Abstract
Background
Trials of tranexamic acid (TXA) in acute intracerebral hemorrhage (ICH) have focused on the imaging outcomes of intraparenchymal hematoma growth. However, intraventricular hemorrhage (IVH) growth is also strongly associated with outcome after ICH. Revised definitions of hematoma expansion incorporating IVH growth have been proposed.
Aims
We sought to evaluate the effect of TXA on IVH growth.
Methods
We analyzed data from the STOP-AUST trial, a prospective randomized trial comparing TXA to placebo in ICH patients presenting ≤ 4.5 h from symptom onset with a CT-angiography spot sign. New IVH development at follow-up, any interval IVH growth, and IVH growth ≥ 1 mL were compared between the treatment groups using logistic regression. The treatment effect of TXA against placebo using conventional (> 6 mL or 33%), and revised definitions of hematoma expansion (> 6 mL or 33% or IVH expansion ≥ 1 mL, > 6 mL or 33%, or any IVH expansion, and > 6 mL or 33% or new IVH development) were also assessed. Treatment effects were adjusted for baseline ICH volume.
Results
The analysis population consisted of 99 patients (50 placebo, 49 TXA). New IVH development at follow-up was observed in 6/49 (12%) who received TXA and 13/50 (26%) who received placebo (aOR: 0.38 [95% CI: 0.13–1.13]). Any interval IVH growth was observed in 12/49 (25%) who received TXA versus 26/50 (32%) receiving placebo (aOR: 0.69 [95% CI: 0.28–1.66]). IVH growth ≥ 1 mL did not differ between the two groups. Using revised definitions of hematoma expansion, no significant difference in treatment effect was observed between TXA and placebo.
Conclusions
IVH may be attenuated by TXA following ICH; however, studies with larger cohorts are required to investigate this further.
Registration
http://www.clinicaltrials.gov; Unique identifier: NCT01702636.
Keywords
Introduction
The use of tranexamic acid (TXA) in acute intracerebral hemorrhage (ICH) has been associated with modest reductions in hematoma growth,1,2 but no major effect on functional outcomes. To date, assessments of TXA and hematoma growth have focused on changes in intraparenchymal hemorrhage volume. However, a number of studies have shown that intraventricular hemorrhage (IVH) growth is a predictor of poor outcome, independent of intraparenchymal volume change or confounding conditions such as obstructive hydrocephalus.3–6 Furthermore, revised definitions of hematoma expansion that incorporate IVH growth have demonstrated improved associations with long-term outcome compared to conventional models.7,8 The effect of TXA on IVH growth remains unclear. Using data from STOP-AUST (the Spot sign and Tranexamic acid On Preventing ICH growth–Australasia Trial), 2 we sought to investigate the treatment effect of TXA on IVH growth. We hypothesized that incorporating IVH growth to measures of hematoma expansion would provide a more detailed measure of TXA performance.
Methods
All supporting data and methodological detail are available within the article and online-only supplement. Access to data can be obtained from the study authors upon request. Patients in this analysis were participants in the STOP-AUST trial with hemorrhage volumes measured at baseline and 24-h follow-up. STOP-AUST was a phase 2, investigator-led, international multicenter, randomized placebo-controlled trial, evaluating TXA versus placebo in patients presenting with acute ICH ≤ 4.5 h of symptom onset with a spot sign on CT-angiography.2,9 The primary outcome of the original trial was hematoma expansion, defined as > 6 mL absolute hematoma growth or > 33% relative hematoma growth, when comparing baseline to follow-up scans at 24 h. ICH and IVH volumes were measured using semi-automated planimetry by an imaging expert blinded to treatment allocation. 10 In this study, we first compared changes in IVH between the treatment and control groups. We assessed new IVH development (defined as the presence of IVH at follow-up where no IVH was present on the initial scan), any interval IVH growth, and IVH growth ≥ 1 mL using binary logistic regression models. These measures of IVH growth are similar to thresholds shown to be associated with long-term clinical outcome in previous studies.3,7,8 Second, we compared the treatment effect of TXA against placebo using conventional and revised definitions of hematoma expansion that incorporate ICH and IVH growth (> 6 mL or 33% or new IVH development, > 6 mL or 33% or any IVH growth, and > 6 mL or 33% or IVH growth ≥ 1 mL). Treatment effects were adjusted for baseline ICH volume in all models. Statistical analyses were performed using STATA v17 (StataCorp, TX, USA). Local research ethics board approval was obtained at all STOP-AUST enrolling sites.
