Abstract
Background and purpose:
Vasospasm is a common complication of endovascular therapy (EVT). There is a lack of understanding of risk factors for periprocedural vasospasm. Here, we aimed to identify factors associated with vasospasm in patients with acute ischemic stroke who undergo EVT.
Methods:
We conducted a retrospective single-center analysis of patients receiving EVT for anterior circulation vessel occlusion between January 2015 and December 2021. Patients were excluded if they showed signs of intracranial atherosclerotic disease (ICAD) or if they underwent intra-arterial thrombolysis. Study groups were defined as patients developing vasospasm during EVT (V+) and patients who did not (V−). The study groups were compared in univariable analysis. Multivariable regression models were developed to predict the patient’s risk for developing vasospasm based on pre-identified potential prognostic factors. The secondary endpoint was clinical outcome defined as the modified Rankin Scale (mRS) difference between pre-stroke mRS and discharge mRS (delta mRS) and likelihood of successful reperfusion (TICI 2b/3).
Results:
In total, 132/1768 patients (7.5%) developed vasospasm during EVT. Vasospasm was more likely to occur in EVT with multiple thrombectomy attempts and after several stent retriever maneuvers. Factors associated with developing vasospasm were younger age (OR = 0.967, 95% CI = 0.96–0.98) and lower pre-stroke mRS (OR = 0.759, 95% CI = 0.63–0.91). The prediction model incorporating patient age, pre-stroke mRS, stent retriever thrombectomy attempts, and total attempts as prognostic factors was found to predict vasospasm with good accuracy (AUC = 0.714, 95% CI = 0.709–0.720). V+ patients showed higher median (IQR) delta mRS (2 (1–4) vs 2 (1–3); p = 0.014). There was no difference in successful reperfusion (TICI 2b-3) between those with or without vasospasm.
Conclusion:
Vasospasm was a common complication in EVT affecting younger and previously healthy patients. Presence of vasospasm did not reduce the likelihood of successful reperfusion. As independent predictors, patient age, pre-stroke mRS, thrombectomy maneuvers, and stent retriever attempts predict the occurrence of vasospasm during EVT with good accuracy.
Introduction
The description of vasospasm as a complication in endovascular stroke treatment traces back to as early as 2009. Gupta 1 described arterial vasospasm after stent retriever thrombectomy in 2009. Since 2015, with the establishment of endovascular treatment (EVT) as a first-line therapy for acute ischemic stroke, more research emerged concerning the management of procedure failure and complications.2,3 In the early randomized multicenter trials of 2015, REVASCAT and SWIFT PRIME reported on vasospasm with 14 cases in the intervention group of REVASCAT (14/103, 13.6%), four of whom required treatment with a vasodilator medication, and four cases of vasospasm (4/98, 4%) in the intervention group of SWIFT PRIME.4,5 More studies showed the occurrence of intracranial vasospasm in 3%–19% of EVT cases.6,7 A study by Akins et al. 8 described vasospasm as a common complication during EVT noted in 16% of all cases.
Vasospasm during EVT is typically considered to be caused by the mechanical irritation of the vessel by thrombectomy attempts using stent retrievers, aspiration catheters, or both. A single center study of 240 patients found a rate of vasospasm of 2.1% (1/47 patients) in EVT without the use of stent retrievers and a rate of 11.3% (17/151 patients) with stent retriever. 9 In SWIFT PRIME the authors regard the occurrence of intraprocedural vasospasm as nonserious and a transient adverse event without further clinical sequelae in the four cases they observed in their study. 10 Emprechtinger et al. 4 considered vasospasm as a complication of EVT and possible cause for recurrent stroke.
In patients receiving EVT due to acute intracranial vessel occlusion, predictors for the occurrence of vasospasm during EVT have not been defined. The influence of the presence of vasospasm on the patient’s clinical outcome or the success of the target vessel reperfusion is also not understood.
The aim of this study was to determine predictors for the development of vasospasm in acute ischemic stroke patients who undergo anterior circulation EVT as well as to define the influence of vasospasm during EVT on the target vessel reperfusion and the clinical outcome.
Methods
This was a retrospective, single-center study with data recorded from a prospective data base. The study was approved by the Ethics Committee of the University of Heidelberg (ethics approval number S-2472009). Patient consent was waived due to the retrospective nature of this study. All consecutive patients with acute ischemic stroke in the anterior circulation and at least one intracranial thrombectomy attempt were selected from a tertiary stroke center in Germany treated between January 2015 and December 2021. This study is reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline. Data are available upon reasonable request to the corresponding author.
