Abstract
Introduction:
Recent anticoagulant intake represents a contraindication for thrombolysis in acute ischemic stroke. Idarucizumab reverses the anticoagulant effect of dabigatran, potentially allowing for thrombolysis. This nation-wide observational cohort study, systematic review, and meta-analysis evaluated the efficacy and safety of thrombolysis preceded by dabigatran-reversal in people with acute ischemic stroke.
Patients and methods:
We recruited people undergoing thrombolysis following dabigatran-reversal at 17 stroke centers in Italy (reversal-group), people on dabigatran treated with thrombolysis without reversal (no-reversal group), and age, sex, hypertension, stroke severity, and reperfusion treatment-matched controls in 1:7 ratio (control-group). We compared groups for symptomatic intracranial hemorrhage (sICH, main outcome), any brain hemorrhage, good functional outcome (mRS 0–2 at 3 months), and death. The systematic review followed a predefined protocol (CRD42017060274), and odds ratio (OR) meta-analysis was implemented to compare groups.
Results:
Thirty-nine patients in dabigatran-reversal group and 300 matched controls were included. Reversal was associated with a non-significant increase in sICH (10.3% vs 6%, aOR = 1.32, 95% CI = 0.39–4.52), death (17.9% vs 10%, aOR = 0.77, 95% CI = 0.12–4.93) and good functional outcome (64.1% vs 52.8%, aOR = 1.41, 95% CI = 0.63–3.19). No hemorrhagic events or deaths were registered in no-reversal group (n = 12). Pooling data from 3 studies after systematic review (n = 1879), reversal carried a non-significant trend for sICH (OR = 1.53, 95% CI = 0.67–3.50), death (OR = 1.53, 95% CI = 0.73–3.24) and good functional outcome (OR = 2.46, 95% CI = 0.85–7.16).
Discussion and conclusion:
People treated with reperfusion strategies after dabigatran reversal with idarucizumab seem to have a marginal increase in the risk of sICH but comparable functional recovery to matched patients with stroke. Further studies are needed to define treatment cost-effectiveness and potential thresholds in plasma dabigatran concentration for reversal.
Introduction
Despite optimal treatment, people with atrial fibrillation (AF) can still experience stroke while taking oral anticoagulants, a type of medication that also limits the access to intravenous thrombolysis (IVT) in the acute setting. 1 Among direct oral anticoagulants (DOACs) used for stroke prevention in AF, dabigatran can be reverted in the acute setting by administering its binding antibody, idarucizumab. 2 Since recent DOAC intake represents a contra-indication to IVT, a reversal agent can be a useful tool to remove circulating dabigatran and then proceed, safely, to IVT. 3 However, despite some preliminary data supporting such treatment paradigm,3–5 current international guidelines could not formulate a recommendation in favor or against the treatment.6,7 Previous studies were limited in sample size, lacked coagulation tests, long-term outcome assessment and treatment data, substantially limiting the interpretation of results.4,8
Here we report the results of a nation-wide observational cohort study on IVT following dabigatran-reversal, comparing its efficacy and safety with IVT in people not taking dabigatran and people on dabigatran at the time of stroke undergoing IVT without reversal. We integrate our results via systematic review and meta-analysis to provide more solid estimates for functional outcome and risk of bleeding.
Methods
Cohort and outcomes
The primary aim of this study was to define safety and efficacy of IVT after dabigatran reversal. This study was part of national policies on monitoring IVT, according to the Safe Implementation of Thrombolysis in Stroke (SITS). 9 All centers adhering to the Italian Stroke Association were invited to express their interest in participating in the DAB-IVT study. Seventeen centers accepted to participate, providing anonymized center-specific data on patients undergoing IVT after dabigatran-reversal (reversal-group) and patients with stroke while on dabigatran and undergoing IVT without reversal (no-reversal group). A control cohort, consisting of patients receiving IVT who were not taking dabigatran at the time of stroke (control-group), was generated by pooling consecutive patients from prospective registries of four stroke centers (Cesena, Perugia, Gubbio-Città di Castello) adhering to the SITS study. 9 Patients in the reversal group were matched with consecutive control patients undergoing IVT without dabigatran therapy in a 1:7 fashion, taking into account age, sex, hypertension, National Institute of Health Stroke Scale (NIHSS) at presentation, and reperfusion treatment (control-group; Supplemental eFigure 1 for STROBE cohort flow-chart).
