Abstract
Abstract
The first European Stroke Organization (ESO) standard operating procedure (SOP) published in 2015 aimed at the implementation the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology to provide evidence-based guidelines for stroke management. This second ESO-SOP is aiming at further increase of the practicability of ESO guidelines and its technical implications. Authors comprised of the members of the ESO guideline Board and ESO Executive Committee. The final document was agreed on by several internal reviews. The second SOP comprises of the following aspects: rational for the SOP, the introduction of expert consensus statements, types of guideline documents, structures involved and detailed description of the guideline preparation process, handling of financial and intellectual conflicts of interest (CoI), involvement of ESO members in the guideline process, review process, authorship and publication policy, updating of guidelines, cooperation with other societies, and dealing with falsified data. This second SOP supersedes the first SOP published in 2015.
Keywords
Introduction
The European Stroke Organisation (ESO) guidelines are based on the method which was developed by the GRADE (Grading of Recommendations Assessment, Development and Evaluation) working group, as a tool for developing recommendations on treatment 1–16 and on diagnostic strategies. 17 The implementation of the GRADE system into the ESO guideline development process was described in the first ESO standard operation procedure (SOP) in 2015, 18 which was applied to numerous guidelines (https://eso-stroke.org/eso-guideline-directory). We now present the second version of this ESO-SOP.
The second version of the SOP was deemed relevant for several reasons: 1) avoid unnecessary delays which were encountered by adopting the previous procedure (e.g revisions of the guideline in different steps); 2) identify external reviewers in the early stage of the process to allow for revision of the PICO questions and avoid criticism to PICO questions raised only after the full guideline was developed; 3) improve formatting and general presentation of the guideline; 4) review the time schedule of the process; 5) introduce “expert consensus statements” to further increase the practicability of ESO guidelines and provide clinical guidance even where evidence is scarce, of low quality or difficult to interpret.
This second SOP supersedes the aforementioned SOP published in 2015. The mission statement is: Providing evidence-based guidance for stroke management throughout medical systems.
