Abstract
Background:
Covert brain infarction (CBI) is highly prevalent and linked with stroke risk factors, increased mortality, and morbidity. Evidence to guide management is sparse. We sought to gain information on current practice and attitudes toward CBI and to compare differences in management according to CBI phenotype.
Methods:
We conducted a web-based, structured, international survey from November 2021 to February 2022 among neurologists and neuroradiologists. The survey captured respondents’ baseline characteristics, general approach toward CBI and included two case scenarios designed to evaluate management decisions taken upon incidental detection of an embolic-phenotype and a small-vessel-disease phenotype.
Results:
Of 627 respondents (38% vascular neurologists, 24% general neurologists, and 26% neuroradiologists), 362 (58%) had a partial, and 305 (49%) a complete response. Most respondents were university hospital senior faculty members experienced in stroke, mostly from Europe and Asia. Only 66 (18%) of respondents had established institutional written protocols to manage CBI. The majority indicated that they were uncertain regarding useful investigations and further management of CBI patients (median 67 on a slider 0–100, 95% CI 35–81). Almost all respondents (97%) indicated that they would assess vascular risk factors. Although most would investigate and treat similarly to ischemic stroke for both phenotypes, including initiating antithrombotic treatment, there was considerable diagnostic and therapeutic heterogeneity. Less than half of respondents (42%) would assess cognitive function or depression.
Conclusions:
There is a high degree of uncertainty and heterogeneity regarding management of two common types of CBI, even among experienced stroke physicians. Respondents were more proactive regarding the diagnostic and therapeutic management than the minimum recommended by current expert opinions. More data are required to guide management of CBI; meantime, more consistent approaches to identification and consistent application of current knowledge, that also consider cognition and mood, would be promising first steps to improve consistency of care.
Keywords
Introduction
Covert brain infarction (CBI), previously termed silent stroke or silent brain infarction, is among the most frequent focal incidental finding on brain imaging1,2 with even higher numbers for incidentally discovered cerebrovascular white matter disease. 3 CBI is defined as a clear post-ischemic lesion on brain imaging that has not caused acute neurological dysfunction. 4 CBI has a strong age-dependence and affects approximately 30% of people at age 705 with even higher estimates for populations with elevated cardiovascular risk.6,7 The estimated annual incidence for CBI is substantially greater than for manifest acute ischemic stroke. 8 On an epidemiological level, the associations of CBI with a high-risk vascular risk profile 2 as well as its link to detrimental outcomes including ischemic stroke, dementia, all-cause death 9 , and further neurological deficits (gait dysfunction, mood disorders and frailty)9–14 are well established. Similar associations have been reported for incidentally discovered CBI in clinical practice. 3
Surprisingly, these epidemiological insights have not led to definitive clinical trials, specifically tackling CBI patients. Hence, the European Stroke Organization guideline 13 on covert cerebrovascular small-vessel disease (SVD), which addresses lacunes and undifferentiated white matter hyperintensities, provides some guidance on therapeutic strategies, but does not cover diagnostic evaluations for CBI. The American Heart and Stroke Association 15 scientific statement (which also addresses cortical and other embolic phenotypes of CBI as well as diagnostic work-up of such phenotypes) remains indeterminate and expert opinions differ on diagnostic work-up and preventive management of patients with CBI. It is thus unclear, how CBI is currently perceived and managed among physicians in clinical neuroscience.
The primary objective of this survey was to gain information on attitudes toward CBI and current management of those colleagues who are commonly confronted with CBIs in their daily work. Survey questions were created to examine potential differences in management according to embolic versus small-vessel disease (SVD) CBI phenotype.
Methods
Design
We followed the Checklist for Reporting Of Survey Studies (CROSS) recommendations. We defined all relevant survey items including demographic data, experience with CBI, attitudes toward CBI, diagnostic, and therapeutic strategies in the management of CBI patients according to a literature review on the topic.13–17
We then reduced the items to those most likely to be relevant for management decisions. We performed an expert validation to review the draft survey for clarity, relevance to the construct and cognitive difficulty. 18 Five board-certified experts in stroke neurology and neuroradiology monitored the integrity of the survey items and the use of scales. Ambiguous items were revised for clarification. Lastly, we performed cognitive interviews with three representatives of the target population to ensure correct interpretation of the items and response options. 19 We ascertained correct branching logic, so that all necessary input fields only appeared as appropriate based on prior input (e.g. further investigations only needed to be filled if the tickbox “further investigations” was selected).
We created and distributed the cross-sectional survey using REDCap (Research Electronic Data Capture) hosted at the Clinical Trials Unit Bern. 20 An online format was used to maximize response return and ease of dissemination. The survey was available in English only.
