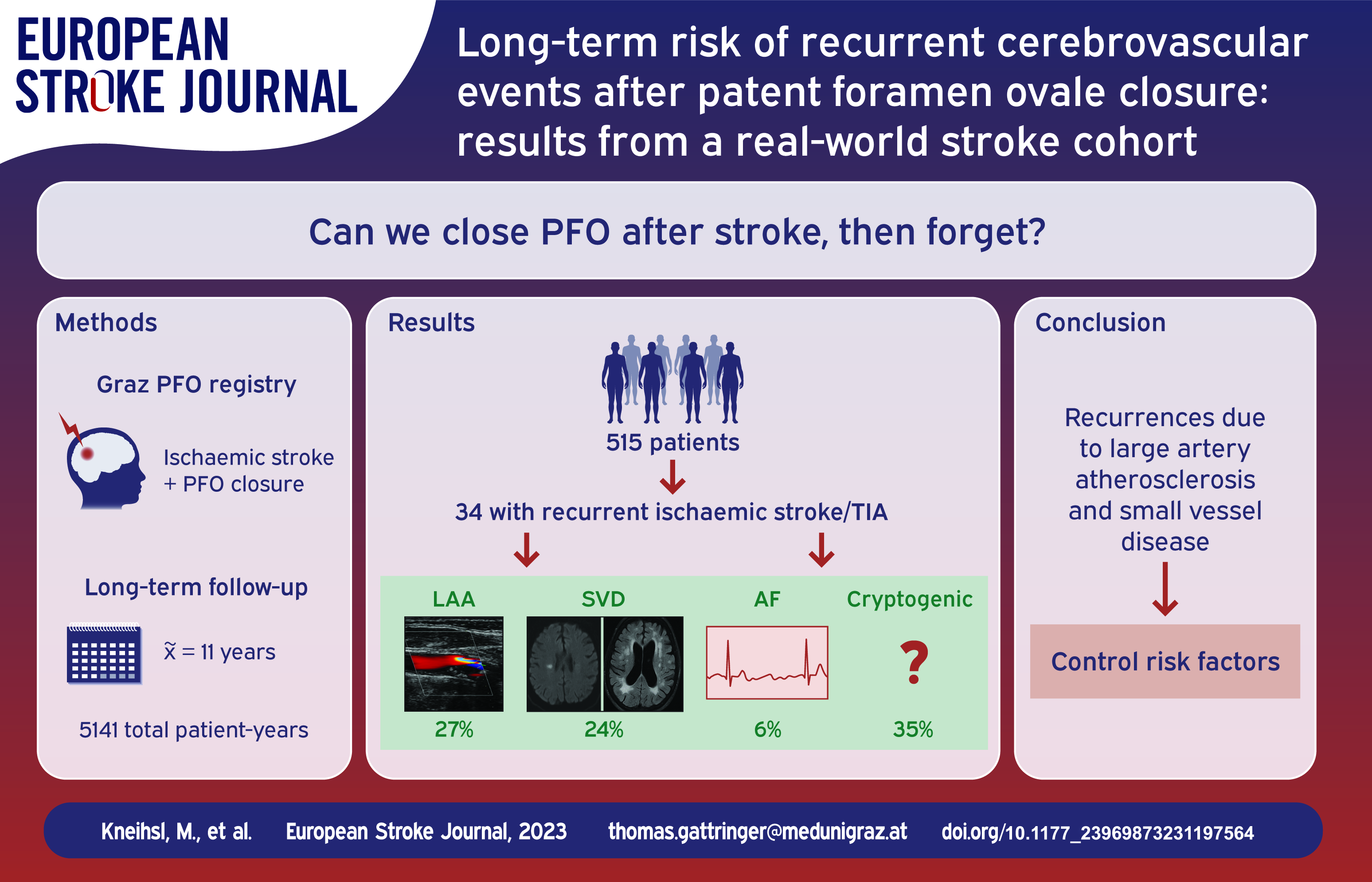
Abstract
Introduction:
Patent foramen ovale (PFO)-closure is recommended for stroke prevention in selected patients with suspected PFO-associated stroke. However, studies on cerebrovascular event recurrence after PFO-closure are limited by relatively short follow-up periods and information on the underlying aetiology of recurrent events is scarce.
Patients and methods:
All consecutive patients with a cerebral ischaemic event and PFO-closure at the University Hospital Graz were prospectively identified from 2004 to 2021. Indication for PFO-closure was based on a neurological-cardiological PFO board decision. Patients underwent standardized clinical and echocardiographic follow-up 6 months after PFO-closure. Recurrent cerebrovascular events were assessed via electronical health records.
Results:
PFO-closure was performed in 515 patients (median age: 49 years; Amplatzer PFO occluder: 42%). Over a median follow-up of 11 years (range: 2–18 years, 5141 total patient-years), recurrent ischaemic cerebrovascular events were observed in 34 patients (ischaemic stroke: n = 22, TIA: n = 12) and associated with age, hyperlipidaemia and smoking in multivariable analysis (p < 0.05 each). Large artery atherosclerosis and small vessel disease were the most frequent aetiologies of recurrent stroke/TIA (27% and 24% respectively), and only two events were related to atrial fibrillation (AF). Recurrent ischaemic cerebrovascular event rates and incident AF were comparable in patients treated with different PFO occluders (p > 0.1).
