Abstract
Spontaneous (non-traumatic) intracerebral haemorrhage (ICH) affects ~3.4 million people worldwide each year, causing ~2.8 million deaths. Many randomised controlled trials and high-quality observational studies have added to the evidence base for the management of people with ICH since the last European Stroke Organisation (ESO) guidelines for the management of spontaneous ICH were published in 2014, so we updated the ESO guideline. This guideline update was guided by the European Stroke Organisation (ESO) standard operating procedures for guidelines and the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) framework, in collaboration with the European Association of Neurosurgical Societies (EANS). We identified 37 Population, Intervention, Comparator, Outcome (PICO) questions and prioritised clinical outcomes. We conducted systematic literature searches, tailored to each PICO, seeking randomised controlled trials (RCT) – or observational studies when RCTs were not appropriate, or not available – that investigated interventions to improve clinical outcomes. A group of co-authors allocated to each PICO screened titles, abstracts, and full texts and extracted data from included studies. A methodologist conducted study-level meta-analyses and created summaries of findings tables. The same group of co-authors graded the quality of evidence, and drafted recommendations that were reviewed, revised and approved by the entire group. When there was insufficient evidence to make a recommendation, each group of co-authors drafted an expert consensus statement, which was reviewed, revised and voted on by the entire group. The systematic literature search revealed 115,647 articles. We included 208 studies. We found
Plain language summary
Background
Every year, around 3.4 million people have a type of stroke caused by bleeding in the brain that is not due to injury or another medical condition. The main causes of this kind of stroke include getting older, health issues like high blood pressure, and being exposed to air pollution. However, doctors and researchers are learning more and more about how to treat and prevent this condition, helping patients recover better and reducing the chances of it happening again. This guideline is an update of the last European Stroke Organisation guideline for people with bleeding in the brain, published in 2014.
How We Created This Guide
To make sure this guide is based on the best available evidence, we followed a structured process recommended by the European Stroke Organisation (ESO) and the European Association of Neurosurgical Societies (EANS). We focused on finding the highest quality evidence about what care works best for patients with bleeding in the brain, and made recommendations guided by a framework called GRADE.
We started with 37 important questions about care for people with stroke due to bleeding in the brain. To answer these, we looked at thousands of research papers and focused on the best available studies, especially ones where a treatment was compared reliably with an alternative. If there was not enough strong evidence to form a recommendation for clinical practice, we used expert opinions to create a consensus about a statement to guide clinical practice.
What We Found
After looking at 115,647 studies, our findings for people with bleeding in the brain were:
• What works best: We found strong evidence that patients get better when treated in specialized stroke units, and when their blood pressure is reduced to prevent more strokes.
• What might help: There is weaker evidence supporting certain treatments, such as using scores to predict the cause of bleeding, early lowering of blood pressure, early use of some drugs to promote blood clotting, surgery to remove the bleeding (including approaches that use only a small hole in the skull), surgery to decompress the skull, drainage of blood in the fluid around the brain with a clot-busting drug, prevention of clots in veins by compression devices, and restarting blood-thinning medications for those who need them. There is also weaker evidence for patients getting better when a care bundle is used. These types of care require further study.
• What should be avoided: We found strong evidence that anti-inflammatory drugs should not be used unless it’s part of a research study.
• What might not help: We found weaker evidence against routine policies to limit treatment, controlling body temperature, controlling blood sugar, and routine treatment to prevent seizures, as well as evidence against giving a platelet transfusion (a type of blood product).
• Uncertain areas: We did not find enough reliable evidence about tests to look for causes of bleeding, scores to predict outcome, early use of several drugs to promote blood clotting, surgery with drainage of fluid with a clot-busting drug, drainage of blood in the fluid around the brain, brain pressure monitoring, blood thinning drugs to prevent clots in veins, routine use of medicine to prevent seizures, blood thinning drugs and devices to prevent strokes and heart attacks for people with an irregular heartbeat, and statins to prevent strokes and heart attacks. In these cases, we provide expert opinions to help guide medical decisions and encourage more reliable research to be done.
Why This Matters
This guideline summarises the best available evidence and expert opinions, to inform the care of people with stroke due to bleeding in the brain. This guideline may help doctors and other healthcare professionals to improve care for people with bleeding in the brain. Although a lot of progress has been made since the last edition of this guideline, more large, reliable, definitive clinical trials are needed to identify ways of improving outcome after bleeding in the brain.

Table of Contents
Composition and approval of the Module Working Group---------------------------------------------------------------------------------------------------------------------5
Development and approval of clinical questions---------------------------------------------------------------------------------------------------------------------6
Evaluation of the quality of evidence and formulation of Evidence-based Recommendations and Consensus Statements---------------------------------------------------------------------------------------------------------------------7
Guideline structure---------------------------------------------------------------------------------------------------------------------7
Management on specialised units---------------------------------------------------------------------------------------------------------------------7
Radiological investigation for underlying cause---------------------------------------------------------------------------------------------------------------------8
Imaging Scores of acute ICH---------------------------------------------------------------------------------------------------------------------8
Type of imaging for acute ICH---------------------------------------------------------------------------------------------------------------------9
Outcome prediction---------------------------------------------------------------------------------------------------------------------9
Outcome scores---------------------------------------------------------------------------------------------------------------------9
Withdrawal of treatment---------------------------------------------------------------------------------------------------------------------10
Acute blood pressure alteration---------------------------------------------------------------------------------------------------------------------11
Spontaneous ICH not associated with antithrombotic drug use---------------------------------------------------------------------------------------------------------------------16
rFVIIa---------------------------------------------------------------------------------------------------------------------16
Tranexamic acid for spontaneous ICH---------------------------------------------------------------------------------------------------------------------18
Spontaneous ICH associated with antiplatelet drug use---------------------------------------------------------------------------------------------------------------------20
Platelet transfusion---------------------------------------------------------------------------------------------------------------------20
Desmopressin---------------------------------------------------------------------------------------------------------------------21
Anticoagulant-associated ICH---------------------------------------------------------------------------------------------------------------------22
ICH associated with use of vitamin K-antagonists---------------------------------------------------------------------------------------------------------------------22
Prothrombin complex concentrate (PCC) versus fresh frozen plasma (FFP)---------------------------------------------------------------------------------------------------------------------22
ICH associated with use factor Xa inhibitors (FXaI)---------------------------------------------------------------------------------------------------------------------23
Prothrombin complex concentrates---------------------------------------------------------------------------------------------------------------------23
Andexanet alfa---------------------------------------------------------------------------------------------------------------------24
Tranexamic acid---------------------------------------------------------------------------------------------------------------------26
ICH associated with use factor II inhibitors---------------------------------------------------------------------------------------------------------------------26
Idarucizumab---------------------------------------------------------------------------------------------------------------------26
Supratentorial ICH---------------------------------------------------------------------------------------------------------------------27
Any surgery aimed at haematoma removal---------------------------------------------------------------------------------------------------------------------27
Haematoma removal by means of craniotomy and open standard surgical technique---------------------------------------------------------------------------------------------------------------------28
Minimally invasive surgical removal (MIS)---------------------------------------------------------------------------------------------------------------------29
Surgery with catheter placement plus thrombolysis---------------------------------------------------------------------------------------------------------------------31
Decompressive craniectomy---------------------------------------------------------------------------------------------------------------------32
ICH with intraventricular extension---------------------------------------------------------------------------------------------------------------------33
External ventricular drainage with or without combined lumbar drainage---------------------------------------------------------------------------------------------------------------------33
External ventricular drainage with intraventricular thrombolysis---------------------------------------------------------------------------------------------------------------------34
Surgical removal of the intraventricular blood---------------------------------------------------------------------------------------------------------------------35
Infratentorial ICH---------------------------------------------------------------------------------------------------------------------37
Single measures for prevention and management of complications---------------------------------------------------------------------------------------------------------------------38
Physical measures to prevent venous thromboembolism ---------------------------------------------------------------------------------------------------------------------38
Short-term antithrombotic therapy to prevent DVT/PE---------------------------------------------------------------------------------------------------------------------40
Temperature management--------------------------------------------------------------------------------------------------------------------- 42
Blood glucose control---------------------------------------------------------------------------------------------------------------------43
Intracranial pressure (ICP) monitoring---------------------------------------------------------------------------------------------------------------------43
Anti-inflammatory treatment---------------------------------------------------------------------------------------------------------------------44
Anti-seizure medications---------------------------------------------------------------------------------------------------------------------47
Care bundles---------------------------------------------------------------------------------------------------------------------49
Blood pressure---------------------------------------------------------------------------------------------------------------------50
Oral anticoagulation (OAC)---------------------------------------------------------------------------------------------------------------------52
Left atrial appendage occlusion (LAAO) for atrial fibrillation---------------------------------------------------------------------------------------------------------------------54
Antiplatelet therapy---------------------------------------------------------------------------------------------------------------------55
Statins---------------------------------------------------------------------------------------------------------------------56
Introduction
Spontaneous intracerebral haemorrhage (ICH) is defined as non-traumatic intraparenchymal haemorrhage (which may expand into the cerebral ventricles) that is assumed to be due to underlying cerebral small vessel disease in the absence of a detected underlying macrovascular, neoplastic, infectious/inflammatory or haemodynamic cause. 1 The five leading risk factors for disability-adjusted life years (DALYs) lost due to ICH are high systolic blood pressure, high body-mass index, ambient particulate matter pollution, high fasting plasma glucose and smoking, although the contributions of these risk factors vary between global regions. 2
In 2019, the global incidence of ICH was estimated at 3.41 million cases (95% CI 2.97–3.91), accounting for 28.8% of all strokes (Figure 1).2,3 The global prevalence was 20.6 million (18.0–23.3). Spontaneous ICH resulted in 2.89 million deaths (2.64–3.10) and was associated with 68.57 million (63.27–73.68) disability-adjusted life-years (DALY).2,3 Despite the decline in age-standardised rates for ICH incidence, from 1990 to 2020, the absolute number of new cases is increasing due to ageing populations globally. This trend is projected to persist in the European Union (EU) between 2019 and 2050, including an increase in related deaths, due to population growth, ageing and increased exposure to several important risk factors. 4 Of the 16 identified risk factors for ICH, the 5 most important are elevated systolic blood pressure, high body-mass index, air pollution, diabetes mellitus and smoking. However, the relative importance of these factors varies across different countries and populations. 3
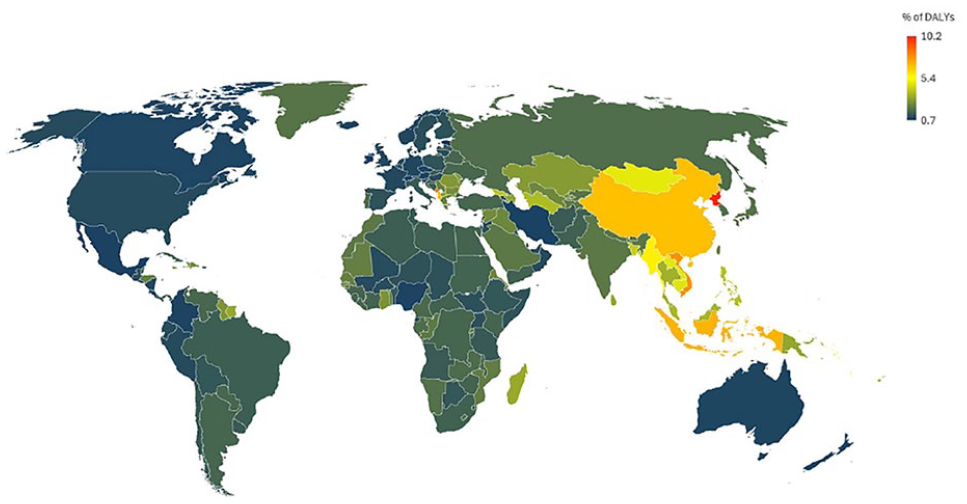
Percent of total DALYs lost to intracerebral haemorrhage by country in 2021. 3
Since the last European Stroke Organisation (ESO) guidelines for the management of spontaneous ICH were published in 2014, many randomised controlled trials (RCTs) and high-quality observational studies have added to the evidence base for the management of people with ICH. 5 This update focused on adding the most reliable subsequent evidence from randomised controlled trials, or systematic reviews of observational studies if RCTs were not available. This update was done in collaboration with the European Association of Neurosurgical Societies (EANS).
Methods
This guideline was developed in accordance with ESO standard operating procedures (SOP), which are based on the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) framework.6,7
Composition and approval of the Module Working Group
This guideline was initiated by the ESO. Two chairpersons (TS, RASS) were selected to assemble and coordinate the guideline’s Module Working Group (MWG). The final group contained 23 experts within the following areas: neurology/neurointensive care (18), neurosurgery (2) and methodology (1). The EANS later joined the ESO MWG in a collaboration between EANS and ESO. The ESO Guideline Board and Executive Committee and the EANS council reviewed the intellectual and financial disclosures of all MWG members and approved the group’s composition. The full details of all MWG members and their disclosures are included in the Supplemental materials.
Development and approval of clinical questions
The MWG developed a list of topics and corresponding questions of highest clinical relevance. Questions were formatted using the PICO (Population, Intervention, Comparator and Outcome) approach. These were reviewed by two external reviewers, as well as members of the ESO Guideline board and Executive Committee. A range of clinical outcomes were rated for each PICO by MWG members as critical, important or of limited importance according to GRADE criteria. Final decisions on outcomes were made using a Delphi consensus approach. The outcome ratings for each PICO question can be found in the Supplement.
Literature search
The main priorities for this guideline update were the reliability and relevance of recent evidence. Consequently, the literature search prioritised RCTs, which we pooled in meta-analyses. If RCTs were unavailable, we reverted to meta-analyses of observational studies, or individual observational studies if meta-analyses were not available. The minimal criteria for including observational studies in quantitative meta-analyses were as follows: the presence of a control group, a reasonable number of events and patients to address the question of interest, and the absence of evidence of major bias (other than those attributable to non-randomised evaluations of the effects of treatments).
For each PICO question, groups of members of the MWG and the guideline methodologist developed search strategies for the following databases: Medline (via Ovid), Embase (via Ovid) and Cochrane Library. Where a search strategy from an existing systematic review was available, this was used or adapted. Where there was a recent relevant systematic review on the question of interest, the corresponding search strategy and results were used and updated as necessary. The search strategies are shown in the Supplement.
The ESO Guideline methodologist (LH) searched databases from January 2013 to May 2024, and PICO 6.2 (oral anticoagulation for atrial fibrillation after ICH) was updated in line with a recent RCT that was included in the guideline and had searched the literature up to January 2025. 8 Additionally, reference lists of review articles, the authors’ personal reference libraries and previous guidelines were searched for further relevant records.
The search results were loaded into the web-based Covidence platform (Health Innovation, Melbourne, Australia) for assessment by the MWG. Two or more MWG members were assigned to screen the titles and abstracts of publications registered in Covidence independently, followed by full text assessment of potentially relevant studies. Disagreements were resolved by discussion between the two reviewers or by a third MWG member.
Data extraction
Data extraction and analysis was performed by the ESO methodologist (LH) and checked by the corresponding MWG members for each PICO. In the case that relevant data were not reported in an eligible study, the corresponding author was contacted. In case of no response, the co-authors of the study were also contacted. If no answer was received, data were considered as missing.
Data analysis
Before conducting our meta-analyses, we carefully considered both fixed-effect and random-effects models. Given the heterogeneity in study design, populations, interventions, and comparators, we selected a random-effects model to estimate the average intervention effect (RevMan; https://revman.cochrane.org/). This approach aligns with the Cochrane Handbook and ESO guidelines, which recommend random-effects models when variability across studies is expected. While fixed-effect models assume identical studies and estimate a common effect size, this was not appropriate due to differences among independently conducted trials. 9 We acknowledge that random-effects models give relatively more weight to smaller studies compared to fixed-effect models. 9
Results were presented as odds ratios with associated 95% confidence intervals (95% CIs). Statistical heterogeneity across studies was assessed using the I2 statistic, and classified as moderate (⩾30%–49%), substantial (⩾50%–74%) or considerable (⩾75%). 3 Where appropriate, subgroup analyses were performed. The risk of bias as indicated in each forest plot was classified according to the following categories: (A) Random sequence generation (selection bias), (B) Allocation concealment (selection bias), (C) Blinding of participants and personnel (performance bias), (D) Blinding of outcome assessment (detection bias), (E) Incomplete outcome data (attrition bias), (F) Selective reporting (reporting bias), (G) Other bias. The risk of bias domains as per the ROBINS-I tool (for non-randomised studies) were as follows: (A) Bias due to confounding, (B) Bias in selection of participants into the study, (C) Bias in classification of interventions, (D) Bias due to deviations from intended interventions, (E) Bias due to missing data, (F) Bias in measurement of outcomes, (G) Bias in selection of the reported result. In the risk of bias assessment, green label indicates ‘low risk of bias’, yellow label indicates ‘unclear risk of bias’ (or ‘moderate risk of bias’, in case of ROBINS-I assessment) and red label indicates ‘serious risk of bias’ (or, when specified, ‘very serious risk of bias’, in case of ROBINS-I).
Evaluation of the quality of evidence and formulation of evidence-based recommendations and consensus statements
The risk of bias of each included RCT or observational study was assessed with the Cochrane Rob2 tool and the ROBINS-I tool (Risk Of Bias In Non-randomised Studies of Interventions), respectively. 10 As recommended, the evidence synthesis did not use a quality ‘score’ threshold but classified overall risk of bias at study level and then in aggregate.
The results of the data analyses were imported into the GRADEpro Guideline Development Tool (McMaster University, 2015; developed by Evidence Prime, Inc.). For each PICO question, and each outcome, the following were considered: risk of bias based on the type of available evidence (RCT or observational studies); consideration of inconsistency of results; indirectness of evidence, imprecision of results and other possible bias. LH generated the GRADE evidence profiles/summary of findings tables, which MWG members used to prepare ‘Evidence-based Recommendations’, based on the GRADE approach. The direction, strength and formulation of the recommendations (Table 1) were determined according to the GRADE evidence profiles and the ESO guideline SOP. 11

Finally, Expert Consensus Statements were added whenever the PICO group considered that there was insufficient evidence available to provide Evidence-based Recommendations and where practical guidance for routine clinical practice was regarded necessary. The Expert Consensus Statements were based on the opinions of the MWG members allocated to each PICO, with revision in discussion with TS and RASS where necessary, and voting by all expert MWG members to quantify the extent of consensus.
Guideline structure
For practicality, the guideline is structured so that each PICO section begins with the Evidence-based Recommendation(s) and, where necessary, Expert Consensus Statement(s). A further paragraph describes ‘current evidence’, summarising the evidence from RCTs that supports the recommendation with respect to the prespecified outcomes of that PICO. Detailed descriptions of single studies or meta-analyses are provided in the Supplement. If relevant information beyond the prespecified outcomes is deemed important for forming the recommendation, an ‘additional information’ paragraph is included after the current evidence section.
Results
General management
Management on specialised units

Analysis of current evidence
A single systematic review and meta-analysis was identified. 12 The study was based on 8 randomised controlled trials (RCTs) including a total of 2657 patients. The RCTs recruited patients with ICH as well as patients with ischaemic stroke, the intervention was stroke unit care, and the comparator was general ward care. Death, or death/dependence were defined as critical outcomes. Stroke unit care reduced death or dependence overall (relative risk (RR) 0.81; 95% confidence interval (CI) 0.47–0.92; p = 0.0009) with no difference in benefits for patients with ICH (RR, 0.79; 95% CI 0.61–1.00) than for patients with ischaemic stroke (RR 0.82; 95% CI 0.70–0.97). Stroke unit care reduced death overall (RR 0.79; 95% CI 0.64–0.97; p = 0.02) and for patients with ICH (RR 0.73; 95% CI 0.54–0.97). No RCTs compared the effects of ICH management in an intensive care unit (ICU), neuro-intensive care unit (NICU) or high dependence unit (HDU) with those in a normal ward or a stroke unit.
Additional information
In these RCTs, stroke unit care involved organised inpatient care in a hospital stroke unit provided by a multidisciplinary team with expertise in stroke management, a specialist in stroke and regular multidisciplinary team meetings. 13 In the RCTs underlying the meta-analysis, care included managing vital signs, preventing complications and initiating rehabilitation efforts.
In a large observational study, consecutive non-ventilated and non-comatose patients with ICH in a German statewide stroke registry, outcomes were compared according to admission ward. After adjusting for stroke severity, age, sex, pre stroke modified Rankin scale (mRS) score, comorbidities, referring medical unit, hospital level, complications during treatment, time to admission and length of hospital stay, treatment of people with ICH in an ICU and normal ward (NW) compared with admission to a stroke unit (SU) was associated with a higher risk of death in hospital (ICU vs SU: Odds ratio (OR) 2.11; 95% CI 1.75–2.55; NW vs SU: OR 1.52; 95% CI 1.23–1.89) and higher odds of an unfavourable functional outcome (ICU vs SU: OR 1.27; 95% CI 1.09–1.46; NW vs SU: OR 1.28; 95% CI 1.08–1.52). A subgroup analysis of severely affected ICH patients (NIHSS of 10–25) showed a lower risk of poor outcome when treatment took place in dedicated neurological ICUs (NICU) compared with stroke units. 14
Radiological investigation for underlying cause
Imaging scores of acute ICH

Analysis of current evidence
The performance (calibration, discrimination and clinical utility) of predicting the underlying cause of ICH was defined as a critical outcome. Our literature search did not identify any RCTs or comparative diagnostic test accuracy studies comparing algorithms with standard care for targeted investigation and detection of ICH cause. Various scores have been developed but their impact on patient’s management remains uncertain.
Additional information
To target the use of intra-arterial digital subtraction angiography (IADSA) at people with ICH according to their probability of harbouring a macrovascular cause (e.g. arteriovenous malformation, intracranial aneurysm, dural arteriovenous fistula, cavernous malformation or cerebral venous thrombosis), 15 the diagnostic angiography to find vascular malformations (DIAGRAM) prediction score may be used to predict the probability of a macrovascular cause in people with ICH based on age (18–50 years vs 51–70 years), ICH location (lobar, posterior fossa, deep), small vessel disease signs on plain computed tomography (CT, yes vs no) and CT angiography (CTA, abnormal vs normal).15,16 DIAGRAM showed good performance in a derivation cohort (c-statistic 0.83, 95% CI 0.78–0.88) and moderate performance in an external validation cohort (c-statistic 0.66, 95% CI 0.58–0.74), but performance improved when CTA results were added (c-statistic 0.91 (95% CI 0.88–0.94), and 0.88 (95% CI 0.83–0.94) respectively). Other scores such as the simple ICH score and secondary ICH score have been developed to predict the probability of a macrovascular cause of ICH.17–19 After performing a non-contrast CT, the guideline authors suggest that younger people with lobar (or cerebellar) ICH, and absence of a history of hypertension should be further investigated with CTA, magnetic resonance imaging (MRI) or MR-angiography (MRA), or IADSA. Rather than using history of hypertension to guide investigation (since it is a highly prevalent risk factor), searching for markers of small vessel disease on brain imaging (CT or MRI) seems more appropriate. 20 However, these prediction scores have been developed in selected populations, and generalisability of their diagnostic performance needs to be investigated.
Type of imaging for acute ICH


