Abstract

1665
HEALTH-RELATED QUALITY OF LIFE AND PREVALENCE OF DEPRESSION IN PATIENTS WITH HEREDITARY AND SPORADIC CEREBRAL AMYLOID ANGIOPATHY (CARE-STUDY)
1 Leiden University Medical Center, Neurology, Leiden, Netherlands
1667
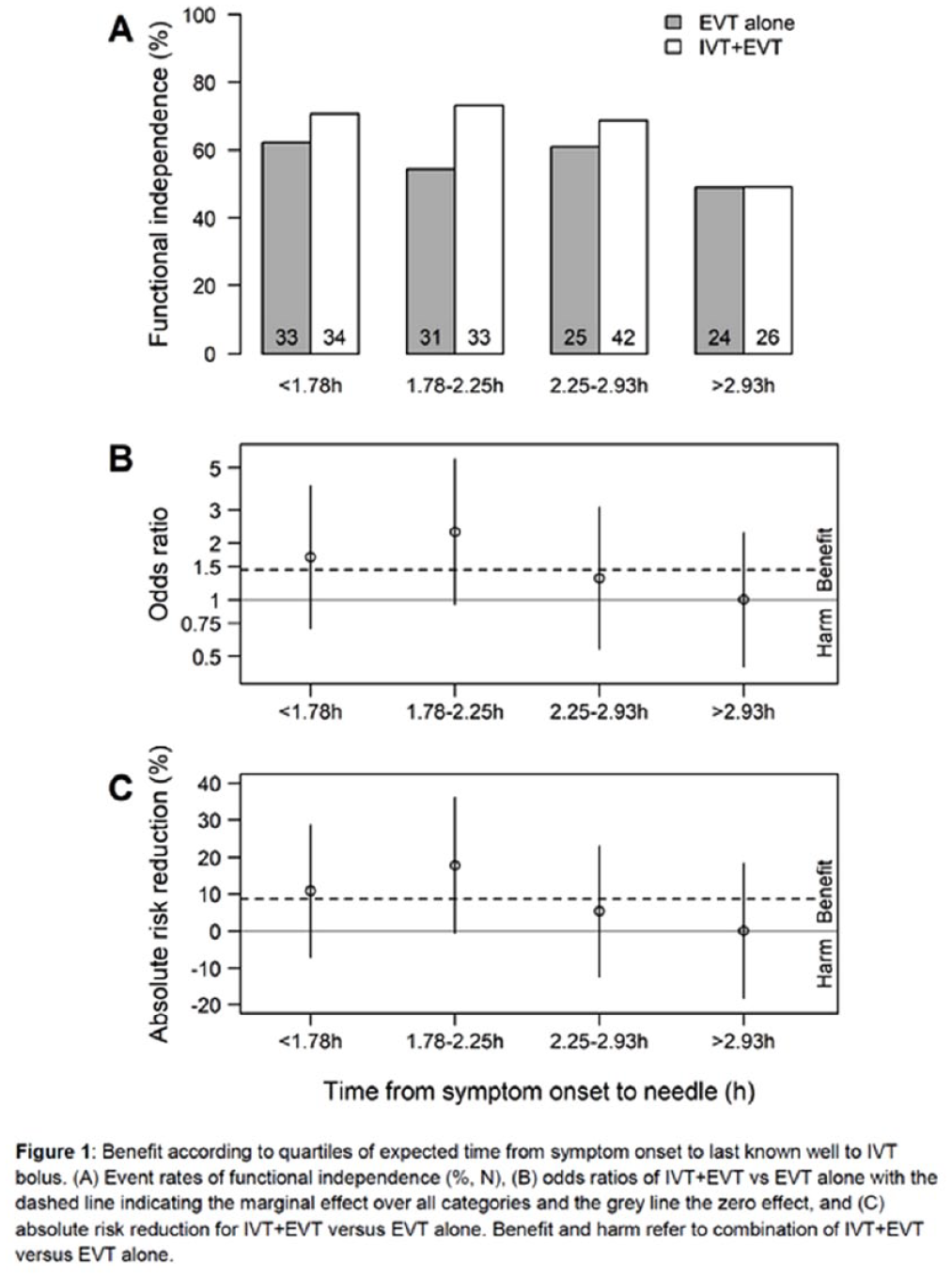
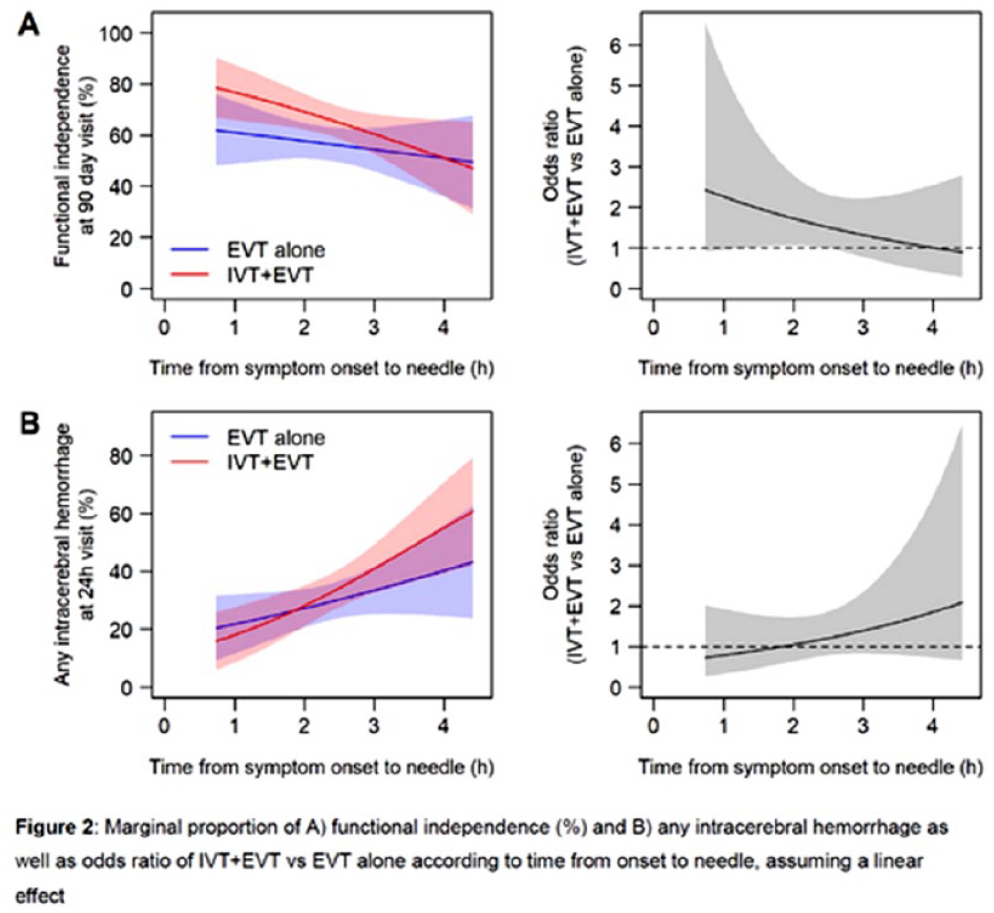
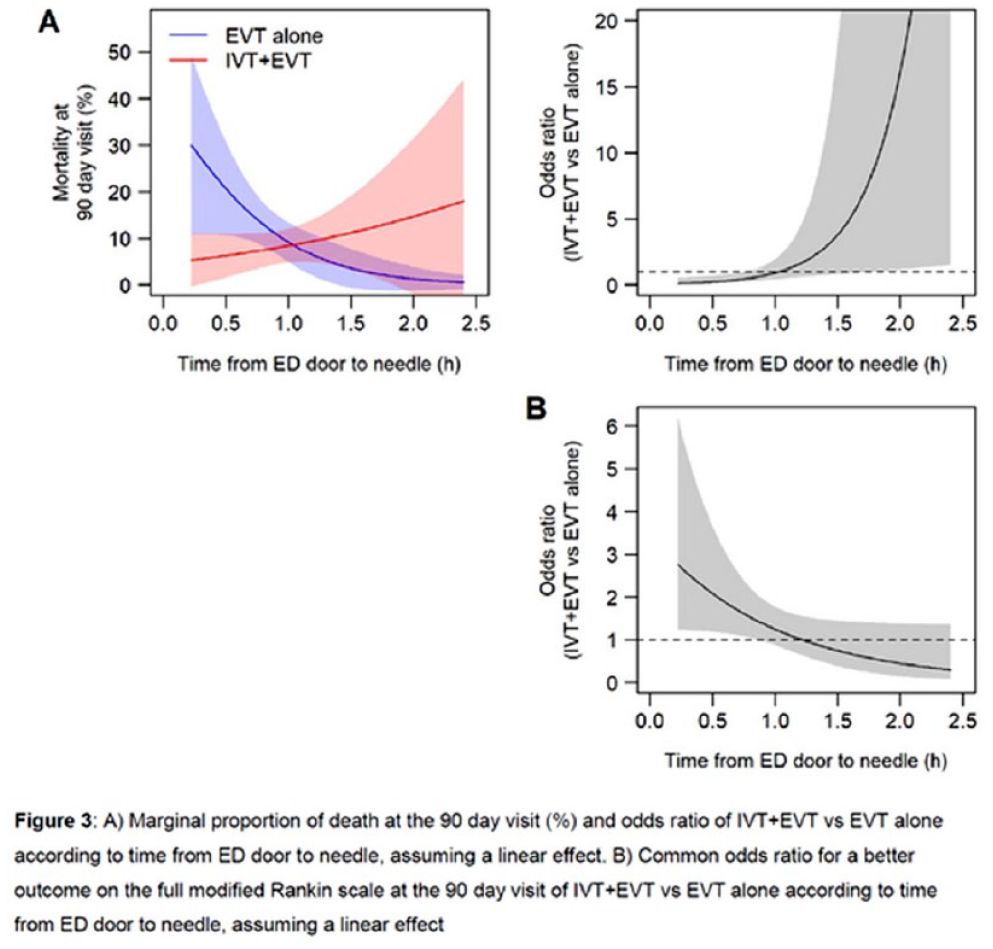
TIME TO TREATMENT WITH BRIDGING INTRAVENOUS ALTEPLASE PRIOR TO ENDOVASCULAR TREATMENT – SUBANALYSIS OF THE RANDOMIZED CONTROLLED SWIFT-DIRECT TRIAL
1Stroke Research Center Bern, Inselspital, University of Bern, Bern, Switzerland, 2University of Bern, Switzerland, University Institute of Diagnostic and Interventional Neuroradiology, Bern, Switzerland, 3CTU Bern, University of Bern, Bern, Switzerland, Bern, Switzerland, 4Department of Neurology, Helsinki University Hospital and University of Helsinki, Finland., Helsinki, Finland, 5Department of Neuroradiology, Hospices Civils de Lyon, Lyon, France, Lyon, France, 6Department of Diagnostic and Therapeutic Neuroradiology, Centre Hospitalier Universitaire de Toulouse, Toulouse, France., Toulouse, France, 7University Institute of Diagnostic and Interventional Neuroradiology, Inselspital, Bern University Hospital, and University of Bern, Bern, Switzerland, 8Neuro Clinical Trial Unit, Department of Neurology, Inselspital, Bern University Hospital, and University of Bern, Switzerland., Bern, Switzerland, 9Division of Neuroradiology and Division of Neurosurgery, Departments of Medical Imaging and Surgery, Toronto Western Hospital, University Health Network, University of Toronto, Toronto, Canada, Toronto, Canada, 10Department of Neurology, Centre Hospitalier Universitaire de Toulouse, Toulouse, France, Toulouse, France, 11Department of Diagnostic and Interventional Neuroradiology, Centre Hospitalier Universitaire de Nantes, Nantes Université, Nantes, France, Nantes, France, 12Department of Neurology, Centre Hospitalier Universitaire de Nantes, Nantes Université, Nantes, France ., Nantes, France, 13Department of Radiology, CHU Rouen, Rouen, France, Rouen, France, 14Department of Neurology, CHU Rouen, Rouen, France, Rouen, France, 15Department of Neurology, Helsinki University Hospital and University of Helsinki, Finland, Helsinki, Finland, 16Department of Diagnostic and Therapeutic Neuroradiology, CHRU-Nancy, Université de Lorraine, INSERM U1254, Nancy, France, Nancy, France, 17Department of Neurology, Stroke Unit, CHRU-Nancy, Université de Lorraine, INSERM U1116, Nancy, France, Nancy, France, 18Department of Neurology, University Medical Center Goettingen, Germany, Goettingen, Germany, 19Department of Neuroradiology, University Medical Center Goettingen, Germany, Goettingen, Germany, 20Department of Neurology, CHU Caen Normandie, University Caen Normandie, INSERM U1237, Caen, France, Caen, France, 21Department of Neuroradiology, CHU Caen Normandie, University Caen Normandie, INSERM U1237, Caen, France, Caen, France, 22Department of Vascular Neurology, Hospices Civils de Lyon, Lyon, France, Lyon, France, 23Department of Neurology, St George’s University Hospital NHS Foundation Trust, London, UK., London, United Kingdom, 24Department of Interventional and Diagnostic Neuroradiology, CHU Bordeaux, University of Bordeaux, Bordeaux, France, Bordeaux, France, 25Stroke Unit, CHU Bordeaux, University of Bordeaux, Bordeaux, France, Bordeaux, France, 26Department of Neuroradiology, University Hospital RWTH Aachen, Aachen, Germany, Aachen, Germany, 27Department of Neurology, University Hospital RWTH Aachen, Aachen, Germany., Aachen, Germany, 28Department of Stroke and Diagnostic and Interventional Neuroradiology, Foch Hospital, Suresnes, France, Suresnes, France, 29Stroke Unit. Department of Neurology. Hospital Vall d’Heborn. Barcelona, Spain, Barcelona, Spain, 30Interventional Neuroradiology. Department of Radiology. Hospital Vall d’Heborn. Barcelona, Spain., Barcelona, Spain, 31Department of Neuroradiology, University Hospital of Limoges, France, Limoges, France, 32Department of Neurology, University Hospital of Limoges, France., Limoges, France, 33Department of Neurology, CHU Reims, Reims, France., Reims, France, 34Department of Neuroradiology, CHU Reims, Reims, France, Reims, France, 35Service of Interventional and Diagnostic Radiology, Centre Hospitalier Universitaire Vaudois and University of Lausanne, Lausanne, Switzerland, Lausanne, Switzerland, 36Department of Neurology, Hôpitaux Universitaires de Genève, Geneva, Switzerland., Geneva, Switzerland, 37Department of Diagnostic and Interventional Neuroradiology, Tours University Hospital, Tours, France., Tours, France, 38Stroke Unit, Department of Neurosciences, University Hospital Germans Trias i Pujol, Barcelona, Spain., Barcelona, Spain, 39Department of Interventional Neuroradiology, Strasbourg University Hospitals, Strasbourg, France., Strasbourg, France, 40Department of Neurology, Lille University Hospital, Lille, France, Lille, France, 41Department of Neurology, University Hospital of Zurich, Zurich, Switzerland. Cereneo, Center for Neurology and Rehabilitation, Vitznau, Switzerland., Zurich, Switzerland, 42Department of Neurology, Inselspital, Bern University Hospital, and University of Bern, Switzerland, Bern, Switzerland, 43Departments of Neurology, Lariboisière hospital and Interventional Neuroradiology, Adolphe de Rothschild Hospital Foundation, University of Paris, FHU NeuroVasc, INSERM 1148, Paris, France., Paris, France, 44Department of Neurology and Comprehensive Stroke Center, David Geffen School of Medicine, University of California, Los Angeles, USA, Los Angeles, United States, 45Department of Neurology, University Hospital Basel, University of Basel, Basel, Switzerland, Basel, Switzerland, 46Department of Neurology, Inselspital, Bern University Hospital, and University of Bern, Switzerland., Bern, Switzerland, 47University Institute of Diagnostic and Interventional Neuroradiology, Inselspital, Bern University Hospital, and University of Bern, Switzerland., Bern, Switzerland
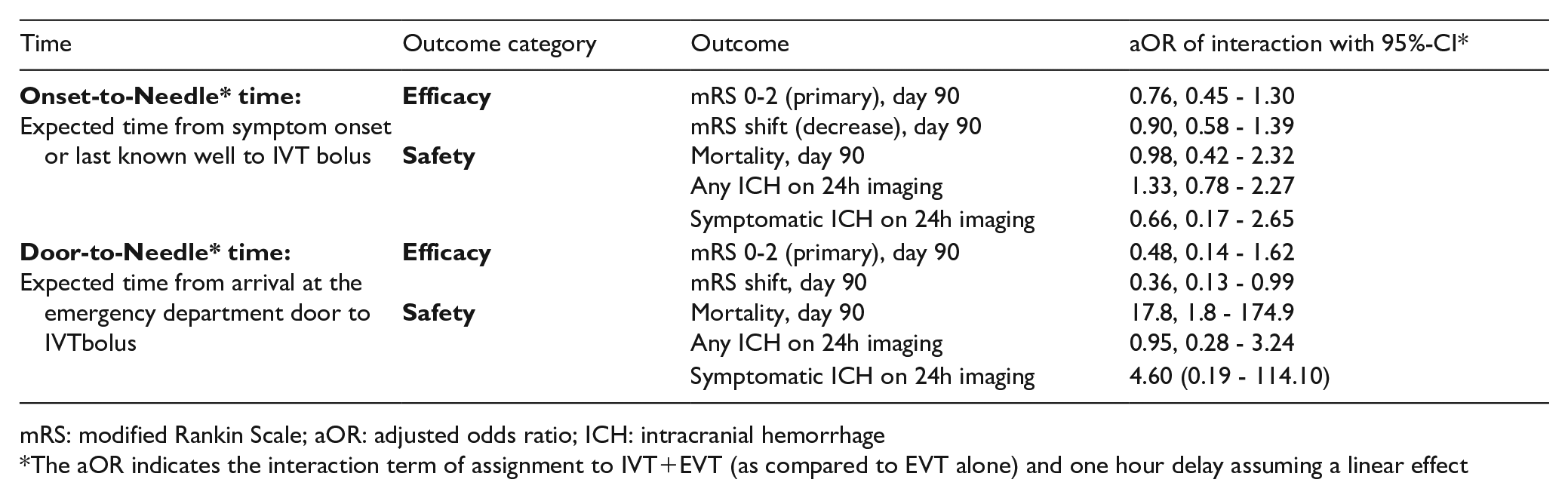
mRS: modified Rankin Scale; aOR: adjusted odds ratio; ICH: intracranial hemorrhage
The aOR indicates the interaction term of assignment to IVT+EVT (as compared to EVT alone) and one hour delay assuming a linear effect
1670
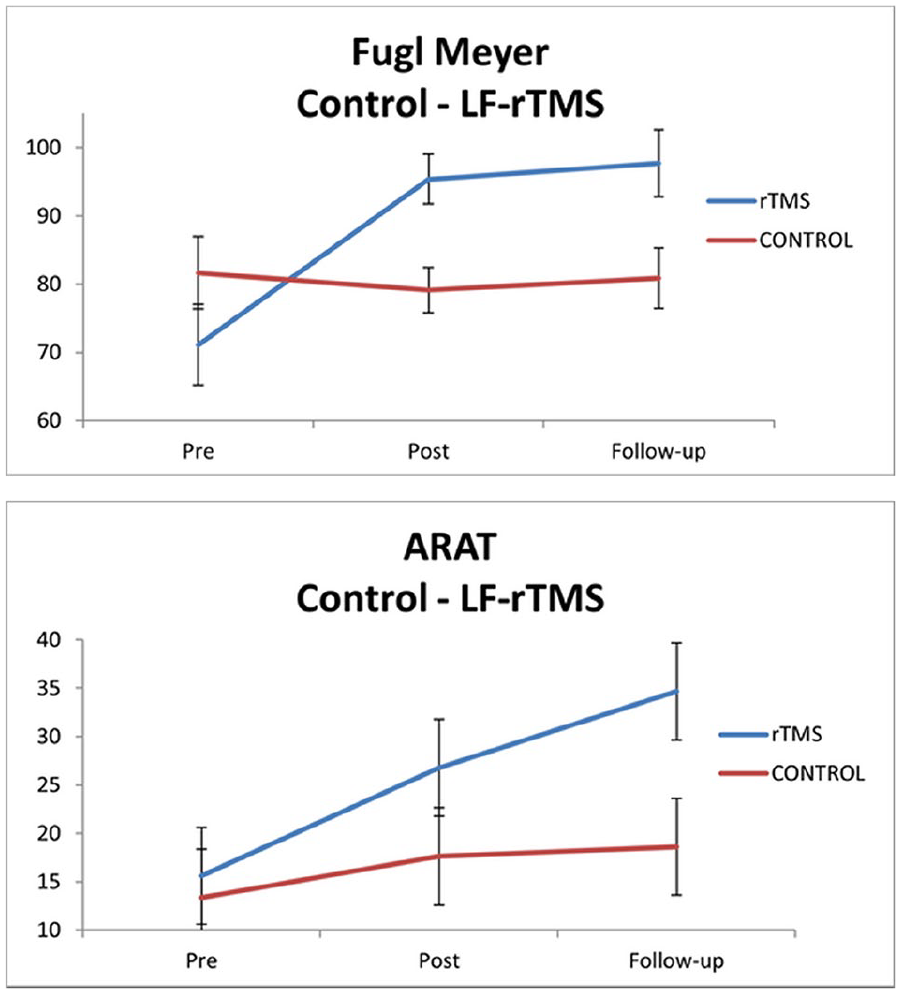
EFFICACY AND SAFETY OF LF-RTMS ASSOCIATED WITH INTENSIVE NEUROREHABILITATION TO IMPROVE UPPER EXTREMITY FUNCTIONALITY IN PATIENTS IN A SUBACUTE PHASE OF STROKE. A PILOT STUDY
1Nadia Sánchez, Research and Innovation Office - Neurorrehabilitation Department, Badalona, Spain, 2Institut Guttmann. Universitat Autònoma Barcelona (UAB), Research and Innovation Office - Neurorrehabilitation Department, Badalona, Spain
1671
APIXABAN FOR TREATMENT OF EMBOLIC STROKE OF UNDETERMINED SOURCE - ATTICUS RANDOMIZED TRIAL
1University of Tübingen, Department of Neurology & Stroke, Tübingen, Germany, 2University of Tübingen, Institute for Clinical Epidemiology and Applied Biometry, Tübingen, Germany, 3Klinikum Stuttgart, Department of Neurology, Stuttgart, Germany, 4Krankenhaus Martha-Maria Halle-Dölau, Department of Neurology, Halle-Dölau, Germany, 5Rems-Murr-Kliniken Winnenden, Department of Neurology, Winnenden, Germany, 6Carl-von-Basedow-Clinic Saalekreis, Department of Neurology, Merseburg, Germany, 7University of Göttingen, Department of Neurology, Göttingen, Germany, 8University of Göttingen, Department of Cardiology, Göttingen, Germany, 9Schwarzwald-Baar Hospital, Department of Neurology, Villingen-Schwenningen, Germany, 10Schwarzwald-Baar Hospital, Department of Cardiology, Villingen-Schwenningen, Germany, 11Medical Campus Lake Constance, Department of Neurology, Friedrichshafen, Germany, 12Marien Hospital Stuttgart, Department of Neurology, Stuttgart, Germany, 13University of Ulm, Department of Neurology, Ulm, Germany, 14University of Kiel, Department of Neurology, Kiel, Germany, 15Klinikum Ludwisburg, Department of Neurology, Ludwigsburg, Germany, 16University of Bonn, Department of Neurology, Bonn, Germany, 17Regiomed Kliniken Coburg, Department of Cardiology, Coburg, Germany, 18Medical Park Berlin Humboldtmühle, Department of Neurology, Berlin, Germany, 19University of Tübingen, Department of Cardiology, Tübingen, Germany
Aim of ATTICUS was to determine whether apixaban, initiated within 28 days after ESUS, is superior to ASA in preventing new ischemic lesions on 12-month follow- up MRI (primary endpoint) in subjects with remote cardiac monitoring.
