Abstract
Background:
While thrombectomy benefits patients with large infarcts, it is unclear whether this benefit persists across different levels of reperfusion.
Aims:
This study investigates how the degree of reperfusion influences the effectiveness of endovascular thrombectomy (EVT) combined with best medical treatment (BMT), compared to BMT alone, in patients with large infarcts.
Methods:
This post hoc analysis of the TENSION trial, a randomized controlled study, assessed EVT versus BMT in patients with extensive infarction (Alberta Stroke Program Early CT Score (ASPECTS) 3–5). Primary outcome was the modified Rankin Scale (mRS) score at 90 days. Secondary outcomes included infarct volume at 24 h, mortality, and symptomatic hemorrhage. Outcomes were stratified by final reperfusion level, measured with the modified thrombolysis in cerebral infarction (mTICI) scale. Confounder-adjusted common odds ratios (cORs) and average treatment effects (ATEs) were estimated using inverse probability weighting with regression adjustment.
Results:
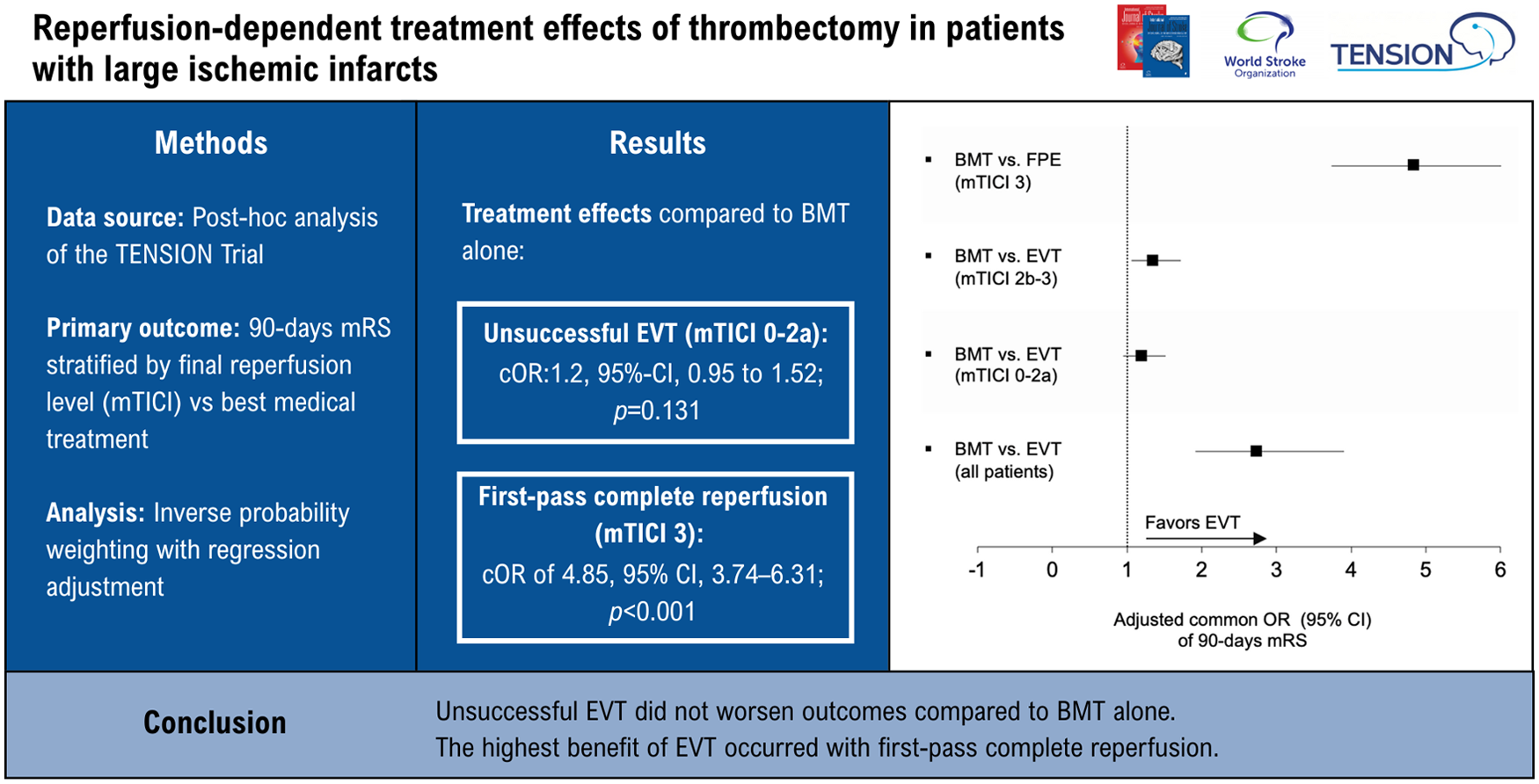
A total of 246 patients (median age, 74 years (interquartile range (IQR), 65–80); median baseline ASPECTS, 4 (IQR, 3–5)) were included. Compared to BMT alone, unsuccessful EVT (mTICI ⩽ 2a) was not associated with worse functional outcomes (cOR:1.2, 95% CI, 0.95 to 1.52; p = 0.131), higher mortality (ATE: –11.6%; 95% CI, –28.82 to 5.61; p = 0.187), or larger infarct volumes on follow-up (ATE:0.99 mL; 95% CI, –45.30 to 45.32; p = 0.965). First-pass complete reperfusion (mTICI 3) showed the greatest treatment benefit, significantly improving all endpoints, with a cOR of 4.85 (95% CI, 3.74–6.31; p < 0.001) for improved mRS scores and a 29% absolute reduction in mortality.
