Abstract
Background:
Computed tomography angiography (CTA) of the supraaortic arteries is commonly used for acute stroke workup and may reveal apical pulmonary lesions (APL).
Aim:
To determine the prevalence, follow-up algorithms, and in-hospital outcomes of stroke patients with APL on CTA.
Methods:
We retrospectively included consecutive adult patients with ischemic stroke, transient ischemic attack, or intracerebral hemorrhage and available CTA at a tertiary hospital between January 2014 and May 2021. We reviewed all CTA reports for the presence of APL. APL were classified as malignancy suspicious or benign appearing based on radiological-morphological criteria. We performed regression analyses to investigate the impact of malignancy suspicious APL on different in-hospital outcome parameters.
Results:
Among 2715 patients, APL on CTA were found in 161 patients (5.9% [95%CI: 5.1–6.9]; 161/2715). Suspicion of malignancy was present in one third of patients with APL (36.0% [95%CI: 29.0–43.7]; 58/161), 42 of whom (72.4% [95%CI: 60.0–82.2]; 42/58) had no history of lung cancer or metastases. When performed, further investigations confirmed primary or secondary pulmonary malignancy in three-quarters (75.0% [95%CI: 50.5–89.8]; 12/16), with two patients (16.7% [95%CI: 4.7–44.8]; 2/12) receiving de novo oncologic therapy. In multivariable regression, the presence of radiologically malignancy suspicious APL was associated with higher NIHSS scores at 24 h (beta = 0.67, 95%CI: 0.28–1.06, p = 0.001) and all-cause in-hospital mortality (aOR = 3.83, 95%CI: 1.29–9.94, p = 0.01).
Conclusions:
One in seventeen patients shows APL on CTA, of which one-third is malignancy suspicious. Further work-up confirmed pulmonary malignancy in a substantial number of patients triggering potentially life-saving oncologic therapy.
Introduction
Computed tomography angiography (CTA) of the brain-supplying arteries is an essential component of multimodal stroke imaging. The CTA examination window typically extends to the aortic arch. Due to the anatomical proximity, apical lung sections are co-scanned. As a result, incidental pulmonary lesions can be discovered, some of which may conceal potentially treatable lung cancer.
Current recommendations for further management of incidental pulmonary lesions on CTA are primarily based on the risk of malignancy. 1 The risk assessment includes lesion characteristics such as size, location, and morphology, but also individual risk factors such as age and smoking history. 2 However, little is known on the prevalence of apical pulmonary lesions (APL) in CTA performed for acute stroke evaluation. In a recent study, APL were incidentally detected on CTA in 5.7% of patients with acute ischemic stroke. 3 Bentsen et al. 4 found an incidence of approximately 1% of pulmonary malignancies detected on CTA in stroke patients, with smoking being more common in patients with findings suspicious for malignancy.
Unfavorable short- and long-term outcomes have already been described for stroke patients with confirmed cancer.5,6 However, these studies did not focus on pulmonary malignancies but considered cancers in general. There is limited information about how many stroke patients have APL with imaging-based suspicion of malignancy, how many of these patients receive further diagnostic workup and therapy, and whether radiologically malignancy-suspicious APL have an adverse impact on hospital course.
Therefore, we aimed to assess: (i) the prevalence of APL detected on routinely acquired CTA in patients with ischemic and hemorrhagic stroke as well as transient ischemic attack (TIA); (ii) the proportion of patients with APL that are radiologically suspected malignant and receive further diagnostic evaluation and therapy; and (iii) the implications of radiologically malignancy suspicious APL on in-hospital outcomes.
Methods
Study design and baseline characteristics
The study was conducted at the University Hospital Basel, a Swiss tertiary hospital treating more than 1000 stroke cases annually and approved by the Ethics Committee Northwestern Switzerland (ID 2021-01185).
We included all consecutive adults (⩾18 years) from the Swiss Stroke Registry (EKNZ UBE 15/30) who were admitted with a final diagnosis of acute ischemic stroke (AIS), TIA, or intracerebral hemorrhage (ICH) between January 2014 and May 2021. We excluded patients without available CTA during the index hospitalization and those without consent. For patients who were repeatedly hospitalized for suspected stroke during the study period, we considered only the first case with available CTA.
Baseline data collected included demographics (sex, age), hospital diagnosis (AIS, TIA, or ICH), oncologic comorbidities (history of pulmonary and extrapulmonary cancer), medical history (smoking history, chronic obstructive pulmonary disease, deep vein thrombosis, previous cerebrovascular event, atrial fibrillation, diabetes, arterial hypertension), clinical characteristics at admission (National Institutes of Health Stroke Scale [NIHSS] score, premorbid modified Rankin Scale [mRS] score, witnessed stroke symptom onset), stroke treatment characteristics (intravenous tissue-type plasminogen activator [tPA], endovascular treatment), and laboratory parameters including inflammatory (c-reactive protein) and coagulation tests (international normalized ratio, d-dimers) on admission.
