Abstract
Background:
Uncontrolled blood pressure (BP) in intracerebral hemorrhage (ICH) survivors is common and associated with adverse clinical outcomes. We investigated whether characteristics of the ICH itself were associated with uncontrolled BP at follow-up.
Methods:
Subjects were consecutive patients aged ⩾18 years with primary ICH enrolled in the prospective longitudinal ICH study at Massachusetts General Hospital between 1994 and 2015. We assessed the prevalence of uncontrolled BP (mean BP ⩾140/90 mmHg) 6 months after index event. We used multivariable logistic regression models to assess the effect of hematoma location, volume, and event year on uncontrolled BP.
Results:
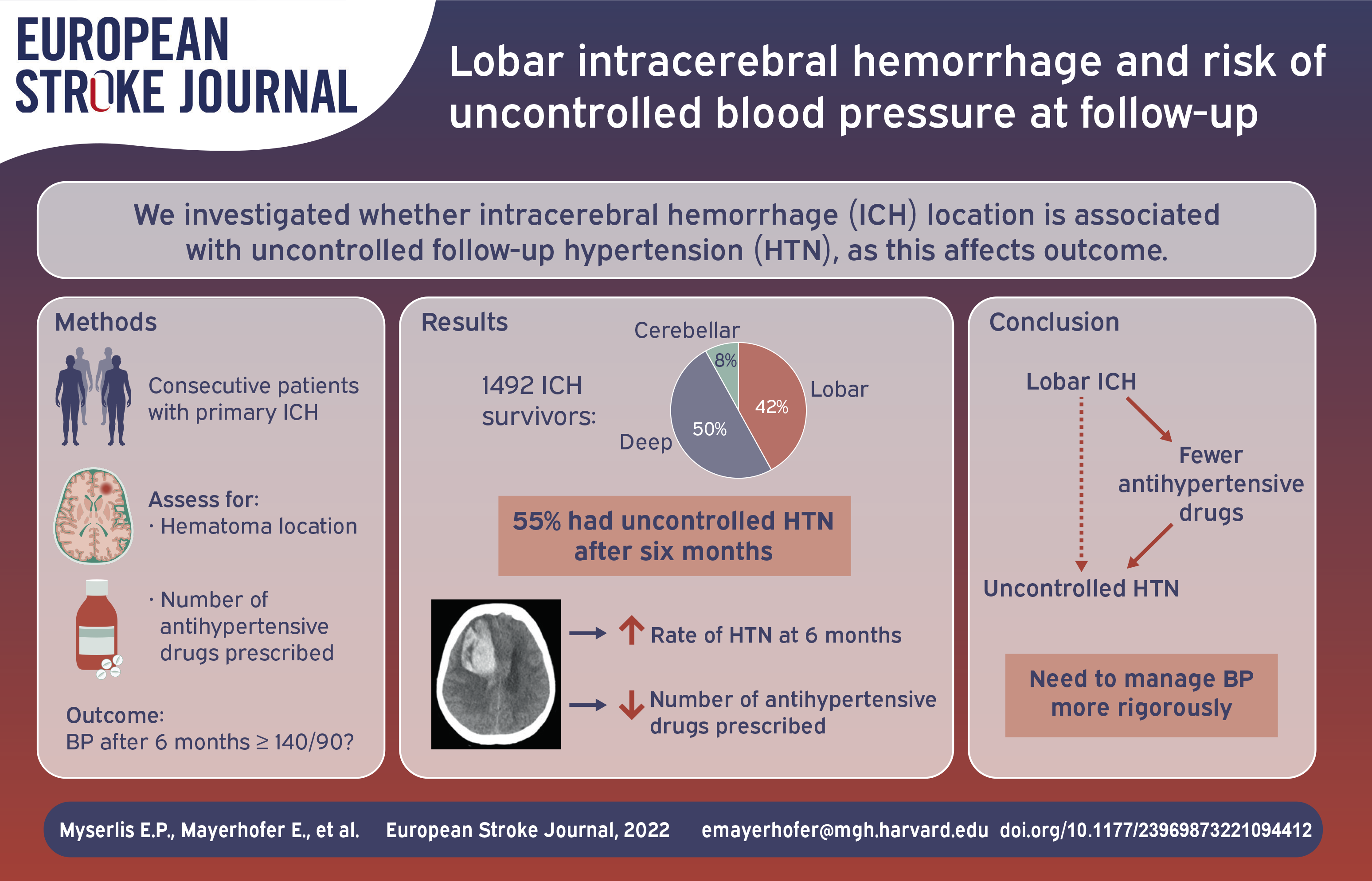
Among 1492 survivors, ICH was lobar in 624 (42%), deep in 749 (50%), cerebellar in 119 (8%). Lobar ICH location was associated with increased risk for uncontrolled BP after 6 months (OR 1.35; 95% CI [1.08–1.69]). On average, lobar ICH survivors were treated with fewer antihypertensive drugs compared to the rest of the cohort: 2.1 ± 1.1 vs 2.5 ± 1.2 (p < 0.001) at baseline and 1.8 ± 1.2 vs. 2.4 ± 1.2 (p < 0.001) after 6 months follow-up. After adjustment for the number of antihypertensive drugs prescribed, the association of lobar ICH location with risk of uncontrolled BP was eliminated.