Conclusion:
In this post hoc analysis of the TENSION trial, unsuccessful EVT did not worsen outcomes compared to BMT alone. The highest benefit of EVT occurred with first-pass complete reperfusion, emphasizing the importance of achieving optimal reperfusion in this vulnerable stroke subgroup. These findings do not justify general treatment recommendations.
Keywords
Introduction
Evidence from randomized trials supporting efficacy and safety of endovascular thrombectomy (EVT) continues to grow, extending across multiple subgroups, including patients with large acute ischemic strokes. Treatment success of EVT is primarily attributed to reperfusion of ischemic brain due to the efficacy of mechanical thrombus removal, facilitated by advancements in modern catheter and device technology.1–5 Consequently, higher degrees of vessel reperfusion (i.e. successful EVT) and subsequent improved reperfusion of the affected hypoperfused arterial territory, are among the strongest procedure-related predictors of favorable long-term outcomes. 6 Conversely, unsuccessful EVT without substantial reperfusion restoration is strongly associated with poorer clinical outcomes. Recent data from randomized trials show that EVT remains unsuccessful in approximately 20 % of cases, defined by a modified thrombolysis in cerebral infarction (mTICI) scale scores of 2a or lower at the end of the procedure. 7
While results from the TENSION trial 2 confirmed that patients with large infarcts benefit from EVT, extensive ischemic damage may also increase tissue vulnerability to the mechanical stresses of the procedure, potentially increasing the risk of periprocedural adverse events such as vessel perforation, dissection, and intracerebral hemorrhage. Therefore, the subgroup of stroke patients with large ischemic lesions may be more susceptible to EVT-related adverse events, particularly, in cases of unsuccessful EVT with incomplete reperfusion. 8
We hypothesized that both treatment effects and adverse effects of EVT in the TENSION population depend on reperfusion degrees achieved, and that even lower degrees of reperfusion (i.e. unsuccessful EVT) do not result in worse outcomes compared to best medical treatment (BMT).
Methods
Study design and population
This study is a post hoc analysis of the TENSION (The Efficacy and Safety of Thrombectomy in Stroke with extended lesion and extended time window; NCT03094715) Trial. 2 The TENSION Trial was an investigator-initiated, prospective, randomized, open-label, blinded endpoint (PROBE) trial conducted in Europe and Canada designed to compare treatment effects of BMT only (control group) to EVT plus BMT (intervention group) in patients with large ischemic strokes. The TENSION trial protocol and main results have been published previously.2,9 Reporting followed the CONSORT guideline.
Clinical assessment
All patients underwent clinical assessments at baseline, at 24 (±6) h, at 7 days or at hospital discharge, and at 90 days (±14). Clinical assessment at 90 days was performed in-person or via telephone interview by trained personnel who were unaware of the treatment-group assignment.
Image assessment
Imaging parameters were assessed by a central independent core laboratory (Eppdata, Hamburg, Germany). Post-treatment flow restoration following EVT was evaluated using the mTICI scale, ranging from “no” to “complete reperfusion” of the relatively hypoperfused territory distal to the occlusion site. Successful EVT was defined as mTICI 2b-3 on the final angiogram and unsuccessful EVT as no to partial reperfusion mTICI 0 to 2a. The first-pass effect was reported for mTICI 3 after the first EVT maneuver. Follow-up infarct volume was measured by independent neuroradiologists on follow-up imaging (computed tomography (CT) or magnetic resonance imaging (MRI), according to the standard of care) performed at 24 ± 8 h using semiautomatic segmentation algorithms (ITK-SNAP 3.8.0 (17) Bleeding events were considered symptomatic (sICH) in accordance with the Second European-Australasian Acute Stroke Study (ECASS II) with a clinical worsening of at least 4 points on the National Institutes of Health Stroke Scale (NIHSS) scale most likely related to hemorrhage.
Outcome measures
Primary outcome was the functional neurological disability across the entire range of modified Rankin Scale (mRS) scores at 90 days (±14) between treatment groups in ordinal shift analysis.
Secondary outcome was independent ambulation defined as an mRS of 0 to 3 at 90 days follow-up. Imaging outcome was defined as 24 h follow-up infarct volume. Safety was evaluated by in-hospital and 90-day mortality rates, incidence of sICH, and malignant middle cerebral artery (MCA) infarction, defined as >½ MCA-territory infarct on follow-up CT with herniation requiring decompressive hemicraniectomy or causing death from edematous mass effect.
Statistical analysis
This post hoc analysis focuses on a modified as-treated population, excluding all patients in which the primary functional endpoint was not available (Supplemental Figure S1).
Standard descriptive statistics were applied for all endpoints. Univariable distribution of metric variables was described with median and interquartile range (IQR) and categorical variables as absolute and relative frequencies.
All outcome measures were analyzed based on different degrees of reperfusion achieved through EVT and compared to control. To account for the loss of randomization and subgroup heterogeneity, treatment effects associated with varying levels of reperfusion were estimated using double-robust inverse probability weighting with regression adjustment (IPWRA), adjusting for key confounding variables, with BMT as the reference comparator.
IPWRA-based adjusted common odds ratios (cORs) and average treatment effect estimates (ATEs) were adjusted for clinically relevant confounders known to influence post-interventional outcomes and safety. These included age, sex, pre-stroke mRS status (0–2), admission NIHSS scores, baseline signs of ischemic infarction assessed via ASPECTS, administration (yes/no) of intravenous thrombolysis (IVT), and occlusion location (internal carotid artery or M1/M2 segments of the MCA). Adjusted cORs and ATEs, along with 95% confidence intervals (CIs), were calculated for selected outcome variables.
For the primary outcome, the mRS score at 90 days, adjusted cORs were reported for ordinal outcome measures. Adjusted cORs greater than 1 indicated a shift in the distribution of 90-day mRS scores toward better functional outcomes (i.e. lower mRS scores) compared to control.