Data collection was based on a predefined form. We collected demographic, clinical, neuroradiological data, and functional status measured with modified Rankin Scale (mRS, before stroke and at 90-day follow-up). Baseline and 90-day NIHSS were used to monitor clinical variation. IVT was performed with standard dosage. Dabigatran reversal was obtained with full-dose (5 mg IV) Idarucizumab. The primary outcome was symptomatic intracranial hemorrhage (sICH) according to ECASS-II based definition. 10 Secondary outcomes were any hemorrhagic event, good functional outcome, defined as modified Rankin scale (mRS) 0–2 at 90-day follow-up, and mortality. We also registered recurrent stroke events as a safety surrogate measure.
Systematic review strategy, selection criteria, bias assessment
The search strategy followed a registered protocol (CRD42017060274, 3 OSF-https://osf.io/amdkz/) and PRISMA 11 and MOOSE 12 guidelines. We searched the Cochrane Library, MEDLINE, EMBASE databases, and GoogleScholar for articles published up to 1st March 2022, limiting the results to studies in English language involving human subjects. The search strategy involved the following terms: (i) “Pradaxa” OR “dabigatran” OR “non vitamin k dependent” OR “new oral anticoagulant,” (ii) “idarucizumab” OR “praxbind” OR “reversal” OR “antidote”; (iii) “stroke” OR “ischemic stroke” OR “acute stroke” OR “cerebrovascular event” OR “cerebrovascular accident.” Three authors (MR, EM, DG) independently performed the literature search. Reference lists of all eligible papers were screened for further articles missed during the original search.
The titles and abstracts of the retrieved articles were screened to exclude irrelevant articles and avoid duplications. Any disagreement was resolved by consensus. Data from the included articles were extracted by three authors (MR, EM, DG), including demographics, dabigatran dose and reversal, laboratory tests, stroke features and treatment timing, functional outcome, mortality, and sICH. Case-control studies were selected, and data were extracted for the purpose of meta-analysis of the odds ratio (OR). Case series (without control group) with low risk of bias (moderate or high quality after systematic bias assessment) were selected for the purpose of additional meta-analysis of proportion for the outcome of interest.
The main outcome was sICH, while additional outcomes were good functional outcome, defined as mRS 0–2 at follow-up, and mortality. We reported the lack of data on outcomes, when appropriate. The risk of bias was assessed and reported according to the Newcastle-Ottawa Scale for bias assessment. 13 Publication bias is reported graphically with Funnel plots for outcomes with n > 5 studies available.
Statistical analysis
Descriptive statistics are presented for continuous variables as means and standard deviations, while medians and interquartile ranges are used for discrete and non-normally distributed variables. Categorical variables are presented as counts and percentages. The main analysis was designed to compare reversal versus control-group, with additional analysis included to compare reversal vs no-reversal group. Χ2, Fisher exact test and Student t-test were used for comparison of categorical and continuous variables. The distribution of outcomes is reported as crude OR and 95% confidence interval (95% CI), and as adjusted odds ratio (aOR) according to logistic regression. For the latter purpose, logistic regression was modeled for each outcome. 14 All factors emerging as potential predictors (p < 0.1) of outcome on univariate analysis were imputed in logistic regression for the specific outcome, with treatment group (reversal vs control-group) and age imported a priori. The sample size was calculated based on a potential 15% increase in sICH risk in reversal-group compared to the standard of care, which could be detected at standard significance level with α = 0.75 with a 1:7 matched control population (n = 286). Control population was derived from a pooled stroke registry population, with matching variables including age, sex, hypertension, NIHSS score at presentation, and reperfusion treatment type. Matching was performed with dabigatran-reversal cases to ensure adjustment for confounders; the control population had a zero probability to receive idarucizumab.
Meta-analysis of OR was used to compare the outcome distribution between reversal and control groups in case-control studies. Heterogeneity was calculated and reported according to I2 (moderate = 30%–50%, substantial = 50%–75%, considerable = 75%–100%). We calculated OR with random effects model for all outcomes, and reported results graphically using forest plots. Additional analysis included meta-analysis of proportions to derive pooled estimates including case series with low risk of bias.
Data availability
Data can be made available upon reasonable request from qualified investigator.