Conclusions:
ICH survivors with lobar hemorrhage were more likely to have uncontrolled BP after 6 months follow-up. This appears to be a result of being prescribed fewer antihypertensive medications. Future treatment strategies should focus on aggressive BP control after ICH independent of hemorrhage location.
Introduction
Intracerebral hemorrhage (ICH), while accounting for only 10%–20% of all stroke cases, is associated with ~50% of stroke-associated morbidity and mortality.1–3 Survivors are at high risk for more severe recurrent stroke, cognitive decline, and depression as a result of progressive cerebral small vessel disease (CSVD).4–6
Blood pressure after ICH is a strong predictor of severe adverse events such as cognitive decline and recurrent ICH, 7 ischemic stroke, dementia, depression, and gait impairment, 6 and major adverse cardiovascular and cerebrovascular events. 8 Despite the association of high BP with long-term deterioration after ICH7,8 and recommendations in widely endorsed clinical guidelines, inadequate BP control remains a major challenge in this population with rates ranging from 50% to 80%.7,9–11 Additionally, clinical trials of BP control interventions in stroke survivors have persistently failed to improve BP control.12,13 Therefore, gaining better insight into the determinants of better BP control after ICH is critical to developing targeted therapeutic strategies that could effectively preserve brain health in survivors of ICH.
It is likely that rates of uncontrolled BP after ICH are determined by a wide variety of factors, including the BP before the hemorrhage, effects of the hemorrhage itself, the nature of the underlying CSVD, patient co-morbidities, genetics, social determinants of health, and provider behavior. Previous findings from our group demonstrated that the acute, ICH-related hypertensive response is associated with post-ICH BP control levels, potentially reflecting the burden of a chronic state of untreated or undertreated hypertension. 11 We hypothesized that other characteristics available in the acute inpatient setting could provide clinically useful predictive information. The aim of this study was to determine whether hematoma location and volume predict post-ICH BP control in ICH survivors.
Methods
Patient recruitment and baseline data collection
We analyzed post-discharge follow-up data from patients enrolled in the Massachusetts General Hospital (MGH) ICH study, a longitudinal, prospective, single-center study of ICH, between July 1994 and December 2015. 7 Initial inclusion criteria were acute primary ICH (onset of symptoms < 24 h before presentation) confirmed by CT scan, age 18 years or older, and survival of at least 90 days. Patients with secondary hemorrhage resulting from trauma, conversion of ischemic infarct, rupture of a vascular malformation or aneurysm, or brain tumor were excluded.
Baseline data were prospectively collected at index ICH by trained study staff during in-person interview of patients and/or reliable surrogates. Baseline data included age, race/ethnicity, prior medical history [history of atrial fibrillation, diabetes mellitus, hypertension, hypercholesterolemia, previous ischemic cerebrovascular accident, coronary artery disease, admission systolic BP (SBP), and admission diastolic BP (DBP)]. Further data were collected by review of medical records. For admission SBP and DBP, we utilized measurements obtained at any hospital the patient first received medical care; if this information was not available, we utilized measurements obtained during admission at MGH. ICH location and volume were analyzed by study investigators blinded to clinical and follow-up data. Lobar location was defined as one or more ICH involving the cerebral cortex, underlying white matter, or both; deep location was defined as one or more ICH involving the putamen, thalamus, brainstem or primary IVH; cerebellar ICH was defined as ICH involving the cerebellum. 14 The study protocol was approved by the institutional review board at MGH and informed consent was obtained from all participants or their authorized surrogates.
Longitudinal follow-up, blood pressure, and medication data capture
The follow-up data collection process has previously been described in detail. 7 Briefly, ICH survivors and caregivers were contacted and interviewed by trained study staff at 3, 6, 9, and 12 months after index ICH and every 6 months thereafter. Study investigators inquired about the most recent BP measurement recorded in a medical setting (no inquiry was made for home- or self-obtained BP measurement). If patients or caregivers were unable to provide exact measurements, medical records from reported encounters falling within the prespecified follow-up periods were obtained via (1) manual review of the electronic medical records of MGH and affiliated hospitals or (2) by patient-provided external medical records. Patients were assigned a single BP value per each follow-up interval; if multiple measurements were available, the average was used for that follow-up period. Patients were interviewed about drug intake, combined with chart review of the prescriptions. Because prescription data was systematically ascertained at 6 months, we used 6 month BP as our primary endpoint.
Variable definition
Controlled BP after 6 months was the primary outcome, defined as SBP ⩾ 140 mmHg or DBP ⩾ 90 mmHg according to the AHA/ASA recommendations during the study period. 15 For the subset of patients where medication data was available, we divided uncontrolled BP in resistant (⩾3 antihypertensive agents) and undertreated (⩽2 antihypertensive agents) hypertension as recently described. 16 For sensitivity analyzes we used the current 130/80 mmHg cut-off recommended by the 2015 AHA/ASA guidelines for prevention of ICH recurrence. 2
Statistical analysis
Age at index ICH, admission SBP, and admission DBP were analyzed as continuous variables and are expressed as mean ± one standard deviation (SD) or median with interquartile range (IQR), depending on whether they had a normal or skewed distribution assessed by Shapiro-Wilk test. All other variables were analyzed as categorical variables and are expressed as number followed by percentages. Unpaired Student’s t-test and Chi-square test were used in univariable analyzes. Multivariable linear and logistic regression models were used for prediction of outcomes by exposures.