Results
Of the 100 patients recruited into STOP-AUST, our primary analysis includes 99 patients (50 placebo, 49 TXA). One patient was excluded due to missing follow-up imaging. Baseline characteristics are presented in Supplemental Table 1. The median time from scan to treatment in all patients was 41 min (IQR: 29–56). Overall, 22 (22%) patients had IVH at initial presentation, and 40 (40%) patients had IVH at 24 h. IVH clot retraction (reduction of IVH volume on follow-up scans) was observed in 13 patients including one patient with full clot resolution. Twenty-eight (28%) patients experienced any degree of IVH growth; 19 of these were cases of new IVH development.
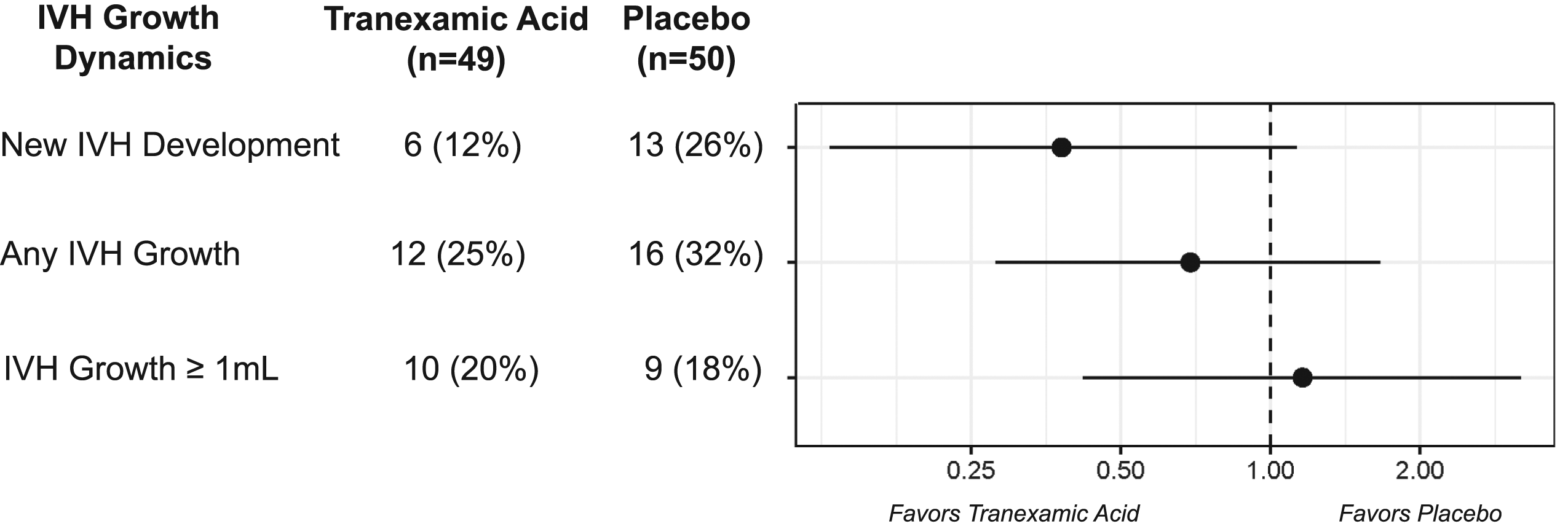
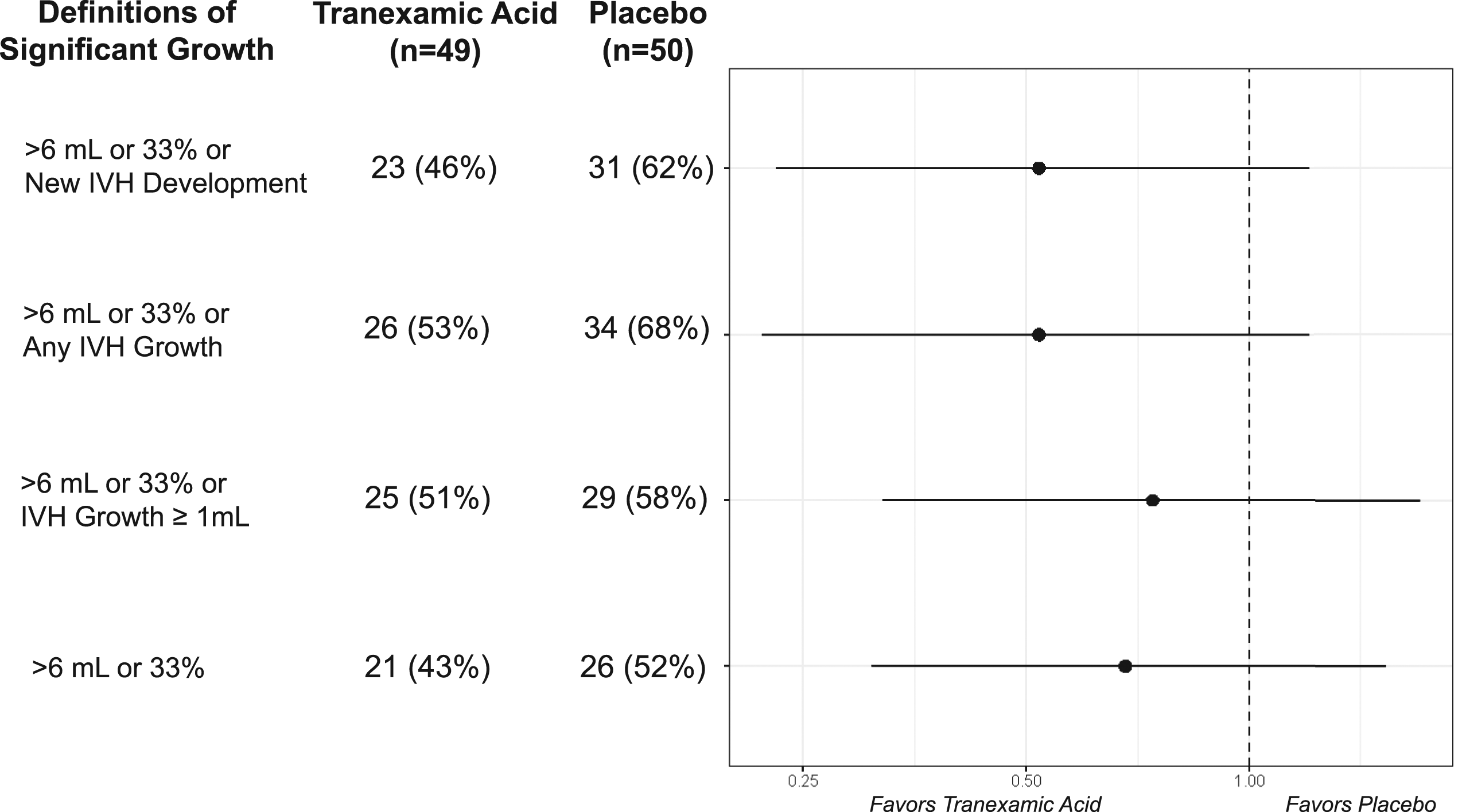