Results
DAB-IVT cohort and outcomes
This study included thirty-nine patients receiving IVT following dabigatran-reversal with Idarucizumab (reversal-group), 300 matched controls (control-group), and 12 patients taking dabigatran at the time of stroke and receiving IVT without reversal (no-reversal-group) (STROBE diagram in Supplemental eFigure 1). All patients received IVT at standard dosage (0.9 mg/kg, max 90 mg). Reversal and control groups were similar in age, gender, hypertension, previous myocardial infarction, baseline NIHSS, and rates of endovascular thrombectomy (EVT). People in reversal-group more frequently suffered from diabetes, had longer activated partial thromboplastin time and international normalized ratio at admission, and shorter onset-to-needle time (Table 1).
Cohort characteristics.
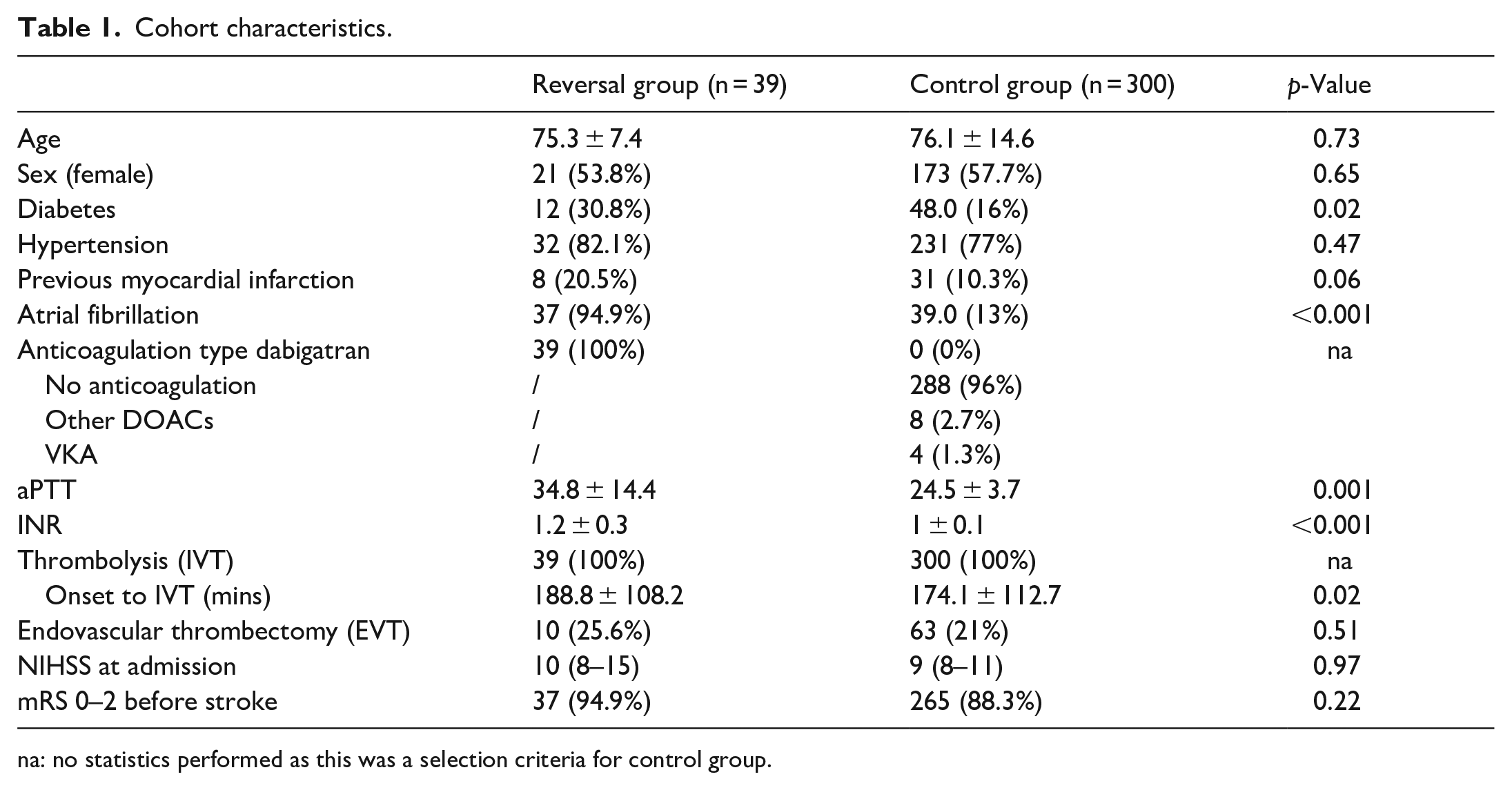
na: no statistics performed as this was a selection criteria for control group.