Analysis of current evidence
Our literature search and a Cochrane review did not identify any RCTs or appropriate diagnostic studies comparing different imaging modalities with a reference standard of IADSA. 21 The reference standard to disclose underlying intracranial vascular malformations remains IADSA as sensitivity and specificity is higher compared with other modalities. As IADSA is more invasive than MRI or CT, the clinical dilemma lies in availability and in determining for which patients the potential diagnostic gain outweighs the potential harms.22,23 No studies have systematically examined all ICH patients using both MRI and CTA compared with the reference standard of IADSA.
Additional information
ICH is a heterogeneous disease, and clinicians should investigate the underlying cause of the bleeding, 24 as this will guide acute management as well as secondary prevention. For example, some intracranial vascular malformations are associated with a high risk of rebleeding and will require specific management. Data are mostly available in convenience samples of people aged <70 years of age without hypertension, who were selected for investigation in everyday practice. Compared with IADSA, CTA and MRA have a sensitivity and specificity of >90% to detect intracranial vascular malformations in these highly selected populations. 21 However, in the DIAGRAM prospective study of 298 adults aged <70 years, CTA/CTV performed within the first 7 days had a sensitivity of 74% and a specificity of 91%, suggesting that when CTA is normal, more investigations should be performed including IADSA, which therefore remains the reference standard. 15 MRI can also provide information about underlying cavernous malformation or cerebral small vessel diseases. 24
Clinical complications from IADSA, most frequently significant puncture site haematomas and transient neurological events, are reported in approximately 1% of patients at experienced centres with a slightly higher probability in acute ICH. In the DIAGRAM study, the rate of complications of IADSA was 0.6%. 15 These procedural risks must be weighed against the higher diagnostic value of IADSA on a case-by-case basis. 25
Outcome prediction
Outcome scores


Analysis of current evidence
Our updated systematic search was based on the systematic review and meta-analysis by Gregório et al. 26 up to September 2016. The study identified 72 prognostic tools across 59 studies, with corresponding discrimination and/or calibration values available for death and/or functional outcomes. The update of this search identified an additional 28 papers evaluating 65 tools (Supplemental Tables 1 and 2, PICO 1). We defined a prediction tool as including at least three variables, and as being used within 48 h of admission to hospital. Only long-term outcomes were assessed: death and functional outcome occurring ⩾30 days. Prediction tools developed using machine learning methods and artificial intelligence software were not included in the present focused update due to a lack of clear best practice guidance.
Of note: Our analysis examines the prognostic value of various scales for predicting mortality and functional outcomes in patients with intracerebral haemorrhage. However, it does not constitute a direct comparison between the predictive models embedded in these scales and the clinical assessments made by treating physicians. Our analysis focuses solely on evaluating the methodological properties of the prognostic tools, particularly their discriminatory ability, validity and applicability, without analysing clinical decision-making or assessing the potential superiority of one approach over the other.
A total of 97 prediction tools were evaluated: death prediction tools (N = 30), functional outcome prediction tools (N = 20), and combined death and functional outcome prediction tools (N = 47). The large number of differing tools highlights the absence of consensus around predicting ICH outcomes. Prognostic discrimination varied between the tools, the Area Under the Receiver Operator Characteristic (AUROC) ranged from 0.64 to 0.93. Half of the tools assessed prediction tool calibration (N = 52, 54%).
We used the Prediction model risk of Bias assessment Tool (PROBAST) to evaluate the quality of evidence (Supplemental Table 3, PICO-1). 27 Sources of bias included a lack of high-quality external validations, a lack of blinding, analysis of small sample sizes and ambiguous reporting of missing data. 26 For guideline recommendations we only considered tools with at least one external validation and where the evidence was categorised as low risk of bias and high applicability to the specific PICO question. We identified 14 tools in 11 publications that met our criteria.28–38
For our quantitative analysis of the prognostic accuracy of selected tools, we assessed eligible development and validation studies. The original ICH score was described in six papers with an AUROC range of 0.82–0.92.28,31,34–37 This was followed by the max-ICH score described in three papers with an AUROC range of 0.77–0.88.34,35,38 The calibration of the ICH score was assessed using the Hosmer-Lemeshow test, yielding varying results (p = 0.37, p > 0.1, p < 0.001),31,35,37 and the max-ICH score was similarly assessed once (p = 0.89). 35 A meta-analysis was deemed unfeasible due to significant heterogeneity, driven by the limited number of studies using the same tool and the variations in assessed outcomes and their respective endpoints.
Additional information
The ICH score is determined by age, ICH volume, infratentorial origin, Glasgow Coma Score (GCS), and intraventricular haemorrhage (IVH). 28 The max-ICH score, also includes age, ICH volume and IVH, as well as NIHSS and use of oral anticoagulation. 38 Several other reviews have looked at this question with similar findings, finding that the ICH score has undergone the most extensive external validation.39–41 Witsch et al. 42 favoured the max-ICH score according to their bespoke criteria for complete score validation. These reviews also highlight the lack of, and need for, further external validation of ICH prediction tools.39–42
The properties of these prediction tools may be falsely improved due to the bias caused by withdrawing treatment from people with worse predicted outcome. The effect of this was not taken into consideration when the ICH score was initially developed, while the max-ICH score actively excluded patients with early care limitations. 38 It is important to note that the original ICH score was created in 2001 and first validated in 2002, so there are limitations to this score given its historical development.28,43 This period predates the widespread implementation of stroke unit care, and death rates were higher. 44 Thus, a prediction tool developed and validated in such cohorts may have resulted in predictive properties which do not accurately reflect the tool’s current performance.
While ICH prediction tools have been extensively developed, uncertainty remains regarding their function and impact on management in clinical practice. 45 Nonetheless, these tools are considered useful in clinical practice to inform outlook for individuals and clinicians, who must be aware of the limitations of the evidence available, and aid discussions with patients’ families.
Withdrawal of treatment


Analysis of the current evidence
No RCTs were identified from our systematic review that examined limitation of treatment, do not attempt cardiopulmonary resuscitation (DNR), and/or initiation of palliative care order within 24 h of acute spontaneous ICH. RCTs are unlikely to be acceptable to healthcare professionals or relatives, and therefore it is probable that high-quality evidence will never be available on this topic.
Additional information
All studies identified were considered of very low-quality evidence, as they were all retrospective observational studies that examined associations between early (i.e. within 24 h) limitation of care, DNR or palliative care with various outcomes for adults with ICH. All studies examined associations between initiation of such orders with outcomes (predominantly death or dependence) after adjustment for various other predictors. However, residual confounding is undoubtedly present in these studies, and a central issue surrounds the concept of ‘self-fulfilling’ prophecy, whereby initiation of withdrawal/limitation of care or DNR orders based on perceived predicted poor outcome inevitably increases the likelihood of worse outcomes.
Some important conceptual differences between DNR and limitation/withdrawal of care should be noted. DNR refers to orders that prohibit resuscitation efforts. In the present discussion, we refer to the initiation of DNR after presentation/admission to hospital, whereby patients or substitute decision-makers put DNR orders in place after ICH onset. On the other hand, limitation or withdrawal of care, or initiation of palliative care, typically occurs following admission in people deemed to be terminally ill based on factors such as the predicted outcome, age and comorbidity/multimorbidity status.
Identified observational studies have highlighted some differences in outcomes among ICH patients whereby DNR (Supplement on PICO 1.3.2: description of single studies), limitation or withdrawal of care was initiated within 24 h. For instance, DNR orders started within 24 h of admission has been associated with lower odds of receiving guideline-based treatment (such as admission to a stroke unit or DVT prophylaxis). 46 People with ICH have also been found to have palliative care initiated within 24 h of admission at greater rates than people with ischaemic stroke, after adjustment for multiple covariates. Further, some existing prognostic models for outcomes after ICH are confounded by the use of early DNR orders or limitation of care.47,48 Additionally, some retrospective observational data also suggest potentially different use of DNR, limitation or withdrawal of care, or palliative care between men and women.49,50
The non-randomised ABC-ICH care bundle study included a secondary outcome of early (<24 h) DNR orders, although a DNR policy was not part of the care-bundle itself; the care bundle was associated with a reduction in early DNR orders, and this reduction was a key mediator of the association between bundle implementation and reduced 30-day case fatality. 51 (see also PICO 5.2)
Blood pressure management
Elevated blood pressure (BP) is common in patients with ICH and is associated with increased risk of haematoma expansion (HE), death, and dependence.52–54 The primary rationale for lowering BP in acute ICH is to prevent haematoma expansion, and thereby clinical deterioration.
Acute blood pressure alteration


Analysis of current evidence
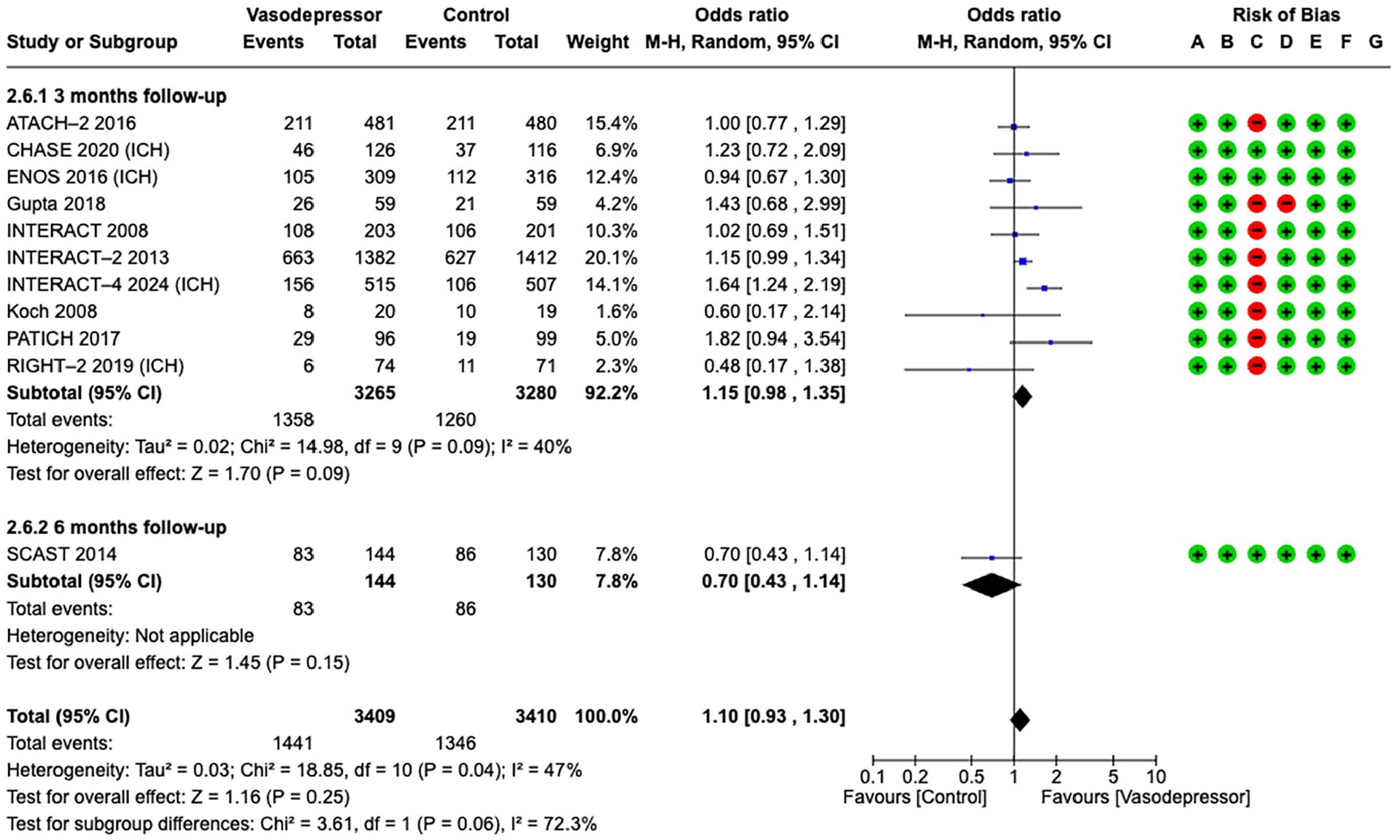
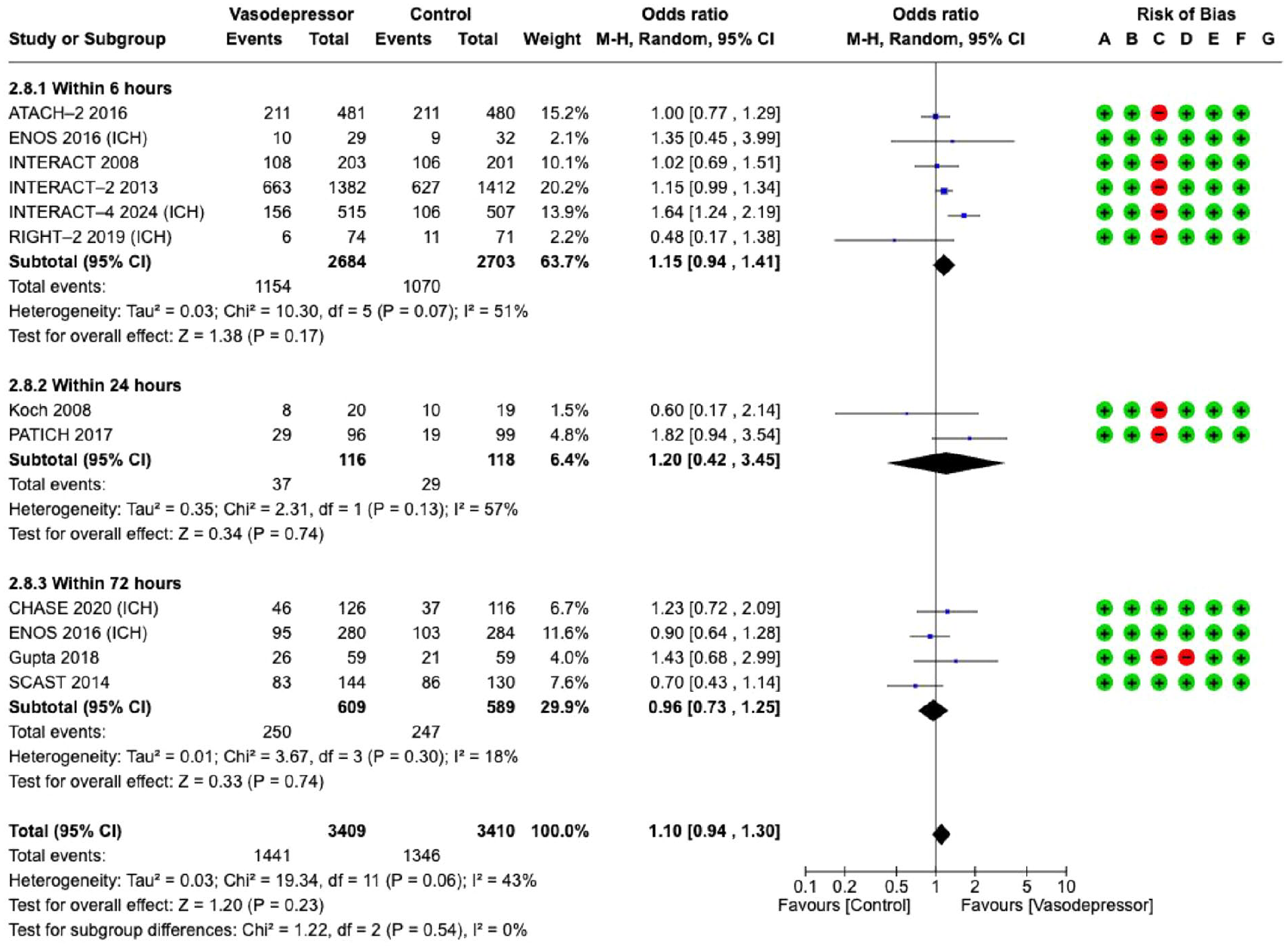
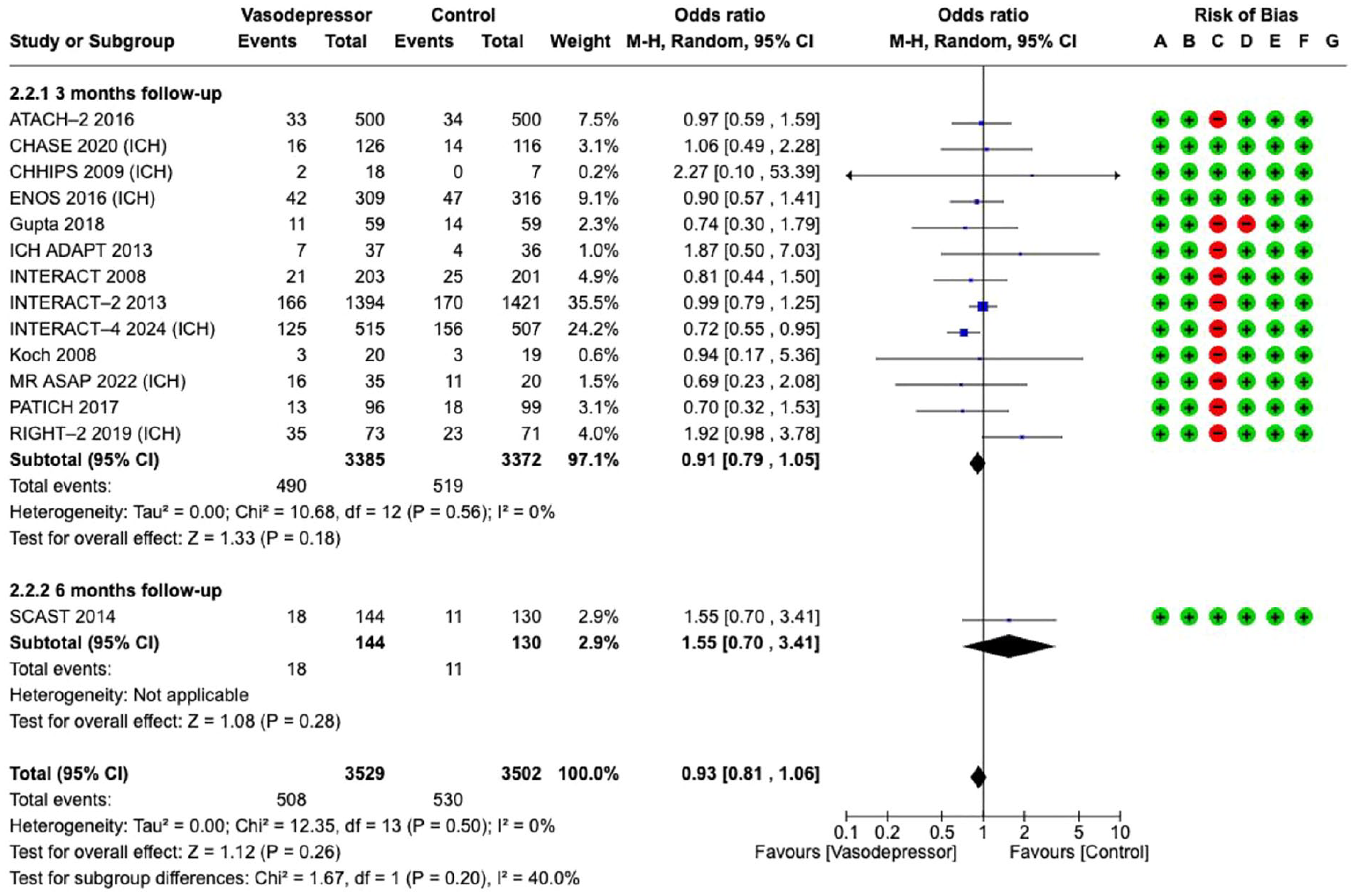
This analysis builds upon the ESO guidelines on blood pressure management in acute ischaemic stroke and ICH that included 12 RCTs. 55 For the current guideline, two additional RCTs were identified.56,57 Fourteen RCTs were included in the updated analyses for this ICH guideline, comparing blood pressure lowering (either titrated to an intensive blood pressure target or using a fixed antihypertensive drug) with control (contemporaneous guideline standard blood pressure target or placebo, respectively) in 7031 participants assessed within 2–72 h of symptom onset (Supplement: description of single studies).56–69 Good functional outcome (defined as mRS 0–2) and death at 3–6 months after ICH were prioritised as critical outcomes, and haematoma expansion within 6 and 24 h as an important outcome.
Blood pressure lowering treatment targeting lower BP compared with control did not improve
Effect on
The effect of blood pressure lowering on good functional outcome did not differ according to time from symptom onset to treatment/randomisation (6, 24 and 72 h, Figure 3).
Effect on
Blood pressure lowering treatment compared with control did not affect
Effect on
The effect of blood pressure lowering on
Effect on
Antihypertensive treatment targeting lower BP, regardless of time to treatment, compared with control showed a non-significant reduction in
Effect on
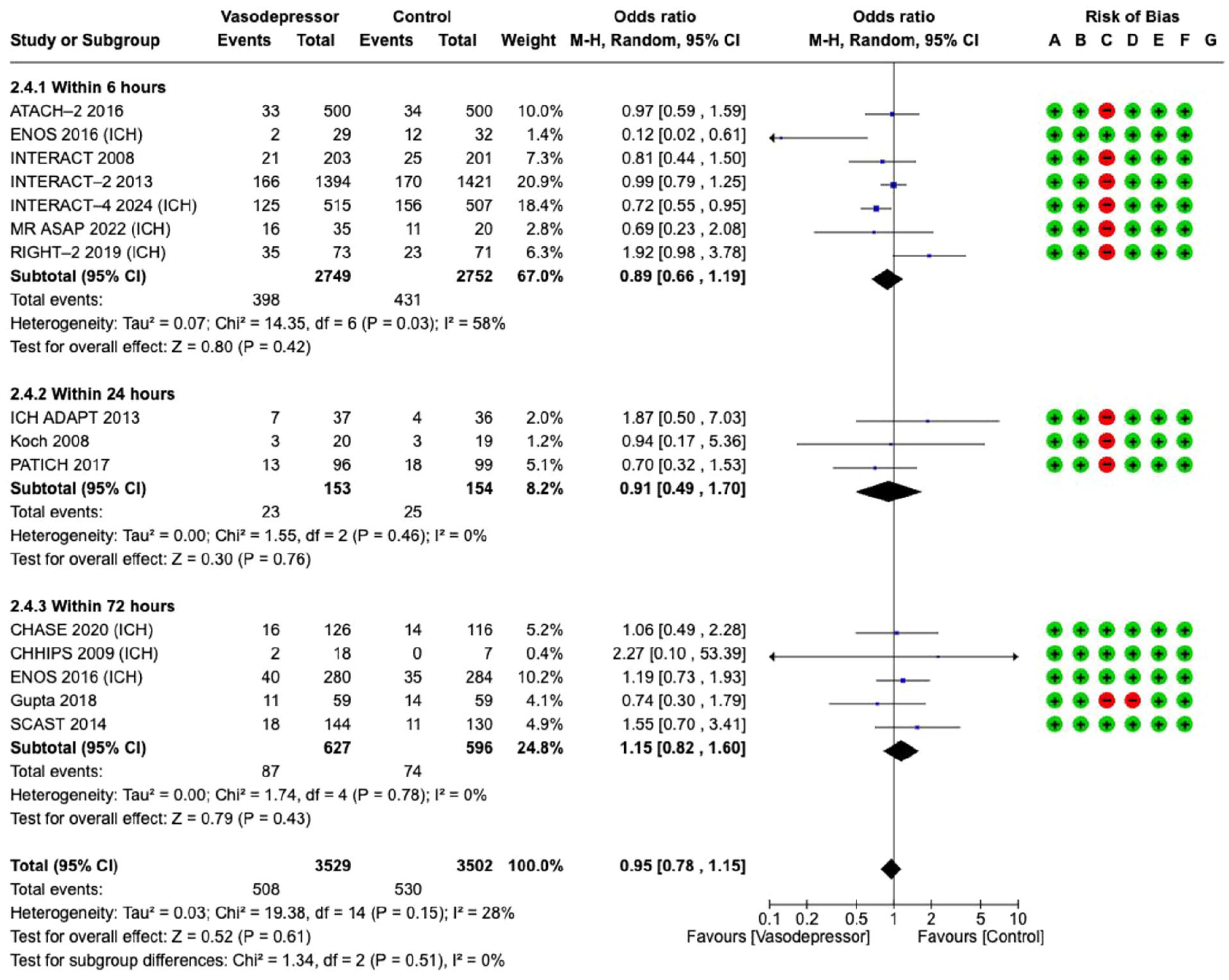
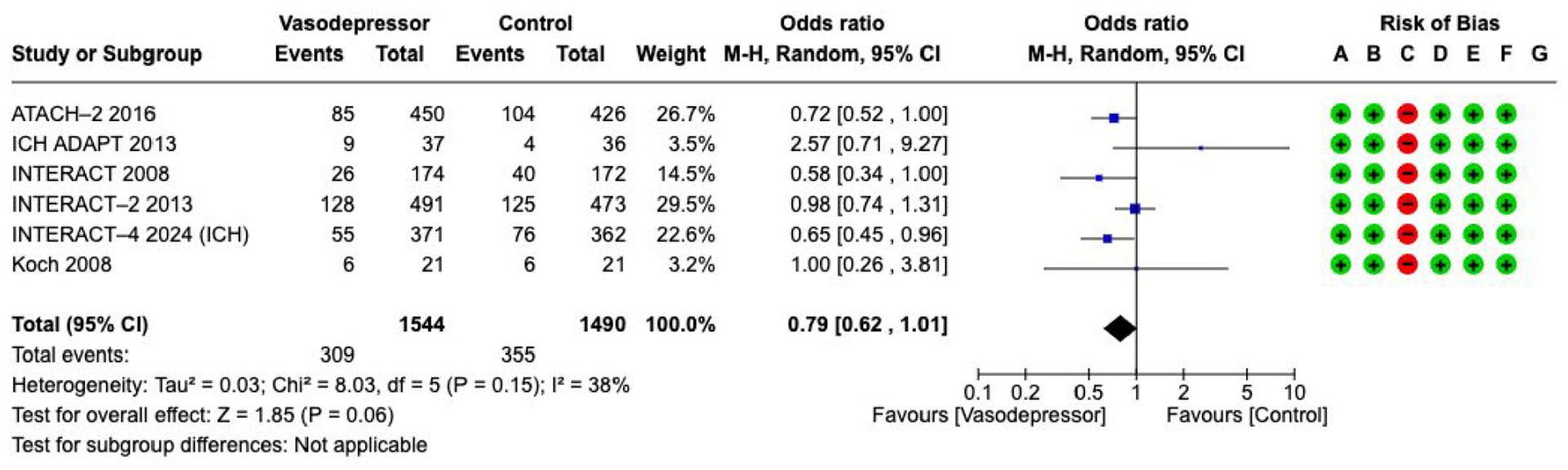
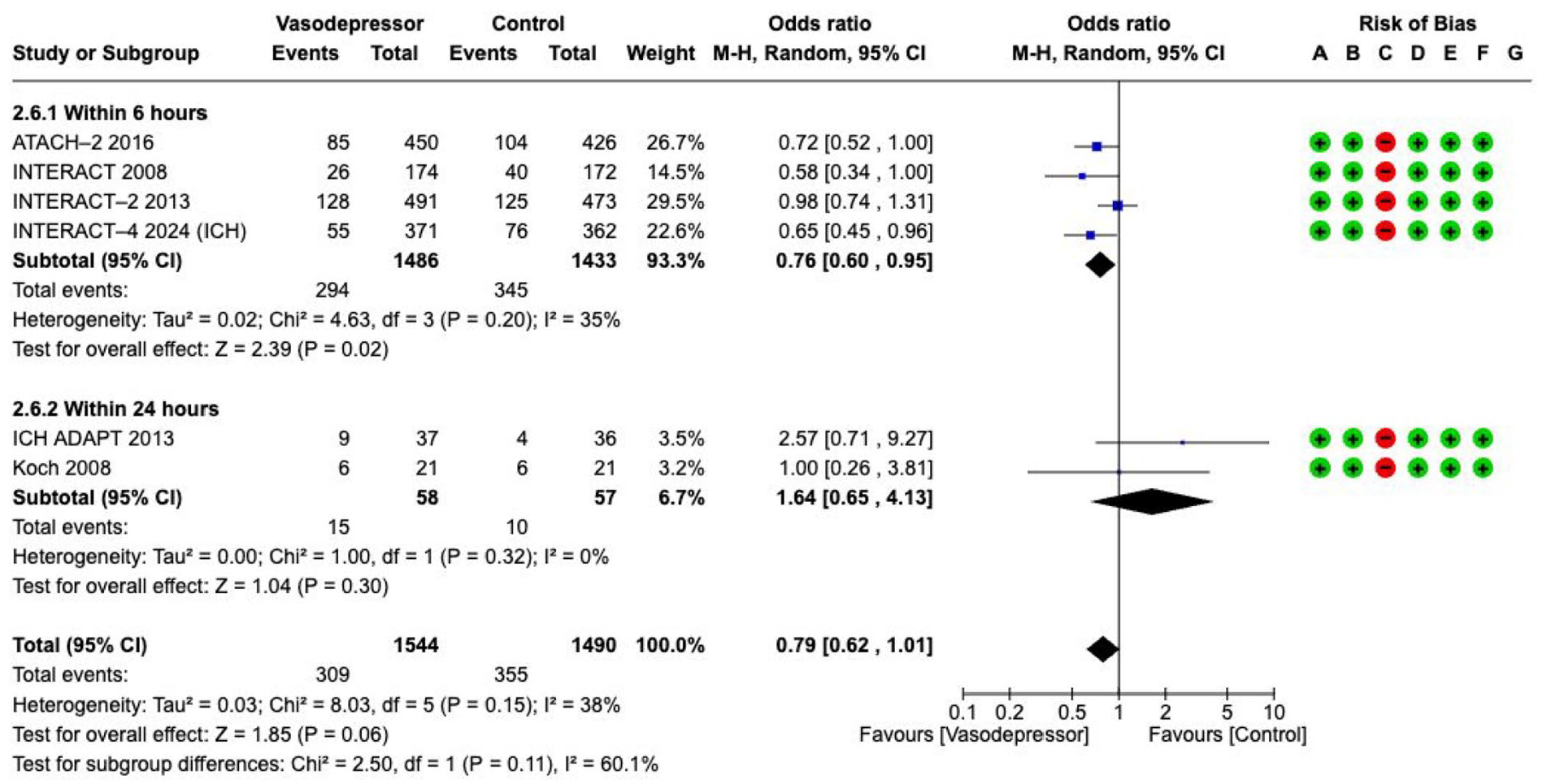
However, blood pressure lowering within 6 h of symptom onset was associated with lower odds of haematoma expansion (OR 0.76, 95% CI 0.60–0.95, 4 RCTs, 2919 participants, low certainty, with moderate heterogeneity within subgroups, Figure 7).
The effect on
This meta-analysis suggests that early BP lowering, within 6 h of symptom onset limits HE in patients with minor to moderate ICH. However, due to substantial clinical and methodological heterogeneity across included studies – such as differences in intervention protocols, patient characteristics, timing, and outcome definitions – interpretation of the pooled effect sizes must be approached with caution. Notably, standard random-effects models may inadequately account for heterogeneity, and early small studies can anchor pooled estimates, limiting the influence of larger, later trials. Although there was a tendency towards better outcomes with lower BP targets, no statistically significant effect on functional outcomes or death was observed at 3 months. The quality of evidence is low or very low, as detailed in the evidence profile table (Supplemental Table 4). The evidence mainly applies to conscious patients with systolic BP below 220 mmHg. The effects on patients with large haematomas, those requiring surgical decompression, severely elevated BP (>220 mmHg) or severe premorbid disabilities remain uncertain.
Additional information
Most HE occurs within the first 3 h. 71 Among the RCTs included in the meta-analysis, only those conducted in the prehospital phase (RIGHT-2, MR ASAP and INTERACT-4) recruited patients within an average of 3 h, although the INTERACT-4 trial managed to recruit all patients within 2 h.56,57,68 However, BP lowering in undifferentiated stroke should be avoided because its effect on people with ischaemic stroke appears to be harmful.72,73
High systolic BP (SBP)
The optimal choice of antihypertensive drug(s) and the optimal duration of intensive BP lowering remain uncertain. To achieve rapid and sustained BP reduction without excessive lowering, the drugs used should be fast-acting with a short half-life. Various antihypertensives, meeting these criteria to varying degrees, have been tested in RCTs. These include labetalol, nicardipine, lisinopril, enalapril, candesartan, urapidil, hydralazine, glyceryl trinitrate and sodium nitroprusside, with their selection often guided by local availability. With the exception of venous vasodilators (such as nitrates), no safety concerns have been noted for these antihypertensives. 85 The calcium channel antagonist clevidipine, with a half-life of 1.5 min, may be particularly effective in BP control. 86 A novel strategy for BP lowering will address multiple factors by integrating analgesia, sedation and anti-sympathetic effects. 87
For blood pressure reduction as part of care bundle treatment, please refer to Section ‘Care bundles.’
Given the uncertainties about the effects of blood pressure reduction on our chosen clinical outcomes overall and in sub-groups, and in patients with haematomas >30 mL, further research is warranted. The ICH ADAPT II trial was published after the completion of this guideline. 88 The ongoing CLUTCH (NCT06402968) is comparing the effect of the short-acting clevidipine vs. standard antihypertensive therapy on SBP target with stability, which is defined as achieving a SBP of less than 150 mm Hg, but greater than 130 mm Hg, plus two subsequent consecutive recordings, taken at least 15 minutes apart, remaining within that 130-150 mm Hg range. Other ongoing trials are TIME-ICH (NCT06760078), looking at the efficacy of TXA vs. placebo both groups including plus intensive blood pressure. and the observational study of Efficacy and Safety Study of Urapidil Alone or With Esmolol in Treating Acute Hypertensive Intracerebral Hemorrhage (NCT06635707).
Haemostatic therapies
Haematoma expansion is associated with worse functional outcome and death.89–94 Haemostatic therapies aim to reduce the risk of haematoma expansion after acute ICH. The type and effects of haemostatic therapy may vary in studies that include ICH not associated with antithrombotic therapy, ICH associated with antiplatelet therapy, and ICH associated with anticoagulation. 72 Clotting factors and antifibrinolytics have been used to treat acute spontaneous ICH. Platelet concentrates, and desmopressin have been used for ICH associated with antiplatelet therapy. For ICH associated with vitamin K-antagonists (VKAs), prothrombin complex concentrate (PCC) or fresh frozen plasma (FFP) have been used. Andexanet alfa has been used for ICH associated with factor Xa inhibitors, while idarucizumab has been used for ICH associated with the direct thrombin inhibitor dabigatran.
Analysis of current evidence
The literature search was based on the Cochrane systematic review, which included a literature search from 1949 to September 2022. 95 The literature search for this guideline was updated from September 2022 to 31 May 2024 and identified four additional RCTs,96–99 resulting in the inclusion of 23 RCTs with 5495 participants (Supplement for PICO 3: description of single studies). The systematic search included RCTs of any haemostatic intervention (i.e. procoagulant treatments such as clotting factor concentrates, antifibrinolytic drugs, platelet transfusion or agents to reverse the action of antithrombotic drugs) compared with placebo, open control or an active comparator for acute spontaneous ICH. A description of the RCTs is provided in the Supplement. The guideline group graded the following outcomes to be critical or important for haemostatic therapies: death, death or dependence (modified Rankin Scale (mRS) 4–6) by day 90, haematoma expansion at 24 h, and thromboembolic adverse events. Most of our recommendations differ from those in the ESO Guideline on Reversal of Oral Anticoagulants in Acute Intracerebral Haemorrhage published in 2019. 94 This is mainly due to a shift in the evaluation of the benefits versus risks of treatments, with thromboembolic adverse events specifically identified as a critical outcome.
Spontaneous ICH not associated with antithrombotic drug use
rFVIIa

