Abstract
Background
Patients with migraine might be more susceptible of spreading depolarizations, which are known to affect vascular and neuronal function and penumbra recovery after stroke. We investigated whether these patients have more severe stroke progression and less favorable outcomes after recanalization therapy.
Methods
We included patients from a prospective multicenter ischemic stroke cohort. Lifetime migraine history was based on the International Classification of Headache Disorders II criteria. Patients without confirmed migraine diagnosis were excluded. Patients underwent CT angiography and CT perfusion <9 h of onset and follow-up CT after three days. On admission, presence of a perfusion deficit, infarct core and penumbra volume, and blood brain barrier permeability (BBBP) were assessed. At follow-up we assessed malignant edema, hemorrhagic transformation, and final infarct volume. Outcome at three months was evaluated with the modified Rankin Scale (mRS). We calculated adjusted relative risks (aRR) or difference of means (aB) with regression analyses.
Results
We included 600 patients of whom 43 had migraine. There were no differences between patients with or without migraine in presence of a perfusion deficit on admission (aRR: 0.98, 95%CI: 0.77–1.25), infarct core volume (aB: -10.8, 95%CI: -27.04–5.51), penumbra volume (aB: -11.6, 95%CI: -26.52–3.38), mean blood brain barrier permeability (aB: 0.08, 95%CI: -3.11–2.96), malignant edema (0% vs. 5%), hemorrhagic transformation (aRR: 0.26, 95%CI: 0.04–1.73), final infarct volume (aB: -14.8, 95%CI: 29.9–0.2) or outcome after recanalization therapy (mRS > 2, aRR: 0.50, 95%CI: 0.21–1.22).
Conclusion
Elderly patients with a history of migraine do not seem to have more severe stroke progression and have similar treatment outcomes compared with patients without migraine.
Keywords
Introduction
Migraine patients, especially with aura, have an increased risk of ischemic stroke. 1 The association between ischemic stroke and migraine is complex and probably multifactorial.2,3 One interesting mechanism involved in the association is spreading depolarization (SD). SDs are waves of depolarizing neuronal and glia cells, spreading through brain tissue and are the presumed underlying neurovascular mechanism of a migraine aura. SDs have been linked directly to ischemia in pure migrainous infarction. Besides that, it has been shown in mice that micro-emboli can also trigger SDs, with or without causing microinfarctions4,5 and increased migraine with aura (MA) and stroke occurrence is reported in patients with patent foramen ovale. 6 SDs in healthy brain tissue are probably a benign phenomenon but in ischemic tissue they may increase (secondary) tissue changes after stroke.2,7,8
Within minutes after an ischemic event, cells contributing to the infarct core become necrotic with membrane breakdown, dysfunctional cellular metabolism and energy supply, disturbed ion homeostasis, and loss of cell integrity. The tissue surrounding the core (the penumbra) is “struggling to survive” due to collateral blood supply being borderline sufficient.9,10 Cells in the penumbra are metabolically still active for some time, until the disruption of the cellular homeostasis in these cells also leads to cell death. Within minutes after vessel occlusion, a first depolarization wave travels through the penumbra, triggered by loss of membrane integrity due to hypoxia and energy depletion. Multiple SDs can follow thereafter, resulting in an additional reduction of blood flow and increased ischemic damage with each wave.11–14 These peri-infarct polarizations have been found in patients with malignant middle cerebral artery (MCA) infarctions. 12 Also, SDs possibly affect blood brain barrier (BBB) permeability leading to increased secondary hemorrhagic changes, malignant edema, and faster penumbra turnover into infarct core.15–17
Although SDs also occur in stroke patients without migraine, stroke patients with a history with migraine may be more susceptible for SDs. Mice carrying the familial hemiplegic migraine gene have a decreased cortical SD threshold and increased infarct sizes compared with wild-type mice. 7 In a recent retrospective MRI study of 72 ischemic stroke patients, patients with migraine had a faster penumbra turnover than patients without migraine.18,19 This fast penumbra turnover might restrict the time window for revascularization therapies and decrease the chance of good functional recovery. 3
We investigated with modern CT-stroke imaging techniques in a prospective clinical stroke cohort, whether stroke progression is more severe in patients with migraine than in those without and whether migraine patients have poorer outcomes after intravenous thrombolysis or mechanical thrombectomy.
