Abstract
Background:
Fatigue is a common and disabling symptom following stroke, but its underlying mechanisms are unknown. Associations with a number of imaging features have been proposed.
Aims:
We aimed to assess whether neuroimaging parameters could better inform our understanding of possible causes of post-stroke fatigue (PSF) through systematic review and meta-analysis.
Methods:
Using a predefined protocol registered with PROSPERO (ID: CRD42022303168), we searched EMBASE, MEDLINE, PubMed, and PsycInfo for studies assessing PSF and computerized tomography (CT), magnetic resonance (MR), positron emission tomography (PET) imaging, or diffusion tensor imaging (DTI). We extracted neuroimaging parameters and narratively analyzed study results to assess any association with PSF. Where there were 3+ similar studies, we carried out a meta-analysis using inverse-variance random-effects model to estimate the total association of each neuroimaging parameter on PSF. The risk of bias was assessed using the Newcastle and Ottawa Scale.
Results:
We identified 46 studies (
Conclusion:
There was little evidence for the association between any neuroimaging parameters and PSF. Future studies should utilize advanced imaging techniques to fully understand the role of lesion location in PSF, while the role of altered brain networks in mediating PSF merits further research.
Introduction
Fatigue is a common and often debilitating symptom following both ischemic and hemorrhagic stroke. It has a major impact on quality of life1,2 and has been identified as one of the unmet needs in stroke research. 3 Depending on the definition used, estimates of the prevalence of post-stroke fatigue (PSF) are around 50%.4,5 Despite its importance, there are no proven treatments for PSF, a fact partly explained by a lack of understanding of its underlying pathophysiology. Potential suggested mechanisms include altered cortical excitability, involvement of specific brain regions, disruption of complex brain networks, and systemic inflammation. 6
If the involvement of specific brain regions or altered brain network activity does play a role, then one might expect neuroimaging to provide important insights into the pathophysiology of PSF. This includes identifying associations with structural anatomical features such as infarct location and size, as well as associations with network integrity and “brain activity.” Most studies describing associations of neuroimaging features with fatigue have been small, and while some have reported associations, for example, with white matter hyperintensity (WMH), 7 and subcortical infarcts7,8 others have not replicated these.1,9
To better understand the relationship between brain structure and function and fatigue and gain insights into the pathophysiology of PSF, we performed a systematic review of neuroimaging variables and their relationship to PSF.
Methods
In line with PRISMA guidelines, 10 the systematic review protocol was preregistered on PROSPERO (ID: CRD42022303168). 11
Searches
We searched four electronic databases using our predefined search strategy (available in Supplemental Appendix 1)) on 20 May 2023. These databases were: EMBASE (from 1974), MEDLINE (from 1946), PubMed (from 1950), and PsycInfo (from 1955). Studies had to assess fatigue and stroke and neuroimaging to be included in record screening (see search terms, Supplemental Appendix 1).
Inclusion and exclusion criteria
We included randomized controlled trials, observational, cohort, case–control, and cross-sectional studies. Only studies in the English language were included.
Studies were included if they assessed stroke patients over 16years old, with any subtype of stroke (e.g., ischemic or intracranial hemorrhage), and any duration since stroke. Studies had to include any assessment of fatigue and at least one of the following imaging modalities: computerized tomography (CT), magnetic resonance (MR), positron emission tomography (PET) imaging, or diffusion tensor imaging (DTI).
Studies were excluded if they assessed patients: under 16, included transient ischemic attack or subarachnoid hemorrhage, or did not assess any of the variables listed above. Gray literature, unpublished literature, abstracts, narrative reviews, and case reports and series in <5 subjects were excluded.
Data extraction
Two reviewers (A.A.J. and A.Z.) independently screened all titles and abstracts. Any disagreement between reviewers was resolved by discussion between the two reviewers and, if no consensus was obtained, by discussion with a third reviewer (H.S.M.).
