Abstract
Background
Balance and gait impairments are the most common motor deficits due to stroke, limiting the patients' daily life activities and participation in society. Studies investigating effect of task-specific training using biomechanical balance and gait variables (i.e. kinetic and kinematic parameters) as well as posturography after stroke are scarce.
Objectives
The primary aim of this study is to assess the efficacy and long-term outcome of task-specific training based on motor relearning program (MRP) on balance, mobility and performance of activities of daily living among post-stroke patients.
Methods
In this two-armed randomised controlled clinical trial, a total of 66 sub-acute stroke patients who meet the trial criteria will be recruited. The patients will randomly receive task-specific training based on MRP or a conventional physical therapy program (CPT). Twenty-four physiotherapy sessions will be conducted, divided into three training sessions per week, 1 h per session, for 8 weeks, followed by an analysis of changes in patient’s balance, gait and performance of activates of daily living at three time periods; baseline, post-intervention and follow-up after 3-months, using clinical outcome measures and instrumental analysis of balance and gait.
Discussion
The results of this study can guide to better understanding and provide an objective clinical basis for the use of task-specific training in stroke rehabilitation. Also, it intends to help bridge the current knowledge gap in rehabilitation and training recommendations to provide a therapeutic plan in post-stroke rehabilitation.
Trial registration
ClinicalTrials.gov (NCT05076383). Registered on 13 October 2021 (Protocol version: v2.0).
Introduction
Background and rationale
The number of people living with stroke is estimated to increase by 27% between 2017 and 2047 in the European Union, mainly because of population ageing and improved survival rates. 1 Stroke often results in functional motor impairments that affect the patients' mobility and ability to perform daily tasks, and full recovery is achieved in only a small proportion of stroke survivors. 2 At 6 months after stroke, 40% of stroke survivors have difficulties with essential self-care (e.g. dressing and feeding), 3 and more than 30% of stroke survivors report participation restrictions (e.g. fulfilling societal roles) even at 4 years after stroke.3,4 All of these factors contribute to the low overall quality of life. 5
The limited walking ability that follows the stroke restricts a patient’s independent mobility in the home and community. Regaining the ability to walk independently is the most critical functional goal in the rehabilitation of stroke patients. 6 In addition to walking ability, patients must adapt physically and cognitively to sudden disturbances in body movement when they encounter environmental barriers and unexpected events during community ambulation. 6 Additionally, stroke frequently results in postural disorders characterised by a mediolateral deviation towards the unaffected lower limb and a greater instability of the centre of pressure. 7 These dysfunctions lead to balance disorders responsible for an increased risk of falls and a lower level of activity and participation in stroke patients. 8 Balance is associated with walking abilities, 9 and it is a predictor for achieving the ability to walk. 10
The motor relearning program (MRP) is one of the rehabilitative strategies used primarily with the post-stroke population. 11 This approach includes many aspects of motor learning theory and provides practical guidelines for retraining functional skills (e.g. balanced sitting, sitting and standing, transfer skills and gait). 11 This approach focuses on task-specific learning through effective feedback and practice development of active movement control. 12 Facilitation techniques are deemphasised, whereas verbal instruction, demonstration and manual guidance are emphasised. The approach is based on four distinct steps: (1) analysis of the essential components of the task; (2) practice of the missing component, that is, when a patient cannot control the necessary muscles to perform a task, this component is practiced separately before incorporating it into the complex task; (3) practice of the task; (4) transference of training to practice in context. 11
A review of the literature revealed that MRP was effective for enhancing the functional recovery of patients who had a stroke. 13 Furthermore, MRP was found to be more effective than the Bobath approach in the early enhancement of activities of daily living (ADL’s) in acute stroke rehabilitation. 14 Moreover, MRP was more effective than proprioceptive neuromuscular facilitation 12 and conventional training 15 for improving basic and functional mobility. Only a few clinically controlled trial studies on the effectiveness of the MRP approach on balance and postural control have been conducted. For instance, a study by Chan et al. (2006) demonstrated a significant improvement on the Berg Balance Scale (BBS) following 6 weeks of MRP. 14 More recently, Khallaf (2020) concluded that task-specific training based on motor learning effectively improved the static and dynamic postural control and trunk ranges of motion among sub-acute stroke patients. 16
Despite that, the available evidence reveals that MRP effectively improves functional abilities and balance in stroke individuals, further investigation is needed to determine the effectiveness of task-specific training based on MRP. The current study will examine the effectiveness and long-term outcome of task-specific training based on MRP on balance, mobility and performance of activities of daily living among post-stroke patients, using not only traditional outcome measures but using instrumental evaluation tools to assess the biomechanical balance and gait variables (i.e. kinetic and kinematic parameters). The results of this study can guide a better understanding of stroke rehabilitation. In addition, the present study is intended to help bridge the current knowledge gap in rehabilitation and exercise recommendations in the stroke population.