For the secondary binary outcome of favorable functional status (mRS 0–3 at 90 days), ATEs were expressed as the mean increase in the probability of achieving mRS 0–3, presented in percentage points, compared to control.
ATEs for the imaging outcome, follow-up infarct volume, were reported as the mean reduction in infarct volume (measured in milliliters). For safety outcomes, ATEs were reported as the mean change in the probability of mortality and symptomatic intracranial hemorrhage (sICH), also expressed in percentage points, compared to control.
No adjustment for multiple testing was performed, and the analyses were regarded as explorative. A two-tailed p-value <0.05 was considered significant for all statistical analyses. Statistical analyses were performed with Stata/MP (version 18.0; StataCorp LLC) and R statistical software (version 4.3.2; R Project for Statistical Computing).
Results
Study population
A total of 246 patients (EVT, n = 126; BMT, n = 120) from the TENSION trial met the inclusion criteria and were analyzed (Supplemental Figure S1). The median age was 74 years (IQR, 65–80), 48.8% (120) were female, and the median baseline NIHSS score was 18 (IQR, 15–22). Most patients had arterial hypertension (80.6%, 191) and atrial fibrillation (33.5%, 77). The majority of occlusions were located in the M1 segment of the MCA (61.8%, 152), with a median ASPECTS of 4 (IQR, 3–5). IVT was administered in 36.2% (89) of patients.
Among EVT patients, successful reperfusion (mTICI 2b-3) was achieved in 83.3% (105), including 29.4% (37) with first-pass effect (mTICI 3 after first pass). Unsuccessful reperfusion (mTICI 0-2a) occurred in 16.7% (21; Supplemental Figure S2). Table 1 summarizes baseline characteristics by treatment and reperfusion success.
Baseline characteristics stratified by reperfusion treatment levels in comparison with best medical treatment.
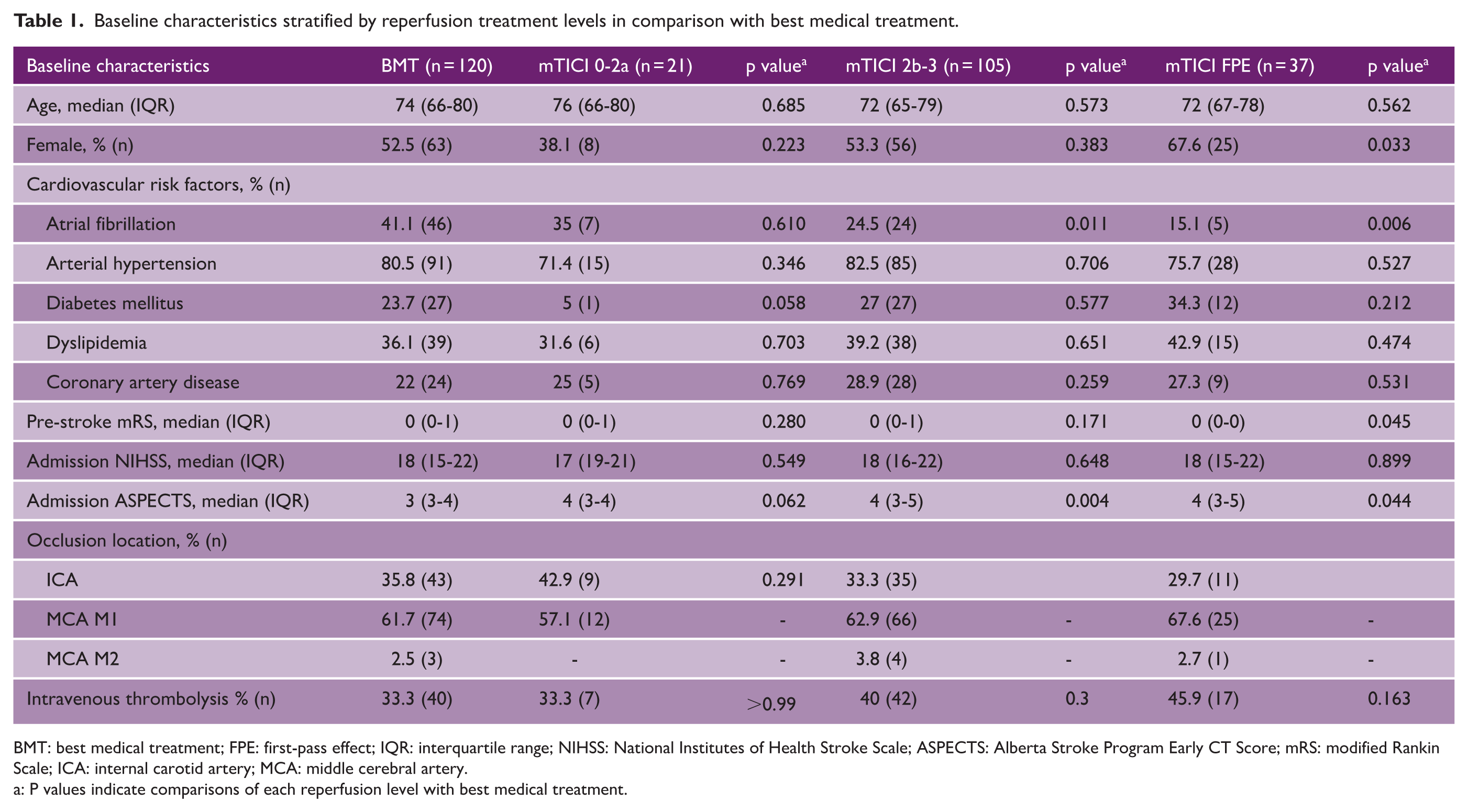
BMT: best medical treatment; FPE: first-pass effect; IQR: interquartile range; NIHSS: National Institutes of Health Stroke Scale; ASPECTS: Alberta Stroke Program Early CT Score; mRS: modified Rankin Scale; ICA: internal carotid artery; MCA: middle cerebral artery.
a: P values indicate comparisons of each reperfusion level with best medical treatment.