Risk of bias
The Newcastle and Ottawa scale (NOS) 12 was utilized to assess the risk of bias. We used the original NOS 12 and the modified NOS for cross-sectional research 13 (Supplementary sTable 1). There are no set cut-offs, but higher scores indicate higher-quality studies. 14
Data synthesis
As this was an exploratory review, we planned to look narratively at studies that corrected for confounders and those that did not together. Main findings for each outcome measure were narratively described. Where appropriate, if three or more studies were sufficiently similar, in both fatigue measure used and neuroimaging parameter, meta-analysis was performed with R (version 4.2.2) package metafor (v3.8-1),
15
using an inverse-variance random-effects model, to estimate the total effect of each neuroimaging measure on PSF. To retain the maximum number of studies in our meta-analyses, we included studies binarizing fatigue using validated cut-offs across multiple measures. We included only raw data or univariate odds ratios in our meta-analyses to ensure consistency in included studies. A
Results
Identified studies
Our search strategy identified 5769 records; 1858 were duplicates and were removed. Of the remaining 3911, through title and abstract screening, we identified 46 records that met inclusion criteria. The most common reasons for exclusion were the wrong study population (
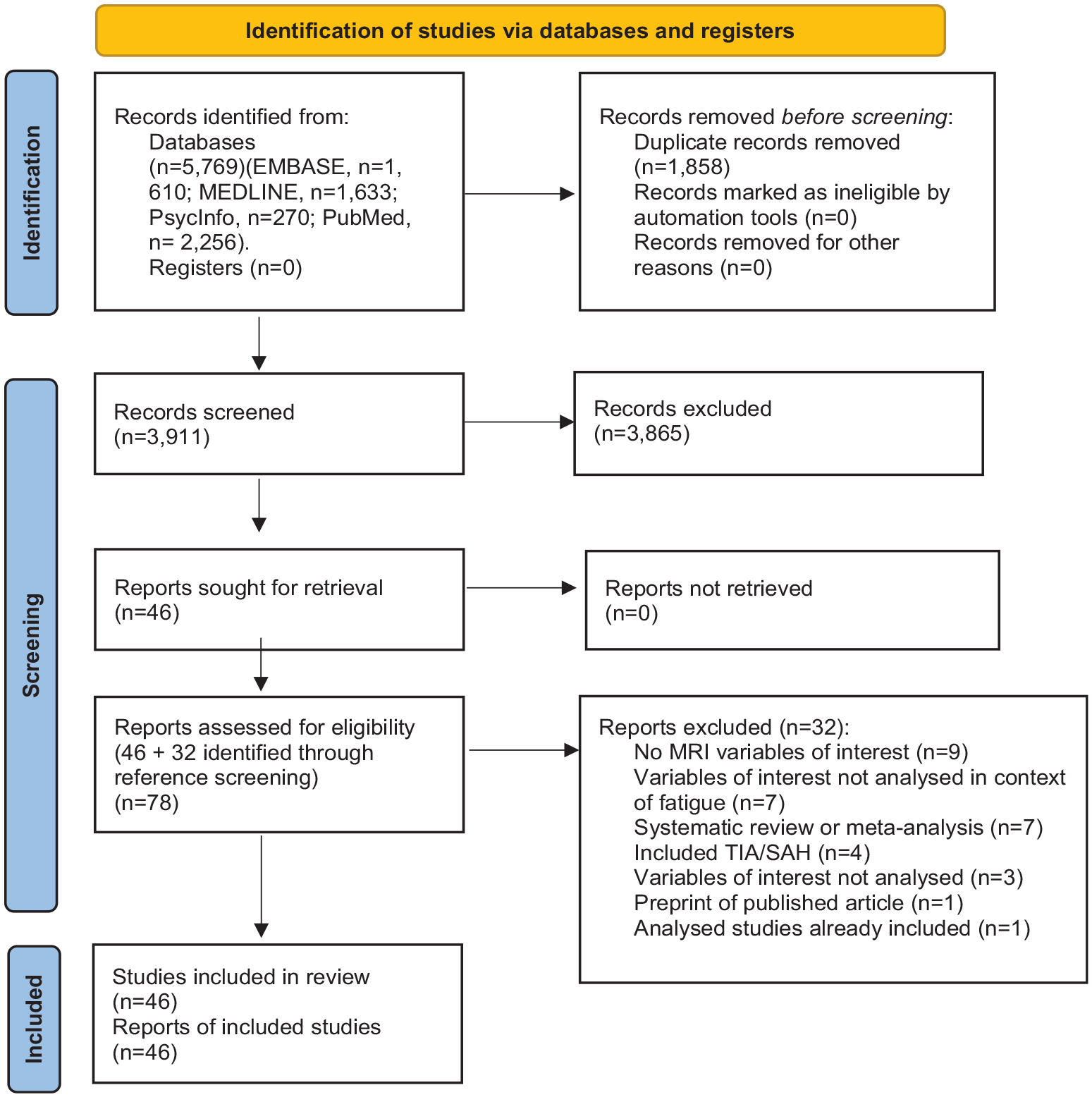
PRISMA flow diagram 10 showing the selection process.