Evaluation of computed tomography angiographies
The electronic radiology reports of all patients with available CTA were reviewed (TDD, MA) for the presence of APL. Incidental pulmonary findings were systematically recorded by the internal reporting scheme for CTA of the neck vessels and assessed by two radiologists as part of clinical routine. For CTA, a standardized acquisition window is used at our clinic. This window covers the aortic arch (including the main trunk vessels for the arterial supply of the head, neck, and upper extremity) and extends to the vertex. For this study, APL were divided into malignancy-suspicious and likely benign lesions by two independent reviewers (TDD, MA) blinded for medical history according to the following CT criteria: presence of indistinct margins, absence of calcifications, and spicules radiating from the lesions, applying criteria published in the literature. 7 A possible underlying malignancy was suspected if any of the above radiological criteria were met. Lesion t was not considered in the assessment of malignancy since the lung sections captured on CTA may not represent the entire lesion, potentially preventing reliable size statements. In cases with APL, radiological evidence of emphysema and the presence of inflammatory infiltrative findings were assessed as well.
Standard diagnostic algorithm for apical pulmonary lesions suspected of malignancy
In patients with APL on CTA, we evaluated whether further oncological investigations were recommended. Recommendations were based on expert consensus from the hospital’s pulmonary center until 2018 and were replaced by the Fleischner criteria in 2018 (Supplemental Table 1). 2 We assessed whether a recommended workup was performed either during the index hospitalization or later. In addition, we examined the reasons for not performing a recommended assessment. Modalities of workup included: chest CT, abdomen CT, whole-body PET-CT, and histological and/or cytological investigations.
In-hospital outcome assessment
The examined in-hospital outcome parameters included the NIHSS score at 24 h, (recurrent) ischemic stroke or symptomatic intracranial hemorrhage (sICH; intracranial bleeding occurring within 7 days after the index event that is associated with ⩾4 points worsening in NIHSS), length of hospitalization and all-cause mortality.
Follow-up of patients with apical pulmonary lesions
All records of patients with APL were reviewed (TDD and MA) to determine whether clinical and/or radiologic follow-up had been performed in either pulmonology or oncology (during index hospitalization or later). In cases with malignancy suspicious APL confirmed by further radiological and/or (cyto-)histological examination, it was assessed whether oncologic treatment (i.e., surgical resection, chemotherapy, and/or radiotherapy) had been performed.
Statistical analysis
For group comparisons of baseline, imaging, and outcome characteristics between radiologically malignancy suspicious versus likely benign APL, the chi-squared test was used for categorical variables and the Whitney-U for continuous variables. Uni- and multivariable linear and logistic regression models were used for outcome analyses. Following covariables were included in the final multivariable models, based on the results of the univariable analyses and other known outcome influencing factors: age, sex, admission event type (ischemic vs hemorrhagic), premorbid mRS score, admission NIHSS score, i.v. thrombolysis and endovascular treatment, coronary artery disease, atrial fibrillation, diabetes, history of stroke (ischemic [including transient ischemic attack] and hemorrhagic), TOAST etiology, history of cancer, and presence of not priorly known pulmonary lesions radiologically suspected of malignancy. We included only cases with complete variables of interest in the primary analyses. In addition, multiple imputations of missing covariables were performed as secondary sensitivity analyses.
For right-skewed data, log-transformation was performed for continuous outcome variables with values not including zero, and square root-transformation for outcome variables including zero. p-Values <0.05 were considered significant, we report 95% confidence intervals. Statistical analyses were performed using R statistical software (version 1.4.1106, R Foundation for Statistical Computing, Austria; the packages used are listed in Supplemental Text Section 1).
Results
Baseline characteristics
We studied 7737 patients with AIS, TIA, or ICH at our center between January 2014 and May 2021 (Figure 1). CTAs including apical lung sections were available for 2851 (36.8% [95%CI: 35.8–37.9]; 2851/7737). After accounting for multiple hospitalizations, 2715 patients remained in whom CTA findings were assessed for the presence of APL. APL were present in 5.9% (95%CI: 5.1–6.9; 161/2715). Radiologically, malignancy was suspected in 2.0% off the overall cohort or 36.0% of patients with APL (95%CI: 29.0–43.7; 58/161), with 72.4% of patients (95%CI: 59.8–82.2; 42/58) having no prior history of primary or secondary pulmonary malignancy. Supplemental Figure 1 illustrates an example of an APL classified as malignancy suspicious and an APL classified as likely benign.