Primary and secondary outcomes
In univariable comparison, median functional outcomes as defined by the mRS at 90 days, were significantly better in the EVT cohort compared to the control group (EVT: 4 (IQR, 3–6), vs control: 4 (IQR, 4–6); p < 0.001; Figure 2). In addition, IPWRA estimates of adjusted cOR for improvement of mRS scores at 90 days favored EVT over control with a cOR of 2.74 (95 % CI, 1.92 to 3.90; p < 0.001). The probability for independent ambulation mRS 0–3 favored also intervention (EVT: 30.9 %, 39 vs control: 13.3 %, 16; p < 0.001, Figure 1 and Table 2).
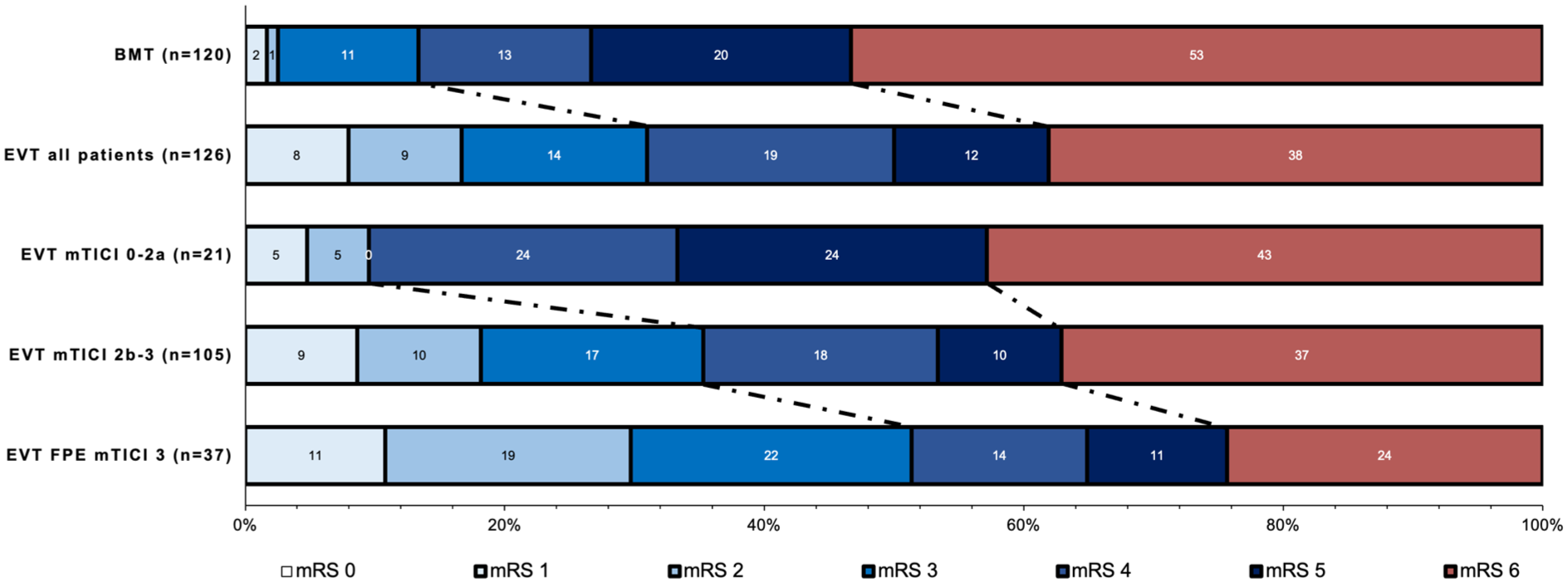
Distributions of modified Rankin Scale scores at 90 days stratified by treatment modality and reperfusion degree post-endovascular thrombectomy.
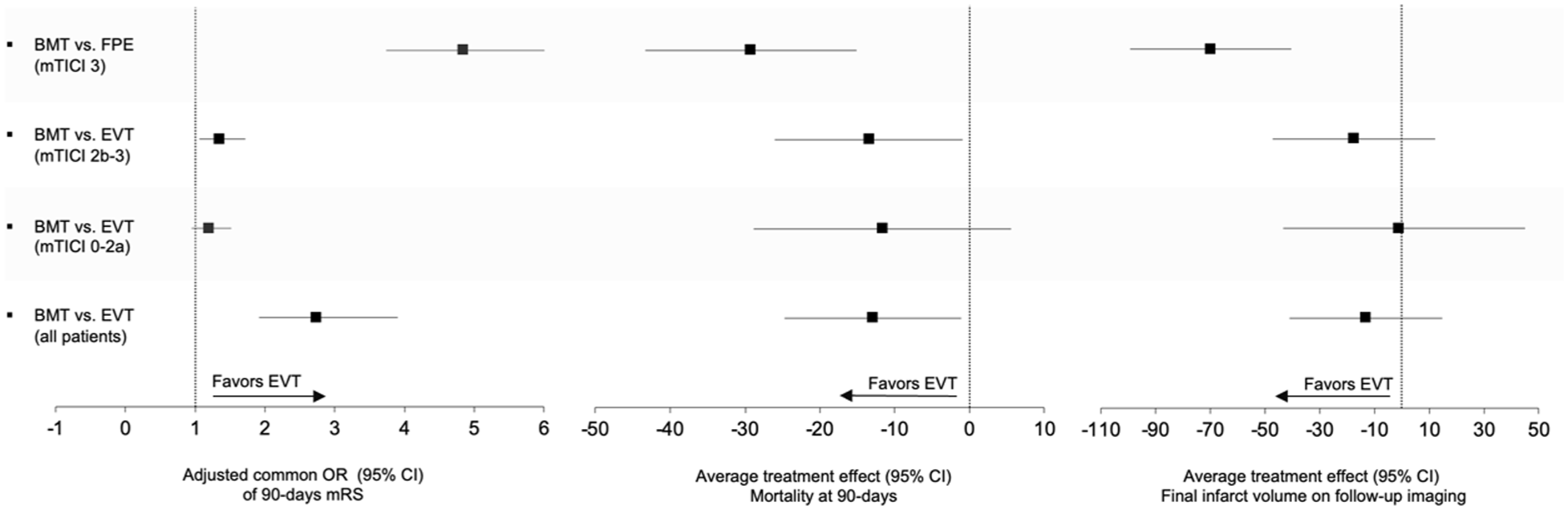
Adjusted common odds ratios and average treatment effects in the study population of functional outcome, mortality, and final infarct volume stratified by treatment modality and reperfusion degree post-endovascular thrombectomy.