Neuroimaging variable categories and

WMH: white matter hyperintensity.
The 46 studies comprised a total of 6543 participants. The most common scale used to assess fatigue was the Fatigue Severity Scale (FSS) (
Associations of PSF with specific neuroimaging variables
Lesion lateralization
Twenty-nine studies (
Sixteen studies had group data or summary data suitable for meta-analysis.9,16,17,19,21,24,25,27,28,32,34,37,38,43,52,54 Two studies had data for both baseline and follow-up,9,43 we used only the earliest timepoint to remain consistent with the other studies. One study looked at fatigue across three groups (low, moderate, and severe). We collapsed these into two groups: low-moderate (FSS score < 4) versus severe (FSS score > 4), in line with other studies. 25 One study had data presented as percentages for two measures; we only included the measure for which percentages could be converted to the total sample size. 34
On meta-analysis (Figure 2), there was no significant difference between stroke lateralization and fatigue; OR for left-side lateralization 0.88 (95% CI 0.64, 1.22) (
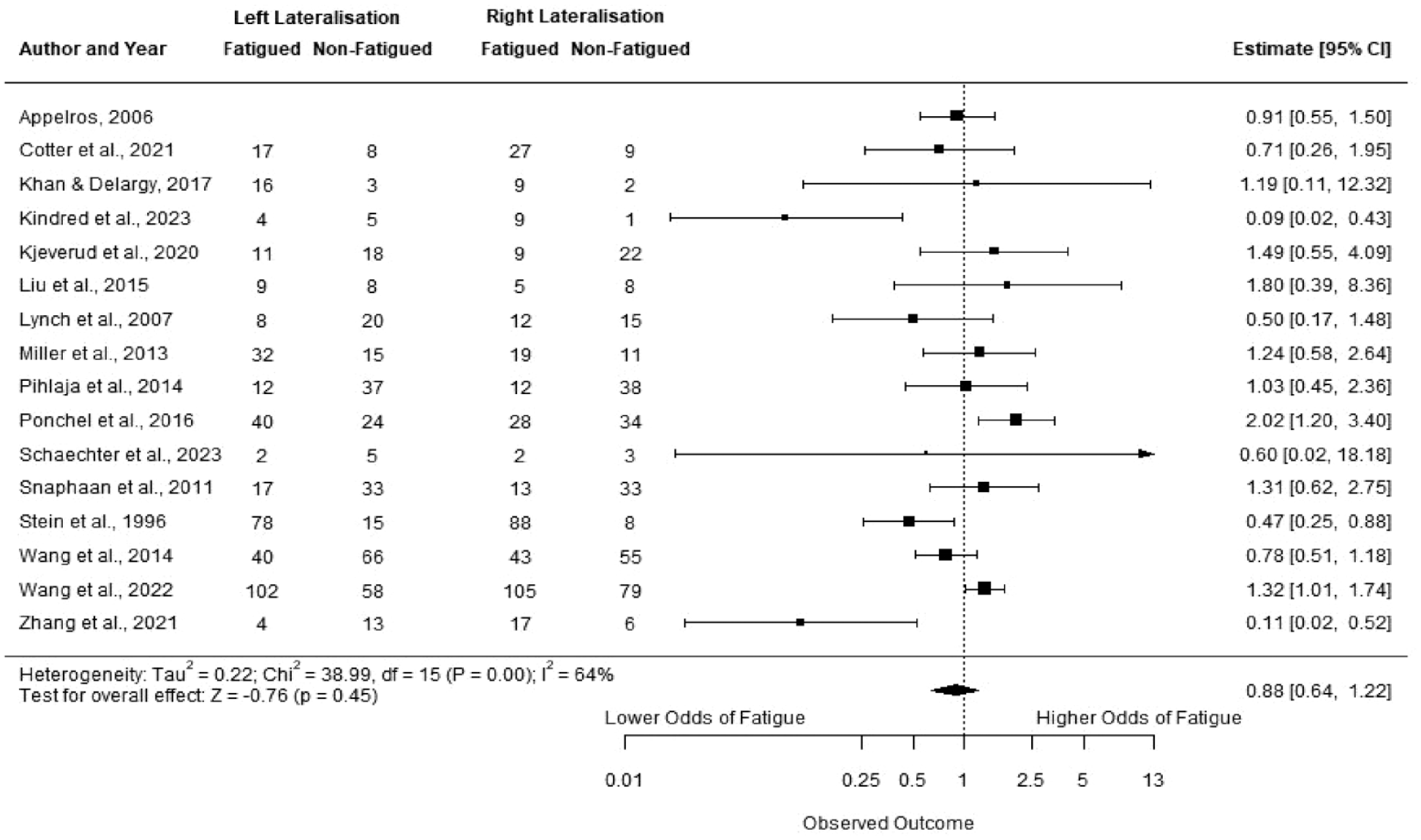
To assess whether the measure of fatigue used altered results, we performed a sensitivity analysis including only a single measure of fatigue (