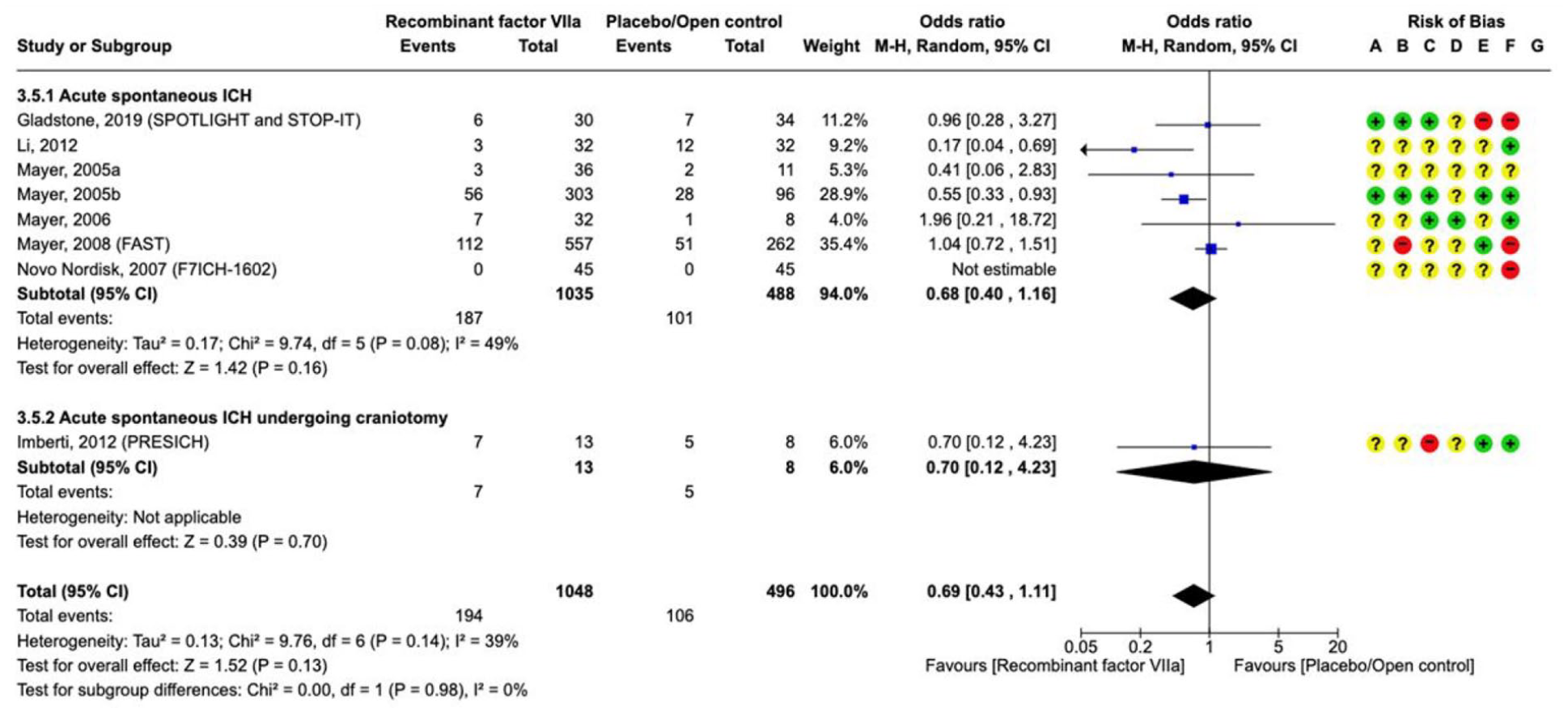
Analysis of current evidence
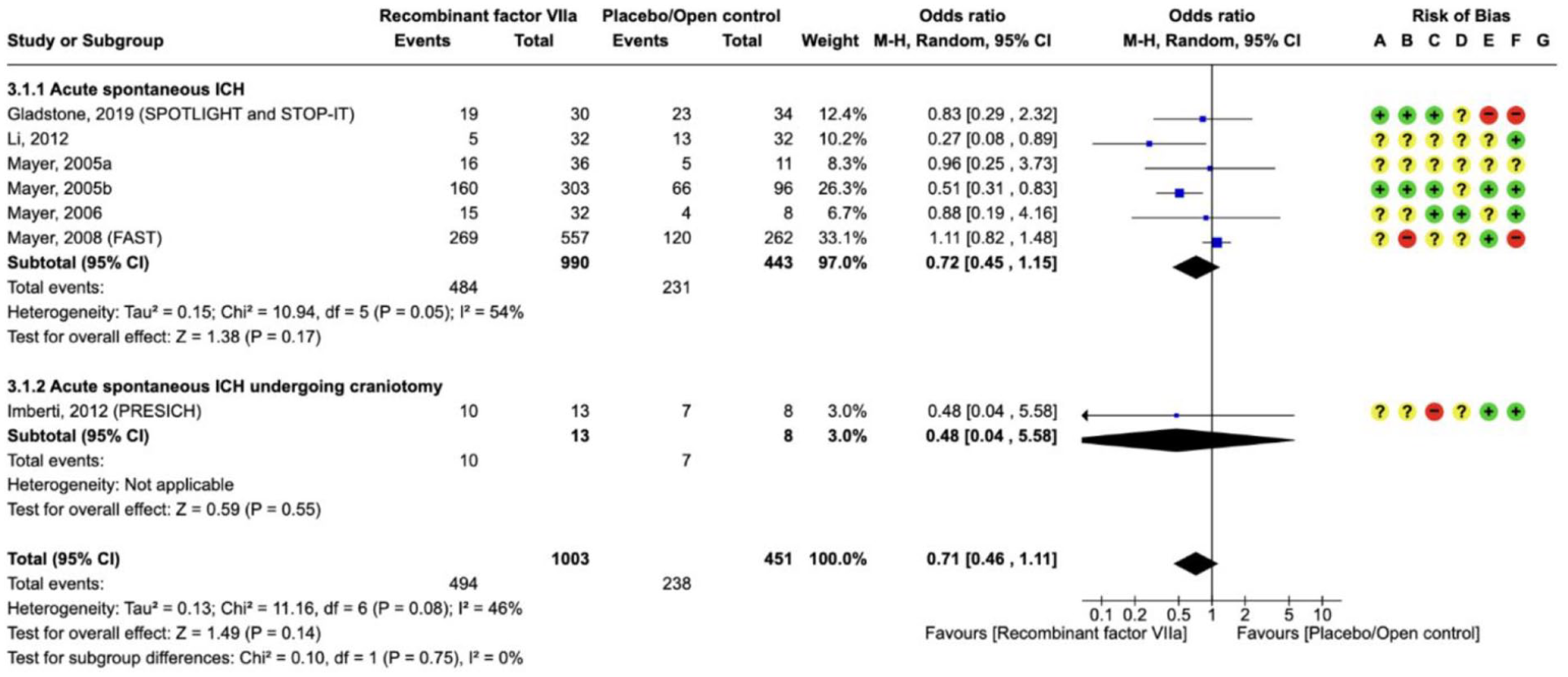
Nine RCTs (1549 participants) compared rFVIIa versus placebo or open control,90,100–106 which are described in the Supplement (Supplement for PICO 3: description of single studies). The quality of evidence is low due to heterogeneity and risk of bias (Supplement for PICO 3 GRADE evidence profile rFVIIa).
For the efficacy outcomes of death, death or dependence at 90 days and haematoma expansion, there are no statistically significant differences between rFVIIa and placebo/open control, but the direction of the effect favours rFVIIa on
Effect on
Effect on
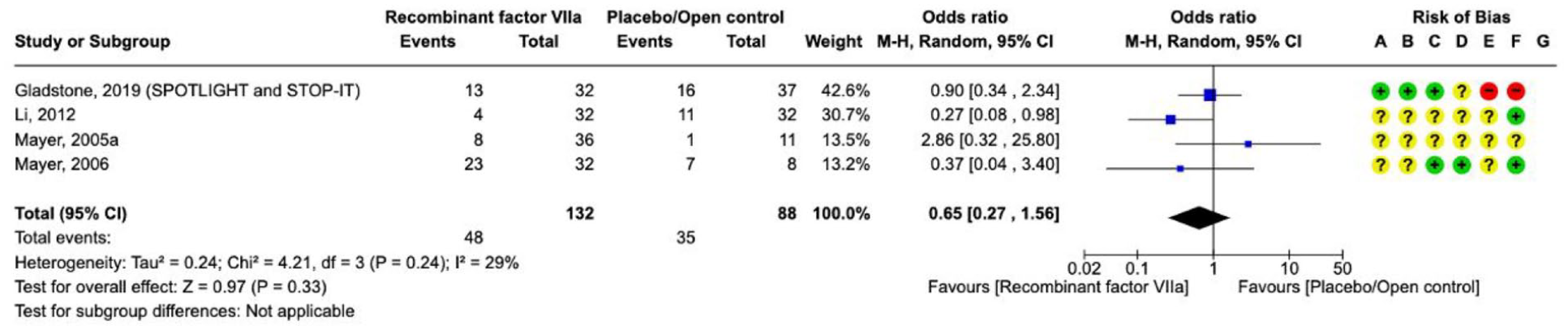
Effect on
For the safety outcome of
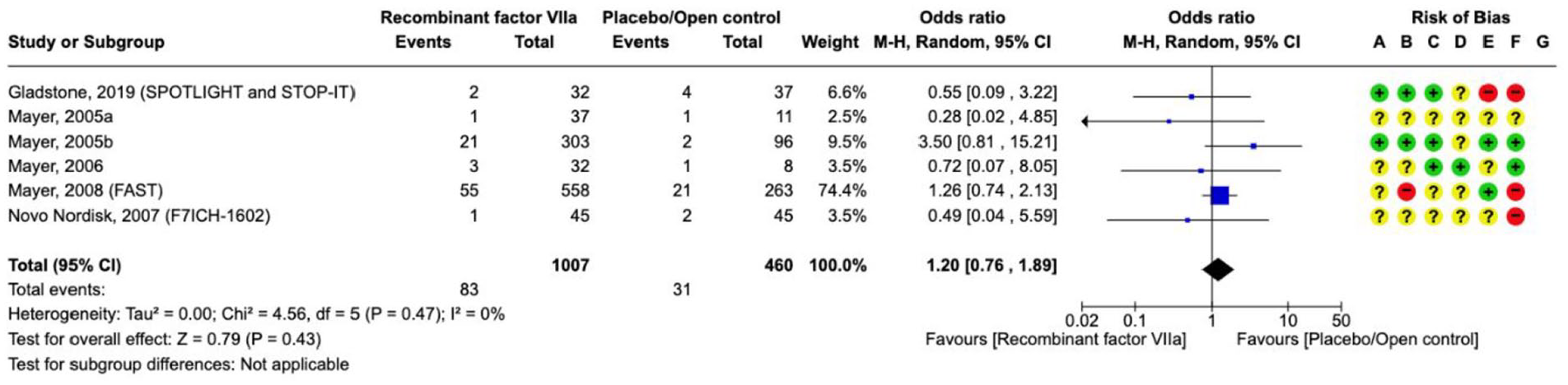
Effect on
Additional information
Further analyses on death or dependence (measured by extended Glasgow Outcome Scale (GOS-E) 1–4) at day 90 show a direction of the effect favouring rFVIIa. There was no clear effect on all serious adverse events (Supplement PICO 3, forest plots on rFVIIa).
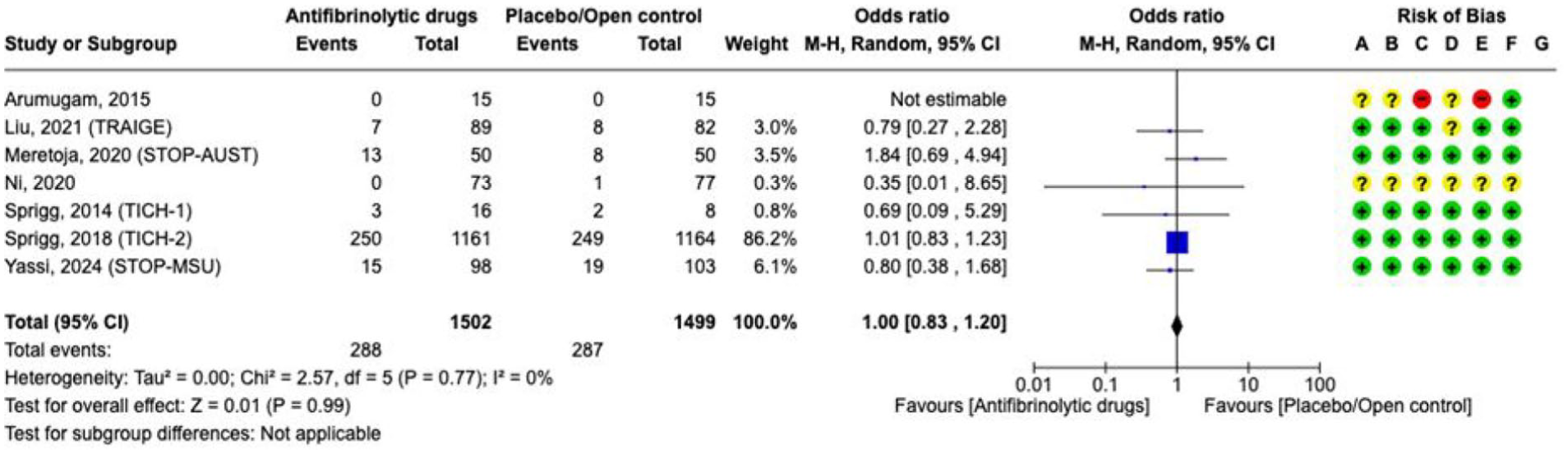
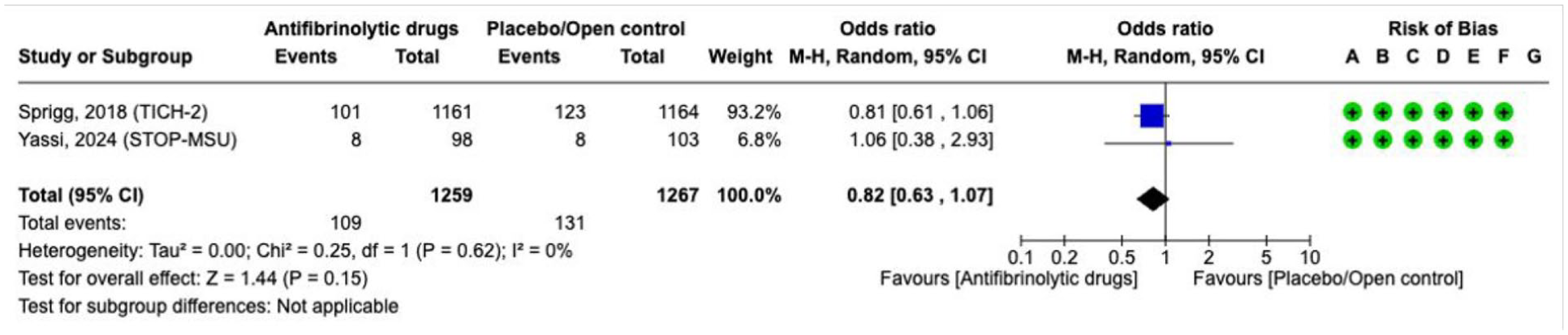
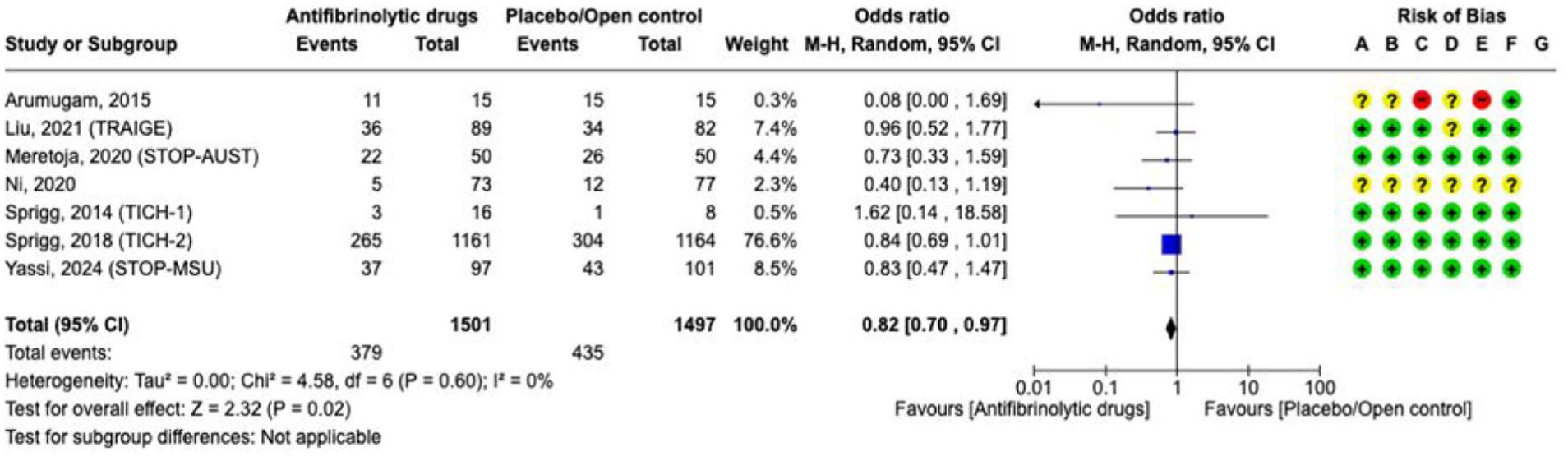
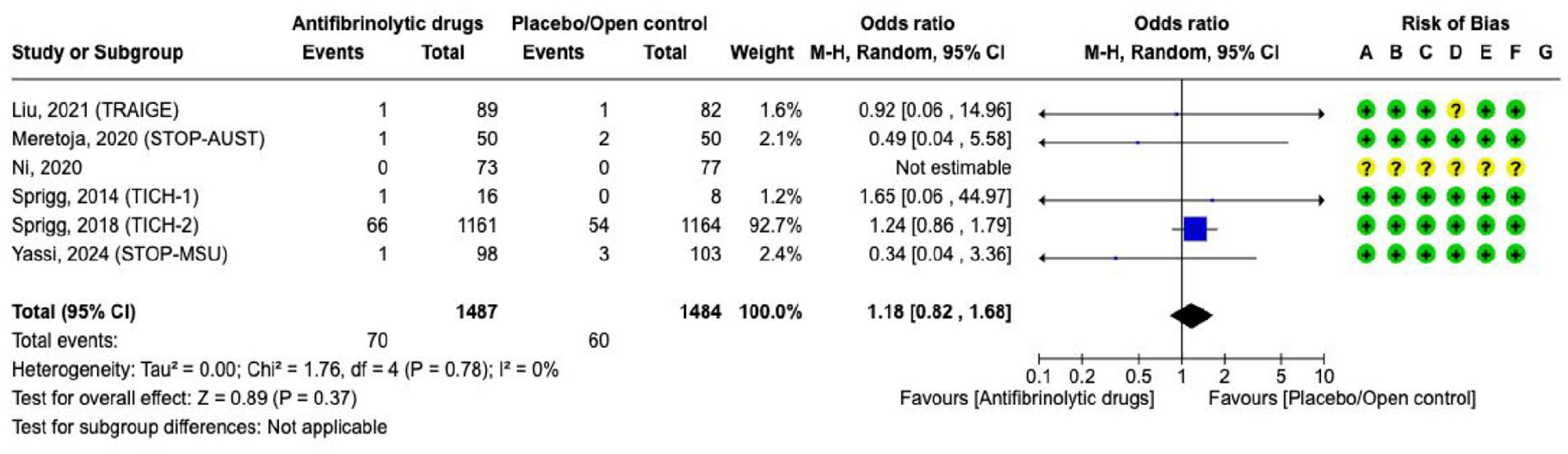
Tranexamic acid for spontaneous ICH