sICH occurred in 4 (10.3%) patients receiving reversal and 18 (6%) controls (aOR = 1.32, 95% CI = 0.39–4.52, p = 0.65; Table 2). Compared to controls, reversal group had non-significant increase in hemorrhagic transformation (23.1% vs 15%, aOR = 0.84, 95% CI = 0.28–2.59), death (17.9% vs 10%, aOR = 0.77, 95% CI = 0.12–4.93) and good functional outcome (64.1% vs 52.8%, aOR = 1.41, 95% CI = 0.63–3.19; Table 2). One patient in each group had a recurrent stroke. No adverse or venous thrombotic events were reported among patients treated with dabigatran-reversal.
Outcome distribution across treatment groups.
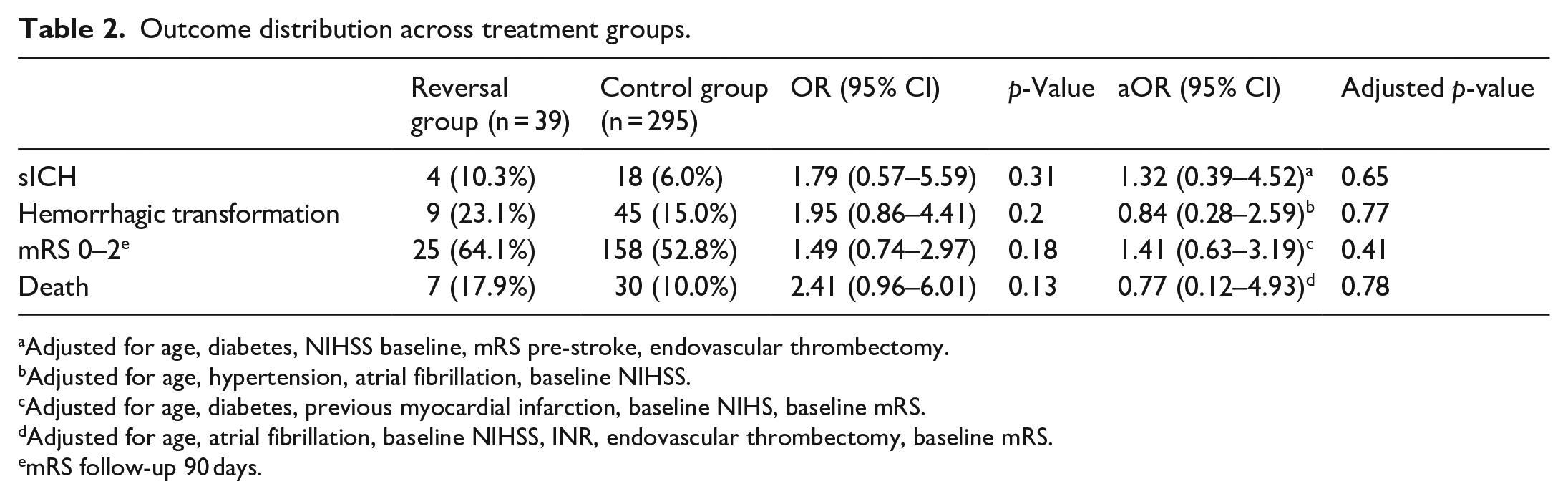
Adjusted for age, diabetes, NIHSS baseline, mRS pre-stroke, endovascular thrombectomy.
Adjusted for age, hypertension, atrial fibrillation, baseline NIHSS.
Adjusted for age, diabetes, previous myocardial infarction, baseline NIHS, baseline mRS.
Adjusted for age, atrial fibrillation, baseline NIHSS, INR, endovascular thrombectomy, baseline mRS.
mRS follow-up 90 days.
People in no-reversal group (n = 12) had lower dabigatran plasmatic concentration compared to people receiving reversal (20 vs 99 ng/ml, p = 0.07), and longer time from last dabigatran intake (1082 vs 404 min, p = 0.02; Supplemental eTable 1). In no-reversal group nine patients had good functional outcome (75%), and no further outcomes were registered. No significant difference in the outcomes considered was found between reversal and no-reversal group (mRS 0–2, 75% vs 64.1%, p = 0.7; death 0% vs 17.9%, p = 0.2; hemorrhagic transformation 0% vs 23.1%, p = 0.1; sICH 0% vs 10.3%, p = 0.6; Supplemental eTable 1).