Trial objectives
The primary objective of this study is to assess the efficacy and long-term outcome of task-specific training based on MRP on balance, mobility and performance of activities of daily living among post-stroke patients. This study will be followed by a 3-months follow-up to evaluate the retention of gains.
Methods
Trial design
This study is a two-armed randomised controlled clinical trial (RCT) of parallel design, using standardised outcome measures and instrumental analysis for balance, postural control and gait to collect data on the improvements. Patients who meet the defined eligibility criteria will be randomly assigned to the study group (MRP) (G
1
) or control group (CPT) (G
2
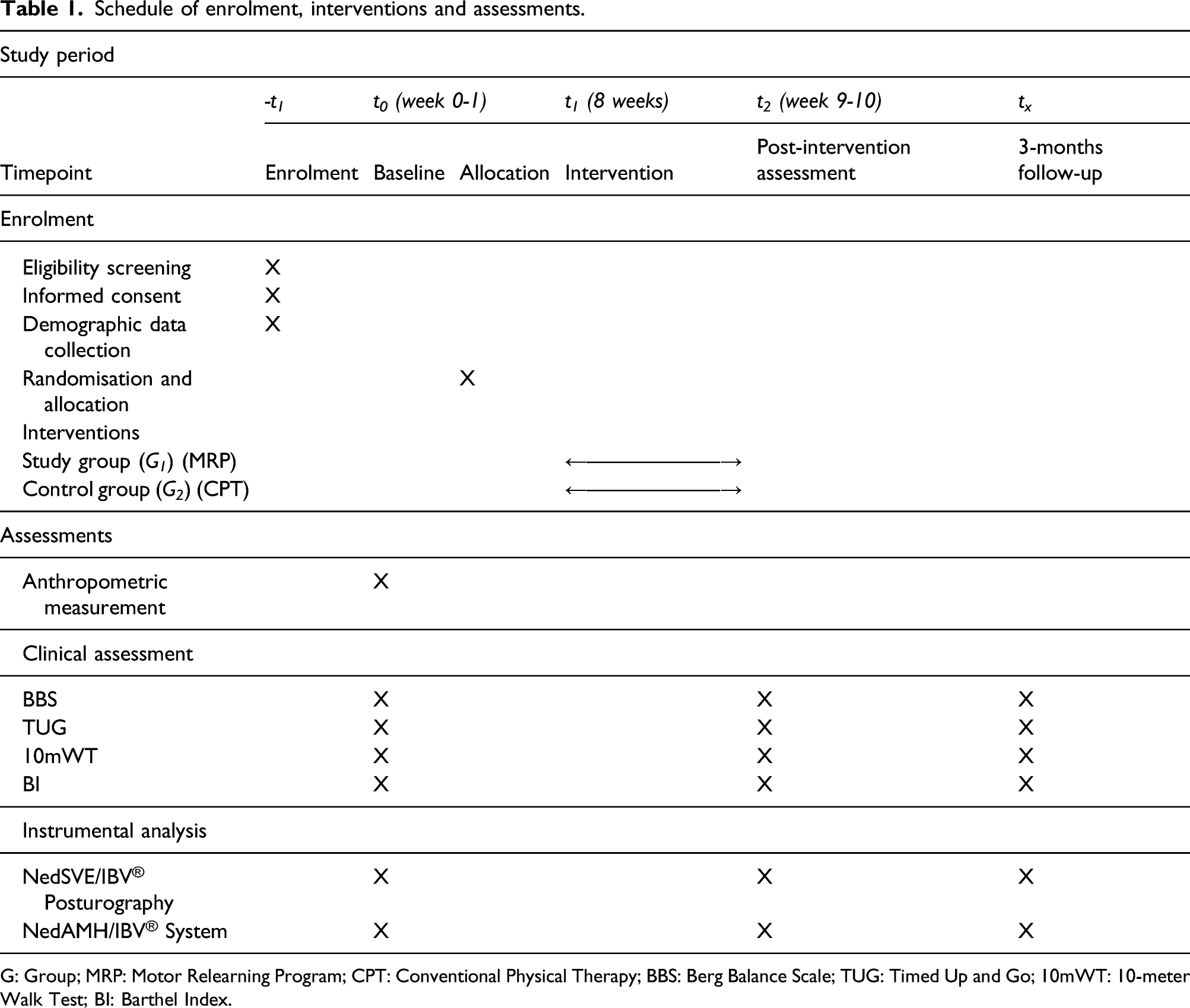
), with an allocation ratio of 1:1. The trial’s Consolidated Standards of Reporting Trials (CONSORT) flow chart is displayed in Figure 1. The development of the study protocol is described following the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) guidelines
17
and a schedule of enrolment, interventions and assessments is outlined in Table 1. The Consolidated Standards of Reporting Trials (CONSORT) flow chart. Schedule of enrolment, interventions and assessments. G: Group; MRP: Motor Relearning Program; CPT: Conventional Physical Therapy; BBS: Berg Balance Scale; TUG: Timed Up and Go; 10mWT: 10-meter Walk Test; BI: Barthel Index.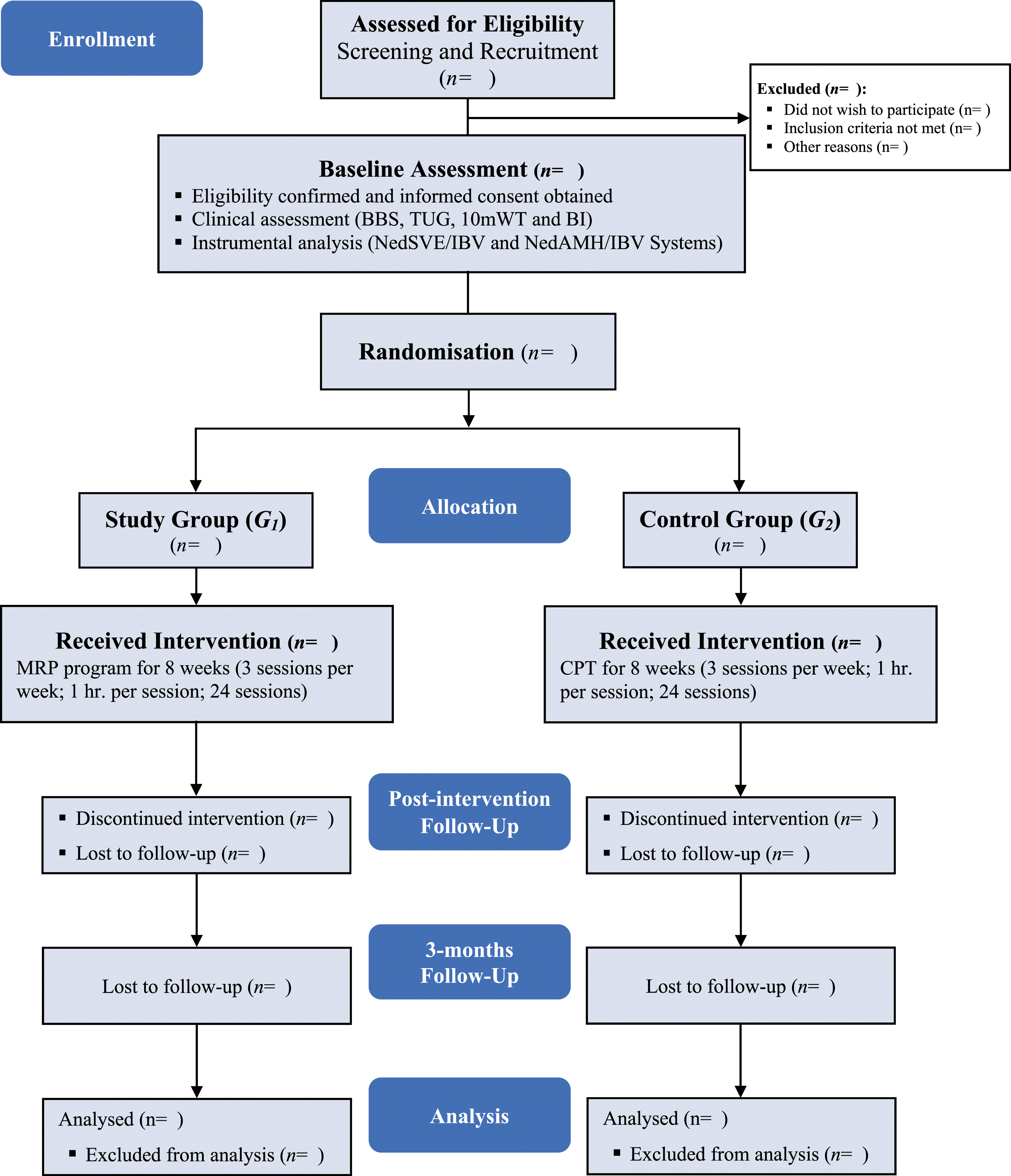
Participant