Analysis of current evidence
The literature search identified 8 RCTs (3061 participants) comparing tranexamic acid versus placebo/open control (Supplement for PICO 3: description of single studies).96,98,107–112 We did not include the ATICH trial of aminocaproic acid because it included only three patients and it was never published.
113
The overall quality of evidence is moderate due to the high risk of bias, limited number of events and/or small sample size (Supplement for PICO 3 GRADE evidence profile TXA). Tranexamic acid for spontaneous ICH compared with placebo/open-label control had no statistically significant effect on
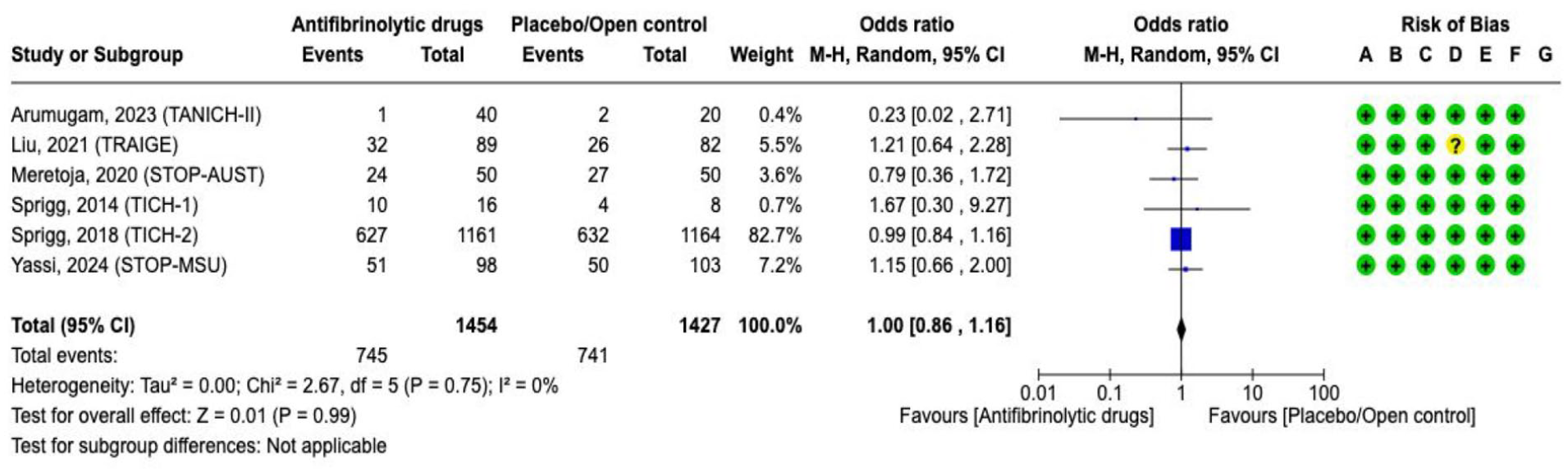
Effect on
Effect on
Effect on
Effect on
Effect on
Spontaneous ICH associated with antiplatelet drug use
Platelet transfusion

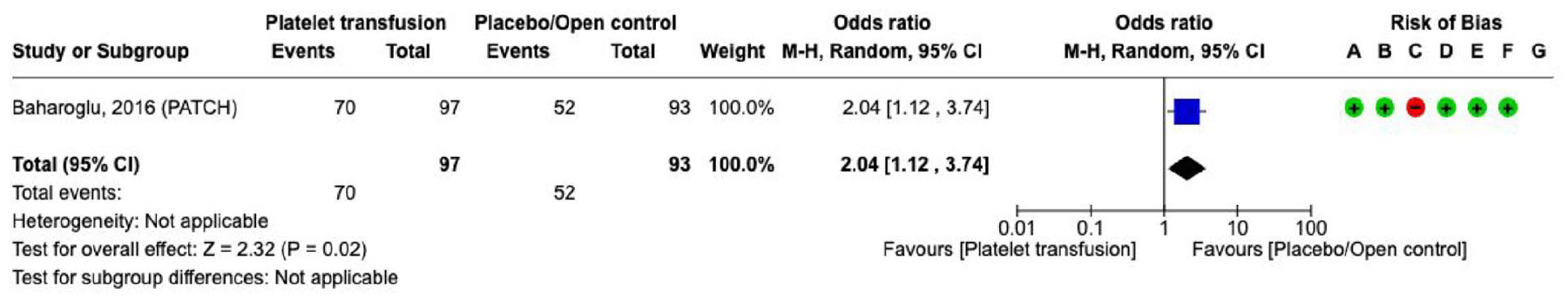
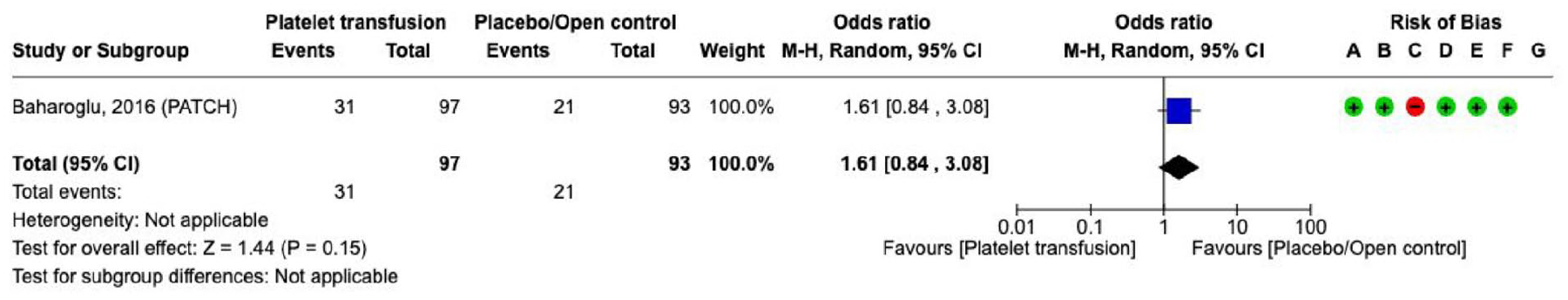
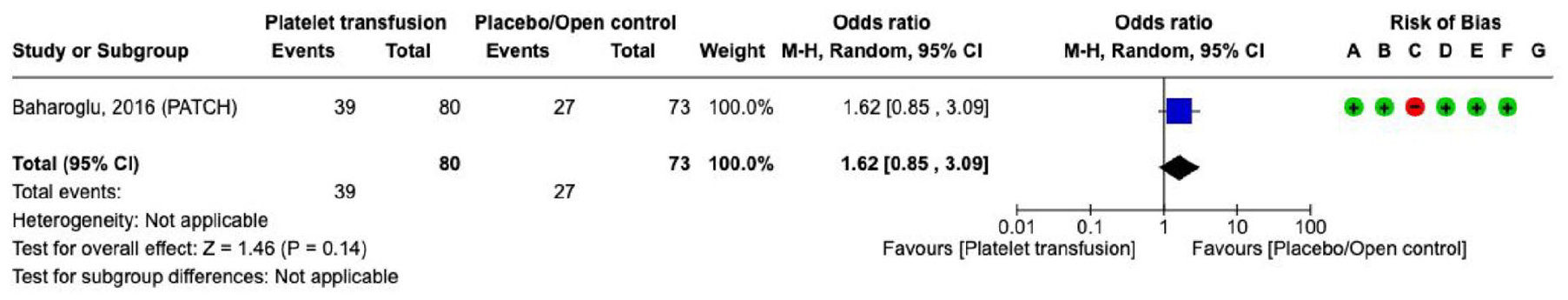
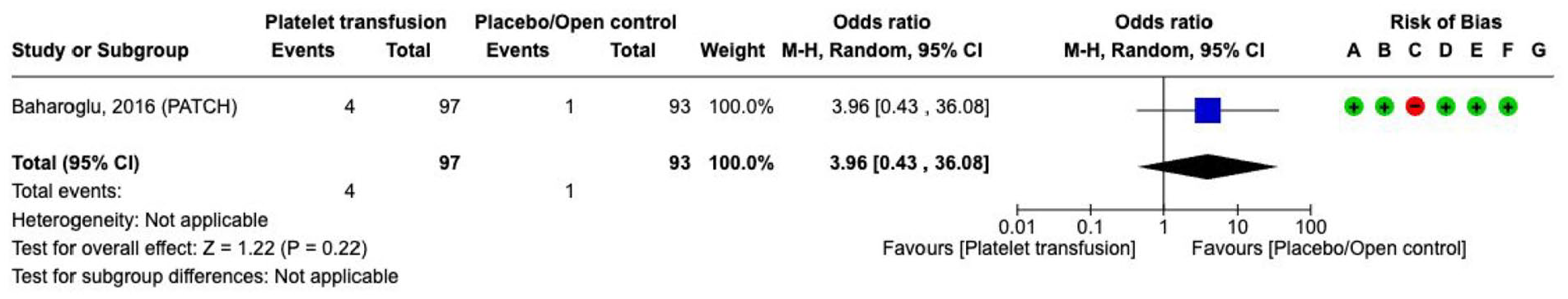
Analysis of current evidence
The literature search identified one RCT that evaluated the effect of platelet transfusion in patients with spontaneous ICH associated with antiplatelet therapy (Supplement for PICO 3 description of single studies, and GRADE evidence profile for platelet transfusion).
114
Platelet transfusion increased the likelihood of
Effect on
Effect on
Effect on
Effect on
Additional information
The analysis of all serious adverse events revealed a direction of the effect favouring placebo/open control (Supplement figure PICO 3).
Desmopressin

Analysis of current evidence
The literature search identified one RCT evaluating desmopressin in patients with spontaneous ICH associated with antiplatelet therapy (n = 54). That trial randomised 27 patients to desmopressin and 27 patients to placebo (Supplement for PICO 3 description of single studies).
97
The overall quality of evidence is very low due to the risk of bias and the small number of events (Supplement for PICO 3, GRADE evidence profile for desmopressin). There were neutral results for the efficacy outcomes
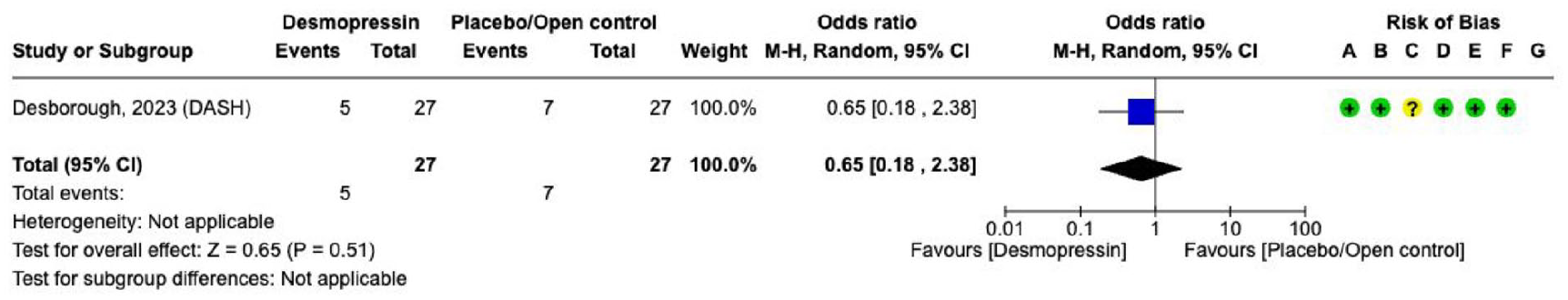
Effect on
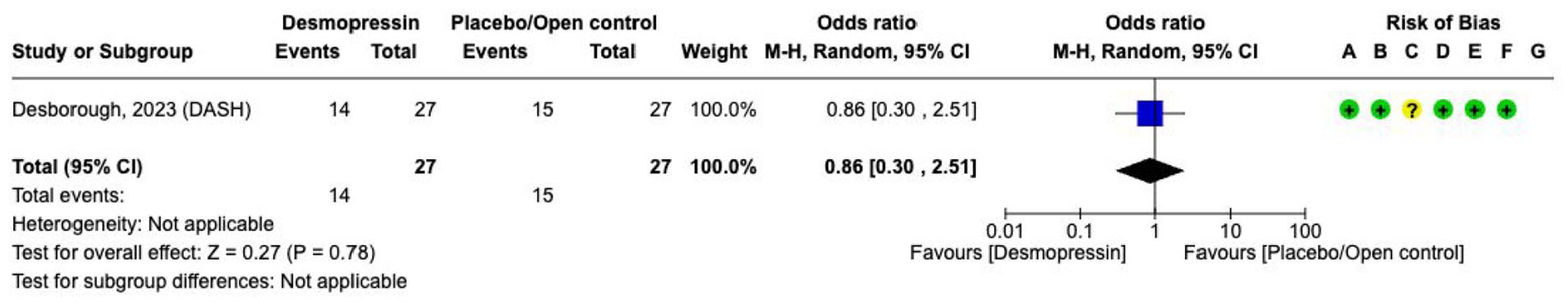
Effect on
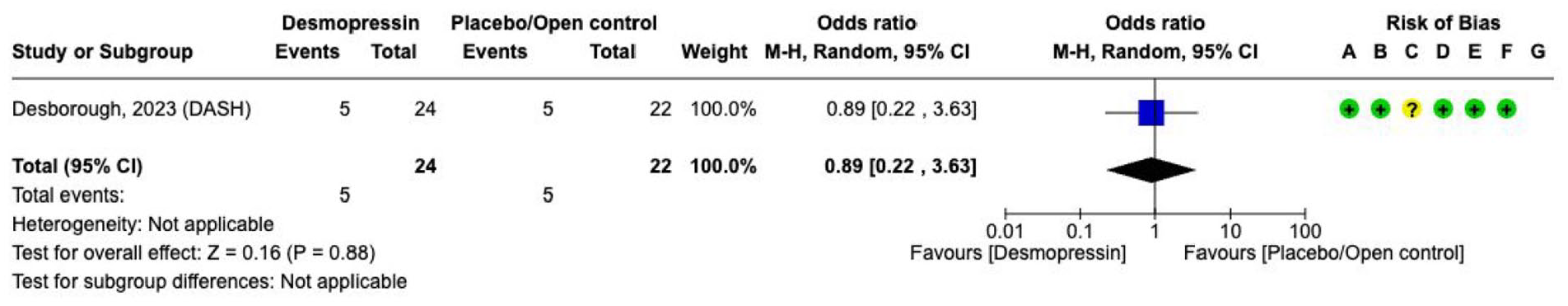
Effect on
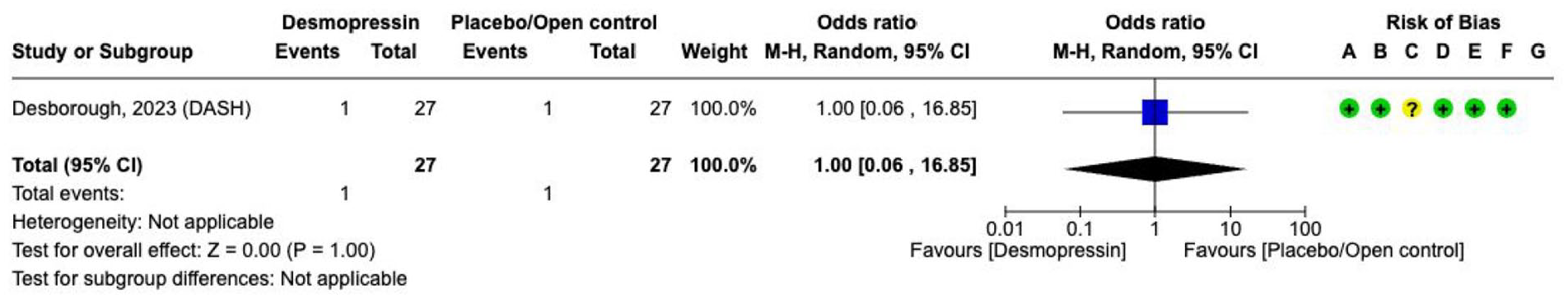
Effect on
Anticoagulant-associated ICH
ICH associated with use of vitamin K-antagonists
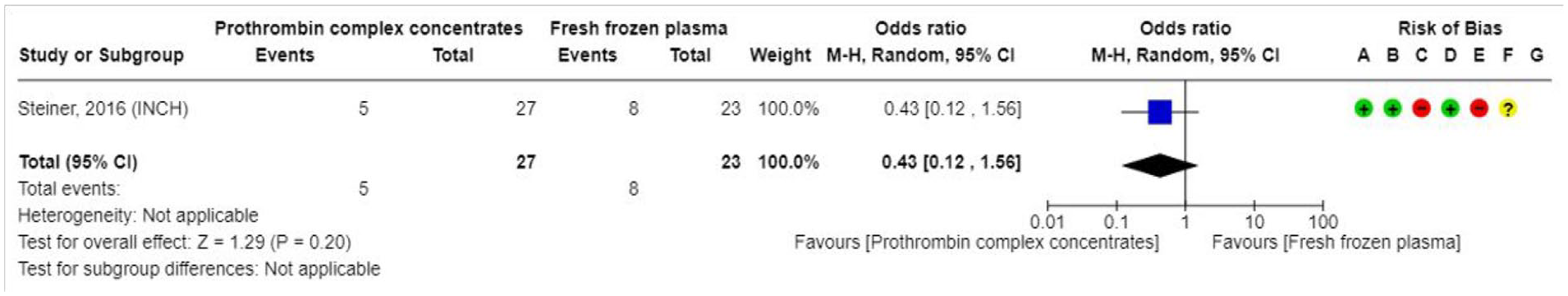
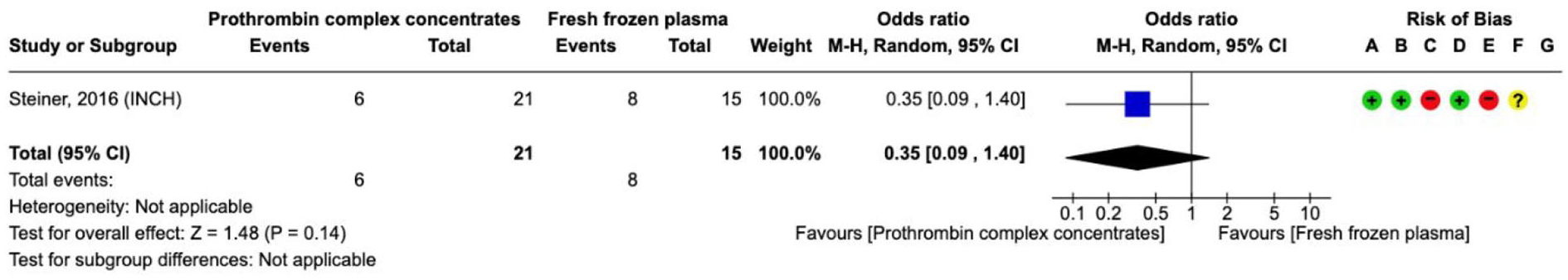
Prothrombin complex concentrate (PCC) versus fresh frozen plasma (FFP)