Types of ESO-supported guideline documents
The ESO supports
Types of guideline documents supported by the ESO.

Guideline documents initiated and prepared by the ESO based on the present SOP; non-ESO specialists from other organisations may be invited to participate.
Guideline documents prepared in collaboration with one or more other scientific organisations; the methodological approach will follow the GRADE approach.
Guideline documents prepared by another organisation that will be endorsed by ESO after agreement between ESO and the other organisation(s) that one or several ESO-members will co-write the manuscript prospectively and will be included in the list of authors. We strongly recommend following the GRADE approach also for these guidelines.
Structures involved in the development of ESO guideline documents
The development of ESO guidelines is driven by the
Structures involved in the development of the ESO guidelines.
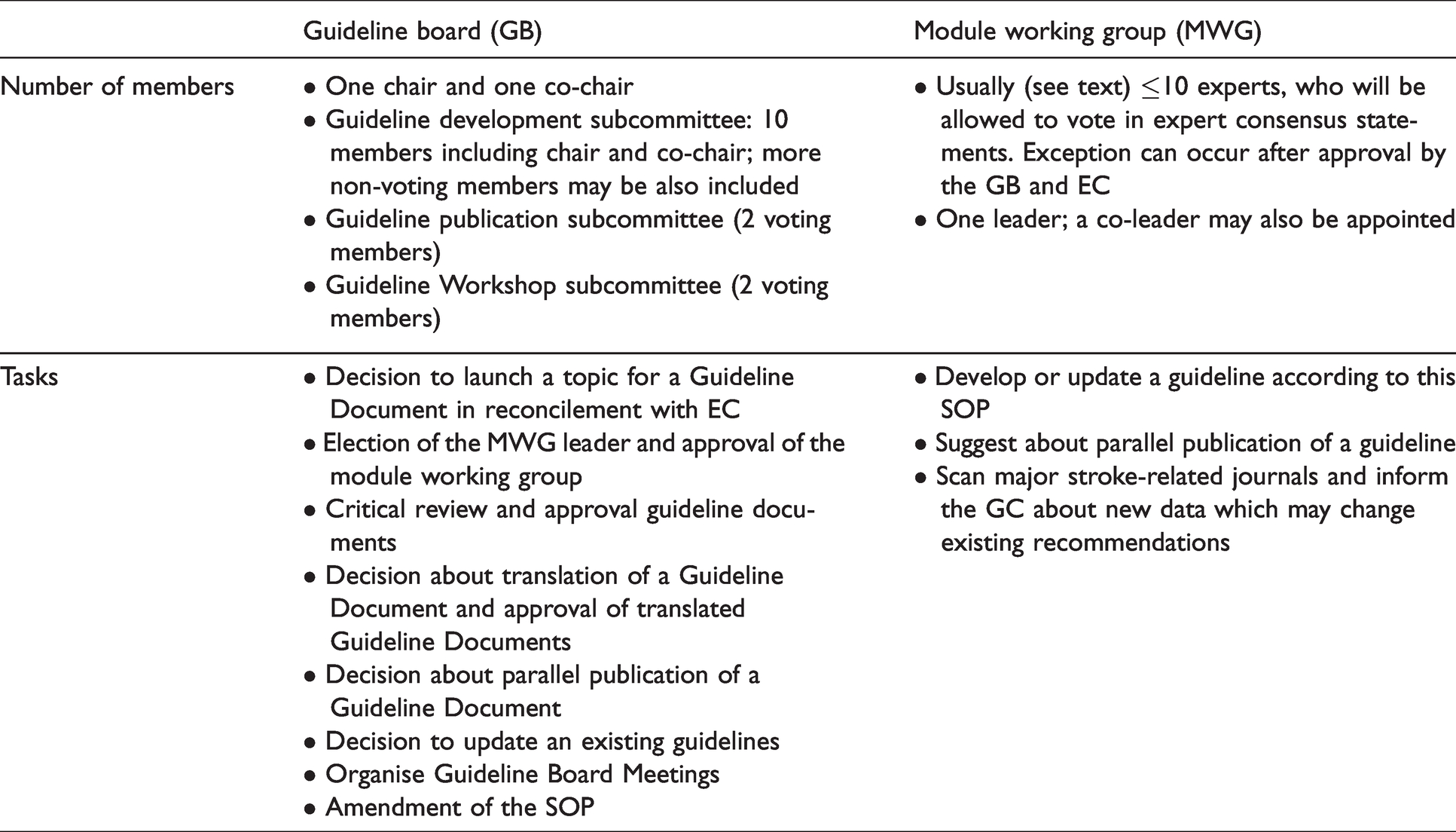
The
A
The
Conflict of interest
A conflict of Interest (CoI) is a set of circumstances that creates a risk that professional judgment or actions regarding a primary interest will be unduly influenced by a secondary interest.19,20 A comprehensive and rigorous process for disclosure of interests and management of COI is essential for the development of high-quality clinical guidelines, as the absence of trust may serve as a barrier to implementation. CoIs are distinguished into “intellectual” and “financial” CoIs – the latter sometimes referred to as “relationships with industry”. CoIs will be assessed by the GB and approved by the EC. The ESO supports the standardization of declarations of CoIs of the international organization and journal participating in the Committee of Medical Journal Editors (ICMJE). All approved CoIs will be declared using the official ICMJE form (www.icmje.org). The derived statements will be published as a table in the addendum of a guideline.
For assessment of financial CoIs these will be differentiated in “moderate” (equal or less than 10,000 €per year) and “significant” (more than 10,000 €per year) within the last three years.
The following Being a principal investigator of an industry-initiated and industry-sponsored trial, registry, other scientific work that relates to a specific guideline, Financial interests in a pharma company of importance to a specific guideline, as personal stocks, ownership, or similar. The leader should not have significant CoI related to the specific Guideline topic.