The survey included 32 questions on 5 distinct web pages (Introduction, Demographics and Workplace, Attitudes, Case 1, Case 2) with estimated completion time of 10 min. The first questions explored the demographic and educational background of the respondents. Further questions explored attitudes toward CBI. The last part included two short case scenarios designed to examine management decisions taken upon incidental detection of an embolic-phenotype of CBI versus a SVD phenotype. The imaging appearance and clinical description of the embolic-phenotype of CBI was compatible with cardio-embolism, but also with other more proximal sources of embolism, including large artery disease.
The description of the embolic case was
“70-year-old patient, MRI done for work-up of normal pressure hydrocephalus without vascular imaging of cervical arteries. Cardiovascular risk factors: obesity, arterial hypertension, diabetes mellitus type 2. Cardiovascular events: heart attack 4 years ago. Medication: antihypertensives, oral antidiabetic drug therapy, aspirin cardio 100mg/d. CBI phenotype: the CBI is new compared to a MRI 3 years ago”
The description of the SVD phenotype was
“65-year-old female, MRI performed for sudden onset of spinning vertigo, final diagnosis benign paroxysmal positional vertigo. Cardiovascular risk factors: arterial hypertension, smoking. Cardiovascular events: none. Medication: antihypertensives”
Free text sections were permitted to include unforeseen management strategies. Furthermore, we asked about uncertainty regarding this scenario. The full survey questionnaire and data report are presented in the online supplemental appendix.
Distribution
We launched the open survey on November 1st, 2021; the database was closed and locked on February 28th, 2022. We invited physicians across the world involved in care for patients undergoing brain imaging (general neurologists, vascular neurologists, neuroradiologists, other neurology subdisciplines, and neurosurgery) to participate via a web-based survey. To maximize participation, we approached national and international stroke organizations to distribute the survey link among their members along with a cover letter detailing the background of the project. These included the European Academy of Neurology, the Asia Pacific Stroke Organisation, the Swiss Neurological Society, and the German Neurological Society. Reminder emails were sent at the discretion of the organization. Due to societal privacy laws, a complete list of invited email contacts could not be recorded. A subset of respondents (convenience sample) was invited individually by e-mail through lists of contacts curated by the authors, with reminders at 3 weeks. We invited survey respondents to distribute the survey link amongst their network.
We designed the survey questions according to recent recommendations. 21 Neutral wording was used to avoid anchoring bias. We provided slider tools to reduce imprecision bias, and incorporated branching logic to minimize incomplete responses. We aimed to reduce sampling and selection bias, by approaching multiple organizations globally. We prohibited duplicate responses by banning repeat responses with the same email address. Respondents had the option to remain anonymous, responses were handled confidentially and the databank prohibited unauthorized access.
Statistical analysis
We used Stata 16 for the statistical analysis (StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC) including the table 1_mc package. We use descriptive statistics (Fisher’s exact test, Wilcoxon rank-sum) as appropriate and report numbers (%) for categorical variables and medians (interquartile range, IQR) for continuous variables. For slider questions (minimum 0, maximum 100), we report the median and 95% confidence interval to report the agreement of the respondent with the given statement. Whereas a complete survey response was defined as information provided for all sections of the survey, a partial response was defined as a response to only a subset of the sections (e.g. the first section). A significance level of 0.05 was used for all tests with no adjustments for multiple testing. Complete case analysis was performed without imputation of missing data. Exploratory post-hoc subgroup analyses were done comparing the responses according to subspecialty (vascular neurologists vs non-vascular neurologists), workplace (University hospital vs non-academic), seniority level (senior position vs non-senior position), and geographic region (Europe vs Asia).
Ethics
The local research ethics board of Bern, Switzerland exempted this study from approval.
Data sharing
Data is available from the corresponding author upon reasonable request.
Results
Demographic distribution
We estimate, that the survey approached about 3′000 physicians, but given the distribution pathways including websites, this was impossible to ascertain.
Of 627 respondents, 362 (58% completion rate) had a partial and 305 (49% completion rate) had a complete response. Thirty-eight percentage of respondents were vascular neurologists, 24% general neurologists, and 26% neuroradiologists. Most respondents were working at a university hospital where a high volume of brain imaging was performed, were at senior appointment levels and had many years of experience caring for stroke patients. Most respondents were from Europe and Asia (Table 1).
