Abstract
Introduction:
Acute ischemic stroke remains the major cause of death and disability and conclusive evidence of Tenecteplase in treating stroke is lacking.
Objective:
To conduct a meta-analysis to determine whether Tenecteplase produces better outcomes than Alteplase and a network meta-analysis comparing the different dosing regimens of Tenecteplase.
Methods:
Searches were made in MEDLINE, CENTRAL, and ClinicalTrials.gov. The outcome measures are recanalization, early neurological improvement, functional outcomes at 90 days (modified Rankin Scale 0–1 and 0–2), intracranial hemorrhage, symptomatic intracranial hemorrhage, and mortality within 90 days from treatment.
Results:
Fourteen studies are included in the meta-analyses and 18 studies in the network meta-analyses. In the meta-analysis, Tenecteplase 0.25 mg/kg has significant results in early neurological improvement (OR = 2.35, and 95% CI = 1.16–4.72) and excellent functional outcome (OR = 1.20, and 95% CI = 1.02–1.42). In the network meta-analysis, Tenecteplase 0.25 mg/kg produces significant results in early neurological improvement (OR = 1.52 [95% CI = 1.13–2.05],
Conclusion:
While not conclusive, our study lends evidence to 0.25 mg/kg Tenecteplase dose for ischemic stroke treatment. Further randomized trials need to be done to validate this finding.
Registration:
International prospective register of systematic reviews (PROSPERO) – CRD42022339774
URL: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=339774
Introduction
Acute ischemic stroke (AIS) remains the major cause of death and disability worldwide.1,2 While Alteplase (rt-PA) shows improved outcomes in AIS, it is associated with hemorrhage and recanalization rates are low, especially in severe strokes.3 –7
Tenecteplase (TNK) is a genetically modified tissue plasminogen activator and has been investigated in AIS. Till date, 11 randomized controlled trials (RCTs)8 –18 have been performed to study the efficacy and safety of TNK. Although several trials achieved significant results for their outcomes of interest,8,9,12,13,15,16,18 other efficacy and safety outcomes were underpowered due to the limited sample sizes.8,10,19
Several meta-analyses20 –24 have been performed to address this shortcoming, however some grouped all TNK patients into a single cohort for analysis, regardless of the different dose regimes. While other meta-analyses have looked into different dosing regimens,20,22,23 the limitations of the component trials included restricted the analysis that could be performed and the sample sizes of the different pooled doses had difficulty reaching significance.
To overcome these shortcomings, we have performed a new network meta-analysis (NMA) that included the latest trials with sub-group analysis on the different dosing regimens of TNK. We aim to give an updated and better powered analysis on TNK.
Methods
The authors declare that all supporting data are available within the article and Supplemental Files. Present meta-analyses and NMAs follow the 2020 guidelines of Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) 25 and has been registered in International Prospective Register of Systematic Reviews (PROSPERO). Registration number: CRD42022339774.
Data search
A search was made in Medline, Cochrane Central Register of Controlled Trials (CENTRAL) and ClinicalTrials.gov up to 12th July 2022. The search words used included the following and were combined with appropriate Boolean operator: “tenecteplase,” “TNK,” “stroke,” “cerebral infarction,” and “ischaemia/ischemia,” and excluded results on “Myocardial infarction,” or “Myocardial ischaemia/ischemia.” Moreover, a further search for relevant studies was made from references of published reviews and meta-analyses. Two investigators (EZT and LLLY) did full text reading for data extraction separately and resolved any disagreements by discussion. Summary of methods involved is shown in Supplemental Table S1.
Inclusion and exclusion criteria
Inclusion criteria are (1) individuals with ischemic stroke, (2) rt-PA and/or TNK given intravenously, (3) within explicitly defined time periods, and (4) dosage used is explicitly defined as per kilogram body weight.
Studies are excluded if (1) Comparison between treatment with rt-PA and TNK or between different dosages of TNK is not possible or (2) There is no specified dosage of either treatment or separation between different dosages of TNK is not possible or (3) It does not report outcome measures specified in this meta-analysis or NMA or (4) Contains overlapping cases or (5) Treatment given time is not explicitly defined or (6) A study has less than five cases (Supplemental Table S1).
Outcome measures
Our outcomes studied includes (1) Recanalization of occluded vessel, (2) Early neurological improvement, (3) Intracranial hemorrhage (ICH), (4) Symptomatic intracranial hemorrhage (SICH), (5) Excellent functional outcome – modified Rankin Scale (mRS) 0–1 – at day 90 from treatment given, (6) Good functional outcome (mRS 0–2) at day 90 from treatment given, (7) Mortality within 90 days from treatment given. The definition used are as defined in the primary studies (Table 1). We have also stated the preferred definitions when there was some heterogeneity in the definition for the outcome measures between the different studies.
Definitions of outcome measures.