The following Active financial relationship (e.g. currently serving on an advisory board for pharmaceutical company) related to a topic of a guideline Relationships with entities that may seek to profit by association with guidelines but are not vested in clinical conclusions of guidelines (e.g., proprietary interest in health IT software related to clinical decision making)
The following Intellectual interest that may lead to perceptive bias (e.g. for a guideline on secondary prevention on BP management after stroke, served as investigator on a study testing BP lowering treatment in stroke patients within previous 3 years) Any inactive high-level conflict (e.g. stock ownership) Any intellectual interest that is only indirectly related to the clinical topic area.
The policy should be implemented across all levels, including the participants reporting the CoIs and the graders of CoIs.
Identification of new topics for an ESO guideline document
New topics for ESO guidelines may be identified during meetings supported by the ESO or may be directly suggested to the GB by any ESO member. Input from the aforementioned sources is transmitted to the GB, which decides with a majority vote for new module topics for a new guideline. The GB will prioritize potential topics according to the importance of clinical implications or to the extent of associated controversy. The decision of the GB needs to be approved by the EC.
Preparation of the guideline document
Workflow and methodology.

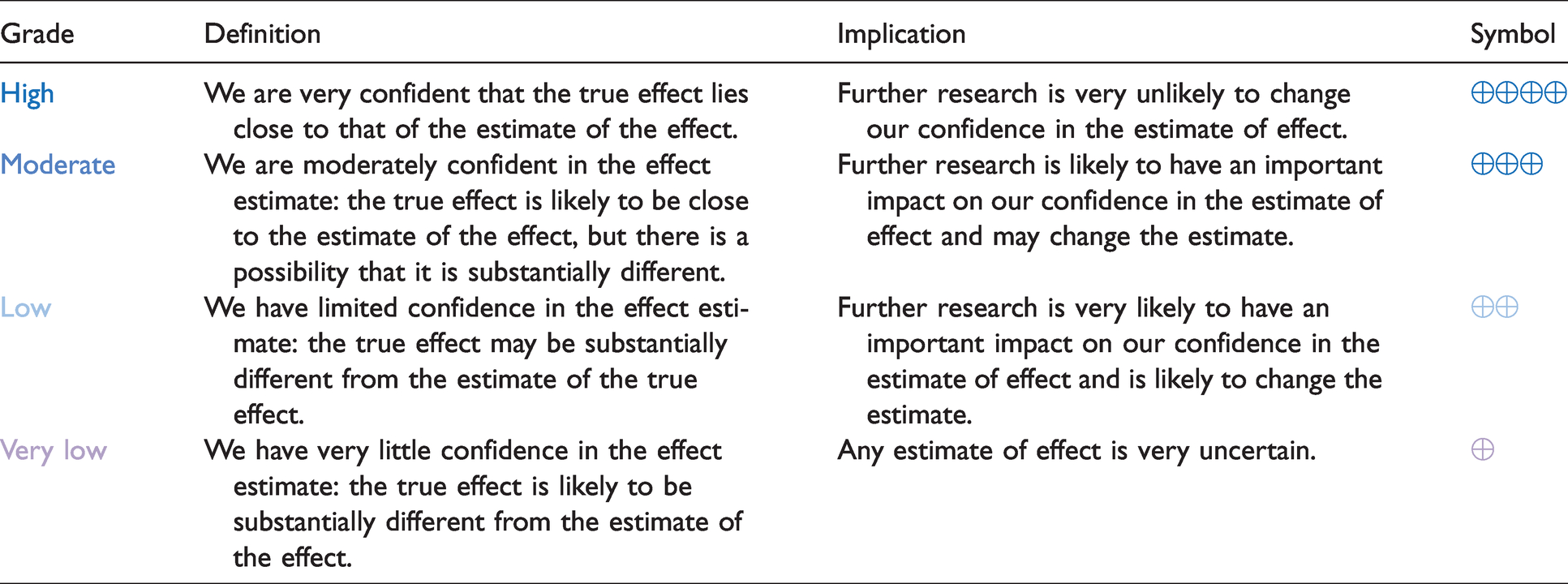
Definitions, implications and symbols of grades of quality of evidence.
Definitions and symbols of categories of strength of recommendation.

Summary of actions towards a guideline document.