Unadjusted and adjusted outcomes stratified by treatment modality and reperfusion degree post-thrombectomy.
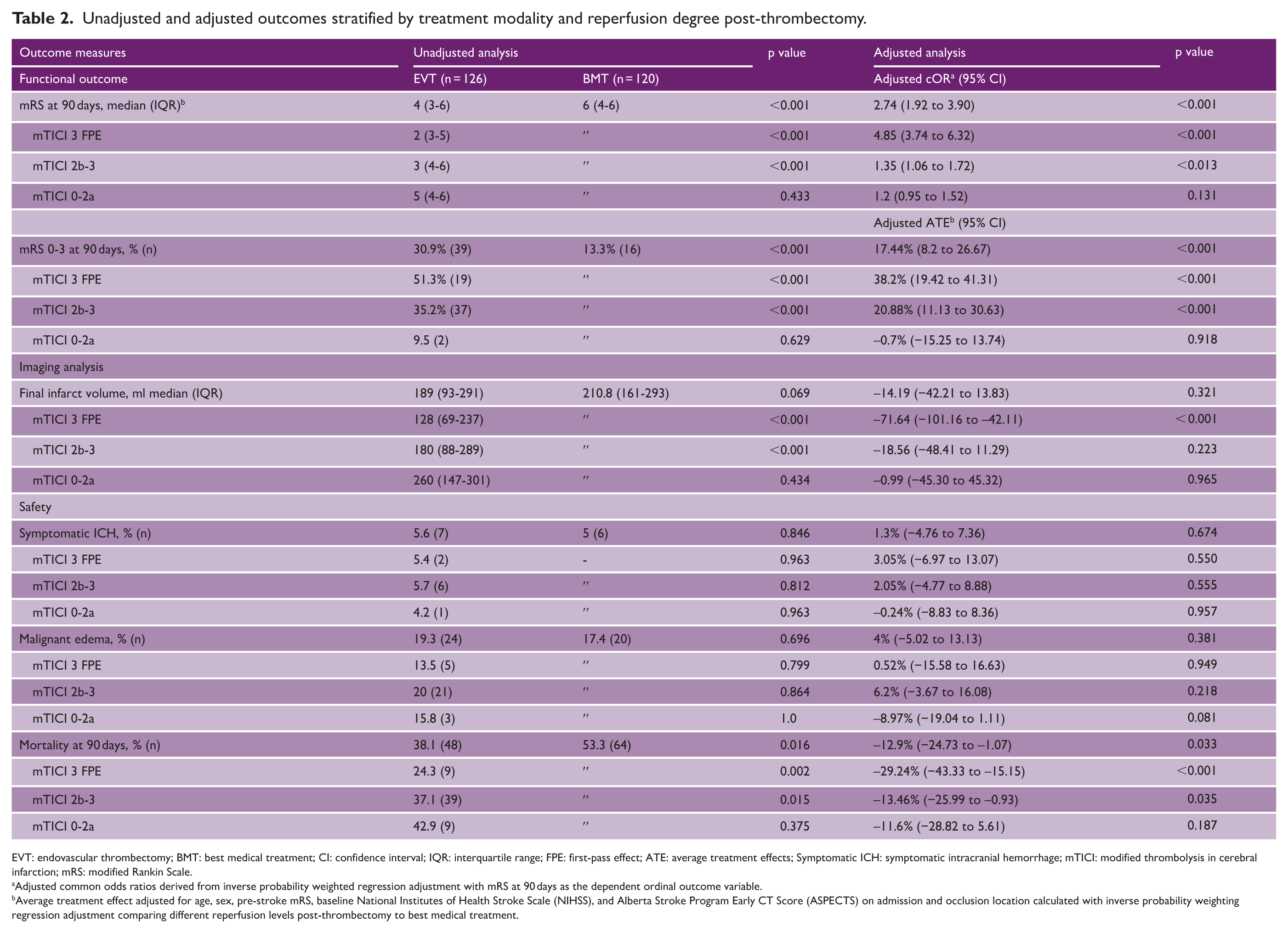
EVT: endovascular thrombectomy; BMT: best medical treatment; CI: confidence interval; IQR: interquartile range; FPE: first-pass effect; ATE: average treatment effects; Symptomatic ICH: symptomatic intracranial hemorrhage; mTICI: modified thrombolysis in cerebral infarction; mRS: modified Rankin Scale.
Adjusted common odds ratios derived from inverse probability weighted regression adjustment with mRS at 90 days as the dependent ordinal outcome variable.
Average treatment effect adjusted for age, sex, pre-stroke mRS, baseline National Institutes of Health Stroke Scale (NIHSS), and Alberta Stroke Program Early CT Score (ASPECTS) on admission and occlusion location calculated with inverse probability weighting regression adjustment comparing different reperfusion levels post-thrombectomy to best medical treatment.