Patient selection and study groups
Inclusion criteria for this study were an intracranial target vessel occlusion in the anterior circulation with at least one thrombectomy attempt. Patients were excluded if they showed signs of intracranial atherosclerotic disease (ICAD) as verified by reviewing the DSA imaging (two neuroradiologists as independent raters). This includes patients, who received percutaneous transluminal angioplasty (PTA) with or without intracranial stenting. Also, patients who underwent intraarterial thrombolysis with or without any additional thrombectomy attempt were excluded (Figure 1). Vasospasm was defined as a concentric vessel constriction (>75% stenosis) visualized on angiography after an EVT maneuver, either during access with a thrombectomy catheter or after a thrombectomy pass. Supported by local guidelines (the study facility’s standard operating procedure (SOP)), the appearance of vasospasm prompted local infusion of intra-arterial nimodipine via microcatheter or via the intermediate catheter flush. Treating intracranial vasospasms, is not on choice of the interventionalist but routinely performed at the study facility. Nimodipine was not routinely added to the flush infusions at the start of EVT.
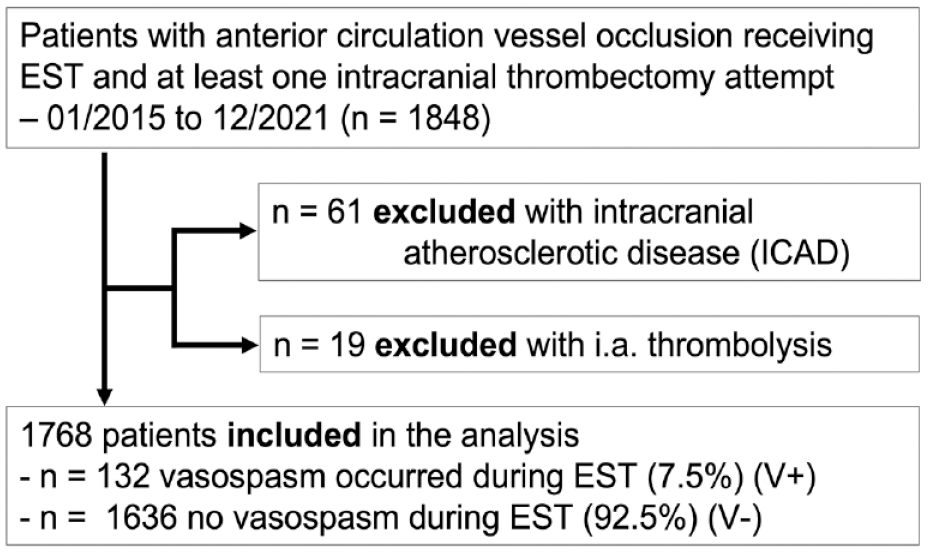
Patient selection for comparison of patients with vasospasm (V+) during endovascular stroke treatment versus no vasospasm (V−) during endovascular stroke treatment; EVT, endovascular stroke treatment.
Two groups were defined according to the presence (V+) or absence (V−) of vasospasm. At the study facility, vasospasm is a complication, that is documented in the obligatory EVT’s interventional report. All cases with documented vasospasm were reviewed by two independent raters (JJ, CW). The two groups were compared in univariable analysis by their relevant clinical and imaging data.
Statistical analysis
Patients’ demographics and clinical examination data were assessed descriptively in both study groups. Continuous variables were described using the median with interquartile range (IQR). For categorical variables, absolute and relative frequencies were provided.
A two-sided Welch’s two-sample t-test and a test of homogeneity (the Fisher’s exact test or Boschloo’s test) were performed for continuous and categorical variables, respectively, to assess whether both groups differed regarding the variables of interest. p-Values of the tests were reported alongside 95% confidence intervals for the mean difference (Welch’s two-sample t-test) and the proportion difference (Fisher’s exact test) or odds ratio (Boschloo’s test). All p-values were interpreted descriptively.
A multivariable analysis was conducted to evaluate whether pre-identified potential prognostic factors impacted on the odds for developing vasospasm. Missing values were imputed 10 times using the predictive mean matching method. For each imputed data set, a variable selection was performed using the elastic net regression with repeated five-fold cross-validation. The set of candidate prognostic factors were the patient age, pre-stroke mRS, stent retriever thrombectomy attempts, total thrombectomy attempts, coronary heart disease, arterial hypertension, number of aspirations maneuvers, patient sex, onset to puncture (in min), site of vessel occlusion, and stenting. At each iteration of the cross-validation procedure, the model and per extension, the subset of prognostic factors which maximized the predictive performance as measured by the Area Under the Curve (AUC) were selected. For each potential prognostic factor, the relative frequency of its inclusion in the best subset of prognostic factors over all 500 cross-validation iterations (100 iterations of five-fold cross validation) and all 10 imputed data sets was then calculated allowing to identify the prognostic factors with the highest predictive value.
Further, to evaluate how the selected prognostic factors impacted the odds of the development of vasospasm, the original data were imputed 10 times again using predictive mean matching and a logistic regression model fitted to each imputed data set with the selected sets of prognostic factors as independent variables. The resulting regression coefficients of all 10 logistic regression models were pooled together following Rubins’ rules and interpreted.
The predictive performance of the fitted regression models was assessed separately using the AUC. Here, a logistic regression analysis with repeated five-fold cross-validation was conducted on each imputed data set. For each imputed data set, the obtained AUC values were averaged over all five cross-validation folds and then over 100 cross-validation iterations. The aggregated AUC values were then logit-transformed, pooled over all imputed data sets using Rubins’ rules and back-transformed to the original scale to obtain a single AUC value and the corresponding 95% confidence interval for each set of selected predictors.