Systematic review and meta-analysis
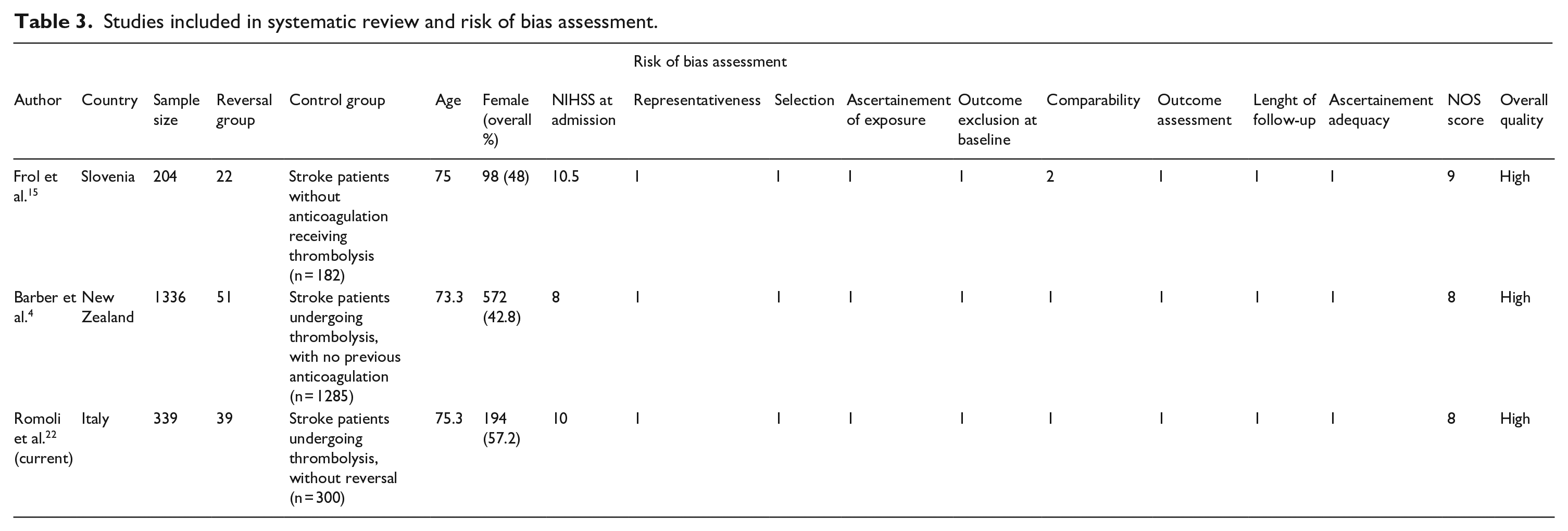
The systematic search retrieved 284 studies, with two studies reaching quantitative synthesis in main analysis,4,15 and four additional case series included in additional analysis (Supplemental eFigure 2 for PRISMA flow-chart). Only two case-control studies were available, one with prospective 15 and one with retrospective design, 4 both of high quality (Table 3). Pooling data from 1879 patients, no significant difference was found for the outcomes of interest between reversal versus control group (Figure 1). However, thrombolysis after reversal carried a non-significant trend for increased risk of sICH (OR = 1.53, 95% CI = 0.67–3.50), death (OR = 1.53, 95% CI = 0.73–3.24), and good functional outcome (OR = 2.46, 95% CI = 0.85–7.16, I2 = 57% with pheterogeneity = 0.13; Figure 1).
Studies included in systematic review and risk of bias assessment.