When stratified by reperfusion success, successful reperfusion (mTICI 2b-3) was associated with a cOR of 1.35 for a shift in the distribution of mRS scores at 90 days (95% CI, 1.06 to 1.72; p < 0.013; Figure 2 and Table 2). Highest treatment effects were observed in patients with a first-pass effect (cOR: 4.85, 95 % CI, 3.74 to 6.32; p < 0.001). Unsuccessful EVT was not associated with worse outcomes compared to control (cOR, 1.2, 95% CI, 0.95 to 1.52; p = 0.131).
Safety analysis
Mortality rates were significantly lower in the EVT compared to control (EVT: 38.1%, 48, vs control: 53.3%, 64; p = 0.016). This effect was significant across the subgroups of successful reperfusion (ATE: –13.46 percentage points probability of mortality, 95 CI, –25.99 to –0.93; p = 0.035) and first-pass effect (ATE: –29.24%, 95 CI, –43.33 to –15.15; p < 0.001). For the EVT subgroup with unsuccessful reperfusion (mTICI 0-2a), no significant difference in mortality rates compared to control were observed. sICH occurred in seven patients (5.6%) of the EVT cohort and in six patients (5%) of the control (p = 0.846) without a significant difference of ATE (1.3%, 95 CI, –4.76 to 7.36; p = 0.674; Figure 2 and Table 2). Malignant edema occurred in 19.3% (24) of EVT patients versus 17.4% (20) of controls (p = 0.696; ATE: 4.0%, 95% CI, –5.02 to 13.13; p = 0.381), with no significant differences across subgroups (Table 2).
Imaging analysis
Follow-up infarct volume in the EVT cohort compared to control group was numerically smaller; however, the difference was not statistically significant (189 mL vs 210.8 mL, p = 0.069; ATE –14.19 ml, 95% CI, –42.21 to 13.83; p = 0.321). For the subgroup of patients with first-pass effect, significantly smaller follow-up infarct volumes were observed (128 mL vs 210.8 mL, p < 0.001; ATE –71.64 mL, 95 CI, –101.16 to –42.11; p < 0.001; Figure 2 and Table 2).
Discussion
This post hoc analysis of the TENSION trial revealed the following main findings for patients with large ischemic lesions at admission: (1) unsuccessful EVT did not result in worse outcomes or increased mortality compared to BMT alone; (2) The EVT-related improvement in functional outcome gradually increased in conjunction with higher degrees of reperfusion; (3) The first-pass effect, defined as mTICI 3 after one pass, led to the greatest improvement in functional outcomes; (4) substantial reductions of final infarct volumes compared to BMT were only observed in patients with a first-pass effect.
The impact of incomplete and unsuccessful EVT on functional outcomes and safety remains unclear, particularly in patients with established large infarcts. For patients with mild to moderate sings of early infarction, data from the HERMES trial showed that unsuccessful EVT in LVO stroke patients did not yield in better outcomes compared to control. 10 In addition, unsuccessful EVT is generally associated with a higher risk of complications. These cases often involve complex occlusions requiring multiple retrieval attempts that can lead to subsequent vascular damage. Furthermore, it is often necessary to perform EVT under general anesthesia during these procedures that may introduce additional risks, including hypotension, which can exacerbate ischemic damage.11,12 In patients with extensive signs of infarction, the fragility of large ischemic tissue lesions may further increase the risk of procedural complications, potentially leading to worse functional outcomes in this subgroup. 8 This raises the critical question of whether unsuccessful EVT in patients with extensive infarction might not only fail to improve outcomes but could potentially result in worse outcomes compared to control.
Our results did not reveal evidence of potential harmful treatment effects of unsuccessful EVT in patients with extensive sings of infarction at baseline. In addition, functional outcome, mortality and final infarct volumes did not differ significantly between patients with unsuccessful EVT and BMT cohort alone. This finding highlights the safety of EVT and indicates that the procedure itself should not be considered a risk factor when evaluating whether to treat patients with low ASPECTS endovascularly. Our results align with previous studies, including an analysis of data from the ESCAPE/NA-1 trial that showed similar results outside the low ASPECTS cohort.13,14
The analysis of treatment effects of EVT compared to control, stratified by reperfusion degree revealed a significant impact of reperfusion success on all study endpoints. Higher reperfusion degrees resulted in significant improvement in functional outcomes, favoring EVT over control. These gradually increasing effects underline the substantial benefit of achieving high reperfusion degrees, even in this subgroup with generally very poor outcome prognosis. 15 In addition, successful reperfusion (mTICI 2b-3) was associated with a significant reduction in 90-day mortality, lowering the risk by approximately 16% compared to control. Importantly, this was achieved without an associated increase of the socioeconomically most unfavorable functional post-stroke status of mRS 5. 16 Overall, the greatest effect sizes across all endpoints were observed when EVT was successful after the first pass (i.e. first-pass effect). This procedural surrogate is known to be a favorable predictor for outcomes in ischemic stroke patients receiving endovascular treatment. 17 In this subgroup of patients, the first-pass effect increased the probability of independent ambulation (mRS 0-3) by 38 percentage points, while mortality decreased by 29 percentage points compared to control. Therefore, the theory of maximizing efforts using most advanced techniques and device technology for achieving mTICI 3 at the first EVT maneuver instead of achieving partial reperfusion as fast as possible seems to be the highest benchmark in the subgroup of low ASPECTS.6,18
Interestingly, although a trend toward gradually increasing effects in terms of infarct volume reduction was observed, a statistically significant effect on follow-up infarct volume was noted exclusively in the subgroup with first-pass effect, consistent with previous findings. 19 However, these observed effects remain underpowered and should be interpreted with caution. This observation resumes the debate on the discrepancy between follow-up infarct volume and clinical outcome.20,21 The pathophysiology of large ischemic strokes, particularly in the context of acute reperfusion therapies, urgently requires further understanding.
