Abstract
Introduction
The risk of intracerebral haemorrhage (ICH) associated with hypertension (HTN) is well documented. While the prevalence of HTN increases with age, the greatest odds ratio (OR) for HTN as a risk for ischemic stroke is at an early age. We sought to evaluate if the risk for ICH from HTN was higher in the youngest patients of each race.
Patients and methods
The Ethnic/Racial Variations of ICH (ERICH) study is a prospective multicenter case-control study of ICH among whites, blacks, and Hispanics. Participants were divided into age groups based on race-specific quartiles. Cases in each race/age group were compared to controls using logistic regression (i.e., cases and controls unmatched). The probability of ICH among cases and controls for each race were compared against independent variables of HTN, quartile of age and interaction of quartile and age also using logistic regression.
Results
Overall, 2033 non-lobar ICH cases and 2060 controls, and 913 lobar ICH cases with 927 controls were included. ORs were highest in the youngest age quartile for non-lobar haemorrhage for blacks and Hispanics and highest in the youngest quartile for lobar haemorrhage for all races. The formal test of interaction between age and HTN was significant in all races for all locations with the exception of lobar ICH in whites (p = 0.2935).
Discussion
Hypertension is a strong independent risk factor for ICH irrespective of location among persons of younger age, consistent with the hypothesis that first exposure to HTN is a particularly sensitive time for all locations of ICH.
Introduction
Intracerebral haemorrhage (ICH) is well recognized as a devastating neurological disease, and it is the subtype of stroke with the highest associated morbidity and mortality. Hypertension (HTN) has long been associated with ICH, and according to recent SMASH-U classification, up to 35% of all ICH can be etiologically attributed to HTN. 1 While the prevalence of hypertension increases with age, the risk for stroke from hypertension varies. For example, in small vessel ischemic stroke, 45 to 65 year olds have a 2.29 fold higher risk of stroke from hypertension compared to only 1.28 times higher risk for those greater than 74 years old. 2 Thus, while the actual prevalence of hypertension increases with age, it may be that impact of hypertension as a risk factor for stroke varies by age. Because ICH shares many of the same risk factors as lacunar small vessel stroke, representing a spectrum of cerebral small vessel diseases, this pattern may be present with ICH as well.
One of the theories to explain this phenomenon involves the autoregulatory response to different blood pressures. Over many years of exposure, it is established that the autoregulatory curve shifts to a new steady state, maintaining stable cerebral blood flow for a range of higher blood pressures.3–6 Yet the period of first exposure, before the curve has had adequate time to shift, may be a time of particular risk. This effect proposed in lacunar small vessel ischemic stroke could similarly explain a potential for increased risk of developing ICH related to HTN as well. 7 Alternatively, it may be that the aging process introduces poorly defined “wear and tear” on blood vessels and other bodily systems that gradually overwhelms traditionally defined risk factors, even among generally healthy individuals.
Because of this, we sought to investigate whether the relative potency of HTN as a risk for ICH was highest in the youngest compared with the oldest patients in each race, and describe differences seen in the risk attributed to HTN based on race and location of haemorrhage. Additionally, for those patients who had HTN diagnosis data available, we sought to identify the risk of ICH based on years from diagnosis of HTN, to assess whether the time nearest to diagnosis is associated with higher risk.
Methods
Ethnic/Racial Variations of Intracerebral Hemorrhage (ERICH) study methods have been described previously. 8 In brief, ERICH is a multicenter, prospective, case-control study, designed to recruit 1,000 non-Hispanic white patients, 1,000 non-Hispanic black patients, and 1,000 Hispanic patients with ICH, along with matched ICH-free controls, for the identification of genetic and epidemiologic risk factors for ICH and outcomes after ICH. Participants were recruited from 19 US sites comprising 42 hospitals. Controls were identified through random digit dialing to match cases by age (±5 years), sex, race/ethnicity, and geographic area. Inclusion into the study requires age >18 years; residency within 50 miles of the recruitment center (100 miles for population centers <1 million); white, black, or Hispanic race/ethnicity; and a spontaneous ICH, defined as the sudden onset of severe headache, altered level of consciousness, or focal neurologic deficit associated with a focal collection of blood within the brain parenchyma, seen on neuroimaging or at autopsy, and not attributable to haemorrhagic conversion of a cerebral infarction or other structural vascular anomalies. All participants or designated proxies underwent a standardized data collection protocol including a personal interview and medical chart abstraction. During the study period, each center reviewed 6 months of true ICH cases that were not enrolled (patient/proxy refusal, out of time window, died before enrolling). Over the 6 month period, there were 388 cases that were true ICH cases but were not enrolled. The study over-recruited to meet the demographic targets and for exclusion of not-a-case after phenotype verification steps. Overall, enrolled cases were younger, more often minorities, less often on Medicare and had a higher mortality rate. However, there were no differences in hypertension, anticoagulant use by abstract/interview, heavy drinking or discharge disposition. Further details are provided in Supplementary Table 1.