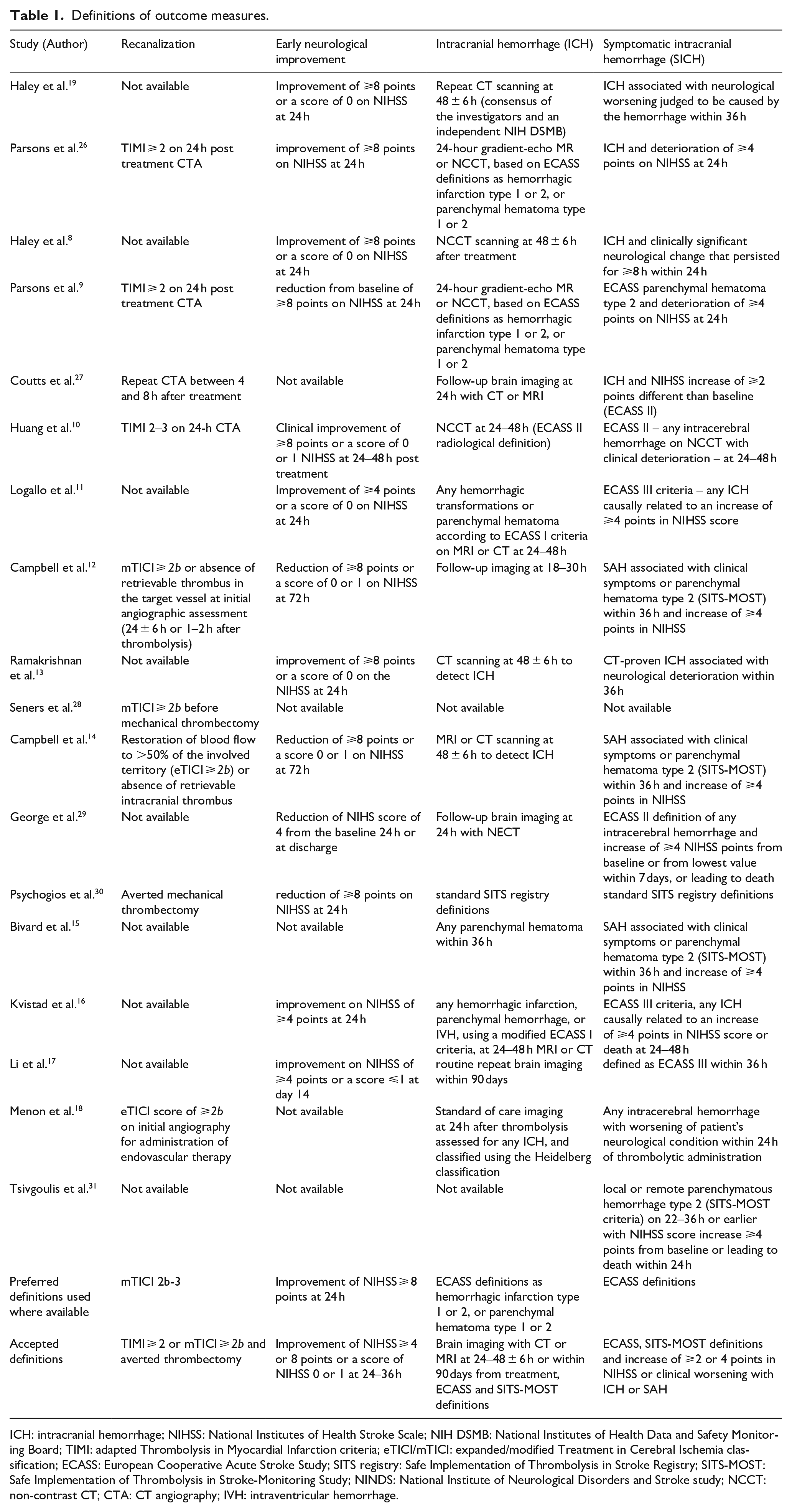
ICH: intracranial hemorrhage; NIHSS: National Institutes of Health Stroke Scale; NIH DSMB: National Institutes of Health Data and Safety Monitoring Board; TIMI: adapted Thrombolysis in Myocardial Infarction criteria; eTICI/mTICI: expanded/modified Treatment in Cerebral Ischemia classification; ECASS: European Cooperative Acute Stroke Study; SITS registry: Safe Implementation of Thrombolysis in Stroke Registry; SITS-MOST: Safe Implementation of Thrombolysis in Stroke-Monitoring Study; NINDS: National Institute of Neurological Disorders and Stroke study; NCCT: non-contrast CT; CTA: CT angiography; IVH: intraventricular hemorrhage.
Data extraction
Data are extracted from selected studies with the use of a preformed data extraction excel sheet. Raw data, for example, counts of outcome event and sample size, are preferred over calculated data, for example, odds ratio (OR) and risk ratio (RR) and per-protocol data are preferred over intention-to-treat (ITT) data. 32
Data analysis
Data analyses were done with R 33 within RStudio interface. 34 Meta-analyses were done with “Meta” package 35 and NMAs were done with “Netmeta” package 36 (Supplemental Information on data analysis).
Risk of bias assessment
Assessment of risk of bias in studies were done, in Review Manager software, 37 by GRADE assessment tool for included RCTs. 38 Assessment of risk of bias in included observational studies was done by Newcastle-Ottawa Scale (NOS). 39 The quality of the observational study was considered high quality if it attains seven or more stars.
Results
The searches returned 232 results from Medline, 172 from CENTRAL and 34 from ClinicalTrials.gov. There were 114 duplicate results. Scanning the titles, abstracts, and full text availability of the search results gave 122 articles. Additional searches in the references, meta-analyses and included papers were performed. After removing study protocols (
Included studies
Primary studies available for the analyses include RCTs (
Risk of bias
Most of the included studies did not perform blinding of treatment allocation. Nevertheless, the assessors were blinded to treatment allocation in detecting outcomes in many of the studies including the observational studies (Supplemental Figure S1 and Table S3).