Lesion location
Nineteen studies in 3036 subjects assessed whether there was an association between lesion location and fatigue.1,7–9,19,27,29,31,33,38,43,46–53 Lesion locations and study results are summarized in Table 2.
Association of lesion location to PSF categorized by brain region.
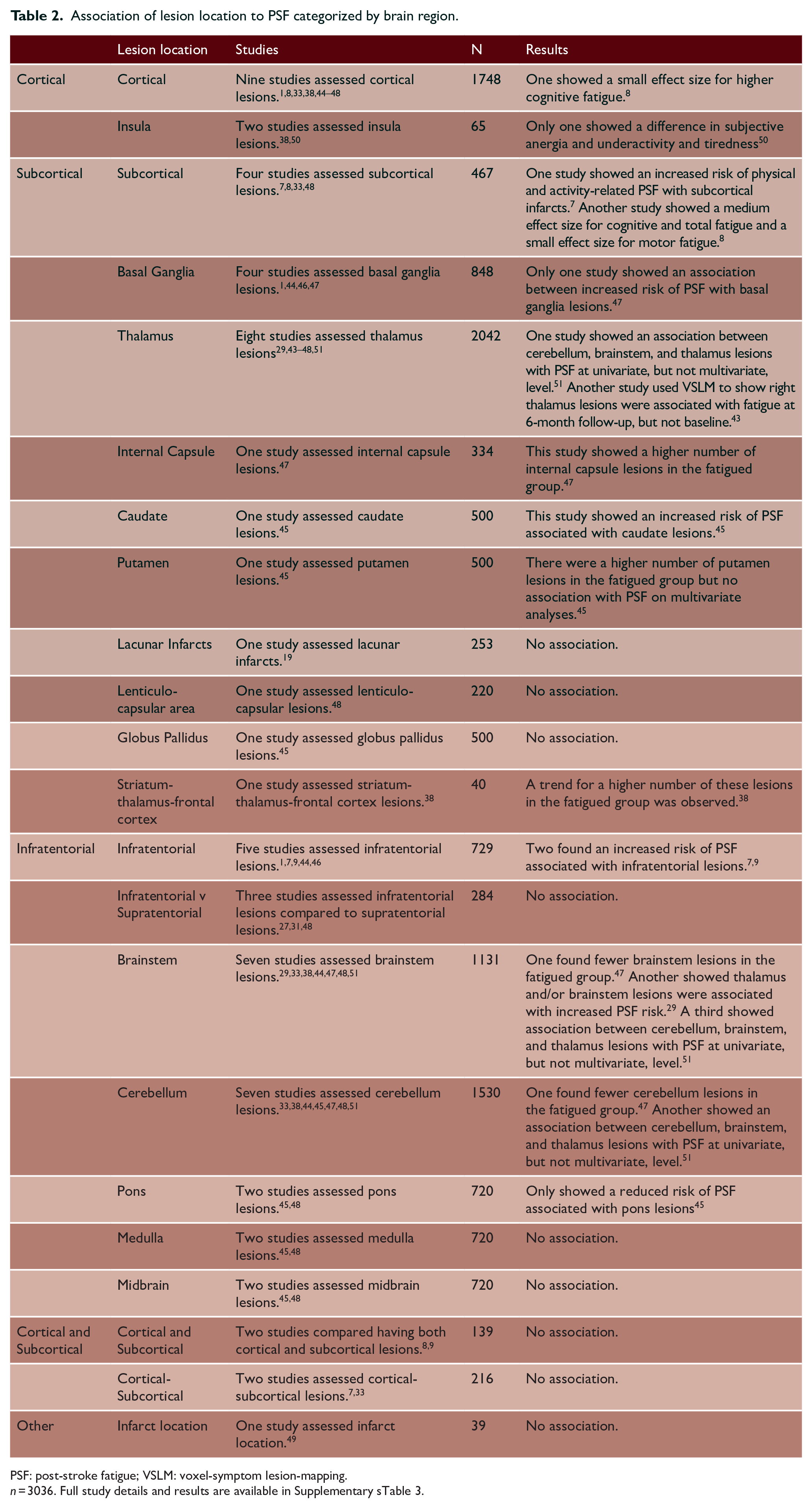
PSF: post-stroke fatigue; VSLM: voxel-symptom lesion-mapping.
Cortical
Nine studies assessed cortical infarcts, with only one (
Subcortical
For the purposes of this review, we excluded brainstem and cerebellum from the category-defined subcortical structures and rather discussed them in the infratentorial section (see Table 2).
Four studies looked at associations with any subcortical lesions, of which two larger studies (
Four studies assessed basal ganglia lesions.1,44,46,47 Three of these studies were conducted by the same research group; we contacted the corresponding author to enquire whether study populations were overlapping but did not receive a reply. Only one out of the three showed an association between acute basal ganglia infarcts and increased PSF risk (OR: 2.08, 95% CI (1.16, 3.75),
Eight studies assessed thalamic lesions,29,43–48,51 five studies, four by the same group, showed no association.44–48 One study showed thalamus and/or brainstem lesions were associated with PSF,
29
while another found thalamus lesions to be associated with PSF in 230 participants.
51
Another study used voxel-symptom lesion-mapping (VSLM) to show right thalamus lesions predicted PSF at follow-up (
Only one study (
Infratentorial
Infratentorial lesions were defined as: brainstem, cerebellum, pons, medulla, and midbrain (Table 2). Infratentorial lesions were investigated in eight studies. Six (
Of seven studies assessing brainstem lesions,29,33,38,44,47,48,51 one showed thalamus and/or brainstem lesions predicted general fatigue,
29
while another (
Four studies had either infratentorial group data or summary statistics suitable for meta-analysis.7,9,27,46 Two studies had data for multiple timepoints; we included only the earliest timepoint for consistency with other studies.7,9 On meta-analysis (sFigure 1), there was no significant association between infratentorial lesions and fatigue; OR: 1.83, 95% CI [0.63, 5.32] (
Lesion volume
Sixteen studies with 2755 participants tested the association between fatigue and lesion diameter or volume. 1,7,20,21,31,32,35,43,44–49,52,53 None identified any significant associations (see Supplementary sTable 4).