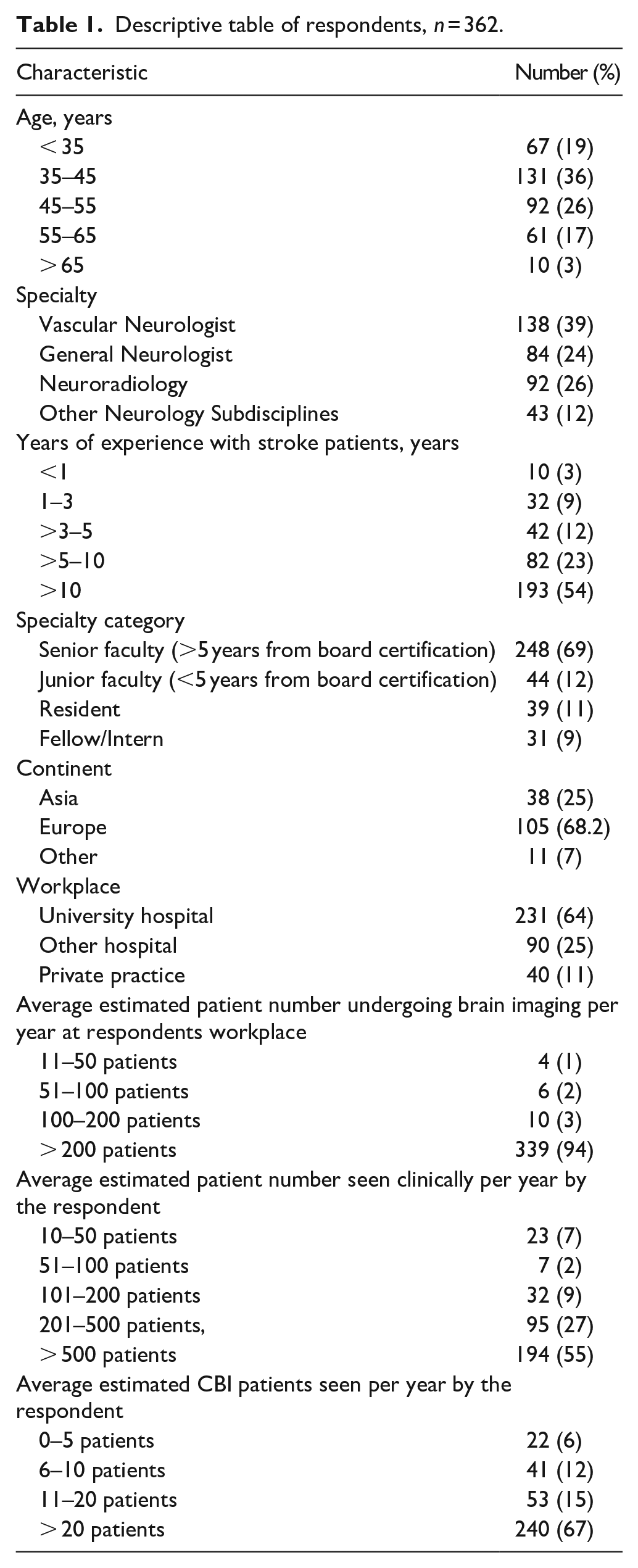
Descriptive table of respondents, n = 362.
Although most respondents stated that brain lesions compatible with CBI were consistently diagnosed as such by neuroradiologists, a concerningly large subgroup indicated that such brain lesions were only inconsistently (23%) or not at all (5%) diagnosed. Even though most respondents stated that they see more than 20 CBI patients per year, only 18% of respondents indicated that they had institutional written protocols for the management of CBI. There was no association between the type of workplace and the availability of institutional written protocols.
Respondents reported strong agreement concerning an association of CBI with future risk of stroke and death (median 85, 95% CI 71–98) as well as CBI with future risk of dementia (median 81, 95% CI 70–97). The majority responded that they had a high degree of uncertainty regarding useful investigations and further management of CBI patients (median 67, 95% CI 35–81). There was no difference in the degree of uncertainty according to the years of experience, senior position holders, type of workplace, or vascular subspecialty.
Whereas almost all respondents (97%) indicated that they would assess vascular risk factors and most would do a work-up analogous to manifest ischemic stroke, only a minority would assess cognitive function and depression (42%, Figure 1).