Recanalization
Eight studies9,10,12,18,26,28
–30 are included in the meta-analysis for recanalization outcome. The current meta-analysis considered not only complete recanalization (TIMI = 3 or mTICI = 3) but also recanalization more than 50% (TIMI ⩾ 2 or mTICI ⩾
There is total 1255 individuals and no significant differences in recanalization for three different dosage subgroups of TNK compared to rt-PA nor between these TNK subgroups (
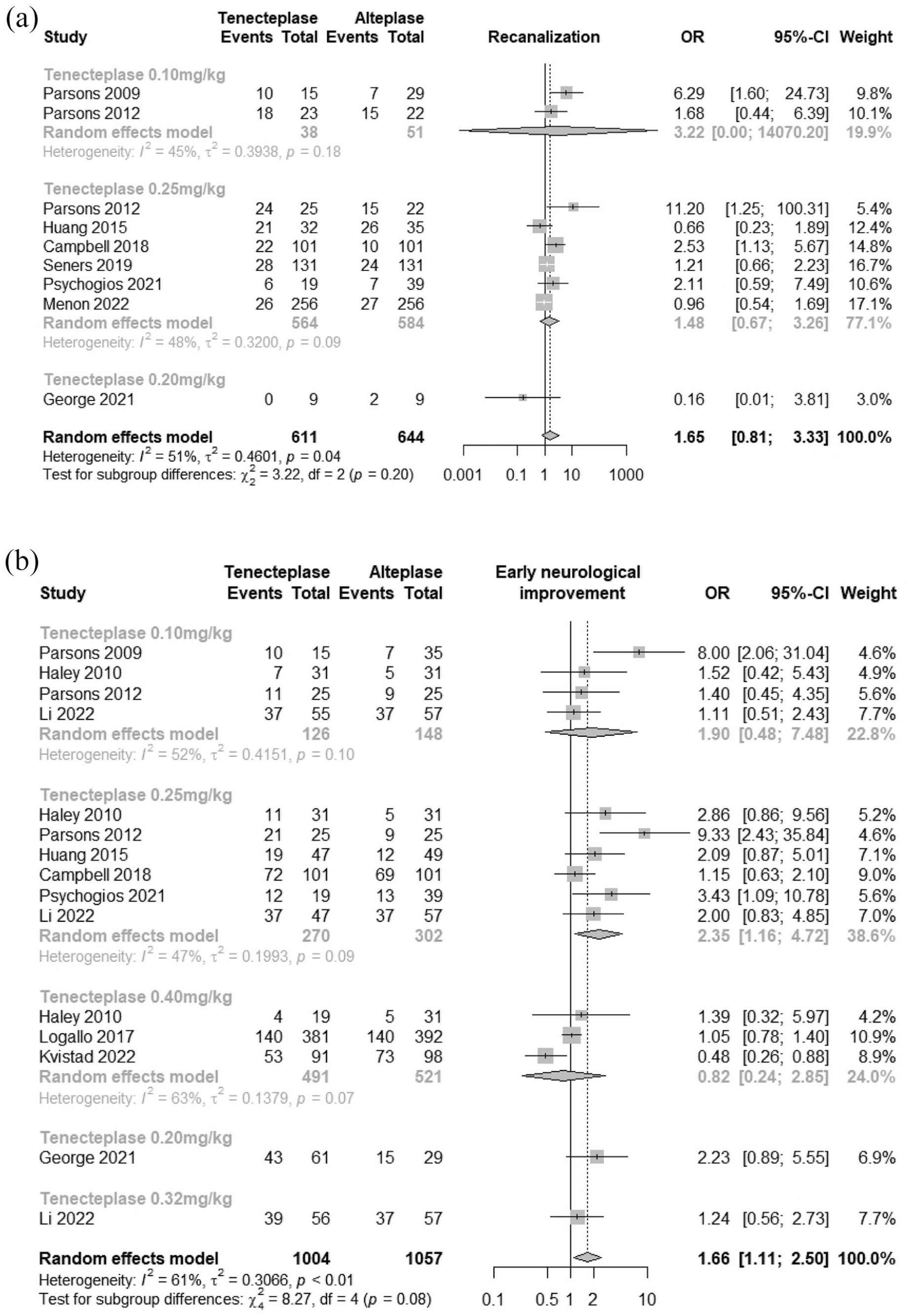
Forest plots of functional outcomes: (a) recanalization and (b) early neurological improvement.