The secondary endpoint was the modified Rankin Scale (mRS) difference before the stroke mRS and at discharge (delta mRS) and was investigated by means of a two-sided Welch’s two-sample t-test. The significance level was set to p = 0.05. A p-value was reported alongside a 95% CI for the mean delta mRS difference between both groups. Statistical analyses were conducted using R version 4.1.3.
Data acquisition
Source data were generated from a prospectively collected stroke database. Additionally, all data included in the present analysis were validated retrospectively to minimize incorrect or missing data (JJ, CW). After review of the reports, the angiographic imaging was reviewed by two independent researchers to assess the occurrence and potential resolution of vasospasm (JJ, CW). Clinical data were assessed by the treating neurologist during the patient’s acute hospital stay.
Results
In this study cohort of 1768 patients with acute ischemic stroke, target vessel occlusion in the anterior circulation and EVT, 132 patients (7.5%) developed vasospasm (V+) during EVT. In 113 cases (6.3%) vasospasm was located intracranially, in 29 cases (1.6%) extracranially, and in 7 cases (0.4%) both intra- and extracranially. In all cases the vasospasm appeared in the vessel segments of the target occlusion, where thrombectomy devices were placed or had to be retrieved from for thrombus removal.
Patients developing vasospasm were younger (median age (IQR), 66 (57–76) vs 78 (67–84); p < 0.001) and with a lower pre-stroke mRS (median (IQR), 0 (0–1) vs 1 (0–2); p < 0.001) compared to patients without vasospasm. Vasospasm patients had less comorbidities such as coronary artery disease (15.2% vs 25.4%, p = 0.01), arterial hypertension, atrial fibrillation, and diabetes type II (Table 1).
Baseline characteristics, procedural metrics, and outcomes of patients with vasospasm (V+) and no vasospasm (V−) during endovascular stroke treatment.
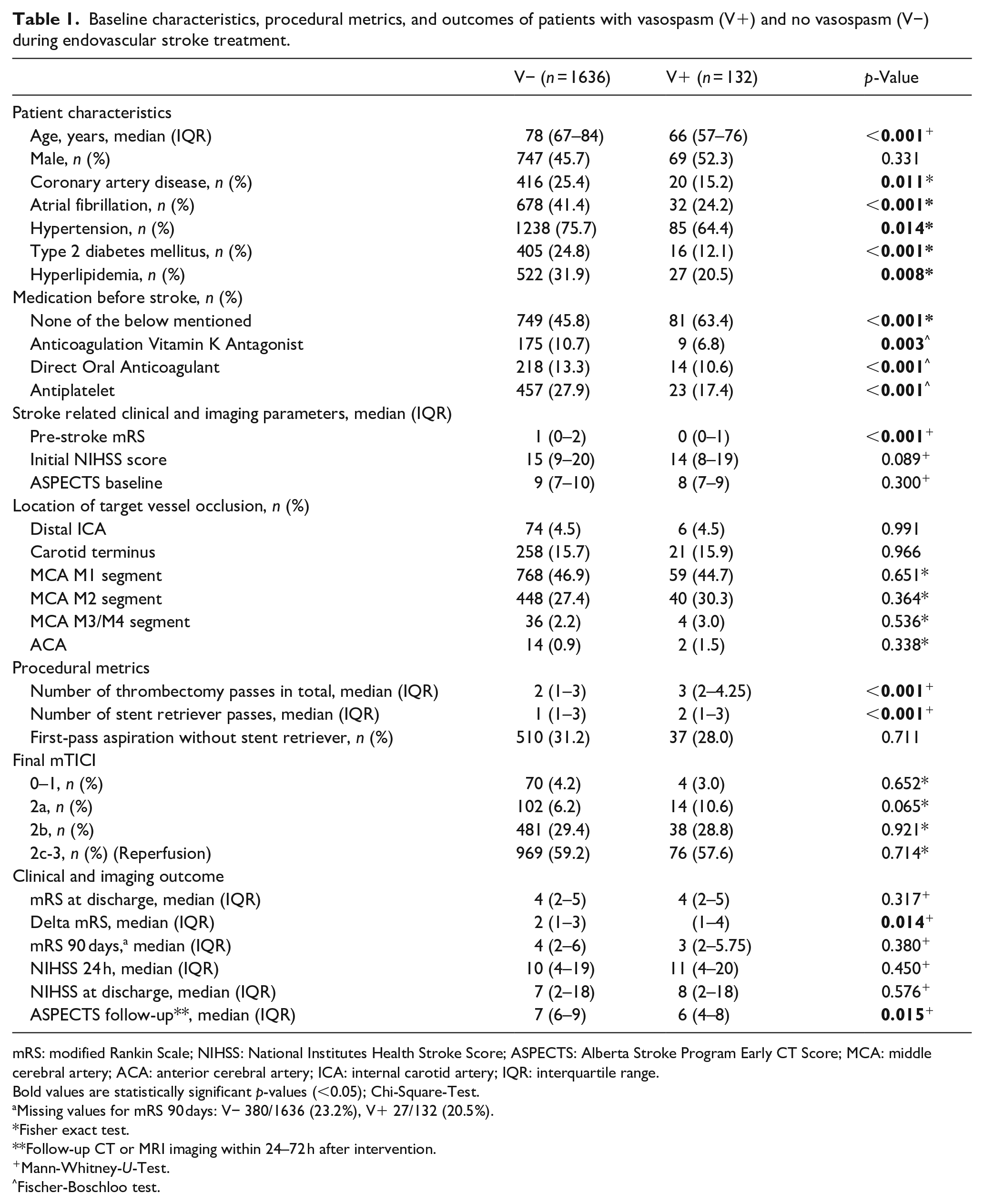
mRS: modified Rankin Scale; NIHSS: National Institutes Health Stroke Score; ASPECTS: Alberta Stroke Program Early CT Score; MCA: middle cerebral artery; ACA: anterior cerebral artery; ICA: internal carotid artery; IQR: interquartile range.
