Abstract
Background and Purpose
Post-stroke fatigue is a debilitating and long-lasting condition. However, there are uncertainties regarding its prevalence and variability between studies. This review aims to estimate the prevalence of fatigue and determine the factors responsible for its variation in the literature.
Methods
A systematic review was conducted for all published studies (search to November 2020) using AMED, CINAHL, EMBASE, MEDLINE, PsycINFO, SCOPUS and Web of Science. Papers were included if they recruited participants with stroke, used a validated scale to measure fatigue and were in English. Two reviewers screened and assessed the relevant studies for eligibility (n = 96). The included papers were appraised using the Joanna Briggs Institute (JBI) tool for prevalence studies, and data were extracted by one reviewer. To understand the variation in PSF prevalence between papers, data were pooled and analysed based on relevant methodological (e.g. time of assessment) or clinical factors (e.g. depression) using Review Manager 5.4 software.
Results
While 48 studies were included and summarised (N = 9004), only 35 were appropriate for the meta-analysis (N = 6851). The most frequently used tool to measure fatigue was the Fatigue Severity Scale (FSS) (n = 31). The prevalence was calculated with a cut-off point of four or more using FSS and resulted in an estimate of 48% (95% CI 42–53%). Time of assessment (<6 vs ≥6 months), stroke type (ischaemic vs haemorrhagic/subarachnoid haemorrhage) and geographical location (East Asia vs Europe) could explain the prevalence variation between studies.
Conclusions
Fatigue is prevalent among stroke survivors. This condition varies in terms of occurrence between studies; however, time of assessment, stroke type and geographical location might explain this variation. As this review estimates the overall burden of fatigue after stroke, it provides a useful indicator to inform policy, planning and healthcare professionals. Further efforts are required to investigate the mechanisms that lead to PSF, particularly in the groups that show high prevalence, in order to prevent or alleviate it.
Introduction
Stroke has many complications, one of which is fatigue. A recent scoping review reported that fatigue is one of the commonest secondary conditions among stroke survivors. 1 It has also been ranked as one of the top research priorities in the United Kingdom. 2 Although there is no consensus on its definition, a practical one is ‘a sensation of exhaustion during or after usual activities, or a feeling of inadequate energy to begin these activities’.3,4 Since defining the condition is open to personal interpretations, this has reflected on the methods that are been used to measure it. These methods are usually subjective and consist of a self-reported questionnaire. Some of the frequently employed tools are the Fatigue Severity Scale (FSS) and Multi-dimensional Fatigue Inventory (MFI).5,6
Post-stroke fatigue has a negative impact on patients’ quality of life and independence, and the evidence of its association with high morbidity and mortality rates is well-established.7–10 This condition has been reported to affect around 29–68% of stroke survivors. 11 However, there is a noticeable variability in this range. This variation has been suggested to be due to multiple reasons, broadly the methodological and clinical differences between studies. One factor that has been claimed to play a role is the selection of a scale to measure fatigue, which might be influenced by the fact that there is neither a clear definition of PSF nor a standardised tool to assess it. 12 Nevertheless, even in studies that used the same scale and cut-off point, there was still a considerable variation between their estimates. Moreover, it has been proposed that the time of assessment might affect the occurrence, resulting in more patients reporting fatigue in later months as they start resuming their normal activities. But the evidence around this factor was conflicting for some cohorts showed an increasing trend in PSF prevalence, while others a downward or stable one. 4 Finally, it has been proposed that other methodological factors such as geographical locations or eligibility criteria, specifically related to depression, disability or cognitive impairment, are somehow related. However, there is still a lack of comprehensive, systematic evidence to support that.13–16
Post-stroke fatigue has increasingly drawn researchers’ attention, particularly for the last decade, but there is still a scarcity of extensive evidence to estimate its prevalence and determine the factors of its variability. Since this condition has multiple implications on stroke survivors and their families’ lives, and there is a need for evidence synthesis regarding its occurrence and the causes of its variation between studies, we aim to address this gap by conducting a systematic review of the literature.
Methods
The data that support the findings of this study are available from the corresponding author upon reasonable requests. This review is complied with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, and it is registered with the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42020201168).17,18
Eligibility criteria
Studies were eligible if they recruited adults (≥18) with stroke, used a validated fatigue measurement scale with a cut-off point, were published in a peer-reviewed journal and in English. Studies were excluded if they recruited participants with other brain injuries (e.g. transient ischaemic stroke, traumatic brain injury), were non–peer-reviewed publications (e.g. conference abstracts) or did not use a structured and validated tool to measure fatigue.