In NMA, nine studies9,10,12,14,18,26
–28,30 (total 1561 individuals) were involved. There were no significant differences seen between TNK dosages and rt-PA for recanalization. Test of inconsistency between designs show statistical significance (
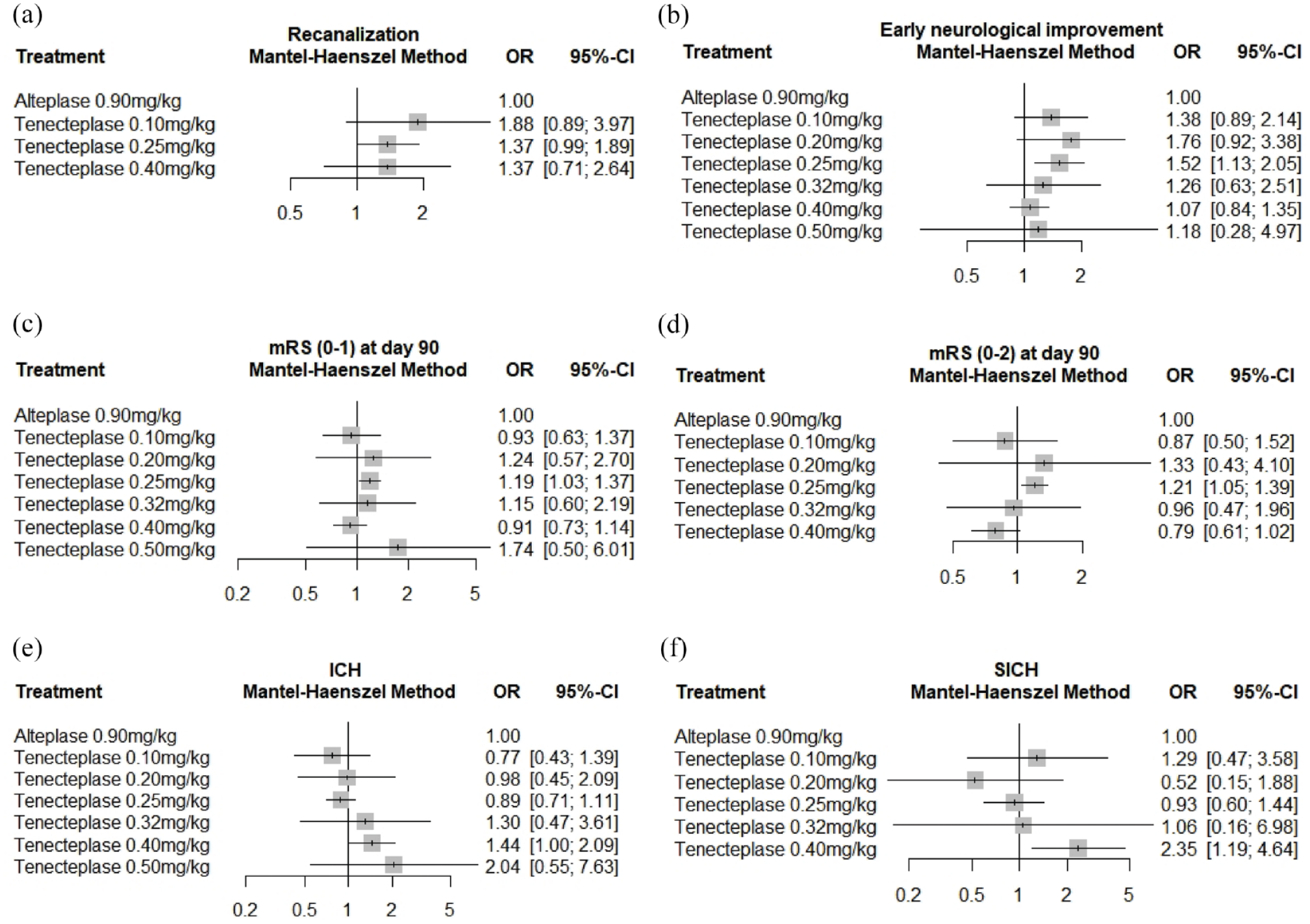
Network meta-analysis results on (a) Recanalization, (b) Early neurological improvement, (c) Excellent functional outcome (mRS 0–1) at day 90, (d) Good functional outcome (mRS 0–2) at day 90, (e) Intracranial hemorrhage (ICH), (f) Symptomatic intracranial hemorrhage (SICH).
Early neurological improvement
Ten studies8
–12,16,17,26,29,30 (2061 individuals) were included in meta-analysis. The results are significant for TNK 0.25 mg/kg subgroup (OR = 2.35 [95% CI = 1.16–4.72]) and in the overall grouped comparison comprising all TNK dosages (OR = 1.66 [95% CI = 1.11–2.50],
Additional analysis excluding the study that measured outcomes at 14 days was performed.
17
The overall group comparison was still significantly in favor of TNK treatment (OR = 1.81 [95% CI = 1.07–3.06],
For NMA, 13 studies8–14,16,17,19,26,29,30 (2359 individuals) are available. TNK 0.25 mg/kg has significantly increased odds of early neurological improvement (OR = 1.52 [95% CI = 1.13–2.05],
Excellent functional outcome (mRS 0–1) at day 90
Total 11 studies8 –12,15 –18,26,31 (4419 individuals) were available for meta-analysis. TNK 0.25 mg/kg showed significantly higher odds for excellent functional outcomes compared to rt-PA (OR = 1.20 [95% CI = 1.02–1.42]) (Figure 3(a)).