Enrollment stopped after interims analysis due to futility. Of 373 patients screened, 178/175 were enrolled into the apixaban/ASA treatment arm.
The ATTICUS trial is funded by the Bristol-Meyers Squibb/Pfizer Alliance (Euro 2,164,650) and Medtronic.
1672
GLOBAL IMPACT OF THE COVID-19 PANDEMIC ON CEREBRAL VENOUS THROMBOSIS AND MORTALITY
1Boston University School of Medicine, Boston Medical Center, Neurology, Radiology, Boston, United States, 2Boston Medical Center, Radiology, Radiation Oncology, Boston, United States, 3Boston University School of Medicine, Boston, United States, 4NHO Osaka National Hospital, Neurology, Osaka, Japan, 5Boston Medical Center, Boston University School of Medicine, Radiology, Boston, United States, 6International Clinical Research Centre, St Anne’s University Hospital and Faculty of Medicine, Masaryk University, Neurology, Brno, Czech Republic, 7Alexandria University Stroke and Neurointervention Unit, Neurology, Alexandria, Egypt, 82nd Department of Neurology, Institute of Psychiatry and Neurology, Neurology, Warsaw, Poland, 9University of British Columbia, Department of Medicine, Division of Neurology, Vancouver, Canada, 10Imperial College Healthcare NHS Trust, Charing Cross Hospital, Stroke Medicine, London, United Kingdom, 11Rhode Island Hospital, Brown University, Neurology, Providence, United States, 12Cooper Neurological Institute, Cooper University Hospital, Neurology, Camden, United States, 13Hospital de Santa Maria - CHULN, Neurology, Lisbon, Portugal, 14Hospital de Egas Moniz, Centro Hospitalar Lisboa Ocidental, Neurology, Lisbon, Portugal, 15Leuven University Hospital, Neurology, Leuven, Belgium, 16University of Tennessee Health Science Center, Neurology, Tennessee, United States, 17University of Colorado School of Medicine, Neurology, Colorado, United States, 18St George’s University Hospital, London, United Kingdom, 19Erasmus MC, University Medical Center Rotterdam, Neurology, Radiology and Nuclear Medicine, Rotterdam, Netherlands, 20University of L’Aquila, Department of Biotechnological and Applied Clinical Sciences, L’Aquila, Italy, 21Akershus University Hospital, Neurology, Oslo, Norway, 22University of Florida, Gainesville, United States, 23Attikon University Hospital, National and Kapodistrian University of Athens, Neurology, Athens, Greece, 24University Clinic of Neurology, University Ss. Cyril and Methodius-Faculty of Medicine, Skopje, North Macedonia, The Republic of, 25Hillel Yaffe Medical Center, Neurology, Hadera, Israel, 26Complejo Hospitalario Universitario de Albacete, Albacete, Spain, 27Heidelberg University Hospital, Neurology, Heidelberg, Germany, 28Eberhard-Karls University, Neurology and Stroke, Tuebingen, Germany, 29Hôpital Lariboisière, Assistance Publique - Hôpitaux de Paris, Université Diderot, Neurologie, Paris, France, 30Hospital das Clínicas, São Paulo University, Neurology, São Paulo, Brazil, 31Royal North Shore Hospital, Sydney, Australia, 32Ohio Health Riverside Methodist Hospital, Neurology, Riverside, United States, 33Amita Health and University of Illinois-Chicago, Neurology, Chicago, United States, 34UT Houston, Neurology, Houston, United States, 35Bon Secours Mercy Health St Vincent Hospital, Neurology, Toledo, United States, 36National University of Malaysia, Kuala Lumpur, Malaysia, 37Lahore General Hospital, Lahore, Pakistan, 38County Clinic Hospital, Faculty of Medicine, Transilvania University, Neurology, Brasov, Romania, 39University of Siena, Siena, Italy, 40Faculty Hospital Trnava, Trnava, Slovakia, 41Bach Mai Hospita, Neurology, Hanoi, Vietnam, 42Christchurch Hospital, Neurology, Christchurch, New Zealand, 431. Wellington Regional Hospital, Capital and Coast District Health Board, 2. University of Otago, Medicine, Wellington, New Zealand, 44Lausanne University Hospital and University of Lausanne, Neurology, Lausanne, Switzerland, 45Erasmus MC, University Medical Center Rotterdam, Neurology, Rotterdam, Netherlands, 46University of Pittsburgh Medical Center, Neurology, Pittsburgh, United States
During the first pandemic year, CVT mortality was higher in patients who were COVID positive compared to COVID negative patients (8/53(15.0%) vs. 41/910 (4.6%), p=0.004). There was an increase in CVT mortality during the first 5 months of pandemic years 2020 and 2021 compared to the first 5 months of the pre-pandemic year 2019 (2019 vs. 2020: 2.26% vs. 4.74%, p=0.05; 2019 vs. 2021: 2.26% vs. 4.99%, p=0.03). In the first 5 months of 2021, there were 26 cases of VITT, resulting in 6 deaths.
Dr. Blasco: speaker and CEC fees from Stryker and Medtronic, respectively.
Dr. Bolognese: participation in the advisory board (AstraZeneca) and speaker fee (Roche).
Dr Dippel: unrestricted grants for research, paid to institution, from Stryker, Medtronic, Cerenovus, Penumbra.
Dr. Falup-Pecurariu: royalties from Springer Nature Publishing Group and Elsevier, Research Grant from Transilvania University Brasov, Speaker fees and honoraria from International Parkinson and Movement Disorders Society, AbbVie.
Dr. Field: in-kind study medication from Bayer Canada, consultation fees from HLS Therapeutics and is on the board of Destine Health.
Dr. Linfante: consulting fees from Penumbra, Medtronic, Stryker, Microvention, InNeuroCo, and Three Rivers.
Dr Michel: grants from Swiss National Science Foundation and Swiss Heart Foundation.
Dr Mikulik, Dr. Sedova: project No. CA18118, IRENE COST Action, by the IRIS-TEPUS Project No. LTC20051 from
the INTER-EXCELLENCE INTER-COST Program of the Ministry of Education.
Dr Nagel: consultancy for Brainomix; speaker bureaus with Boehringer Ingelheim and Pfizer.
Dr Nguyen: research support Medtronic and SVIN (related).
Dr Nogueira: consulting fees for advisory roles with Anaconda, Biogen, Cerenovus, Genentech, Hybernia, Imperative Care, Medtronic, Phenox, Philips, Prolong Pharmaceuticals, Stryker, Shanghai Wallaby, and Synchron.
Dr Ortega-Gutierrez: consultant for Medtronic, Stryker Neurovascular; received grants from Stryker, IschemiaView, Viz.ai, and Siemens.
Dr Siegler: consulting fees (Ceribell), speakers’ bureau (AstraZeneca)
Dr. Yamagami: research grants (Bristol-Myers Squibb), lecturer’s fees (Bayer, Daiichi-Sankyo, Stryker), advisory boards (Daiichi-Sankyo).
Dr Zaidat: research grants from Stryker, Medtronic, Cerenovus, Penumbra, and Genentech.
1680
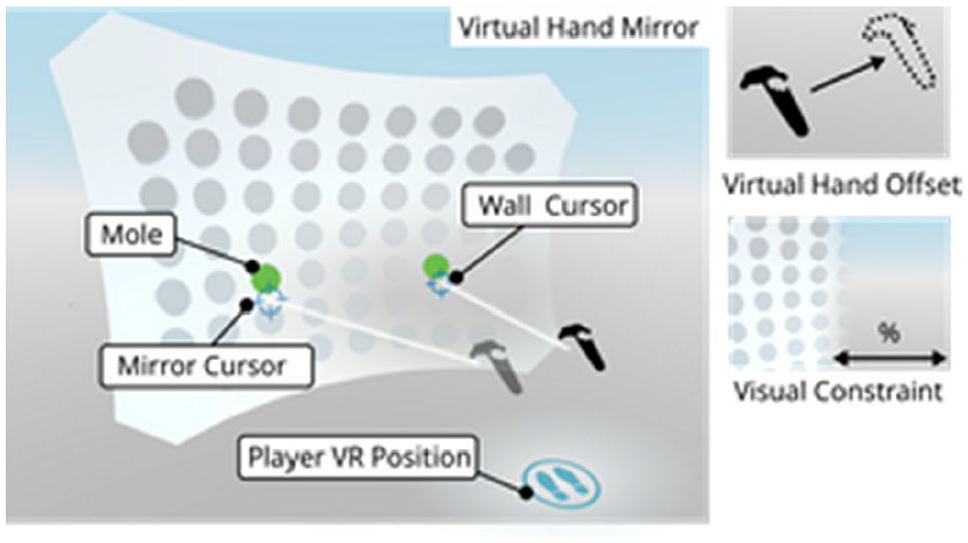
VIRTUAL MIRROR THERAPY IN A VR POINTING TASK FOR STROKE REHABILITATION
1Aalborg University, Department of Architecture and Media Technology, Aalborg, Denmark, 2Hammel Neurorehabilitation Centre and University Research Clinic, Hammel, Denmark
1681
THE LAC UNCAR INTERVENTION TRIAL 2 (LACI-2). A PROPECTIVE RANDOMISED OPEN LABEL BLINDED ENDPOINT PARTIAL FACTORIAL TRIAL TESTING CILOSTAZOL AND/OR ISOSORBIDE MONONITRATE IN LACUNAR STROKE
1 University of Edinburgh, Edinburgh Imaging, Edinburgh, United Kingdom
1683
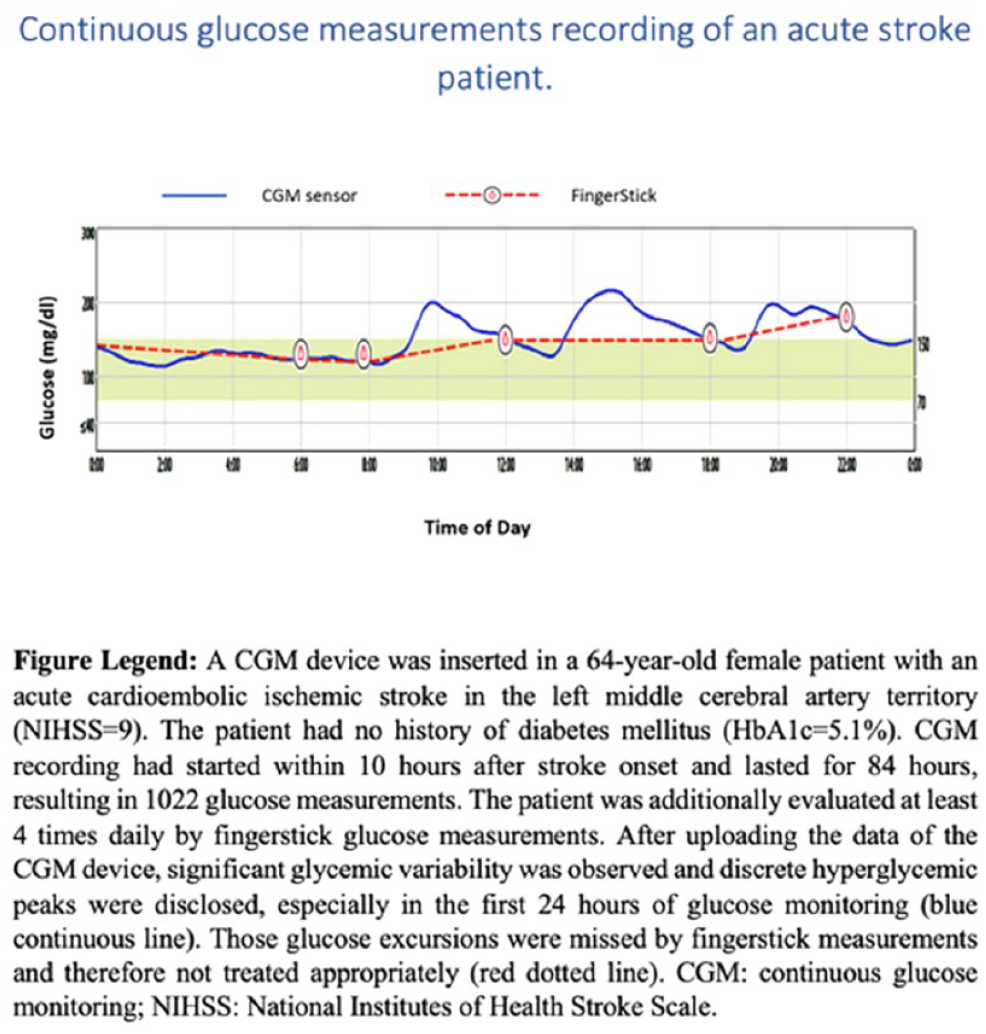
GLYCEMIC VARIABILITY OF ACUTE STROKE PATIENTS AND CLINICAL OUTCOMES: A CONTINUOUS GLUCOSE MONITORING STUDY
1National and Kapodistrian University of Athens, Athens, Greece, Second Department of Neurology, School of Medicine, University General Hospital ‘Attikon’, Athens, Greece, 2Harvard Medical School, Boston, MA, USA, Department of Neurology, Beth Israel Deaconess Medical Center, Boston, United States, 3National and Kapodistrian University of Athens, Athens, Greece, Second Department of Internal Medicine-Propaedeutic and Diabetes Center, Medical School, University General Hospital ‘Attikon’, Athens, Greece, 4Pediatric Hospital of Athens, Agia Sophia, Athens, Greece, Department of Neurosurgery, Athens, Greece, 5National and Kapodistrian University of Athens, Athens, Greece, First Department of Psychiatry, Aiginition Hospital, School of Medicine, Athens, Greece
1685
BEST REVASCULARISATION APPROACH FOR POSTERIOR CIRCULATION STROKES WITH ISOLATED VERTEBRAL ARTERY OCCLUSION: DESIGN OF THE “BRAVO” STUDY
1Lausanne University Hospital and University of Lausanne, Stroke Center, Department of Clinical Neurosciences, Lausanne, Switzerland, 2Lausanne University Hospital and University of Lausanne, Department of Diagnostic and Interventional Radiology, Lausanne, Switzerland
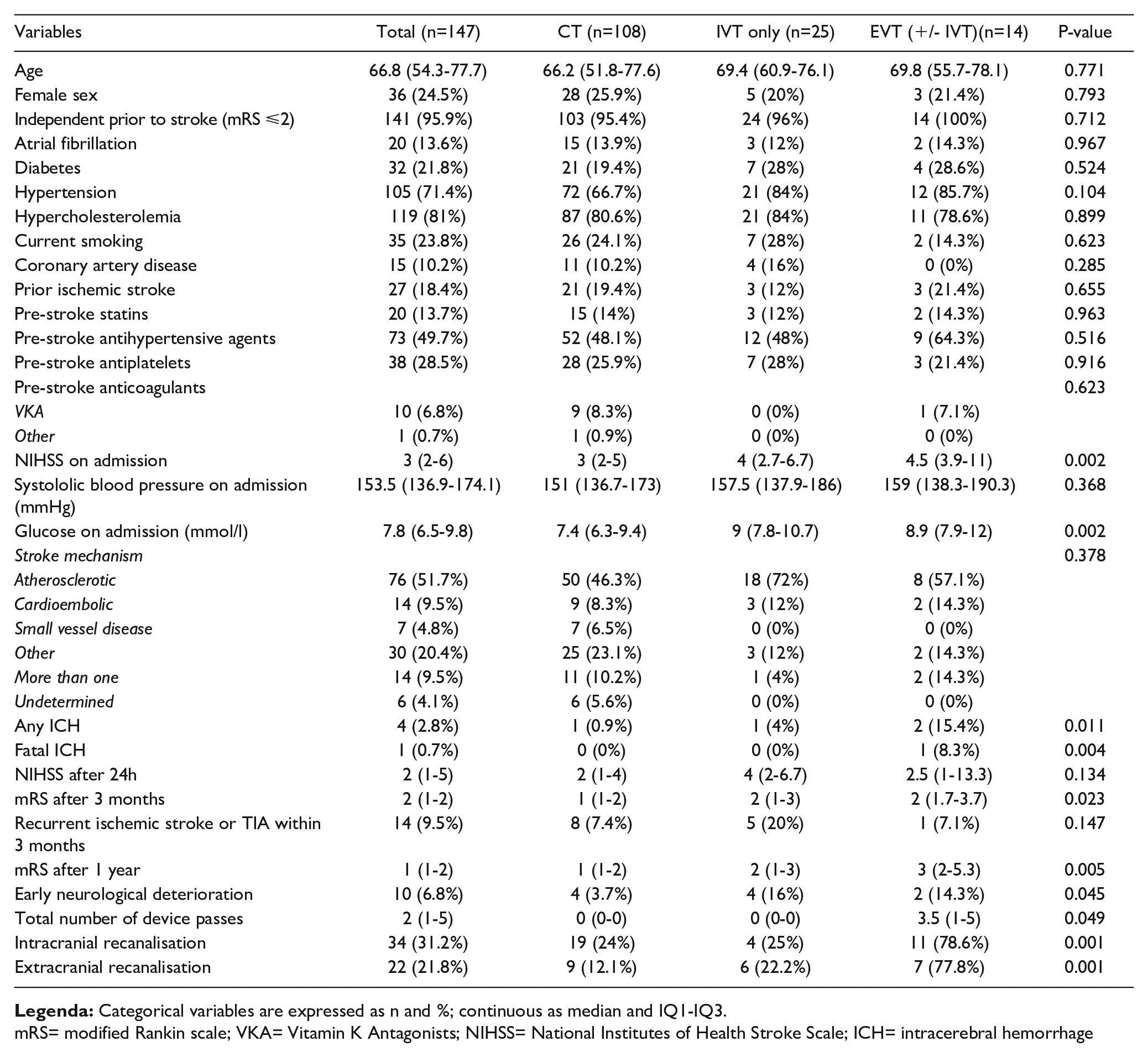
mRS= modified Rankin scale; VKA= Vitamin K Antagonists; NIHSS= National Institutes of Health Stroke Scale; ICH= intracerebral hemorrhage
1686
DRIP, SHIP, RETRIEVE BUT DON’T LEAVE: HOW OFTEN DO REGIONAL REPATRIATION PATHWAYS WORK AS INTENDED?
1 Royal Victoria Hospital, Comprehensive Stroke Centre, Belfast, United Kingdom
1687
GLOBAL IMPACT OF THE COVID19 PANDEMIC ON SUBARACHNOID HEMORRHAGE ADMISSIONS, ANEURYSM TREATMENT AND IN-HOSPITAL MORTALITY: 1 YEAR FOLLOW-UP
1Boston University School of Medicine, Boston Medical Center, Neurology, Radiology, Boston, United States, 2Boston Medical Center, Radiology, Radiation Oncology, Boston, United States, 3Boston University School of Medicine, Boston, United States, 4International Clinical Research Centre, St Anne’s University Hospital and Faculty of Medicine, Masaryk University, Neurology, Brno, Czech Republic, 5National Hospital Organization, Osaka National Hospital, Stroke Neurology, Osaka, Japan, 6Mannheim University Hospital, Neurosurgery, Mannheim, Germany, 7Boston Medical Center, Boston University School of Medicine, Radiology, Boston, United States, 8Akershus University Hospital, University of Oslo, Oslo, Norway, 9Alexandria University Stroke and Neurointervention Unit, Neurology, Alexandria, Egypt, 10Leuven University Hospital, Neurology, Leuven, Belgium, 11Turku University Hospital, Radiology, Turku, Finland, 12Université de Paris, Radiology, Paris, France, 13Heidelberg University Hospital, Radiology, Heidelberg, Germany, 14University of Siena, Neurology, Siena, Italy, 15Luzerner Kantonsspital, Luzerne, Switzerland, 16Lahore General Hospital, Lahore, Pakistan, 17Bach Mai Hospital, Neurology, Hanoi, Vietnam, 18Transilvania University, Faculty of Medicine, Brasov, Romania, 19Royal North Shore Hospital, Sydney, Australia, 20University of Tennessee Health Science Center, Neurology, Tennessee, United States, 21Spectrum Health and Michigan State University College of Human Medicine, Neurosciences, Michigan, United States, 22University of Massachusetts Medical Center, Radiology, Worcester, United States, 23University Hospital/ Goethe University, Neurology, Frankfurt, Germany, 24Nottingham University Hospitals NHS Trust, Neuroradiology, Nottingham, United Kingdom, 25Rush University, Chicago, United States, 26University Hospital St Ivan Rilski, Radiology, Sofia, Bulgaria, 27St George’s University Hospital, London, United Kingdom, 28Centre Hospitalier de l’Universite de Quebec, Neurochirurgie, Quebec, Canada, 29Rede Mater Dei de Saúde, Interventional Neuroradiology, Belo Horizonte, Brazil, 30University Hospital Zurich, Zurich, Switzerland, 31National institute of Neurology Tunis, Tunis, Tunisia, 32Grant Medical College and Sir JJ Hospital, Mumbai, India, 33Instituto de Neurocirugia Dr. Asenjo, Radiology, Santiago, Chile, 34Royal London Hospital, Barts Health NHS Trust, London, United Kingdom, 35Sestre Milosrdnice University Hospital Center, Zagreb, Croatia, 36Ohio Health Riverside Methodist Hospital, Riverside, United States, 37University of Kansas Medical Center, Kansas, United States, 38Hue Central Hospital, Hue, Vietnam, 39Institute of Medical Sciences, Medical College of Rzeszow University, Rzeszow, Poland, 40Specialist Hospital Konskie, Collegium Medicum, Jan Kochanowski University, Kielce, Poland, 41Hôpitaux Universitaires de Genève, Radiology, Geneva, Switzerland, 42Miami Cardiac & Vascular Institute, Miami Neuroscience Institute, Miami, United States, 43Royal University Hospital, Neurosurgery, Saskatoon, Canada, 44Cleveland Clinic Abu Dhabi, Abu Dhabi, United Arab Emirates, 45Attikon University Hospital, National and Kapodistrian University of Athens, Neurology, Athens, Greece, 46Heidelberg University Hospital, Neurology, Heidelberg, Germany, 47University of Pittsburgh Medical Center, Neurology, Pittsburgh, United States
This study is registered NCT04934020.
Dr. Falup-Pecurariu reported royalties from Springer Nature Publishing Group and Elsevier, Research Grant from Transilvania University Brasov, Speaker fees and honoraria from International Parkinson and Movement Disorders Society, AbbVie, outside the submitted work.
Dr Mikulik was supported by project No. CA18118, IRENE COST Action funded by COST Association, by the IRIS-TEPUS Project No. LTC20051 from the INTER-EXCELLENCE INTER-COST Program of the Ministry of Education, Youth and Sports of the Czech Republic, and by STROCZECH within CZECRIN Large Research Infrastructure No. LM2018128 funded by the state budget of the Czech Republic
Dr Nogueira reported consulting fees for advisory roles with Anaconda, Biogen, Cerenovus, Genentech, Hybernia, Imperative Care, Medtronic, Phenox, Philips, Prolong Pharmaceuticals, Stryker Neurovascular, Shanghai Wallaby, and Synchron and stock options for advisory roles with Astrocyte, Brainomix, Cerebrotech, Ceretrieve, Corindus Vascular Robotics, Vesalio, Viz-AI, RapidPulse, and Perfuze; and investments in Viz-AI, Perfuze, Cerebrotech, Reist/Q’Apel Medical, Truvic, and Viseon.