Search strategy
An extensive literature search was conducted between October and November 2020. Search terms were developed based on three key words, namely, stroke, fatigue and prevalence. Databases included AMED (inception to October 2020), CINAHL (1937 to November 2020), EMBASE (1947 to October 2020), MEDLINE (1865 to November 2020), PsycINFO (1806 to October 2020), SCOPUS (1960 to November 2020) and Web of Science (1900 to November 2020). The search was also complemented by scrutinising key papers’ reference lists (i.e. snowballing). A full search strategy of one of the databases is in the online supplementary materials.
Procedure
Two authors (IA and AW) conducted the search and initially screened results’ titles and abstracts. Once the irrelevant papers were excluded, full texts were obtained for the remaining studies to be assessed for eligibility. Any exclusion occurred at this stage was recorded and presented on the PRISMA flow chart (Figure 1). Due to time restrictions, the methodological quality of the included studies was appraised by one reviewer (IA) using the Joanna Briggs Institute (JBI) checklist which is designed for systematic reviews of prevalence data.
19
This tool has nine anchoring statements ranging from the appropriateness of the sample frame to the validity of the methods used to identify the condition. The checklist is explained in depth in the JBI reviewers’ manual.
20
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMAs) flow diagram.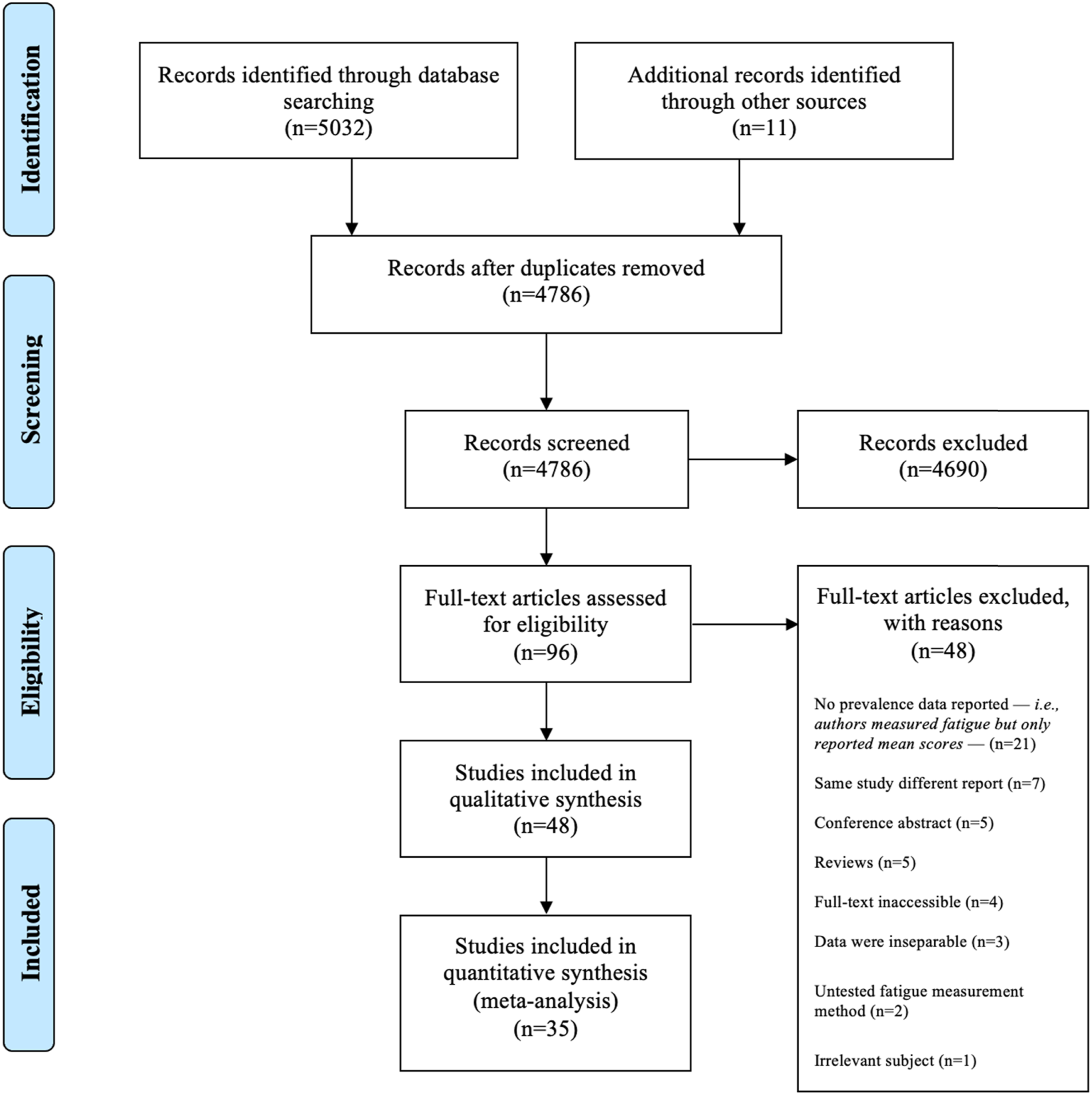
Data extraction
Data were extracted by (IA) using an adapted JBI form which included descriptive and analytic details such as study design; setting and location; eligibility criteria; population demographics; number of participants; number of fatigued participants; fatigue scale and data analysis (e.g. measures of associations). Any further data or clarifications needed were requested directly from the authors via emails. The extraction form can be found in the online supplementary materials.