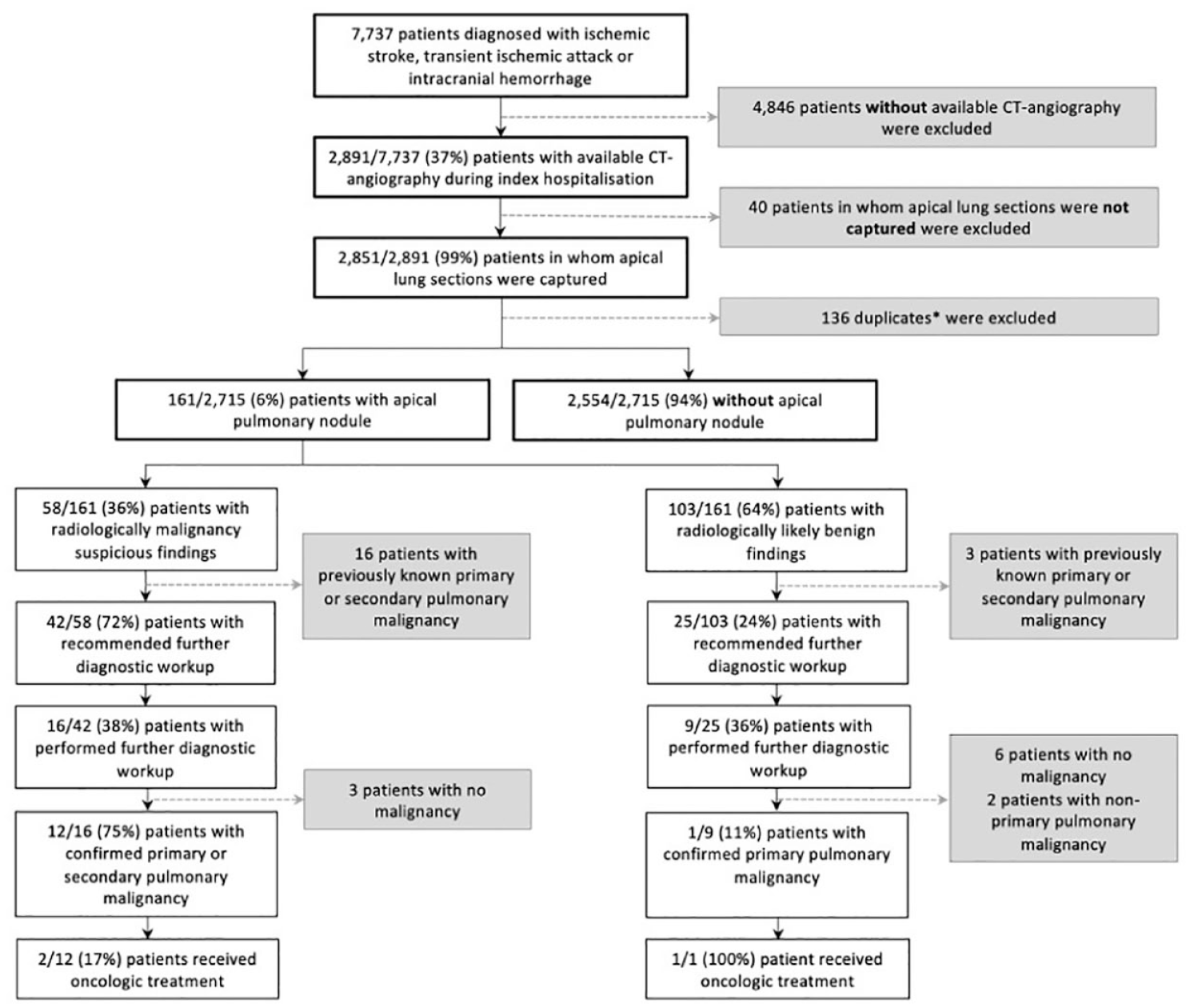
Flow chart of patient selection.
Table 1 shows the baseline characteristics of patients with captured apical lung segments on CTA according to the presence and type of APL. Patients with malignancy suspicious APL were – compared to those with benign APL – younger (median [IQR]: 78 [68–84] vs 80 [73–85], p = 0.048), had more often a history of cancer (pulmonary: 26% vs 3%, p ⩽ 0.001; extrapulmonary: 36% vs 18%, p = 0.02), and of deep vein thrombosis (19% vs 7%, p = 0.04). Concerning the index event, patients with radiological malignancy suspicious APL were clinically more severely affected than those with benign appearing lesions (NIHSS at admission: 9.5 [4.0–15.8] vs 5.0 [2.0–11.0], p = 0.03) and showed higher c-reactive protein levels on admission (8.1 [3.1–34.3] vs 3.0 [1.4–9.7], p ⩽ 0.001).
Baseline characteristics of patients with captured apical pulmonary segments on computed tomography angiography.