Dr. Ajit S. Puri is a consultant for Cerenovus, CereVasc, Merit and Medtronic; research grant from Medtronic. Stocks in InNeuroCo, Galaxy, Agile, Perfuze, and NTI. Dr. Yamagami reported research grants from Bristol-Myers Squibb, lecturer’s fees from Bayer, Daiichi-Sankyo, Stryker, and membership of the advisory boards for Daiichi-Sankyo outside the submitted work.
1688
SECONDARY PREVENTION BY STRUCTURED SEMI-INTERACTIVE STROKE PREVENTION PACKAGE IN INDIA (SPRINT INDIA): A RANDOMIZED CONTROLLED TRIAL
1Christian Medical College & Hospital, Ludhiana, Department of Neurology, Ludhiana, India, 2University of Alberta, Department of Medicine, Canada, Canada, 3Sree Chitra Tirunal Institute for Medical Sciences and Technology, Department of Neurology, Thiruvananthapuram, India, 4Post Graduate Institute of Medical Education and Research, Department of Neurology, Chandigarh, India, 5Lalitha Super Speciality Hospital, Department of Neurology, Guntur, India, 6Bangur Institute of Neurosciences, Department of Neurology, Kolkata, India, 7Amrita Institute of Medical Sciences, Department of Neurology, Kochi, India, 8Christian Medical College, Department of Neurology, Vellore, India, 9St Stephens Hospital, Department of Neurology, Delhi, India, 10Government General Hospital, Department of Neurology, Guntur, India, 11Kasturba Medical College and Hospital, Department of Neurology, Manipal, India, 12Ramesh Hospital, Department of Neurology, Gunutr, India, 13CARE Hospitals, Department of Neurology, Hyderabad, India, 14Jawaharlal Institute of Postgraduate Medical Education and Research, Department of Neurology, Puducherry, India, 15Guwahati Neurological Research Centre, Department of Neurology, Guwahati, India, 16National Institute of Mental Health and Neurosciences, Department of Neurology, Bangalore, India, 17Mazumdar Shaw Medical Centre, Narayana Hrudayalaya, Department of Neurology, Bangalore, India, 18St John’s Medical College, Department of Neurology, Bangalore, India, 19All India Institute of Medical Sciences, Department of Neurology, New Delhi, India, 20Grant Government Medical College and Sir J.J. Group of Hospitals, Department of Neurology, Mumbai, India, 21Institute of Neurosciences, Department of Neurology, Kolkata, India, 22Baptist Christian Hospital, Department of Medicine, Tezpur, India, 23Vadodara Institute of Neurosciences, Department of Neurology, Vadodara, India, 24Zydus Hospital, Department of Neurology, Ahmedabad, India, 25Nizam’s Institute of Medical Sciences, Department of Neurology, Hyderabad, India, 26Institute of Medical Sciences, Banaras Hindu University, Department of Neurology, Varanasi, India, 27All India Institute of Medical Sciences, Department of Neurology, Bhubaneswar, India, 28Indira Gandhi Medical College & Hospital, Department of Neurology, Shimla, India, 29GGS Medical College, Department of Neurology, Faridkot, India, 30KLE Academy of Higher Education & Research, Department of Neurology, Belagavi, India, 31Fortis Escorts Hospital, Department of Neurology, Jaipur, India, 32BGS Global Hospital, Department of Neurology, Bangalore, India, 33Indian Council of Medical Research, Non-Communicable Diseases Section, New Delhi, India
Methods: SPRINT INDIA is a multicentre, randomized, parallel-design, adaptive and blinded end-point clinical trial under INSTRuCT network. First-ever stroke patients, age ⩾18years, who had a cellular device, were randomized centrally. Intervention arm received an educational workbook (12 languages), short messages and videos regularly concentrating on risk factor mitigation. Control group received standard of care. Primary outcome was a composite of recurrent stroke, high-risk transient ischemic attack, acute coronary syndrome, and death at one year. Key secondary outcome measures include medication compliance, FBS, Lipids, smoking and alcohol cessation at one year.
1692
ESTABLISHING ORGANIZED STROKE CARE IN LOW-AND-MIDDLE INCOME COUNTRIES: FROM TRAINING OF NON-SPECIALIST TO IMPLEMENTATION (ON BEHALF OF WORLD STROKE ORGANISATION-FUTURE STROKE LEADERS)
1St. Stephen’s Hospital, Neurology, Delhi, India, 2All India Institute of Medical Sciences, Neurology, Delhi, India, 3University of Southern California, California, United States, 4National Institute of Health, Yerevan, Armenia, Neurology, Yerevan, Armenia, 5Pusat Perubatan Universiti Kebangsaan, Malaysia, Neurology, Malaysia, Malaysia, 6University of Mauritius, Mauritius, Mauritius
This ongoing project aims to:
Build capacity among non-specialists through a set of training modules on evidence-based stroke-care
Develop a mobile Application (Android and iOS) to deliver and monitor stroke-care
Implement and assess the efficacy of the non-specialist model of stroke-care
Evaluate the effectiveness of the virtual training modules for capacity building, by assessing a pre-and-post knowledge, attitude and practices (KAP) of the non-specialists in training.
Evaluate the effectiveness of the non-specialist driven model of stroke-care,by assessing and comparing pre-and-post implementation data on Key quality indicators collected from the hospitals.(Table1)
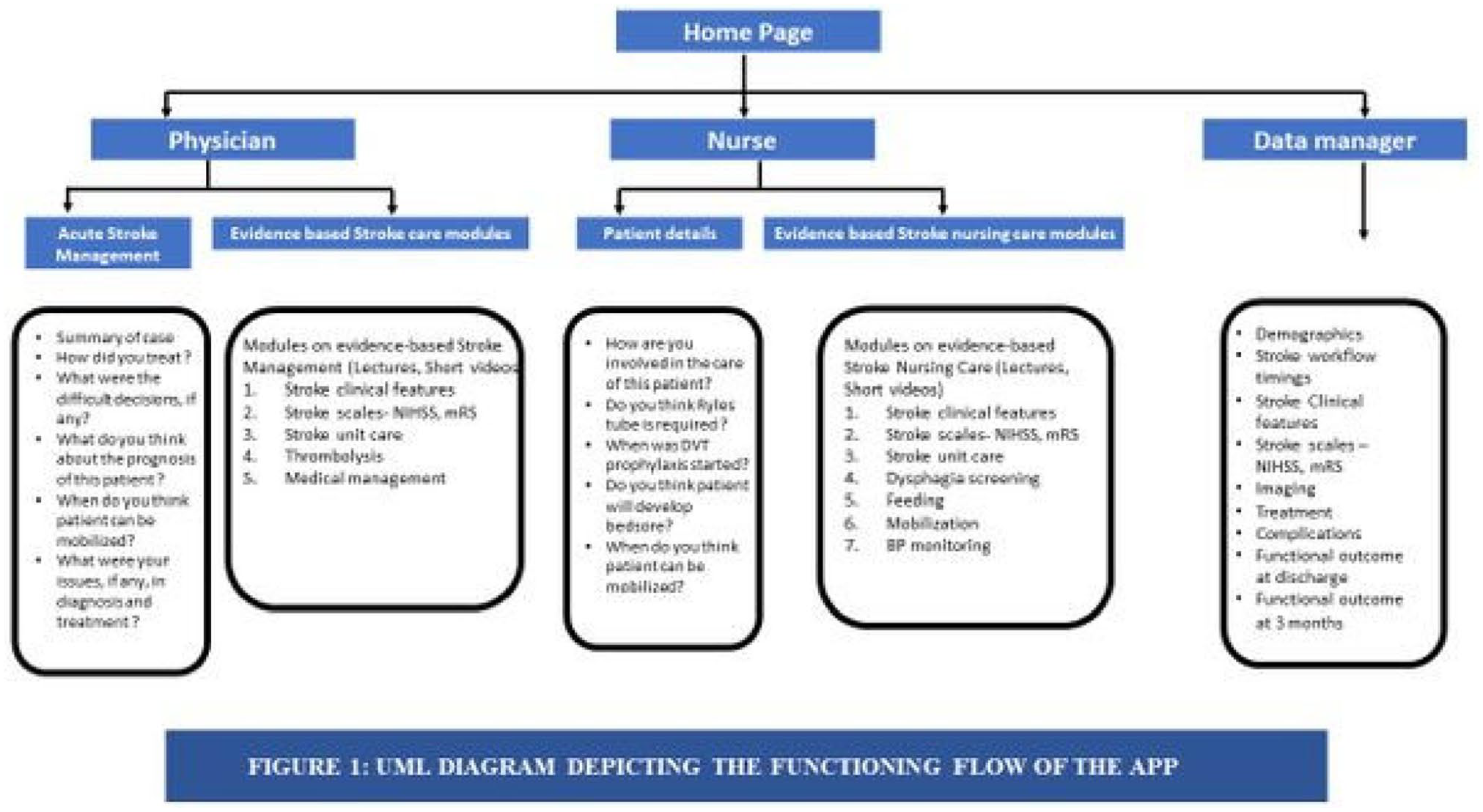
Key Quality Indicators.

1694
FUNCTIONAL RECOVERY FOLLOWING THROMBECTOMY IN THE LATE WINDOW WITH PRE-STROKE DISABILITY: A COLLABORATION OF THE CLEAR AND RESCUE-JAPAN INVESTIGATORS
1Cooper University Hospital, Cooper Neurological Institute, Camden, United States, 2Boston Medical Center, Boston, United States, 3Grady Memorial Hospital, Atlanta, United States, 4National Cerebral and Cardiovascular Center, Suita, Japan, 5Heidelberg University Hospital, Heidelberg, Germany, 6Universite de Lausanne, Lausanne, Switzerland, 7Hospital Vall d’Hebron, Barcelona, Spain, 8NHO Osaka National Hospital, Osaka, Japan, 9Hyogo College of Medicine, Nishinomiya, Japan, 10Boston Medical Center, Department of Neurology, Boston, United States, 11University of Cambridge, Cambridge, United Kingdom, 12University of Lille, Lille, France, 13University of Texas Health Sciences Center, Houston, United States, 14University of Iowa Hospitals and Clinics, Iowa City, United States, 15University of Toledo, Toledo, United States, 16Kobe City Medical Center General Hospital, Kobe, Japan, 17University of Bern, Bern, Switzerland, 18University of Massachusetts Memorial Medical Center, Worcester, United States, 19Seisho Hospital, Odawara, Japan, 20Centre Hospitalier de l’Université de Montréal, Montréal, Canada, 21SUNY New York Upstate Medical University, Syracuse, United States, 22Yokohama Shintoshi Neurosurgical Hospital, Yokohama, Japan, 23Ise Red Cross Hospital, Ise, Japan, 24Sapporo Shiroishi Memorial Hospital, Sapporo, Japan, 25University Hospital Basel, Basel, Switzerland
1703
SPECIALIZED PRO-RESOLVING MEDIATOR AND LIPID SIGNATURE IN SUBARACHNOID HAEMORRHAGE
1Amsterdam UMC (VUmc), Dept. Molecular Cell Biology and Immunology, Amsterdam, Netherlands, 2Amsterdam UMC (AMC), Dept. Neurosurgery, Amsterdam, Netherlands, 3Amsterdam UMC (AMC), Dept. Biomedical Engineering and Physics, Amsterdam, Netherlands, 4Amsterdam UMC (AMC), Dept. Neurology, Amsterdam, Netherlands, 5Leiden University Medical Center, Center for Proteomics and Metabolomics, Leiden, Netherlands
1705
NOVEL ROBOTIC-ASSISTED TRANSCRANIAL DOPPLER VERSUS TRANSTHORACIC ECHOCARDIOGRAPHY TO DETECT RIGHT-TO-LEFT SHUNTS
1University of Tennessee Health Science Center, Neurology, Memphis, United States, 2CHI Memorial, Chattanooga, United States, 3Barrow Neurological Institute, Phoenix, United States, 4Houston Methodist, Houston, United States, 5Swedish Memorial, Seattle, United States, 6Providence, Portland, United States, 7National and Kapodistrian University of Athens, Athens, Greece, 8University of Tennessee Health Science Center, Memphis, United States
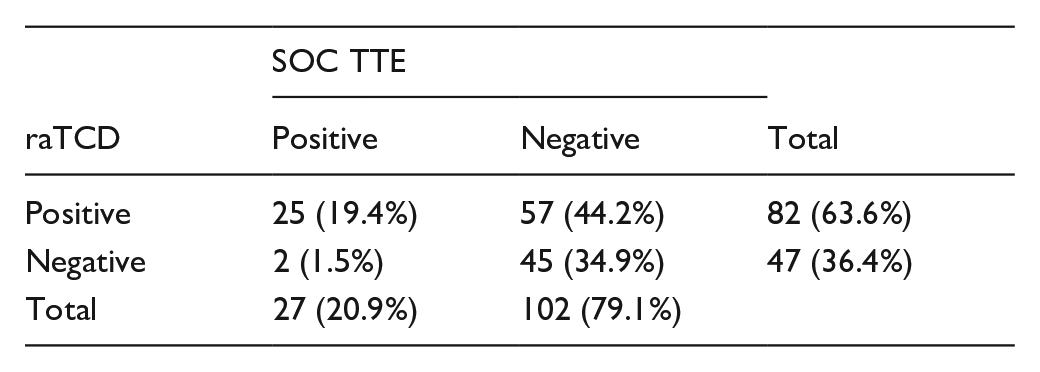

1706
MORTALITY PREDICTION AMONG PATIENTS WITH ACUTE ISCHEMIC STROKE USING MACHINE LEARNING ALGORITHMS: ANALYSIS OF THE MIMIC-IV DATABASE
1Changhua Christian Hospital, Department of Neurology, Changhua, Taiwan, 2Kaohsiung Medical University, School of Medicine, Kaohsiung, Taiwan, 3National Defense Medical Center, Tri-Service General Hospital, Department of Internal Medicine, Taipei, Taiwan, 4National Tsing Hua University, Department of Computer Science, Hsinchu, Taiwan, 5National Formosa University, 5. Department of Computer Science and Information Science, Yunlin, Taiwan, 6Tri-Service General Hospital, National Defense Medical Center, Department of Surgery, Taipei, Taiwan
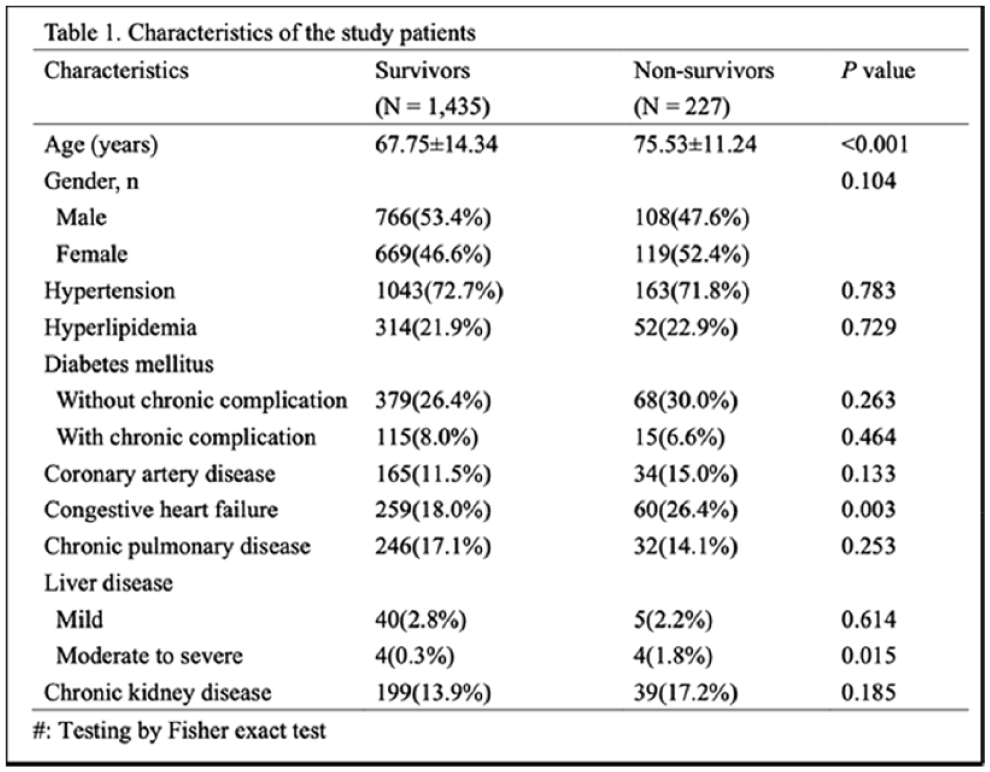
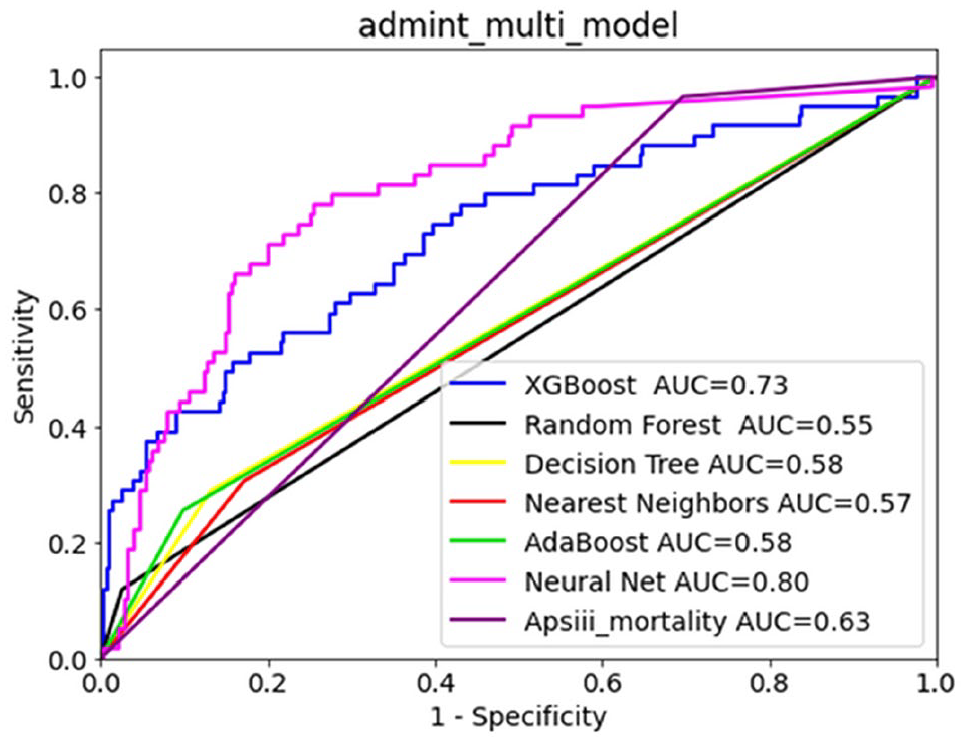
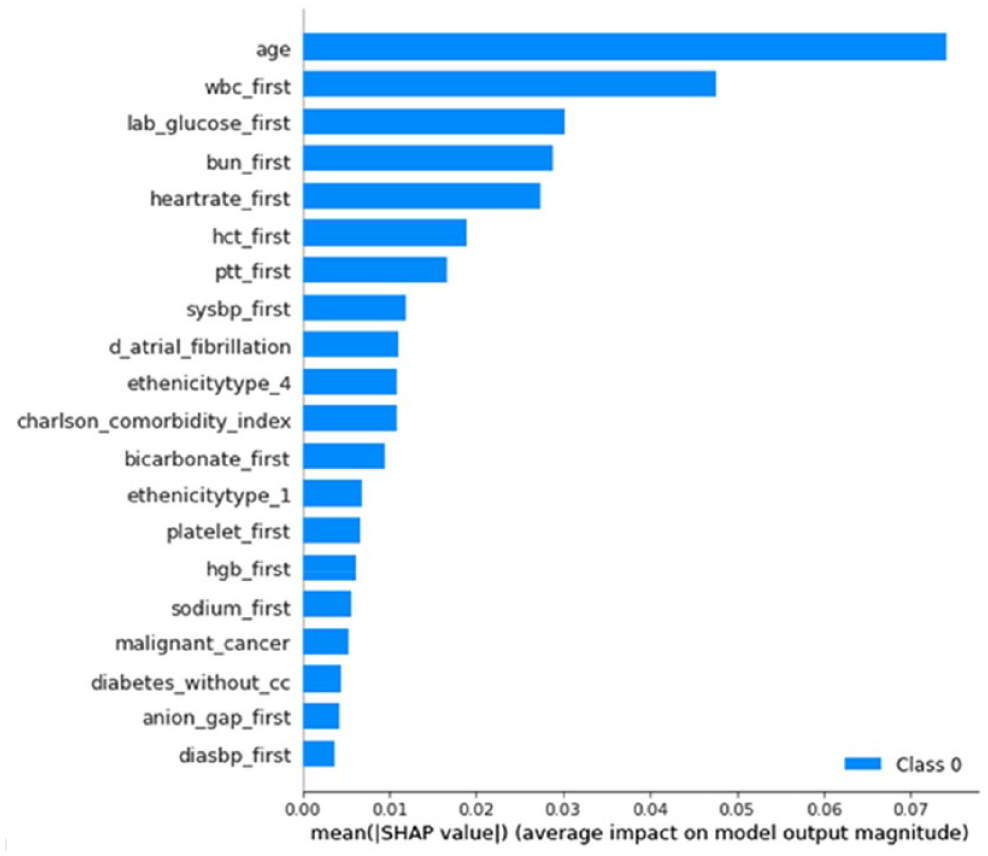
1707
INTRA-ARTERIAL BONE MARROW MONONUCLEAR CELLS (BM-MNCS) TRANSPLANTATION IN ACUTE ISCHEMIC STROKE (IBIS TRIAL). A PHASE IIB RANDOMIZED,DOSE-FINDING, SINGLE-BLIND CONTROLLED MULTICENTER TRIAL
1Hospital Universitario Virgen del Rocio, Stroke Unit, Seville, Spain, 2Instituto de Biomedicina de Sevilla-IBiS, Neurovascular Lab, Sevilla, Spain, 3Hospital Reina Sofia, Stroke Unit, Cordoba, Spain, 4Hospital Universitario Virgen del Rocio, Hematology Department, Seville, Spain, 5Hospital Universitario Virgen del Rocio, Interventional Neuroradiology, Seville, Spain, 6Hospital Universitario Virgen Macarena, Stroke Unit, Seville, Spain, 7Hospital Universitario Puerta del Mar, Stroke Unit, Cadiz, Spain, 8Hospital Universitario Virgen del Rocio, Radiology Department, Seville, Spain
1709
FOUR-MONTH TREATMENT WITH NEW ANTIDIABETIC AGENTS (GLP-1/SGLT-2I) IMPROVES CARDIOVASCULAR FUNCTION IN PATIENTS WITH TYPE 2 DIABETES MELLITUS AND ISCHEMIC STROKE
1Attikon University Hospital, Second Department of Internal Medicine, Medical School, National and Kapodistrian University of Athens, Athens, Greece, 2Attikon University Hospital, Second Cardiology Department, Laboratory of Preventive Cardiology, Medical School, National and Kapodistrian University of Athens, Athens, Greece, 3Attikon University Hospital, Second department of Neurology, Medical School, National and Kapodistrian University of Athens, Athens, Greece
1711
THE EFFECTIVENESS OF ADDITIONAL CORE STABILITY EXERCISES IN IMPROVING DYNAMIC SITTING BALANCE, COORDINATION AND LOWER LIMB SPASTICITY FOR SUBACUTE STROKE-SURVIVORS (CORE-TRIAL). A RANDOMIZED CONTROLLED TRIAL
1Universitat Internacional de Catalunya, Physical Therapy, Sant Cugat del Valles, Spain, 2Hospital Universitari de Vic, Rehabilitation, Vic, Spain, 3Germanes Hospitalaries Sagrat Cor, Rehabilitation, Martorell, Spain, 4Hospital de Tarrassa, Rehabilitation, Terrassa, Spain, 5Centre Fòrum | Consorci MAR Parc de Salut de Barcelona, Rehabilitation, Barcelona, Spain, 6Hospital Sociosanitari Mutuam Girona, Rehabilitation, Girona, Spain, 7Parc Taulí Hospital Universitari, Rehabilitation, Sabadell, Spain, 8Centro Cochrane Iberoamericano - Institut d’Investigació Biomèdica Sant Pau (IIB Sant Pau), CIBERESP, Public Health, Barcelona, Spain
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee Research of each hospital. It was funded by Fundació Marató TV3, grant number 201737-83. Protocol registration is in ClinicalTrials.gov number Identifier NCT03975985.