All subjects had neuroimaging confirmation of the ICH. A centralized neuroimaging core reviewed all imaging to confirm the diagnosis of spontaneous, non-traumatic ICH, and to determine location (lobar or non-lobar). Location of ICH was adjudicated by each enrolling site and by a blinded neuroimaging trained reviewer. Arterial maps of expected deep versus lobar arterial supplies were used, with the location being determined by where the epicenter of the haemorrhage was closest to. Discrepancies were reviewed by a panel of experts. The initial plan set forth in the ERICH protocol was for at least 1 of every 5 enrollees to undergo MRI scan, if they didn’t receive one as clinical standard of care. Overall, more than 50% of the cohort had an MRI scan available for review. Further details on neuroimaging diagnostics are included in the ERICH study protocol. Additionally, while one of the goals of ERICH was to investigate genetic variations that increase risk of ICH, another primary goal was to identify differences in the risk factors and imaging characteristics of ICH in a multiracial cohort. As it was outside the scope of the present analysis, no genetic information was analyzed.
Variable definition
The ERICH protocol used standardized definitions for hypertension, diabetes, hypercholesteremia, anticoagulation use, antiplatelet use, statin use, length of stay, ICH score, EVD placement, craniotomy, do-not-resuscitate (DNR) order and comfort-measures-only (CMO) order. These were additionally confirmed by self report on the case and control interview.
For the purposes of this analysis, both cases and controls were asked if they had ever been diagnosed with hypertension and they were asked to provide a list of all medications they took prior to ICH for cases or prior to interview for controls. Hypertension itself was defined by the self-report question on the interview questionnaire. From the list of medications taken prior to the stroke, if the list included any of ace inhibitors, alpha blockers, alpha adrenergic agonist, angiotensin II receptor blocker, beta blocker, calcium channel blocker, diuretic, renin inhibitor, vasodilator, antianginal nitrate or non-specific antihypertensive, then they were considered treated hypertension. If they did not take any of the anti-hypertensives listed and were hypertensive by self-report they were considered untreated hypertension. Elevated blood pressure at the time of study enrollment was not used to define “hypertension” for the present analysis because, for cases, ICH itself can result in elevated blood pressure. The cases and controls were also asked the date of diagnosis of hypertension. For cases the years since diagnosis is calculated from the date of diagnosis to the date of ICH. For controls it is calculated from the date of diagnosis to the date of interview.
Statistical analysis
Descriptive statistics are presented by lobar and non-lobar locations separately. They are presented as frequencies and percentages for all variables except ICH volume which is presented as median and interquartile range. Initially, it was planned that the entire cohort would be divided into quartiles for the analysis, but the ages of the separate ethnicities/races within each cohort was too disparate to allow for an appropriate analysis. Therefore, the quartiles of age were determined by each race individually and age is presented as percentages in quartiles. Additionally, analyzing race specific quartiles’ risk of ICH would eliminate possible race/ethnicity related confounders that would be included when analyzing whole cohort quartiles. The cases and controls are compared by race. The percentages of hypertensive individuals in each quartile in cases and controls are presented in Tables 1 and 2 along with OR(CI) for the probability of ICH in each quartile by race. Pairwise comparisons of the quartiles are also provided.