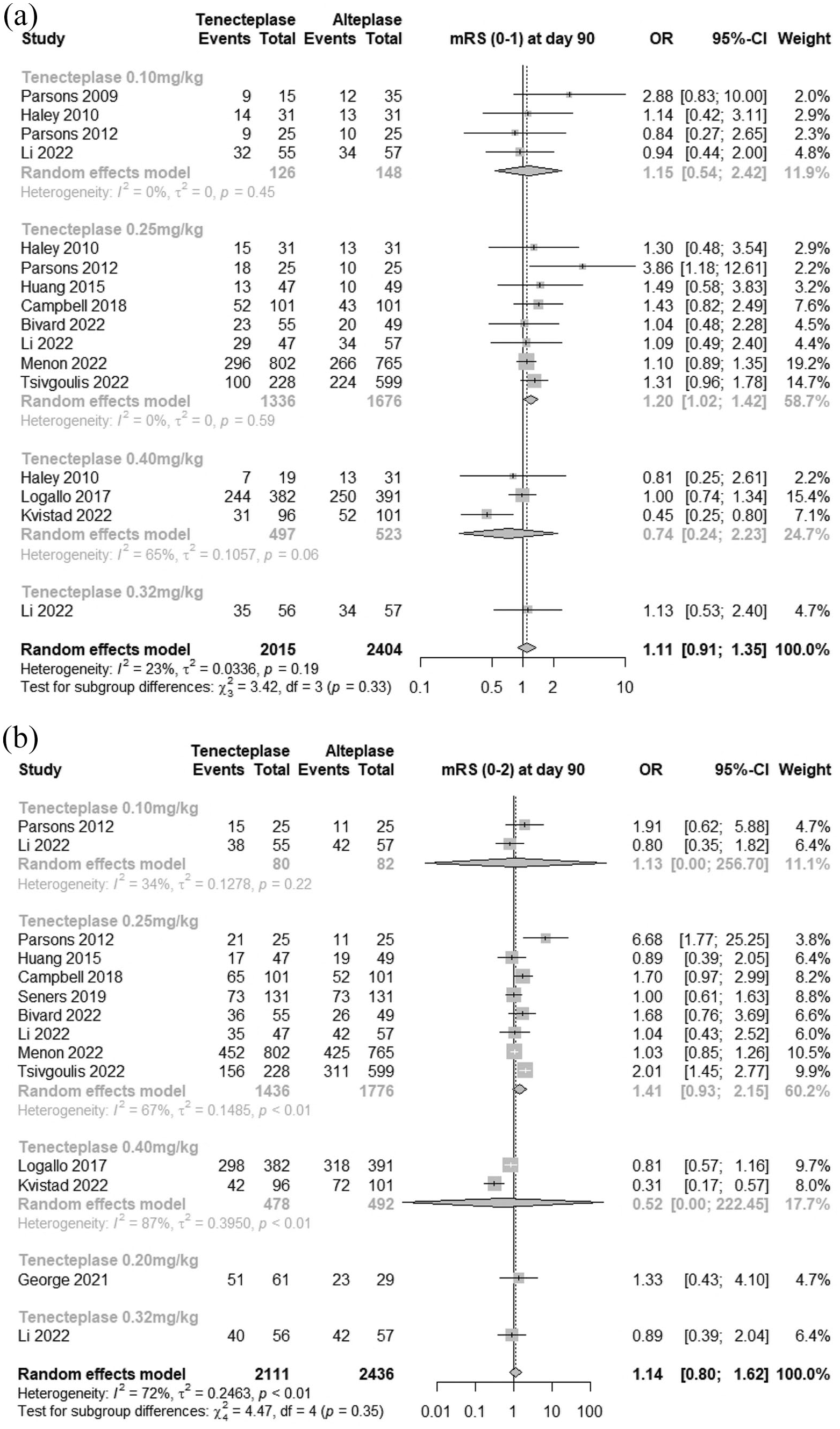
Forest plots of functional outcomes. (a) Excellent functional outcome (mRS 0–1) at day 90. (b) Good functional outcome (mRS 0–2) at day 90.
Total 15 studies,8
–19,26,27,31 (4767 individuals) are available for NMA. TNK 0.25 mg/kg shows statistically significant excellent functional outcomes (OR = 1.19 [95% CI = 1.03–1.37],
Good functional outcome (mRS 0–2) at day 90
For the meta-analysis on good functional outcomes (mRS 0–2), 11 studies9
–12,15
–18,28,29,31 (4547 individuals) were available. The overall combined TNK cohort was not significant (OR = 1.14 [95% CI = 0.80–1.62],
In the NMA, there are 13 studies9
–12,14
–18,27
–29,31 (4756 individuals). The odds of good functional outcomes for TNK 0.25 mg/kg is significantly higher than rt-PA (OR = 1.21 [95% CI = 1.05–1.39],
Intracranial hemorrhage
Most of the studies used outcomes within 48 h from treatment except Li et al. 17 which included outcomes up to 90 days (Table 1). For the purpose of meta-analysis, hemorrhagic infarction reported in some studies has been taken as ICH.