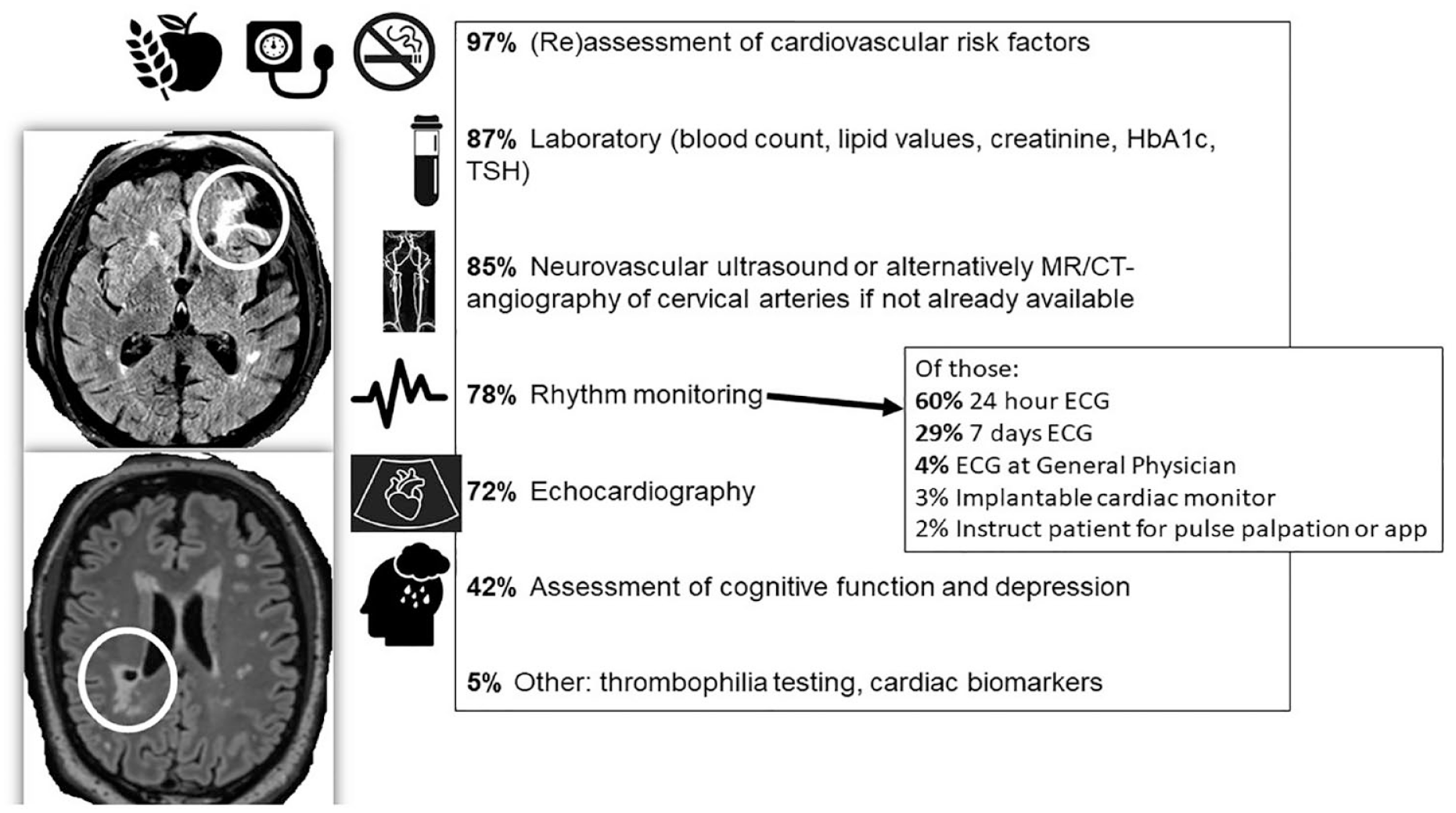
“If a CBI is seen on cerebral imaging, what investigations do you consider to be indicated?”
Diagnostic evaluation
When comparing diagnostic evaluation between the two phenotype case studies, significantly fewer respondents thought that further investigations were indicated in the SVD case scenario (79.5%) than for the embolic CBI scenario (91.3%). However, still a vast majority considered further investigations indicated, also in the SVD case scenario (Table 2). Respondents were more likely to order prolonged rhythm monitoring (e.g. 24 h-ECG 48% vs 36%, p = 0.003), echocardiography (transthoracic echocardiography 61% vs 45, p < 0.001) and non-invasive cervical artery angiography (80% vs 65%, p < 0.001) for the embolic CBI phenotype scenario. Surprisingly, less than half (47.6%) would order a 12-channel and 24 h-ECG and less than a third (31.5%) would order prolonged 7-day ECG monitoring for a likely embolic CBI. However, a substantial proportion felt that a cardiac work-up should be ordered in the SVD case scenario (i.e. 24 h-ECG 36% and transthoracic echocardiography 45%). There was no difference in the rate of respondents that thought that an extended laboratory including lipid profile and HbA1 c should be done between the two scenarios (66% vs 65%, p = 0.97). More respondents thought that in the embolic CBI scenario, revascularization of an ipsilateral cervical artery stenosis would be appropriate even if recommendations for primary prevention by revascularisation were not fulfilled (80% vs 65%, p < 0.001).
Comparison of management according to phenotype of covert brain infarction.