Brain atrophy
Four studies in 540 subjects examined brain atrophy measures in relation to the presence and severity of PSF1,7,9,39 (Supplementary sTable 5). All examined associations with global brain volumes, including some subcortical and cortical volumes, and none reported association with PSF. One study (
Number of infarcts
Four studies, all from the same author, assessed whether the number of acute infarcts was associated with PSF44–47 (
Four studies in 922 subjects assessed the relationship between the number of old infarcts and the presence of PSF (Supplementary sTable 6). All studies showed no significant relationship.1,39,44,45
Cerebral microbleeds
Only one study assessed cerebral microbleeds (CMBs) and PSF in 199 subjects.
46
Deep CMBs (basal ganglia, external capsule, internal capsule, and thalamus) were associated with fatigue on univariate analysis (
WMHs
Seven studies in 1336 subjects assessed the relationship between WMH and PSF1,7,9,39,44–46 (Supplementary sTable 7). All used visual rating scales to assess WMH severity (Fazekas scale in 5, ARWMC scale in 1, and one bespoke grading scale). Six showed no association between WMH severity and PSF.1,9,39,44–46 One reported an association with a Fazekas score of 1 and mental fatigue at 3 (OR: 1.54, 95% CI (1.05, 2.21),
Three studies had group data or summary statistics available for meta-analysis.7,9,45 On meta-analysis (sFigure 2), there was no association between WMH and fatigue; OR: 1.21, 95% CI [0.84, 1.75], (
Diffusion tensor imaging (DTI) of white matter ultrastructure
DTI is a sensitive marker of white matter ultrastructural damage in cerebrovascular disease and has been correlated with symptoms such as cognition58,59 and apathy 60 in stroke. At the time of the search strategy, we were unable to find any papers assessing DTI parameters and PSF.
Brain connectivity and network analysis
It has been suggested that cognitive and psychological symptoms arise from damage to anatomically distributed brain networks caused by focal lesions rather than specific lesion locations themselves. 59 Network disruption is important in mediating the effect of cerebral small-vessel disease on both cognition58,61 and apathy. 60 Networks can be assessed structurally using diffusion-weighted imaging and tractography and functionally using functional MRI (fMRI).
Two studies investigated structural tractography-based networks and their relationship to PSF.35,52 The first study constructed individual-level whole-brain disconnectivity probability maps based on lesion maps from 84 stroke patients using normative data from healthy controls. 35 Nonparametric permutation-based inference was used to conduct voxel-wise analyses on disconnectome maps and estimate regional disconnectivity. 35 There was no association between PSF and any disconnectome measures after accounting for multiple comparisons and controlling for depression. 35 A second smaller study used diffusion MRI data from 420 healthy controls to map structural disconnection caused by lesions to the white matter in 12 post-stroke participants. 52 When all lesions were lateralized to the same hemisphere, voxel-wise correlations showed higher fatigue correlated with greater structural disconnection in an ipsilesional cluster (rostral middle frontal cortex and superior frontal cortex) and a contralesional cluster containing six frontal cortex regions (rostral middle frontal cortex, caudal middle frontal cortex, superior frontal cortex, inferior frontal cortex (pars triangularis and pars opercularis), and caudal anterior cingulate cortex). 52 When lesions were not lateralized to the same hemisphere, no significant associations were observed. 52
Three studies assessed resting-state brain activity markers of PSF using fMRI.21,38,52 In 63 stroke subjects, PSF was associated with posterior hypoactivity and prefrontal hyperactivity, as assessed by the amplitude of low-frequency fluctuations (ALFF), which was suggested to reflect dysfunction within large-scale brain systems such as fronto-striatal-thalamic and frontal-occipital networks.