Eligibility criteria for participants
Inclusion criteria
Participants that meet the following criteria will be included in the study: (1) first-ever sub-acute (1–6 months) stroke patients; (2) able to give informed consent; (3) patients with hemiparesis; 2–4 muscle power in the affected side (Medical Research Council-MRC Muscle Scale 2–4); (4) able to stand independently for at least 1 min; and (5) can ambulate 25 feet/10 m (with or without the assistive device).
Exclusion criteria
Potential participants will be excluded for meeting any of the following criteria: (1) post-stroke patients with major cognitive deficits (Montreal Cognitive Assessment-MoCA score ≤20) and/or communication impairments that do not allow patients to follow directions (i.e. deafness and aphasia); (2) those who are receiving other related therapy through the study, which may affect the efficacy of this study; (3) those with any contraindications to start rehabilitation (i.e. severe uncontrolled hypertension, uncontrolled diabetes or unstable angina); and (4) those with a history of neurological deficits other than stroke.
Study setting
The study will be carried out at the Department of Physical Medicine and Rehabilitation of the Parc de Salut Mar Consortium (Hospital de l'Esperança and Centre Fòrum-Hospital del Mar), Barcelona, Spain. The clinical outcome measures and instrumental analysis will be conducted in the Functional and Movement Analysis Laboratory at Centre Fòrum-Hospital del Mar.
Recruitment and consent
Participants will be referred by a specialist physician to be recruited in the study. The researcher will screen the enrolled patients who meet the selection criteria and provide an information sheet of the study and possible benefits of the rehabilitation and the relevant safety during the trial to obtain informed consent. Once the consent is obtained, the researcher will proceed with the baseline assessment. Participants who completed the baseline assessment will be randomly assigned into the two groups and subjected to rehabilitation programs.
Randomisation and allocation
Sequence generation
The eligible participants will be randomly allocated to one of the two groups (study group or control group), using a web-based tool (www.sealedenvelope.com). The random sequence will be generated using permuted block randomisation with a block size of 4 and a 1:1 allocation ratio.
Concealment mechanism and implementation
The results of sequence generation will be distributed and stored in sealed envelopes and kept confidential by a member of the research team who conducted the randomisation procedure; this person will be blind to the participants’ identities and will not be involved in recruitment, assessment or intervention. For allocation in groups, each participant blindly will pull up a sealed envelope indicating one of the intervention groups. Participants will be informed about their assigned intervention group.