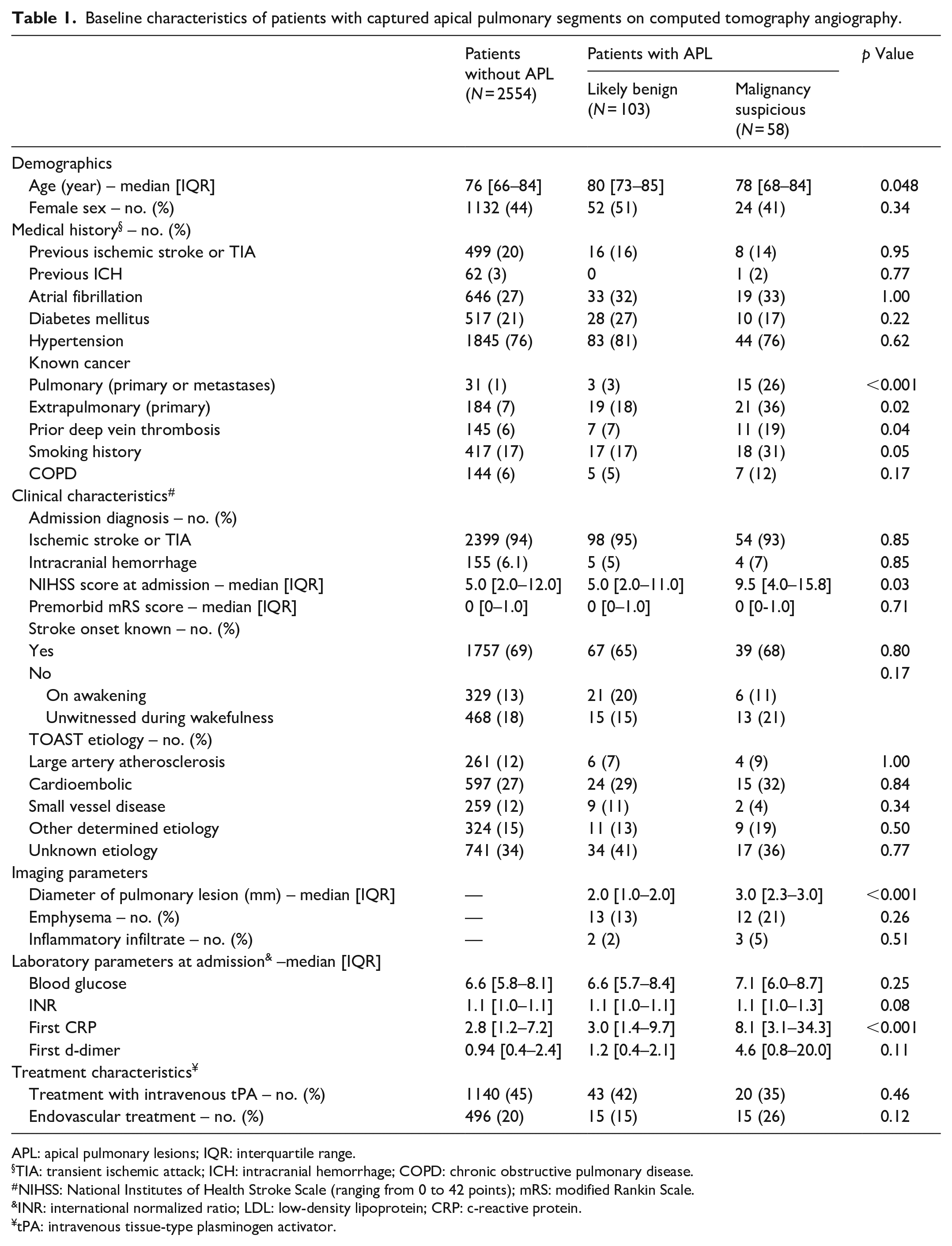
APL: apical pulmonary lesions; IQR: interquartile range.
TIA: transient ischemic attack; ICH: intracranial hemorrhage; COPD: chronic obstructive pulmonary disease.
NIHSS: National Institutes of Health Stroke Scale (ranging from 0 to 42 points); mRS: modified Rankin Scale.
INR: international normalized ratio; LDL: low-density lipoprotein; CRP: c-reactive protein.
tPA: intravenous tissue-type plasminogen activator.
Further oncological diagnosis and treatment
The most common reasons for not performing a workup for cancer (both during the index hospitalization or during follow-up at our clinic) were the recommendation of outpatient re-evaluation of further diagnostic clarification (42.9% [95%CI: 24.5–63.5]; 9/21)
Oncological diagnosis and treatment of patients with confirmed pulmonary malignancy.