Analysis of current evidence
Two RCTs have compared PCC with fresh frozen plasma (FFP) for treatment of patients with ICH associated with use of VKA (Supplement for PICO 3 description of single studies, and GRADE evidence profile for PCC vs FFP).115,116 In the FFP versus PCC in patients with intracranial haemorrhage related to vitamin K-antagonists (INCH) trial all patients received 10 mg intravenous vitamin K. In the study by Boulis et al., only patients in the control group received subcutaneous vitamin K. The overall quality of evidence is very low due to high risk of bias and limited number of events. The RCTs had the speed of normalisation of INR as the primary outcome and were not powered to detect differences in clinical outcomes. The evidence was uncertain for the effect on
Effect on

Effect on
Effect on
Additional information
In the INCH trial, patients randomised to FFP treatment received rescue therapy with PCC if the INR had not normalised by 3 h after start of FFP-infusion, which was the primary outcome event. PCC was superior to FFP in normalising INR levels (aOR 30.6, 95% CI 4.7–197.9). In the expert consensus statement, we suggest a fixed dose of 30 or 50 U/kg. The fixed dose of 30 U/kg was used in INCH, the higher dose of 50 U/kg is recommended by the manufacturer. The analysis of all serious adverse events revealed a direction of the effect in favour of PCC (Supplemental Figure PICO 3).
Our systematic literature search did not identify any RCT comparing vitamin K with placebo or open control. Four-factor PCC includes factors II, VII, IX and X, with factor VII having the shortest half-life time of approximately 4–6 h. The half-life of the effects of various vitamin K-antagonists ranges from 8 h to 6 days (acenocumarol: 8–11 h, warfarin: 37–48 h, phenprocoumon: 96–155 h). An increase in INR 12–24 h after reversal therapy with FFP or PCC without the addition of vitamin K has been reported. 117 Consequently, the immediate intravenous administration of vitamin K is recommended, and may require repetition in order to prevent a rebound of the INR. The INR must be monitored until sustained normalisation is demonstrated.
ICH associated with use factor Xa inhibitors (FXaI)
Prothrombin complex concentrates


Analysis of current evidence
PCC has been used for the treatment of ICH associated with factor Xa-inhibitor use due to the historical lack of an alternative. The literature search did not identify any RCTs comparing the efficacy of PCC with other standard therapies (except for a subgroup of patients included in the ANNEXA-I trial).
Additional information
Observational data (see Supplement) have not shown a clear association between PCC treatment and improved outcomes, such as reduced mortality, improved functional status or reduced haematoma expansion rates, compared with usual care.118,119 Limited observational data suggest that thromboembolic adverse events occur in a low rate after administration of PCC.120,121
Andexanet alfa


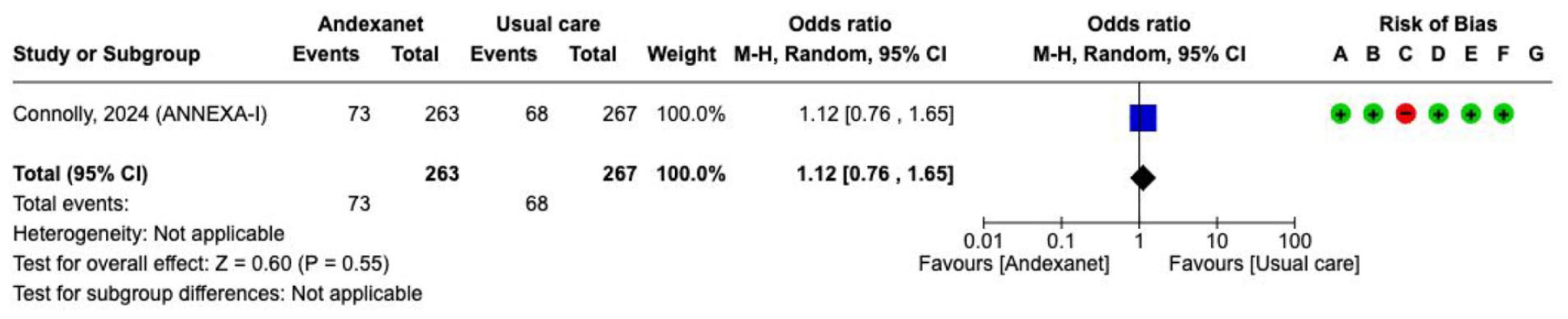
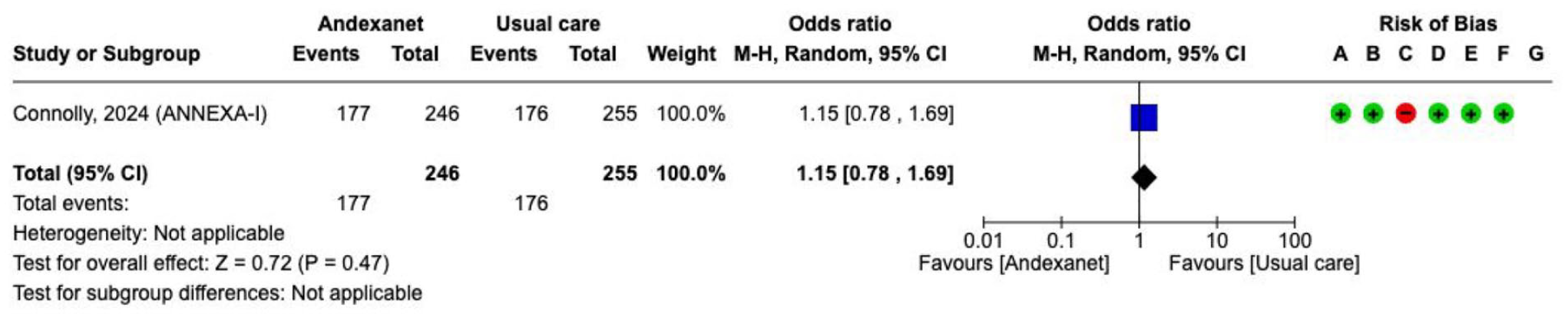
Analysis of current evidence
One RCT (n = 530) investigated the effect of andexanet alfa versus usual care (Supplement for PICO 3 description of single studies, and GRADE evidence profile for andexanet alfa).
99
The primary outcome was haemostatic efficacy, defined as a combination of three different measures at 12 h after baseline: expansion of the haematoma volume of ⩽35%, an increase in NIHSS ⩽ 7 points, and no receipt of medical or surgical rescue therapy. The primary outcome was met with a significantly higher proportion of patients receiving andexanet alfa compared with usual care (150/224 (67%) vs 121/228 (53%), adjusted difference 13.4%, 95% CI 4.6–22.2). There was no difference between groups for
Effect on
Effect on
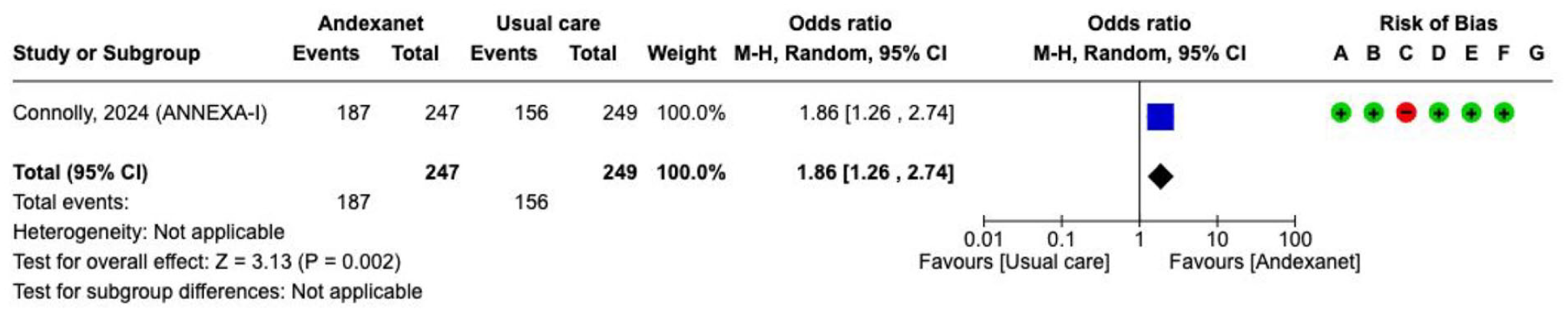
Effect on
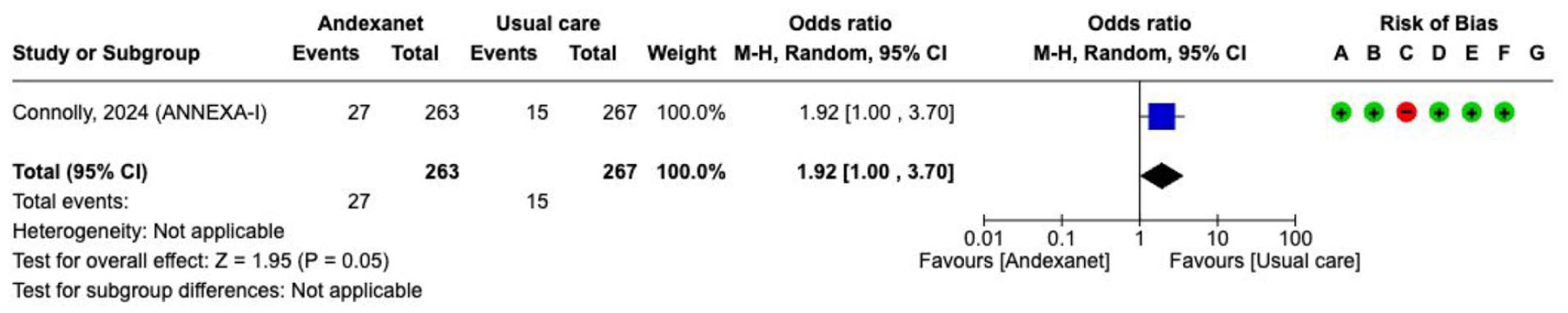
Effect on
Additional information
The secondary endpoint of reduction in factor Xa activity within 2 h was achieved in 94.5% of patients treated with andexanet alfa compared with 26.9% of those who received standard care. 99
Tranexamic acid
PICO 3.3.2.3 In adults with ICH associated with use of factor Xa-inhibitor (FXaI, apixaban, edoxaban, rivaroxaban) does tranexamic acid compared with standard care reduce death or dependence, death or haematoma expansion and not increase risk of thromboembolic adverse events?

Analysis of current evidence
One RCT investigated tranexamic acid versus placebo in patients with ICH associated with FXaI (Supplement: Description of single studies). 112 There was no difference between TXA and placebo in death or dependence by 90 days (OR 1.23, 95% CI 0.40–3.74, 1 RCT, 63 participants), death by 90 days (OR 1.22, 95% CI 0.45–3.31, 1 RCT, 63 participants) or haematoma expansion (OR 0.73, 95% CI 0.27–1.99, 1 RCT, 63 participants). There was no difference between TXA and placebo in thromboembolic adverse events (OR 2.07, 95% CI 0.35–12.22, 1 RCT, 63 participants). The overall quality of evidence is low due to the identification of only one RCT with a small sample size and a limited number of events.
The ongoing TICH-3 RCT is including people with ICH associated with FXaI use (ISRCTN97695350).
ICH associated with use factor II inhibitors
Idarucizumab


Analysis of current evidence
Therapy with idarucizumab, a monoclonal antibody fragment which inactivates the anticoagulant effect of dabigatran, has been licensed worldwide since 2016. No completed or ongoing RCTs exploring the efficacy and/or safety of idarucizumab versus usual care for reversal of direct factor IIa-inhibitor therapy in adults with ICH were found in the literature search.
The REVERSE-AD study was a prospective case series (n = 503) that evaluated a primary outcome of maximum percentage reversal of the effect of dabigatran within 4 h after administration of idarucizumab. The primary outcome was determined by normalisation of two coagulation parameters: the dTT (diluted thrombin time) and the ECT (ecarin clotting time). Fifty-three of the included patients suffered an ICH. All patients who suffered an ICH reached the primary endpoint of either normalisation of dTT or ECT. There were no serious thromboembolic adverse events within the first 5 days.122,123
Additional information
A systematic review of 30 observational studies, involving 3602 patients, reported good haemostatic efficacy in 77.7% of patients with bleeding, regardless of location. 124 The pooled proportion of thromboembolic events at any follow-up time was 2.0%.
Ongoing trials
There are 10 ongoing RCTs investigating haemostatic therapy in intracerebral haemorrhage:
Surgical management
Supratentorial ICH
Any surgery aimed at haematoma removal

Analysis of current evidence
This PICO addresses available evidence on the general hypothesis that surgical haematoma removal for supratentorial ICH may prevent secondary injury. Death or dependence were assessed as critical outcomes. A total of 17 RCTs involving 3927 participants compared the effect of any surgical approach aimed at haematoma removal against no surgery on the risk of death (Supplement PICO 4: description of studies).126–142 The effect of surgery was evaluated at 3 months (four studies), 6 months (nine studies) or 12 months (four studies). Surgery aimed at haematoma removal compared with no surgical treatment reduced the risk of
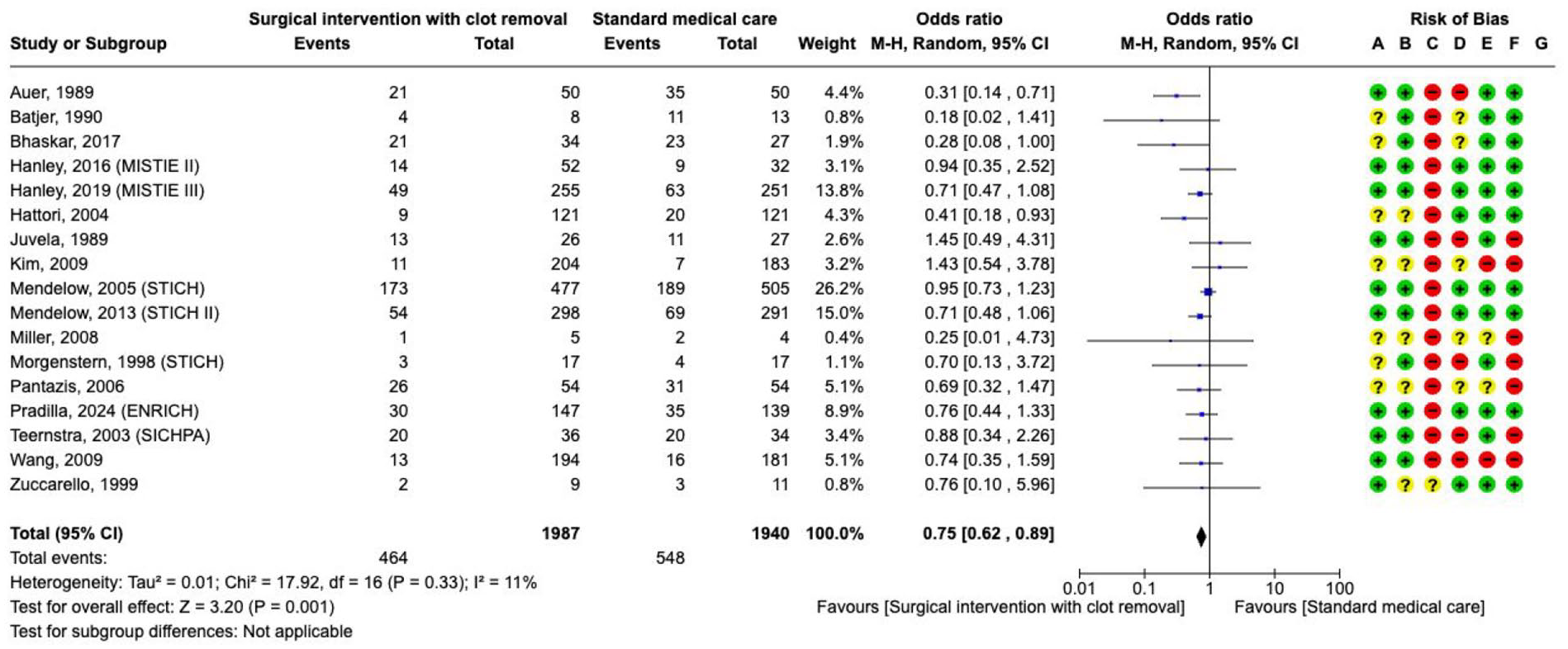
Effect on
The effect of surgery aimed at haematoma removal against no surgery on
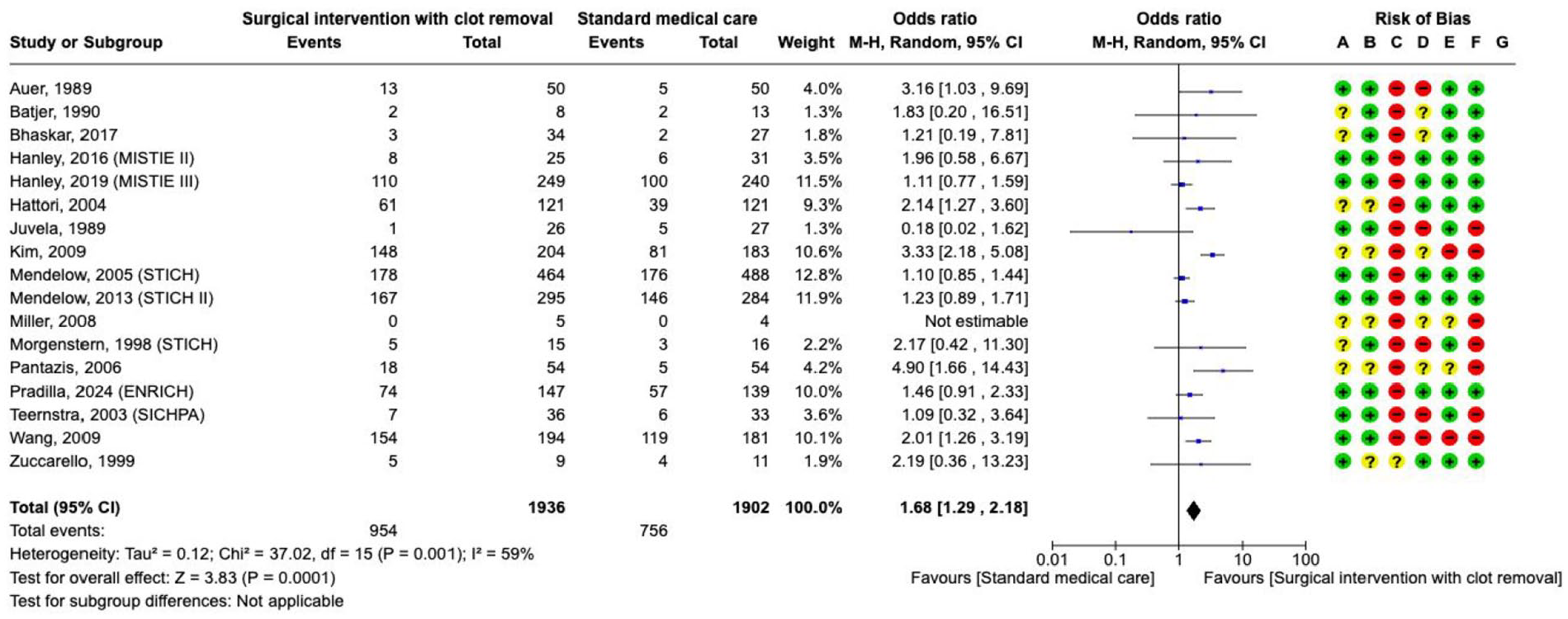
The effect on 3–12 months
Additional information
ICH volume is an important predictor of death and functional outcome for patients with supratentorial ICH. 143 Besides the direct brain injury by compression and disruption of the parenchyma, blood degradation products and plasma-derived components trigger an inflammatory response leading to additional injury. Neurosurgical haematoma evacuation can affect clinical outcome, either via direct alleviation of the mass effect or by ameliorating secondary brain injury after ICH. The importance of haematoma volume reduction was shown in an exploratory, observational analysis of MISTIE-III, in which an end-of-treatment ICH volume of 15 mL or less was associated with a 10.5% additional increase in mRS score 0–3 at 1 year. 130
In addition to the extent of haematoma volume reduction, other factors might influence the outcome after surgery, including ICH location (lobar vs non-lobar), Glasgow Coma Score (GCS) at presentation and time between symptom onset and surgery. In an individual patient data meta-analysis (IPDMA) comprising 8 RCTs published from 1985 to 2010 involving 2186 participants, randomisation within 8 h of ICH onset and a GCS between 9 and 12 were associated with better outcome after surgery. 139
Results on the modifying effect of ICH location are conflicting. An exploratory subgroup analysis of STICH 134 suggested a possible benefit of surgical treatment on lobar ICH, which was not confirmed in STICH-II. 135 The IPDMA, which included both STICH trials, indicated a non-significant benefit for more superficial lobar haematomas without intraventricular haemorrhage with surgery (OR 0.68, 95% CI 0.44–1.06, 4 RCTs, 340 participants). 144 More recently, minimally invasive surgery has been shown to improve functional outcome measured with utility-weighted mRS compared with no intervention, which appears attributable to intervention for lobar ICH. 138
Haematoma removal by means of craniotomy and open standard surgical technique