Bold values are statistically significant p-values (<0.05); Chi-Square-Test.
Missing values for mRS 90 days: V− 380/1636 (23.2%), V+ 27/132 (20.5%).
Fisher exact test.
Follow-up CT or MRI imaging within 24–72 h after intervention.
Mann-Whitney-U-Test.
Fischer-Boschloo test.
The target vessel occlusion site did not differ between the two groups (Table 1). Vasospasm patients had more thrombectomy attempts with contact aspiration or stent retriever thrombectomy (median (IQR); 3 (2–4.3) vs 2 (1–3); p < 0.001) as well as more stent retriever passes (median (IQR); 2 (1–3) vs 1 (1–3); p < 0.001). The initial thrombectomy approach – namely contact aspiration or stent retriever thrombectomy – did not influence the occurrence of vasospasm (first maneuver contact aspiration V− 31.2% vs V+ 28.0%, p = 0.711). The prevalence of tandem occlusion and Stent-assisted angioplasty for extracranial high grade ICA stenosis or occlusion was comparable between study groups (n, %; V− 209, 12.8% vs V+, 22, 16.7%, p = 0.21). Vasospasm was treated by local standard operating procedure protocol with intra-arterial nimodipine in 77.8% (105/132) of patients, resulting in near-complete or complete resolution of vasospasm in the majority of cases (86.7%, 91/105) and a comparable mTICI score at the end of the EVT compared to patients without vasospasm (mTICI 2b-3, 86.4% vs 88.6%, p = 0.82).
Elastic net regression analysis revealed that with a 100% selection probability of over 5000 iterations, age, pre-stroke mRS, stent retriever maneuvers, and total number of thrombectomy attempts, had the highest predictive value for developing vasospasm (Table 2). Logistic regression analysis revealed younger patient age and lower