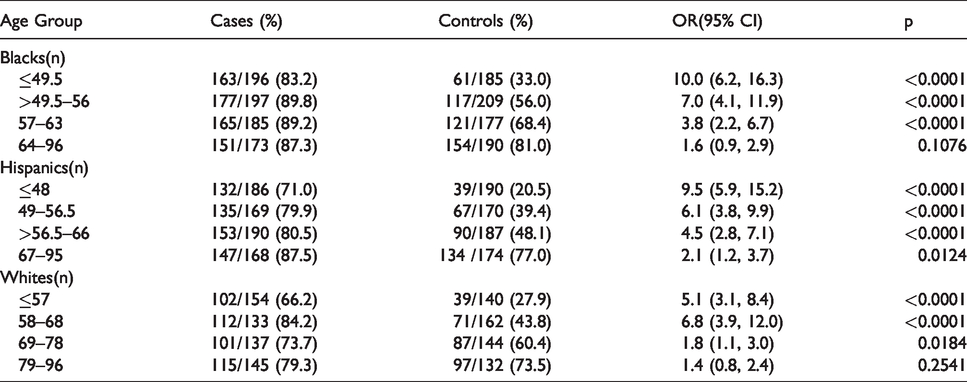
Risk of non-lobar ICH related to HTN.
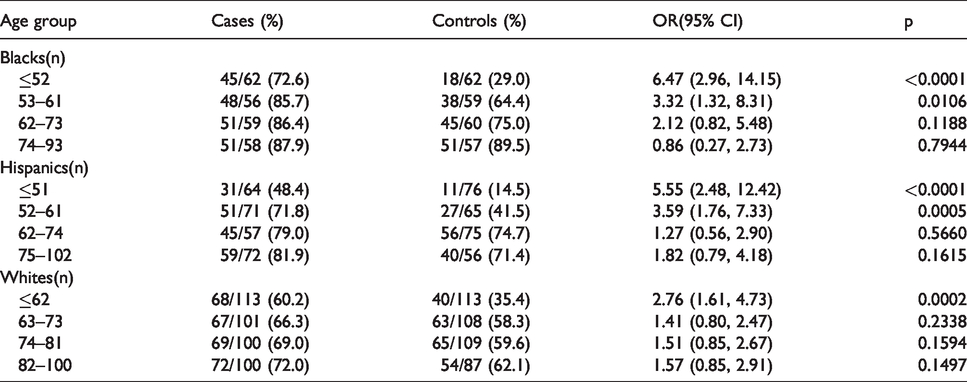
Risk of lobar ICH related to HTN.
Logistic regression models, modeling the probability of ICH using only those who had previously been diagnosed as hypertensive were constructed in lobar and non-lobar locations against decades since diagnosis of hypertension. The predicted probabilities along with upper and lower 95% confidence levels from those models were output and graphed against decades since diagnosis of hypertension. The test for overall interaction of HTN by quartiles was done by logistic regression modeling the probability of ICH against presence of hypertension, quartile of age and interaction of hypertension and quartile of age separately in lobar and non-lobar ICH by each race/ethnic category.
Results
A total of 3,000 ICH patients were enrolled in the ERICH study. Enrolled ICH cases were equally distributed across blacks (n = 1000), whites (n = 1000) and Hispanics (n = 1000). These were stratified based on location of ICH, either lobar or non-lobar and the baseline characteristics for cases and controls are seen in Supplementary Tables 2 and 3.
Demographic and baseline characteristics for non-lobar ICH cases and controls were divided by race. There were 2033 cases included, with 2060 controls. There were 569 white ICH cases, 751 black cases and 713 Hispanic cases included in the analysis. Overall, cases had significantly more HTN (81.3% vs 52.3% p=<0.0001) and significantly more untreated HTN (36.6% vs 6.0%, p=<0.0001). They were more likely to use alcohol heavily, take warfarin and to have a family history of ICH. They were less likely to be insured, and had less education. They were also less likely to have hyperlipidemia. Whites did not have a statistically significant difference between cases and controls regarding hyperlipidemia, but both blacks and Hispanics did (see supplementary table for more information).
Demographic and baseline characteristics for lobar ICH cases and controls were also divided by race. There were 913 cases included in the analysis, with 927 controls. Of these, 414 were white, 235 were black and 264 were Hispanic. Overall, the cases were more likely to have hypertension and more likely to have untreated HTN. They were also more likely to use alcohol heavily, use warfarin or other anticoagulants (not including directly acting oral anticoagulants). They were less likely to have advanced education and were less likely to be insured. There was no statistically significant difference in diabetes or hyperlipidemia, antiplatelet use and family history of ICH (see supplementary table for more information).