In univariable analyzes, characteristics of patient groups were compared: uncontrolled vs. controlled blood pressure after 6 months follow-up; lobar vs. non-lobar ICH; hematoma volume >30 vs ⩽30 cm3. Variables that differed (p < 0.1) between the patient groups were treated as potential confounders and added to the respective regression models for assessing their association with the outcome of interest. Furthermore, because the observation covered a long time period, ICH event year was included to investigate potential time-dependency of treatment patterns. Only significant predictors of the outcome were kept in the final models for quantification of effect sizes. For investigating the effect of the number of antihypertensive drugs on uncontrolled blood pressure after 6 months, we calculated separate models due to the smaller sample size. All analyzes were performed using R software version 4.1.1 (R Foundation for Statistical Computing), and p < 0.05 (2-tailed) was considered statistically significant.
Results
Study participants
A total of 1690 patients met inclusion criteria. 1492 ICH survivors were available for analysis after exclusion of those with missing CT scan (n = 15), loss to follow-up (n = 177), or mixed lobar and deep hemorrhage (n = 6, Figure 1). Median age was 72 (IQR 62–80) years, 688 (46%) patients were female, and 1272 (85%) were white. There were 624 (42%) ICH survivors with lobar, 749 (50%) survivors with deep, and 119 (8%) with cerebellar ICH. Hematoma volume was available for 1364 (91.4%) of the patients. Median ICH volume was 10 (IQR, 3.5–24) cm3, and 264 (19.4%) patients had an ICH volume ⩾ 30 cm3 (Table 1). Data on antihypertensive prescriptions was available for 779 (52.2%) patients at 6 month follow-up.
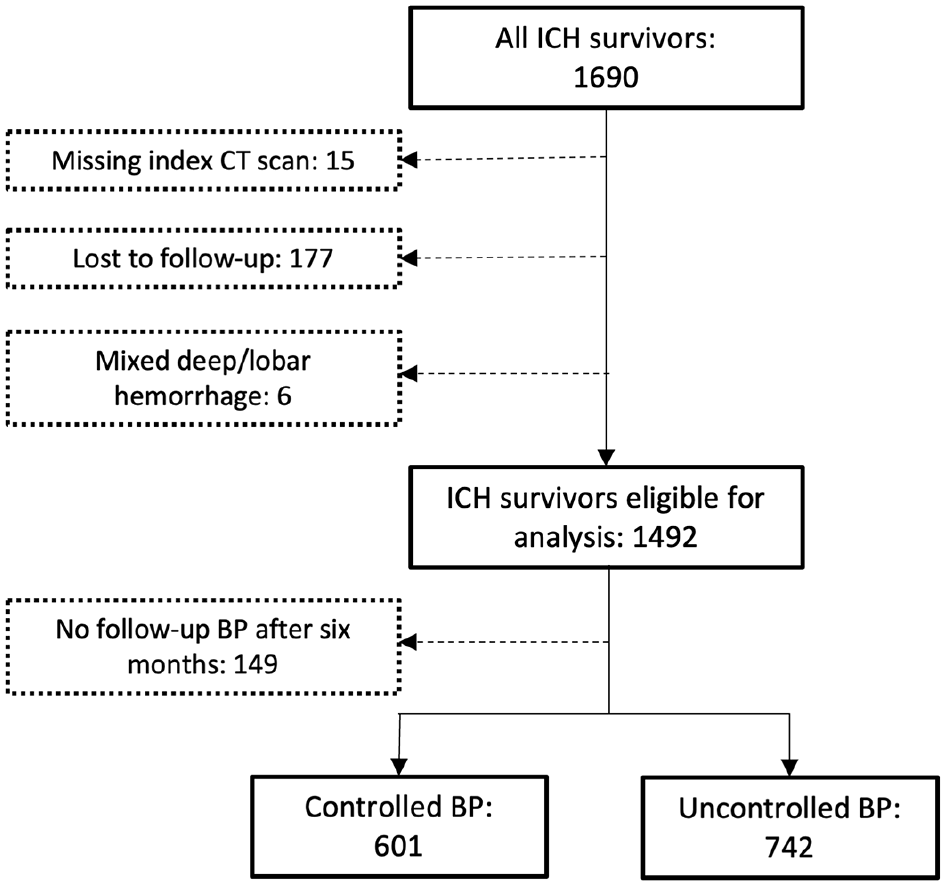
Study flow chart.
Sample characteristics by location of hemorrhage (N = 1492).