Discussion and conclusion:
In this long-term follow-up-study of patients with a cerebral ischaemic event who had received PFO-closure with different devices, rates of recurrent stroke/TIA were low and largely related to large artery atherosclerosis and small vessel disease. Thorough vascular risk factor control seems crucial for secondary stroke prevention in patients treated for PFO-related stroke.
Keywords
Introduction
Patent foramen ovale (PFO) associated paradoxical embolism is an important cause of stroke mainly in young ischaemic stroke patients.1,2 Large randomized controlled trials in the past decade identified a more than 60% relative risk reduction of recurrent cerebrovascular events after PFO-closure compared to best medical treatment in selected cryptogenic stroke patients (number needed to treat: 13 during 10 person-years of follow-up).3–6 International secondary stroke prevention guidelines recommend PFO-closure in cryptogenic stroke patients with suspected paradoxical embolism aged 18–60 years.7,8 However, clinical trials did not provide follow-up data of more than 5 years after the intervention and therefore long-term safety/complications and recurrent vascular event rates in patients who had undergone PFO-closure are limited.3–5 Moreover, previous studies did not report on the aetiologies of recurrent ischaemic strokes,9–11 which could add valuable information on the general clinical management of cryptogenic stroke patients with suspected paradoxical embolism. Furthermore, information on the long-term safety and outcome after PFO-closure with different occluder devices is lacking. Although various PFO occluder systems are used in clinical practice, only real-world long-term follow-up data >10 years of patients treated with Amplatzer PFO occluder are available thus far.12–14
The aim of the present project was (1) to evaluate the rate of long-term complications and recurrent vascular events of patients who underwent PFO-closure for suspected PFO-associated stroke over a follow-up period of more than 10 years, (2) to report the aetiologies of recurrent stroke or transient ischaemic attacks (TIA) and (3) to gather long-term follow-up data on complications and prognosis of patients treated with different PFO-closure devices.
Materials and methods
Data from this study are available from the corresponding author upon reasonable request. The study was approved by the ethics committee of the Medical University of Graz (number: 34-079 ex 21/22).
Selection of participants and data collection
This study is based on the Graz PFO Registry that prospectively documented all consecutive patients who underwent PFO closure at the University Hospital Graz between 2004 and 2021 and contains data on demographics, cerebrovascular risk factors, preinterventional ROPE Score, 15 PFO-closure procedure, the type of PFO occluder and postinterventional complications.
Patients under the age of 18 years were excluded from the study cohort, as were patients who did not undergo a clinical follow-up in the federal state of Styria, of which Graz is the capital (for details see Figure 1).
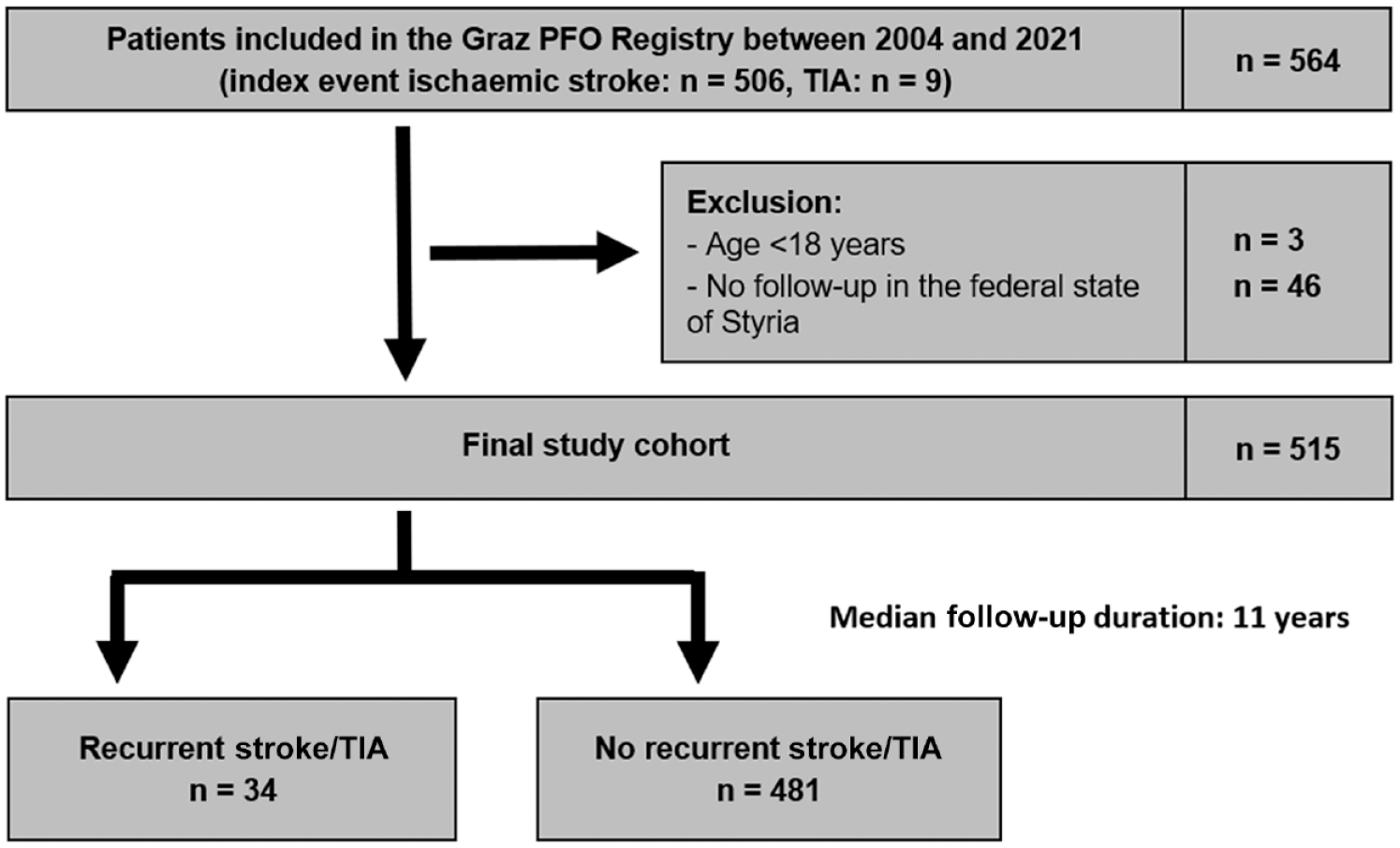
Flow diagram of selection of study participants.