Table 1 shows the unadjusted odds ratios of non-lobar ICH from HTN separated by race. The quartiles were created based on each individual race’s age composition, and therefore the ages that define the youngest quartile for blacks (≤49.5) is different than Hispanics (≤48) and different from whites (≤57). Overall, whites were older than both blacks and Hispanics leading to a youngest age difference between quartiles of almost 10 years. The OR for blacks was highest in the youngest quartile (OR = 10 (95% CI 6.2–16.3)) and decreased in subsequent quartiles, with the oldest quartile (64–96 yo, OR = 1.6 (95% CI 0.9–2.9), p = 0.1076) being the only quartile which was not statistically significant. Hispanics similarly show the highest risk in the youngest quartile (OR= 9.5, (95% CI 5.9–15.2)) and decreased with increasing age, but all quartiles show a statistically significant interaction with HTN. Whites also have higher OR point estimates (OR = 5.1 for the youngest quartile, OR= 6.8 in the second youngest quartile) in the younger two quartiles than the oldest two (OR 1.8 for 69–78 yo, OR = 1.4 for 79–96 yo) and all but the oldest quartile were statistically significant.
Table 2 shows the unadjusted odds ratios of lobar ICH from HTN separated by race. Again, the quartiles were divided based on each individual race’s age composition, with the youngest black quartile being ≤52 years old, Hispanics ≤51 years old and whites ≤62 years old. As with non-lobar ICH, white quartiles were roughly 10 years older than both blacks and Hispanics. The OR for the youngest black quartile was 6.47 (95% CI 2.96, 14.15) and was similar to the youngest Hispanic quartile (OR = 5.55 (95% CI 2.48–12.42)). The second quartiles for both were lower (OR = 3.32 for blacks, OR = 3.59 for Hispanics) and both were statistically significant. The oldest two quartiles for both blacks and Hispanics did not reach statistical significance. Whites also had a statistically significant youngest quartile (OR = 2.74, 95% CI 1.61–4.73, p = 0.0002) but a lower point estimate than the other two races. No other quartile for whites reached statistical significance.
Table 3 shows the quartile comparisons of risk for non-lobar ICH for each race and overall p-value for interactions between quartiles for each race. Blacks and Hispanics had the highest OR seen between quartiles 1 and 4 (OR= 2.72 for blacks, OR = 3.08 for Hispanics), with decreased risks seen for the comparison between quartile 1 and quartiles 2 and 3. Additionally, there was a larger risk for quartile 2 compared to quartile 4 for blacks and Hispanics (OR= 1.54 for blacks, OR = 1.84 for Hispanics) than quartile 2 compared to quartile 3 (OR= 1.11 for blacks, OR = 1.18 for Hispanics).The p-value for the overall trend was significant in all races.
Comparison of different quartiles within individual races of non lobar ICH risks.
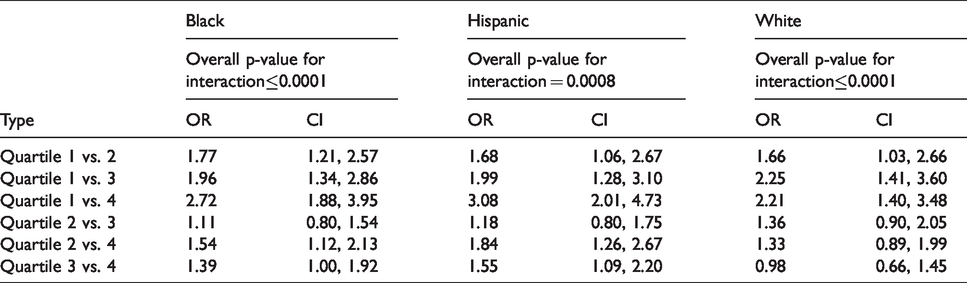
Table 4 shows the quartile comparisons of risk for lobar ICH for each race and overall p-value for interactions between quartiles for each race. The highest OR for blacks was seen comparing quartile 1 vs quartile 4 (OR = 2.50), with decreasing OR as the comparison in ages because closer (quartile 1 vs quartile 3 OR = 2.21, quartile 1 vs quartile 2 OR = 1.98). The highest OR for Hispanics was seen when quartile 1 was compared to quartile 3 (OR = 3.51), followed by when quartile 2 was compared to quartile 3 (OR = 2.35). Whites had the highest OR when comparing quartile 1 vs quartile 2 and quartiles 1 vs quartile 3 (OR = 1.60), with all OR confidence intervals crossing 1. The p-value for the overall trend was significant for blacks and Hispanics (p = 0.0326, p = 0.0466 respectively). The p-value for the overall trend for lobar ICH related to HTN for whites was the only location and race where the interaction did not reach statistical significance (p = 0.2935).