Patients Characteristics at baseline (n=87).
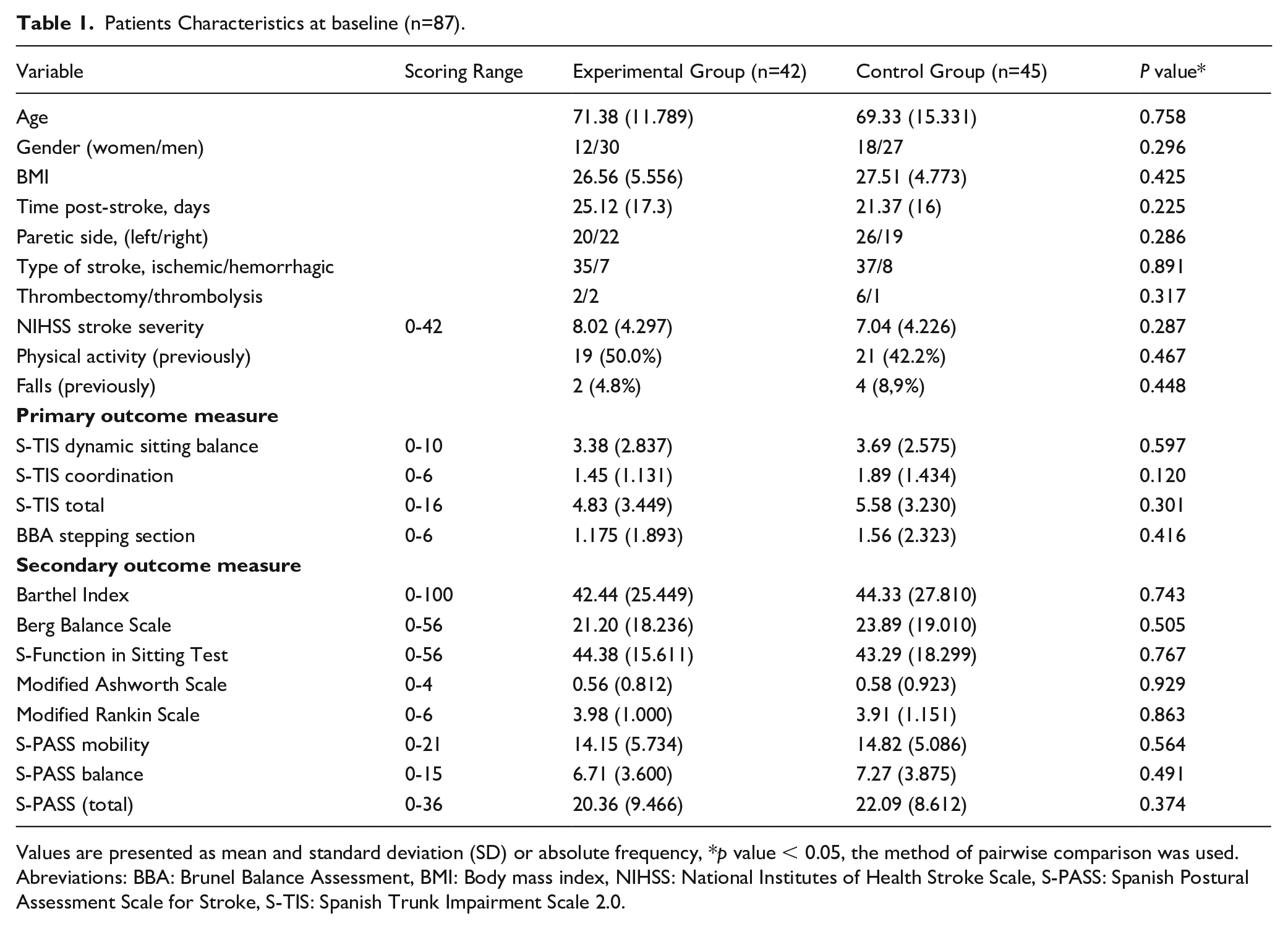
Values are presented as mean and standard deviation (SD) or absolute frequency, *p value < 0.05, the method of pairwise comparison was used. Abreviations: BBA: Brunel Balance Assessment, BMI: Body mass index, NIHSS: National Institutes of Health Stroke Scale, S-PASS: Spanish Postural Assessment Scale for Stroke, S-TIS: Spanish Trunk Impairment Scale 2.0.
Post-treatment values.
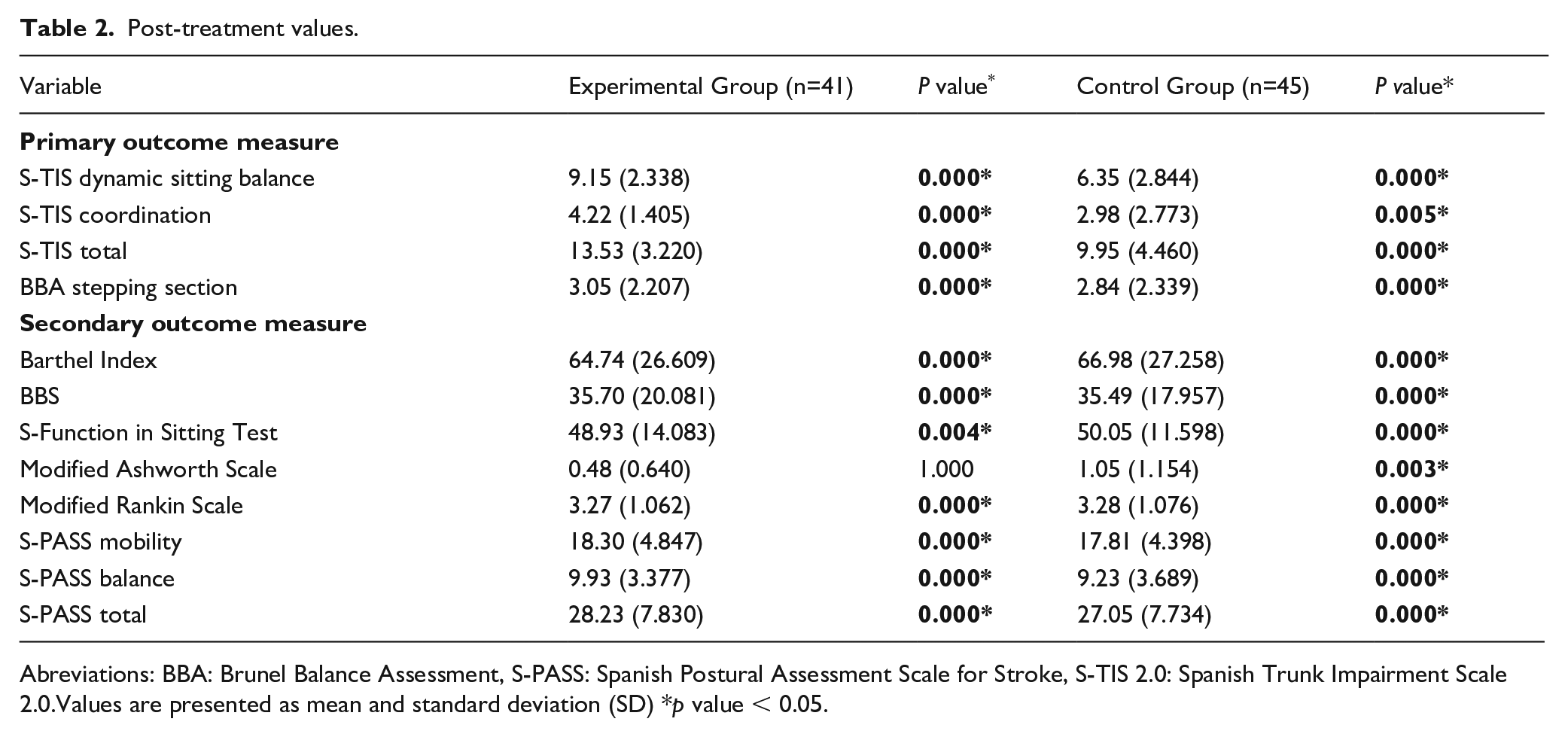
Abreviations: BBA: Brunel Balance Assessment, S-PASS: Spanish Postural Assessment Scale for Stroke, S-TIS 2.0: Spanish Trunk Impairment Scale 2.0.Values are presented as mean and standard deviation (SD) *p value < 0.05.
Comparison between experimental and control groups post-treatment.
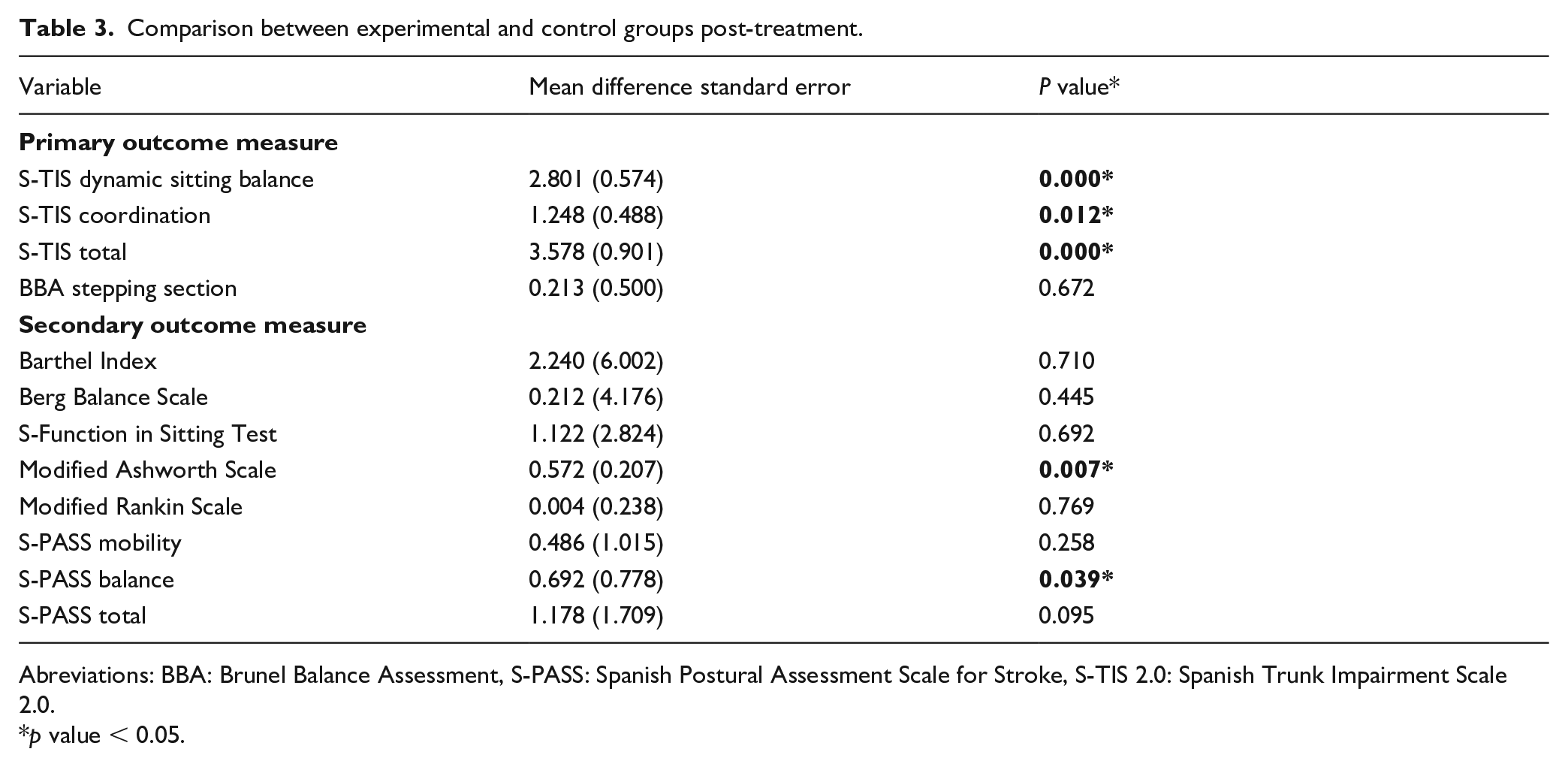
Abreviations: BBA: Brunel Balance Assessment, S-PASS: Spanish Postural Assessment Scale for Stroke, S-TIS 2.0: Spanish Trunk Impairment Scale 2.0.
p value < 0.05.

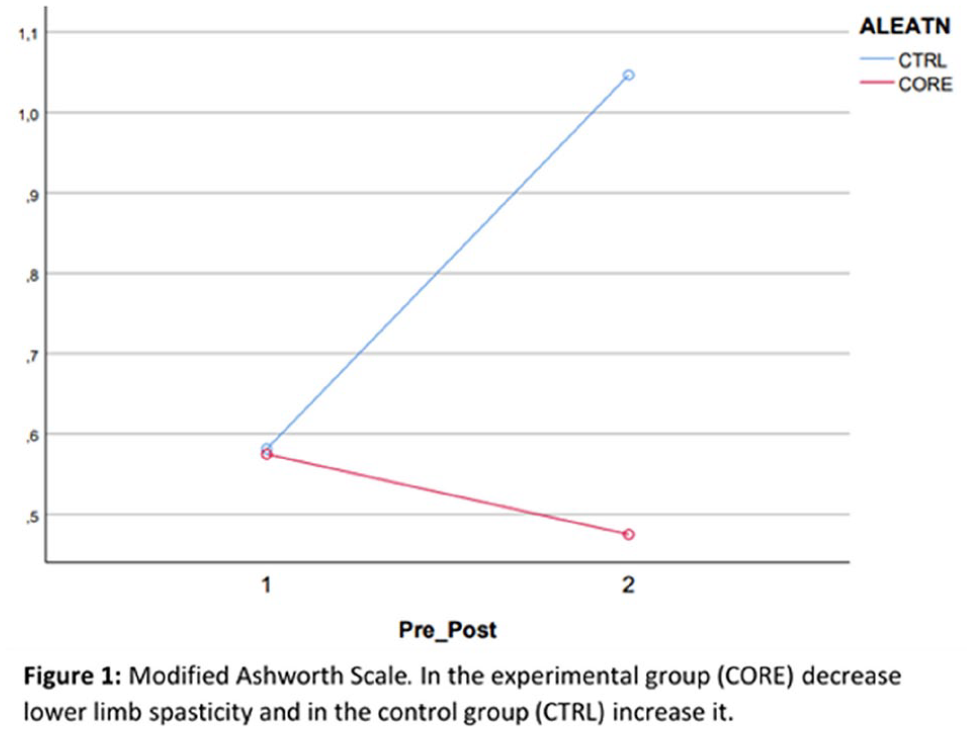
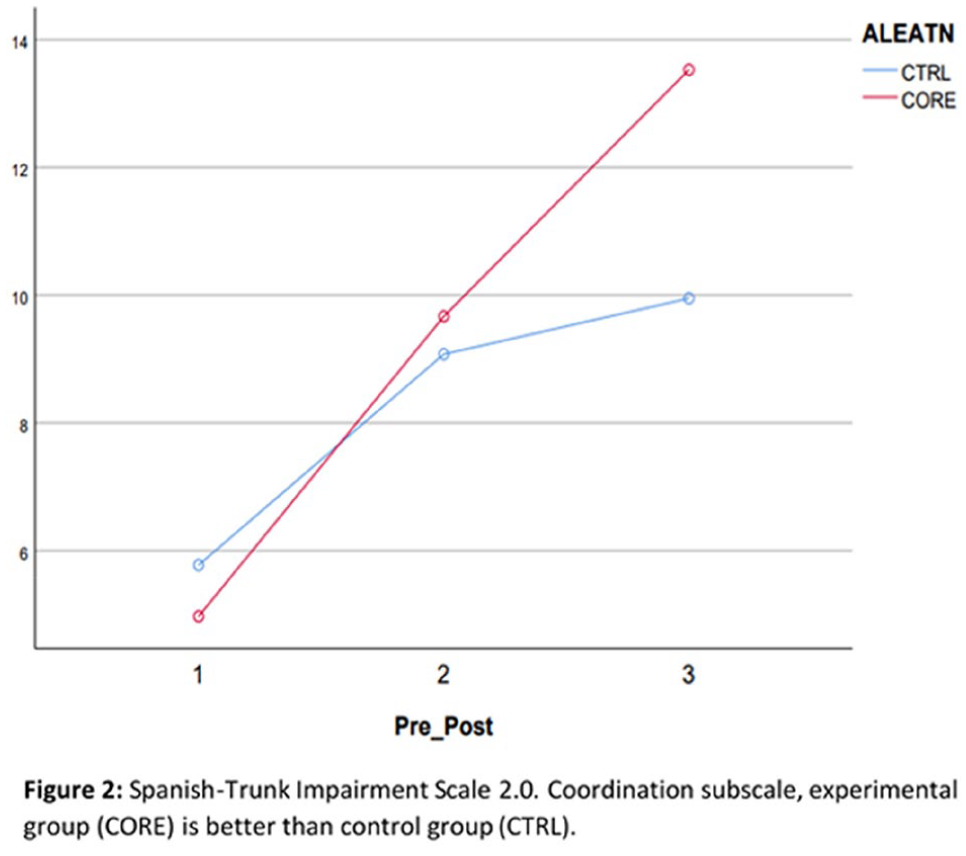
1712
ALTEPLASE COMPARED TO TENECTEPLASE (ACT) RANDOMIZED CONTROLLED TRIAL
1University of Calgary, Neurosciences, Radiology and Community Health Sciences, Calgary, Canada, 2Hotchkiss Brain Institute, Calgary, Canada, 3University of Alberta, Division of Neurology, Department of Medicine, Edmonton, Canada, 4University of Calgary, Calgary, Canada, 5Centre Hospitalier de l’Université de Montréal (CHUM), Montreal, Canada, 6Université de Montréal, Montreal, Canada, 7Sunnybrook Health Science Center, Toronto, Canada, 8University of Toronto, Toronto, Canada, 9Queen’s University, Division of Neurology, Department of Medicine, Kingston, Canada, 10Universite’ de Sherbrooke, Sherbrooke, Canada, 11Queen Elizabeth Health Sciences Center, Halifax, Canada, 12Kelowna General Hospital, Kelowna, Canada, 13Hamilton Health Sciences Center, Hamilton, Canada, 14McMaster University, Hamilton, Canada, 15University of Ottawa, Department of Medicine, Ottawa, Canada, 16Ottawa Heart Research Institute, Ottawa, Canada, 17University of British Columbia, Fraser Health Authority, New Westminster, Canada, 18London Health Sciences Center, London, Canada, 19Western University, London, Canada, 20Toronto Western Hospital, Toronto, Canada, 21University of Manitoba, Winnipeg, Canada, 22Queen Elizabeth Hospital, Charlottetown, Canada, 23University of British Columbia, Vancouver Stroke Program, Division of Neurology, Vancouver, Canada, 24Medicine Hat Regional Hospital, Medicine Hat, Canada, 25Gray Nuns Community Hospital, Edmonton, Canada, 26St. Michael’s Hospital, Toronto, Canada, 27Red Deer Regional Hospital, Red Deer, Canada, 28University of Saskatchewan, Saskatoon, Canada, 29Centre de recherche du CHUS, Centre Intégré Universitaire de Santé et des Services Sociaux de l’Estrie, Sherbrooke, Canada
1713
PRESENT STATUS OF THE STRUCTURED AMBULATORY POST-STROKE CARE PROGRAM (SANO) – A CLUSTER-RANDOMIZED MULTIMODAL POST-STROKE CARE INTERVENTION TRIAL
1University of Wuerzburg, Institute for Clinical Epidemiology and Biometry, Wuerzburg, Germany, 2Klinikum Ludwigshafen am Rhein, Ludwigshafen am Rhein, Germany, 3Charité Berlin, Department of Neurology with Experimental Neurology, Berlin, Germany, 4Bezirkskrankenhaus Günzburg, Günzburg, Germany, 5University Medical Center Würzburg, Würzburg, Germany, 6Bayerischer Hausärzteverband, Würzburg, Germany, 7University Medical Center Mainz, Mainz, Germany, 8Benedictus Krankenhaus Tutzing, Tutzing, Germany, 9University Hospital Basel, Basel, Switzerland, 10Imperial College London, Department of Brain Sciences, London, United Kingdom, 11German Stroke Society, Berlin, Germany, 12Universitätsklinikum Jena, Institut für Medizinische Statistik, Informatik und Datenwissenschaften, Jena, Germany, 13Institut für Arbeit und Technik, Gelsenkirchen, Germany
1715
A MULTIMODAL INDIVIDUALIZED INTERVENTION TO PREVENT FUNCTIONAL DECLINE AFTER STROKE. A RANDOMISED CONTROLLED TRIAL ON LONG-TERM FOLLOW-UP AFTER STROKE (THE LAST-LONG TRIAL)
1NTNU-Norwegian University of Science and Technology, Department of Neuromedicine and Movement Science, Trondheim, Norway, 2Akershus University Hospital, Department of Neurology, Lørenskog, Norway, 3City of Trondheim, Department of Health and Social Services, Trondheim, Norway, 4St. Olavs Hospital, Trondheim University Hospital, Department of Stroke, Trondheim, Norway, 5Bærum Hospital, Department of Stroke, Bærum, Norway, 6NTNU-Norwegian University of Science and Technology, Regional Centre for Child and Youth Mental Health and Child Welfare, Trondheim, Norway, 7NTNU-Norwegian University of Science and Technology, Department of Public Health and Nursing, Trondheim, Norway, 8St. Olavs Hospital, Trondheim University Hospital, Department of Geriatrics, Trondheim, Norway, 9Møre and Romsdal Health Trust, Department of Medicine, Ålesund, Norway
The intervention consists of regular meetings with a community-based stroke-coordinator, who will use a checklist to assess the patients’ risk- profile within physical health and lifestyle, mobility and ADL function, cognitive function, and social function. Accordingly, the coordinator will set up an action-plan based on national guidelines aiming to achieve the treatment goals.