In general, all ischaemic stroke/TIA patients (imaging-based definition) underwent an intense stroke work-up consisting of brain imaging (mostly magnetic resonance imaging (MRI)), clinical and laboratory examinations (including haematological assays and coagulation tests), ultrasound examinations or computed tomography (CT)/MRI – angiography of the brain-supplying extracranial arteries, CT/MRI-angiography of intracranial blood vessels, transthoracic echocardiography and a continuous cardiac rhythm monitoring of at least 48 h (longer in individual patients with high clinical suspicion of atrial fibrillation). Transoesophageal echocardiography was additionally performed in all cryptogenic stroke patients aged <60 years or in older patients if clinical signs (e.g. Valsalva-manoeuvre prior to the stroke/TIA) or imaging features (e.g. multiterritorial brain infarct phenotype) were indicative for a cardiac/paradoxical embolism.
If a PFO was identified, all those patients were discussed in a regular neurological-cardiological PFO board meeting. In brief, these board meetings always included a vascular neurologist and experienced interventional cardiologists. Decision on PFO-closure was based on latest national and international consensus statement recommendations with consideration of age, cerebrovascular risk factors, brain infarct morphology and anatomic characteristics of the PFO (e.g. shunt size, diameter, presence of atrial septal aneurysm). The same applied to patients aged >60 years, who were also included in the board meetings after individual referral of the treating neurologist. In general, PFO-closure was only conducted in patients in whom no competing cause of stroke (according to the TOAST classification) was found in the detailed stroke work-up. 16
PFO-closure and medical treatment
PFO-closure was conducted by cardiological specialists with large expertise in interventional procedures. Interventional PFO-closure was performed via femoral venous access. The PFO was crossed by a guidewire, followed by an 8–10 F delivery guiding catheter. Through the catheter a nitinol-framed Amplatz-like occluder was delivered with first a disc opened on the left atrial side, followed by a second disc opened on the right atrial side of the septum. Transoesophageal echocardiography was used to confirm the correct position of the occluder before release.
After the index stroke/TIA, patients were routinely given antiplatelet therapy (aspirin 100 mg QD or clopidogrel 75 mg QD). At the day of PFO-closure, heparin was given in a therapeutic dosage until 24 h after the intervention. Postinterventional medical treatment regimens (mostly antiplatelet monotherapy) were recommended at the discretion of the treating physicians (cardiologist and neurologist) and in accordance with current expert recommendations/consensus statements.7,8,17,18 In general, antiplatelet monotherapy was continued throughout the entire follow-up period.
Postinterventional management and follow-up
All PFO-closure patients routinely underwent echocardiography within 24 h after the intervention. Moreover, daily pulse controls were conducted during hospital stay and patients underwent a further 24-h ECG after hospital discharge to identify subclinical cardiac arrhythmias such as atrial fibrillation (AF).
At 1 and 6 months after PFO-closure, regular clinical controls were conducted. Transoesophageal echocardiography was routinely performed 6 months after PFO occlusion. Post-interventional complications, a residual shunt or vascular events (i.e. ischaemic stroke, TIA, myocardial infarction, systemic embolism) were documented in the patient records.
For long-term follow-up data on AF and recurrent vascular events, the medical and nursing documentation and communication network of Styria (MEDOCS) was used. MEDOCS covers medical information collected in all public hospitals (which includes all hospitals providing general emergency and acute neurological care) in the entire federal state of Styria. 19 Further relevant follow-up data were retrieved from the Austrian electronic health record (ELGA) database, an electronic information system documenting medical records from non-public hospitals and healthcare facilities in Austria. If no current medical records were available by the electronical databases (e.g. based on patient’s opt-out option for data storage in ELGA), patients were contacted by phone for a follow-up visit. Recurrent vascular events and aetiology of recurrent stroke/TIA were assessed by two raters (IH, NS) and – in case of uncertainty – discussed with an experienced stroke neurologist (MK). Tissue-based definition by brain imaging was used to diagnose TIA. Diagnostic work-up and TOAST-based aetiological classification of recurrent stroke/TIA were identical to the procedures at baseline. 16
Statistics
Statistical analyses were conducted using the software IBM SPSS Statistics, version 28 (IBM Corp, Armonk, NY, USA). Nominal data are presented in count and percentage while median and range was used to present ordinal and continuous variables.