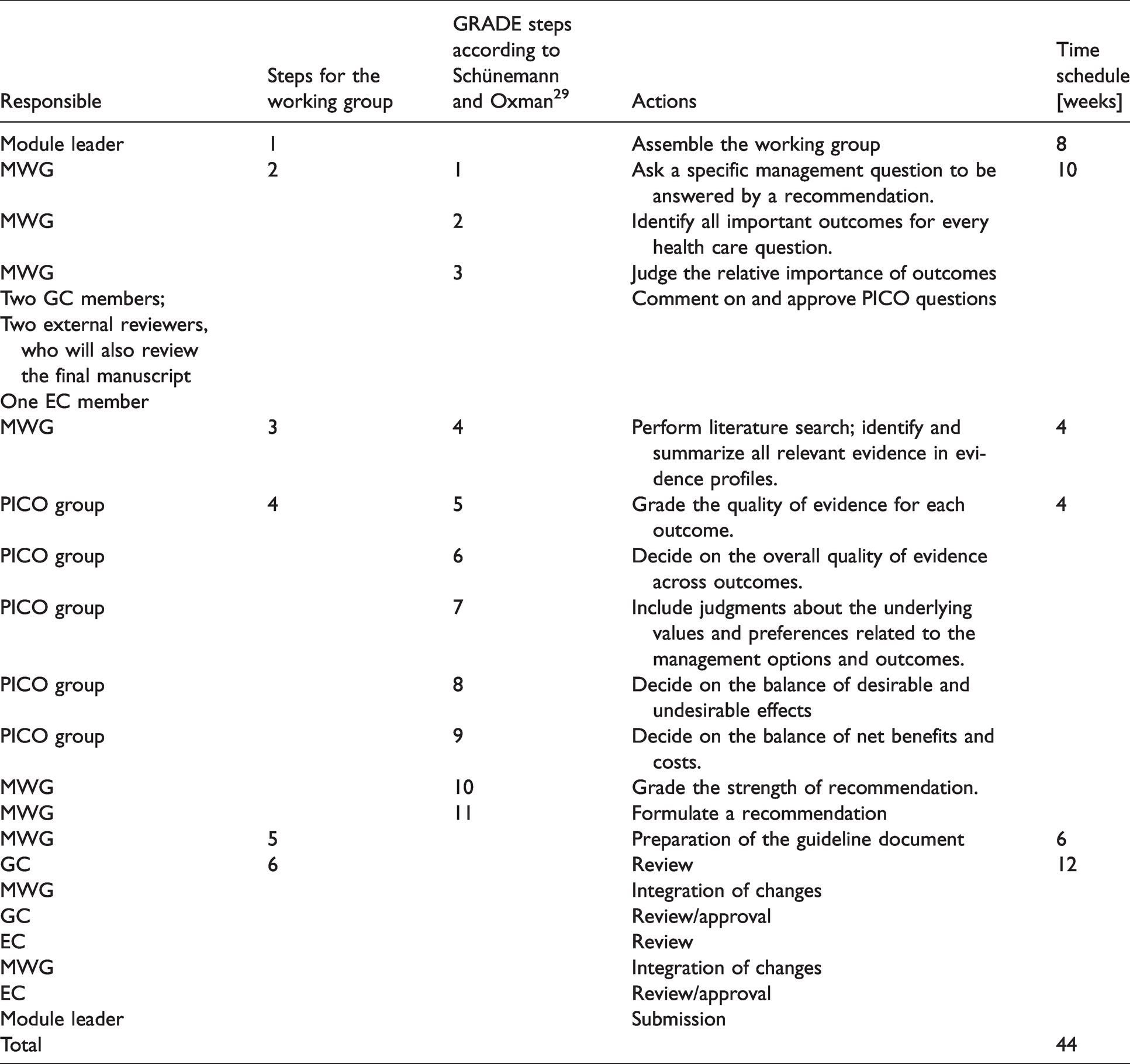
The web-based MAGICapp solution (http://magicproject.org/magicapp/) will be used during the guideline process development. MAGICapp has been specifically designed to facilitate the preparation, dissemination and update of clinical guidelines, with a special focus on the GRADE methodology. The whole guideline manuscript can be directly prepared in MAGICapp (formulation of PICO questions, preparation of descriptive tables, evaluation of the quality of evidence, formulation of recommendations and supporting text, and inclusion of references). Instructions for using MAGICapp will be presented during the ESO Guideline Development Workshop.