Mixed models will be used to evaluate differences between the groups for the primary (modified Rankin Scale) and secondary endpoints (cognition, motor function, extended ADL, self-perceived health, quality-of-life, frailty, vascular events, caregivers burden, health costs, etc.) across the 4 time points.
ClinicalTrials.gov identifier: NCT03859063
1718
A RANDOMIZED CONTROLLED TRIAL TO TEST EFFICACY AND SAFETY OF THROMBECTOMY IN STROKE WITH EXTENDED LESION AND EXTENDED TIME WINDOW (TENSION)
1University Hospital Heidelberg, Neuroradiology, Heidelberg, Germany, 2Centre hospitalo-universitaire Reims Université Reims Champagne Ardenne Hôpital Maison Blanche, Neuroradiology,, Reims, France, 3CHARLES UNIVERSITY, Faculty Hospital, Radiology, Hradec Kralove, Czech Republic, 4Comenius University’s Jessenius Faculty of Medicine and University Hospital, Radiology, Neurology, Martin, Slovakia, 5Aarhus University Hospital, Department of Neurology, Aarhus, Denmark, 6Oslo University Hospital, Rikshospitalet, Neurology, Oslo, Norway, 7Medical University Innsbruck, Department of Neuroradiology, Innsbruck, Austria, 8La Paz University Hospital, Neuroradiology, Madrid, Spain, 9University of Calgary, Alberta, Canada, 10Centre Hospitalier Lyon Sud, Service de Biostatistique, Pierre-Bénite, France, 11University Medical Center Hamburg, Neuroradiology, Hamburg, Germany, 12University Medical Center Hamburg, Neurology, Hamburg, Germany
TENSION is registered at ClinicalTrials.gov (ClinicalTrials.gov Identifier NCT03094715).
1722
PRECIOUS: PREVENTION OF COMPLICATIONS TO IMPROVE OUTCOME IN ELDERLY PATIENTS WITH ACUTE STROKE. A RANDOMISED, OPEN, PHASE III, CLINICAL TRIAL WITH BLINDED OUTCOME ASSESSMENT
1University Medical Center Utrecht, Neurology, Utrecht, Netherlands, 2University of Nottingham, Stroke Trials Unit, Division of Clinical Neuroscience, Nottingham, United Kingdom, 3Amsterdam University Medical Centers, Location AMC, Neurology, Amsterdam, Netherlands, 4Oslo University Hospital, Neurology, Oslo, Norway, 5University Medical Center Hamburg-Eppendorf, Clinical Trial Center North, Hamburg, Germany, 6ASST di Mantova, Neurology and Stroke Unit, Mantua, Italy, 7University of Debrecen, Neurology, Debrecen, Hungary, 8Network & Institut National de la Santé et de la Recherche Médicale, European Clinical Research Infrastructure, Paris, France, 9Erasmus MC, University Medical Center, Rotterdam, Department of Neurology,, Rotterdam, Netherlands, 10University of Tartu, Department of Neurology and Neurosurgery, Tartu, Estonia, 11Institute of Psychiatry and Neurology, 2nd department of Neurology, Warsaw, Poland, 12University of Glasgow, Institute of Cardiovascular and Medical sciences, Glasgow, United Kingdom, 13University of Edinburgh, Centre for Clinical Brain Sciences, Edinburgh, United Kingdom, 14Larissa University Hospital, University of Thessaly, Department of Medicine, Larissa, Greece, 15Stroke Alliance For EUROPE (SAFE), Brussels, Belgium, 16University Medical Center Hamburg-Eppendorf, Department of Neurology, Center for Clinical Neurosciences, Hamburg, Germany
1723
OPTIMAS: A RANDOMISED CONTROLLED TRIAL TO ESTABLISH THE OPTIMAL TIMING OF ANTICOAGULATION AFTER ACUTE ISCHAEMIC STROKE
1University College London, Comprehensive Clinical Trials Unit, London, United Kingdom, 2University College London, Stroke Research Centre, London, United Kingdom
On behalf of the OPTIMAS investigators.
1728
EFFECT OF TIME WINDOW ONENDOVASCULAR THROMBECTOMY WITH OR WITHOUTINTRAVENOUS THROMBOLYSISIN ACUTE ISCHEMIC STROKE: RESULTS FROM DIRECT-MT
1Nanjing First Hospital, Nanjing Medical University, Department of Neurology, Nanjing, China, 2Naval Medical University Changhai Hospital, Shanghai, China
1730
ENDO VASCULAR THERAPY FORLOW NIHSS ISCHEMIC STROKES (ENDOLOW): CLINICAL TRIAL UPDATE
1Heidelberg University Hospital, Neurology, Heidelberg, Germany, 2University of Cincinnati, Cincinnati, United States, 3University of Calgary, Calgary, Canada, 4University of Pittsburgh, Pittsburgh, United States, 5University of Heidelberg, Heidelberg, Germany, 6Emory University, Atlanta, United States, 7Cincinnati Children’s Hospital Medical Center, Cincinnati, United States
1732
A NOVEL INTERNET PLATFORM FOR STROKE RESEARCH COLLABORATION AND FUNDING: INITIAL INSIGHTS
1University of Calgary, Department of Radiology, Calgary, Canada, 2University of Calgary, Calgary, Canada, 3University of Calgary, Department of Clinical Neurosciences, Calgary, Canada, 4University Hospital Basel, Department of Neurology, Basel, Switzerland, 5University of Amsterdam, Department of Radiology and Nuclear Medicine, Amsterdam, Netherlands, 6University Hospital Basel, Department of Neuroradiology, Basel, Switzerland, 7David Geffen School of Medicine, UCLA, Department of Neurology, Los Angeles, United States
RM: CEO and shareholder of Collavidence Inc.
1734
ENDOVASCULAR TREATMENT FOR ACUTE BASILAR ARTERY OCCLUSION - A MULTICENTER RANDOMIZED CONTROLLED TRIAL (ATTENTION)
1The first affiliated Hospital of USTC, Department of Neurology, Hefei, China, 2Lehigh Valley Health Network, Allentown, United States, 3Zeenat Qureshi Stroke Research Center, Morris, United States, 4UPMC Stroke Institute, Pittsburgh, United States
1737
THE MELBOURNE MOBILE STROKE UNIT TENECTEPLASE VERSUSALTEPLASE FOR STROKE THROMBOLYSIS EVALUATION TRIAL IN THE AMBULANCE (TASTE-A)
1University of Melbourne, Department of Medicine and Neurology, Melbourne Brain Centre at the Royal Melbourne Hospital, Melbourne, Australia, 2Ambulance Victoria, Melbourne, Australia, 3The Walter and Eliza Hall Institute of Medical Research, 3. Population Health and Immunity Division, Melbourne, Australia, 4Alfred Health, Department of Neurology, Melbourne, Australia, 5Austin Health, Department of Neurology, Melbourne, Australia, 6Box Hill Hospital, 5. Department of Neurology, Melbourne, Australia, 7Monash Health, 7. Department of Medicine, Melbourne, Australia, 8Western Health, Department of Neurology, Melbourne, Australia, 9University of New South Wales, Department of Neurology, Sydney, Australia
1739
TENECTEPLASE VERSUS ALTEPLASE FOR MANAGEMENT OF ACUTE ISCHEMIC STROKE (NOR-TEST 2 PART A): A PHASE 3, RANDOMISED, OPEN-LABEL, BLINDED ENDPOINT TRIAL
1University of Bergen, Institute of clinical medicine, Department of Neurology, Bergen, Norway, 2Haukeland University Hospital, Department of Neurology, Bergen, Norway
EudraCT No 2018-003090-95. ClinicalTrials.gov No NCT03854500
1740
EARLY VS STANDARD TRACHEOSTOMY IN VENTILATED PATIENTS WITH SEVERE STROKE: RESULTS OF THE RANDOMIZED, MULTICENTER, INTERNATIONAL TRIAL SETPOINT2
1University of Heidelberg, Neurology, Heidelberg, Germany, 2Maine Medical Center, Neurology and Intensive Care, Portlande, United States
We aimed to test whether early versus standard tracheostomy improved functional outcome.
Speaker honoraria and travel support from BMS, Boehringer, Medtronic unrelated to the trial.
1741
ENDOVASCULAR VS MEDICAL MANAGEMENT OF POSTERIOR CEREBRAL ARTERY OCCLUSION STROKE: A META-ANALYSIS
1University Hospital Heidelberg, Neurology, Heidelberg, Germany, 2Aristotle University of Thessaloniki, Radiology, Thessaloniki, Greece, 3Lausanne University Hospital, Neurology, Lausanne, Switzerland, 4Heidelberg University Hospital, Neuroradiology, Heidelberg, Germany, 5Hamburg University Hospital, Radiology, Hamburg, Germany, 6Helsinki University Hospital, Neurology, Helsinki, Finland, 7Boston Medical Center, Radiology, Boston, United States, 8University of Pittsburgh Medical Center, UPMC Stroke Institute, Pittsburgh, United States, 9Dresden University Hospital, Neurology, Dresden, Germany, 10Dresden University Hospital, Neuroradiology, Dresden, Germany, 11Centro Hospitalar Universitario de Lisboa Central, Neuroradiology, Lisboa, Portugal, 12Hospital de Egas Moniz, Centro Hospitalar Lisboa Ocidental, Neurology, Lisbon, Portugal, 13IRCCS Istituto delle Scienze Neurologiche di Bologna, University of Perugia, Neurology, Bologna, Italy, 14Heidelberg University Hospital, Neurology, Heidelberg, Germany, 15Radiology Boston Medical Center, Neurology, Boston, Germany, 16Klinikum Ludwigshafen, Neurology, Ludwigshafen/Rhein, Germany
1750
EDOXABAN FOR STROKE PREVENTION IN INTRACRANIAL HEMORRHAGE SURVIVORS WITH ATRIAL FIBRILLATION: DESIGN OF THE ONGOING ENRICH-AF GLOBAL RANDOMIZED TRIAL
1McMaster University / Population Health Research Institute, Hamilton, Canada, 2Hospital Universitari Vall d’Hebron, Barcelona, Spain, 3Royal Cornwall Hospital, Truro, United Kingdom, 4Nobel Medical College Teaching Hospital, Biratnagar, Nepal, 5Hospital Universitario Ramón y Cajal / Universidad de Alcalá, Madrid, Spain, 6Hospital de la Santa Creu I Sant Pau, Barcelona, Spain, 7Victoria Hospital Kirkcaldy, Fife, United Kingdom, 8Royal United Hospital Bath, Bath, United Kingdom, 9University of Antwerp, Antwerp, Belgium, 10B P Koirala Institute of Health Sciences, Dharan, Nepal, 11Inselspital, University Hospital Bern, Bern, Switzerland, 12Center for Stroke Research Berlin / Charité-Universitätsmedizin Berlin, Berlin, Germany, 13Blue Cross Brain Hospital Affiliated to Tongji University, Shanghai, China, 14University Hospital of Wales, Cardiff, United Kingdom, 15Xinhua Hospital, Chongming Branch, Shanghai, China, 16King’s Mill Hospital, Sutton-in-Ashfield, United Kingdom, 17CUB - Hôpital Erasme, Brussels, Belgium, 18Antwerp University Hospital/University of Antwerp, Antwerp, Belgium, 19University of Saskatchewan, Saskatoon, Canada, 20Alexandria University, Alexandria, Egypt, 21Katholieke Universiteit Leuven, Leuven, Belgium, 22University of Edinburgh, Edinburgh, United Kingdom, 23University of Ottawa, Ottawa, Canada, 24University of A Coruna, A Coruna, Spain, 25Ain Shams University, Cairo, Egypt, 26GNRC Hospital, Guwahati, India, 27Donostia University, San Sebastian – Donostia, Spain, 28La Fe University and Polytechnic Hospital, Valencia, Spain, 29Complejo Hospitalario Universitario de Caceres, Caceres, Spain, 30University of Calgary, Calgary, Canada