To compare nominal variables, Pearson’s chi-square test was utilized. In quantitative continuous variables normal distribution was tested with histograms, the Q-Q plot and Kolmogorov-Smirnov tests. If normal distribution was identified, comparison of variables was performed with parametric analysis using the unpaired student’s test. Not normally distributed continuous variables were tested with the nonparametric Mann-Whitney-U test.
A multivariable cox regression model was calculated for stroke/TIA during follow-up as the target variable and adjusted for age, sex, arterial hypertension and all variables that were associated with recurrent stroke/TIA in univariable analysis (p < 0.1). A p-value of <0.05 was set to identify significant differences between studied variables without adjustment for multiple testing.
Results
A total of 564 patients with cerebral ischaemic events had undergone PFO-closure between 2004 and 2021. Of those, three patients were aged <18 years (1.8%) and 46 (8.9%) had no clinical follow-up in the federal state of Styria and were therefore excluded from the study. The final study cohort comprised 515 patients (median age: 49 years, female: 40.4%), of which 506 patients had an ischaemic stroke as the index event (98.4%, TIA: n = 9) (Figure 1).
Baseline
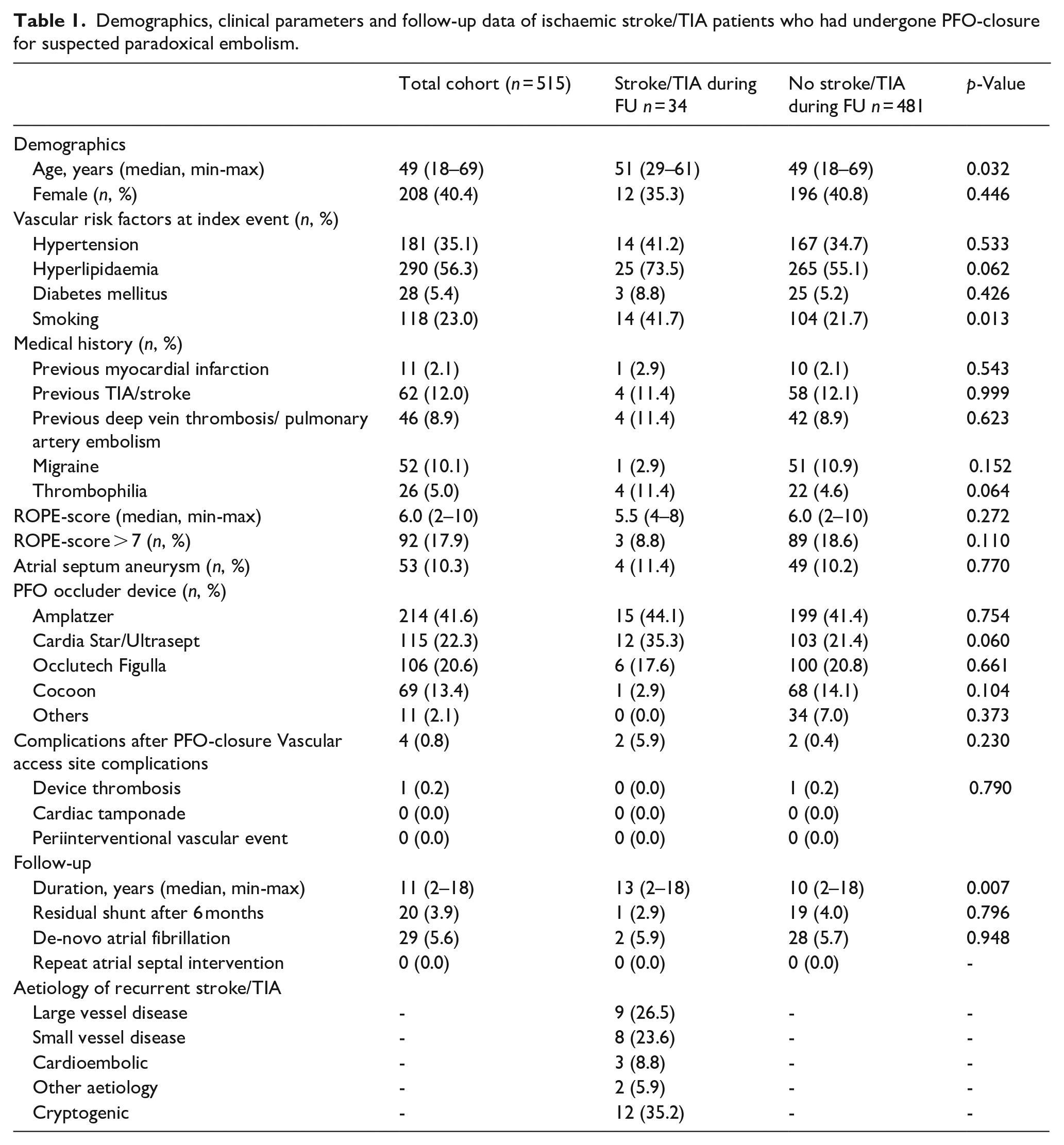
Of all included study participants, hyperlipidaemia was the most prevalent vascular risk factor (n = 290, 56.3%) followed by arterial hypertension (n = 181, 35.1%) and smoking (n = 118, 23.0%) and the median preinterventional ROPE-Score was 6 (range: 2–10). Amplatzer PFO occluder was the most often used device for PFO-closure (n = 214, 41.6%). Only five patients had a major periinterventional complication (1.0%; device thrombosis: n = 1; vascular access site complications: n = 4) which did not lead to a recurrent vascular event. The vast majority of included patients (98.7%) was discharged on antiplatelet monotherapy (ASS: n = 449, 87.2%; Clopidogrel: n = 59; 11.5%). No patient died during the hospital stay. More information is given in Table 1.