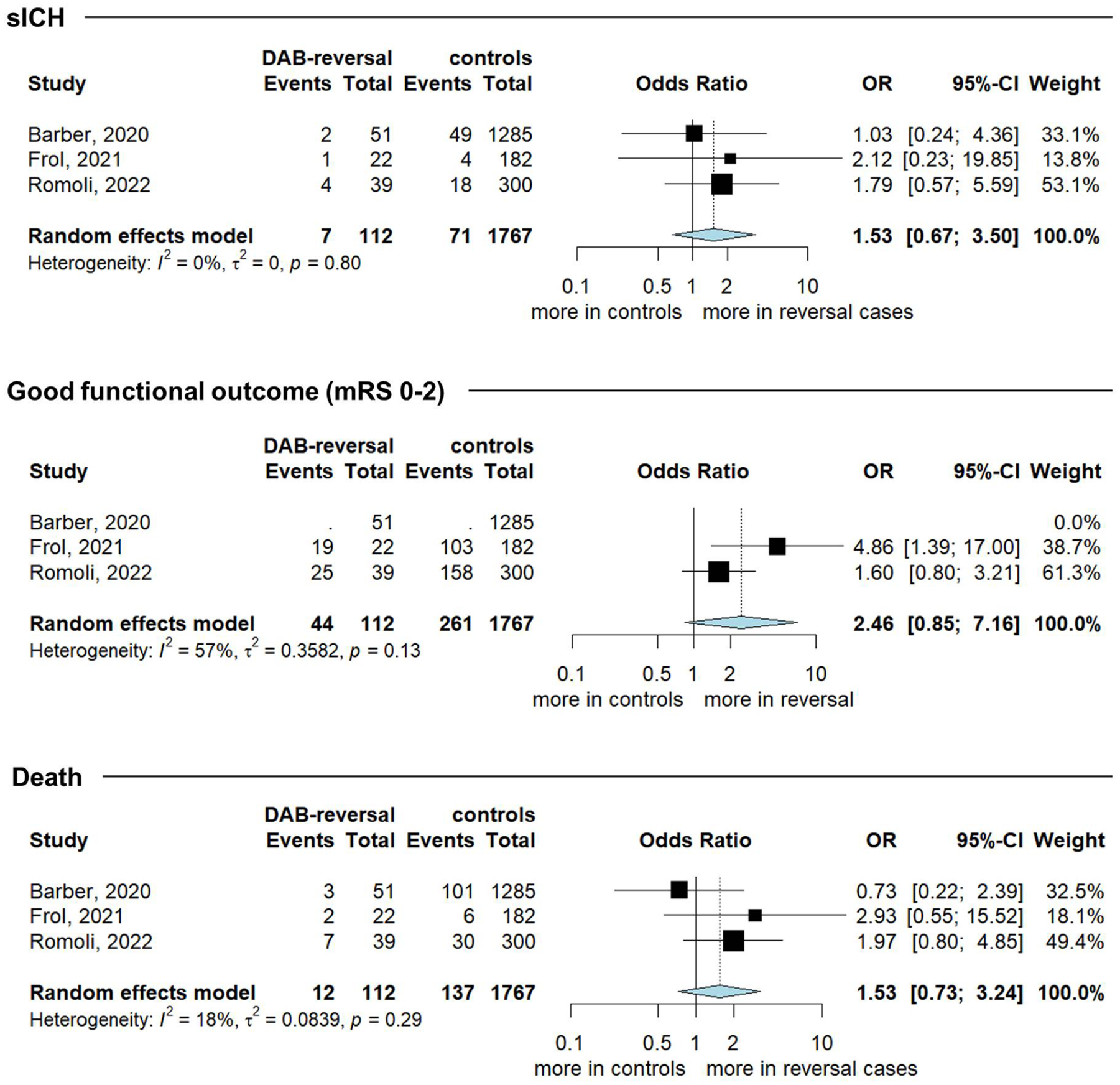
Pooled risk estimate for symptomatic intracranial hemorrhage (sICH), good functional outcome and death for dabigatran reversal versus control group.
Among case series retrieved, two were excluded for potential duplication, and eight for high risk of bias (Supplemental eTable 1), leaving four to be included in additional analysis,5,16–18 all of moderate quality in relation to potential selection bias (Supplemental eTable 2). Funnel plots and Egger’s test revealed no critical publication bias (Supplemental eFigure 3). Meta-analysis of proportions among people undergoing IVT after reversal (n = 223) revealed a 4% rate of sICH (95% CI = 2%–9%, pheterogeneity = 0.95), a 74% rate of good functional outcome (95% CI = 59%–85%, pheterogeneity = 0.18), and a 9% risk of death (95% CI = 5%–15%, pheterogeneity = 0.18) (Supplemental eFigure 4).