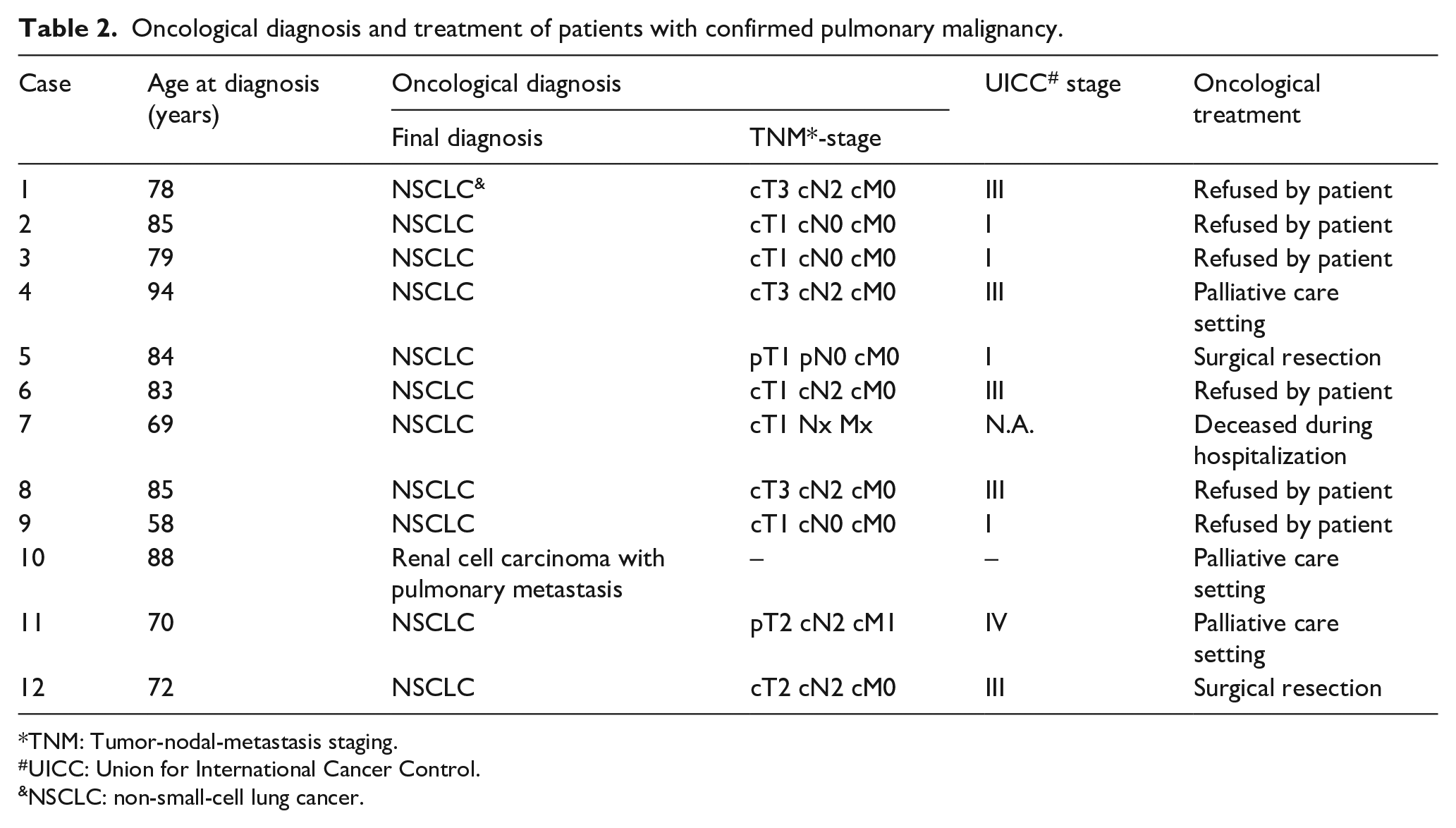
TNM: Tumor-nodal-metastasis staging.
UICC: Union for International Cancer Control.
NSCLC: non-small-cell lung cancer.
In-hospital outcomes
At 24 h after hospital admission, NIHSS of patients with malignancy suspicious APL was higher (7.0 [3.0–13.0] vs 2.0 [0–6.0], p ⩽ 0.001) and so was all-cause in-hospital mortality (19% vs 3%, p = 0.002) compared with those with radiologically likely benign lesions (Table 3). There were no significant differences regarding the frequency of recurrent ischemic stroke, sICH, or hospital length stay. The results of the univariable analyses are presented in Supplemental Table 3. Multivariable regression analyses showed an association between malignancy suspicious APL and higher NIHSS scores at 24 h (square root-beta = 0.67, 95%CI: 0.28–1.06, p = 0.001) and all-cause mortality (aOR = 3.83, 95%CI: 1.29–9.94, p = 0.01), but not for length of hospitalization (log-beta = −0.10, 95%CI: −0.34–0.15, p = 0.43; Tables 3 and 4). Due to limited case numbers, further regression analyses were omitted for the remaining outcome parameters.
In-hospital clinical course of patients according to the presence of pulmonary lesion.