Analysis of current evidence
Surgical haematoma removal for supratentorial ICH by means of craniotomy compared with no surgical treatment did not reduce the risk of
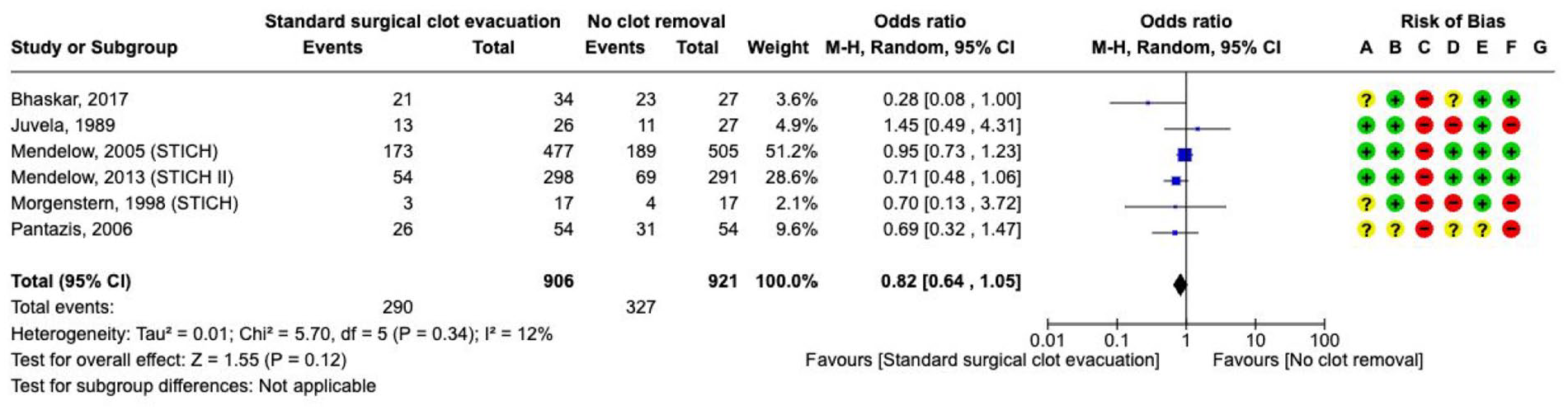
The effect on
A total of 5 RCTs involving 1753 participants with supratentorial ICH compared the effect of surgery aimed at haematoma removal against no surgery on
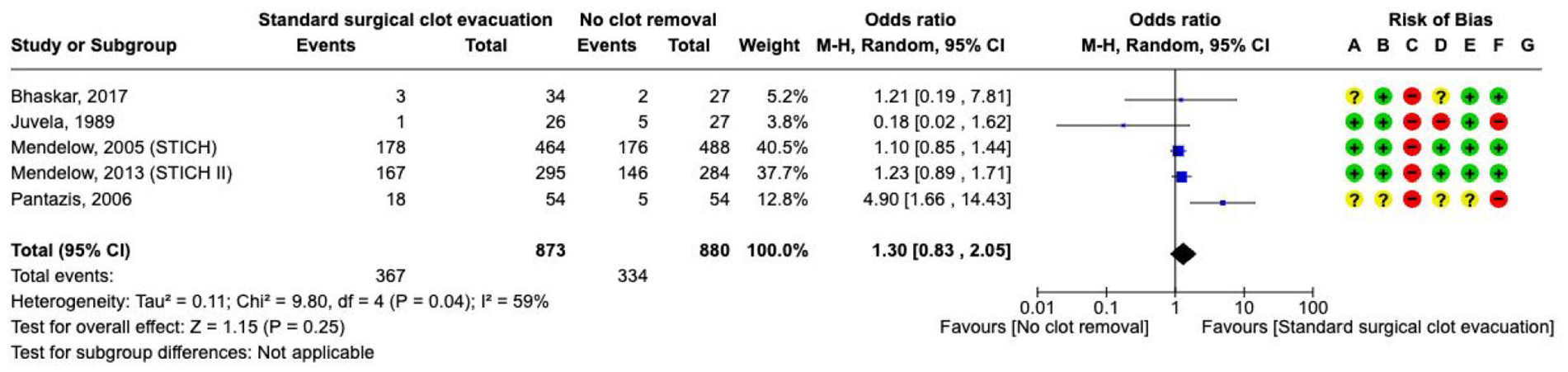
The effect on
Additional information
The role of craniotomy to perform surgical evacuation of supratentorial ICH remains uncertain. Results from two large RCTs have not shown a clear benefit on death or functional outcome. In STICH, 1033 patients with spontaneous supratentorial ICH that had occurred within 72 h, were randomised to surgery for evacuation of the haematoma (within 24 h of randomisation) versus initial conservative treatment. 134 Primary outcome was the prognosis-based favourable outcome from the eGOS at 6 months, which was comparable between the two groups (26% in surgical group vs 24% in medical management group; OR 0.89, 95% CI 0.66–1.19, p = 0.414). It was considered that these results might have been attributed to the inclusion of patients with intraventricular extension and the late timing of the intervention.
In STICH II, 601 patients with spontaneous superficial ICH affecting the lobar region within 1 cm of the cortex and without ventricular extension that had occurred within 48 h, were randomised to early craniotomy to evacuate haematoma (within 12 h after randomisation) or conservative treatment. The primary outcome was prognosis-based favourable outcome from the eGOS (extended Glasgow Outcome Scale) at 6 months, which was similar in both treatment arms (41% surgical group vs 38% medical management group; OR 0.86, 95% CI 0.62–1.20, p = 0.367). 135
As mentioned in PICO 4.1.1, an IPDMA from 8 studies published between 1985 and 2010 involving 2186 participants demonstrate that randomisation within 8 h of ictus and a GCS between 9 and 12 were associated with better outcome, whilst there was non-significant evidence that lobar ICH without intraventricular extension may benefit from surgery. 144
Minimally invasive surgical removal (MIS)

Analysis of current evidence
There are four RCTs comparing minimal invasive surgical techniques with medical management (Supplement for PICO 4.1.3 GRADE evidence profile).126,131,136,138 Minimal invasive surgery aimed at haematoma removal compared with no surgical treatment improved
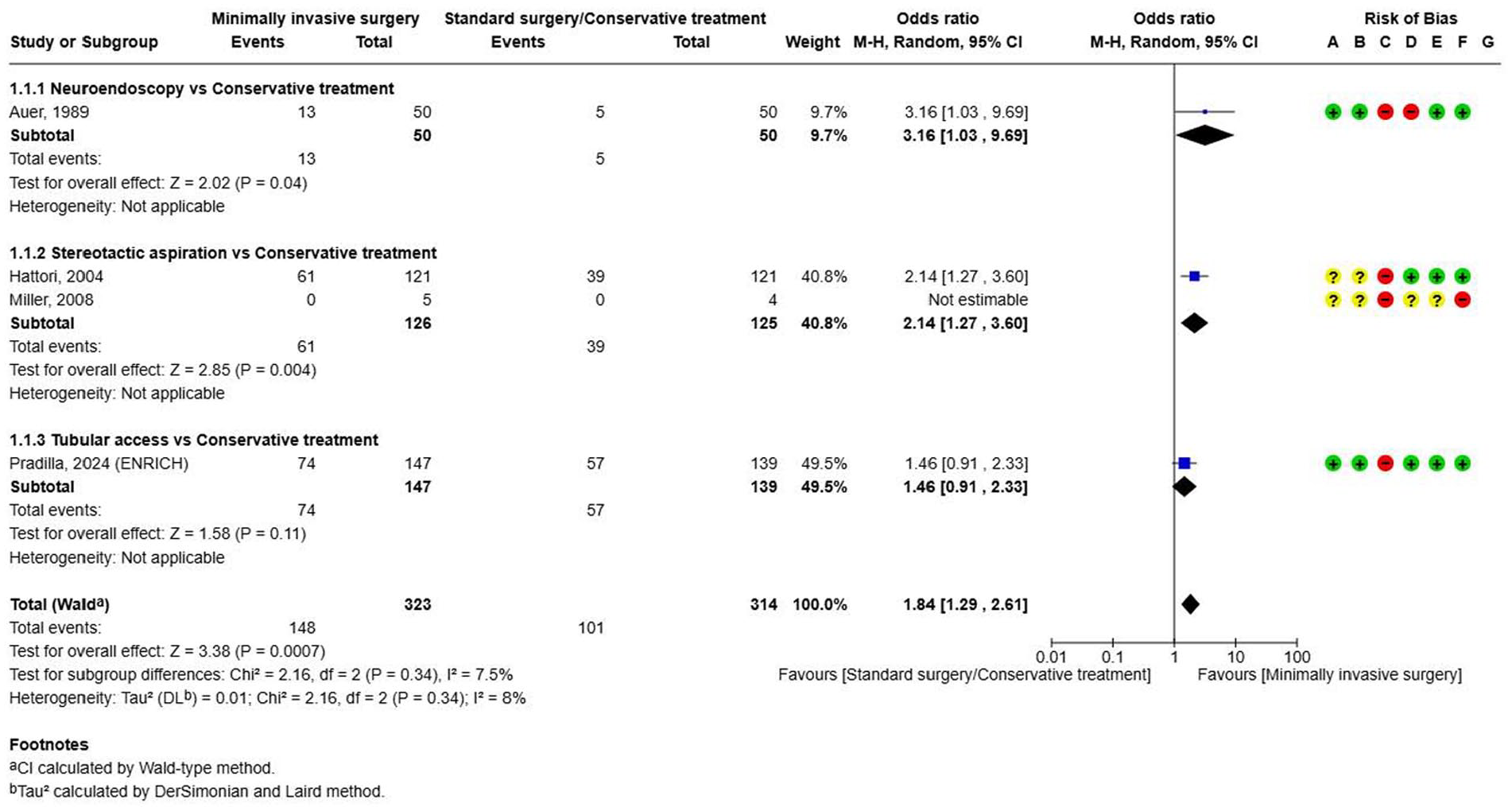
The effect on
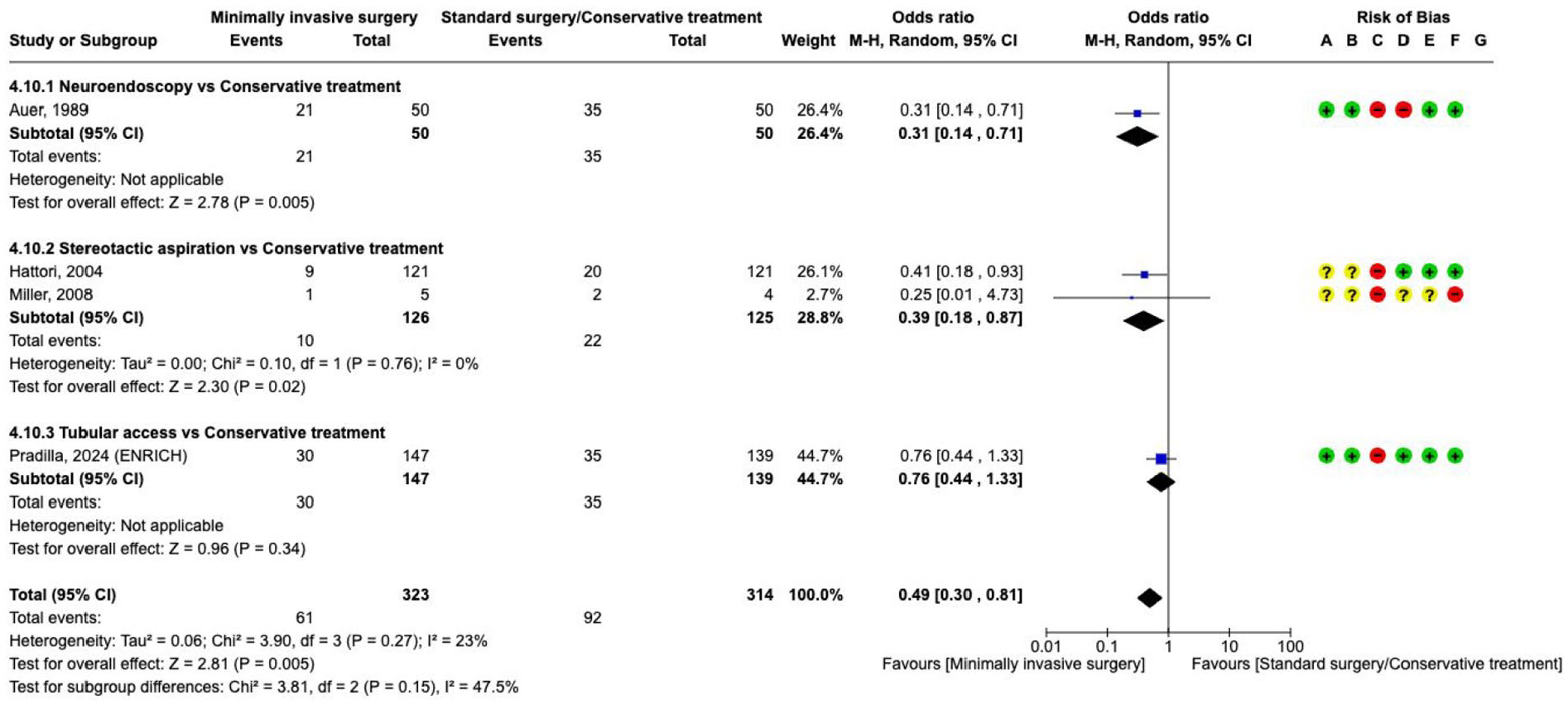
The effect on
Our recommendation is based on the ENRICH trial investigating 300 people with lobar or basal ganglia haemorrhage. 138 This trial described itself as minimally-invasive because it took a trans-sulcal approach to the ICH with a tubular access device, and special attention was paid to align the approach along the long axis of the white matter tracts (parafascicular). Surgery had to be started within 24 h after the onset of stroke symptoms or the time the subject was last known to be well. At an interim analysis, the inclusion of deep ICH was stopped for futility based on a prespecified adaptation rule. The authors reported a better functional outcome with mean score on the utility-weighted modified Rankin scale at 180 days (the primary efficacy endpoint) of 0.458 in the surgery group compared with 0.374 in the control group, for a between-group difference of 0.084 (95% Bayesian credible interval, 0.005–0.163) in the total population. Death by 30 days occurred in fewer patients in the surgery group than in the control group (30/147 vs 35/139, OR 0.76, 95% CI 0.44–1.33; Figure 37). The results of the analysis appeared to be attributable to the surgery effect in the lobar haemorrhage location only. The generalisability of these results is limited to the restricted population meeting the trial entry criteria with respect to the haematoma volume (30–80 mL as calculated by the ABC/2 method), level of consciousness (GCS 5–14), and a time from onset of symptoms to surgery (<24 h).
Additional information
The term ‘minimally-invasive surgery’ is used for a combination of a mindset and surgical techniques with a special focus on maintaining tissue integrity and function. Historically, it developed from so-called standard surgery when additional imaging or intraoperative techniques and devices were developed. In ICH, the mindset and techniques that justify the use of the term minimally-invasive are a combination of localising eloquent cortex or subcortical tracts using imaging, navigation and mapping, minimising cortical incision, planning trajectories along the white matter fibres, respecting tissue at the haematoma borders, and avoiding coagulation close to small vessels (i.e. performing the surgery as if an eloquent tumour would be removed), with the aim to decompress the brain tissue and to significantly reduce the haematoma volume. Therefore, this approach should be analysed as a category separate from open surgery, where a combination of these methods might have been applied, but without prespecifying for all surgeries in the studies mentioned under PICO 4.1.2. The device used by the ENRICH group is, therefore, only an example of the minimally-invasive surgery mindset and surgical technique.
Stereotactic aspiration and endoscopic removal of the haematoma may also improve functional outcome; however, the latter is based on an old, small underpowered RCT, and further trials are justified. 131 In our analysis of other surgical techniques such as endoscopic haematoma removal or stereotactic aspiration, we excluded RCTs at high risk of bias or poor methodology.
Surgery with catheter placement plus thrombolysis

Analysis of current evidence
The combination of a small burr hole craniotomy with navigated or stereotactically-guided insertion of a catheter with subsequent, repeated lysis and drainage of the haematoma over time (several days) was evaluated in three RCTs with low to moderate bias due to heterogeneity and limited number of events (Supplement 4.1.4 GRADE evidence profile).124,125,129,130,134,139 The combined meta-analysis of three studies did not show an effect on
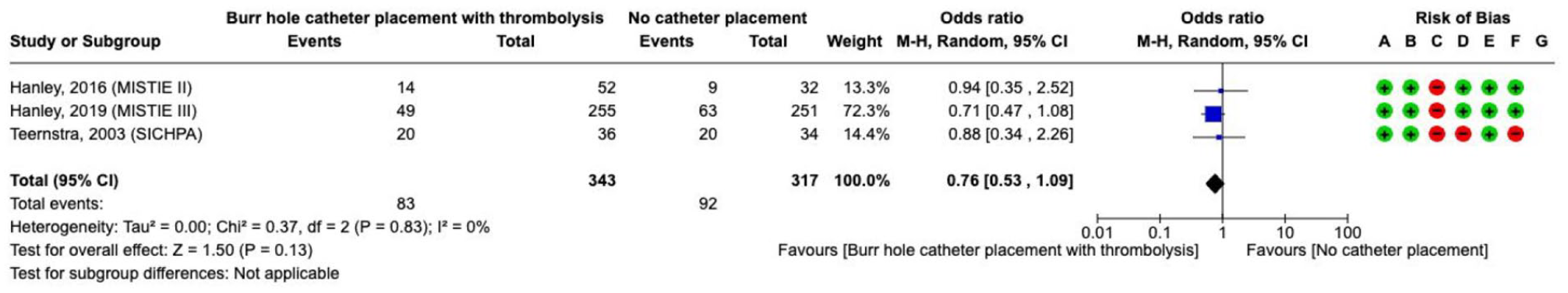
The effect on
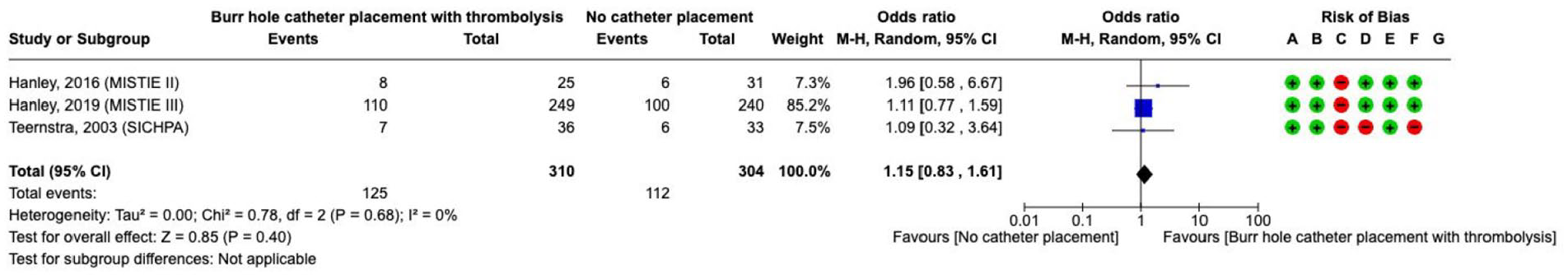
The effect on
Additional information
Of the three available RCTs,129,130,139 MISTIE III is the largest trial. MISTIE III analysed minimally-invasive catheter placement with aspiration and subsequent (repeated) thrombolysis using recombinant tissue plasminogen activator (rt-PA) of the haematoma. 130 MISTIE III used neuronavigation to plan an entry point and select a trajectory along the long axis of the haematoma avoiding eloquent tissue, waited for haematoma stability, controlled treatment progress by repeated imaging, trained surgeons and assessed long-term outcome, among others. The outcome, despite a median ICH volume at admission of 41.8 mL, was better as expected and at 1 year 43% of patients had good functional outcomes (mRS 0–3), and 80% of patients were living at home or in active rehabilitation. MISTIE III did not follow an early treatment paradigm, instead, surgery commenced after a median of 58 h after symptom onset and ended after 123 h. The treatment effect was not significant. As a secondary endpoint MISTIE III could show a slight reduction in mortality in the treatment group, however, due to multiple testing, this finding should be interpreted with some caution. MISTIE III provided an exploratory observational analysis of the end-of-treatment (EOT) volume and showed that haematoma size reduction to 15 mL or less was associated with better functional outcome at 1 year. This finding and analysis of EOT volumes should be evaluated in future trials.
Decompressive craniectomy

Analysis of current evidence
To date, only the SWITCH trial, has investigated the role of decompressive craniectomy without haematoma evacuation for 201 patients with ICH involving the basal ganglia or thalamus (Supplement 4.1.5 GRADE evidence profile). 145 The main inclusion criteria included age ⩽ 75 years, ICH volume of 30–100 mL and surgery had to be performed ⩽72 h of ictus. Primary outcome was a score on the mRS of 5–6 at 180 days. Recruitment was prematurely stopped before reaching the planned sample size of 300 participants.
Concerning
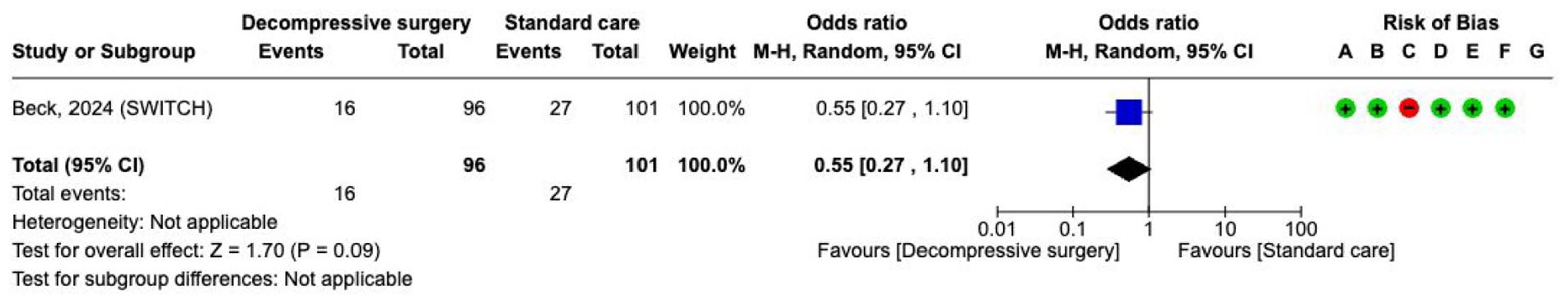
The effect on

The effect on
ICH with intraventricular extension
External ventricular drainage with or without combined lumbar drainage


Analysis of current evidence
To date, no RCTs and no prospective studies have compared the use of external ventricular drain (EVD) versus no EVD for acute ICH. However, placement of an EVD in patients with overt hydrocephalus and decreased level of consciousness is regarded as a life-saving emergency procedure in clinical practice.
Additional information
Intraventricular extension occurs in a significant proportion of patients with ICH (30%–40%). The mechanical obstruction and the blood breakdown products prevent resorption of cerebrospinal fluid (CSF), causing hydrocephalus in about 50% of these patients. 146 The insertion of an EVD is usually recommended in patients with large IVH who developed a hydrocephalus which contributes to a decreased level of consciousness.147,148 It rapidly decreases intracranial pressure and is considered a life-saving emergency procedure in these patients. There are currently no specific recommendations for selecting patients with IVH for EVD in terms of timing or volume of the IVH, except for the presence of hydrocephalus and reduced level of consciousness. In some retrospective series, EVD was associated with reduced mortality at hospital discharge.147,148 Best available evidence is a propensity score-adjusted model of retrospective data of 540 patients showing lower 30-day mortality in patients treated with an EVD compared with no EVD that had an ICH volume >11 mL (OR = 0.47, 95% CI 0.25–0.88), lower initial GCS (<13, OR 0.38, 95% CI 0.20–0.71) or IVH with increasing modified Graeb score (OR 1.09 per point, 95% CI 1.04–1.13). There was no benefit for functional outcome in patients receiving an EVD. 148
Ongoing clinical trials: The Lumbar Drainage of Intraventricular Hemorrhage (DRAIN IVH, clinicaltrials.gov NCT06510842) trial investigates whether early insertion of a lumbar drainage in addition to the EVD compared to EVD alone leads to better functional outcome and reduced shunt dependency.
External ventricular drainage with intraventricular thrombolysis