Methods
Patients
We included patients from a large prospective multicenter cohort study in the Netherlands (Dutch Acute Stroke Trial (DUST) 20 ). The main goal of the DUST study was to analyze the predictive value of computed tomography angiography (CTA) and perfusion (CTP) on clinical outcome in patients with ischemic stroke. Inclusion criteria were: age ≥18 years, stroke symptom onset at time of imaging <9 h, National Institutes of Health Stroke Scale (NIHSS) ≥2 or ≥1 if intravenous thrombolysis (IVT) with rtPA was indicated, and known absence of renal failure or allergy to contrast agent. Patients with discharge diagnoses other than ischemic stroke (for example TIA without CT perfusion defect) were excluded from analysis. Non-contrast CT (NCCT), CTP, and CTA were performed on admission, with scan protocols standardized between centers. Study approval was received from the institutional medical ethics committee of all participating hospitals. Ethical approval was obtained from the medical ethics committee of the University Medical Center Utrecht (UMCU), the Netherlands, in addition to local approval from all participating hospitals. Written informed consent was obtained from patients or their legal representative. The medical ethics committee UMCU waived the need for informed consent for patients who died before informed consent could be obtained.
Follow-up NCCT was repeated around three days with (depending on the consent) additional CTP and CTA or MRI if clinically indicated. CT protocols were adjusted within centers according to the hardware used for comparable results, described in detail previously. 20
Migraine diagnosis
Migraine (past and present) was diagnosed upon admission with a 5-point questionnaire Migraine Screener for Stroke (MISS) 21 by trained headache research nurses. Although this questionnaire has a high negative predictive value (0.99), the positive predictive value (0.80 overall and 0.38 for MA) is less accurate. To exclude false positive diagnoses, we approached all patients with a positive migraine screener (at least 1 question answered with “yes”) for an extensive telephone interview to verify the migraine diagnosis. Interview questions were based on International Classification of Headache Disorders II criteria (ICHD-II) migraine and aura criteria in a (semi-) structured manner. In case of ambiguity, a headache neurologist (GMT) was consulted. Patients were excluded when they were unable to answer screener questions due to severe aphasia or decreased cognitive abilities and when migraine diagnosis could not be verified due to loss of follow-up or refusal to participate in the interview.
Clinical characteristics and outcome
We collected data about cardiovascular risk factors, NIHSS, and modified Rankin Scale (mRS) prior to stroke on admission. Stroke-to-imaging time and mode of treatment (intravenous treatment, mechanical thrombectomy or no revascularization therapy) were recorded. Stroke etiology was classified with the Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification, 22 and infarct localization was scored with the Oxfordshire classification 23 consisting of the subtypes partial anterior circulation infarct (PACI), total anterior circulation infarct (TACI), lacunar infarct (LACI), and posterior circulation infarct (POCI).
After a follow-up period of three months, the mRS was prospectively assessed by a telephone interview performed by a trained research nurse. An mRS > 2 was considered a poor clinical outcome.
Radiological characteristics
All radiological assessments were performed blinded for clinical diagnosis and migraine diagnosis (except for side of symptoms) by trained observers with at least five years of experience in neurovascular imaging. Upon admission, early ischemic changes were evaluated on NCCT with the Alberta Stroke Program Early CT score (ASPECTS) scale. The ASPECTS is a 10-point (0 = all areas involved, 10 = no areas of the MCA territory involved in the infarct) quantitative topographic CT scan score evaluated on two standardized levels. 24 The score was dichotomized ≤7 or > 7 which has previously been identified as a useful predictive cutoff point for clinical functional outcome. 25
Clot Burden Score was determined on CTA as the absence of opacification in the anterior circulation on a 10-point scale where a score of 10 indicates the contrast opacification of all vessel segments and 0 lack of opacification of any vessel segment and dichotomized in <7 and ≥7. 26 Collaterals were determined on CTA in patients with anterior circulation infarction (ACA and MCA) due to a large vessel occlusion. Subsequently collaterals were defined as poor when collateral filling was ≤50% of the affected area.26–28
The CTP parameters cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), and time to peak (TTP) were automatically calculated (using Extended Brilliance Workstation 4.5; Philips Healthcare software and in-house developed perfusion software applying a bSVD algorithm) and displayed in color coded maps; MTT and CBV thresholds to define infarct core and penumbra were applied as described previously. 29 Our primary outcome variable presence of a perfusion deficit (yes or no) at entry was visually assessed using CBF, CBV, MTT, and TTP color coded maps. Infarct core and penumbra area at entry were classified into two levels by applying the ASPECTS to the MTT and CBV thresholds and again dichotomized in <8 and ≥8. 30 Volumes of infarct core and penumbra were calculated within the whole scan range using in-house software, with: Tmax > 6 s for total infarct area and a relative CBF threshold of 30% was used to differentiate between core and penumbra.31,32
We calculated the BBB permeability surface area product with CTP data according to the nonlinear regression method described in detail previously. 33 A BBB permeability ratio >1 indicates that the BBB permeability of the ipsilateral hemisphere is higher compared with that in the contralateral hemisphere.