1751
HYBRIDMACHINE LEARNING-BASED PREDICTION OF FUTILE RECANALIZATION AFTER ENDOVASCULAR TREATMENT IN ACUTE ISCHEMIC STROKE: RESCUE-FR INDEX
1Beijing Tiantan Hospital, Capital Medical University, Beijing, China, Department of Neurology, Beijing, China, 2China National Clinical Research Center for Neurological Diseases, Beijing Tiantan Hospital, Beijng, China, BioMind Technology AI Center, Beijing, China
Important Characteristics in Derivation Cohort and Validation Cohort.
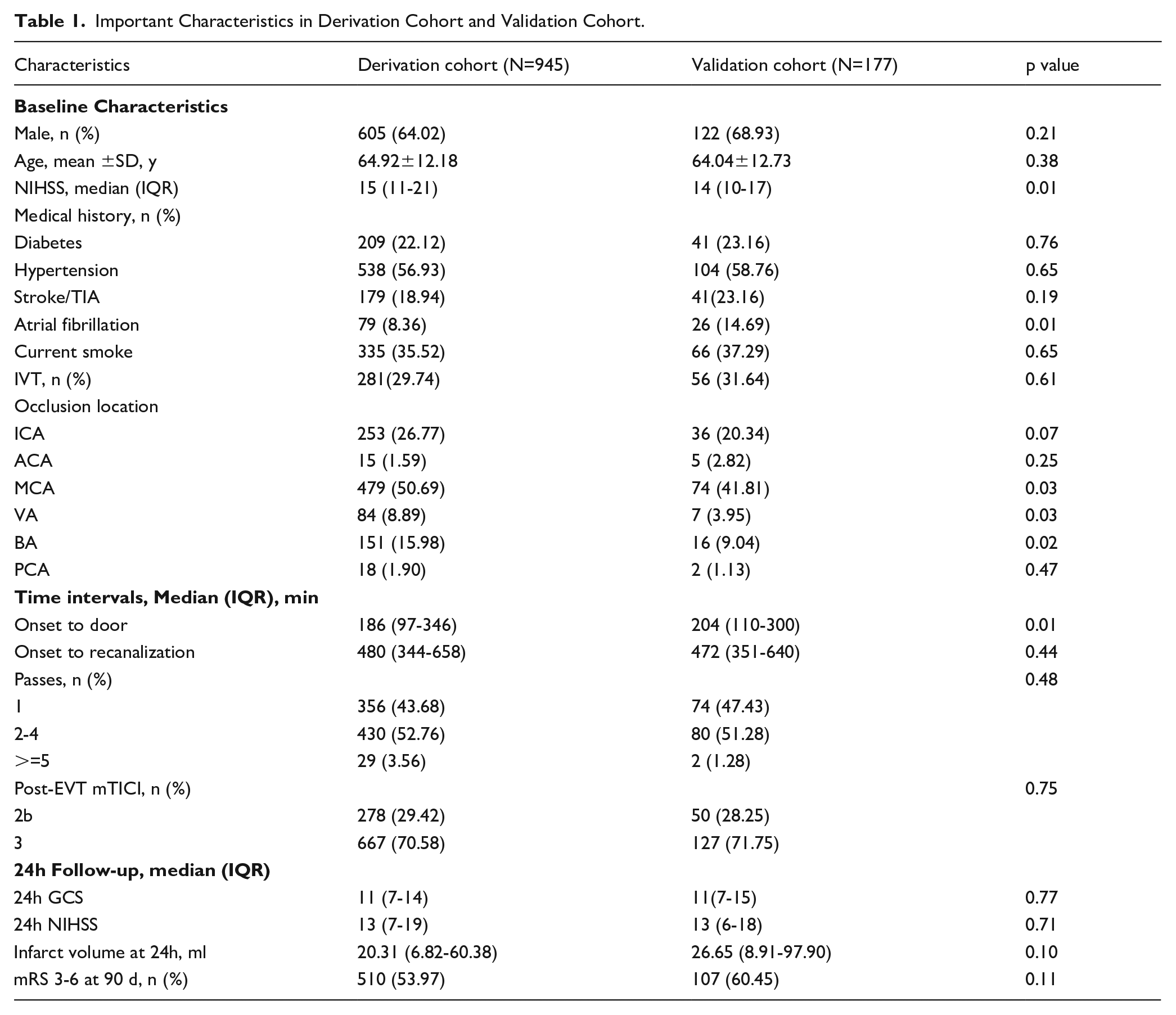
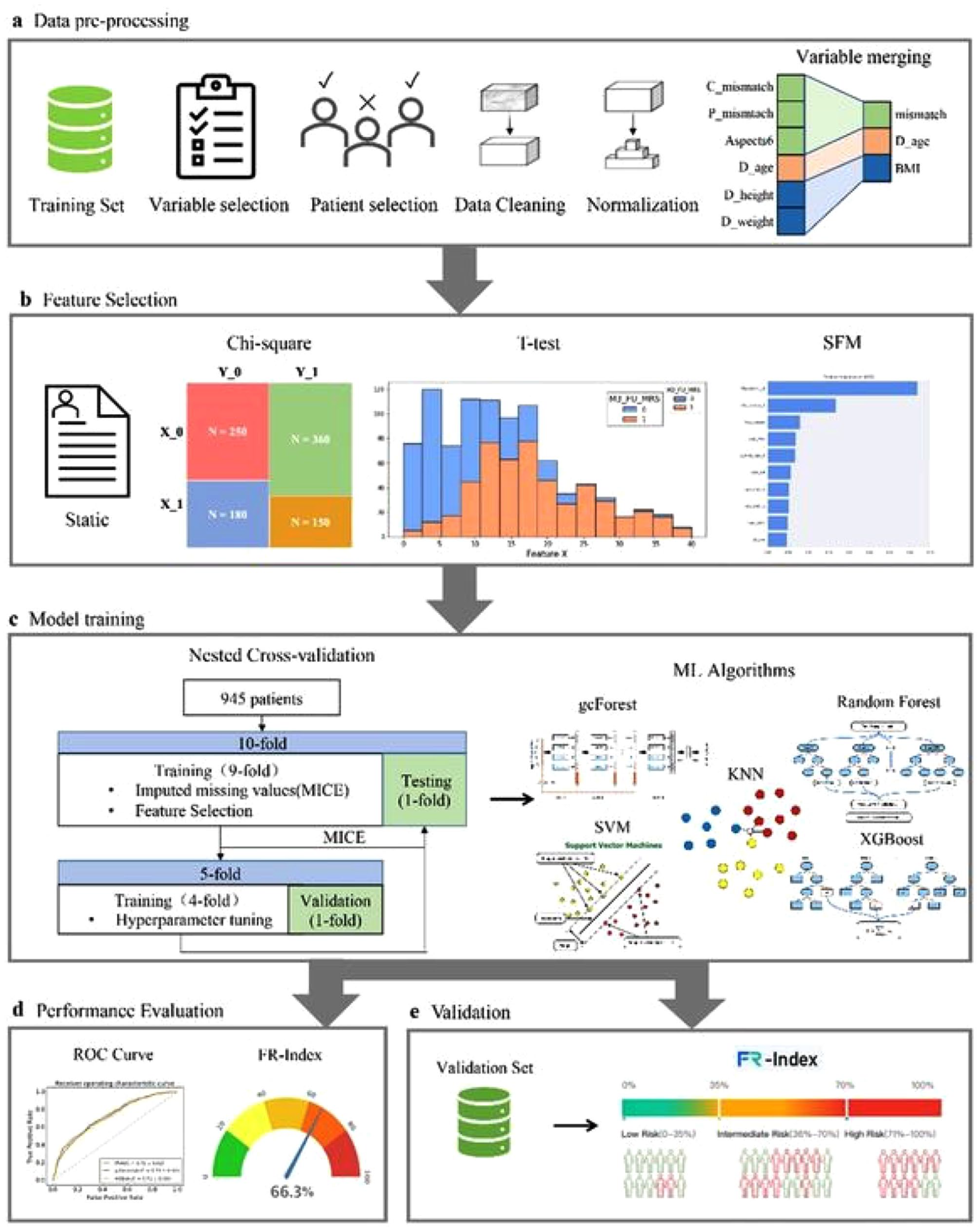
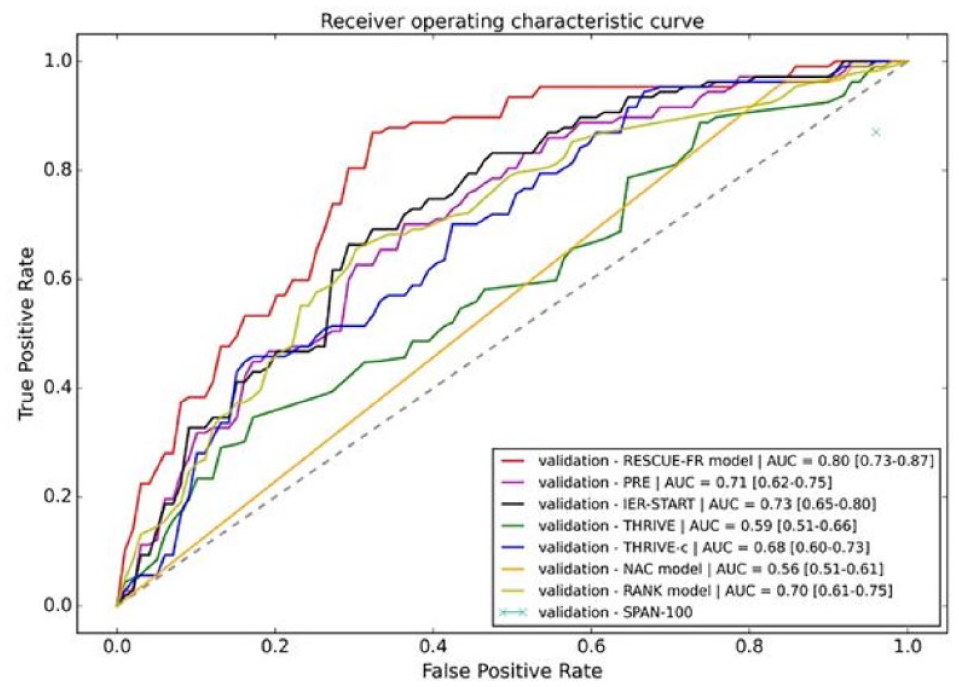
1753
EASI-TOC: A TRIAL OF ACUTE CERVICAL INTERNAL CAROTID ARTERY STENTING DURING ENDOVASCULAR THROMBECTOMY FOR ANTERIOR CIRCULATION STROKE
1Université de Montréal, Neurosciences, Montreal, Canada, 2Université de Montréal, Radiology, Montreal, Canada, 3Université de Montréal, Biostatistics, Montreal, Canada, 4Centre de Recherche du CHUM, Montreal, Canada, 5University of Calgary, Clinical Neurosciences, Calgary, Canada, 6Université Laval, Medicine (Neurology), Quebec, Canada, 7McMaster University, Hamilton, Canada, 8University of British Columbia, Vancouver, Canada, 9Queen’s University, Kingston, Canada, 10McGill University, Montreal, Canada, 11Université Laval, Surgery, Quebec, Canada, 12University of Ottawa, Medicine (Neurology), Ottawa, Canada, 13Queen’s University, Radiology, Kingston, Canada, 14Dalhousie University, Radiology, Halifax, Canada
1755
ENDOVASCULAR TREATMENT VERSUS BEST MEDICAL MANAGEMENT IN ACUTE BASILAR ARTERY OCCLUSION STROKES: RESULTS FROM THE MULTICENTER ATTENTION REGISTRY
1The first affiliated hospital of USTC, department of neurology, Hefei, China, 2Zeenat Qureshi Stroke Research Center, Morris, United States, 3Maoming People’s Hospital, Guangdong, China, 4Zhoukou Central Hospital, Zhoukou, China, 5Nanjing First Hospital, Nanjing, China, 6Cangzhou Central Hospital, Cangzhou, China, 7Suzhou Municipal Hospital, Suzhou, China, 8Sir Run Run Shaw Hospital, Zhejiang University, Hangzhou, China, 9The Second Affiliated Hospital of Soochow University, Soochow, China, 10Qingdao Central Hospital, Qingdao University, Qingdao, China, 11Taizhou First People’s Hospital, Taizhou, China, 12Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, China, 13Lehigh Valley Health Network, Allentown, United States, 14UPMC Stroke Institute, Pittsburgh, United States
1756
TREATMENT OF HYPERGLYCAEMIA IN ACUTE STROKE: RESULTS FROM THE TRIAL OF EXENATIDE IN ACUTE ISCHAEMIC STROKE (TEXAIS)
1Monash University - Eastern Health Clinical School, Melbourne, Australia, 2Westmead Hospital & University of Sydney, Centre for Diabetes and Endocrinology Research, Centre for Diabetes and Endocrinology Research, Sydney, Australia, 3Monash University, Eastern Health Clinical School, Melbourne, Australia, 4University of Melbourne - Melbourne Medical School, Faculty of Medicine, Dentistry and Health Sciences, Melbourne, Australia, 5Australian Catholic University/St Vincent’s Health Australia (Sydney) - Nursing Research Institute, Sydney, Australia, 6University of Melbourne - Florey Institute of Neuroscience and Mental Health, Melbourne, Australia, 7University of Melbourne - Department of Medicine, Department of Endocrinology, Austin Health, Melbourne, Australia, 8University of Newcastle, Department of Medicine, Newcastle, Australia, 9The University of Sydney - Sydney Medical School, Sydney, Australia, 10University of Melbourne - Melbourne Brain Centre, Melbourne, Australia, 11Sydney University - Ingham Institute for Applied Medical Research, South Western Sydney Clinical School, Sydney, Australia, 12University of Melbourne - Melbourne Brain Centre, Department of Neurology, Melbourne, Australia, 13University of Queenland - Royal Brisbane and Women’s Hospital, Department of Neurology, Brisbane, Australia, 14University of Helsinki, Department of Neurology, Helsinki, Norway, 15University of New South Wales - Liverpool Hospital, Department of Neurology, Sydney, Australia, 16University of Queensland - Princess Alexandra Hospital, Department of Neurology, Brisbane, Australia, 17Sunshine Coast University, Sunshine Coast, Australia, 18University of Western Australia - Fiona Stanley Hospital, Perth, Australia, 19University of Melbourne - St Vincent’s Hospital, Department of Neurology, Melbourne, Australia, 20Monash University - Alfred Hospital, Department of Neurology, Melbourne, Australia, 21St John Of God Hospital, Perth, Australia, 22University of Otago, Department of Neurology, Christchurch, New Zealand, 23University of Tasmania, Department of Medicine, Launceston, Australia
1757
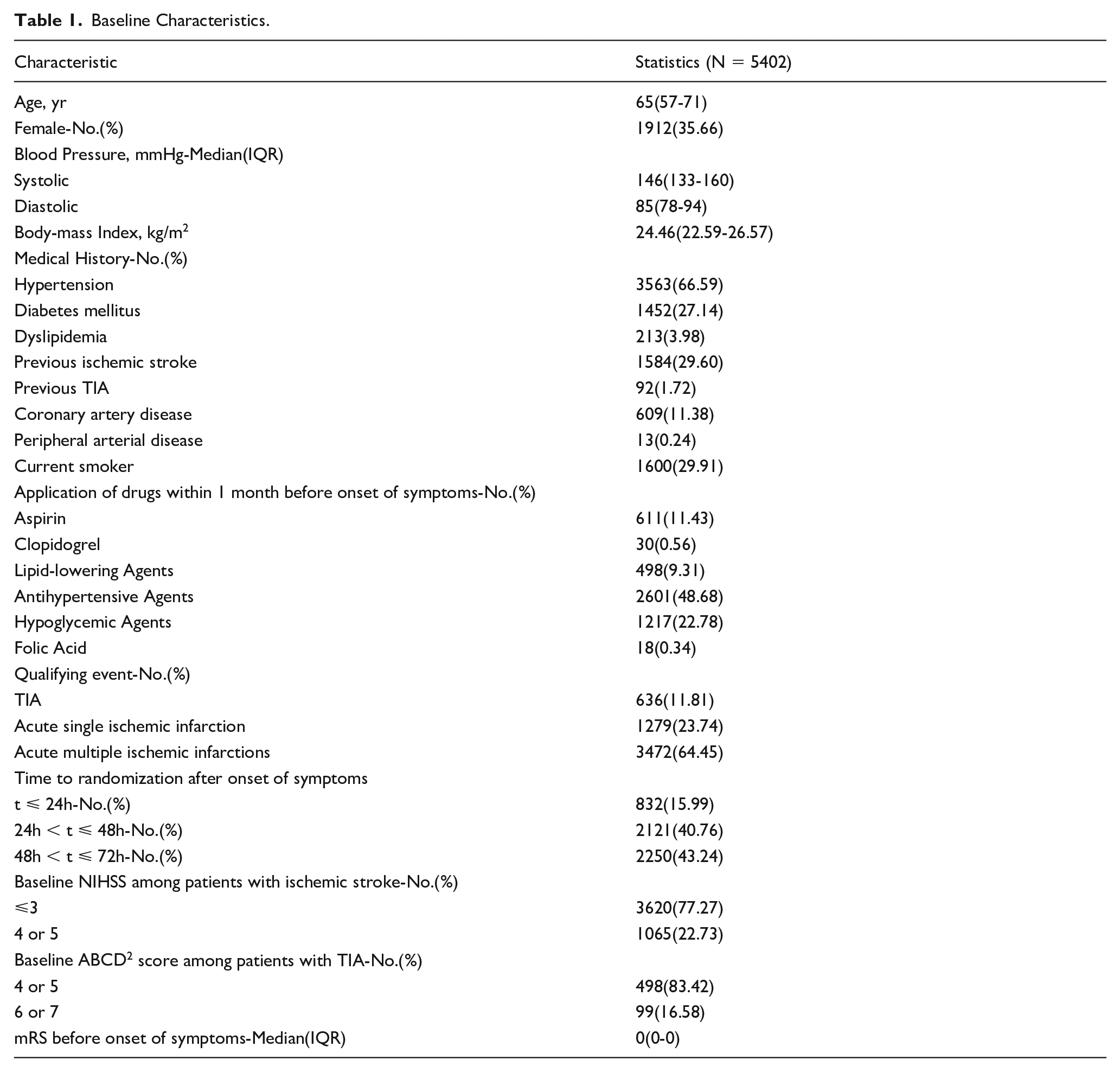
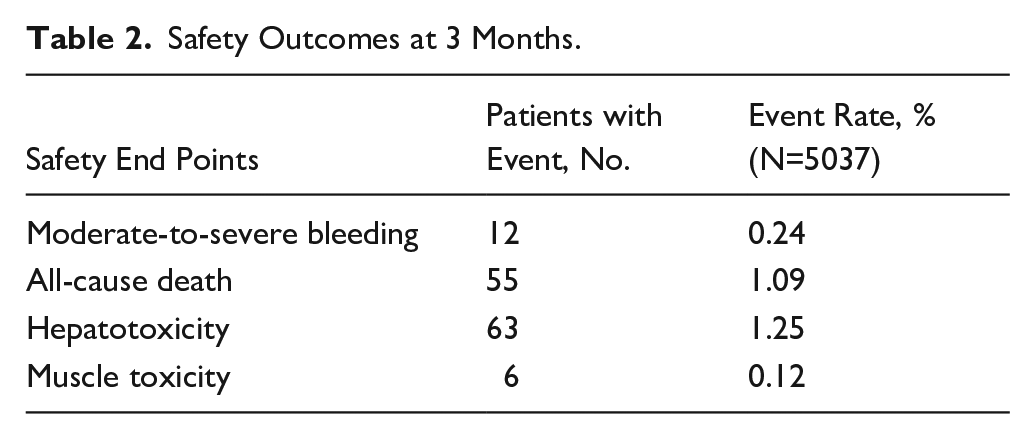
INTENSIVE STATIN AND ANTIPLATELET THERAPY FOR ACUTE HIGH-RISK SYMPTOMATIC INTRACRANIAL OR EXTRACRANIAL ATHEROSCLEROSIS: A RANDOMIZED, DOUBLE-BLIND, PLACEBO-CONTROLLED, MULTICENTER, 2*2 FACTORIAL DESIGN TRIAL
1Beijing Tiantan Hospital, Capital Medical University, Neurology, Beijing, China, 2Chinese Institute for Brain Research, Beijing, China, 3China National Clinical Research Center for Neurological Diseases, Beijing, China, 4Capital Medical University, Advanced Innovation Center for Human Brain Protection, Beijing, China, 5Beijing Key Laboratory of Translational Medicine for Cerebrovascular Disease, Beijing, China
Baseline Characteristics.
Safety Outcomes at 3 Months.
1758
GLENZOCIMAB, A NOVEL ANTITHROMBOTIC, IS ASSOCIATED WITH REDUCED INTRACRANIAL HEMORRHAGE AND MORTALITY RATES WHEN COMBINED WITH STANDARD-OF-CARE REPERFUSION THERAPIES: THE ACTIMIS STUDY
1Hôpital Lariboisière, Service de Neurologie Unité NeuroVasc, Paris, France, 2CHRU de Nancy - Hôpital Central, Unité Neurovasculaire - Service de Neurologie, Nancy, France, 3Hospital Vall d’Hebron, Stroke Unit, Barcelona, Spain, 4Policlinico Umberto I, Unità di Trattamento Neurovascolare, Roma, Italy, 5Universitätsspital Basel, Stroke Unit, Basel, Switzerland, 6UZ Leuven, Department of Neurology, Leuven, Belgium, 7Acticor Biotech, Paris, France, 8University of Rouen, Rouen, France, 9LVTS, UMR_S1148 INSERM, Paris, France, 10Universitätsklinikum Essen (AöR), Klinik für Neurologie, Essen, France, 11Memorial Hermann Hospital Texas Medical Center, Clinical Innovation and Research Institute, Houston, United States
Avenard G. is the Chief Executive Officer of Acticor Biotech.
Plétan Y. is the Chief Medical Officer of Acticor Biotech.
Jandrot-Perrus M. is the co-founder of Acticor Biotech.
Mazighi M. and Grotta JC are ACTIMIS study coordinators, a study sponsored and funded by Acticor Biotech.
1759
A PROSPECTIVE, MULTICENTER, RANDOMIZED, PARALLEL-CONTROLLED CLINICAL STUDY TO EVALUATE THE SAFETY AND EFFICACY OF INTRACRANIAL BALLOON ANGIOPLASTY COMBINED WITH INTENSIVE DRUG THERAPY FOR SICAS (BASIS)
1 Capital Medical University, Interventional Neuroradiology, Beijing, China
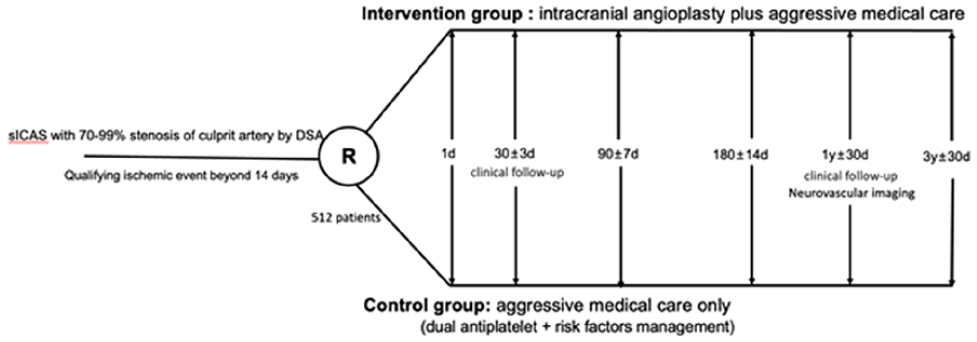
1760
EARLY VERSUS LATE INITIATION OF ANTICOAGULATION IN PATIENTS WITH ATRIAL FIBRILLATION AFTER ACUTE STROKE OR TIA – THE PRODAST STUDY
1University of Duisburg-Essen, Institute for Medical Informatics, Biometry and Epidemiology, Essen, Germany, 2Hannover Medical School, Department of Neurology, Hannover, Germany, 3BDH Clinic Elzach, Elzach, Germany, 4Center for Clinical Trials Essen, Essen, Germany
1761
LEFT ATRIAL APPENDAGE OCCLUSION VERSUS STANDARD OF CARE IN PATIENTS WITH ATRIAL FIBRILLATION AND PRIOR THROMBO-EMBOLIC EVENT DESPITE ANTICOAGULATION THERAPY: A PROPENSITY SCORE MATCHED COMPARISON
1St. Antonius Ziekenhuis Nieuwegein, Cardiology, Nieuwegein, Netherlands, 2Amsterdam UMC, Cardiology, Amsterdam, Netherlands, 3Inselspital, Neurology, Bern, Switzerland, 4San Raffaele Research Hospital, Cardiology, Milan, Italy, 5Centro Cardiologica Monzino, Cardiology, Milan, Italy, 6National Institute of Cardiology, Cardiology, Warsaw, Poland, 7Rigshospitalet, Cardiology, Copenhagen, Denmark, 8Aarhus University Hospital, Cardiology, Aarhus, Denmark, 9Complexo Hospitalario Universitario de Vigo, Cardiology, Vigo, Spain, 10University Hospital of Leon, Cardiology, Leon, Spain, 11Hospital Clinico San Carlos, Cardiology, Madrid, Spain, 12Centro Medico Teknon Barcelona, Cardiology, Barcelona, Spain, 13CHI Health Creighton University Medical Center, Cardiology, Omaha, United States, 14University Heart Center Lübeck, Cardiology, Lübeck, Germany, 15Hospital Clinic de Barcelona, Cardiology, Barcelona, Spain, 16St. Joseph’s Medical Center, Cardiology, Stockton, United States, 17Cardiologicum Hamburg, Cardiology, Hamburg, Germany, 18International Heart Institute of Montana, Cardiology, Montana, United States, 19Emory University Hospital, Cardiology, Atlanta, United States, 20Spedali Civili di Brescia, Cardiology, Brescia, Italy, 21Rush University Medical Center, Cardiology, Chicago, United States, 22University Hospital Basel, Neurology, Basel, Switzerland, 23University Hospital Erlangen, Neurology, Erlangen, Germany, 24University Hospital Perugia, Neurology, Perugia, Italy, 25National Cerebral and Cardiovascular Center, Neurology, Osaka, Japan, 26University College London, Neurology, London, United Kingdom, 27University Hospital Verona, Neurology, Verona, Italy
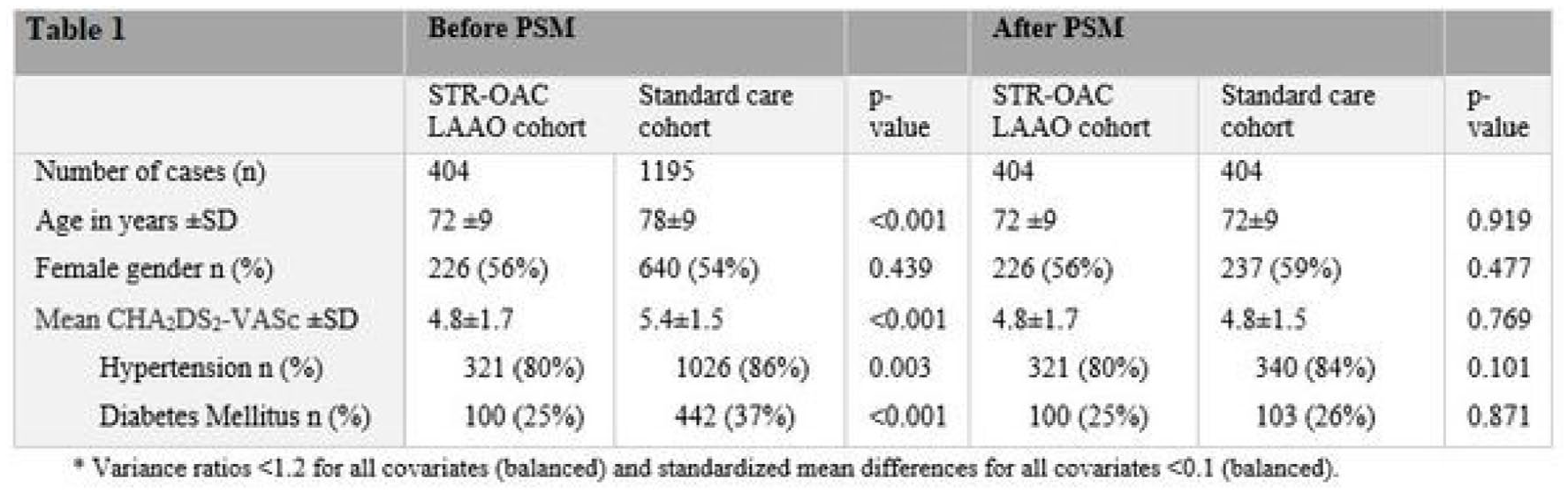
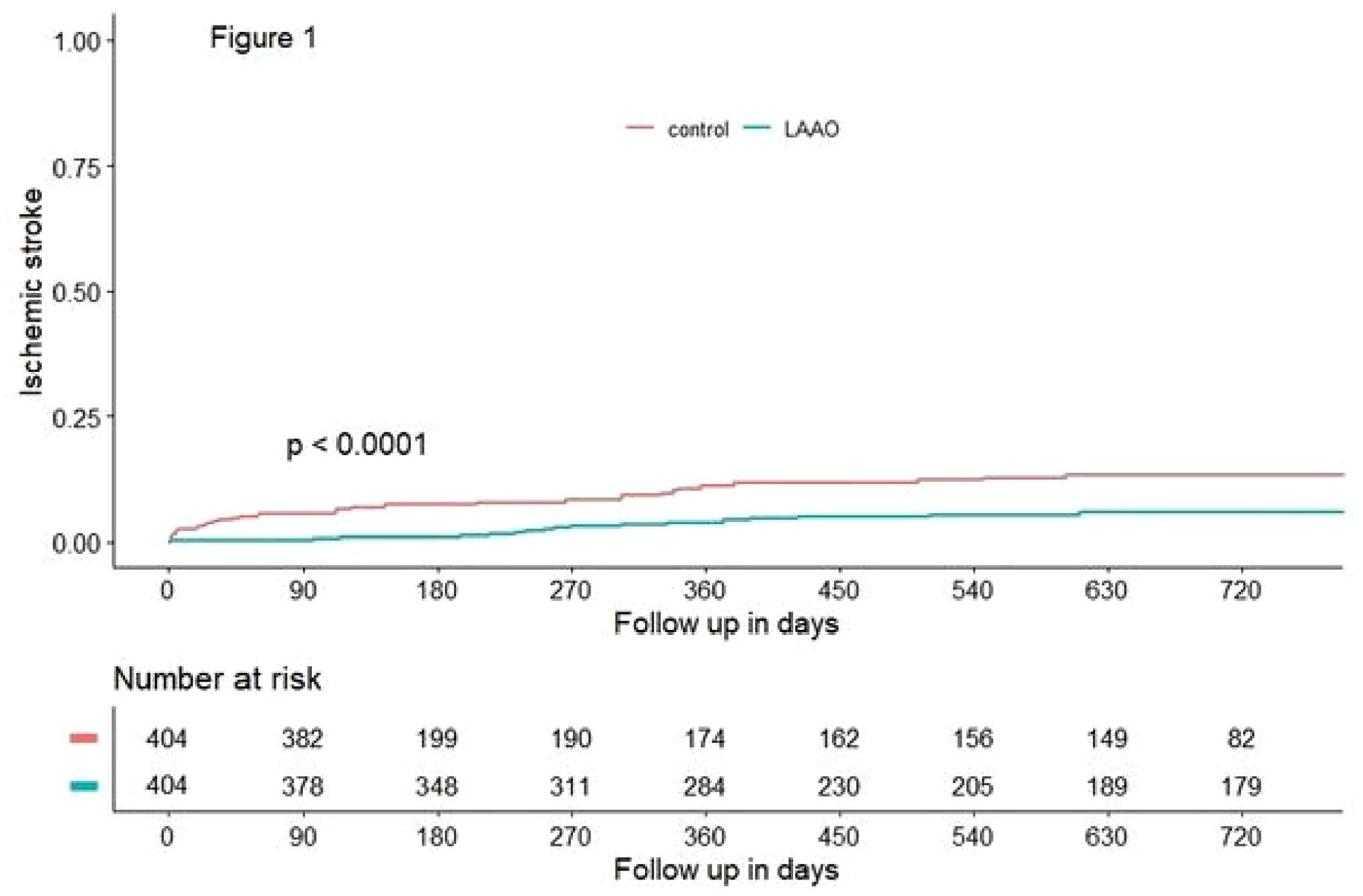
1762
PREDICTIVE VALUE OF AUTOMATED ASSESSED CT PERFUSION ON DELAYED CEREBRAL ISCHEMIA IN MODERATE-TO-POOR GRADE ANEURYSMAL SUBARACHNOID HEMORRHAGE
1 Beijing Tiantan Hospital, Neurology, Beijing, China
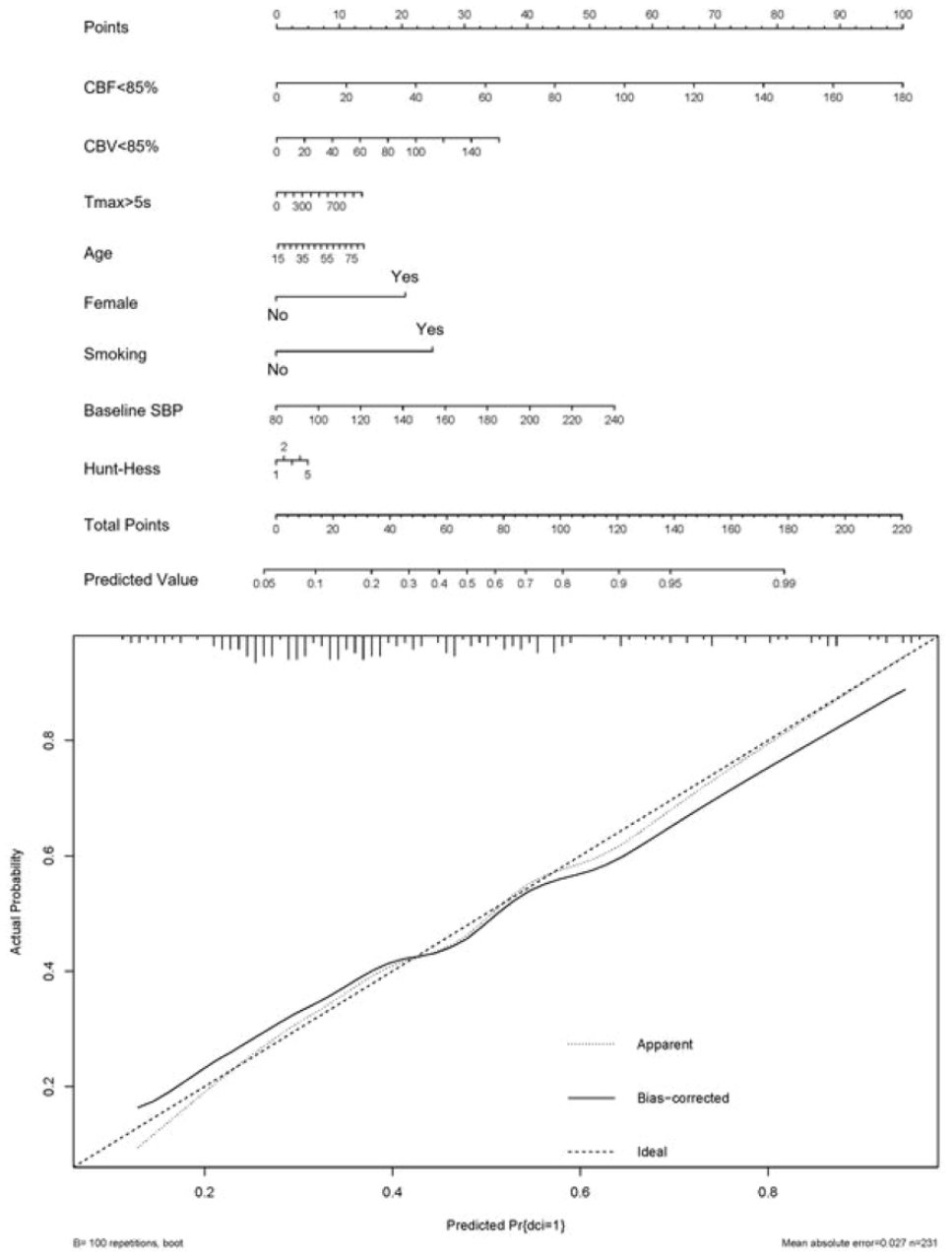
1763
HEART RATE VARIABILITY PREDICTS RECURRENCE IN PATIENTS WITH ACUTE MINOR STROKE OR TRANSIENT ISCHAEMIC ATTACK STRATIFIED BY ABCD2 SCORE
1 Beijing Tiantan Hospital of Capital Medical University, Neurology, Beijing, China
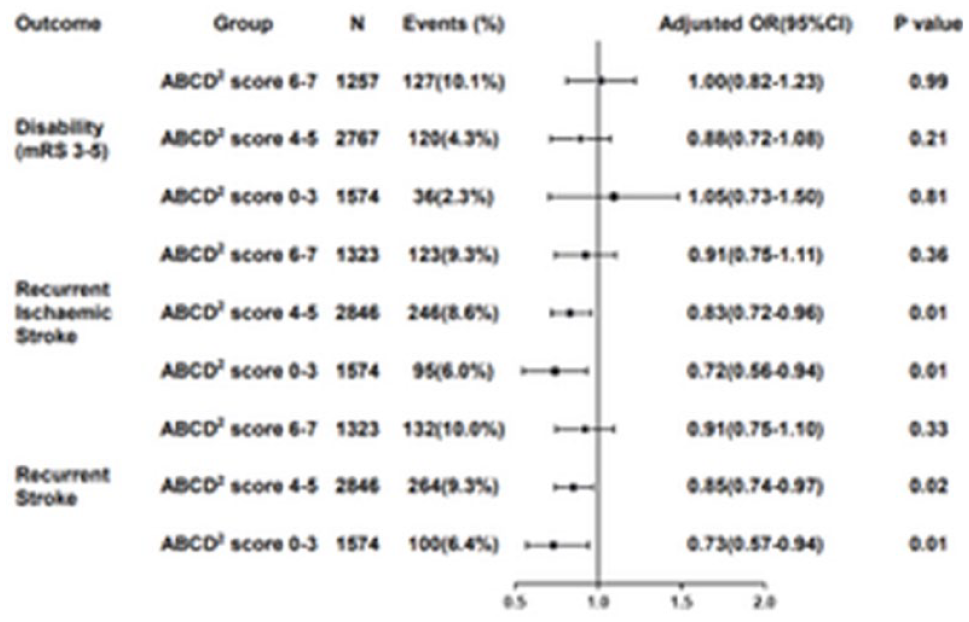
Risk of outcomes in patients with MIS/TIA divided into six groups by different SDNN and ABCD2 socre at 1-year follow-up.