Introducing expert consensus statements
The overall goal of ESO guidelines is to provide practical and evidence-based recommendations based on a systematic literature search and analysis of available evidence. Randomized controlled trials (RCT) and meta-analyses of RCTs are the primary targets of systematic literature search, assessment of studies (up- and downgrading of quality of evidence), and evidence-based recommendations. If RCTs and meta-analyses are not available, other available data should be included and graded. Minimal criteria for observational studies to be included in quantitative meta-analysis are: presence of a control group, reasonable number of events and patients to address the question of interest, and no evidence of major bias. The ROBINS-I tool may be used for this purpose (https://methods.cochrane.org/methods-cochrane/robins-i-tool).
Working groups will be confronted with a situation where there are neither RCTs, meta-analyses of RCTs nor observational studies that fulfil the above-mentioned criteria or situations were RCTs are inconclusive. In these situations, we allow for an “expert consensus statement”. An expert consensus statement is a practical advice on a management or procedural point. The expert consensus process starts with an open discussion on a specific question within the MWG. Thereafter the Delphi method and secret ballot voting are used to agree on a final proposal and to avoid the bandwagon effect. The Delphi-method, is a widely used and accepted method to achieve convergence of opinion by using a series of questionnaires to collect data by participants and allowing for reassessment of initial judgments by all experts of the working group. At final stage MWG members will be requested to vote for or against the final proposal of statement. Only statements which will reach the majority of vote for will be reported in the guideline. An expert consensus statement should clearly state the direction as “suggest for” or “suggest against”.
Each expert consensus statement will be preceded by a short paragraph - called “additional information” - that summarizes the literature/scientific rational for the statement.
Review process
The review process of an ESO guideline will include two steps: first, the review of PICO questions (at the very beginning of the guideline development process), and second the review of the final manuscript Figure 1. Both reviews will be done by internal (2 members of the GB and 1 member of the EC) and 2 external reviewers, the latter according to a list of external reviewers that was agreed upon between the ESO and the editor of the ESJ. Quality criteria for reviewers are: experience with the GRADE methodology, declaration of CoIs to the ESJ editor, and availability to provide their comments within the given time frame (Table 7). Internal and external reviewers will be mentioned in the acknowledgement of the final publication.