On follow-up we assessed imaging markers of secondary brain damage including presence of malignant edema, defined as a midline shift ≥5 mm; hemorrhagic transformation, defined using the European Cooperative Acute Stroke Study criteria and included the presence of any subtype; 34 and final infarct volume (cc), quantified by manually delineating hypodense territory on NCCT. 35 Presence of a persistent occlusion was defined as no recanalization visible on follow-up CTA in the subgroup of patients with vessel occlusion at entry.
Data analysis
The primary outcome variable for our study was considered to be the presence of a perfusion deficit. A post-hoc power analysis was performed for this outcome, resulting in sufficient precision to detect a 15% difference in perfusion deficit presence (in 60% of the patients without migraine vs. 75% in those with migraine).
Poisson and linear regression (all univariable and multivariable) analyses with migraine as the determinant were used to calculate (adjusted) risk ratios (RR) or beta-coefficients (Β) including 95% confidence intervals (95% CIs). Adjustments were made for age, sex, stroke-to-imaging time (STI), NIHSS on admission, smoking, and a history of hypertension. In addition, for outcome variables at follow-up, an adjustment for IVT and/or mechanical thrombectomy treatment was added. Three subgroup analyses were performed: (1) for patients with MA because patients with MA might be more susceptible for SDs, (2) for patients with large infarcts (based on Oxfordshire classification at discharge: excluding LACI and unknown) because secondary changes are likely to be dependent on infarct size, and (3) for patients with proven vessel occlusions. Missing values were imputed with single imputation for multivariable analyses. Missing values were not imputed when comprising more than 20% of the data (in case of the BBB permeability ratio), when random imputation would lead to improbable results (“infarct locations” and “occlusion”) and when variables were not missing at random (follow-up imaging variables “malignant edema”, “hemorrhagic transformation”, “final infarct volume”, and “persistent occlusion”).
Results
Patients
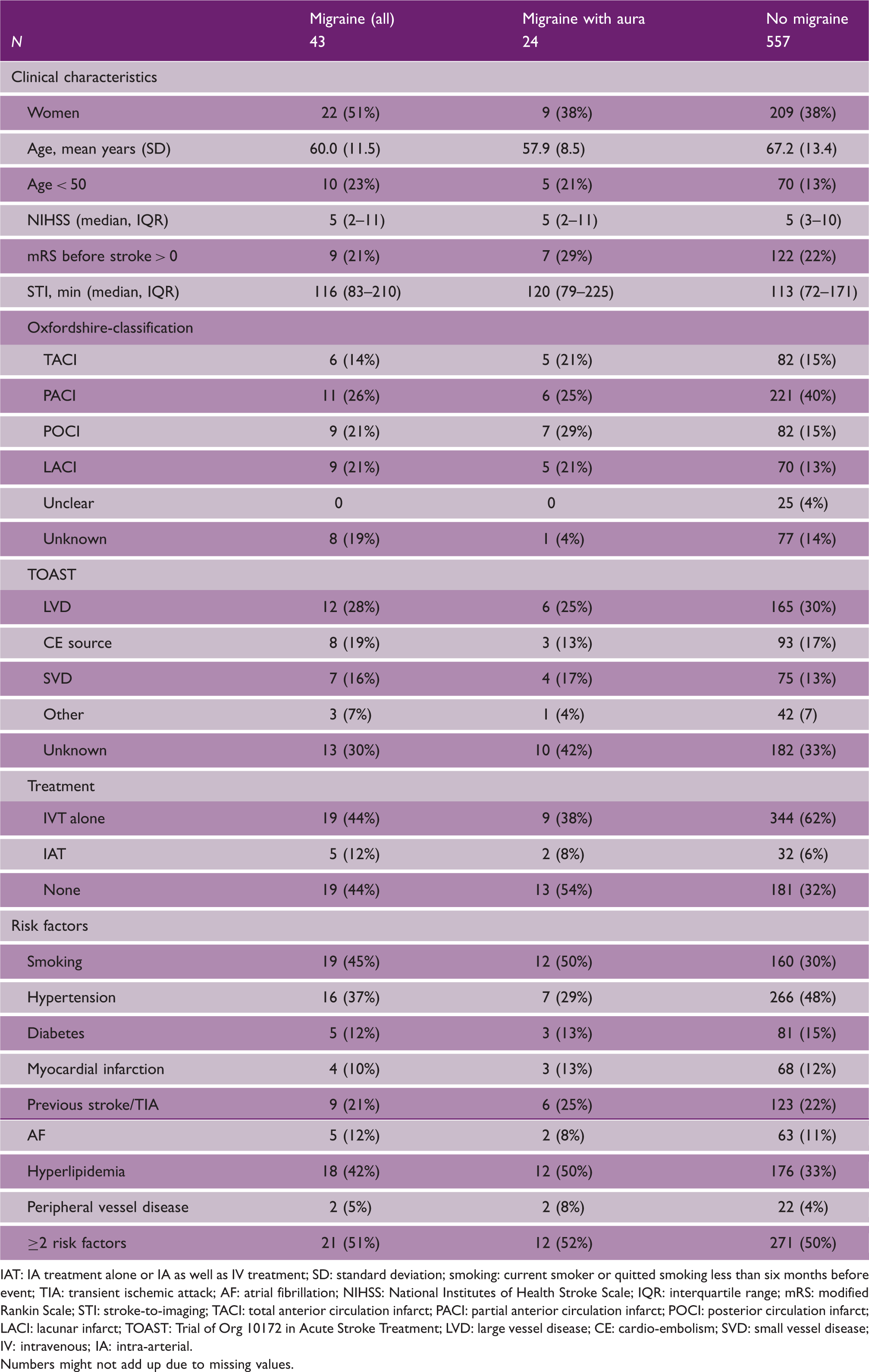
We included 600 patients; 43 (7%) with migraine (24 with aura and 19 without aura) and 557 patients without migraine (Figure 1). Baseline characteristics were comparable between patients with and without migraine, except that patients with migraine were in general younger at time of stroke onset, more often female and more often had lacunar or vertebrobasilar infarctions compared with patients without migraine (Table 1).