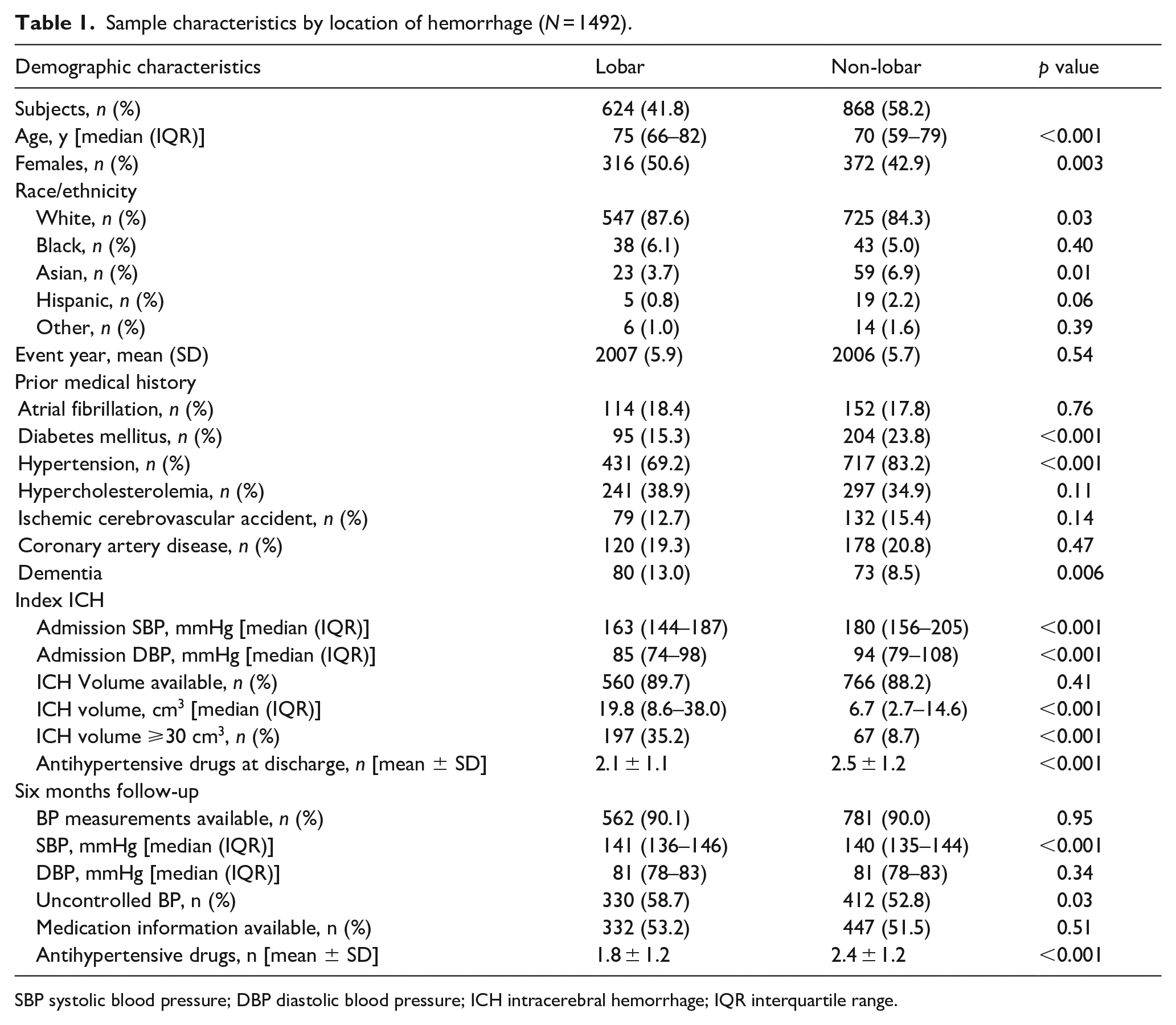
SBP systolic blood pressure; DBP diastolic blood pressure; ICH intracerebral hemorrhage; IQR interquartile range.
Patients with lobar hemorrhage had larger hematomas (19.1 [8–38] cm3) than patients with deep hemorrhage (6.2 [2.0–14.0] cm3, p < 0.001) and those with cerebellar hemorrhage (6.0 [2.0–13.4] cm3, p < 0.001). Similarly, the majority of the patients with hematoma volume ⩾30 cm3 (n = 264, 19.4%) had lobar ICH (n = 197, 74.6%; Table 2). After adjusting for age and sex in a linear regression model, ICH in the lobar location were significantly larger compared to the rest of the sample (β = 16.3 cm3, 95% CI [14.2, 18.4]).
Sample characteristics for patients with available hematoma volume (N = 1326).