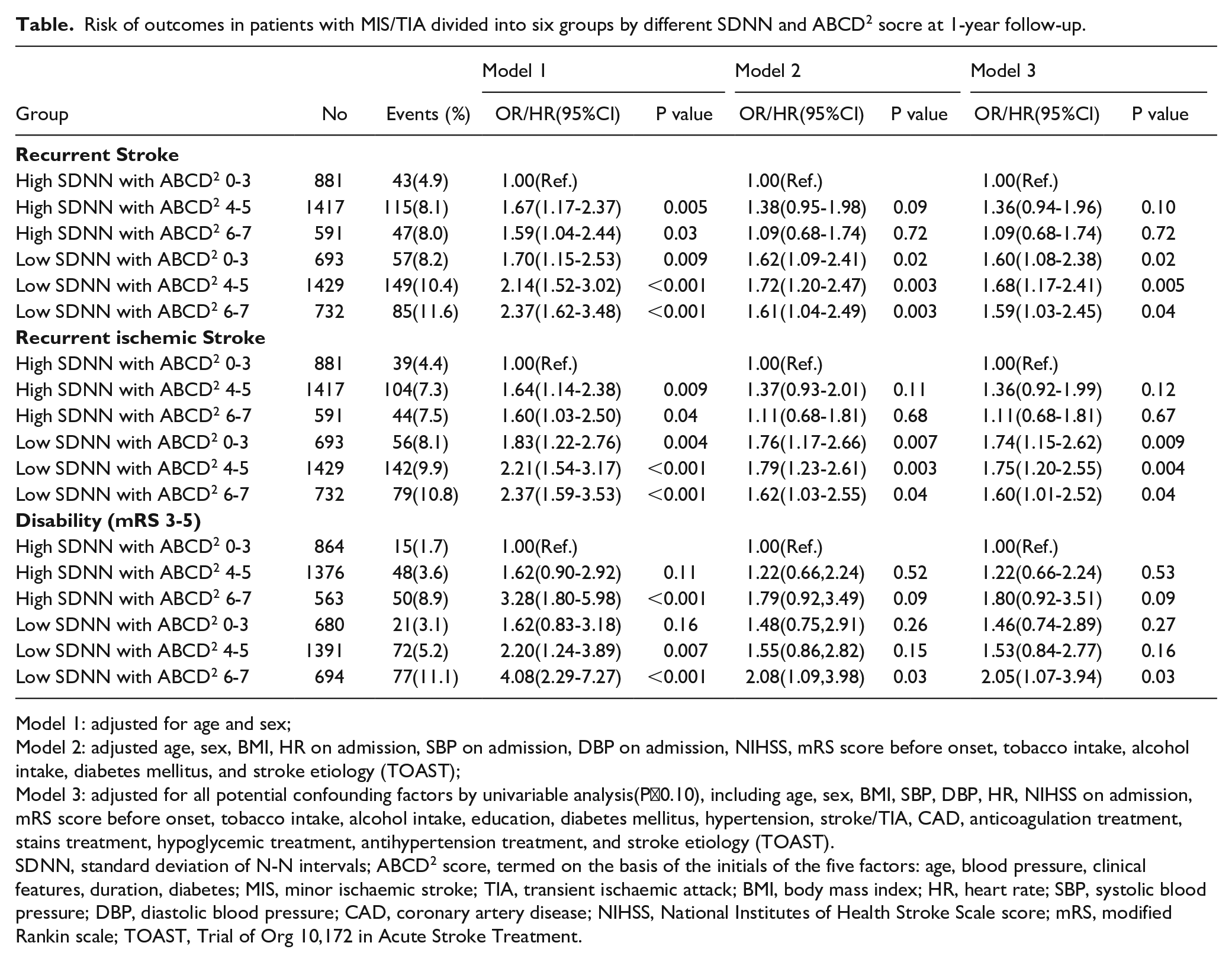
Model 1: adjusted for age and sex;
Model 2: adjusted age, sex, BMI, HR on admission, SBP on admission, DBP on admission, NIHSS, mRS score before onset, tobacco intake, alcohol intake, diabetes mellitus, and stroke etiology (TOAST);
Model 3: adjusted for all potential confounding factors by univariable analysis(P<0.10), including age, sex, BMI, SBP, DBP, HR, NIHSS on admission, mRS score before onset, tobacco intake, alcohol intake, education, diabetes mellitus, hypertension, stroke/TIA, CAD, anticoagulation treatment, stains treatment, hypoglycemic treatment, antihypertension treatment, and stroke etiology (TOAST).
SDNN, standard deviation of N-N intervals; ABCD2 score, termed on the basis of the initials of the five factors: age, blood pressure, clinical features, duration, diabetes; MIS, minor ischaemic stroke; TIA, transient ischaemic attack; BMI, body mass index; HR, heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; CAD, coronary artery disease; NIHSS, National Institutes of Health Stroke Scale score; mRS, modified Rankin scale; TOAST, Trial of Org 10,172 in Acute Stroke Treatment.
1765
EFFECTS OF INTENSIVE BLOOD PRESSURE LOWERING ON CEREBRAL ISCHAEMIA: INSIGHTS FROM THE ENCHANTED TRIAL
1Shanghai East Hospital, School of Medicine, Tongji University, Shanghai, China, 2The George Institute for Global Health, University of New South Wales, Sydney, Australia, 3The George Institute China at Peking University Health Sciences Center, Beijing, China, 4Ingham Institute for Applied Medical Research, Liverpool Hospital, UNSW, Sydney, Australia
1771
INTRAVENOUS THROMBOLYSIS IN PATIENTS WITH ISCHAEMIC STROKE AND RECENT DIRECT ORAL ANTICOAGULANTS INTAKE – AN INTERNATIONAL COLLABORATION
1Heidelberg University Hospital, Department of Neurology, Heidelberg, Germany, 2Christchurch Hospital, Department of Neurology, Christchurch, New Zealand, 3New Zealand Brain Research Institute, Christchurch, New Zealand, 4Inselspital University Hospital Bern, Department of Neurology, Bern, Switzerland, 5University Hospital Basel, Department of Neurology and Stroke Center, Basel, Switzerland, 6Charité-Universitätsmedizin Berlin, Klinik und Hochschulambulanz für Neurologie, Berlin, Germany, 7Charité-Universitätsmedizin Berlin, Center for Stroke Research Berlin, Berlin, Germany, 8University of Basel, University Department of Geriatric Medicine Felix Platter, Neurology and Neurorehabilitation, Basel, Switzerland
Wilson, D: No COIs
Meinel, T: No COIs
Gensicke, H: has received research support from the Swiss National Science Foundation, advisory board honoraria from Daiichi Sankyo and funding for travel from BMS/Pfizer and AbbVie.
Scheitz, JF: No COIs
Nolte, CH: received research grants from German Ministry of Research and Education, German Center for Neurodegenerative Diseases, German Center for cardiovascular Research, and speaker and/or consultation fees from Abbott, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Pfizer Pharma, and Alexion.
Engelter, S: STE has received funding for travel or speaker honoraria from Bayer, Boehringer Ingelheim and Daiichi-Sankyo. He has served on scientific advisory boards for Bayer, Boehringer Ingelheim, BMS/Pfizer, and MindMaze and on the editorial board of Stroke. His institutions have received an educational grant from Pfizer, compensation from Stago for educational efforts and research support from Daiichi-Sankyo, the Science Funds [Wissenschaftsfonds] of the University Hospital Basel, the University Basel, from the “Wissenschaftsfonds Rehabilitation” of the University Hospital for Geriatric Medicine Felix Platter, the “Freiwillige Akademische Gesellschaft Basel”, the Swiss Heart Foundation, and the Swiss National Science Foundation.
Ringleb, P: PAR received travel support and lecture fees from Boeheringer Ingelheim, Daiichi Sankyo, Pfizer, and Bayer, outside the submitted work.
Wu, T: No COIs
Seiffge, D: Advisory Board Bayer Switzerland AG and Portola/Alexion, Research funding: Swiss Science Foundation, Swiss Heart Foundation, Bangerter-Rhyner Foundation, Bayer Foundation, Portola/Alexion
1772
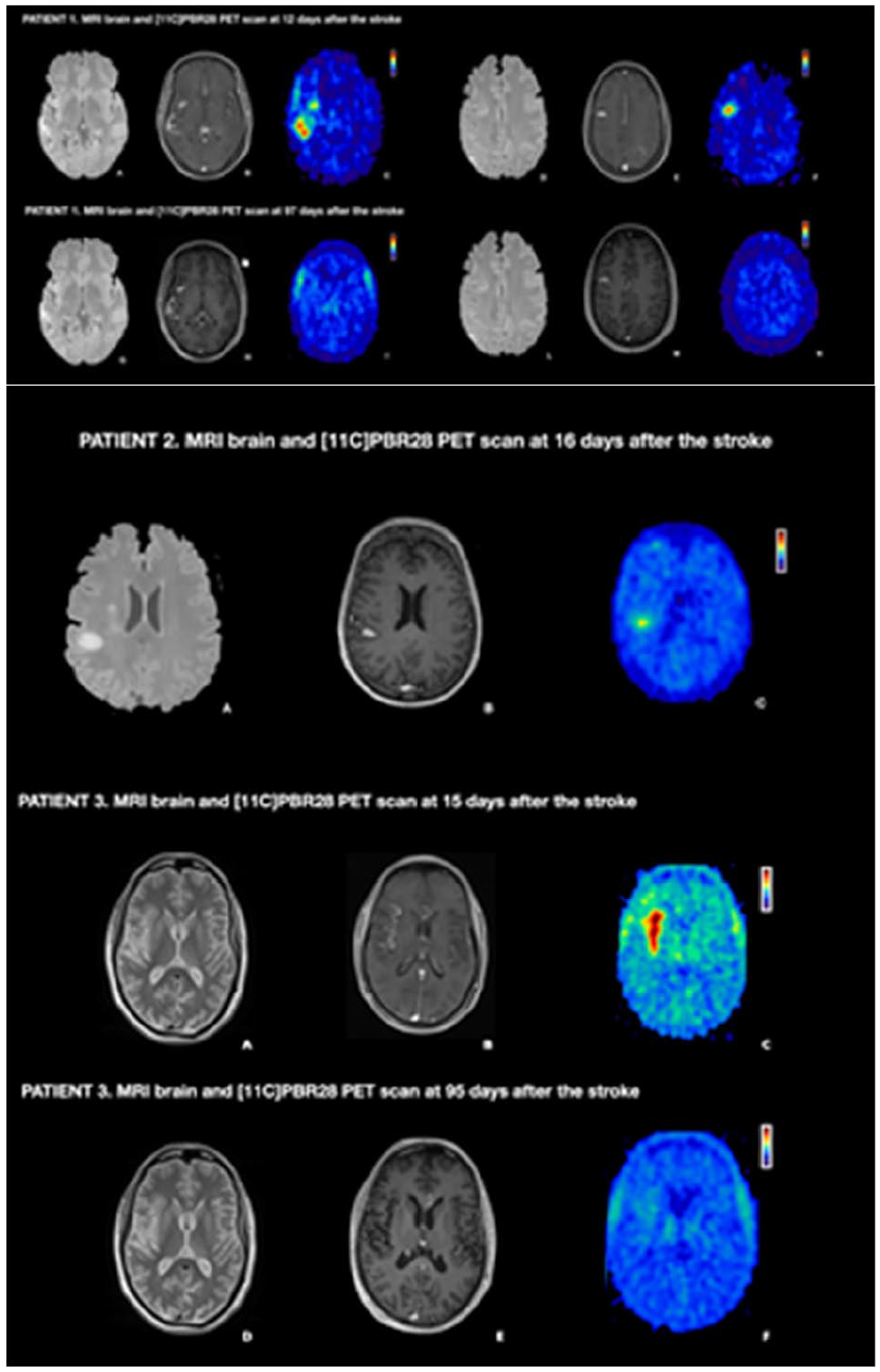
TIME COURSE OFNEUROINFLAMMATION AFTER HUMAN STROKE - A PILOT STUDY USING CO-REGISTERED PET AND MRI
1Imperial College London, BRAIN SCIENCE, London, United Kingdom, 2Imperial College London, London, United Kingdom
1773
ETHICS OUTCOMES OF THE ALTEPLASE COMPARED TO TENECTEPLASE (ACT) TRIAL
1University of Ottawa / Ottawa Hospital Research Institute, Medicine / Neurology, Ottawa, Canada, 2University of Calgary, Calgary Stroke Program, Calgary, Canada, 3University of Alberta, Edmonton, Canada, 4University of Toronto, Sunnybrook Hospital, Toronto, Canada
1774
UNDERLYING CAUSES OF CRYPTOGENIC STROKE AND CRYPTOGENIC TIA IN THE NORDIC ATRIAL FIBRILLATION AND STROKE (NOR-FIB) STUDY: TOPLINE RESULTS. CLINICALTRIALS.GOV IDENTIFIER NCT02937077, EUDRACT 2018-002298-23
1Østfold Hospital Trust, Department of Neurology, Grålum, Norway, 2University of Oslo, Institute of Clinical Medicine, Oslo, Norway, 3Østfold Hospital Trust, Department of Cardiology, Grålum, Norway, 4Nordlandssykehuset, Department for Neurology, Bodø, Norway, 5Innlandet Hospital Trust, Lillehammer Hospital, Department of Neurology, Lillehammer, Norway, 6Molde Hospital, Department of Neurology, Molde, Norway, 7Diakonhjemmet Hospital, Department of Internal Medicine, Oslo, Norway, 8Oslo University Hospital, Ullevål, Stroke Unit, Department of Neurology, Oslo, Norway, 9Vestre Viken Hospital Trust, Bærum Hospital, Department of Internal Medicine, Gjettum, Norway, 10University Hospital of North Norway, Department of Neurology, Tromsø, Norway, 11Oslo University Hospital, Rikshospitalet, Department of Neurology, Oslo, Norway, 12Skåne University Hospital, Department of Neurology, Malmö, Sweden, 13Vestfold Hospital, Department of Neurology, Tønsberg, Norway, 14Herlev Gentofte Hospital, Department of Neurology, Herlev, Denmark, 15Stavanger University Hospital, Department of Neurology, Stavanger, Norway, 16Vestre Viken Hospital Trust, Drammen Hospital, Department of Neurology, Drammen, Norway, 17Haukeland University Hospital, Department of Neurology, Bergen, Norway, 18Telemark Hospital, Department of Neurology, Skien, Norway, 19Rigshospitalet University Hospital, Department of Neurology, Copenhagen, Denmark, 20Bispebjerg University Hospital, Department of Neurology, Copenhagen, Denmark, 21Oslo University Hospital, Ullevål, Department of Cardiology, Oslo, Norway
Majority of NOR-FIB patients did not have AF and 33% did not have any significant arrhythmia. The non-arrhythmia patients were significantly younger (P< 0.001) and had the lowest rates of all measured comorbidity, especially dyslipidaemia (P = 0.045) and previous stroke / TIA (P = 0.018). In 13.5% patients, other possible stroke causes were revealed (small vessel disease, large artery disease, hypercoagulable states, cardioembolism). Another 6.6% of patients, none of them having AF, had cancer diagnosis, as a possible cause of their ischaemic event.
DA has received honoraria from Actelion, Amgen, AstraZeneca, BMS/Pfizer, Bayer, Boehringer-Ingelheim, MSD, Novartis, Roche Diagnostics, Sanofi and Vifor Pharma. DA has received research grants to the institution from BMS/Pfizer, Bayer, Roche Diagnostics and Medtronic.
AHA has received travel support, honoraria for advice or lecturing from Bayer, Boehringer Ingelheim, BMS, Allergan, Teva, Sanofi-Genzyme, Novartis, Roche, and Teva and research grant from Boehringer Ingelheim.
Devices (ICMs) were partly made available by Medtronic.
1776
MULTICENTER RANDOMIZED CLINICAL TRIAL OF ENDOVASCULAR TREATMENT OF ACUTE ISCHEMIC STROKE IN THE NETHERLANDS FOR LATE ARRIVALS: MR CLEAN-LATE
1Maastricht University Medical Center+ and School for Cardiovascular Diseases (CARIM), Maastricht, Department of Neurology, Maastricht, Netherlands, 2Maastricht University Medical Center+ and School for Cardiovascular Diseases (CARIM), Maastricht, Department of Radiology, Maastricht, Netherlands, 3Leiden University Medical Center, Department of Radiology, Leiden, Netherlands, 4Medical Center Haaglanden, Department of Radiology, The Hague, Netherlands, 5University Medical Center Groningen, Department of Neurology, Groningen, Netherlands, 6Sint Antonius Hospital, Department of Neurology, Nieuwegein, Netherlands, 7Amsterdam University Medical Center, location University of Amsterdam, Department of Radiology, Amsterdam, Netherlands
1778
EXPLORING DONOR BEHAVIOR IN CROWDFUNDING OF STROKE RESEARCH
1University of Calgary, Department of Radiology, Calgary, Canada, 2University Hospital Basel, Department of Neuroradiology, Basel, Switzerland, 3University Hospital Basel, Department of Neurology, Basel, Switzerland, 4University of Calgary, Department of Clinical Neurosciences, Calgary, Canada, 5University of Amsterdam, Department of Radiology and Nuclear Medicine, Amsterdam, Netherlands, 6University of Calgary, Calgary, Canada, 7David Geffen School of Medicine, UCLA, Department of Neurology, Los Angeles, United States
RM: CEO and shareholder of Collavidence Inc.
1779
MEDICAL CROWDFUNDING AS A COMPLEMENTARY FUNDING SOURCE FOR EARLY-CAREER STROKE RESEARCHERS
1University of Calgary, Department of Clinical Neurosciences, Calgary, Canada, 2University Hospital Basel, Department of Neurology, Basel, Switzerland, 3University Hospital Basel, Department of Neuroradiology, Basel, Switzerland, 4University of Amsterdam, Department of Radiology and Nuclear Medicine, Amsterdam, Netherlands, 5University of Calgary, Department of Radiology, Calgary, Canada, 6University of Calgary, Calgary, Canada, 7David Geffen School of Medicine, UCLA, Department of Neurology, Los Angeles, United States
RM: CEO and shareholder of Collavidence Inc.