Blinding
The researcher who will generate the sequence of randomisation will not be involved in recruitment, assessment or intervention. The outcome assessor will be blinded to group allocation. However, as the study is an exercise-based intervention with the active participation of therapists and participants, both therapist and participants cannot be blinded to the intervention after assignment, but the data analysts will be blinded to group allocation and intervention prior to analysis.
Interventions
Individuals from both groups will complete 8-weeks (3 sessions per week; 1 h per session; 24 sessions) of either task-specific training based on the MRP or a conventional physical therapy, which will be performed by physiotherapists of the department of physical medicine and rehabilitation. The interventions in the two groups will be as follows:
Study group (MRP) (G1)11,12,18
Task-specific training based on MRP will be performed for the study group. Each training session will consist of five training tasks: (1) bed mobility and sitting up over the side of the bed: bed mobility exercises (i.e. rolling and bridging), followed by transition from supine position to sitting at the edge of the bed; (2) balanced sitting: head and trunk movements in sitting position with feet and knees approximately 15 cm apart and multidirectional reaching activities; (3) standing up and sitting down: from sitting on a firm flat surface with no arm rests, standing up starts with upper body vertical and feet placed backward, followed by sitting down by flexing the hip, knees and ankles; (4) balanced standing: head and trunk movements while standing with feet apart and multidirectional reaching actions; and (5) practice of walking components is followed by practice of walking itself.
Control group (CPT) (G2)19,20
The CPT exercises will be performed following a standard stroke rehabilitation program for the control group. Each CPT rehabilitation session will include the following exercises: (1) passive and active-assisted range of motion exercises for the upper and lower extremity including the shoulder, forearm, wrist, hip, knee and ankle; (2) gradual progressive stretching of shoulder, elbow, wrist, hamstrings and calf; (3) isometric strengthening exercises of the trunk and quadriceps; (4) balance training including practicing reaching beyond arm’s length while sitting and standing; and (5) walking training that includes challenge to dynamic balance (e.g. overground walking and obstacle courses).
Strategies to improve adherence to the intervention
Various strategies will be adopted to improve adherence to interventions. To begin with, participants will receive a comprehensive, simplified oral and written description about why the study is being done and what it will involve. In addition, to establish a good relationship with the participants and provide needed guidance to gain their trust and cooperation. Furthermore, a flexible schedule of training sessions will be offered to motivate participants to improve adherence.
Assessments and outcomes
Assessment variables for this clinical trial include the patient’s basic information, clinical outcome measures and instrumental analysis of balance, postural control and gait. These assessments will be carried out by an independent researcher, who is not aware of the allocation. Patients from the two groups will be assessed at baseline (t 0 ), post-intervention (t 2 ) and 3-months follow-up (t x ). The patient’s assessment will follow a standard sequence outlined in Table 1. The study outcomes will be
Berg Balance Scale
The Berg Balance Scale (BBS) is a 14-item scale commonly used standardised balance assessment to quantitatively determine through observation the patient’s ability to maintain balance either statically or while performing various predetermined functional movements. 21 It proved to be a valid and a reliable scale for assessing static and dynamic balance after a stroke. 22 Patients will receive a score from 0 to 4 for each scale item, with a total score out of 56. A score of 0 indicates an inability to perform the task, and a score of 4 indicates the task’s independent performance. 21
Timed Up and Go Test
The Timed Up and Go Test (TUG) is a is a reliable, valid and easy-to-administer clinical tool for assessing functional mobility (i.e. mobility, balance and locomotor performance) in people with balance disturbances includes, but is not limited to, individuals with stroke. 22 It requires the participant to stand up from a chair, walk 3 m, turn, return to the chair and sit down again. The required time to perform the test is recorded in seconds. 23
10-meter Walk Test
The 10-meter Walk Test (10 mWT) is a performance test to assess walking speed over a short distance in meters/second. A participant walks 10 m, and the required time for the intermediate 6 m is recorded to allow for acceleration and deceleration. 24 The 10 mWT has demonstrated excellent reliability and feasible for measuring gait speed among individuals with a stroke. 25
Barthel Index