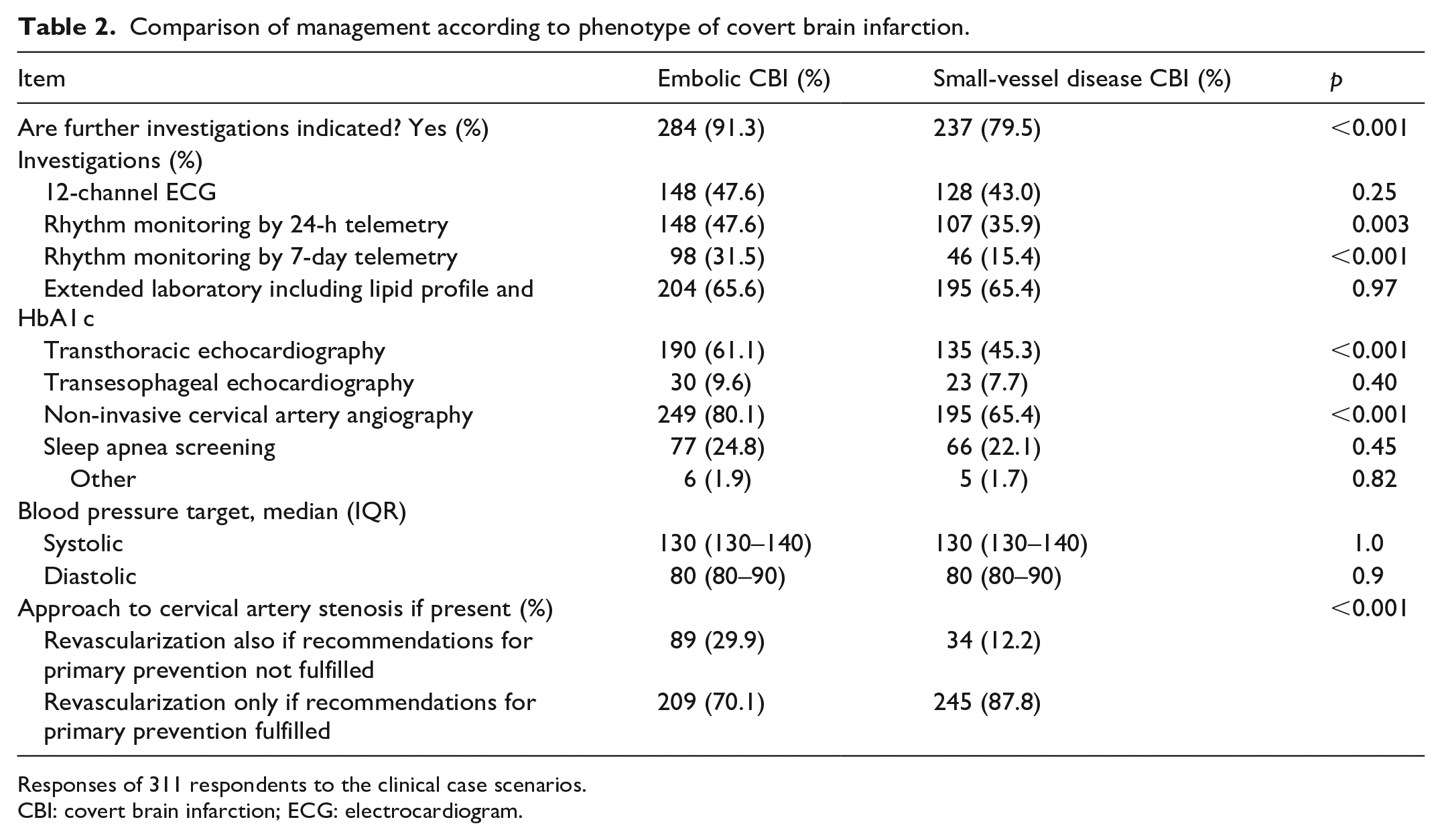
Responses of 311 respondents to the clinical case scenarios.
CBI: covert brain infarction; ECG: electrocardiogram.
Therapeutic management
The majority of respondents said they would initiate lifestyle modification (89%), antithrombotic therapy (77%), lipid-lowering therapy (76%), and antihypertensives (75%). Of those respondents favoring antithrombotic therapy, the majority were in favor of aspirin (81%), whereas few opted for other antiplatelet agents (9%), dual antiplatelet therapy (6%) or direct oral anticoagulants (3%). For the embolic CBI phenotype vignette already taking aspirin, 43% would switch to clopidogrel, whereas 55% would continue aspirin.
Almost twice as many respondents preferred to aim for blood pressure targets according to secondary prevention (64%) as compared to primary prevention (36%) targets. There was a large variation in target low density cholesterol levels with 12% aiming for levels of <3.5 mmol/l (<135 mg/dl), 40% aiming for levels of <2.6 mmol/l (<100 mg/dl), 39% aiming for levels <1.8 mmol/l (<70 mg/dl), and 9% aiming for levels of <1.4 mmol/l (<54 mg/dl).
Sensitivity and subgroup analyses
In the subgroup analysis comparing vascular neurologists and non-vascular neurologists, we made the following observations: vascular neurologists were older, had more years of experience, more often senior positions, and more often worked at university hospitals (Supplemental Material Table 1). Despite this fact, there was no difference in the degree of uncertainty regarding useful investigations and further management of CBI patients (69 points vs 67 points, p = 0.45).