pre-stroke mRS to be factors associated with developing vasospasm during EVT in the anterior circulation (age: OR = 0.967, 95% CI = 0.96–0.98, pre-stroke mRS OR = 0.759, 95% CI = 0.63–0.91). For a 1-year age difference, the odds of vasospasm during EVT were 3.2% higher for the younger patient.
Variable selection probability for predicting Vasospasms (n = 5000 iterations).
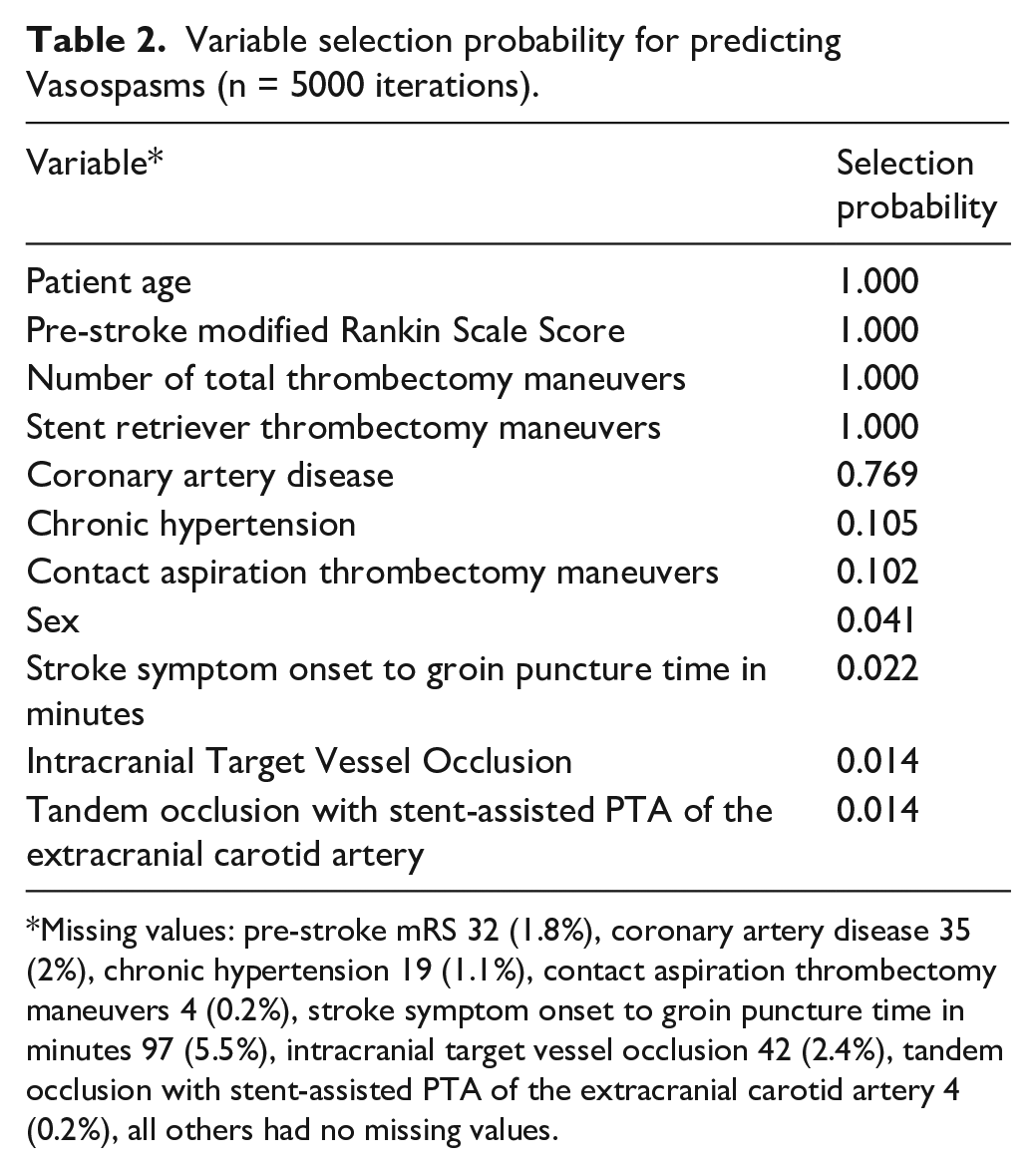
Missing values: pre-stroke mRS 32 (1.8%), coronary artery disease 35 (2%), chronic hypertension 19 (1.1%), contact aspiration thrombectomy maneuvers 4 (0.2%), stroke symptom onset to groin puncture time in minutes 97 (5.5%), intracranial target vessel occlusion 42 (2.4%), tandem occlusion with stent-assisted PTA of the extracranial carotid artery 4 (0.2%), all others had no missing values.
Further analyses revealed that the risk to develop vasospasm during EVT doubled for a patient who was 20 years younger compared to an average patient in this study cohort (Figure 2). Regarding pre-stroke mRS, for two patients with a one-unit difference in pre-stroke mRS score, the odds of vasospasm were 24.1% higher for the patient with the lower score. Although V+ patients showed less comorbidities like coronary artery disease (CAD), including CAD in the model led to only minimal change in the regression coefficients (Table 3).
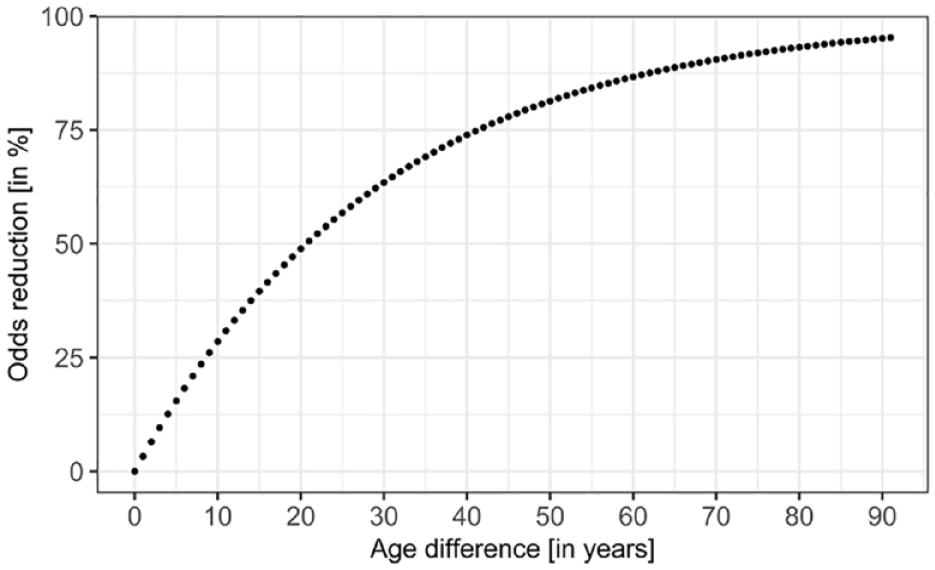
Relationship between patients’ age difference in years and odds reduction for developing vasospasms.
Logistic regression analysis with age, pre-stroke mRS, total number of thrombectomy attempts, stent retriever maneuvers, and coronary artery disease as prognostic factors. Pooled regression coefficients over 10 imputations.