Demographics, clinical parameters and follow-up data of ischaemic stroke/TIA patients who had undergone PFO-closure for suspected paradoxical embolism.
Follow-up
Patients were followed for a median duration of 11 years (range: 2–18 years, 5141 total patient-years of follow-up). Six months after the cerebrovascular index event, 20 patients (3.9%) had a residual shunt on echocardiography control. During the entire follow-up period, 29 patients (5.9%) were identified with new-onset atrial fibrillation, no further PFO-closure (device) specific complications were observed. In total, 40 patients (7.8%) had vascular events during the follow-up period. Of those, 34 patients (6.6%) had a recurrent stroke/TIA, five patients (1.0%) were diagnosed with a myocardial infarction and one patient (0.2%) had a systemic embolism. Five patients died during the follow-up period (1.0%) (Table 1).
Recurrent stroke/TIA
Recurrent ischaemic cerebrovascular events (stroke: n = 22, TIA: n = 12) were most frequently related to atherosclerosis of the large brain-supplying vessels (n = 9, 26.5%) and cerebral small vessel disease (n = 8, 23.6%). Only three patients had a cardioembolic stroke during the follow-up (8.8%, AF: n = 2, ejection fraction <30%: n = 1) and 12 recurrent cerebrovascular events (35.2%) remained cryptogenic (Figure 2). Of note, only one patient with a recurrent stroke/TIA had a residual PFO-based right-to-left shunt (0.3%) and no atrial thrombus formations were observed on echocardiography control.
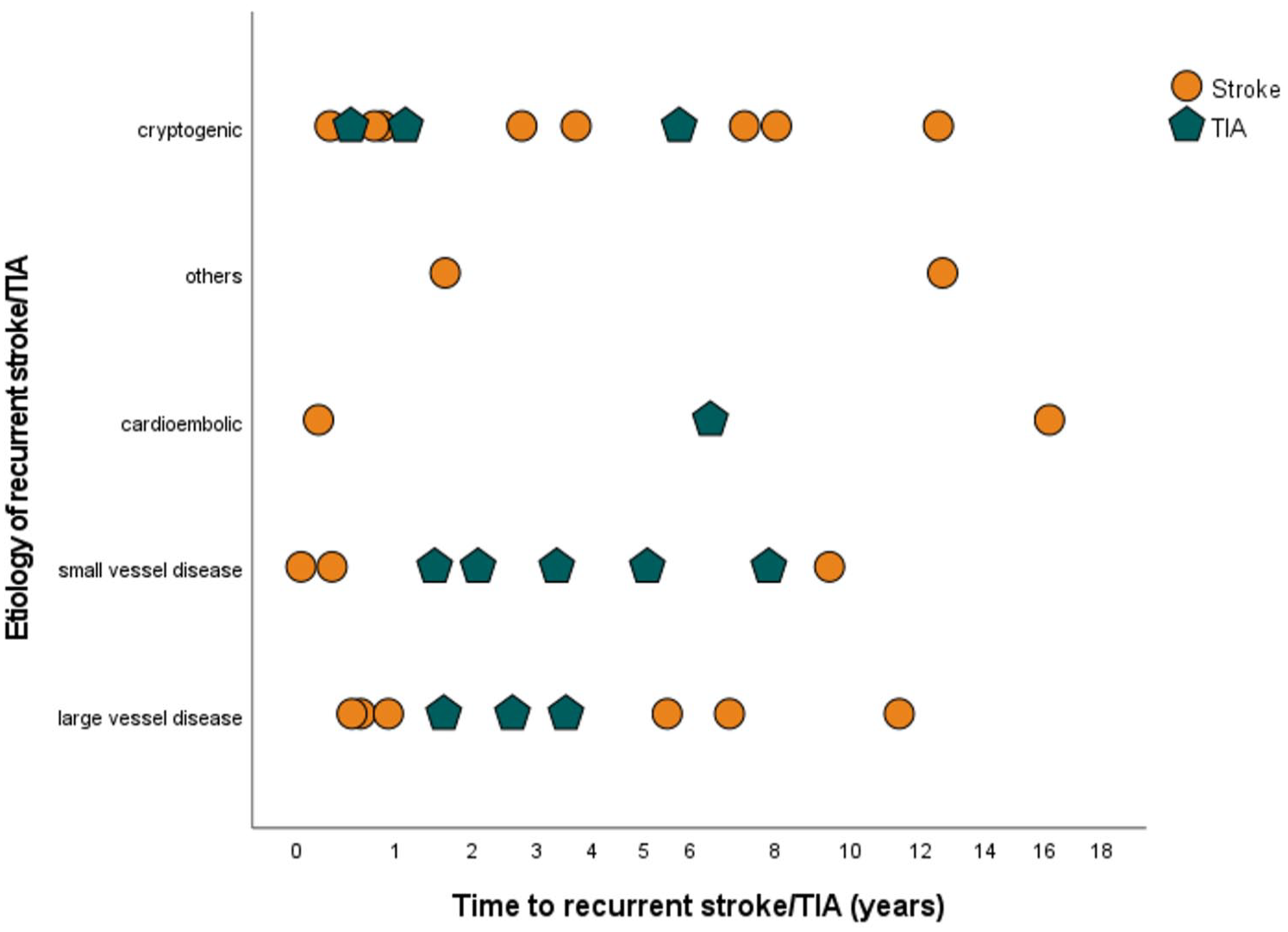
Time trajectories of recurrent cerebrovascular events and their aetiology after PFO-closure for suspected PFO-related cerebral ischaemic event.