Inclusion flow chart. MISS: Migraine Screener for Stroke. Baseline characteristics of patients IAT: IA treatment alone or IA as well as IV treatment; SD: standard deviation; smoking: current smoker or quitted smoking less than six months before event; TIA: transient ischemic attack; AF: atrial fibrillation; NIHSS: National Institutes of Health Stroke Scale; IQR: interquartile range; mRS: modified Rankin Scale; STI: stroke-to-imaging; TACI: total anterior circulation infarct; PACI: partial anterior circulation infarct; POCI: posterior circulation infarct; LACI: lacunar infarct; TOAST: Trial of Org 10172 in Acute Stroke Treatment; LVD: large vessel disease; CE: cardio-embolism; SVD: small vessel disease; IV: intravenous; IA: intra-arterial. Numbers might not add up due to missing values.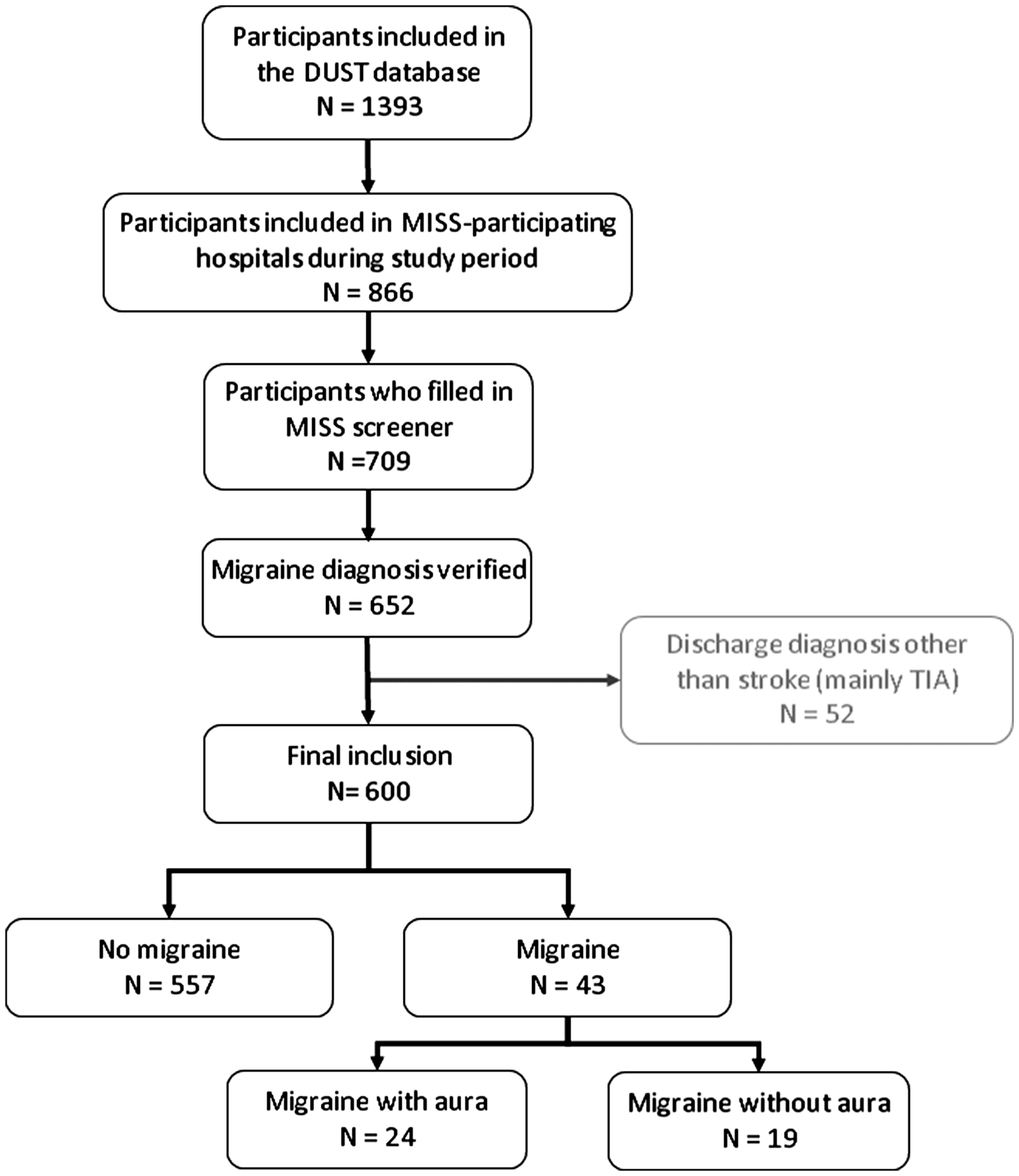
Stroke on admission and follow-up
Radiological characteristics upon admission and follow-up
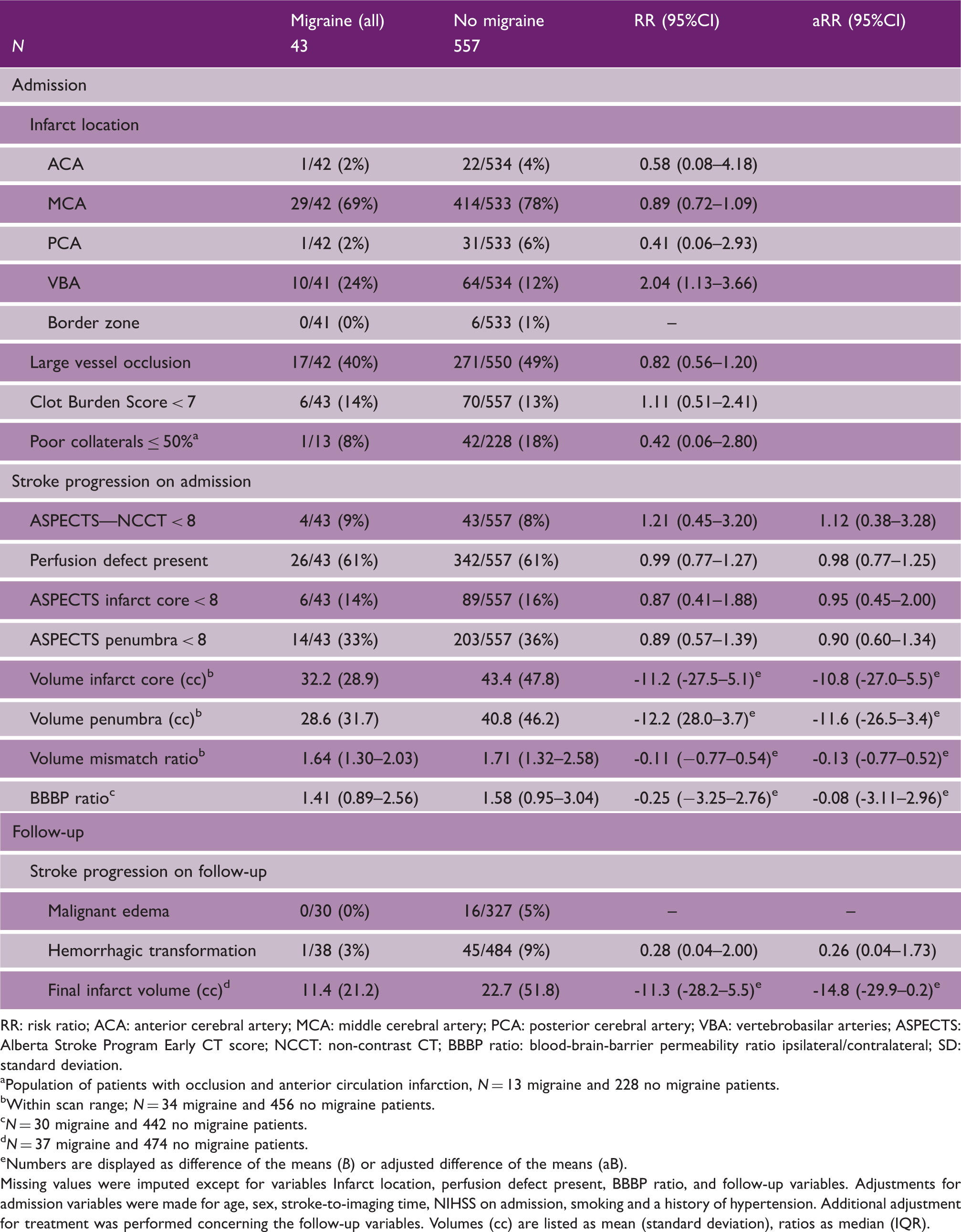
RR: risk ratio; ACA: anterior cerebral artery; MCA: middle cerebral artery; PCA: posterior cerebral artery; VBA: vertebrobasilar arteries; ASPECTS: Alberta Stroke Program Early CT score; NCCT: non-contrast CT; BBBP ratio: blood-brain-barrier permeability ratio ipsilateral/contralateral; SD: standard deviation.
Population of patients with occlusion and anterior circulation infarction, N = 13 migraine and 228 no migraine patients.
Within scan range; N = 34 migraine and 456 no migraine patients.
N = 30 migraine and 442 no migraine patients.
N = 37 migraine and 474 no migraine patients.
Numbers are displayed as difference of the means (B) or adjusted difference of the means (aB).
Missing values were imputed except for variables Infarct location, perfusion defect present, BBBP ratio, and follow-up variables. Adjustments for admission variables were made for age, sex, stroke-to-imaging time, NIHSS on admission, smoking and a history of hypertension. Additional adjustment for treatment was performed concerning the follow-up variables. Volumes (cc) are listed as mean (standard deviation), ratios as median (IQR).
Follow-up imaging on average three days after stroke onset was performed in 522 (87%) patients. In 325 (54%) patients follow-up imaging included CTP. No difference was found between migraine versus non-migraine patients in prevalence of malignant edema (0/30 (0%) 16/327 (5%)) and hemorrhagic transformation (aRR: 0.26, 95%CI: 0.04–1.73) or final infarct volume (aB: -14.8, 95%CI: -29.9–0.2; Table 2, lower part).
Subgroup analysis including only migraine patients with aura or patients with large infarcts or patients with proven vessel occlusions showed essentially similar results (Supplementary Tables 1 and 2).