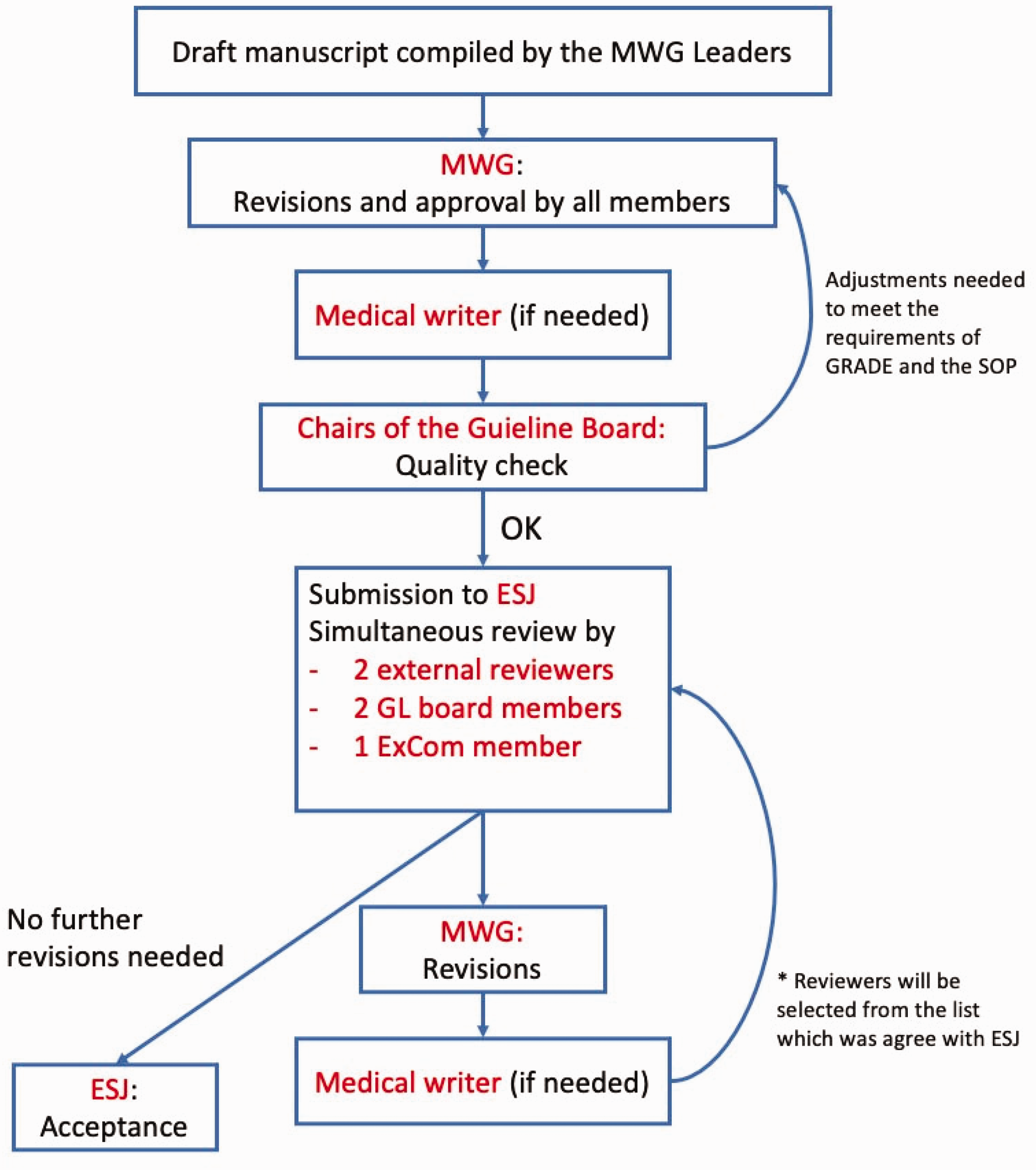
Second step of the reviewing process of an ESO guideline document.
Authorship and publication policy
The title of an ESO Guideline Document should have the following format: “
It is suggested that the MWG leader is first author and the other MWG members are listed in alphabetical order. In case of two leaders/co-chairs, it is suggested that they take the position of the first and last authors. These may change according to the opinions of the MWG if there is full consensus. The list of authors will be approved by the GB and the EC.
An ESO Guideline Document is submitted to the ESJ. Parallel publication in other journal(s) (in addition to the ESJ) is possible to allow for wider dissemination, notably in the case of multi-society guidelines, but will need to be agreed upon by contract(s). An open-access policy is sought for the ESO guideline documents, in order to make them widely available and increase their dissemination. In the same context, the National Stroke Scientific Organisations which are organizational members of the ESO should be asked to circulate guideline documents to their members and post them on the corresponding websites. Recommendations of each ESO guideline will also be made available through MAGICapp and on the ESO website.
Update of an existing ESO guideline document
Each ESO guideline should be revised approximately every three to four years, or earlier if new evidence is published that challenges current guidelines. MWG members are expected to start working on a revision approximately 24 to 36 months after the publication of the previous guideline document or publish a statement that there have been no major changes in the supporting evidence. In the meantime, expedited recommendations may be produced to provide updates on focused topics (see above).
Cooperation and publication process
ESO welcomes the development of guidelines in cooperation with other societies. Prerequisite of a collaboration is that the methodology is based on the GRADE approach and handling of CoIs follow a similar approach as stated in this SOP. The selection of ESO members for a specific working group will be done according to this SOP as stated above. The MWG leader will be appointed by ESO as stated above, unless agreed otherwise between the societies. A co-leader to the module working group may be appointed by the cooperating society. The selection of members for working groups by the cooperating society will follow the rules of that society. The maximum number of working group members shall range from up to 15 to 20 members. The distribution of members will be part of negotiation between the societies. The title of a guideline will mention the names of the cooperating societies in the following order: “ESO - cooperating society (societies) …”, unless agreed upon otherwise between the societies.