Comparison of different quartiles within individual races of lobar ICH risks.
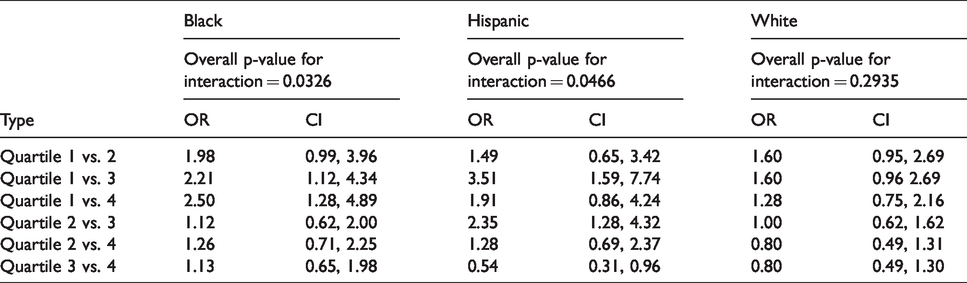
Figure 1 is a graph comparing the predicted probability of ICH to the difference between the time from diagnosis of HTN to ICH/interview in decades. The graph illustrates the steady increase in the probability of non-lobar ICH in the decades closest to the diagnosis of hypertension. The confidence interval around the probability line is narrowest between the first and second decade before the development of ICH. The probability of non-lobar ICH significantly increases in the decades nearest to the ICH/interview. While there is a similar trend in the probability of lobar ICH, it is not statistically significant and not presented here.
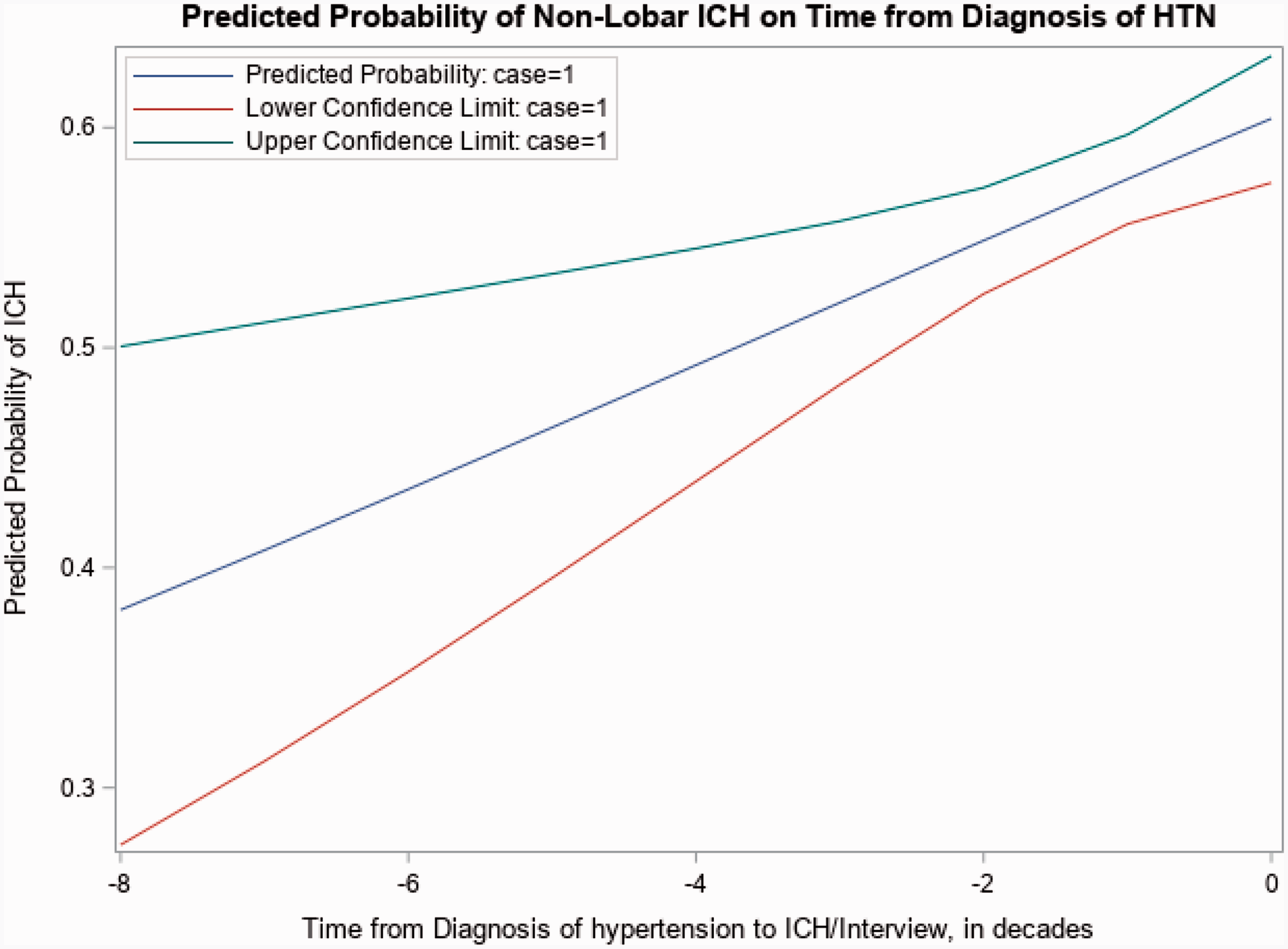
Predicted probability of non-lobar ICH on time from diagnosis of HTN.
Discussion
The present study demonstrated that hypertension is a larger independent risk factor for younger patients versus older patients in developing ICH, regardless of race or location of haemorrhage and that the risk of developing a non-lobar ICH is higher in the time closest to the diagnosis of HTN. This is consistent with our hypothesis that as cerebral vasculature is first exposed to higher blood pressures, the auto-regulatory curve has not yet shifted and the first exposures to these elevated pressures are a particularly high-risk time for developing ICH, identical to its relationship to small vessel ischemic stroke.
The findings we present in non-lobar haemorrhage are striking, showing that HTN in the youngest quartiles of all races equates to a roughly 8x higher risk of developing an ICH at younger ages. This was somewhat to be expected, as the association between HTN and non-lobar ICH has been demonstrated in numerous studies.9–12 However, evaluating the risk between separate age groups and demonstrating that the OR for the youngest versus the oldest in all races is greater than 2, and that the overall trend comparing risk between all age quartiles is significant has not been previously reported extensively. Additionally, when looking at the regression model of risk of ICH compared to time from diagnosis of HTN, it is consistent with our hypothesis that the initial period of HTN is an asymmetric period of higher risk of non-lobar ICH prior to adaptation of cerebral autoregulation.