Semi-quantitative CT-based imaging assessments may be reaching their limitations in distinguishing pan-necrosis versus incomplete infarction at the cellular level. Recent initiated discussions challenging the conventional ischemic core concept, including phenomena like selective neuronal loss and variability in tissue vulnerability, underline the complexity of these processes and highlight the need for further research.22–24
Limitation
This study has all the limitations inherent in a post hoc analysis of randomized trial data. In particular, the analysis may be underpowered, as the study was not designed to compare reperfusion-dependent treatment effects. Importantly, the degree of reperfusion (study groups) was not randomized; although IPWRA was applied, residual confounding cannot be excluded, and causal inference should therefore be made with caution. Additionally, angiographic imaging is generally not included in follow-up imaging protocols; therefore, reperfusion rates and effects are unavailable in the control. In addition, there is currently a lack of a reliable gold standard for determining final infarct size on 24-h follow-up imaging.
Conclusion
This post hoc analysis of the TENSION trial showed that unsuccessful EVT was not associated with worse outcomes or increased mortality compared to control in patients presenting with large signs of infarction. Consequently, EVT should therefore not be considered an independent risk factor in treatment decision-making. The highest benefit of EVT compared to control alone was observed when complete reperfusion was achieved at the first pass. This finding highlights the importance of prioritizing strategies and techniques aimed at achieving first-pass success as the benchmark for optimizing outcomes in this vulnerable stroke subgroup. However, because reperfusion groups were not randomized and residual confounding cannot be excluded, these results should be interpreted with caution and regarded as exploratory.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930251387613 – Supplemental material for Reperfusion-dependent treatment effects of thrombectomy in patients with large ischemic infarcts
Supplemental material, sj-pdf-1-wso-10.1177_17474930251387613 for Reperfusion-dependent treatment effects of thrombectomy in patients with large ischemic infarcts by Lukas Meyer, Susanne Gellißen, Götz Thomalla, Martin Bendszus, Gabriel Broocks, Matthias Bechstein, Christian Thaler, Fabien Subtil, Susanne Bonekamp, Anne H Aamodt, Blanca Fuentes, Elke R Gizewski, Michael D Hill, Antonin Krajina, Laurent Pierot, Claus Z Simonsen, Kamil Zelenˇák, Rolf A Blauenfeldt, Bastian Cheng, Angélique Denis, Hannes Deutschmann, Franziska Dorn, Fabian Flottmann, Johannes C Gerber, Mayank Goyal, Jozef Haring, Christian Herweh, Silke Hopf-Jensen, Vi T Hua, Märit Jensen, Andreas Kastrup, Christiane F Keil, Andrej Klepanec, Egon Kurcˇa, Ronni Mikkelsen, Markus Möhlenbruch, Stefan Müller-Hülsbeck, Nico Münnich, Paolo Pagano, Panagiotis Papanagiotou, Gabor C Petzold, Mirko Pham, Volker Puetz, Jan Raupach, Gernot Reimann, Peter A Ringleb, Maximilian Schell, Eckhard Schlemm, Silvia Schönenberger, Bjørn Tennøe, Christian Ulfert, Katerˇina Vališ, Eva Vítková, Dominik F Vollherbst, Wolfgang Wick, Jens Fiehler and Helge Kniep in International Journal of Stroke
Footnotes
Acknowledgements
We thank the patients and their families for participating in the trial, the members of the data and safety monitoring board, the European Society of Minimally Invasive Neurological Therapy, the International Consortium for Health Outcomes Measurement, and the Stroke Alliance for Europe for their collaboration. Participating centers and site investigators of the TENSION trial are listed in ![]() .
.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H Kniep reports compensation as speaker from Asklepios Kliniken, an ownership stake in Eppdata GmbH and compensation from Eppdata GmbH for consultant services. L Meyer reports compensation from Eppdata GmbH for consultant services. G Thomalla reports funding from the European Commission (EUHorizon 2020 research and innovation program, 754640; payments to the institution); personal consulting fees from Acandis, AstraZeneca, Bayer, Boehringer Ingelheim, and Stryker; personal payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Acandis, Alexion, Marin, Bayer, Boehringer Ingelheim, BristolMyersSquibb/Pfizer, Daiichi Sankyo, and Stryker; participation as DSMB member for the TEA Stroke Trial (no payments) and ReSCInD trial (no payments); work as a speaker of the Commission for Cerebrovascular Diseases of the German Society of Neurology (DGN; no payments); and membership of the Board of Directors of the European Stroke Organisation (ESO; no payments). M Jensen reports grants from the German Research Foundation and by the European Union (Horizon Europe); personal fees from Bristol Myers Squibb. G Broocks reports compensation for consultant services from Eppdata GmbH and Acandis, and a travel grant from the American Society of Neuroradiology. M Bendszus reports funding from EU Horizon 2020 and Deutsche Forschungsgemeinschaft (payments to the institution); honoraria for lectures from Novartis, Boehringer Ingelheim, and Seagen; and consulting fees from NeuroScios and Boehringer Ingelheim and is an editor in chief of Clinical Neuroradiology (Springer). S Bonekamp reports funding from the EU Horizon 2020 research and innovation program (754640; payments made to the institution) and EU HORIZON Research and Innovation Action 2021 (Project 101057263; payments made to the institution) AH Aamodt reports unrestricted research grants from Boehringer Ingelheim; honoraria for lectures from BMS/ Pfizer, Teva, Roche, Abbvie, Lundbeck, and Novartis; and participation in Advisory Boards for MSD, BMS/Pfizer, Lundbeck, Lilly, and Abbvie. B Fuentes reports research grants from Carlos III Institute of Health; personal payment for educational lectures from Servicio Madrileño de Salud; payment for lectures from Euromedice to the institution; personal payment for educational lectures from Takeda; support for attending meetings from Daiichi Sankyo; receipt of materials for research from Abbot. MD