical difficulties in mechanical thrombectomy. Lower recanalization rates have also been reported in previous studies 4,24 and for coronary revascularization therapy in kidney disease patients. 25 Due to the association of KD/CKD with cerebral small vessel disease, 17 recovery of microvasculature after successful recanalization might have been further limited, contributing to bad outcome. Interestingly, we observed a higher initial ASPECTS in the KD group, representing a lesser extent of early ischemic changes. A lower ASPECT Score has been associated with higher risk of mortality and poor outcome in stroke patients. 26 However, some previous studies have shown that this association did not persist when controlling for other factors.27,28 Diabetes was a predictor for mortality when evaluating the association of pre-diagnosed CKD and mortality. Diabetes is a known cause of CKD 29 as well as for intracranial atherosclerosis 30 and cerebral small vessel disease, 31 thus further contributing to bad outcome.
The lack of effect of KD on functional independence in this study is also in line with the results from previous works,8,9,16 however, other studies had conflicting results.4–7
Comparing results across studies is hampered by differences in terminology and underlying definitions. This especially applies for kidney dysfunction with the discrimination of acute kidney injury (AKI) and CKD, as prior laboratory data with creatinine values are often unavailable. This explicitly applies for acute and short inpatient treatments such as strokes. Most studies have thus analyzed a single measurement of GFR at admission to assess baseline kidney function as proposed by the European Renal Best Practice guidelines. Two studies applied a more thorough approach.15,16 The study by Osman et al. did not clarify their definition of CKD, therefore limiting comparability. Fandler-Höfler et al. differentiated acute kidney injury (AKI) at admission from AKI during the hospital stay and CKD, using the highest available GFR during the hospital stay as baseline and quantifying CKD according to Kidney Disease Improving Global Outcomes (KDIGO) criteria. They found that AKI at admission and AKI during the hospital stay predicted worse outcomes after 90 days in multivariate analysis. The authors stress the importance of differentiating between CKD and AKI, however, the used definitions are also limited and not recommended by leading kidney organizations such as KDIGO. In clinical practice, decisions often have to be made based on the information available at a glance. Therefore we decided for a practical approach for this study and estimated kidney function based on the admission creatinine but controlled analyses for a pre-existing coded condition of CKD based on previous documentation, medical history taken from referring ambulatory general health physicians or the referring hospital.
The single center design is the main limitation of this study, restricting generalizability of our results. In sensitivity analysis for the subgroup with data availability for symptom onset to recanalization time, data availability did not come at random in group comparison (data not shown), therefore possibly limiting the meaningfulness of this analysis. The definition of CKD in this study was based on medical records and therefore did not allow for further sub-classification of CKD. Future prospective and multicenter studies should be conducted to investigate this further.
In conclusion, KD did not have an influence on functional independence 90 days after thrombectomy for anterior circulation stroke, but predicted mortality at 90 days. This effect remained when controlling for cardiovascular risk factors and pre-diagnosed CKD. KD on admission should not be an exclusion criteria when making a treatment decision in stroke patients but should inform clinicians to pay increased attention to those patients and might be used for prognostic evaluation.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231224200 – Supplemental material for Kidney dysfunction predicts 90 days mortality after stroke thrombectomy independent of cardiovascular risk factors and chronic kidney disease
Supplemental material, sj-docx-1-eso-10.1177_23969873231224200 for Kidney dysfunction predicts 90 days mortality after stroke thrombectomy independent of cardiovascular risk factors and chronic kidney disease by Marlene Heinze, Maximilian Schell, Felix Leonard Nägele, Bastian Cheng, Fabian Flottmann, Jens Fiehler, Christian Schmidt-Lauber and Götz Thomalla in European Stroke Journal
Footnotes
Acknowledgements
MH and GT conceived and designed the study. MH analyzed and interpreted the data and wrote the first draft of the manuscript. MS, FLN, BC, FF, JF, and CSL acquired and curated data and critically revised the manuscript.
Disclosures
MH, MS, FLN, BC, FF, CSL have no disclosures. JF reports funding unrelated to this study from the German Ministry of Science and Education (BMBF), the German Ministry of Economy and Innovation (BMWi), the German Research Foundation (DFG), the European Union (EU), Hamburgische Investitions- und Förderbank (IFB), Medtronic, Microvention, Route92 and Stryker. JF is a consultant for: Bayer, Boehringer Ingelheim, Cerenovus, Evasc Neurovascular, MD Clinicals, Medtronic, Medina, Microvention, Penumbra, Phenox, Stryker and Transverse Medical. He is a stock holder at Tegus Medical, Vastrax and Eppdata. GT reports personal fees from Acandis, Alexion, Amarin, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb/Pfizer, Daiichi Sankyo, Portola, and Stryker outside the submitted work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Guarantor
MH
Ethical approval
The study was approved by the ethics committee of the chamber of physicians at Ludwig-Maximillians University LMU, Munich (689-15) as the leading ethics committee. Approval by local ethics committees or institutional review boards was obtained for all participating sites according to local regulations.
Contributorship
MH and GT conceived and designed the study. MH analyzed and interpreted the data and wrote the first draft of the manuscript. MS, FLN, BC, FF, JF, and CSL acquired and curated data and critically revised the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