Statistical analysis
Prevalence data were recorded, and standard errors were calculated using the following formula:
Results
The initial search produced 5032 results. After excluding irrelevant studies and duplicates, 96 articles remained, and their full texts were obtained to be assessed for eligibility. Ultimately, 48 studies were included in this review (Figure 1).
Summary of the included studies.
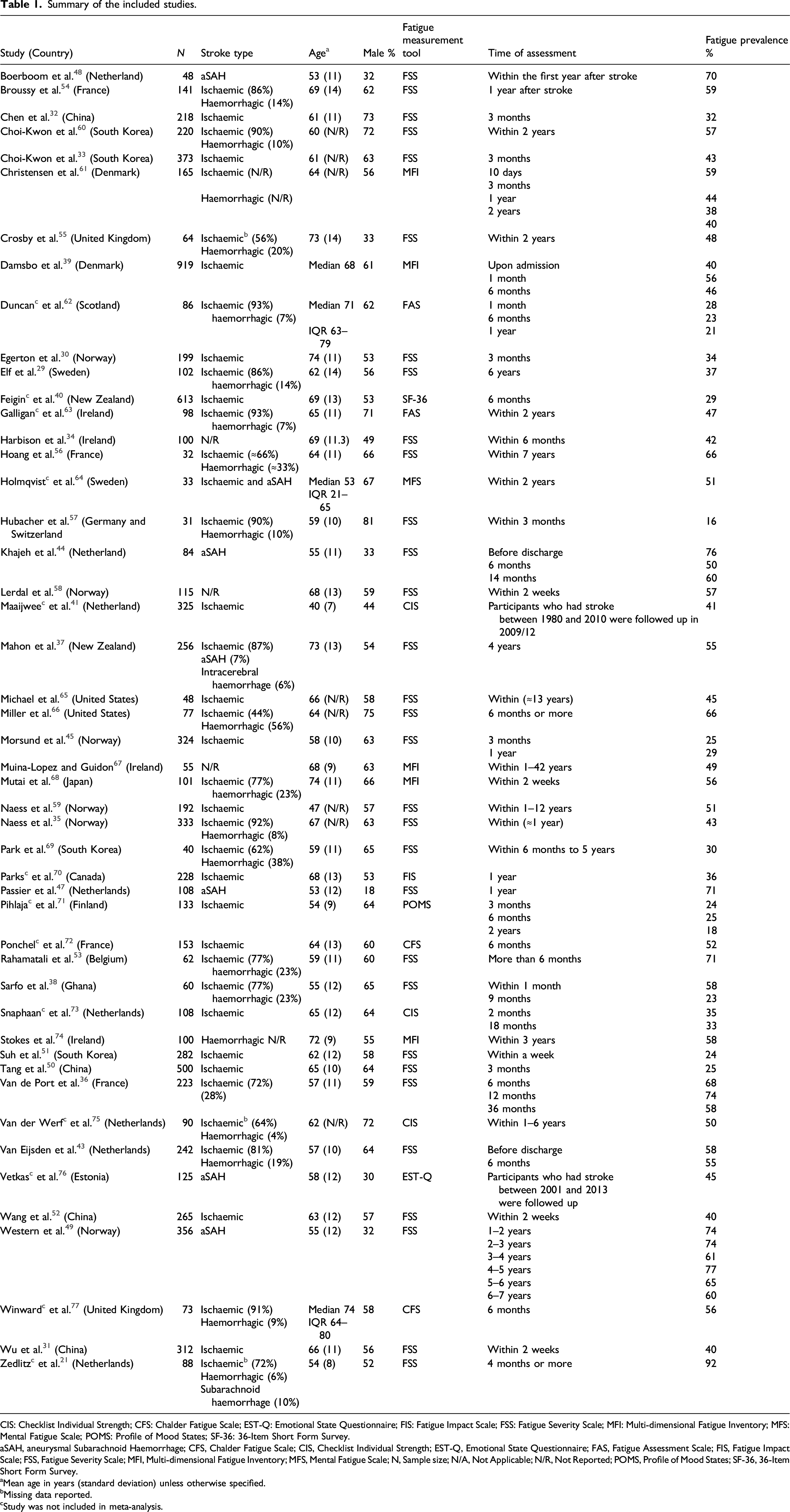
CIS: Checklist Individual Strength; CFS: Chalder Fatigue Scale; EST-Q: Emotional State Questionnaire; FIS: Fatigue Impact Scale; FSS: Fatigue Severity Scale; MFI: Multi-dimensional Fatigue Inventory; MFS: Mental Fatigue Scale; POMS: Profile of Mood States; SF-36: 36-Item Short Form Survey.aSAH, aneurysmal Subarachnoid Haemorrhage; CFS, Chalder Fatigue Scale; CIS, Checklist Individual Strength; EST-Q, Emotional State Questionnaire; FAS, Fatigue Assessment Scale; FIS, Fatigue Impact Scale; FSS, Fatigue Severity Scale; MFI, Multi-dimensional Fatigue Inventory; MFS, Mental Fatigue Scale; N, Sample size; N/A, Not Applicable; N/R, Not Reported; POMS, Profile of Mood States; SF-36, 36-Item Short Form Survey.
aMean age in years (standard deviation) unless otherwise specified.
bMissing data reported.
cStudy was not included in meta-analysis.
Authors who used FSS mostly selected the nine-item version (n = 29), whereas only two authors preferred the seven-item scale.29,30 Most studies had a pre-specified aim to estimate fatigue prevalence except seven papers that focused on