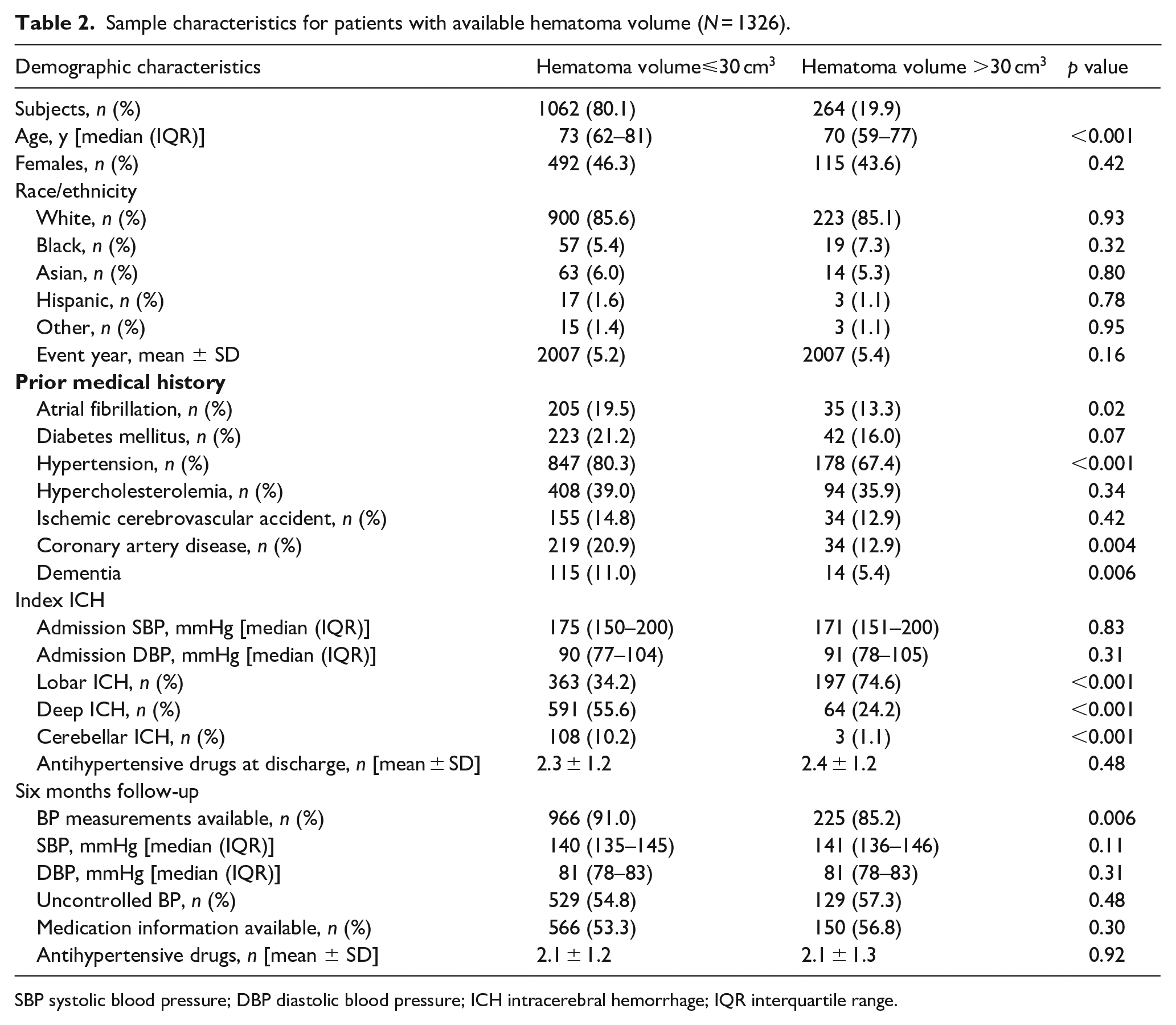
SBP systolic blood pressure; DBP diastolic blood pressure; ICH intracerebral hemorrhage; IQR interquartile range.
Post-ICH BP control status
BP values after 6 months of index ICH event were available for 1343 (90%) of the patients. More than half of the patients had uncontrolled BP after 6 months follow-up (n = 742, 55.1%, Table 3). The prevalence of uncontrolled BP was higher in patients with lobar ICH (n = 330, 58.7%) compared to the rest (n = 412, 52.8%, p = 0.03) but not significantly in patients with hematoma volume ⩾ 30 cm3 (n = 129, 57.3%) compared to the rest (n = 529, 54.8%, p = 0.48).
Baseline parameters of patients with uncontrolled versus controlled BP after 6 months follow-up.
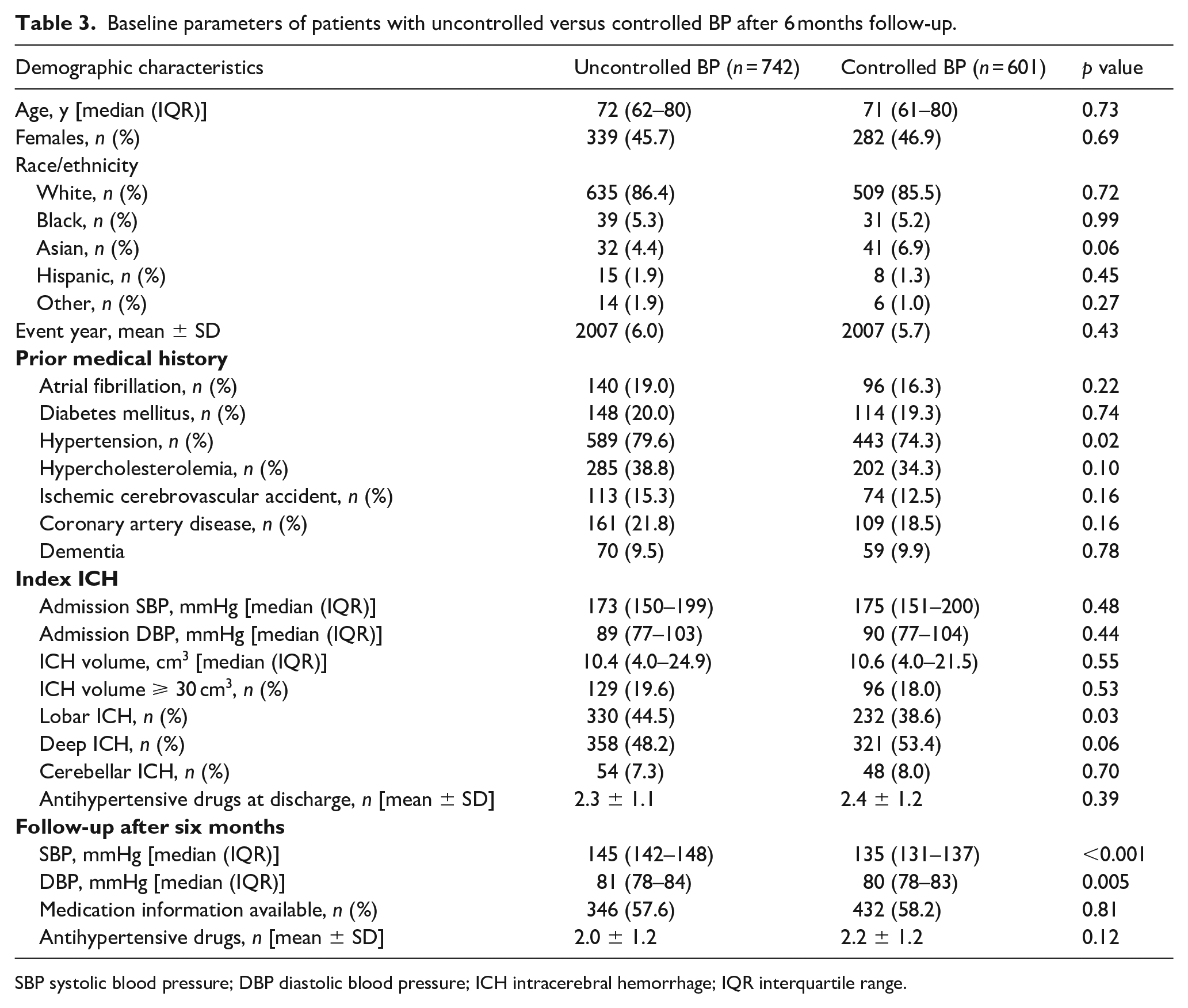
SBP systolic blood pressure; DBP diastolic blood pressure; ICH intracerebral hemorrhage; IQR interquartile range.
We further divided uncontrolled BP into undertreated and resistant BP and assessed their rates after 6 months, where most antihypertensive medication prescriptions were available. Patients with lobar ICH were prescribed significantly lower number of antihypertensive drugs than the rest of the patients: 2.1 ± 1.1 vs 2.5 ± 1.2 (p < 0.001) at baseline and 1.8 ± 1.2 vs 2.4 ± 1.2 (p < 0.001) after 6 months follow-up. As a consequence, the fraction of patients with undertreated BP was higher in those with lobar hemorrhage (n = 152, 45.9 %) compared to the rest (n = 141, 31.5%, p < 0.001), and the fraction of patients with resistant BP after 6 months was higher in patients with non-lobar hemorrhage (n = 92, 20.6%) compared to those with lobar hemorrhage (n = 47, 14.2%, p = 0.02). The rates of controlled, undertreated, and resistant BP after 6 months follow-up in the subsets of patients