Analysis of current evidence
External ventricular drainage with intraventricular thrombolysis was investigated in three small and one larger RCTs with very serious risk of bias and serious imprecision (Supplement 4.2.2 GRADE evidence profile).144–147,149–152 Due to the high risk of bias and limited number of events, the quality of evidence was graded as very low. Meta-analysis of these four trials showed a lower
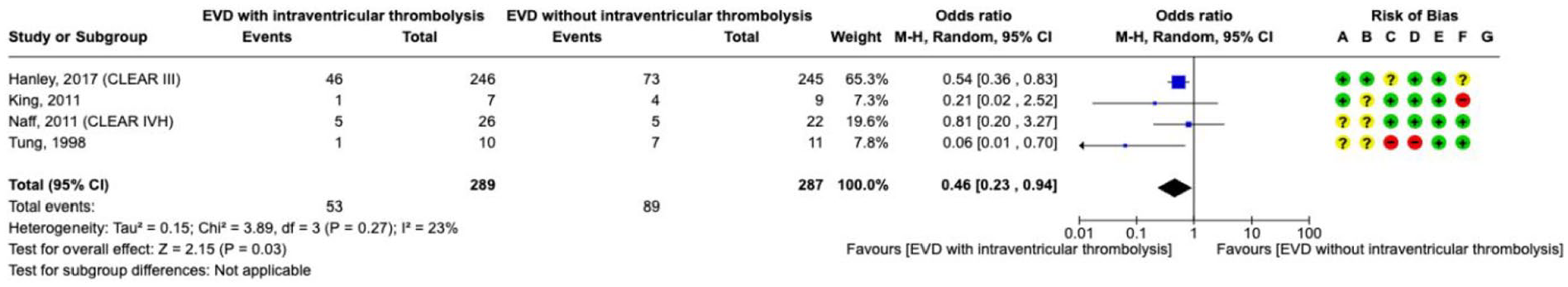
The effect on
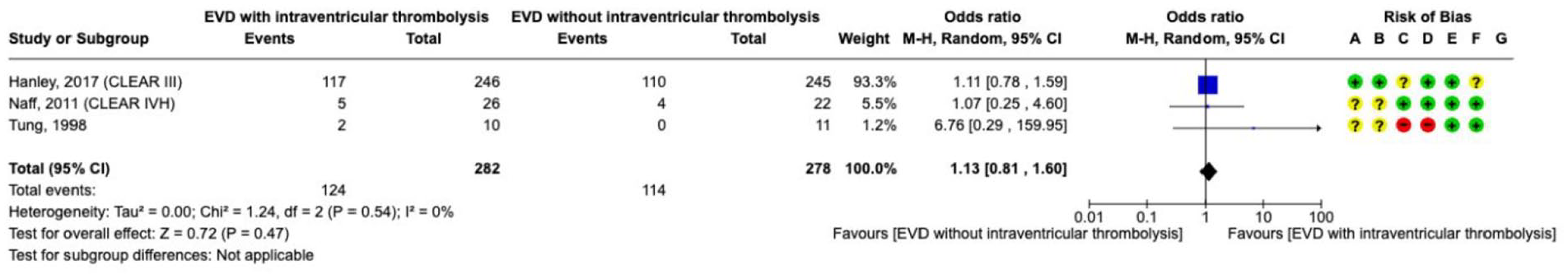
The effect on
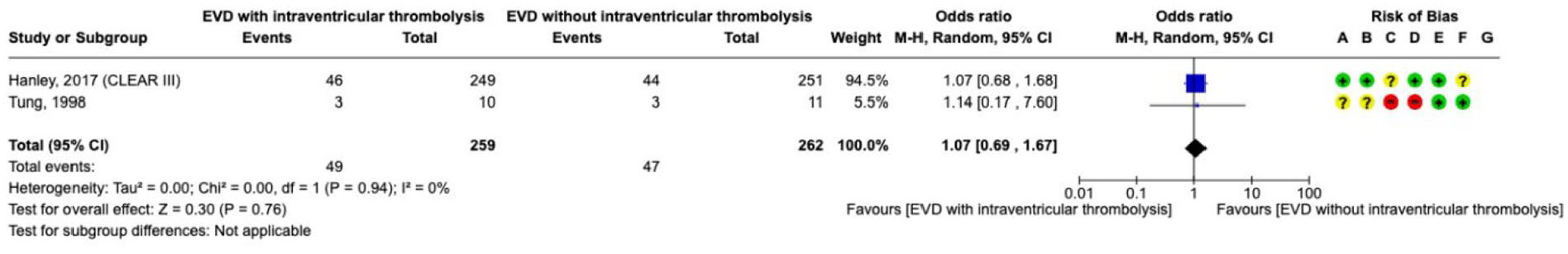
The effect on
Staykov et al. 153 conducted a randomised, open-label, parallel-group study of drainage plus fibrinolysis with or without lumbar drainage after proven patency of the third and fourth ventricles. The trial was stopped prematurely because of significant efficacy of the tested intervention. The primary endpoint, defined as permanent shunt placement (determined by a total of 3 unsuccessful EVD clamping attempts or the need for CSF drainage exceeding 14 days) was observed in 43% (7 of 16) of the control group versus 0% (0 of 14) of the intervention group (p = 0.007).
Additional information
The rationale to remove a larger amount of the intraventricular blood or to remove it faster by adding thrombolysis to EVD alone was investigated in few RCTs with a high risk of bias. In the largest trial, CLEAR III, patients with small to moderate ICH volume (<30 mL) obstructing hydrocephalus, and stable intraventricular haematoma were included. It demonstrated that the intraventricular administration of rt-PA could enhance haematoma resolution compared with saline. 152 Median intraventricular haemorrhage volume before enrolment was 21.8 mL. However, randomisation was done 52 h after the ictus, treatment started 3 h later and commenced only 2.5 days further after randomisation. The primary outcome, that is, good functional outcome (mRS 0–3) at 6 months, in patients receiving rt-PA versus saline was neutral (48% vs 45%; risk ratio 1.06, 95% CI 0.88–1.28). Mortality was reduced by 11% in patients receiving rt-PA versus saline, but there was an almost equivalent increase of 8% of patients with an mRS of 5. This reduction in mortality at the cost of patients surviving with severe disability, is considered a main obstacle for implementation of this rt-PA protocol. Patients should be further included in studies of thrombolysis with refined protocols. Earlier start, larger IVH volume, and less EOT volume may be one of the features to be adapted and analysed. In a secondary analysis, a more effective haematoma clearance (>80%) seemed to improve functional outcome.
Surgical removal of the intraventricular blood

Analysis of current evidence
Death and dependence (defined as mRS 4–6) at 1–6 months were prioritised as critical outcomes, and shunt dependence as an important outcome. Surgical removal of intraventricular blood mainly using neuro-endoscopy was investigated in five RCTs, however, with small sample size, and with a very serious risk of bias (Supplement 4.2.3 GRADE evidence profile).149–158 These studies were performed with endoscopy combined with EVD (with or without fibrinolysis), versus EVD alone (with or without fibrinolysis). All studies investigated mortality, with only three showing a trend towards lower death rate. Our meta-analysis also found a non-significant reduction in
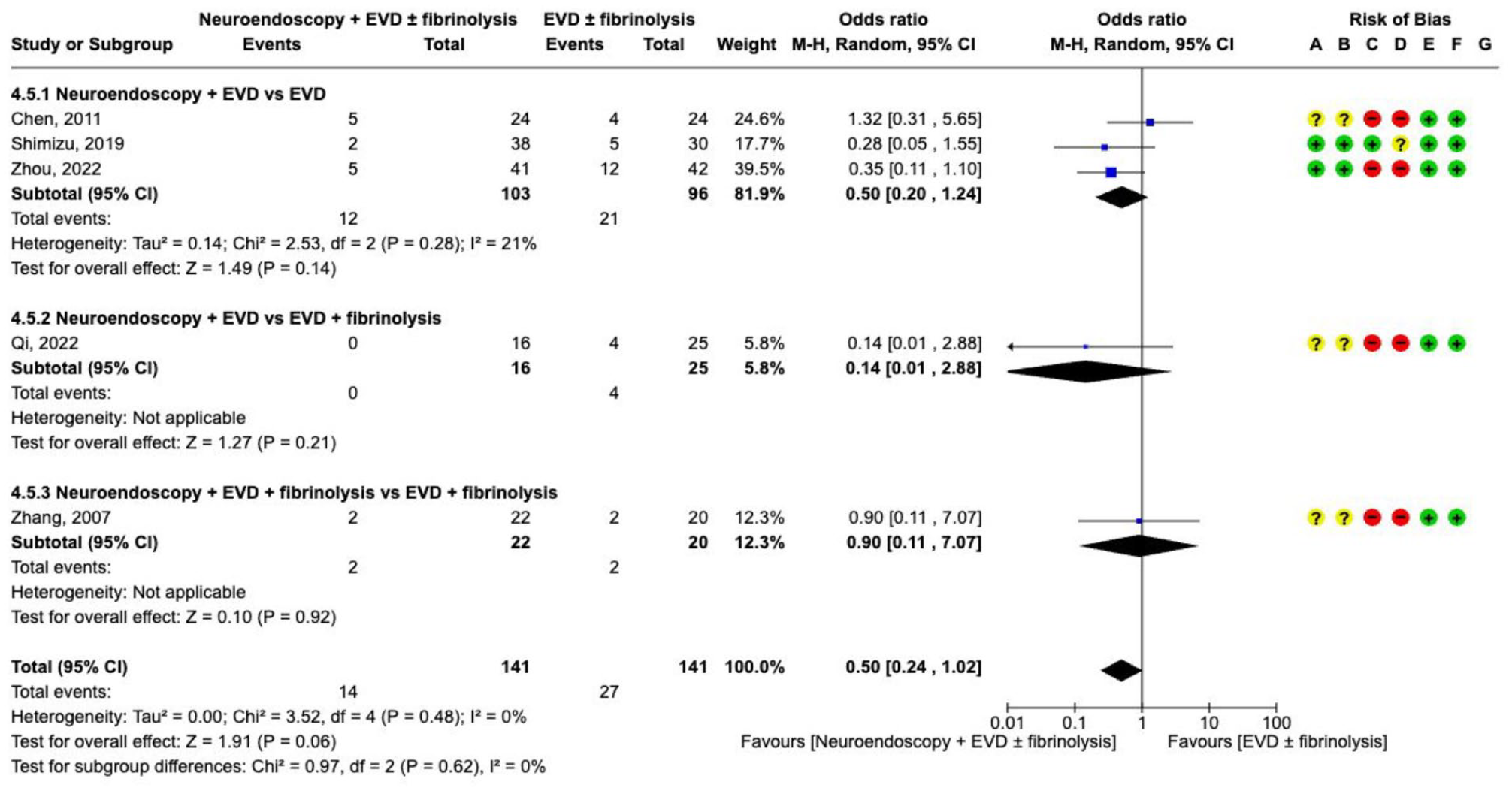
The effect on
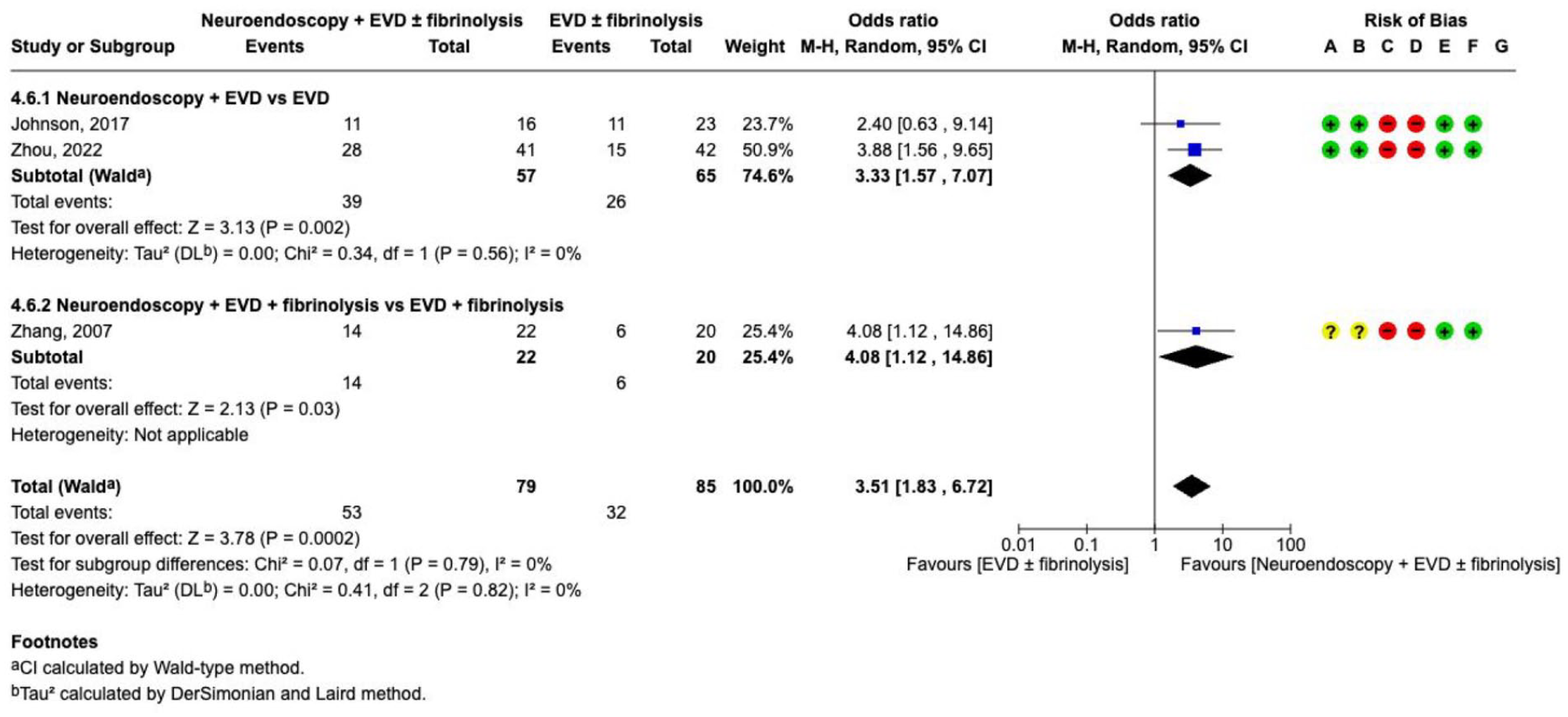
The effect on
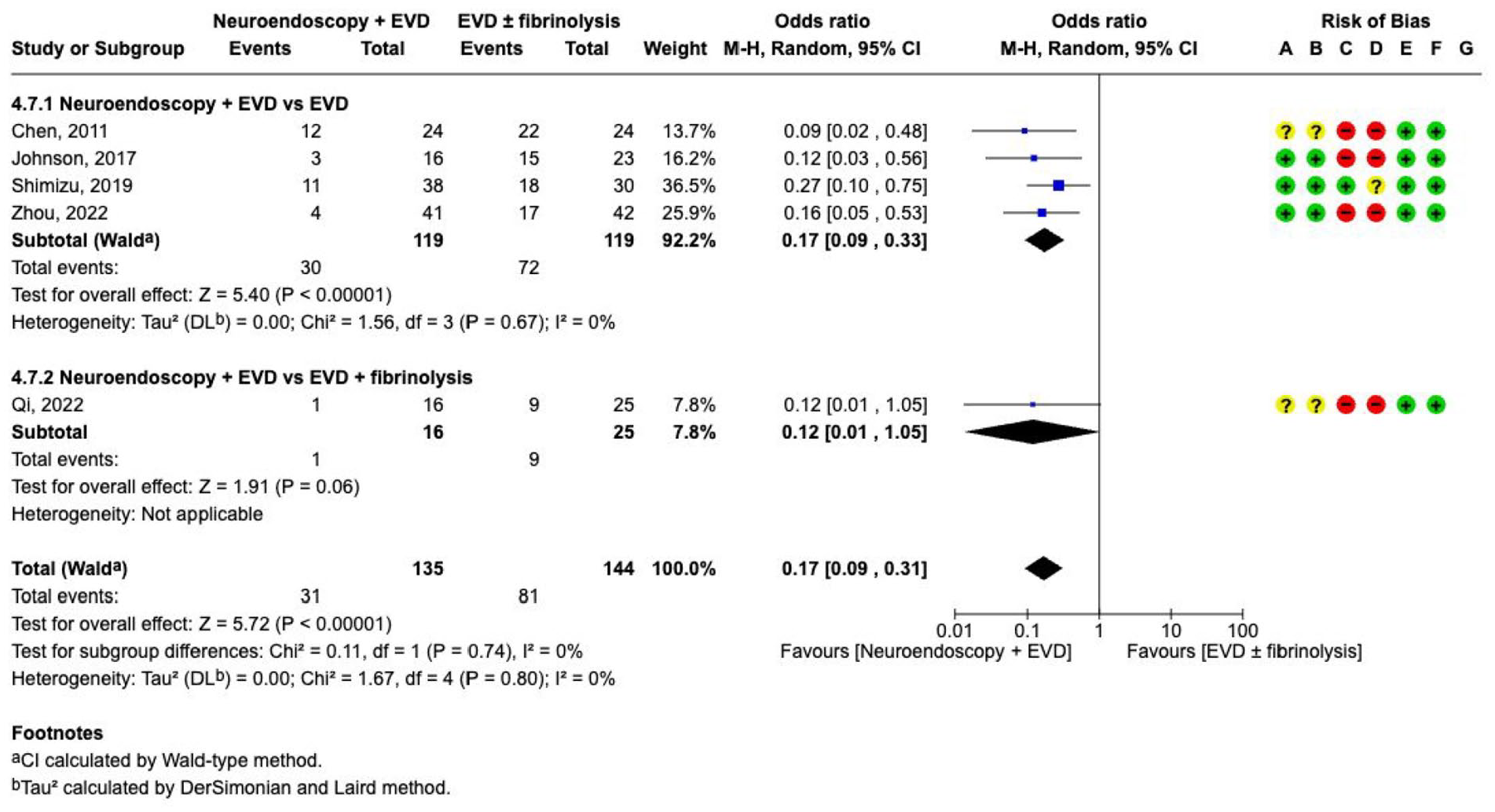
The effect on
Additional information
Intraventricular haemorrhage can result in secondary brain damage due to the presence of blood in the ventricles, the slow resolution and clearance rate, and the development of chronic hydrocephalus. The rationale behind removing a larger amount of the haematoma and to wash-out the ventricular blood was investigated in several RCTs. Despite these trials, the quality of evidence remains very low due to substantial risks of bias because of small sample sizes, and the heterogeneity of trials, which included different intervention and control groups. Therefore, an adequately powered RCT comparing endoscopic removal with EVD versus EVD alone is still lacking. There is also still considerable uncertainty regarding patient selection, intraventricular haematoma volume, ICH volume, timing, and for the surgical procedure itself, including whether to perform septostomy and attempt to clear the contralateral ventricle, or whether to enter the third ventricle.
Infratentorial ICH

Analysis of current evidence
The systematic literature search did not reveal any RCT comparing surgery with conservative management. Our recommendation is based on an IPDMA and expert opinion that surgical removal of the haemorrhage with or without EVD may be considered in patients who are deteriorating neurologically, have brainstem compression and/or hydrocephalus from ventricular obstruction.46,159–161
In the IPDMA of observational studies which included 578 patients with a propensity score matched cohort, the proportion of patients who
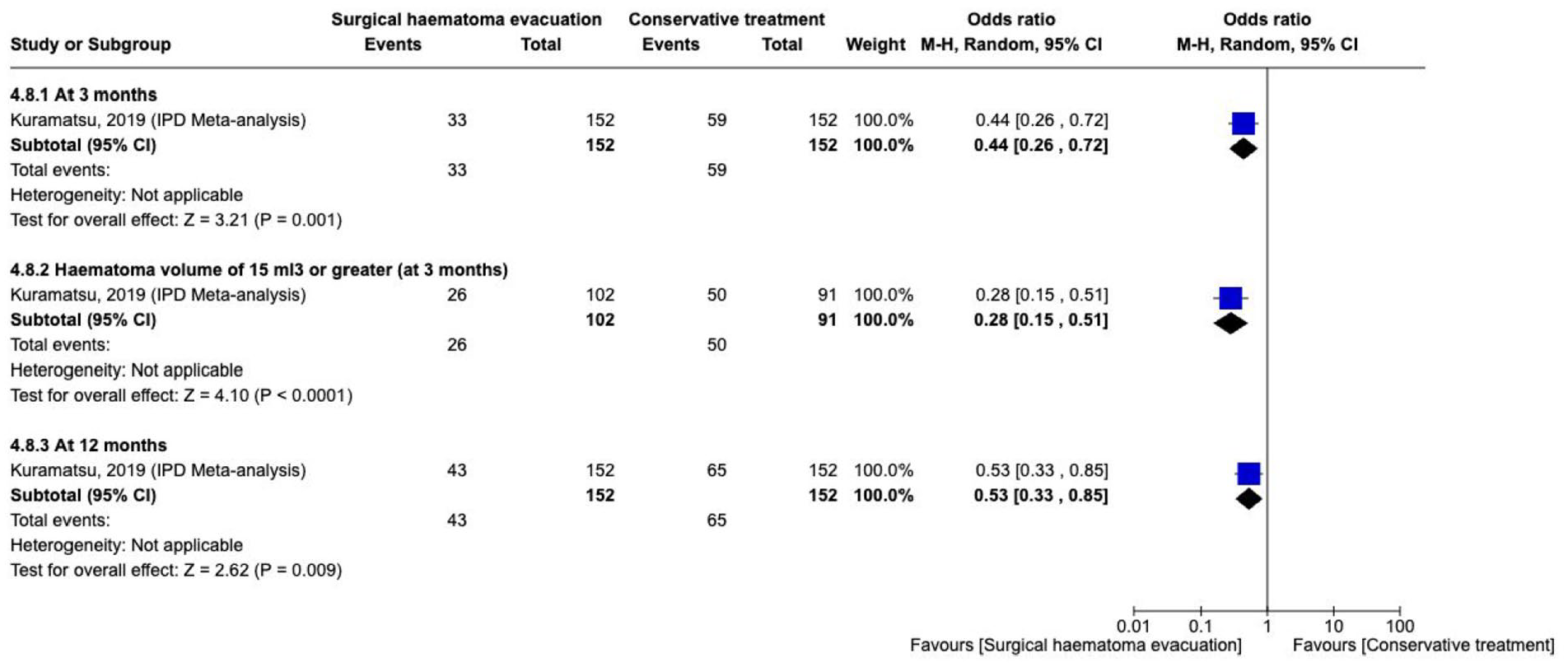
The effect on
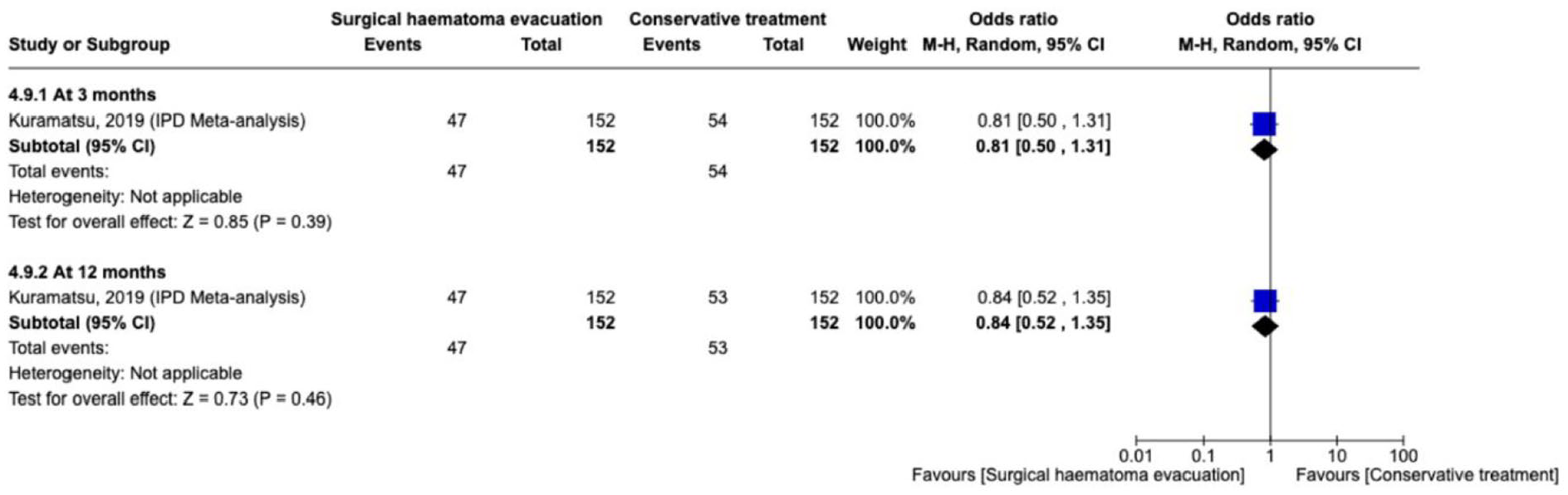
The effect on
Additional information
The long-term prognosis of patients who survive a first spontaneous cerebellar ICH is poor and comparable to that of patients who survive a first supratentorial ICH. 162 Because of the limited infratentorial space, cerebellar ICH is frequently associated with brainstem compression, hydrocephalus and herniation. Therefore, haematoma evacuation is often performed despite a lack of randomised evidence. The Canadian ICH best practice recommendations recommend that EVD should be placed in conjunction with haematoma evacuation in the setting of concurrent hydrocephalus. 159 For patients with cerebellar ICH and ‘clinical hydrocephalus’, EVD alone may be potentially harmful, especially if the basal cisterns are compressed, and EVD alone may be insufficient when intracranial hypertension impedes blood supply to the brainstem. 45
Prevention and management of complications
Single measures for prevention and management of complications
Physical measures to prevent venous thromboembolism