assessing the contributing factors of the condition.21,31–36 In terms of quality, the majority of studies had a medium to high-quality score of five or above out of nine (n = 20) (Figure 2). The most failed criterion was appropriate sampling as only two papers had a representative sample, with either a population-based or randomised sample, whereas the majority had a consecutive sampling.37,38 All studies reported a cut-off point of four or more to identify fatigued participants. There was only one study that exclusively included participants with severe fatigue and reported a PSF prevalence of 92%, as a result, it was not included in the final quantitative synthesis.
21
Data were pooled for 30 studies (N = 5511) to estimate PSF prevalence and resulted in a figure of 48% (95% CI 42–53), with high heterogeneity ( Critical appraisal of the included studies. Random effects meta-analysis of post-stroke fatigue prevalence in studies that used FSS and MFI to measure fatigue. FSS: Fatigue Severity Scale; MFI: Multi-dimensional Fatigue Inventory.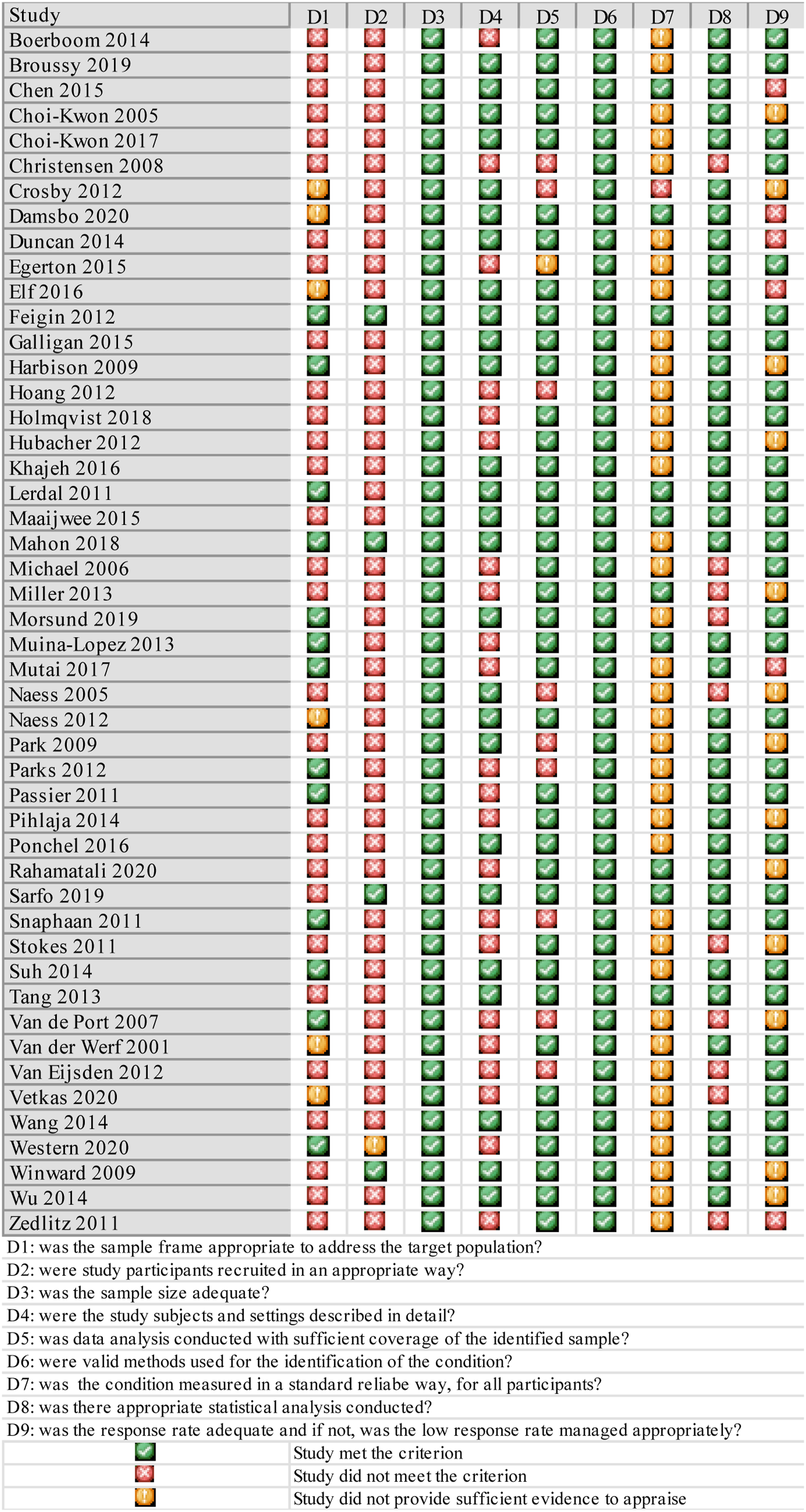
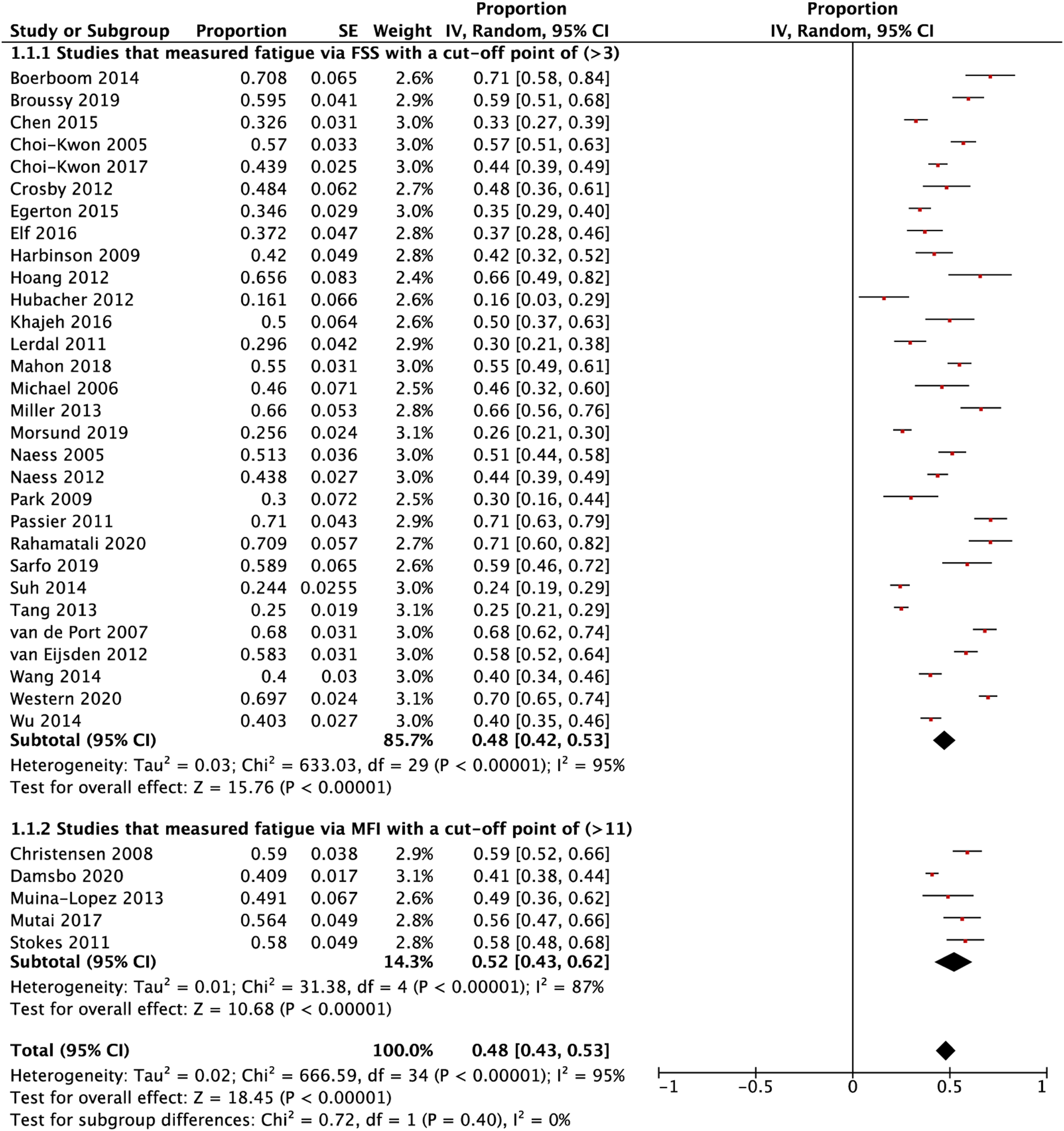
Five studies employed the general fatigue subscale of MFI, with a cut-off point of 12 or more to measure PSF, and three of which had a medium to high-quality score. All studies aimed to estimate fatigue prevalence in stroke survivors except one that aimed to investigate the determinants of the condition.
39