Vascular neurologists more frequently indicated that the detection of any CBI should trigger the (re)assessment of cardiovascular risk factors (98% vs 88%, p = 0.001), were more proactive regarding lifestyle modification (92% vs 75%, p < 0.001), the initiation of lipid-lowering therapy (80% vs 65%, p = 0.008) and antihypertensives (79% vs 64%, p = 0.006). In the non-SVD scenario, they more frequently recommended a 12-channel ECG (59% vs 37%, p < 0.001), extended laboratory investigations (70% vs 58%, p = 0.045) and cervical artery angiography (78% vs 65%, p = 0.026), whereas there were no differences for the SVD scenario.
The main results did not change when excluding neuroradiologists from the analysis regarding the diagnostic and therapeutic management. The subgroup analysis according to workplace showed that those working at a University hospital were more often vascular neurologists and more frequently initiated antithrombotic therapy (73% vs 62%, p = 0.032, Table S2) upon detection of a CBI. The subgroup analysis according to the position showed that those with a senior faculty position more often assess mood and cognition in CBI patients (43% vs 30%, p = 0.025). Senior position holders and Asian respondents also more often initiate lipid-lowering drugs and antihypertensives (Supplemental Tables S3 and S4).
Discussion
Respondents of this international survey were mostly university hospital senior faculty with ample experience in the stroke field and mostly from Europe and Asia. This may suggest lower interest or awareness among practitioners in community-based settings. Even among this experienced group, there was a high degree of uncertainty and heterogeneity at all stages regarding CBI diagnostic evaluations and medical management. While the majority of respondents favored further investigations, uniform consensus on neuroimaging workup, cardioembolic workup with rhythm and echo monitoring and therapeutic approaches varied by embolic versus small-vessel disease CBI.
Given the burden of CBI on brain imaging6,7 and its contribution to neurocognitive decline, cardiovascular outcome events, mobility problems, disability, and mood disorders,9–12 it is surprising that few clinical trials specifically tackle this frequent and easily identifiable population.
Regarding recognition of CBI by the radiology department, there was a significant percentage – even in high-volume academic centers – that reported inconsistent or non-existent mention in reports of brain lesions compatible with CBI in clinical practice. Tools such as structured reporting or natural language processing might assist in reliable CBI recording.22,23 At the neurology level, there was further heterogeneity in response to finding a likely embolic CBI, with surprisingly low rates of basic investigations for cardiac rhythm or structural disorders as compared to manifest stroke. Furthermore, there were significant variations between approaches to embolic and SVD CBI. Slight differences in the case vignettes might explain some of the heterogeneity. This survey identified a discrepancy between the frequency of the problem and the scarcity of institutional written protocols for CBI detection and management.
In accordance to the American Heart and Stroke Association scientific statement, 24 more respondents would include prolonged rhythm monitoring and cardioaortic imaging for the embolic CBI phenotype. However, many would include this also for the SVD CBI case. Such an approach is backed up by a recent study that found similar rates of atrial fibrillation in manifest acute ischemic stroke patients caused by SVD or large-artery stenosis. 25 The subgroup of vascular neurologists had the same level of uncertainty as compared to non-vascular neurologists, despite the fact that they had more years of experience and more often senior positions. The subgroup analyses showed that vascular neurologists were more proactive in ensuring optimal risk factor control as well as work-up for proximal sources of embolism in the non-SVD scenario. Vascular neurologists, senior faculty members, and Asian respondents were also more proactive in initiating secondary prevention strategies including lipid-lowering therapies and antihypertensives.
The majority of respondents indicated that they would start aspirin for any CBI, but there were heterogeneous approaches on which antithrombotic therapy to use. Although a preliminary study in n = 83 patients with CBI found a reduction in manifest acute ischemic stroke over a 4 years follow-up period, 26 larger studies are needed. A subanalysis of the overall neutral Aspirin in Reducing Events in the Elderly (ASPREE) study according to baseline CBI status is ongoing.27–29 However, given the overall harmful effect of Aspirin in the primary prevention setting, even a positive subanalysis would have to be replicated to confirm, whether there are certain phenotypes of CBI that benefit from antiplatelet therapy. Unfortunately, the Antiplatelet Therapy for Silent Brain Infarction (ANTISBI) study (NCT03318744) planned to assess whether aspirin could effectively decrease the incidence of future manifest stroke in CBI patients has not been initiated due to inadequate funding.