There are 12 studies8
–12,15
–18,26,29,30 (3791 individuals) available for meta-analysis. Combined TNK group did not reach statistical significance (OR = 1.05 [95% CI = 0.71–1.55],
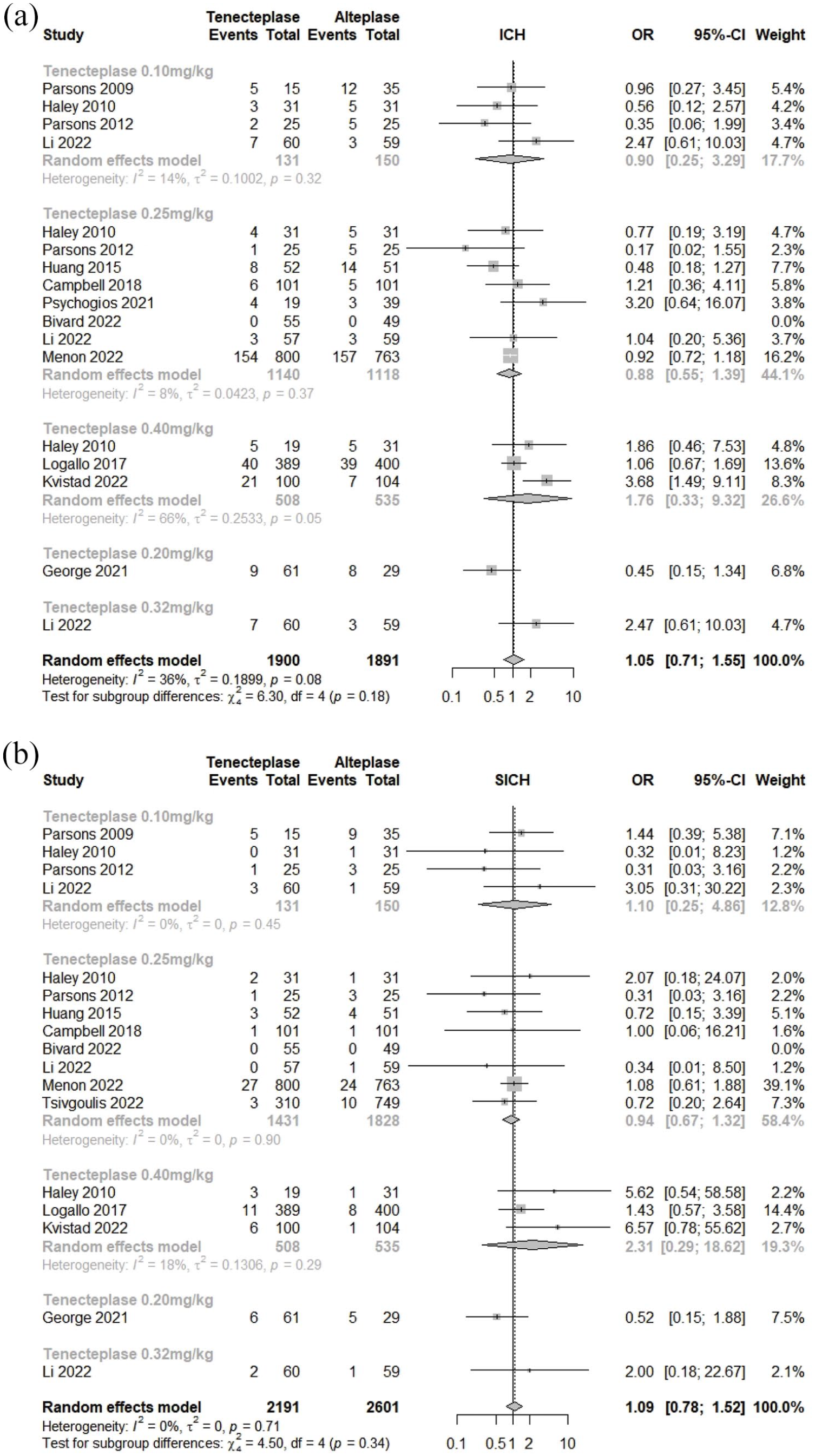
Forest plots of safety outcomes: (a) Intracranial hemorrhage (ICH) and (b) symptomatic intracranial hemorrhage (SICH).
An analysis without Li et al.
17
was done in view of difference in the window for their definition of ICH and again showed non-significant result (OR = 0.95 [95% CI = 0.61–1.49],
For the NMA, 14 studies8
–13,16
–19,26,27,29,30 (3670 individuals) are available. The results are non-significant. There was no significant inconsistency between designs (
Symptomatic intracranial hemorrhage
For this outcome, studies defined it as clinical deterioration of four or more points increase in NIHSS in addition to ICH (Table 1). There are twelve studies8
–12,16
–18,26,29,31 (4792 individuals) available for the meta-analysis. Overall TNK treatment has non-significant higher odds of SICH occurring (OR 1.09 [95% CI = 0.78–1.52],
For NMA, there are twelve studies8
–12,14,16
–18,26,29,30 (4695 individuals). TNK 0.40 mg/kg was significantly associated with SICH (OR = 2.35 [95% CI = 1.19–4.64],
Mortality within 90 days
In this meta-analysis, it is the all-cause mortality that was reported in ten studies8 –12,15 –18,31 (4381 individuals). All treatment groups with TNK are not statistically significant (Figure 5(a)). Additional analysis including George et al. 29 that reported stroke and treatment related mortality are reported in Supplemental Figures S7 and S8.