are depicted in Figure 2. There was a significant decrease in the number of antihypertensive drugs between baseline and after 6 months follow-up for all patients (−0.24, 95% CI: [−0.35, −0.13]), without significant difference between the groups of lobar and non-lobar ICH (p = 0.65).
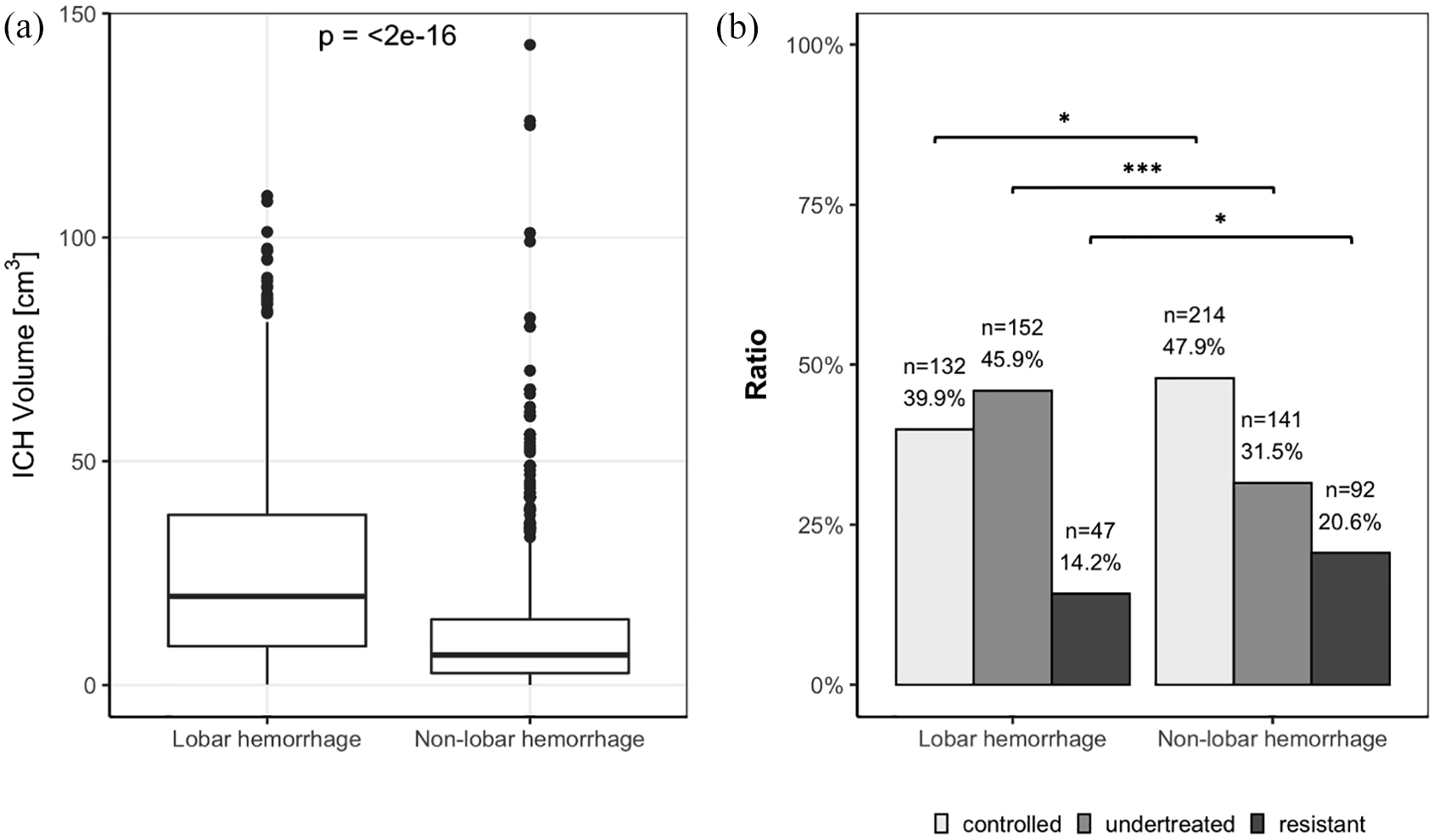
Differences in ICH volume and distribution of controlled, undertreated, and resistant blood pressure after 6 months follow-up by ICH location.
Regarding current treatment guidelines for ICH recurrence, 2 1313 (97.7%) of the patients had uncontrolled follow-up BP after 6 months with no difference between lobar and non-lobar location (p = 0.58). Similar to the guidelines at the time of treatment, the fraction of patients with undertreated BP was higher in those with lobar hemorrhage (n = 248, 74.7 %) compared to the rest (n = 251, 56.2%, p < 0.001), and the fraction of patients with resistant BP was higher in patients with non-lobar hemorrhage (n = 183, 40.9%) compared to those with lobar hemorrhage (n = 77, 23.2%, p < 0.001).
Lobar hematoma location, anti-hypertensive medication prescription, and post-ICH BP control
After adjusting for all potential confounders from univariable analyzes (Tables 1–3), race and presence of hypertension contributed significantly to the primary outcome and were kept in multivariable analyzes. Patients with lobar ICH had higher odds of uncontrolled BP (OR 1.35; 95% CI [1.08–1.69]) after 6 months follow-up compared to the rest of the patients. Patients with deep ICH had lower odds of uncontrolled BP (OR 0.78; 95% CI [0.62–0.96]) compared to the rest of the cohort. There was no association of cerebellar ICH with post-ICH BP control status (p = 0.57). Given the high rate of undertreated BP in patients with