It is reassuring, that at least this neuroscience audience was familiar with the associations of CBI with increased risk of detrimental outcomes, although sampling, and non-response bias might lead to overestimation regarding this knowledge. Nevertheless, only a minority included assessment of cognitive function and depression in clinical practice, which might be equally or an even more important outcome to CBI patients as compared to the prevention of vascular events. 30 Similar gaps in risk of post-stroke dementia would be beneficial, in actual practice, most state that they seldom or never had conversations practice of impairment have addressing cognitive been identified for patients with manifest stroke: whereas most healthcare professionals believe that discussing the risk of post-stroke dementia would be beneficial, in actual practice, most state that they seldom or never had conversations about this topic. 31
A prior small qualitative study among a broader audience not restricted to clinical neurosciences, similarly found diagnostic, prognostic, and therapeutic uncertainty in CBI management. 16 Comparable to the subgroup of vascular neurologists in this smaller study, most respondents of our survey population described practices extrapolated from secondary stroke prevention. 16 Only a small fraction (20% for SVD CBI and 9% for embolic CBI) indicated to refrain from further investigations in our survey. Almost all respondents would (re)assess vascular risk factors in accordance to the American Heart and Stroke Association scientific statement 24 and the recent ESO guideline on covert cSVD. 13
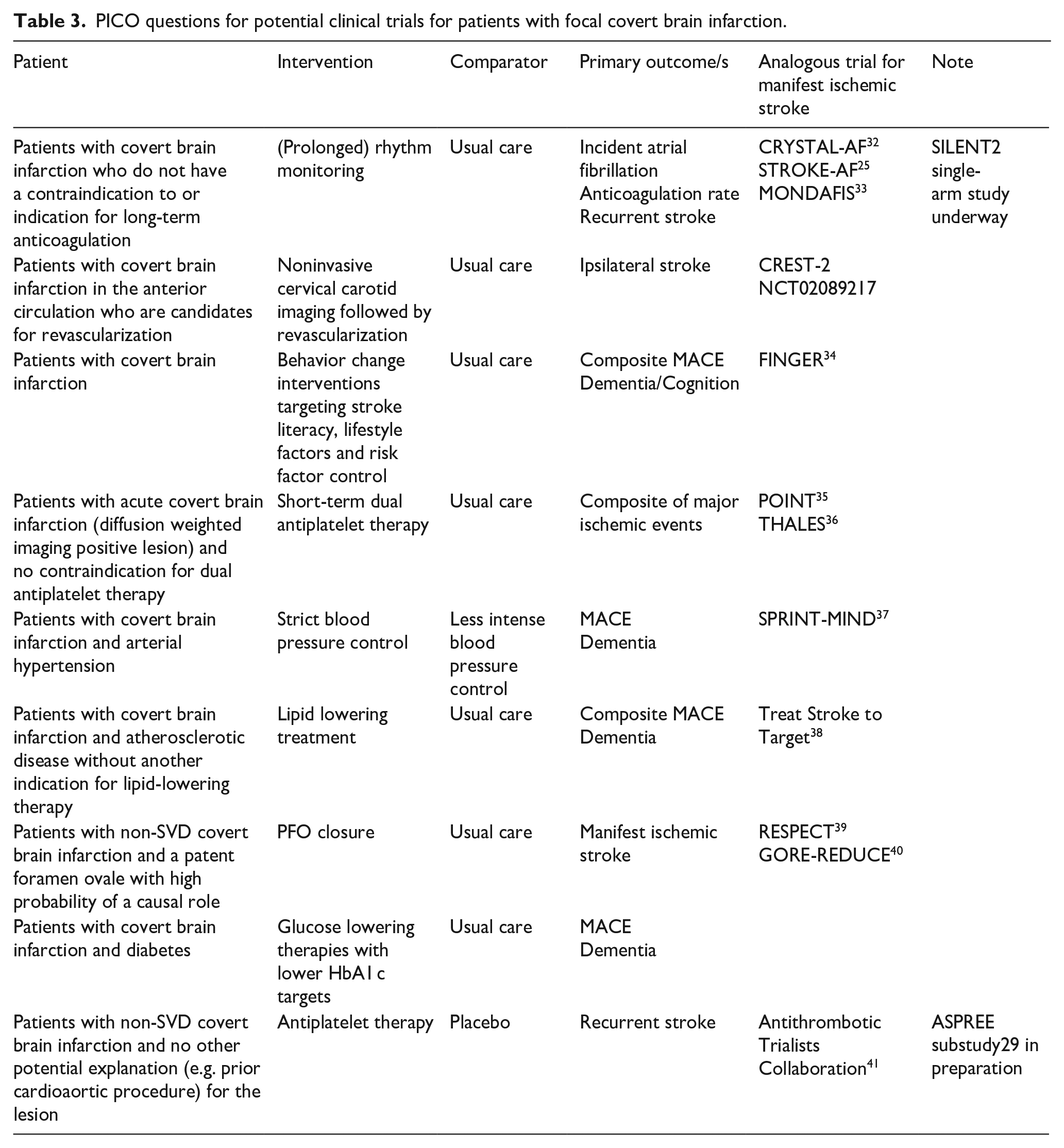
Clinicaltrials.gov lists several studies on covert SVD, but only a handful on patients with potentially embolic CBI: The SILENT prospective observational study aims to assess the yield of likely underlying treatable causes identified through a complete etiological workup including long-term rhythm monitoring, echocardiography, non-invasive angiography of the cervical, and intracranial arteries, and laboratory tests. The embedded SILENT2 study (NCT04449523) uses implantable cardiac monitors to assess the yield of screening for paroxysmal atrial fibrillation in 150 patients with CBI at increased risk. Several trials targeting specifically CBI and its subtypes, could tackle the question, whether recent advances in manifest stroke are also applicable to CBI patients (Table 3).