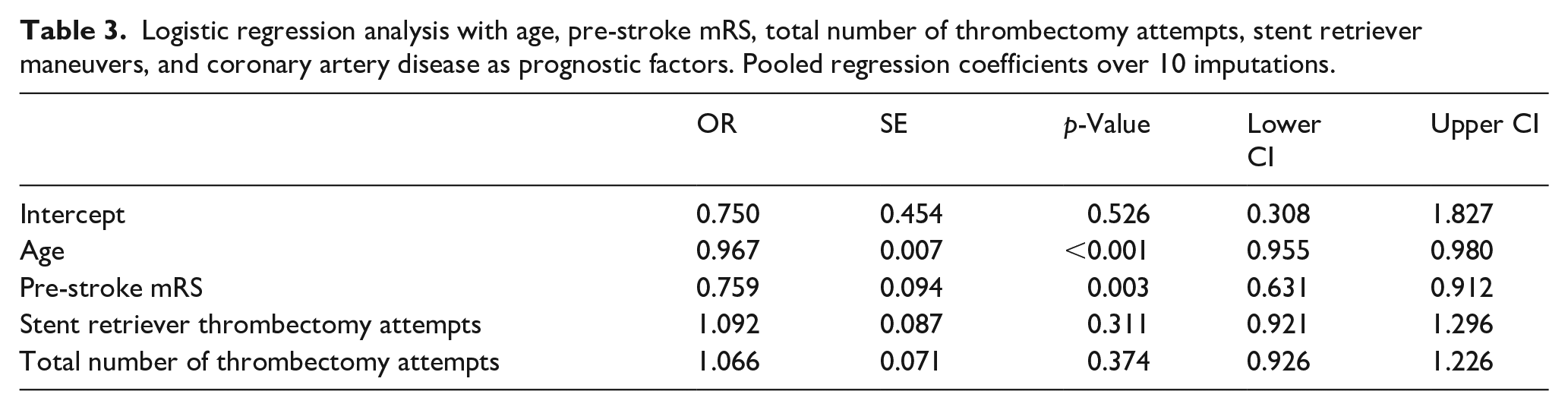
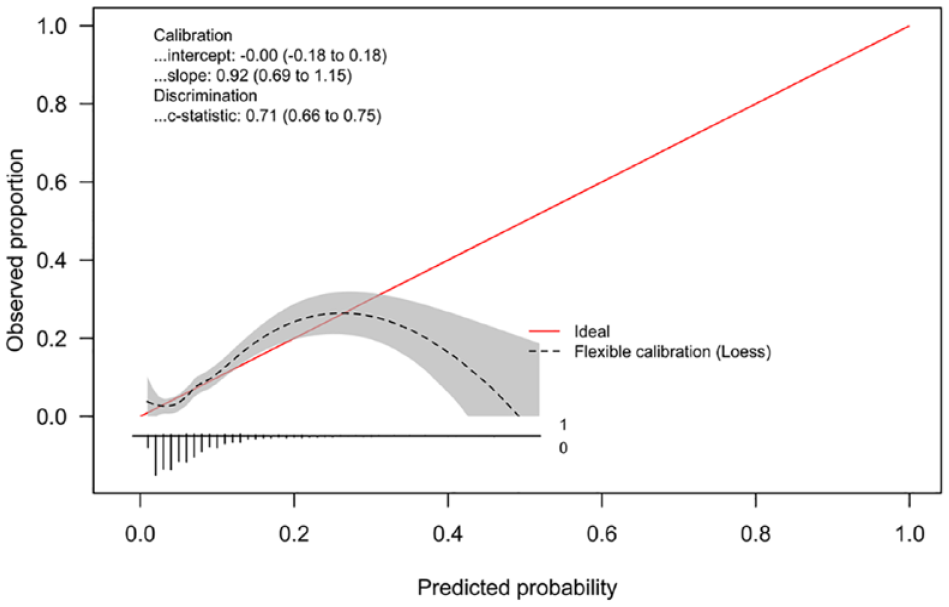
A prediction model comprising patient age, pre-stroke mRS, total number of thrombectomy attempts, and stent retriever maneuvers was found to predict the odds for developing vasospasm with good accuracy (AUC = 0.714, CI = 0.709–0.720). The calibration plot in Figure 3 shows that most of the predicted probabilities were at the lower end and the model seemed to be well calibrated in that area. However, high underpredictions and overpredictions can be observed at higher prediction probabilities. Although the overall calibration as measured by the calibration intercept was zero (intercept = 0, CI = −0.18 to 0.18), the calibration slope was lower than 1 (calibration slope = 0.93, CI = 0.69–1.17) suggesting that the estimated risks were too high for patients who were at high risk and too low for patients who were at low risk. Including CAD in the model did not improve its prediction accuracy (AUC = 0.713, CI = 0.708–0.718, Table 4). The resulting calibration plot was almost identical in shape, and the calibration intercept and slope only changed minimally (Figure 4).