The findings shown here in lobar haemorrhage, however, are more surprising. Especially in blacks and Hispanics, the OR in the youngest quartiles shows a remarkable increase compared to older quartiles (OR of 6.47 for blacks, 5.72 for Hispanics). The risk in the youngest quartile of whites is also elevated, but when evaluating the overall interaction, the trend for lobar haemorrhage related to HTN in whites is not significant. This is the only race and ICH location for which the overall trend that the younger quartiles are at higher risk than older quartiles was not significant. The surprisingly high OR seen for blacks and Hispanics may be related to the relatively lower contribution that the APOE genotype has been shown to have for developing lobar ICH in these patients. 13
The risk of developing ICH at different ages has been examined, but somewhat sparingly, finding that the risk of ICH increases with increasing age.14–17 However, studies have shown that risk attributable to HTN of developing an ICH is higher in younger patients. One such study done in Australia showed that HTN produced greater ORs for ICH for individuals younger than 55 than those older, however it was limited by a somewhat small number of cases. 18 Additionally, a study done in Japanese patients demonstrated that the odds ratio for ICH with HTN is higher in individuals aged ≤69 than in those aged ≥70. 19
While etiologies for lobar and non-lobar haemorrhages have classically been thought and shown to be different (cerebral amyloid angiopathy (CAA) and APOE ε2 or ε4 genotype contributing to a high proportion of lobar haemorrhages), these studies were done in primarily white populations.20,21 Subsequent research has suggested that some lobar haemorrhages are related to HTN as well, especially in blacks and Hispanics. It has been shown that both blacks and Hispanics have a lower attributable risk of developing ICH related to APOE ε2 or ε4 genotypes, 13 suggesting that lobar haemorrhages might not be as closely related to CAA in these populations.