Discussion
DOAC intake in the 48 h preceding stroke represents a contraindication for IVT.6,7 In the current nation-wide study we highlight that reversal with idarucizumab may represent a feasible way to allow IVT in people with acute ischemic stroke while on dabigatran, with little impact on treatment timing. Onset-to-needle time was twelve minutes longer with reversal, similar to what was previously reported in a New Zealand-based study, 4 a delay that can reasonably be shortened with clear administration protocols and careful prehospital triage. Implementing this process may also increase the use of dabigatran-reversal in stroke, which seems rather uncommon as even nation-wide series may be limited in sample size.4,15
In our Italian study, despite not reaching clear statistical significance, people receiving reversal before IVT had marginally higher rates of sICH and hemorrhagic transformation than matched-controls. This to some extent replicates a slight increase in bleeding emerging from previous studies,4,15,16,18 but may derive from confounders, as emerges from regression-adjusted risk. In particular, compared to people treated with reversal, matched-controls had a substantially lower rate of anticoagulation at the time of stroke (<5%), had a lower prevalence of diabetes, displayed normal coagulation testing, and received treatment within a shorter timeframe, all factors known to critically limit the risk of sICH. 19 On the other hand, a biological basis for an increased risk of bleeding can be found in rebound anticoagulation. In particular, as idarucizumab concentration decreases over 12 h, there might be room for late increase in ecarin clotting time due to residual circulating dabigatran. 2 To this extent, the 4% rate of sICH from meta-analysis of proportions may be reassuring, as it seems superimposable to sICH rates reported in large studies including people without reversal. 19 However, given the trend also emerging from OR meta-analysis, such a non-significant 1.5-fold increase in sICH with a general increase in hemorrhagic transformation warrants further investigation.
Beyond sICH, reversal was also associated with a marginal trend in favor of good functional outcome, which may reinforce the concept that reversal with idarucizumab has no detrimental impact on long term functional recovery. Such trend already emerged in two studies included in meta-analysis and is worthy of attention, particularly regarding cost-effectiveness.4,15 The rate of good functional outcome and the uncommon presentation of stroke recurrence (one case only) argue against an increase in pro-thrombotic activity due to idarucizumab administration, 20 which once postulated prevented guidelines from endorsing such treatment strategy. 6 This nationwide study reported no venous thrombotic events or allergic reactions after using idarucizumab. This seems in line with clotting data reported in the RE-VERSE AD trial, 2 where idarucizumab administration restored hemostasis with no rebound in clotting parameters and with only one thrombotic event occurring within the first 72 h from the administration of the antidote. 2 Given the lack of rebound pro-coagulant effect,2,21 dabigatran reversal might be considered reasonably safe regarding thrombotic complications. Further collaborative studies will help in refining the risk/benefit profile of reversal.
The indications to reverse dabigatran may have to adapt to local circumstances and practice. When dabigatran concentration can be tested, it might be worth considering the cost-effectiveness of the reversal in the light of plasmatic concentration and clotting test results. In this study, only twelve patients received IVT without dabigatran-reversal, all strictly related to low plasmatic concentration and unknown intake (n = 1), or low plasmatic concentration and normal coagulation screening with known or remote intake (n = 11). Among those patients no hemorrhagic transformation or death was registered, with 75% reaching good functional outcome. The available sample prevents from drawing any conclusion on whether IVT can be safely administered below a definite dabigatran plasma concentration, but suggests that studies targeting this issue should implement additional cost-effective analysis to define the optimal management.
Some limitations should be taken into account to interpret these results. First, the main limitation of this study is the observational nature, which despite reaching nation-wide adherence, faces issues related to lack of funding, and relies on local collaborative effort. To this extent, we reached 17 centers throughout the nation through iterative campaigns by providing simple dataframe to ease data imputing, and maintaining direct correspondence with local investigators, to limit reporting bias. Second, the control-group was derived only from four centers providing reversal patients with a local ongoing stroke registry, and was matched for some of the most critical features regarding stroke, but was unbalanced for potential modifiers (e.g. diabetes). Nevertheless, the extent of matching and the rates of outcomes in the control group are similar to larger studies, 19 adding robustness to our findings. Third, larger studies are needed to confirm our estimates when taking into account other potential risk factors for bleeding left unmeasured in this study, such as renal function, use of statins, or fibrinogen depletion.6,22,23 Third, the meta-analysis involved only three studies, which, despite being nation-wide collection of cases, still carry an intrinsic risk of bias. The fact that the estimates for the outcomes of interest are well replicated in the meta-analysis of proportions seems to add robustness to our findings. It is paramount to carry on data collection and support international collaboration to clarify the optimal treatment for a subgroup of patients which could hardly be involved in randomized controlled trials.