The Barthel Index (BI) is an ordinal scale that measures performance in activities of daily living (ADL) in the domains of self-care and mobility. It measures the level of assistance required by a patient on 10 items describing ADL and mobility. The score of each item ranges from 0 to 10 depending on the level of patient’s functionality, which will be summed to create a score from 0 to 100, with higher scores indicating greater ability to function independently. 26 It was reported that the BI is a reliable and valid instrument in assessing ADL functions in stroke patients. 27
Instrumental analysis of balance and gait
The instrumental analysis of balance and gait is performed using NedSVE/IBV® and NedAMH/IBV® systems based on the Dinascan/IBV P600 dynamometric platform specifically designed to study balance and gait.28,29 The balance and postural control will be assessed using the computerised posturography-NedSVE/IBV® platform, a software application for analysing balance disorders by comparing them with normality patterns. The system combines static posturography tests with dynamic tests based on gait analysis. 28 The specific tests included in the NedSVE/IBV platform are sensory-dynamic analysis and analysis of the rhythmic and directional postural control. 30 The gait will be assessed using the NedAMH/IBV® system, a software application for the biomechanical gait assessment. The patient is asked to walk through a walking corridor at a comfortable speed to carry out the assessment. 29 The system records the speed and kinetic (reaction forces exerted by the lower limb) parameters as the patient cross the photocell barriers and steps on the dynamometric platform. 31
Sample size
The sample size for this trial is calculated by using balance impairment as the main indicator, that is, Berg Balance Scale (BBS). Based on previous studies, the expected mean effect size is around d = 0.65.13,32,33 Using the statistical program G* power Software (version 3.1; Henrich-Heine-Universitat Dusseldorf, Germany), 34 at alpha level (α) of 0.05 and power (1– β) of 0.80. This generated a sample size of 30 patients in each group. Considering a 10% drop-out, the total sample size for this trial is about 66 patients randomised to one of the two treatment groups (study group or the control group).
Data management
The PI will create the database and be responsible for data entry and keeping the database uploaded and stored in the electronic database. Data will be collected in a prepared SPSS 21.0 form. Double-check will be applied routinely to ensure that the data was entered correctly to promote data quality. Each participant is assigned a unique enrolment identification number at the start of the assessment. The personal information of the participants will not be contained in the database and will not appear in the relevant reports of the trial to ensure participant confidentiality. Data will be stored according to the guidelines of the Regional Research Ethics Committee of the Hospital del Mar Research Institute. The PI will control the use of the study data; access to data, which will only contain coded data, will be restricted to researchers directly involved in the trial analysis. All clinical trial data will be analysed anonymously, and they will be kept for 5 years after the termination of the clinical trial.
Statistical methods
Quantitative variables in the two treatment groups will be statistically described with mean ± standard deviation unless otherwise stated. The univariate analysis will be performed using appropriate statistical tests (i.e. chi-squared (χ2) test, Fisher’s Exact test, Student’s t-test or Mann–Whitney U test), depending on variables analysed. Treatment effect will be analysed by changes in balance and gait parameters pre- and post-intervention. Changes during follow-up will be assessed by analysis of variance (ANOVA) using mixed repeated measures and a one-factor design to analyse values over time. The effect size will be reported using Cohen’s term d index. The data or at least one of the key outcomes will be analysed by intention-to-treat. The results over the follow-up period will be reported considering potential scenarios of noncompliance and non-adherence. The level of significance will be set at p ≤ .05. Data analysis will be performed using IBM SPSS Statistics v.21.
Adverse event reporting and harms
Adverse events, although unlikely, could be related to the rehabilitation exercises such as fatigue, muscle stiffness, ‘spasticity’ and/or falls. All expected exercise-related adverse effects will be explained in the information sheet before the enrolment to the study participants. Adverse events, if any, will be recorded during assessment and rehabilitation sessions.
Auditing
The PI will be responsible for auditing the trial-related activities (assessments and interventions) and documents concerning trial auditing. The study directors and PI will meet bi-monthly to discuss and monitor the progress of the trial-related procedures and documents throughout the trial.