Compared to patients without a recurrent cerebrovascular event during the follow-up period, patients with recurrent stroke/TIA were older (median age: 51 years vs 49 years, p = 0.032), more often smokers (41.7% vs 21.7%, p = 0.013) and tended to a higher rate of hyperlipidaemia (73.5% vs 55.1%, p = 0.062). There were no significant differences in rates of atrial septum aneurysms, residual shunts or new-onset AF (all p > 0.1).
Of interest, only three patients with a recurrent stroke/TIA had a preinterventional ROPE-score > 7 (8.8%) compared to 89 patients (18.6%) who did not have a recurrent cerebrovascular event (p = 0.100) (Table 1).
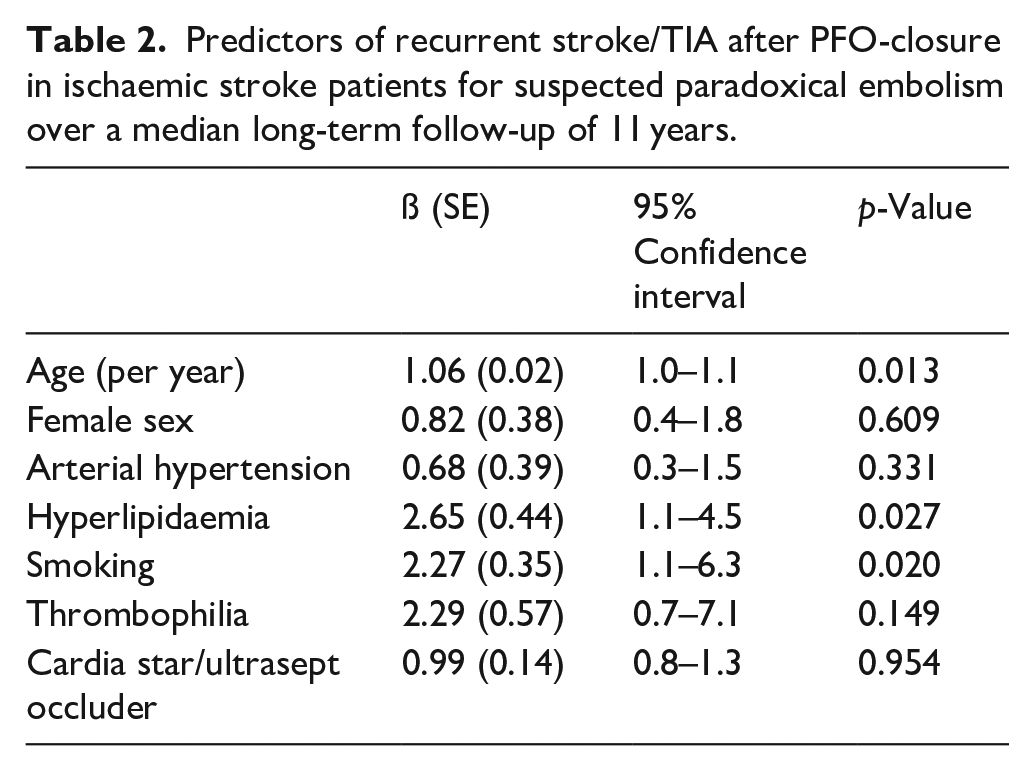
In a multivariable binary logistic regression analysis, higher age (ß: 1.06 ± 0.02 [per year], p = 0.013), smoking (ß: 2.27 ± 0.35, p = 0.020), hyperlipidaemia (ß: 2.65 ± 0.44, p = 0.027) and follow-up time (ß: 1.13 ± 0.04 [per year], p = 0.004) remained associated with recurrent ischaemic stroke/TIA after adjustment for important co-variables (for details see Table 2).
Predictors of recurrent stroke/TIA after PFO-closure in ischaemic stroke patients for suspected paradoxical embolism over a median long-term follow-up of 11 years.