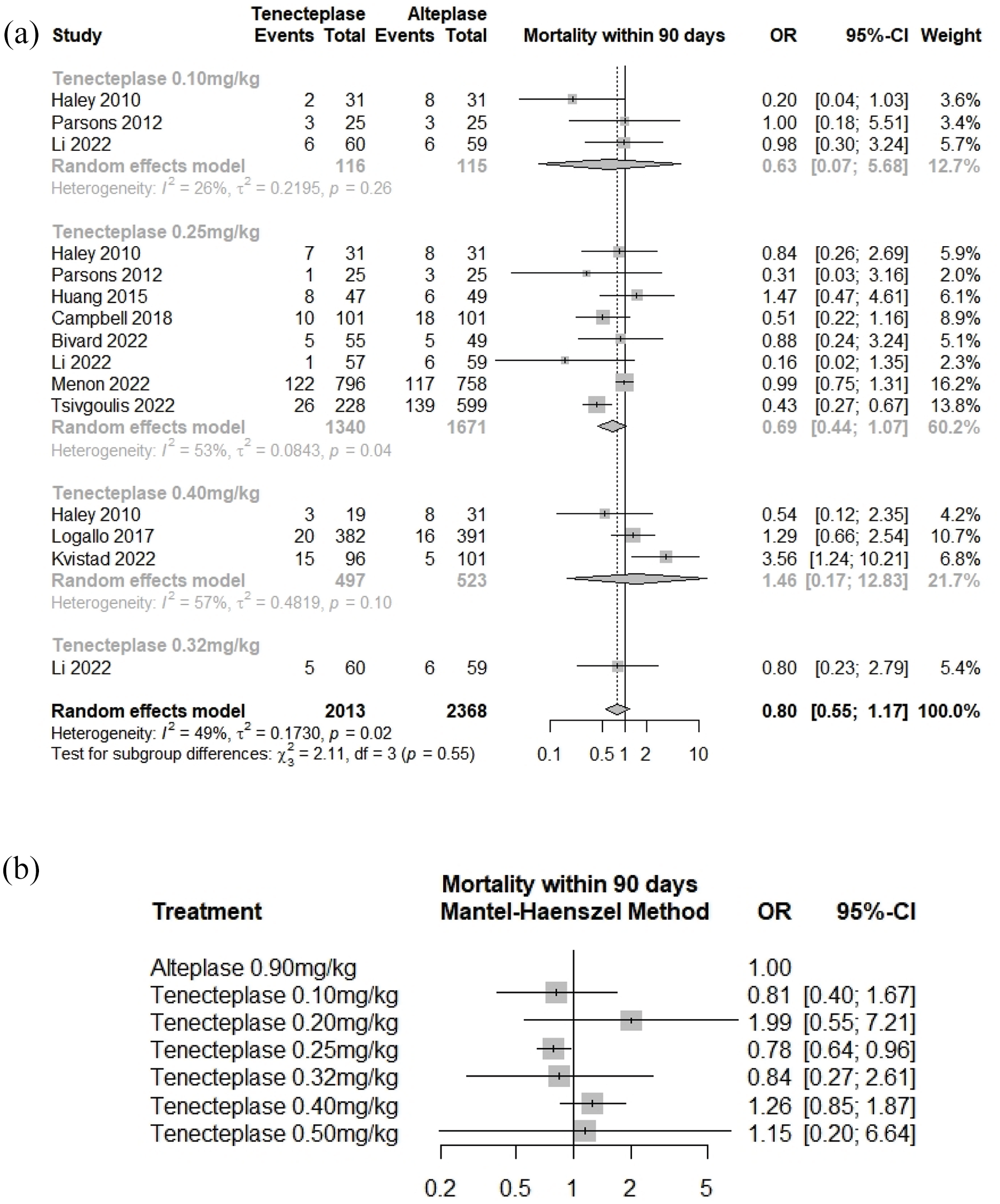
(a) Forest plot of Mortality within 90 days, (b) Network meta-analysis results on Mortality within 90 days.
There are twelve studies8
–12,14
–18,26,31 (4564 individuals) for the NMA. TNK 0.25 mg/kg showed statistically significant reduced mortality (OR = 0.78 [95% CI = 0.64–0.96],
However, there are no significant inconsistency between designs (
Results of analyses limited to RCTs only
We have also performed additional analyses limited to data involving RCTs only (Supplemental Figures S9–S26). To summarize, there are no significant findings in any of the additional meta-analyses. However, in the NMAs, there are significant findings on TNK 0.40 mg/kg on mRS 0–2 outcome (OR = 0.76 [95% CI = 0.59–0.99],
Discussion
In this NMA, we uncovered several major findings. Firstly, in the meta-analysis, TNK at a dose of 0.25 mg/kg was associated with higher rates of early neurological improvement and excellent functional outcomes (mRS 0–1) at 3 months. In the network meta-analysis (NMA), it also showed improved odds of early neurological improvement, mRS 0–1, mRS 0–2 and mortality at 90 days. In contrast, TNK 0.40 mg/kg was associated with a higher risk of SICH. However, there are no significant findings on any of the outcome measures for TNK 0.10, 0.20, 3.2, and 0.50 mg/kg in the meta-analyses nor NMAs. Results of all the analyses are summarized in Supplemental Tables S4 and S5.
TNK is a genetically modified thrombolytic with a longer half-life, better resistance to removal by plasminogen activator inhibitor-1 and increased fibrin specificity.40 –42 This translated into better reperfusion and lower risk of intracranial bleeds in earlier animal models. 43
Recanalization
Averted thrombectomy is possibly a robust measure of the efficacy of bridging thrombolytic, since recanalization associated with thrombectomy may conceal potential differences between TNK and rt-PA. In the EXTEND-IA, there was an increase in the rate of averted thrombectomy of 12% but this did not reach significance. 12
TASTE-A showed a higher incidence of early distal clot migration on repeat imaging. This distal clot migration is highly suggestive that clot lysis had taken place between TNK and repeat scan and may be a surrogate marker of thrombolytic effectiveness. 15 Their findings are consistent with a pooled analysis of two TNK RCTs that showed TNK was associated with significantly higher recanalization at 24 h compared to rt-PA. 44
Early neurological improvement
In our meta-analysis, TNK 0.25 mg/kg was associated with early neurological improvement. Even after removing the study with a different time window (at 14 days after treatment), TNK 0.25 mg/kg showed a strong trend to significance. The NMA also showed that TNK 0.25 mg/kg was associated with early neurological improvement. These findings are supportive of an earlier trial where 0.25 mg/kg of TNK was superior to rt-PA or 0.10 mg/kg TNK. 9
Functional outcomes (mRS 0–1, 0–2) and mortality
In EXTEND-IA TNK trial,12,45 TNK 0.25 mg/kg showed non-significant improvement in functional outcomes at 3 months compared to rt-PA. This finding dovetailed with Alteplase Compared to Tenecteplase (AcT), which compared TNK 0.25 mg/kg against rt-PA and managed to reach the non-inferiority for mRS 0–1 at 90 days and validated the 0.25 mg/kg without an increase in SICH and mortality. 18
With the increased sample size and power in our meta-analysis, TNK 0.25 mg/kg was significantly associated with excellent functional outcome. Our NMA showed TNK 0.25 mg/kg was significantly associated with excellent functional outcomes (mRS 0–1), good functional outcomes (mRS 0–2) at 90 days and improved mortality.