The proportion of patients exhibiting “no IVH growth,” “IVH growth <1 mL,” and “IVH growth ≥1 mL” are presented in Figure 1. IVH growth dynamics are presented in Figure 2 and Supplemental Figure 1. New IVH development at follow-up was observed in 12% of patients who received TXA compared to 26% receiving placebo (aOR: 0.38 [95% CI: 0.13–1.13]). Any IVH growth was observed in 25% of patients who received TXA versus 32% who received placebo (aOR: 0.69 [95% CI: 0.28–1.66]). No major differences in IVH growth ≥ 1 mL were observed between the two groups. Significant hematoma expansion, defined as > 6 mL or 33% or new IVH development; > 6 mL or 33% or any IVH growth; and > 6 mL or 33% or IVH growth ≥ 1 mL was observed in 46% TXA/62% Placebo (aOR 0.52 [95% CI: 0.23–1.19]), 53% TXA/68% Placebo (aOR 0.52 [95% CI: 0.22–1.20]), and 51% TXA/58% Placebo (aOR: 0.74 [95% CI: 0.32–1.70]), respectively (Figure 3). IVH growth dynamics, stratified by baseline ICH location, are provided in Supplemental Table 3. No major differences between cortical and deep ICH locations were observed. Distributions of intraventricular hemorrhage (IVH) growth by treatment. Intraventricular hemorrhage (IVH) growth dynamics stratified by treatment. Definitions of significant hematoma growth, stratified by treatment.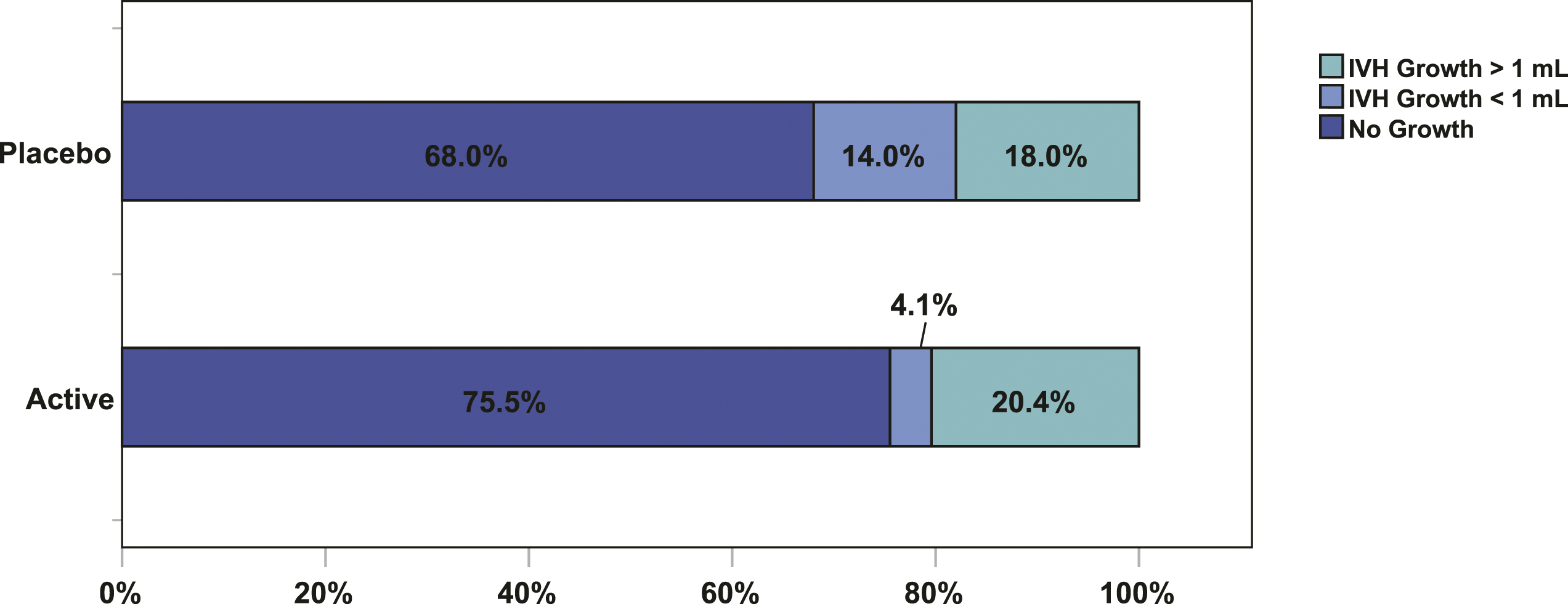
Discussion
In this post-hoc analysis of the STOP-AUST trial, we assessed the treatment effect of TXA on IVH growth. We observed a non-statistically significant reduction in any IVH growth, which appeared mostly driven by growth < 1 mL and a reduction in new IVH development.