Logistic regression calibration plot for the prediction regression model with age, pre-stroke mRS, total number of thrombectomy attempts, and stent retriever maneuvers (event rate = 7%).
Logistic regression calibration plot for the prediction regression model with age, pre-stroke mRS, total number of thrombectomy attempts, stent retriever maneuvers, and coronary artery disease (event rate = 7%).
Prediction model for vasospasm during endovascular stroke treatment.
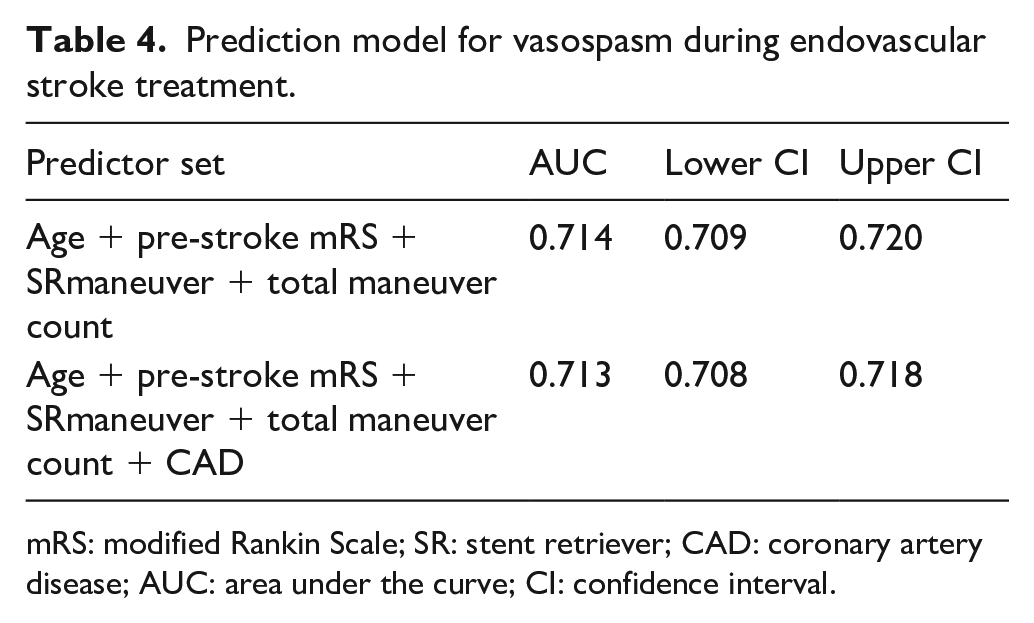
mRS: modified Rankin Scale; SR: stent retriever; CAD: coronary artery disease; AUC: area under the curve; CI: confidence interval.
As for the secondary study endpoint, the difference between discharge mRS and pre-stroke mRS was significantly higher for V+ patients (median (IQR) 2 (1–4) vs 2 (1–3), p = 0.014, Table 1). Follow-up median (IQR) ASPECTS was lower in patients with compared to without vasospasm (6 (4–8) vs 7 (6–9), p = 0.015).
Discussion
Vasospasm was a common complication during EVT in the anterior circulation with a 7.5% rate in this study cohort, which is in line with previous reports ranging from 3% to 23%.6,7 As a main finding, this study showed that vasospasm occurred in younger patients, patients with lower pre-stroke mRS, and are to be expected in EVT procedures with more total passes and especially after multiple stent retriever passes. The first-line thrombectomy technique (contact aspiration or stent retriever thrombectomy under aspiration) did not differ between patients with or without vasospasm. The presence of vasospasm did not affect the likelihood of final successful reperfusion.
To our knowledge, this is the first study presenting predictors for vasospasm during EVT, namely younger patient age, lower pre-stroke mRS, higher number of thrombectomy passes and stent retriever passes. They can predict the risk of vasospasm during EVT with good accuracy (AUC = 0.718). According to this study, the incidence of vasospasm doubles with an age difference of 20 years for the younger patient. The age dependency of cerebral vasospasm is known for patients with subarachnoid hemorrhage11,12 but has not been shown for stroke patients receiving EVT. The younger patients in the V+ group had less comorbidities such as coronary artery disease, which we surmise might be correlated to intracranial arteriosclerosis, decreased vessel wall elasticity and the propensity for the development of vasospasm. However, coronary artery disease did not play a role in the development of vasospasm during EVT in our study.
With regards to procedural factors, with every stent retriever pass, the risk of vasospasm increased by 6%. Previous studies reported a higher rate of vasospasm in patients treated by stent retriever thrombectomy compared to aspiration thrombectomy or treated with more stent retriever passes.9,13 However, this might change in the wake of large-bore aspiration catheters or new stent retriever devices with higher radial force, which are currently developed and might be associated with a higher risk for vasospasm.