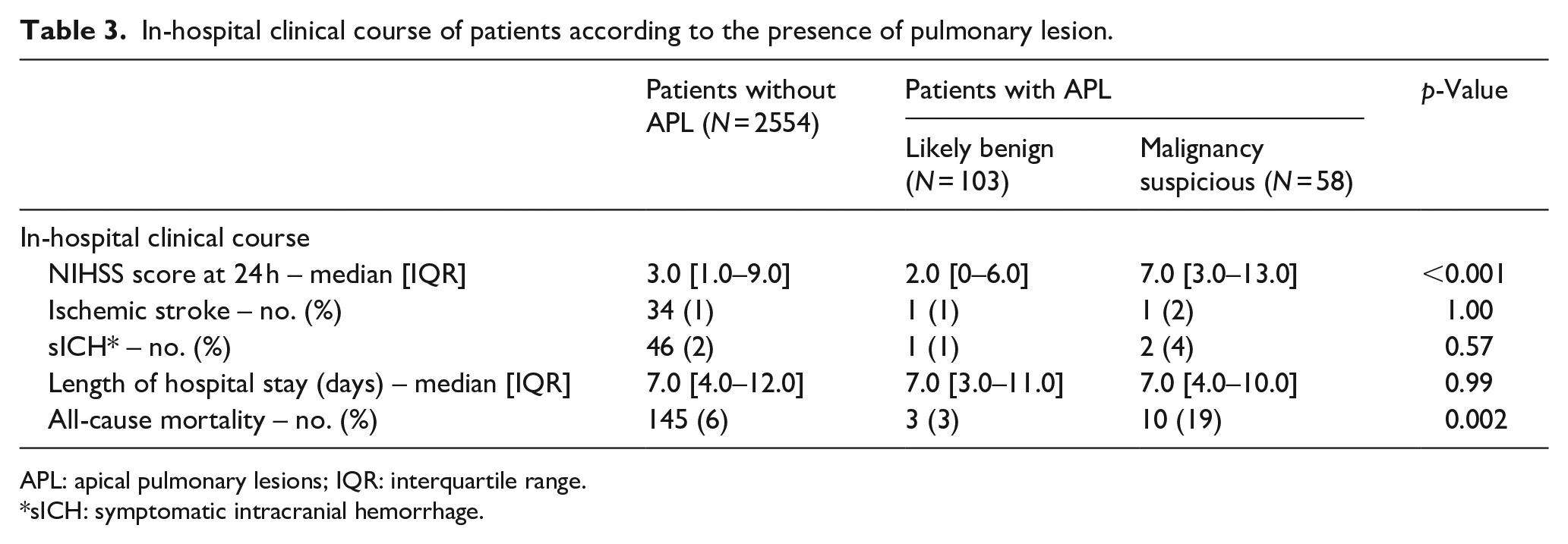
APL: apical pulmonary lesions; IQR: interquartile range.
sICH: symptomatic intracranial hemorrhage.
Multivariable regression outcome analysis according to presence of malignancy suspicious apical pulmonary lesions.
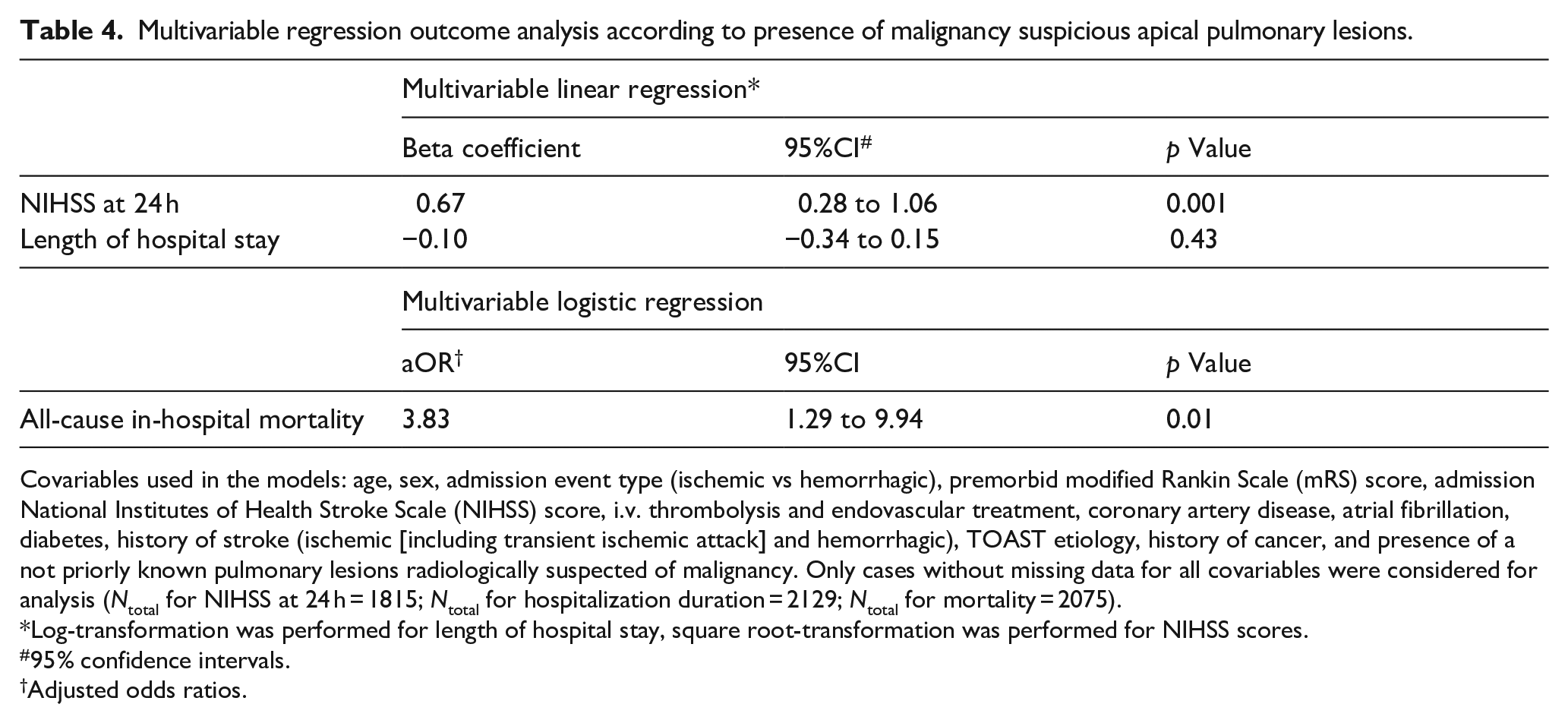
Covariables used in the models: age, sex, admission event type (ischemic vs hemorrhagic), premorbid modified Rankin Scale (mRS) score, admission National Institutes of Health Stroke Scale (NIHSS) score, i.v. thrombolysis and endovascular treatment, coronary artery disease, atrial fibrillation, diabetes, history of stroke (ischemic [including transient ischemic attack] and hemorrhagic), TOAST etiology, history of cancer, and presence of a not priorly known pulmonary lesions radiologically suspected of malignancy. Only cases without missing data for all covariables were considered for analysis (Ntotal for NIHSS at 24 h = 1815; Ntotal for hospitalization duration = 2129; Ntotal for mortality = 2075).