lobar ICH, we further investigated the effect of lobar ICH on antihypertensive treatment. After adjusting for age and presence of hypertension, having a lobar ICH was associated with prescription of fewer antihypertensive drugs both at baseline (β = −0.32, 95% CI [−0.46, −0.18]) and after 6 months (β = −0.41, 95% CI [−0.56, −0.25]), whereas having a deep hemorrhage was associated with an increased number of antihypertensive drugs at baseline (β = 0.17, 95% CI [0.03, 0.30]) and after 6 months (β = 0.31, 95% CI [0.16, 0.47]). Cerebellar hemorrhage was associated with an increased number of antihypertensive drugs at baseline (β = 0.38, 95% CI [0.16, 0.61]) and showed a trend toward association after 6 months (β = 0.28 [0.00, 0.57], p = 0.051).
We wanted to assess the role of management during hospitalization in the differences in antihypertensive drugs after follow-up. There was a strong independent effect of the number of antihypertensive drugs at discharge on the number of antihypertensive drugs after 6 months (β = 0.52, 95% CI [0.46, 0.58]), attenuating the effect of ICH location (lobar: β = −0.16, 95% CI [−0.30, −0.02]; deep: β = 0.19, 95% CI [0.06, 0.33]; cerebellar: β = −0.11, 95% CI [−0.36, 0.13]).
We further explored whether the number of antihypertensive drugs mediated the association between lobar hemorrhage and uncontrolled hypertension. Increasing numbers of antihypertensive drugs after 6 months were associated with lower odds of uncontrolled BP (OR for each antihypertensive drug 0.86 95% CI [0.75, 0.98]). In this model, the effect of lobar ICH on uncontrolled BP was no longer significant (OR 1.33, 95% CI [0.99, 1.81]), indicating that higher risk for uncontrolled BP after lobar ICH is at least partially mediated by antihypertensive treatment.