Amplatzer versus non-Amplatzer PFO occluder
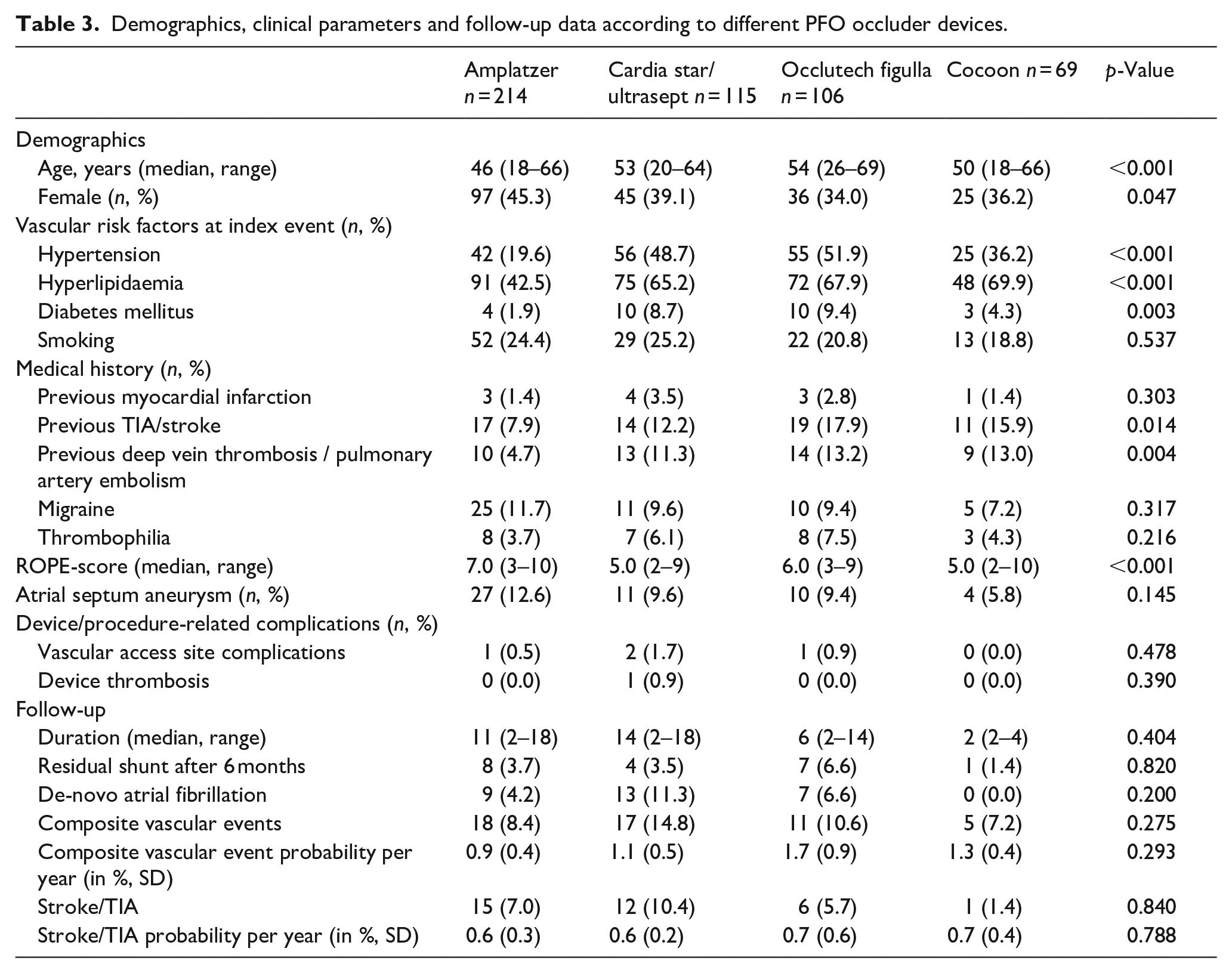
The most frequently used non-Amplatzer occluder devices were Cardia Star/Ultrasept (n = 115, 22.3%), Occlutech Figulla (n = 106, 20.6%) and Cocoon (n = 69, 13.4%). Compared to patients treated with an Amplatzer PFO occluder (41.6%), patients treated with non-Amplatzer devices were older (median age: 50 years vs 46 years, p < 0.001) and had more often at least one classical vascular risk factor at the index event (69.2% vs 53.1%, p = 0.007). Despite these imbalances in baseline characteristics, both subgroups showed comparable rates in procedure-related complications, residual shunts 6 months post intervention, de-novo AF and, most importantly, recurrent vascular events during the follow-up period (p > 0.1; details are presented in Table 3).
Demographics, clinical parameters and follow-up data according to different PFO occluder devices.
Discussion
In this real-world prospective single-centre study on the long-term follow-up of patients with ischaemic cerebrovascular events who were treated with PFO-closure for suspected PFO-related stroke, the rates of long-term adverse events and stroke recurrence were low. Recurrent cerebral ischaemic events were largely related to large artery atherosclerosis and cerebral small-vessel disease.
The majority of stroke/TIA patients treated with PFO-closure are aged <50 years and therefore have a high remaining life expectancy, which supports the need for long-term follow-up studies after PFO-closure. However, randomized controlled trials reporting on the benefit of PFO-closure in cryptogenic stroke patients were terminated at a maximum of 5 years after stroke and data on longer follow-up observations are scarce.4–6 To the best of our knowledge, we here present the longest follow-up study of stroke/TIA patients who underwent PFO-closure for suspected paradoxical cerebral embolism with a median follow-up period of more than 10 years (>5000 total patient-years) and observed a rate of recurrent cerebrovascular events of 7.0%, which is slightly higher compared to previous studies in this field (1.5%–5.5%).4–6,9,10 However, these differences should be mainly attributed to (1) our real-world cohort setting including patients with a higher median age and a higher prevalence of classical vascular risk factors and (2) to the longer median follow-up duration of 11 years after PFO-closure. Notably, the stroke/TIA recurrence rates per 100 patient years were in the range of those reported in previous studies (0.6 vs 0.3–0.6 per 100 patient years) with a substantially shorter follow-up time period.6,9,10,20
To optimize treatment and secondary stroke prevention regimens after PFO-closure, data on the aetiology of recurrent cerebrovascular events are of great importance. To the best of our knowledge, we present the first real-world cohort study that reports on the aetiology of recurrent stroke/TIA and identified small vessel disease and large artery atherosclerosis as the most frequent aetiologies responsible for more than half of all cerebral ischaemic events during the follow-up period. As large artery atherosclerosis and cerebral small vessel disease are strongly related to classical vascular risk factors, it is not surprising that – apart from age – smoking and hyperlipidaemia were the most important predictors of recurrent stroke/TIA in our multivariable model. Our results are supported by a recent study on ischaemic stroke patients who were treated with PFO-closure, which also identified an association of classical vascular risk factors (i.e. diabetes) with incident cerebrovascular events during the follow-up period. 10 According to these data, thorough vascular risk factor control in patients with treatable risk factors seems therefore crucial in all ischaemic stroke patients who had undergone PFO-closure for suspected paradoxical embolism to prevent further cerebrovascular events. Moreover, the presence of classical cardiovascular risk factors should always lead to a detailed work-up of competing causes of ischaemic stroke of presumed PFO-related aetiology.