As for the management of vasospasm-related complications during EVT, our center followed the recommendation of using calcium channel blockers, such as nimodipine. Nimodipine is a dihydropyridine agent that blocks voltage-gated calcium channels and has a dilatory effect on arterial smooth muscle with a half-life of approximately 9 h. 14 Pilgram-Pastor et al. 3 suggest a rate of 0.5–1 mg nimodipine intra-arterial over several minutes and points out the risk of hypotension and steal phenomena. Others suggest the use of 2–5 mg nimodipine intra-arterial as soon as a tendency to focal vasospasm can be detected. 9 In our study nimodipine efficiently resolved vasospasm caused by EVT in 86.7% of cases, but its influence on infarct growth and stroke outcome is unknown. It is also possible that removal of the thrombectomy device alone, resolves the vasospasm over time. We do not know in how many cases or in which subgroup the vasospasm would resolve spontaneously without medical treatment.
Regarding outcome measures, this study showed that lower ASPECTS scores, corresponding to larger ischemic infarcts, were more common after EVT for patients with vasospasm. This seems surprising as patients with vasospasm were treated immediately with nimodipine leading to a restored vessel diameter in the majority of cases and leading to comparable reperfusion rates in the V+ and V− study groups. Supposedly, this could have been related to vessel re-occlusion after EVT in some of the vasospasm patients or a longer time to achieve reperfusion in those who had V+. As vessel imaging after EVT was not routinely performed, this hypothesis could not be verified and should be investigated in further studies. Also, Emprechtinger et al. 4 discussed that recurrent stroke could be caused by vasospasm induced by EVT. Another hypothesis is that vasospasm compromises the macro- and microcirculation, whereas treatment with intra-arterial drug delivery has an effect merely on larger vessels and not on the microcirculation. Thus, there is still impaired blood flow with subsequent infarction of tissue.
Concordant with our imaging finding of larger infarcts in patients with vasospasm, these patients also showed a worse outcome regarding their mRS at discharge as compared to their initially lower mRS before stroke onset (delta mRS) compared to patients without vasospasm. Consequently, the increased infarct volumes and worse short-term outcome should prompt more studies on the monitoring and management of patients with vasospasm after EVT.
In our study, we could not ascertain whether the ability to retrieve thrombus at the time of the pass in which vasospasm was detected was negatively affected by the presence of vasospasm. Nonetheless, the fact that successful final reperfusion was achieved similarly in patients with and without vasospasm indicates that the presence of vasospasm does not hinder one’s ability to achieve final successful reperfusion.
Limitations of this study are related to the single-center, retrospective design. The interventionalist’s choice of material, thrombectomy technique, and personal expertise might certainly influence the occurrence rate of vasospasm. However, this very large study cohort was treated by 18 different interventionalists in total. The findings in our study may not be generalizable as differing procedure management in response to vasospasm might result in different occurrence rates of vasospasm and their related outcome. The exact positioning of the thrombectomy devices as well as the type of the thrombectomy devices are potentially also related to the occurrence of vasospasm but was not analyzed for this study. This retrospective study does not contain information on the influence of vasospasm on further thrombectomy attempts. Future studies are needed to estimate these influences.
Conclusion
Vasospasm during EVT in the anterior circulation represents a complication affecting mainly the younger and previously healthier stroke patient resulting in larger infarcts and worse short-term outcome. Predictors for the development of vasospasm are younger patient age, lower pre-stroke mRS, higher number of total thrombectomy and stent retriever passes, that predict the occurrence of vasospasm during EVT with good accuracy. The presence of vasospasm did not impact on the ability to achieve final successful reperfusion.
Footnotes
Acknowledgements
The authors wish to acknowledge the excellent clinical stroke care by all members of the department of neurology and neuroradiology, University Hospital Heidelberg, which made this research possible and continues to provide stroke patients and their families with the best opportunity to regain as much physical and mental health as possible.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TN: UNRELATED: advisory board with Idorsia. MB: UNRELATED: grants and personal fees from Bayer, Codman, Guerbet, Medtronic, and Novartis; grants from the German Research Council (DFG), European Union, Hopp Foundation, Siemens, and Stryker; personal fees from BBraun, Böhringer Ingelheim, Roche, Teva, and Vascular Dynamics. MAM: UNRELATED: Board Membership: Codman; Consultancy: Medtronic, MicroVention, Stryker; Grants/Grants Pending: Balt*, MicroVention*; Payment for Lectures Including Service on Speakers Bureaus: Medtronic, MicroVention, Stryker. *Money paid to the institution.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The ethics committee of Medical Faculty, University Hospital Heidelberg, approved this study (REC-number s-249).
Informed consent
Informed consent was not sought for the present study because of the retrospective nature of this study. Waving patient informed consent was approved by the local ethics committee.
Guarantor
JJ
Contributorship
CSW, JJ, MAM researched literature, conceived the study, performed data analysis. JJ, TH, MB, MAM, CSW performed data acquisition. SA, JAV, JJ, CSW performed data analysis. MAM and MB supervised and helped with the study’s ethical approval, CSW and JJ wrote the manuscript’s first draft. JJ was as guarantor involved in all steps of this clinical study from research idea to conceptualization, data acquisition, and analysis as well as writing the manuscript’s first draft. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
This study’s data are available from the corresponding author upon reasonable request.