Log-transformation was performed for length of hospital stay, square root-transformation was performed for NIHSS scores.
95% confidence intervals.
Adjusted odds ratios.
In the group of patients with APL, we investigated whether suspected malignancy influenced acute therapy for ischemic stroke (tPA, endovascular treatment; Supplemental Table 4).
Sensitivity analysis
To assess the impact of excluded cases due to incomplete covariable set on the predefined in-hospital outcomes, we repeated the multivariable regression models after multiple imputations (N = 10) of missing covariables. The results were consistent with our primary analyses (Supplemental Tables 5 and 6).
Discussion
Our main findings are: (i) APL were found in 6% of patients undergoing CTA for stroke or TIA; (ii) APL were malignancy suspicious in one-third of patients; and (iii) presence of radiologically malignancy suspicious APL was associated with higher NIHSS scores at 24 h and higher in-hospital mortality.
A recently published randomized trial provides evidence on lung cancer incidence in high-risk male patients (with at least 15 packyears) who received low-dose chest CT. 8 The screening group had a lung cancer incidence rate of 5.6% per 1000 person-years. However, data on the prevalence of APL in CTAs of neck vessels are limited and based on smaller studies than ours. In our cohort, the prevalence of APL was 5.9%, which is in the top end of the range described in the literature (3.3%–5.7%).3,9 This may be partly because incidental findings were often classified according to their clinical significance in previous studies, thus potentially leaving minor findings unaccounted for. In addition, information on the percentage of APL that were radiologically suspicious for malignancy was lacking. In contrast to Chen et al., 9 we studied only patients with a final diagnosis of stroke or TIA. The overlap of risk factors of AIS/TIA and pulmonary malignancies – such as smoking and advanced age10–12 – likely contributed to the higher prevalence in our cohort. Indeed, the proportion of smokers in the APL group was numerically higher compared to the group with probable benign lesions. Of note, the APL group had a significantly more frequent history of deep vein thrombosis. The latter may play a mechanistic role in the context of paraneoplastic thrombophilic diathesis.
Our third finding was that one in three APL was radiologically suspicious of malignancy. Further inpatient or outpatient workup was recommended in most cases but was performed in less than a half. In cases where further investigations were performed, the suspicion of primary or secondary pulmonary malignancy was confirmed in 81.3%. This is a notable result, as there appears to be a discrepancy between general guideline recommendations and clinical practice in stroke patients. The most common reasons for not performing a recommended further evaluation of a suspected malignancy were low urgency (suggesting outpatient re-evaluation) and lack of therapeutic consequence due to severe neurological disability.
These findings underscore the clinical significance of radiologically suspected malignant APL in stroke patients. We demonstrated that radiological suspicion of malignancy is associated with worse in-hospital outcomes; in particular, the adjusted association with higher NIHSS scores at 24 h, which – to our knowledge – has not been described previously.
Our findings are broadly consistent with those of a recent large US study. 6 This study showed that cancer – metastatic and non-metastatic – was associated with higher odds for prolonged hospital stay and in-hospital mortality in patients admitted with acute ischemic stroke. However, in our cohort, we did not observe any differences in hospitalization length. We can only speculate about the underlying reasons: (i) the mostly incidental nature of the malignancy suspicious findings in our cohort may reflect less advanced disease stages in cases with pulmonary malignancy; (ii) some of the radiologically malignancy suspicious APL which were not further investigated may have been benign findings; and (iii) that the recommendation of further outpatient investigations likely resulted in shorter hospitalization durations in patients with malignancy suspicious findings as outpatient evaluations are common practice. Our results underline that stroke patients with malignancy suspicious APL represent a patient group that requires special attention during hospitalization – regardless of whether the suspected cancer is confirmed. Regarding the outcome related to reperfusion therapies in patients with active cancer, a recent large meta-analysis 13 (with a total of around 1000 patients) demonstrated that IVT was associated with an increased risk of symptomatic ICH (OR = 9.80; 95%CI: 3.19–30.13). With respect to EVT, it was shown that it was associated with less favorable outcomes (OR = 0.55; 95%CI: 0.33–0.93) and increased mortality (OR = 2.91; 95%CI: 1.89–4.47). Further studies are needed to clarify whether routine single chest CT screening following CTA might affect long-term outcomes – including mortality – associated with specific types of stroke.