Furthermore, all papers lacked an appropriate sampling method and only one report had a detailed description of the setting and participants. The combined PSF estimate was 52% (95% CI 43–62%) (N = 1199), with high heterogeneity (
As estimates varied across the studies, stratified analyses were carried out. Based on the time of assessment, patients who were interviewed within the first 6 months had a prevalence of 36% (95% CI 30–42%), whereas those who were assessed after that had a higher proportion of 56% (95% CI 50–63%) (p < 0.001). Moreover, while participants with ischaemic stroke had a prevalence of 36% (95% CI 30–42%), those who suffered from haemorrhagic stroke had nearly double that figure 66% (95% CI 59–74%) (p < 0.001). Additionally, studies that were conducted in Asia had a lower estimate 37% (95% CI 29–45) than those carried out in Europe 51% (95% CI 42–59%) (p = 0.02) (Figure 4). Random effects meta-analysis of post-stroke fatigue prevalence stratified according to time of assessment, stroke type and location.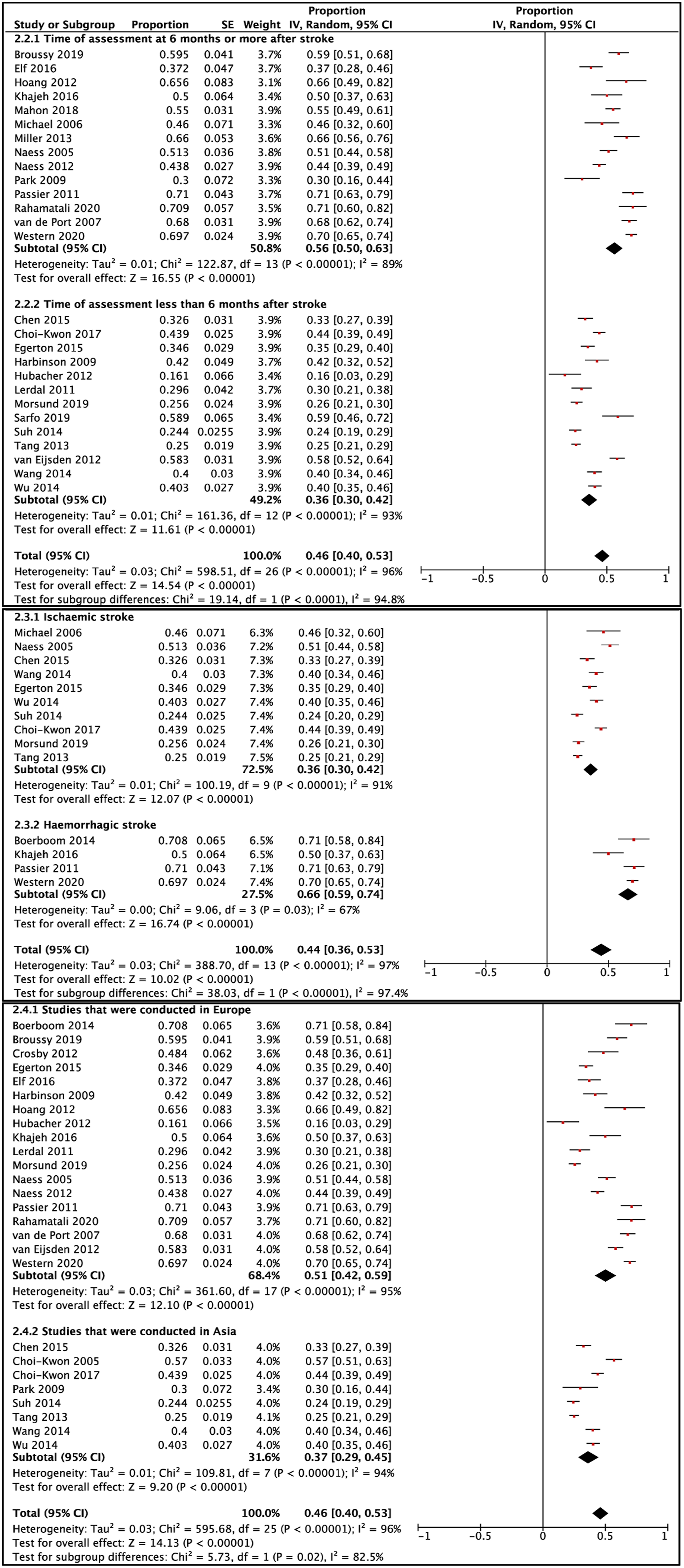
Based on other eligibility criteria and methodological factors, stratified analyses were performed to further understand the variation. There was little to no evidence of a difference between PSF estimate in studies with sample size of 100 or more 45% (95% CI 38–52%) and those with less than that number 52% (95% CI 41–64%) (p = 0.30), though the confidence interval was narrower in the first group. Moreover, studies that excluded patients with depression had a lower proportion 41% (95% CI 32–50%) than those that did not 51% (95% CI 