Another consideration relevant to our findings must be discussed. Identifying HTN as a significant risk factor in younger compared with older patients highlights the fact that younger patients generally have less risk factors than older patients. One of the strongest risk factors identified for ICH, and stroke in general, is age. It may well be that the cumulative impact from a variety of risk factors, the sum of which being greater than the individual components, “overwhelms” the risk imparted solely by HTN and leads to HTN having a relatively lower associated risk in older patients. This theory, however, does not preclude that a disruption in autoregulation in younger hypertensive patients leads to their increased risk of developing ICH compared to non-hypertensive patients.
The presently identified asymmetric risk with HTN in younger patients highlights the need to more aggressively both screen and treat this population. This can be difficult, as younger patients without obvious symptoms related to their HTN may be less inclined to seek out preventive care, and given the high prevalence of HTN in general, the importance and outsized value of aggressively treating HTN in the young from a provider standpoint may not be appropriately realized. Additionally, there are multiple studies22–24 highlighting the effect that untreated HTN in particular can have on the risk of ICH, again emphasizing the importance of aggressively seeking out and treating HTN in younger patients. The value of the present study, and the ERICH study in general, extends to note that there is in fact a racial difference in the risk that HTN poses– suggesting that broader efforts to diagnose and treat a condition that is known to be more prevalent within black and Hispanic communities, which typically are underserved medically for a variety of causes, could have substantially more benefit reducing and possibly preventing the consequences of ICH. Preventing ICH in younger patients also helps to reduce the financial consequences of morbidity and disability related to ICH on a larger societal scale.
The study has several strengths worth noting. First, it uses the largest case-control study of ICH incorporating 3 different races. Cases were identified using a standard definition and methodology and imaging was analyzed centrally, ensuring uniformity in across recruitment centers especially regarding the determination of location of ICH. It is the first study that we are aware of that reports the association between the heightened risk of developing a non-lobar ICH related to HTN closer to the diagnosis of HTN.
This study has limitations. First, this being a case control study, it is susceptible to recall bias. Additionally, despite controlling for certain characteristics expected to influence the evaluation, there may be other variables not included that skew the results. All studies investigating ICH struggle with defining HTN, as most patients present hypertensive during a haemorrhagic stroke, but this difficulty was minimized as much as possible using the standard trial definition used by ERICH.
In this study, we demonstrate that HTN is a significant risk factor for both lobar and non-lobar ICH, regardless of race, but also that the youngest compared to the oldest patients are at increased risk. This highlights the importance of aggressively screening and treating patients for HTN, and suggests new areas for future research efforts to investigate the racial differences in modifying this highly prevalent risk factor to reduce its impact on global morbidity and mortality suffered by ICH patients young and old.
Supplemental Material
sj-pdf-1-eso-10.1177_2396987321994296 - Supplemental material for Risk of intracerebral haemorrhage from hypertension is greatest at an early age
Supplemental material, sj-pdf-1-eso-10.1177_2396987321994296 for Risk of intracerebral haemorrhage from hypertension is greatest at an early age by Robert Stanton, Stacie L Demel, Matthew L Flaherty, Eleni Antzoulatos, Lee A Gilkerson, Jennifer Osborne, Tyler P Behymer, Charles J Moomaw, Padmini Sekar, Carl Langefeld and Daniel Woo in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MLF is a co-founder and has ownership interest in Sense Diagnostics, Inc., a start-up company developing a non-invasive sensor intended to monitor ICH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health NCT 01,202,864.
Ethical approval
Institutional Review Board approval at each participating center was required before the initiation of study enrollment.
Informed consent
Informed consent was obtained from each enrolled subject or legally authorized representative.
Guarantor
RJS.
Contributorship
RJS, SLD, EA, TPB and DW researched literature and conceived the study. DW, MLF, TPB, LAG, JO, CM, PS, CL was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. RJS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