In conclusion, systematic evaluation of apical lung sections included in CTA of stroke patients is essential as it may identify malignancy suspicious APL, which may allow the initial diagnosis of treatable lung cancer in a minority of patients.
Strengths and limitations
Our study has strengths: (i) we used an extensive data set based on the prospectively curated Swiss Stroke Registry; (ii) APL were assessed according to pre-specified criteria by two independent reviewers; (iii) we used simple, comprehensible criteria for radiological malignancy suspicion to make our findings most easily reproducible.
We acknowledge limitations: (i) changes regarding the assessment algorithm of APL took place during the study period due to the introduction of the Fleischner criteria in 2018. Even though there is a considerable overlap between the two algorithms, there was a tendency toward a lower threshold for further examinations in the borderline area of the size categories prior to the introduction of the Fleischner criteria compared to after. This may have influenced the workup rates in our cohort; (ii) lack of further information on stroke treatment related parameters (e.g. recanalization status) and smoking history, especially cumulative dose in packyears. The latter impacts the expected incidence of lung malignancies; (iii) despite the systematic recording of incidental pulmonary findings, it is possible that particularly small lesions were missed or, if detected, de-prioritized because of the focus on acute stroke – not cancer screening; (iv) the actual number of outpatient diagnosed pulmonary malignancies might be higher since we could only record those outpatient investigations that took place at our center.
Conclusion
In acute stroke patients, (i) APL were found in 6% of patients undergoing CTA for stroke or TIA; (ii) APL were malignancy suspicious in one-third of patients, with pulmonary malignancy confirmed in a substantial number of patients undergoing further investigations; (iii) presence of radiologically malignancy suspicious APL was associated with higher NIHSS scores at 24 h and higher in-hospital mortality. Assessing the apical lung segments on CTA can have therapeutic implications.
Supplemental Material
sj-doc-2-eso-10.1177_23969873231151488 – Supplemental material for Apical pulmonary lesions suspected of malignancy visible on neck CT angiography performed for acute stroke: Prevalence, treatment, and clinical implications – the PLEURA study
Supplemental material, sj-doc-2-eso-10.1177_23969873231151488 for Apical pulmonary lesions suspected of malignancy visible on neck CT angiography performed for acute stroke: Prevalence, treatment, and clinical implications – the PLEURA study by Tolga D Dittrich, Mara Aujesky, Salome Rudin, Annaelle Zietz, Benjamin Wagner, Alexandros Polymeris, Valerian L Altersberger, Tim Sinnecker, Henrik Gensicke, Stefan T Engelter, Philippe Lyrer, Viviane Hess, Raoul Sutter, Christian H Nickel, Leo H Bonati, Urs Fischer, Marios Psychogios, Mira Katan and Gian Marco De Marchis in European Stroke Journal
Supplemental Material
sj-docx-1-eso-10.1177_23969873231151488 – Supplemental material for Apical pulmonary lesions suspected of malignancy visible on neck CT angiography performed for acute stroke: Prevalence, treatment, and clinical implications – the PLEURA study
Supplemental material, sj-docx-1-eso-10.1177_23969873231151488 for Apical pulmonary lesions suspected of malignancy visible on neck CT angiography performed for acute stroke: Prevalence, treatment, and clinical implications – the PLEURA study by Tolga D Dittrich, Mara Aujesky, Salome Rudin, Annaelle Zietz, Benjamin Wagner, Alexandros Polymeris, Valerian L Altersberger, Tim Sinnecker, Henrik Gensicke, Stefan T Engelter, Philippe Lyrer, Viviane Hess, Raoul Sutter, Christian H Nickel, Leo H Bonati, Urs Fischer, Marios Psychogios, Mira Katan and Gian Marco De Marchis in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MP has received funds from Medtronic, Stryker, Penumbra, Phenox, Rapid Medical. He has received speakers honoraria from Stryker, Penumbra, Acandis, Phenox, Medtronic. GMDM received speaker honoraria from Medtronic; he declares no other conflicts of interest related to this work. The remaining authors report no conflicts of interest related to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the University Hospital Basel (“The funder”). The funder did not influence the design of the study, data collection, analysis or interpretation of the data.
Informed consent
Patients were informed about the use of their routinely collected data for research purposes. Patients who refused the use of their data were excluded from the analysis.
Ethical approval
The study was approved by the responsible Swiss Ethics Committee (ID: 2021-01185).
Guarantor
TDD.
Author contributions
GMDM and TD planned the work. MA and TD acquired the data and drafted the manuscript. TD performed the analyses. All authors interpreted the results and substantially contributed to the final manuscript.
Data availability
Anonymized records may be provided upon reasonable request by the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