Two further observations of our study data are of interest: First, AF was only present in two patients with recurrent stroke/TIA. Although we had no continuous cardiac rhythm monitoring of more than 72 h available, new-onset AF rates in our study were comparable to a recent real-world data analysis on long-term outcome after PFO-closure. 9 This might be attributed to the availability of all medical data stored from health care providers in the federal state of Styria during the follow-up period. Hence, our findings argue against a major contribution of AF – either induced by the PFO occlusion or occurring spontaneously after the procedure – with recurrent cerebrovascular events during a long-term follow-up >10 years after PFO-closure in cryptogenic stroke patients.
A further strength of our study is that – in comparison to recent investigations – we had not solely data on Amplatzer occluder devices available.9,10 Studies on the comparison between different PFO occluder devices used in daily clinical routine are limited and especially long-term outcome and potential associations with residual shunts and new-onset AF remain largely unknown. 11 Our results indicate that different PFO occluder devices (i.e. Amplatzer, Cardia and Occlutech) have a comparable long-term safety and do not impact on recurrent cerebrovascular event rates. Importantly, residual shunt rates on echocardiography 6 months after the intervention and new-onset AF rates during the follow-up period did also not differ between different devices. Although our results from a real-word cohort study cannot replace head-to-head trial data – which could be a target for future studies – our observations are important for clinical routine as all tested devices are frequently used in daily clinical practice.
The major limitation of this study arises from its design. Although we had prospectively-collected registry-based data on stroke/TIA patients and PFO-closure available and therefore offer a thoroughly-defined cohort of all patients who had undergone PFO-closure over the studied period, some variables including the sizes of the initial PFO and of the occluder devices were not available in all patients and the assessment of recurrent events and new-onset AF was based on electronic health records. Nevertheless, we were able to capture electronic medical records of all acute health care hospitals in the entire federal state and additionally performed telephone follow-up visits in patients with missing data, which should have avoided a relevant information bias.
Second, the real-world cohort setting leaving the selection of PFO occluder devices at the discretion of the treating cardiologist over a two-decade period led to some baseline imbalances. Results based on different underlying PFO occluder devices must therefore be interpreted cautiously. However, as rates of residual shunts, new-onset AF and recurrent vascular events were (very) low in all tested PFO occluder subgroups, we do not think that these baseline imbalances changed the conclusion of our results to a significant extent. Third, although reporting on a median follow-up duration of 11 years, several included patients had undergone a shorter follow-up period <5 years. Nevertheless, this only affected 15% (74 patients) of the total cohort. Lastly, we did not perform multiple testing due to the limited patient numbers included in the subgroups. Our findings should therefore be regarded as hypothesis generating and need to be replicated in a larger study.
In conclusion, this study reporting on >5000 years of follow-up after PFO closure in patients with ischaemic cerebrovascular events presents a low rate of adverse events and recurrent stroke/TIA independent of PFO occluder devices. Intensive risk factor control might be crucial for secondary stroke prevention in patients with treatable risk factors as the most frequent aetiologies of recurrent cerebral ischaemic events were based on large artery atherosclerosis and cerebral small vessel disease.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Fischer reported research support of the Swiss National Science Foundation and the Swiss Heart Foundation; PI of the ELAN trial, Co-PI of the DISTAL, TECNO, SWIFT DIRECT and SWITCH trial; research grants from Medtronic (BEYOND SWIFT, SWIFT DIRECT) and from Stryker, Rapid medical, Penumbra and Phenox (DISTAL); consultancies for Medtronic, Stryker, and CSL Behring (fees paid to institution); participation in an advisory board for Alexion/Portola, Boehringer Ingelheim, Biogen and Acthera (fees paid to institution); member of a clinical event committee (CEC) of the COATING study (Phenox) and member of the data and safety monitoring committee (DSMB) of the TITAN, LATE_MT and IN EXTREMIS trials; vice-presidency of the Swiss Neurological Society. The other authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Ethics Committee of the Medical University of Graz (approval number: 34-079 ex 21/22).
Informed consent
Informed consent was waived by the Ethics Committee.
Guarantor
TG.
Contributorship
MK: data acquisition and interpretation, manuscript preparation. SH: acquisition of data and critical revision of the manuscript content. IH: acquisition of data. NS: acquisition of data. DK: acquisition of data. GTG: critical revision of the manuscript content. GG: critical revision of the manuscript content. GW: acquisition of data. SFH: critical revision of the manuscript content. MH: critical revision of the manuscript content. NB: critical revision of the manuscript content. SV: acquisition of data. UF: critical revision of the manuscript content. CE: critical revision of the manuscript content. TG: critical revision of the manuscript content. All authors have read and approved the final manuscript and agreed to be accountable for all aspects of the work.