Treatment outcome
Outcome in patients including sub-analyses for patients treated with intravenous thrombolysis or mechanical thrombectomy for patients with or without migraine (top); patients with or without migraine with aura (Middle) and large infarcts, with or without migraine (bottom)
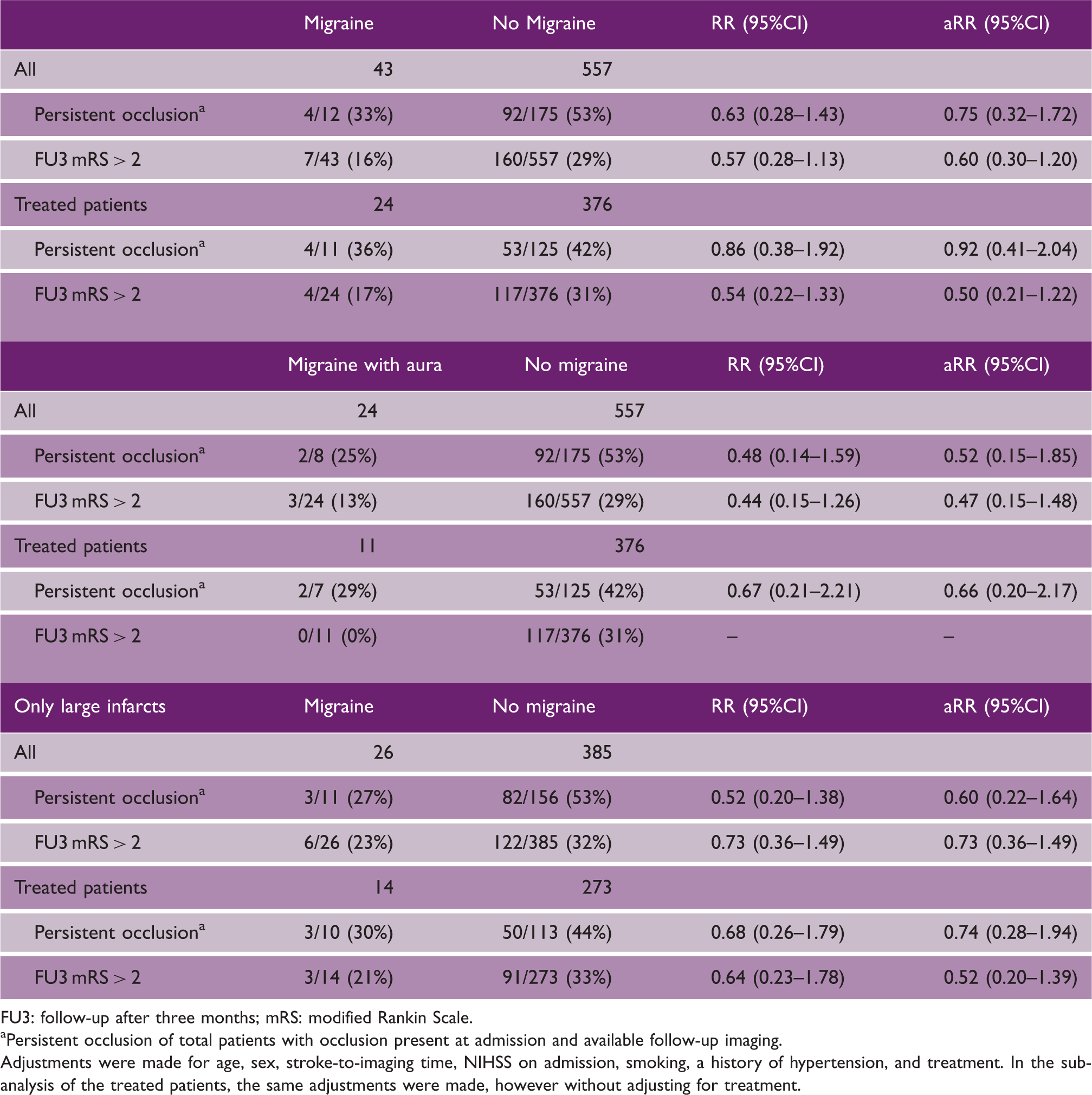
FU3: follow-up after three months; mRS: modified Rankin Scale.
Persistent occlusion of total patients with occlusion present at admission and available follow-up imaging.
Adjustments were made for age, sex, stroke-to-imaging time, NIHSS on admission, smoking, a history of hypertension, and treatment. In the sub-analysis of the treated patients, the same adjustments were made, however without adjusting for treatment.
Discussion
Elderly patients with a history of migraine did not have more severe stroke progression or poorer outcome compared with patients without migraine. Also, no difference in outcome after recanalization treatment was present. If anything, brain injury seemed smaller in patients with migraine although these differences were not statistically significant.