SICH
In NOR-TEST, 0.4 mg/kg TNK showed no difference in ICH, mortality, or other adverse events compared to rt-PA. 11 However a criticism was that the patients included in NOR-TEST had milder strokes, median NIHSS of 4 (IQR = 2–8), and about 1/5th were stroke mimics. The EXTEND-IA TNK trials also reported that 0.4 mg/kg was not associated with worse outcomes. 45
The NOR-TEST 2 trial that used the same protocol as the earlier trial but with a higher NIHSS showed no improvement in recanalization but had more incidence of bleeds in the 0.4 mg/kg TNK group. 16 While these findings appear to be in contrast to those of NOR-TEST and EXTEND-IA TNK studies, nonetheless, there was an increased mortality seen in the NOR-TEST study in the subgroup with severe stroke which had prompted an early safety review to be undertaken in the NOR-TEST 2 trial. 46
In our study, TNK was not associated with increased risk of SICH compared to rt-PA in the meta-analysis. In NMA, TNK 0.40 mg/kg is significantly associated with SICH and validated the results of NOR-TEST 2.
Dosing
The non-inferiority of TNK to rtPA was previously described. 22 The recent three studies enabled us to better understand the optimal TNK dosing. Our study validates higher risk of SICH with 0.4 mg/kg and early neurological improvement, functional outcomes and mortality with 0.25 mg/kg doses.
What we have discussed so far referred to our results inclusive of observational studies. Our analyses limited to RCT data differ in that only TNK 0.40 mg/kg showed significant results in the NMAs on mRS 0–2, ICH (including Li et al. 17 ) and SICH outcomes in which it performed significantly worse than rt-PA.
As with previous meta-analyses,20,22,23 our analyses using RCT data alone have been insufficient to reach significant findings for all of the outcomes in the meta-analyses and some outcomes in the NMAs. Adding observational studies into the analysis provides additional power. However, one should be caution when interpreting analyses inclusive of observational study data and we do not intend for our findings to be taken as conclusive. Our findings will still need to be validated in further randomized studies.
In this study, we included 11 RCTs and 7 observational studies, which is the biggest sample size of any meta-analysis to date and is sufficiently powered to conduct sub-group analyses of different TNK doses. This has an advantage over previous meta-analyses that combined different dosages of TNK into one arm. 21
Limitations
There is considerable heterogeneity across several clinical outcomes and these likely stemmed from the differences in inclusion criteria such as age limits, baseline NIHSS score, previous level of morbidity and imaging modality. The onset-to-treatment timing are also different with three studies looking at a time window later than 4.5 h while the rest of the studies treated patients less than 4.5 h.9,26,27
The studies also had differences in stroke severity with Coutts et al. 27 explicitly studying minor stroke with an intracranial vessel occlusion and the NOR-TEST participants 11 having a median NIHSS of 4 while in contrast, the rest of the trials included had no limitations on the baseline NIHSS. Five studies included LVO stroke patients9,12,14,28,30 while the rest included both LVO and non-LVO patients and this combined variations between studies could potentially introduce substantial heterogeneity into the analyses.
It is also important to note that the various analyses were performed on per-protocol results which could inflate the differences between groups. This approach could introduce additional biases and heterogeneity in addition to those that had existed between individual studies due to their differing per-protocol treatments. 47
Finally, there is the assumption of control arms being comparable in a network meta-analysis, which may not be true. The outcomes for the control arms in the included studies are heterogeneous (Supplemental Table S6). Furthermore, in the NMAs we performed on recanalization, early neurological improvement and mRS 0–2, there was significant differences in the test of inconsistency between study designs.
Conclusion
In our NMA, 0.40 g/kg TNK was associated with significantly more SICH, while 0.25 mg/kg was significantly associated with better early neurological improvement, good and excellent functional outcomes and lower mortality. This study provides further evidence to aid dose selection for TNK in ischemic stroke.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221129924 – Supplemental material for Different dosing regimens of Tenecteplase in acute ischemic stroke: A network meta-analysis of the clinical evidence
Supplemental material, sj-docx-1-eso-10.1177_23969873221129924 for Different dosing regimens of Tenecteplase in acute ischemic stroke: A network meta-analysis of the clinical evidence by Ei Zune The, Ne Naing Lin, Mazen Matar, Hock Luen Teoh and Leonard Leong Litt Yeo in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable since the study uses published data.
Informed consent
Not applicable.
Guarantor
EZT
Contributorship
All authors contributed equally.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