Our study substantially increases the number of patients receiving IVT after reversal reported in previous case-control studies.4,15 The results from meta-analysis suggest that people with stroke while on dabigatran can find the same benefit on functional status from IVT after reversal compared to the general population, although being at marginally higher risk of bleeding. Further studies are needed to refine our findings and provide potential dabigatran plasma thresholds allowing for cost-effective and safe treatment of this patient subgroup.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221131635 – Supplemental material for Thrombolysis after dabigatran reversal: A nation-wide Italian multicentre study, systematic review and meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873221131635 for Thrombolysis after dabigatran reversal: A nation-wide Italian multicentre study, systematic review and meta-analysis by Michele Romoli, Eleonora Matteo, Ludovica Migliaccio, Mauro Gentile, Maria Giulia Mosconi, Giuseppe Maria Scura, Marcello Naccarato, Enrico Colangeli, Paolo Candelaresi, Vincenzo Andreone, Fabrizio Giammello, Rosa Fortunata Musolino, Cristina Dell’Aera, Federica Nicoletta Sepe, Edoardo Pronello, Leonardo Barbarini, Marcella Caggiula, Federica Rizzo, Marco Petruzzellis, Elisa Giorli, Maria Luisa Zedde, Sabrina Anticoli, Marilena Mangiardi, Mario Muto, Francesco Diana, Maria Vittoria De Angelis, Anna Digiovanni, Letizia Concari, Sara La Gioia, Maria Sessa, Sara Biguzzi, Francesco Cordici, Marco Longoni, Maria Ruggiero, Silvia Cenciarelli, Paolo Eusebi, Simona Sacco, Valeria Caso, Maurizio Paciaroni, Stefano Ricci, Andrea Zini, Danilo Toni and David Giannandrea in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873221131635 – Supplemental material for Thrombolysis after dabigatran reversal: A nation-wide Italian multicentre study, systematic review and meta-analysis
Supplemental material, sj-docx-2-eso-10.1177_23969873221131635 for Thrombolysis after dabigatran reversal: A nation-wide Italian multicentre study, systematic review and meta-analysis by Michele Romoli, Eleonora Matteo, Ludovica Migliaccio, Mauro Gentile, Maria Giulia Mosconi, Giuseppe Maria Scura, Marcello Naccarato, Enrico Colangeli, Paolo Candelaresi, Vincenzo Andreone, Fabrizio Giammello, Rosa Fortunata Musolino, Cristina Dell’Aera, Federica Nicoletta Sepe, Edoardo Pronello, Leonardo Barbarini, Marcella Caggiula, Federica Rizzo, Marco Petruzzellis, Elisa Giorli, Maria Luisa Zedde, Sabrina Anticoli, Marilena Mangiardi, Mario Muto, Francesco Diana, Maria Vittoria De Angelis, Anna Digiovanni, Letizia Concari, Sara La Gioia, Maria Sessa, Sara Biguzzi, Francesco Cordici, Marco Longoni, Maria Ruggiero, Silvia Cenciarelli, Paolo Eusebi, Simona Sacco, Valeria Caso, Maurizio Paciaroni, Stefano Ricci, Andrea Zini, Danilo Toni and David Giannandrea in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors have no conflict of interest related to this work. Relevant disclosures outside the submitted work: MP received honoraria from Sanofi-Aventis, Boehringer Ingelheim, Bayer, Bristol Myers Squibb, Daiichi Sankyo, and Pfizer. VC received honoraria from Boehringer Ingelheim, Bayer, and Daiichi Sankyo, and Pfizer. SS reports personal fees and nonfinancial support from Allergan, Abbott, Eli Lilly, Novartis, Teva, Bayer, Pfizer, Medtronic, Starmed, Bristol-Myers Squibb, and Daiichi Sankyo. SR reports nonfinancial support from Bayer. AZ declares consulting fees from Boehringer-Ingelheim, Alexion and CLS Behring, and declares grant from the Italian Ministry of Health as principal investigator for clinical trial (RF-2019-12370834). DT reports fees for advisory board and speaker honoraria from Abbott, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Medtronic, and Pfizer. All other authors have nothing to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and informed consent
This study, being part of the Safe Implementation of Thrombolysis in Stroke (SITS) study, was approved by the Institutional Review Board, with consent waived.
Guarantor
DG
Contributorship
MR and DG: study design, data collection, systematic review design, data extraction, manuscript drafting. MR and PE: statistical analysis. EM: systematic review design, data extraction, manuscript drafting. MP, SR, AZ, DT: study design supervision, revised manuscript for intellectual content. All other authors: data collection, revised manuscript for intellectual content.
List of collaborators available as Appendix.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