21
A second study in 16 post-stroke patients reported fractional ALFF was lower in several brain areas in PSF, including the right frontal, right inner orbital frontal, right orbital inferior frontal, right triangular frontal inferior, right anterior and lateral cingulate, and right medial frontal gyri.
38
A third small study (
Two studies assessed the effect of modafinil on fMRI functional connectivity (FC) in PSF,55,56 using data from the double-blind MIDAS-II trial (
Two studies tested whether disruption of brain asymmetry was associated with PSF.54,57 The first (
Risk of Bias Assessment
Using the NOS, we identified a moderate level of bias; with a maximum score of 8 or 9 (cross-sectional and cohort studies, respectively) mean (SD) quality score was 4.57(1.14) (Supplementary sTable 1 for individual study bias estimates).
Discussion
In this systematic review and meta-analysis, carried out using robust methodology according to PRISMA guidelines and including a total of 46 papers (n
It has been suggested that a number of the cognitive and psychological consequences of stroke may be mediated by damage to distributed brain networks.58,59,61 We found limited studies investigating associations between network integrity and PSF. Structural network integrity relies on the reconstruction of white matter connections using tractography analysis of DTI. One large study utilizing this technique found no association. 35 Of note, the same technique applied to the same dataset was able to show an association between network integrity and post-stroke cognitive impairment, suggesting the technique does have the power to detect associations between symptoms and network dysfunction. 59 However, a second small study found structural disconnection in several ipsilesional and contralesional frontal brain regions, as well as reduced FC in the ipsilesional rostral middle frontal cortex, associated with PSF. 52
Functional network measures rely on the BOLD MRI signal and temporal correlations between activation in different brain regions. In small or moderate-sized cohorts, these did suggest some potential associations with PSF.21,38,52,55,56 It has been hypothesized that fatigue in neurological disorders may relate to alterations in limbic input and motor functions in the basal ganglia, therefore, affecting activity in the fronto-striato-thalamic system. 63 Consistent with this, one fMRI study identified posterior hypoactivity and prefrontal hyperactivity, which was interpreted to reflect dysfunction within fronto-striatal-thalamic and frontal–occipital networks. 21 The MIDAS-II drug trial identified reduced connectivity of the fronto-striatal-thalamus predicted better response to modafinil. 56 With both structural and functional studies implicating frontal regions,21,52,56 future work should further investigate the role of frontal networks in PSF.
Altered perceived effort may also underlie fatigue; it has been suggested that reduced ability to attenuate irrelevant sensory stimuli drives increased perceived effort and causes feelings of fatigue. 64 One functional network study showed those whose fatigue responded to modafinil treatment had significant decreases in rsFC in the somatosensory cortex, inferior parietal lobule, and temporal pole. 55 This may indicate changes within brain areas associated with sensory processing. Specifically, increased activity in these regions at rest, prior to modafinil use and fatigue improvement, could be reflective of overactivity in sensory processing leading to poorer attenuation and, as a result, fatigue. Furthermore, Ondobaka et al. 57 used rs-fMRI to show that IIB in the M1 was associated with, and could predict, fatigue score. The authors suggest that altered brain asymmetry in the M1 may cause sympathetic central nervous system dominance, leading to higher arousal, poorer sensory attenuation, and, thus, fatigue.57,65
Taken together, our review suggests anatomical characteristics of the stroke lesion do not relate to PSF. In particular, lesion size and laterality are not important in the pathogenesis of PSF, suggesting clinical care should focus on screening and management of known risk factors, such as inactivity 66 and mood, 67 in order to manage PSF.
Whether there is a lack of any relation, or whether the analysis techniques applied have been too simplistic remains uncertain. Taken together, lesion location studies were largely inconclusive, with no consistent associations with any particular region; however, one large VSLM study implicated the right thalamus in PSF at 6 months post-stroke but not at baseline. 43 Future research should utilize such advanced imaging techniques to further probe the relationship between fatigue and lesion location. It is also possible a more network-based approach is required to identify relationships, and early studies have implicated the frontal,38,52 fronto-striato-thalamic,21,56 and sensory processing networks,55,57 but further studies are required to replicate these associations.