43–58%); however, there was very weak evidence of difference between them (p = 0.10). Other eligibility criteria, such as disability, cognitive impairment and cancer, demonstrated no evidence of a difference between prevalence data in studies that included affected participants and those that did not with (p = 0.47), (p = 0.42) and (p = 0.69), respectively (see Supplemental material).
Discussion
This review demonstrates that almost half of adults complain from fatigue at some point after stroke, though this estimate should be treated with caution due to high heterogeneity. Time of assessment, stroke type and geographical location could explain the prevalence variation between studies.
Consistent with a previous review, PSF prevalence in this study ranges between 42 and 53% in studies that used FSS, whereas 43–62% in those that employed MFI, but in each group the heterogeneity is high, with
The first methodological factor investigated was the time of assessment. Interestingly, this review finds that PSF proportion is lower within the first 6 months following a stroke than later on (36% vs 56%). This is in contrast to what has been stated in the literature that the course of the condition is generally persistent over time. However, when we individually examine the included longitudinal studies, two papers report a decrease of fatigue levels over time,38,43 while another two show a fluctuation,36,44 and only one study demonstrates an increase. 45 Notably, the studies that demonstrate a decline in fatigue scores followed the same cohort whereas the others do not report if that is the case. It is also worth considering that the two studies that followed the same patients one of them has a relatively small sample size (N = 60), 38 while the other shows that 66% of its cohort has remained either fatigued or non-fatigued during the follow-up period. Factors which influence the time course of fatigue are complex and can be biological or psycho-social, nonetheless, a few have been proposed, namely, post-stroke depression and stroke severity, though the temporal relationship between the former and fatigue lacks sufficient evidence.13,38
One unexpected outcome is the stark difference in PSF proportion between patients with ischaemic stroke and those with haemorrhagic stroke (36% vs 66%). To our knowledge, this area has insufficiently been investigated in the literature. One review that examines this subject at length discusses a few determinants that play a role. 46 One factor that has been reported is the female sex. Consistently in this review, the studies that exclusively recruited patients with haemorrhagic stroke at least two-thirds of their participants were women.44,47–49 Other determinants reported are mood disorders that follow ictus; however, there are not sufficient data to firmly conclude the association. Since this phenomenon is not fully understood, there is still abundant room for further studies to better understand the mechanisms behind those associations and support the proposed contributing factors.