1781
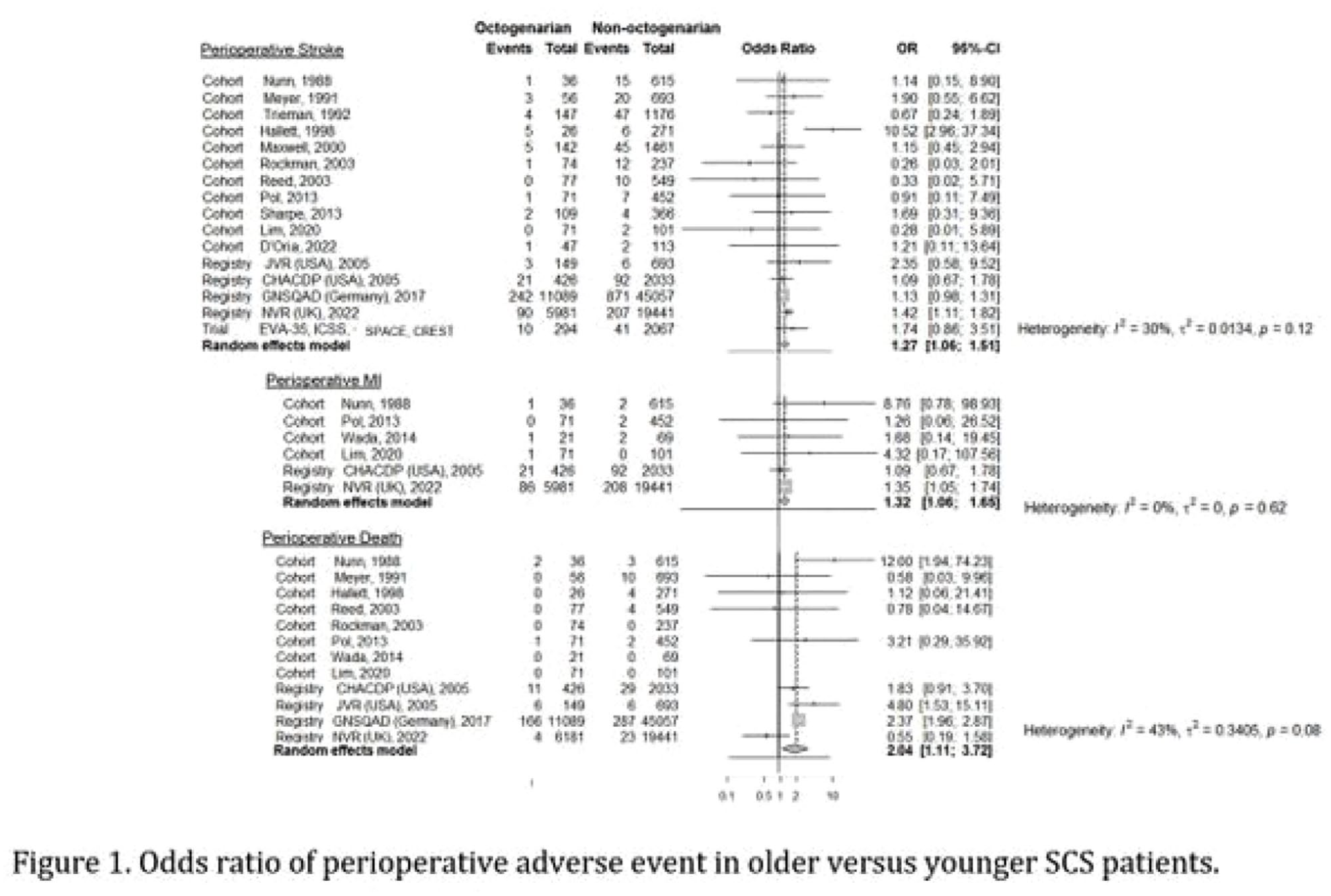
SAFETY AND EFFICACY OF CAROTID ENDARTERECTOMY IN ELDERLY WITH SYMPTOMATIC CAROTID STENOSIS - A SYSTEMATIC REVIEW AND META-ANALYSIS WITH INDIVIDUAL PATIENT DATA
1University of Oxford, Nuffield Department of Clinical Neurosciences, Oxford, United Kingdom, 2University of Oxford, Centre for Prevention of Stroke and Dementia, Oxford, United Kingdom
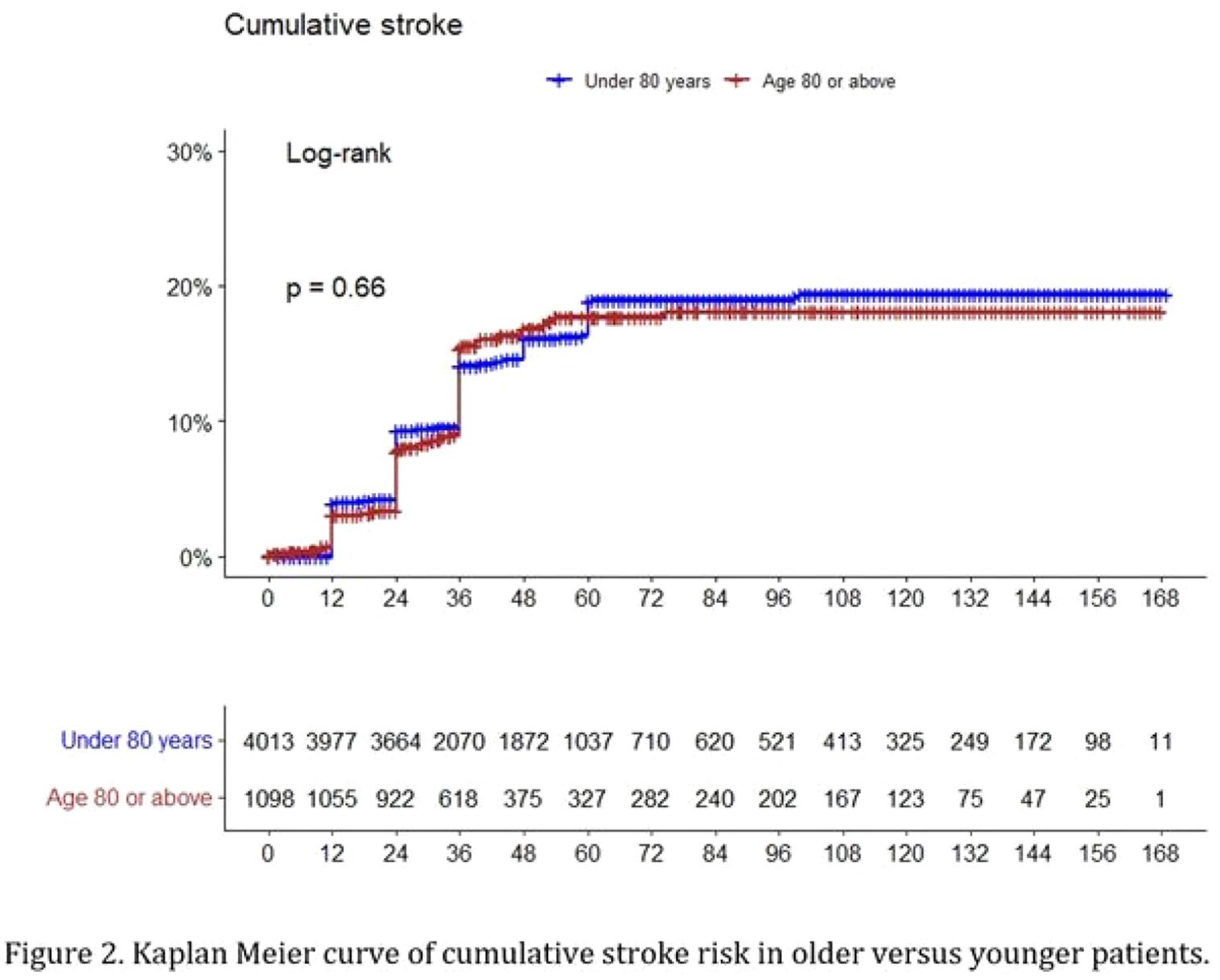
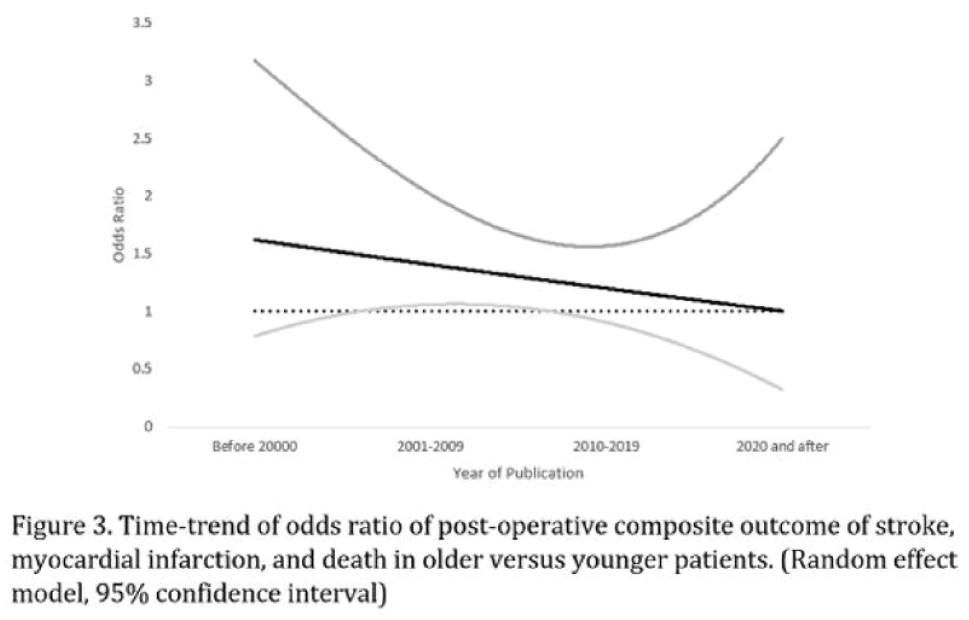
1786
BIOMARKER S100B DURING CAROTID ENDARTERECTOMY
1 University Medical Centre Maribor, Vascular Surgery, Maribor, Slovenia
concentration of S100B protein. Selective shunting was performed when neurological changes occurred, regardless of NIRS.
1788
TYPE 2 DIABETES CHANGES THE IMMUNOMODULATORY AND NEUROMODULATORY EFFECTS OF BRADYKININ IN A MOUSE MODEL OF CEREBRAL AND RETINAL ISCHEMIA
1University of Zagreb School of Medicine, Croatian Institute for Brain Research, Zagreb, Croatia, 2Sestre Milosrdnice University Hospital Center, Department of Ophthalmology, Zagreb, Croatia
1789
MAKING THE INVISIBLE VISIBLE: THE EXPERIENCES OF MILD STROKE PATIENTS
1 Paris-Cité, Department of Neurology and Stroke Center, APHP Bichat Hospital, Paris, France
1792
BLOOD-BRAIN BARRIER LEAKAGE IN CEREBRAL AMYLOID ANGIOPATHY
1Leiden University Medical Center, Neurology, Leiden, Netherlands, 2Leiden University Medical Center, Radiology, Leiden, Netherlands, 3Maastricht University Medical Center, Radiology, Maastricht, Netherlands, 4J. Philip Kistler Stroke Research Center, Neurology, Boston, United States, 5MassGeneral Institute for Neurodegenerative Disease, Charlestown, United States, 6Zuyderland Hospital Heerlen, Neurology, Heerlen, Netherlands, 7Maastricht University Medical Center, Neurology, Maastricht, Netherlands
1794
EFFECT OF INTRA-ARTERIAL ALTEPLASE FOLLOWING SUCCESSFUL THROMBECTOMY ON BRAIN IMAGING: A NESTED STUDY OF THE CHOICE RANDOMIZED TRIAL
1Hospital Clínic, Neurology, Barcelona, Spain, 21 Institut d’Investigacions Biomèdiques Agustí Pi i Sunyer (IDIBAPS), Barcelona, Spain, 3Hospital Clínic, Barcelona, Spain, 4Hospital Universitari Germans Trias i Pujol, Badalona, Spain, 5Institut d’Investigacions Biomèdiques de Barcelona-Consejo Superior de Investigaciones Científicas, Barcelona, Spain
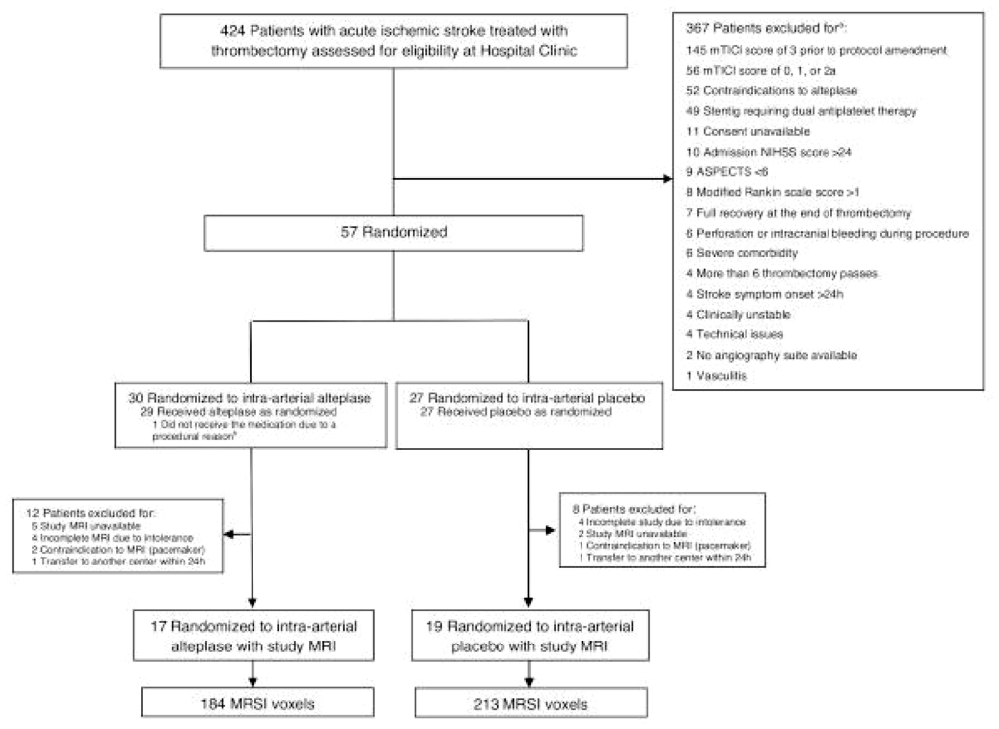
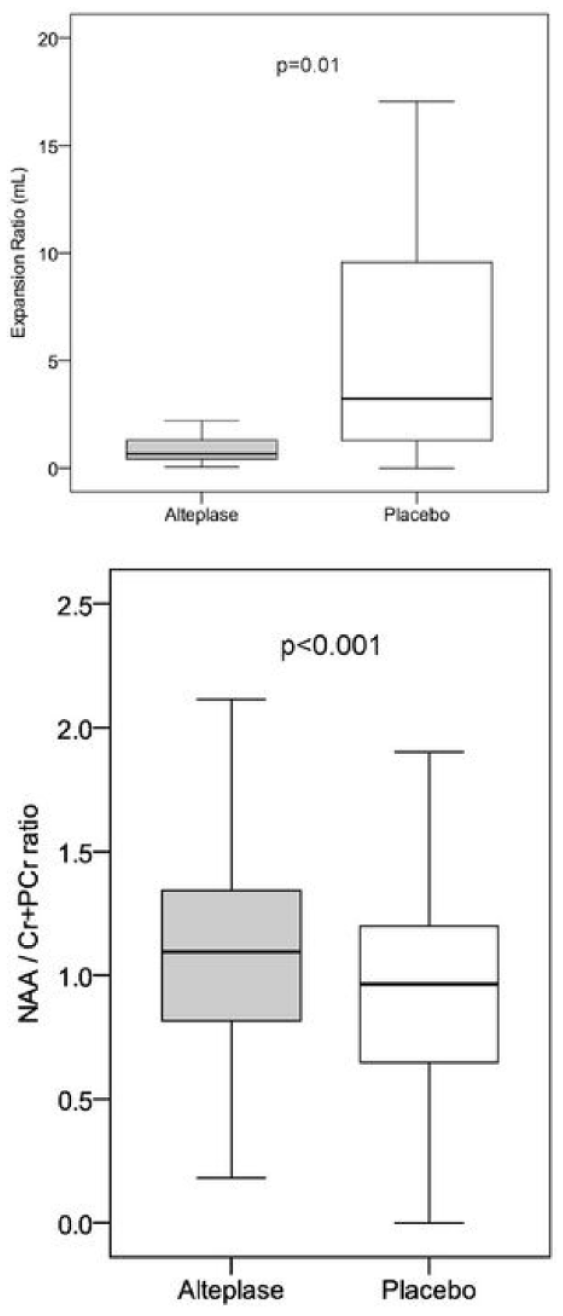
1795
INFECTIONS IN PATIENTS WITH ACUTE STROKE
1Riga Stradins University, Faculty of Medicine, Rīga, Latvia, 2Riga Stradins University, Faculty of Infectology, Rīga, Latvia
The aim of this study was to investigate infections and their frequency in patients with acute stroke.
1797
THE MILD STROKE STUDY 3: LONGITUDINAL STUDY OF CEREBRAL SMALL VESSEL DISEASE IN PATIENTS PRESENTING WITH MILD ISCHAEMIC STROKE
1 University of Edinburgh, Edinburgh Imaging and the UK Dementia Research Institute, Centre for Clinical Brain Sciences, Edinburgh, United Kingdom
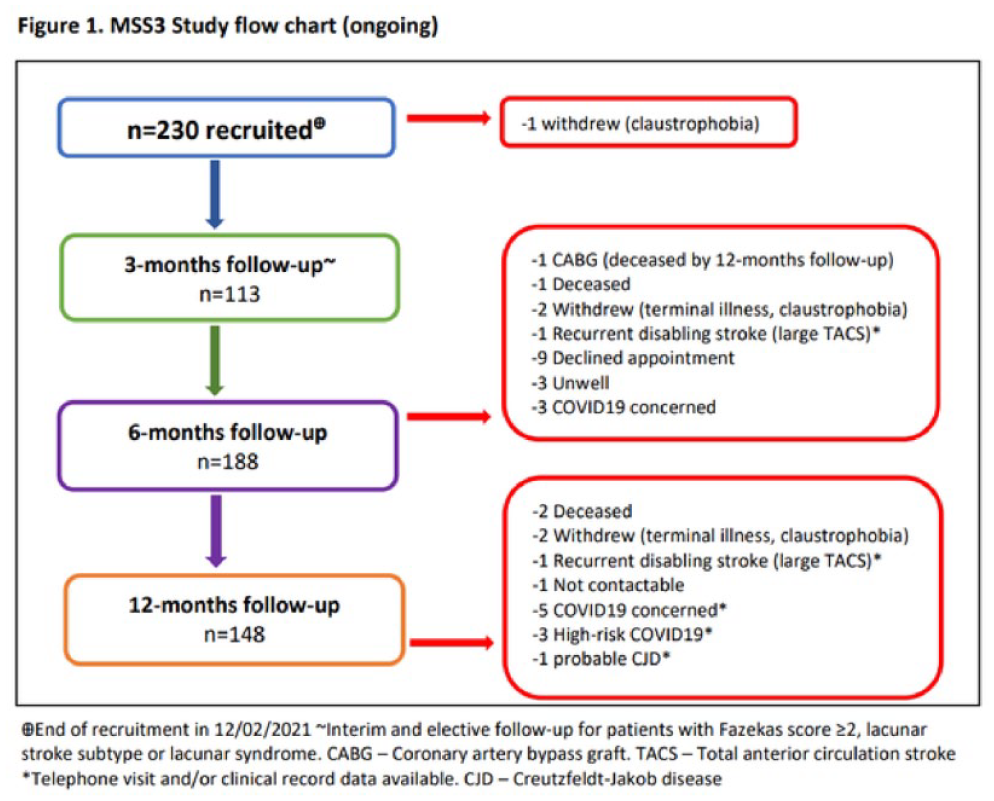
1798
EARLY MINIMALLY-INVASIVE ENDOSCOPY-GUIDED SURGERY FOR SPONTANEOUS SUPRATENTORIAL INTRACEREBRAL HAEMORRHAGE: THE DUTCH ICH SURGERY TRIAL (DIST) PILOT STUDY
1Department of Neurology, Donders Institute for Brain, Cognition and Behaviour, Radboud University Medical Center, Nijmegen, Netherlands, 2Department of Neurosurgery, Radboud University Medical Center, Nijmegen, Netherlands, 3Amsterdam UMC location University of Amsterdam, Neurosurgery, Meibergdreef 9, Amsterdam, Netherlands, 4Amsterdam Neuroscience, Neurovascular disorders, Amsterdam, Netherlands, 5Department of Neurology, Haaglanden Medical Center, The Hague, Netherlands, 6Department of Neurology, Medisch Spectrum Twente, Enschede, Netherlands, 7Department of Neurology, Academic Medical Center, Amsterdam, Netherlands, 8Department of Neurology, Erasmus MC University Medical Center, Rotterdam, Netherlands, 9Department of Neurology, Isala Hospital Zwolle, Zwolle, Netherlands, 10Department of Neurology and Neurosurgery, University Medical Center, Utrecht, Netherlands, 11Department of Public Health, Erasmus MC University Medical Center, Rotterdam, Netherlands, 12Department of Neurology, Elisabeth-Twee Steden Ziekenhuis, Tilburg, Netherlands, 13Department of Medical Imaging, Radboud University Medical Center, Nijmegen, Netherlands, 14Department of Neurology, Maastricht University Medical Center, Maastricht, Netherlands, 15Department of Neurology, Leiden University Medical Center, Leiden, Netherlands, 16Department of Neurosurgery, Erasmus Medical Center, Erasmus MC Stroke Center, Rotterdam, Netherlands
1799
TWIST (TENECTEPLASE IN WAKE-UP ISCHAEMIC STROKE TRIAL)
1University Hospital of North Norway, Department of Clinical Research, Tromsø, Norway, 2University Hospital of North Norway, Department of Neurology, Tromsø, Norway, 3UiT The Arctic University of Norway, Department of Clinical Medicine, Tromsø, Norway, 4Hospital of Southern Norway, Department of Neurology, Kristiansand, Norway, 5Region Skåne, Division of Health Care Management, Malmö, Sweden, 6Lund University, Department of Neurology, Lund, Sweden, 7Uppsala University, Department of Medical Sciences, Neurology, Uppsala, Sweden, 8St. Olavs Hospital, Trondheim University Hospital, Department of Medicine, Trondheim, Norway, 9Bispebjerg Hospital and University of Copenhagen, Department of Neurology, Copenhagen, Denmark, 10Helsinki University Hospital and University of Helsinki, Department of Neurology, Helsinki, Finland, 11Vilnius University, Department of Neurology and Neurosurgery, Center for Neurology, Vilnius, Lithuania, 12Christchurch Hospital, Department of Neurology, Christchurch, New Zealand, 13Riga East University Hospital, Department of Neurology and Neurosurgery, Riga, Latvia, 14Karolinska University Hospital, Department of Neurology, Stockholm, Sweden, 15Karolinska Institutet, Department of Clinical Neuroscience, Stockholm, Sweden, 16Royal Melbourne Hospital, University of Melbourne, Melbourne Brain Centre, Melbourne, Australia, 17University of Basel, Neurology and Neurorehabilitation, University Department of Geriatric Medicine Felix Platter, Basel, Switzerland, 18University Hospital of Basel and University of Basel, Department of Neurology, Basel, Switzerland, 19UiT The Arctic University of Norway, Department of Community Medicine, Tromsø, Norway, 20UCL Queen Square Institute of Neurology, Stroke Research Centre, London, United Kingdom, 21University of Leicester, Department of Cardiovascular Sciences and NIHR Biomedical Research Centre, Leicester, United Kingdom
1800
ACTIVE EXOSKELETON SINGLE-JOINT HYBRID ASSISTIVE LIMB® (HAL) IMPROVES UPPER LIMB FUNCTION AFTER STROKE: A SINGLE CASE EXPERIMENTAL CLINICAL (SCED) STUDY
1Hospices Civils de Lyon, Hôpital Henry Gabrielle, Service de médecine physique et réadaptation, Saint Genis Laval, France, 2Hospices Civils de Lyon, Hôpital Henry Gabrielle, Mouvement et Handicap, Saint Genis Laval, France, 3Hospices Civils de Lyon, Hôpital Henry Gabrielle, Service de médecine physique et réadaptation, Saint Genis Lavalfr, France, 4Université de Lyon, Université Lyon 1, INSERM U1028 ; CNRS UMR5292 ; Lyon Neuroscience Research Center, Trajectoires Team, Lyon, France
1802
COGNITION AS A MEDIATOR OF ACTIVITY LEVELS AND DEPRESSION IN CHRONIC STROKE
1 University of Oxford, Department of Experimental Psychology, Oxford, United Kingdom