Translation of an ESO guideline document into other languages
The ESO welcomes interest for translating ESO guideline documents or at least the executive summary into other languages. Any individual or organisation interested in performing such a translation should first contact the GB. One or two ESO members (whose native language is the requested one) should be assigned by the GB to review and approve the translation before the final approval by the GB and the EC.
Dealing with falsified data after publication of an ESO guideline
Data included in analyses may turn out to be falsified after publication of guidelines or meta-analyses, and sometimes no specific notice addressing this problem is provided in the journal featuring the original publication. 30 We can only acknowledge potential data falsification that was clearly made public, notably through the journal featuring the original publication. Module working group members shall ensure that no notice of retraction or expression of concern has been published about studies that could be featured in guidelines. If such potentially falsified data has already been included in an ESO guideline publication, a short additional statement addressing this point should be published. In particular, a sensitivity analysis excluding such data or studies may be provided.
Amendment of this SOP
This SOP may be amended after discussion and majority vote among the members of the GB and approval by the EC; amendments will be published.
Footnotes
Acknowledgement
We would like to thank Luzia Balmer for her assistance in this article.
Declaration of conflicting interests
AHA is an employee of the Oslo University Hospital, Norway.
AHA reports fee for lecturing from Bayer, Boehringer Ingelheim, BMS, Allergan, Teva, Novartis, Roche, and Teva and research grant from Medtronic and Boehringer Ingelheim outside submitted work.
HC is an employee of Bispebjerg og Frederiksberg Hospital in Copenhagen Denmark. HC has nothing to disclose.
MD is an employee of LMU Klinikum, Institut für Schlaganfall- und Demenzforschung (ISD) in Munich, Germany. MD has nothing to disclose.
BF is an employee of Hospital Universitario La Paz in Madrid, Spain. BF has nothing to disclose.
EK is an employee of the Imperial College London
Charing Cross Hospital, UK. EK has received travel grants from Pfizer and Bayer and reports advisory board for Pfizer outside the submitted work.
PK is an employee of University of Cincinnati, USA. PK has nothing to disclose.
JMF is an employee of the Hospital de la Santa Creu i Sant Pau in Barcelona, Spain. JMF has nothing to disclose.
BN is an employee of the Lund University Hospital in Sweden. BN reports personal fees from Astra Zeneca and Bayer outside the submitted work.
TQ is an employee of the University of Glasgow, Scotland. TQ has nothing to disclose.
SS is an employee of the University of L’Aquila, Italy.
SS reports grants, personal fees and non-financial support from Allergan, Novartis, personal fees and non-financial support from Teva, Eli Lilly, personal fees from Astra Zeneca, personal fees from Abbott, Medscape, other from Pfizer, non-financial support from Bayer, Medtronic, Starmed, Bristol-Myers-Squibb, Daiichi-Sankyo outside the submitted work.
TS is an employee of the Klinikum Frankfurt Höchst, Germany and the Heidelberg University Hospital. TS reports personal fees Boehringer Ingelheim, Bayer, BMS Pfizer, Daiichy Sankyo, Portola outside the submitted work.
DT is an employee of Sapienza University of Rome, Italy. DT report personal fees from Abbott, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Medtronic, Pfizer, outside the submitted work.
GT is an employee of Sainte-Anne hospital, Université de Paris, France. GT reports personal fees from Guerbet France, outside the submitted work.
MZ is an employee of Azienda Unità Sanitaria Locale di Reggio Emilia, Italy. MZ has nothing to disclose.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical Approval
Not applicable.
Informed consent
Informed consent was not sought for this article.
Guarantor
TS.
Contributorship
TS, GT, MD drafted the manuscript, and researched literature. All authors reviewed and edited the manuscript and approved the final version of the manuscript