Dissemination of results
The results of this study will be reported in a PhD thesis by the main author and submitted as manuscripts to peer-reviewed journals for publication. Moreover, the results will be presented at relevant national or international conferences if possible.
Discussion
One of the main motor deficits resulting from stroke is the postural control dysfunction that leads to balance disorders responsible for an increased risk of falls and a lower level of functional activities and participation. 7 Balance is a predictor for achieving the ability to walk among stroke patients. 10 The most crucial functional goal in the rehabilitation of stroke patients is regaining the ability to walk independently. 35 In this regard, the current study examines the effectiveness of task-specific training based on MRP using clinical outcome measures and instrumental evaluation tools to measure the biomechanical balance and gait variables.
Previous studies on stroke have widely used standardised outcome measures to evaluate motor deficits (i.e. balance and gait). In this respect, this study adopted an instrumental balance and gait analysis as part of the approach for evaluating the results. This will provide quantitative data (kinetic and kinematic parameters) about balance and gait and reduce the interference of subjective factors in the evaluation.
The interventional exercises of this study will focus on training sub-acute stroke patients within the task context to improve their motor abilities to enable them to do life functional activities independently. In order to obtain a significant improvement on the study outcomes, the intensity of interventions in this study will be 24 rehabilitation sessions for 8-weeks (3 sessions per week; 1 hr. per session), which is in line with what is recommended by Canadian Stroke Best Practice Recommendations. 36 The interventions will be followed by an analysis of changes in the patients' balance, gait and performance of activities of daily living at two stages: post-intervention and at 3-months from the end of the intervention to determine the long-term outcome obtained.
The results of the several assessment tools (instrumental and outcome measures) will be compared between the two groups in order to verify the efficacy of the task-specific training based on MRP. The findings of this study can guide a better understanding of stroke rehabilitation. Besides, the present study is intended to help bridge the current knowledge gap in rehabilitation and training recommendations in the stroke population, to provide a therapeutic plan in the post-stroke rehabilitation, focused on the use of task-specific training in order to help patients return to their daily functional activities in the community quickly and effectively.
A potential limitation to the trial that must be acknowledged is that the number of tests may pose a challenge to the participants, increasing the risk of drop-out at post-intervention and follow-up. Great efforts will be made to motivate the study participants by explaining the potential benefits to the patients and their families. Participants will also receive feedback on their quality of movement and performance and acknowledging their results after each assessment.
Trial status
The study is registered on ClinicalTrials.gov with register number (NCT05076383). Study participants were being recruited at the time of this submission. The participants' recruitment began on October 2021; the actual trial status is ongoing, and October 2022 is the expected date for the end of recruitment. The study completion date is estimated to be January 2023.
Footnotes
Acknowledgements
Sincere gratitude to the study director, Dr Esther Duarte, for the constant support, insightful readings and comments that have made the completion of this research protocol manuscript possible. Acknowledgements to Dr Ester Marco for this research’s tremendous help.
Declaration of conflicting interests
The author(s) declare that they have no competing interests, and the study has not received external funding.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. This research study is an essential requirement for Amer Ghrouz doctoral study (PhD) in Medicine (Physical Medicine and Rehabilitation) at Universitat Autònoma de Barcelona, Spain and not funded by any party or agency.
Availability of data and materials
Data sharing is not applicable to this article as no datasets have so far been generated or analysed.
Informed consent
Participants will be provided with written and oral information about the study, and a written informed consent to participate will be obtained from all participants before enrolment.
Ethical approval
The Research Ethics Committee of the Hospital del Mar Research Institute, Barcelona, Spain, has approved the study (REC Nº: 2021/9986/I).
Guarantor
AG.
Contributionship
AG and ED contributed to the trial design and procedures, developed the content and drafted the manuscript. ED and EM were responsible for critical revision of the article for important intellectual content, provided feedback on the trial design and managed the project. RB, EM-R and CR-F contributed feedback on assessments and data collection. All authors revised the manuscript for relevant scientific content and approved the final version of the manuscript.