Consistent with the literature, this review finds that studies conducted in Europe have higher PSF proportions than those in East Asia. In one review, this is attributed to cultural differences in psychosocial factors rather than epidemiological determinants such as stroke type, though this review has found it to be a factor. 42 This is evident as our results show that most of the included studies in Asia have participants with only ischaemic stroke (n = 6),31–33,50–52 whereas those in the Europe include patients with either both types of stroke (n = 11) or only haemorrhagic stroke (n = 4).34–36,43,44,47–49,53–58 A post-hoc sensitivity analysis supporting this demonstrates that studies in Europe that only included patients with ischaemic stroke have a similar PSF proportion to the ones conducted in Asia (37% vs 37%).29,30,45,59 To develop a comprehensive picture of why PSF is more prevalent in Europe than in East Asia, and to test our theory, further investigations are recommended.
We further investigated other methodological factors by using some of the eligibility criteria. Despite the narrower range that PSF estimate has in studies with sample size of 100 or more than those with less than that, there is no evidence of a significant difference between the two groups (p = 0.30). This could be explained by the fact that the condition is prevalent unlike other rare illnesses that require larger sample size to identify it. Furthermore, although the proportion is higher in studies that have included participants with depression than those that have not (51% vs 41%), there is still very weak evidence of a significant difference between the two categories (p = 0.10). This is also evident in another review which concludes that depression is not likely to explain the variability in PSF proportions between studies as a considerable number of stroke patients without depressive symptoms suffer from weariness. 42
Although the majority of the included studies have a medium to high quality of evidence, there is no consensus on one validated tool to assess fatigue after stroke. This area requires further research to standardise the method of identifying the condition. Despite this variety, PSF is still common, even if we accept the lower end of its occurrence range among stroke survivors. Therefore, healthcare professionals who care for this specific population should be aware of it. As this review concerns a prevalence of a condition, it will be the first step to inform policy and planning for future care services targeting stroke survivors who are at risk or experience fatigue.
This review has several limitations on both the review and included studies levels. Although it was planned a priori, the search was limited to the published and English literature. This, in theory, could have led to missing out on relevant articles. However, we scrutinised the key references’ lists and this has expanded the comprehensiveness of this review. In addition, SCOPUS had a limited number of keywords per set which affected the number of synonyms searched per key term. Furthermore, although a validated tool was used to appraise the included studies, it was conducted by one reviewer, as a result, this might have introduced a subjective assessment and risk of bias during the critiquing process. Additionally, due to the insufficient number of studies or difficulty in retrieving data, some eligibility criteria were not dissected in the subgroup analyses, such as age and sampling methods. On the study level, fatigue was assessed at separate time points, in different geographical locations or settings and with distinct assessment tools and ascertainment methods, which theoretically might have led to methodological and clinical heterogeneity, though some of these factors were considered in the stratified analyses.
Summary/conclusions
This review demonstrates that fatigue is prevalent among stroke survivors. This condition varies in terms of occurrence between studies; however, time of assessment, stroke type and geographical location might explain this variation. As this review estimates the overall burden of fatigue after stroke, it provides a useful indicator to inform policy, planning and healthcare professionals. Continued efforts are needed to investigate the mechanisms that lead to PSF, particularly in the groups that show high prevalence, in order to prevent or alleviate it.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211047681 – Supplemental Material for Prevalence of fatigue after stroke: A systematic review and meta-analysis
Supplemental Material, sj-pdf-1-eso-10.1177_23969873211047681 for Prevalence of fatigue after stroke: A systematic review and meta-analysis by Ibraheem Alghamdi, Cono Ariti, Adam Williams, Emma Wood and Jonathan Hewitt in European Stroke Journal
Footnotes
Acknowledgements
We thank Mari-Ann Hilliar, a librarian at Cardiff University, for her support in developing the protocol. We also thank the authors who replied to our emails.
Contributorship
The authors have contributed equally to the preparation and writing of this review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
IA
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