Hill reports funding from Nil; grants to the University of Calgary for the TEMPO-2 trial from Boehringer Ingelheim, Biogen, NoNO (ESCAPE-NA1 trial and ESCAPE-NEXT trial), Canadian Institute for Health Research (ESCAPE-NA1 trial and ESCAPE-NEXT trial), Medtronic (HERMES collaboration), Alberta Innovates (QuICR Alberta Stroke Program); that some of the funds were used for the ESCAPE-NA1 trial from Alberta Innovates; consulting fees from Sun Pharma Brainsgate (paid work for adjudication of clinical trial outcomes); US patents 62/086,077 (licensed to Circle NVI) and 10,916,346 (licensed to Circle NVI); private stock ownership from Circle and PUreWeb; participation as data and safety monitoring committee chair of the RACECAT trial (end 2020), the Oncovir Hiltonel trial (ongoing), and the DUMAS trial (ongoing); participation as a data and safety monitoring committee member of the ARTESIA trial (ongoing), and the BRAIN-AF trial (ongoing); and is president of the Canadian Neurological Sciences Federation (not for profit) and a Board member of the Canadian Stroke Consortium (not for profit). A Krajina reports grants from the European Commission for the TENSION study (payment to the institution). L Pierot reports consulting fees from Balt, Microvention, and Phenox; and support for attending meetings or travel for the TENSION investigator meeting (transport and accommodation was reimbursed by the organization). CZ Simonsen reports grants from Novo Nordisk Foundation and Health Research Foundation of Central Denmark Region. RA Blauenfeldt reports speaker fees from Novo Nordisk, Pfizer and Beyer and grants from Novo Nordisk Foundation and Lundbeck Foundation. H Deutschmann reports financial compensation for the start-up fee and the obligatory payment to the hospital administration paid by the sponsor (Medical University Heidelberg) to the clinical division (no personal payments); personal consulting fees from Stryker; speakers honorary from Medtronic; support for attending meetings or travel from Medtronic; and past presidency of the Austrian Society of Interventional Radiology and past presidency Austrian Society of Neuroradiology. F Dorn reports consulting fees from Microvention and Balt, payment for expert testimony from Cerenovus and Microvention; payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Cerenovus, Stryker, Acandis, Asahi, Medtronic, Q’Apel, Tonbridge, Microvention, Inspire and Penumbra; participation in a Data Safety Monitoring Board or Advisory Board at Cerenovus and Microvention; work as an associate editor for Clinical Neuroradiology and Journal of Clinical Medicine and previously for Journal of NeuroInterventional Surgery. C Herweh reports consulting fees from Brainomix and lecture fees from Stryker, Microvention Terumo and Medtronic. S Hopf-Jensen reports funding for data collection, payment, or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Terumo. M Goyal reports research grants from Medtronic and Cerenovus (payments to the University of Calgary); royalties or licenses from Microvention (systems of intracranial access); personal consulting fees from Microvention, Medtronic, Stryker, Mentice, Philips, and Penumbra; and stock or stock options from Circle Neurovascular. CFK was chair of the German stroke registry (unpaid). R Mikkelsen reports payments for a stroke lecture from TMC Academy. M Möhlenbruch reports grants from Balt, Medtronic, MicroVention, and Stryker; consulting fees from Siemens; and support for attending meetings or travel from Europa Group. S Müller-Hülsbeck reports consulting fees from Terumo and Boston Scientific Corporation; payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Terumo and Boston Scientific Corporation. N Münnich reports the provision of study materials. P Pagano reports support for attending meetings or travel for the TENSION investigator meeting (transport and accommodation was reimbursed by the organization). M Pham reports grants from the German Research Foundation (DFG SFB 1158 A10, DFG KFO 5001 P02, DFG KFO 5001 Z, and DFG SFB TR 240 B02); speaker honoraria unrelated from Merck Serono and Bayer; and support for attending meetings or travel from Merck Serono (travel reimbursement) and Bayer (travel reimbursement). V Puetz reports lecturer’s fees from Boehringer Ingelheim, Daichii Sankyo, AstraZeneca and BMS; travel support from NoNo, Inc. PA Ringleb reports consulting fees to the institution from Boehringer Ingelheim and Bayer; and payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Boehringer Ingelheim, Bayer, Pfizer, and BMS (all made to the institution). E Schlemm reports grants from Hamburg Innovation and Hertie Foundation. DV Vollherbst reports research grants from MicroVention; consulting fees from Medtronic; and paid lectures from Cerenovus and Johnson & Johnson. W Wick reports consulting fees to the institution from Abbvie, BMS, GSK, and Servier. J Fiehler reports funding from the European Commission; personal consulting fees from Acandis, Cerenovus, Medtronic, Microvention, Phenox, Stryker, and Roche; consulting at Philips (no payments); payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Penumbra and Tonbridge; support for attending meetings or travel from Medtronic and Penumbra; stock or stock options from Tegus Medical, Eppdata, and Vastrax; and participation in a Data Safety Monitoring Board or Advisory Board at Phenox (personal fees) and Stryker (personal fees) and is a past president of ESMINT. F Flottmann reports consulting fees from Eppdata and support for attending meetings or travel from Microvention, Medtronic, Cerebrovascular Research and Education Foundation (CREF), and Acandis. All other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project received funding from the European Union’s Horizon 2020 Research and Innovation Programme under grant 754640.
ORCID iDs
Data sharing statement
Data that underlie the results reported in this article, after de-identification, will be made available on request beginning 12 months and ending 36 months following publication of the trial to investigators whose proposed use of the data has been approved by the steering committee. 1
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