Our findings seem to contradict previous findings in animal studies. Mice carrying the familial hemiplegic migraine type 1 (FHM1) mutation developed faster and had a larger infarct volume after MCA occlusion compared with control mice. 7 This was most likely caused by the lower threshold for occurrence of SDs.11,36,37 However, in these animal studies, MCA occlusion was performed in a standardized way which always resulted in an infarct at the same location. In our study, reflecting clinical practice, we compared patients with infarcts at very heterogeneous locations and including lacunar infarcts. Unfortunately, we were not able to assess penumbra turnover in our patients because follow-up CTP scans were not available in almost half of the patients and volumes of the perfusion deficits were surrounded by wide CIs. In one retrospective study, a non-mismatch pattern was more often seen in migraineurs with aura within 72 h of stroke onset, based on diffusion and perfusion weighted MRI. 19 Although we could not confirm or refute this finding, we found no differences in the extent of early ischemic changes on NCCT, presence of a perfusion deficit or extent of infarct core, and penumbra on admission (CTP) between patients with and without migraine. A possible explanation for the tendency of even less secondary damage in migraineurs is the hypothesis that migraine attacks could be of homeostatic and neuroprotective response to oxidative stress in the brain. 38
Our findings are important for clinical practice, as we found no diminished treatment effect of IVT or mechanical thrombectomy in patients with migraine. Clinical outcome in general was comparable in patients with and without migraine. This finding is in line with the Women's Health study where no difference was found in functional outcome after TIA or stroke in women with a history of migraine, but no treatment effect was assessed. 39 Based on our data there seems to be no reason to restrict the time window for revascularization treatment in migraineurs.
Strengths of our study are the large number of included ischemic stroke patients in a prospective manner in combination with the extensive standardized CT imaging in the acute phase of ischemic stroke. All life-time migraine diagnoses were confirmed by telephone interview with questions based on the formerly ICHD-II criteria but for migraine diagnoses comparable with the new ICHD-III criteria, which decreased the chance of misclassification bias.
Our study also has limitations. For the primary analyses, we combined migraine with and without aura, which could have diluted the effect of MA on stroke characteristics. Spreading depolarization is a mechanism that is in particular related to MA although some studies suggest that it also occurs in migraine without aura. 40 In a secondary analysis, we compared migraine patients with aura with patients without migraine. The effect estimates obtained pointed in the same direction as our main results.
As secondary tissue changes after stroke are dependent on infarct size at admission we also performed an additional analysis including only large infarcts. This analysis again showed comparable results with the main analyses. However, because of the limited group sizes these additional analyses should be interpreted with great caution and replication in future studies with larger number of MA patients is warranted.
Our population was in general about 65 years old at time of their stroke. Because migraine is most active at younger age recall bias might have occurred. We also did not take into account the (possible) use of prophylactic migraine medications which may have influenced occurrence of SDs. Unfortunately, we had no data on the date of the last migraine attack or the presence of active migraine. The higher susceptibility for SDs in migraineurs might be associated with current status of migraine activity. In another study of our group on the effect of migraine history on delayed cerebral ischemia (DCI) after subarachnoid hemorrhage (SAH) we found a statistically significant interaction of the effect with age. 41 Only in young SAH patients migraine history was related to DCI. 41 As the proportion of young stroke patients in our present study is low we feel more data are needed before the current reassuring results can be generalized to patients under the age of 50 years.
Supplemental Material
Supplemental material for Stroke progression and clinical outcome in ischemic stroke patients with a history of migraine
Supplemental material, Supplemental Material for Stroke progression and clinical outcome in ischemic stroke patients with a history of migraine by Inge A Mulder, Ghislaine Holswilder, Marianne AA van Walderveen, Irene C van der Schaaf, Edwin Bennink, Alexander D Horsch, L Jaap Kappelle, Birgitta K Velthuis, Jan Willem Dankbaar, Gisela M Terwindt, Wouter J Schonewille, Marieke C Visser, Michel D Ferrari, Ale Algra, Marieke JH Wermer and for the DUST investigators in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Wermer: ZonMW-Veni grant, ZonMw-VIDI grant, the Dutch Heart Foundation (2011T055) and the Dutch Brain foundation (F2014(1)-22). DUST study: Dutch Heart Foundation (2008T034) and NutsOhra Foundation (0903-012).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