PICO questions for potential clinical trials for patients with focal covert brain infarction.
For SVD, recently a framework for trial design has been proposed 17 and the ESO guideline on covert SVD also includes recommendations to focus on clinical (not imaging or other intermediary) outcomes in future trials in covert SVD. 13 Importantly, non-SVD phenotypes represent at least 15% of CBI42–44 and should be specifically tackled in clinical trials given the differing pathophysiology. Until more robust evidence is available, this survey identified promising instruments to overcome management uncertainties and facilitate implementation of consistent care:
(1) reliable identification in the (neuro)radiology department, noting in the report to the stroke neurologist, and disclosure to CBI patients to clarify preferences regarding diagnostic and therapeutic management.
(2) composing institutional written protocols that also address associated symptoms such as mobility problems/falls, cognition, and neuropsychiatric disease including depression.
(3) ensuring patients receive and adhere to lifestyle advice
Limitations
First, this survey assessed attitudes and hypothetical management decisions, but did not measure actual clinical practices. Covert cerebrovascular disease comes to attention usually through general internists or other primary providers and is seldom referred to specialists. Hence, the specialist approach might refer to a high-risk group that is based on referral, but probably dramatically underestimates the current practice globally. Furthermore, surveys of clinicians’ practices and attitudes should not be mistaken for optimal evidence-based care and the most frequent responses do not necessarily reflect best practice. The responses may be a reflection of sampling and non-responder as well as partial-response bias potentially affecting the validity and representativeness of the survey. We could not track response rate due to public distribution of the survey via websites of professional associations. The case vignettes were not entirely comparable, since the embolic phenotype already had a cardiovascular event. The survey was only available in English thus with limited generalizability to countries where other languages are spoken, some of which may have much higher prevalence and incidence of CBI. The answers reflect the range in knowledge, personal experiences, and facility resources available to the respondents at the time of the survey. Since we consider this survey hypothesis generating, we did not adjust for multiple testing. Hence, some of our findings might be chance findings.
Conclusion
Even among experienced stroke physicians, there is a high degree of uncertainty and heterogeneity regarding CBI management. Future studies specifically aimed at CBI phenotypes including non-SVD phenotypes are needed. Respondents were more proactive regarding the diagnostic and therapeutic management than the minimum recommended by current expert recommendations. Reliable identification and composing institutional written protocols that also address associated symptoms seem promising first steps to overcome management uncertainties and facilitate implementation of consistent care.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231187444 – Supplemental material for Management of covert brain infarction survey: A call to care for and trial this neglected population
Supplemental material, sj-docx-1-eso-10.1177_23969873231187444 for Management of covert brain infarction survey: A call to care for and trial this neglected population by Thomas R Meinel, Camilla B Triulzi, Johannes Kaesmacher, Adnan Mujanovic, Marco Pasi, Lester Y Leung, David M Kent, Yi Sui, David Seiffge, Philipp Bücke, Roza Umarova, Marcel Arnold, Laurent Roten, Thanh N Nguyen, Joanna Wardlaw and Urs Fischer in European Stroke Journal
Supplemental Material
sj-pdf-2-eso-10.1177_23969873231187444 – Supplemental material for Management of covert brain infarction survey: A call to care for and trial this neglected population
Supplemental material, sj-pdf-2-eso-10.1177_23969873231187444 for Management of covert brain infarction survey: A call to care for and trial this neglected population by Thomas R Meinel, Camilla B Triulzi, Johannes Kaesmacher, Adnan Mujanovic, Marco Pasi, Lester Y Leung, David M Kent, Yi Sui, David Seiffge, Philipp Bücke, Roza Umarova, Marcel Arnold, Laurent Roten, Thanh N Nguyen, Joanna Wardlaw and Urs Fischer in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Swiss National Science Foundation (Grant number 32003B_189077), Swiss Heart Foundation (FF20022), and SAMW/Bangerter Foundation (YTCR 42/19) support multicentre studies on patients with covert brain infarction and the incidence of silent atrial fibrillation in patients with covert brain infarction, led by our group. JMW is part funded by the UK Dementia Research Institute Ltd, which receives its funding from the UK Medical Research Council, Alzheimer’s Research UK and Alzheimer’s Society.’
Ethical approval
The local research ethics board of Bern, Switzerland exempted this study from approval.
Informed consent
Since this is a survey with exemption from ethical approval, no informed consent was needed.
Guarantor
Thomas Meinel is the guarantor for this work.
Authors’ contribution
Design & conduct of the survey: Meinel, Triulzi, Wardlaw, Fischer
Data analysis: Meinel, Triulzi,
Result interpretation: all authors
First manuscript draft: Meinel, Triulzi, Fischer
Manuscript review & critical revisions: all authors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