In the past 10 years, there has been renewed interest in antifibrinolytic and hemostatic therapies for ICH.1,11 The primary imaging endpoint in most trials has been hematoma growth, with the most common definition being > 6 mL or > 33 mL growth in ICH volume. However, ICH is a complex disorder where acute blood product can move between different compartments of the intracranial space, with variable effects on clinical outcomes. Therefore, the incorporation of IVH parameters into clinical trial imaging endpoints has been proposed to improve the understanding of the treatment effect as well as the association between hematoma growth and functional endpoints. Our analysis is limited by a small sample size and potentially reduced generalizability due to the requirement of a CTA spot sign for recruitment. However, given that IVH growth is associated with poorer long-term outcomes, the observed trends are hypothesis generating and require further analysis in larger cohorts.
An additional challenge for future analyses is how to best assess IVH that is not detectable using current approaches. For example, when blood leaks into the intraventricular compartment, its measurement may become less accurate due to dilution within the CSF, redistribution outside of the brain, or even constriction as the composition of blood becomes more coagulated, making it difficult to be confident of the exact volume. The use of “new IVH development” as an alternate outcome measure may be more resilient from this point of view, and this warrants further assessment in future studies. Another area requiring further study is the influence of IVH location on clinical outcome in patients with small-interval IVH growth. Prior analysis of the IVH thresholds was performed in cohorts where patients requiring interventions such as an extra-ventricular drain were excluded.3,7 However, in clinical practice, the strategic presence of blood in certain locations can have an overwhelming effect on outcome. For example, a smaller volume of blood located in the third ventricle may lead to obstructive hydrocephalus, with worsened clinical outcome, compared to the same volume in the lateral ventricle. In our analysis, more patients with deep hematomas had IVH at baseline (27%) compared with those with cortical hematomas (13%). However, the proportion of patients with IVH expansion, using any of the definitions, was not significantly different between deep and cortical locations. This suggests that IVH growth is not purely a reflection of baseline hematoma location.
There is an increasing push to focus on medical therapies for ICH in the hyperacute time frame (0–2 h), as the time from symptom onset to imaging has been shown to be a consistent predictor of hematoma expansion. 12 In STOP-AUST, there was a trend toward a stronger treatment effect of TXA in the < 3-h time window, and particularly in the < 2-h time window. The ongoing STOP-MSU trial (NCT03385928) is a randomized controlled trial of TXA against placebo within 2 h of symptom onset with non-contrast CT selection. In this trial, we plan to further examine IVH growth both as an individual variable and in combination with ICH growth. 13 The FASTEST trial will evaluate rFVIIa within the 2 h of symptom onset and will also aim to assess IVH growth (NCT03496883).
Conclusions
IVH may be attenuated by TXA following ICH. Given the strong associations between IVH and functional outcome, this imaging metric should be further evaluated in future ICH trials.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211072402 – Supplemental Material for Does tranexamic acid affect intraventricular hemorrhage growth in acute ICH? An analysis of the STOP-AUST trial
Supplemental Material, sj-pdf-1-eso-10.1177_23969873211072402 for Does tranexamic acid affect intraventricular hemorrhage growth in acute ICH? An analysis of the STOP-AUST trial by Vignan Yogendrakumar, Teddy Y. Wu, Leonid Churilov, Turgut Tatlisumak, Daniel Strbian, Jiann-Shing Jeng, Timothy J. Kleinig, Gagan Sharma, Bruce C.V. Campbell, Henry Zhao, Chung Y. Hsu, Atte Meretoja, Geoffrey A. Donnan, Stephen M. Davis, and Nawaf Yassi in European Stroke Journal
Footnotes
Author Contributions
VY, TW, LC, and NY conceived the study and drafted the manuscript. HZ, CH, GD, and SMD provided conceptual assistance. All authors reviewed and approved the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AM reports honoraria paid for advisory board, speaker fees, and travel from Stryker and Boehringer Ingelheim. SMD reports lectures and advisory boards activities for Medtronic, Amgen, and Boehringer Ingelheim. All other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: STOP-AUST was funded by the Australian National Health and Medical Research Council, (grant: 1081718), and the Royal Melbourne Hospital Foundation.
Ethical approval
Local research ethics board approval was obtained at all STOP-AUST enrolling sites.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Guarantor
NY
Data Availability
All supporting data and methodological detail are available within the article and online-only supplement. Access to data can be obtained from the study authors upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