To rule out a time-dependent effect in treatment patterns, we added event year as predictor to our models, which had no effect on uncontrolled BP after 6 months in all patients (p = 0.40) and in patients with medication data available (p = 0.51). There was also no interaction between event year and number of antihypertensive drugs after six months (p = 0.69).
Discussion
Our results demonstrate that ICH survivors with lobar ICH are at increased risk for uncontrolled BP and that this risk is substantially mediated by relative undertreatment of hypertension. This effect was driven by the number of antihypertensive drugs prescribed and therefore seems to reflect physician practices in the management of ICH patients. While we did not find evidence that the characteristics of the acute ICH itself (location or volume) were associated with risk of subsequent uncontrolled BP, we did see a trend toward higher rates of treatment-resistant hypertension in patients with ICH in the deep regions.
The role of hypertension in the development of arteriolosclerosis and the risk for spontaneous ICH in the deep structures of the brain is well established.17,18 Thus, guidelines for secondary ICH prevention have primarily focused on managing short- and long-term BP control in patients with non-lobar ICH. 2 In our cohort, the rate of uncontrolled BP ranged from 52% up to 58% for all ICH cases as well as across ICH subtypes. When comparing controlled and uncontrolled BP groups, having a lobar ICH was associated with relatively worse BP control, perhaps reflecting the effectiveness of the guidelines focusing on strict BP management for patients specifically with non-lobar ICH. The treatment differences in patients with lobar ICH were remarkable and began already during hospitalization. It is unclear whether these patients had a higher frequency of conditions such as syncope or orthostatic hypotension that might limit use of antihypertensives. Another potential explanation is reticence to initiate treatment early in larger ICH to maintain cerebral perfusion pressure. However, the observed association raises concern for inadequate intensification of antihypertensive medications in this group of ICH survivors, leading to increased ICH recurrence, small vessel disease outcomes, and major cardiovascular events which we have associated with elevated follow-up BP in our cohort.7,8,16
There have been numerous studies investigating the role of clinical, social, and other determinants of resistant hypertension or uncontrolled BP in the general population.19–22 Among the factors that have been reported are older age, male sex, black race, diabetes mellitus, hyperlipidemia, chronic kidney disease, obesity or higher body mass index (BMI), coronary artery disease, atrial fibrillation, medical history of cerebrovascular accident or transient ischemic attack, peripheral vascular disease, and lower educational attainment and income.19–21,23 Unfortunately, data specifically on the ICH population are sparse and underreported.
Our study has several limitations. Despite the relatively large sample size for a population of ICH survivors, our results represent data from a single center that may not be generalizable to other cohorts of ICH survivors. Our findings therefore need to be replicated in future studies, ideally in populations representative of more diverse racial and ethnic backgrounds, especially in light of the known disparities that have been observed in BP control and ICH recurrence rates.9,24 Although we made an effort to comprehensively evaluate the baseline radiographic predictors of post-ICH uncontrolled BP status as well as potential confounders, a systematic collection of social determinants of health as well as physician practices was limited, which could potentially affect our outcomes of interest. With regards to medication data adjustments, our inferences pertain only to anti-hypertensive medication prescriptions and only in half of the participants, since we could not measure adherence and thus actual medication use. Additionally, due to the observational nature of the study, existence of unmeasured confounders that may have affected the relationship between ICH characteristics and BP control status cannot be ruled out. The lack of a standardized procedure for BP data capture in the post-discharge period and the reliance on BP metrics measured during routine clinical care is another important limitation.
Conclusions
In this cohort of ICH survivors, lobar location of ICH was strongly associated with higher rates undertreated hypertension after 6 months. This finding highlights the importance of emphasizing the treatment of hypertension for survivors of spontaneous ICH, regardless of location.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the US National Institutes of Health (NIH) (R01NS093870, K23NS100816). Dr. Anderson receives sponsored research support from the NIH (R01NS103924, U01NS069763) relevant to this manuscript.
Informed consent
Written informed consent was obtained from all subjects or their legally authorized representatives before the study.
Ethical approval
Ethical approval for this study was obtained from the institutional review board at Massachusetts General Hospital.
Guarantor
JR
Study Concept
EPM, EM, CDA, AB, JR; Study Design: EM, EPM; Acquisition of Data: all authors; Statistical Analysis: EPM, EM; Manuscript Preparation: EPM, EM, CDA, JR; Manuscript Review: all authors; Study Supervision / Coordination: BEM, LS, ADW, JNS, EG, AV, SG, AB, JR, CDA. All author(s) read and approved the final manuscript.
Disclosures
Dr. Goldstein has received research support from NIH, Pfizer, Octapharma, Takeda, and has received consulting from Alexion, CSL Behring, Cayuga, and NControl. Dr. Biffi receives sposored research support from the NIH (K23NS100816) and AHA American Heart Association (AHA). Dr. Anderson receives sponsored research support from the American Heart Association (AHA) (18SFRN3425000) and Bayer AG, and has consulted for ApoPharma, Inc. Dr Rosand receives sponsored research support from the NIH, AHA and OneMind, and consults for Takeda Pharmaceuticals. All other authors have no disclosures to report relevant to this manuscript.
Trial registration
Not applicable.