It is also possible that perhaps the fatigue measures may have been too simplistic to detect associations between neuroimaging parameters and fatigue. Most studies we identified treated fatigue as a single construct, but this may not be the case. Emerging evidence suggests the cause of PSF is multidimensional. 6 Studies have investigated demographic factors, neurological/physical deficits, medical comorbidities, smoking, medications, sleep disturbances, pain, pre-stroke fatigue, depression and anxiety, and cognitive impairment as potential risk factors. 6 It is, therefore, possible that the same subjective experience of fatigue could result from a number of different mechanisms. The varied response to the modafinil treatment in the MIDAS-II trial further corroborates the heterogenous nature of fatigue.55,56
Future research could investigate the inflammation hypothesis: inflammatory response following stroke upregulates proinflammatory cytokines causing sickness behaviors, further leading to feelings of fatigue. 68 Early studies have shown associations between fatigue after stroke and inflammatory markers,6,31,68 suggesting a viable avenue for future research.
One limitation of this review is the moderate risk of bias displayed by studies; this is likely to be partially explained by the nature of included studies. Many studies were not designed to specifically investigate fatigue and neuroimaging, rather a number examined these as secondary or subanalyses within existing research/research questions (28.3%) (Supplementary sTable 9), highlighting the infancy of this field and the need for more robust research assessing the pathogenesis of PSF. Some studies included were primary, looking for associations between neuroimaging parameters and fatigue; in others, it was a secondary question. However, sensitivity analysis removing secondary analyses from the meta-analyses did not significantly alter results for either lesion lateralization or infratentorial location (results not shown). Additional sensitivity analysis excluding lower-quality studies also did not alter the results of the meta-analyses.
The meta-analyses combined studies regardless of fatigue outcome measure used. As noted above, fatigue is a multidimensional construct, and different outcome measures may measure different, and perhaps not overlapping, domains. 66 For the lesion lateralization meta-analysis, there were sufficient studies to perform a sensitivity analysis using only a single measure of fatigue, and this did not alter the results significantly.
Of note, many studies did not control for confounders in the current review; in fact, most carried out only univariate analyses. Some studies did control for key confounders such as age,9,21,35,43–45 sex9,21,43–45 and depression;1,9,21,29,35,43–45 however, the majority did not. Age and sex have both been previously associated with fatigue, 6 while depression is a known major confounder in the assessment of fatigue. 6 It is key that future research should consider and control for these factors, to accurately identify potential correlates of fatigue.
Finally, many studies considered both ischemic and hemorrhagic stroke, it is important to note that differences in stroke pathophysiology could also mean differing pathomechanisms of fatigue between types of stroke. Future research should aim to assess differences in fatigue across types of stroke.
In conclusion, we found little evidence for any association of neuroimaging features with PSF, although for some imaging variables, there was limited data available. Several studies implicated altered functional brain networks in PSF, but more work is required to confirm and fully understand the relevance and clinical implications of such alterations.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231192214 – Supplemental material for Neuroimaging correlates of post-stroke fatigue: A systematic review and meta-analysis
Supplemental material, sj-docx-1-wso-10.1177_17474930231192214 for Neuroimaging correlates of post-stroke fatigue: A systematic review and meta-analysis by Amy A Jolly, Adriana Zainurin, Gillian Mead and Hugh S Markus in International Journal of Stroke
Footnotes
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.A.J., A.Z., and H.S.M. report no relevant disclosures. G.M. receives royalties from Later Life Training and Elsevier and Consultancy fees from Imperative Care.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.A. Jolly’s studentship is supported by the Stroke Association R4VaD grant (RRZA/199); A.Z. is supported by the Tun Taib Foundation Scholarship; G.M. and H.S.M. report no relevant funding. This research was supported by the NIHR Cambridge Biomedical Research Centre (grant no. NIHR203312). The views expressed are those of the author and not necessarily those of the NIHR or the Department of Health and Social Care.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