Analysis of current evidence
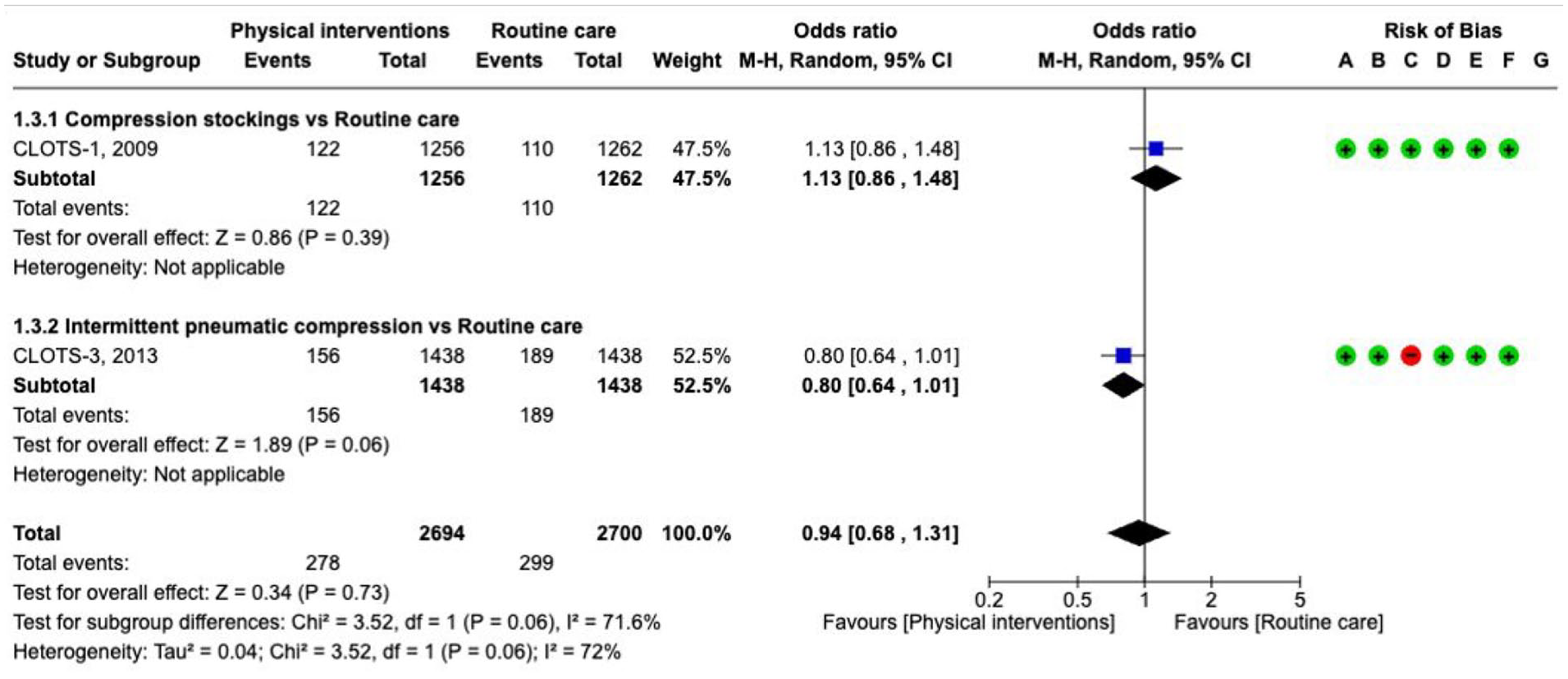
The guideline group graded the following outcomes as critical: venous thromboembolism, symptomatic pulmonary embolism (PE), symptomatic deep venous thrombosis (DVT) and death. Our literature search identified four RCTs that compared physical measures with standard care (CLOTS-1, CLOTS-3), or with different physical measures (CLOTS-2, VICTORIAh) for the prevention of venous thromboembolism in patients with stroke, including ICH (Supplement GRADE evidence profile for PICO 5.1.1).163–166 The RCTs included mixed numbers of ICH patients (N = 232, 9.2% (CLOTS-1); N = 365, 11.7% (CLOTS-2); N = 376, 13.1% (CLOTS-3); N = 151, 100% (VICTORIAh)). The CLOTS-1 study compared graduated compression stockings against ‘routine care’, while the CLOTS-3 study compared intermittent pneumatic compression (IPC) with ‘routine care’. However, uncertainty exists over the actual rate and distribution of additional low molecular weight heparin (LMWH) administrations among the ICH-subgroups in both intervention arms and standard care. The studies included in the meta-analyses are presented separately for the types of physical measures acknowledging the different effects of the ‘passive’ measure of ‘stockings’ compared with the ‘active’ measures of ‘intermittent pneumatic compression’. As demonstrated in the meta-analysis in Figures 50 and 51, physical measures compared with ‘routine care’ tended to prevent
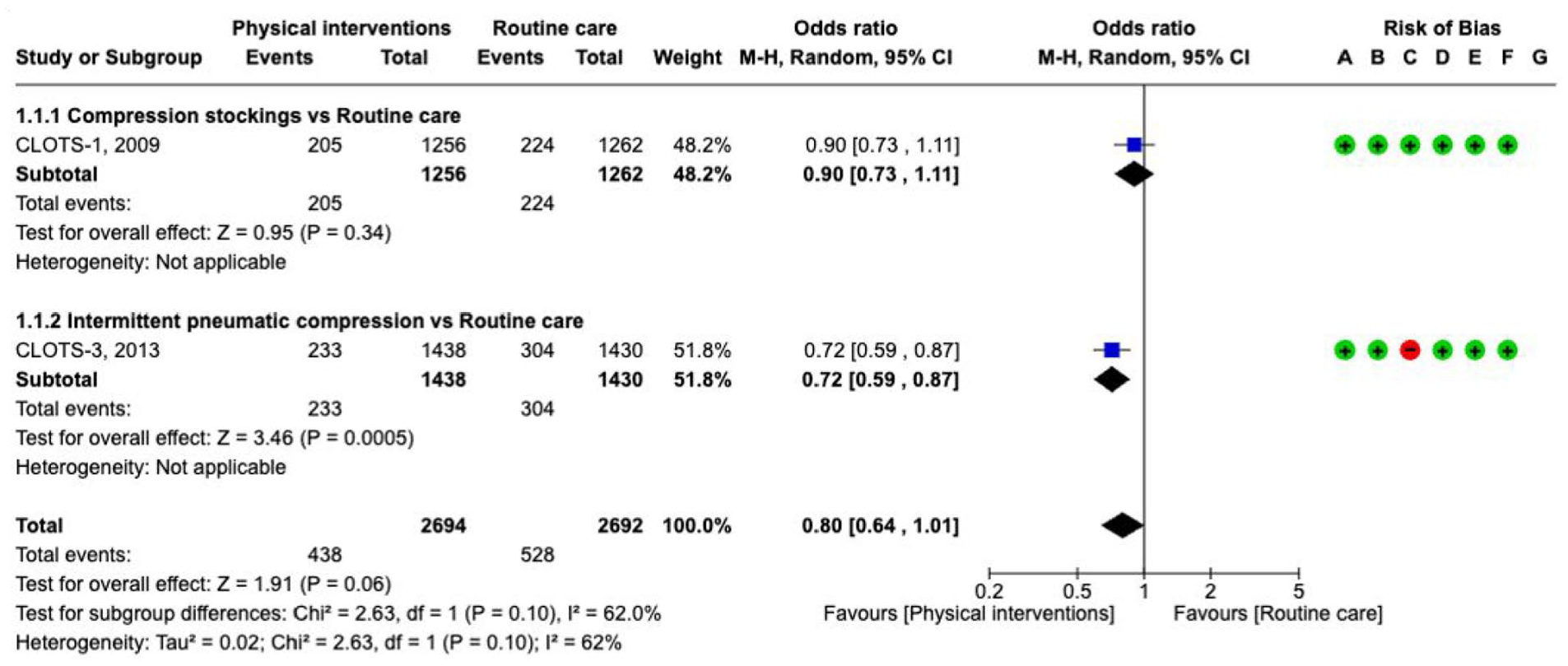
Prevention of
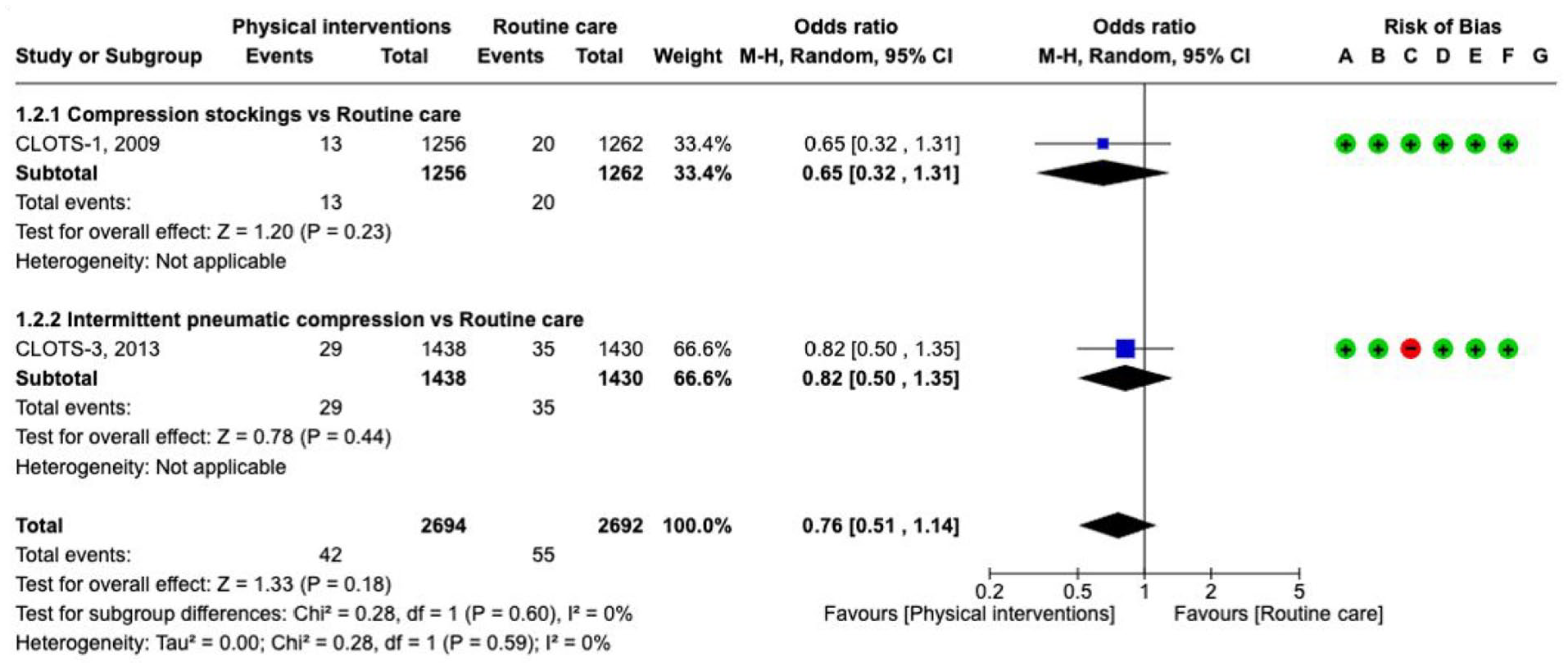
Prevention of
Prevention of
The VICTORIAh trial evaluated the efficacy and safety of intermittent pneumatic compression (IPC) in addition to elastic compression stockings versus elastic compression stockings alone in people with spontaneous or traumatic ICH. The trial reported no symptomatic DVT until day 10, and deaths before day 10 were not attributed to venous thromboembolism. 166 Additional IPC did not reduce the risk of asymptomatic venous thromboembolism in an adjusted analysis. Wearing of IPC was associated with more skin lesions, and the adherence was low. The CLOTS-2 study compared thigh-length stockings with below-knee stockings and found no significant difference in the adjusted odds ratio of any DVT or death. 164
Additional information
A randomised open-label trial (CIREA1) compared IPC with graduated compression stockings at 6 days in patients treated in the intensive care unit. 167 The trial included heterogeneous patient groups but reported relative risk for a composite outcome (fatal PE, symptomatic PE, symptomatic DVT or asymptomatic DVT) also separately for spontaneous ICH patients (N = 135, outcomes IPC plus graduated compression stockings 6.6% vs graduated compression stockings alone 11.9%, RR 0.55, 95% CI 0.19–1.66). Due to the limitation of a low statistical power, the results do not support superiority of the combination of IPC and graduated compression stockings.
Short-term antithrombotic therapy to prevent DVT/PE


Analysis of current evidence
The guideline group graded the following outcomes as critical: venous thromboembolism (VTE), symptomatic pulmonary embolism (PE), symptomatic deep venous thrombosis (DVT) death and recurrent ICH.
Our literature search identified four distinct RCTs that explicitly addressed the early prevention of venous thromboembolism by administering heparin or low-molecular weight heparin (LMWH) to the cohort of acute spontaneous ICH (Supplement PICO 5.1.2: description of single studies).168–172 All trials were included in a Cochrane Review before. 173 A synthesis of the findings reveals considerable heterogeneity amongst the included trials with regard to their inclusion criteria, interventions, and outcome assessments (prevention of death, symptomatic or asymptomatic deep vein thrombosis, and pulmonary embolism; Figures 53–55, quality of evidence table: see Supplement). Trial details are summarised in the Supplement.
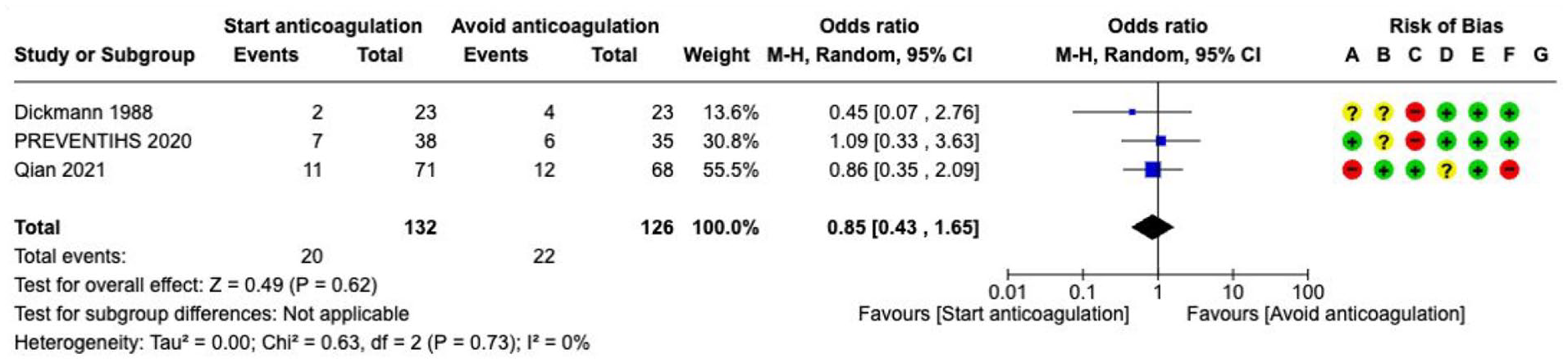
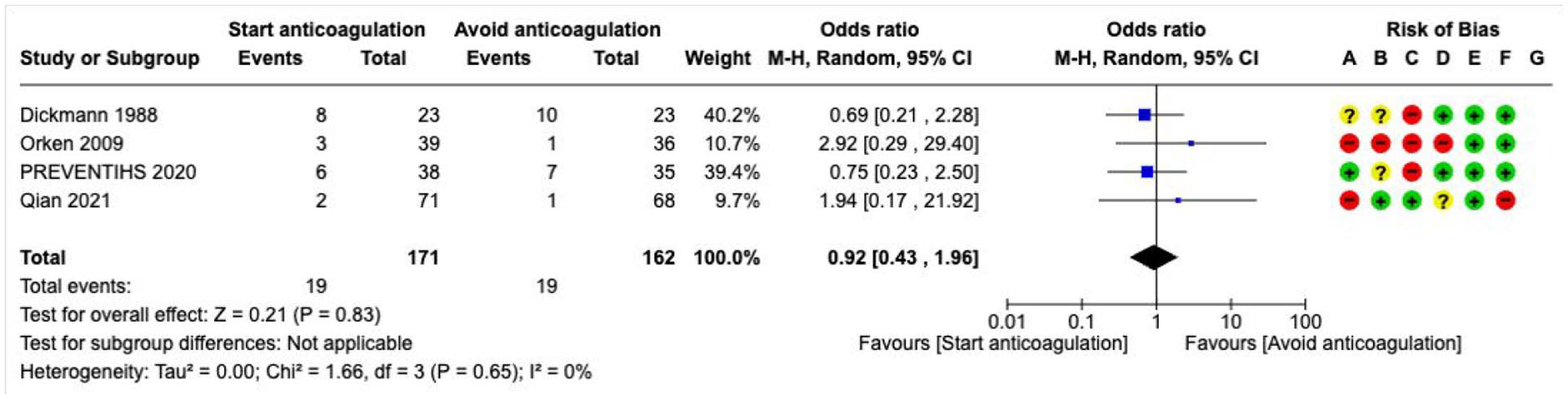
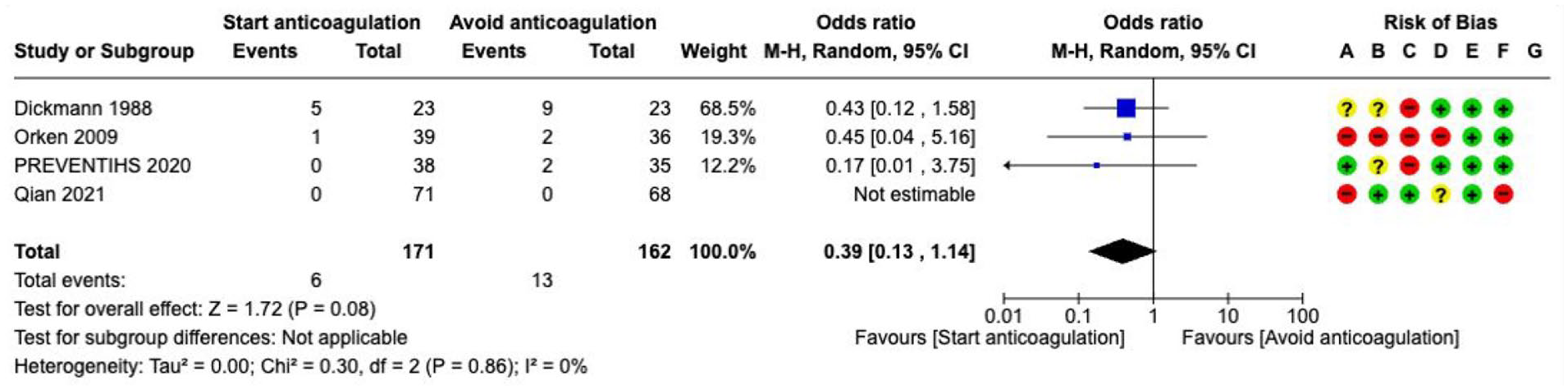
The Cochrane meta-analyses included 4 small RCTs investigating 171 patients treated with early heparin therapy, compared with later start,168,171 co-administration with graduated compression stockings, 169 or co-administration with IPC (PREVENTIHS). 170 Importantly, Boeer et al.168,172 reports on a small two-stage trial (N = 68), primarily comparing heparin (3 × 5000 IE s.c./d) starting at day 4 versus day 10, but secondarily adding a non-randomised third group, starting heparin at day 2. Notably, patients were allowed to receive a potentially prothrombotic high-dose of dexamethasone (starting with 48 mg/d) for treatment of cerebral oedema. Compared with patients with later start of heparin (day 4 vs 10 after diagnosis of ICH), earlier start at day 2 reduced the incidence of pulmonary embolism assessed by pulmonary perfusion scintigraphy at day 10. 164 Bleeding risk was not increased by earlier administration. 168 The non-randomised open-label extension group (commencing at day 2) by Boeer et al., added after knowledge of the outcomes in the intervention group (starting at day 4) in the trial by Dickmann et al. 172 introduces serious bias and was not included in the Cochranes analyses.
The meta-analysis showed no
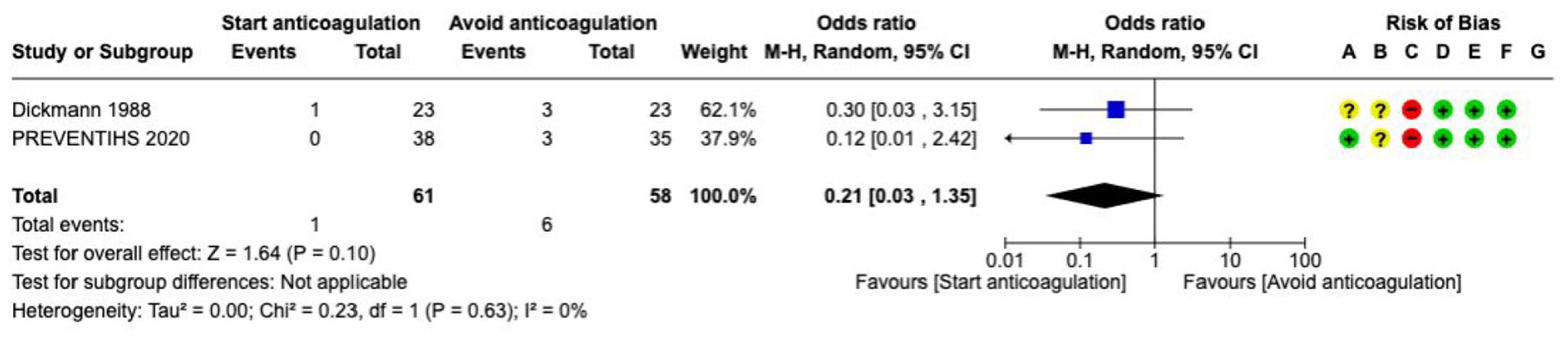
Risk of
Additional information
A retrospective large observational cohort study included 1702 patients with OAC-associated ICH and prospectively 1022 patients with non-OAC-ICH treated with low-dose heparin prophylaxis. 174 This study defined intracranial haemorrhagic complications as their primary outcome (occurring in 1.7% of patients, with no observed differences in crude incidence rates among patients with prior OAC or no OAC). However, the study did not include a control group of patients who did not receive heparin prophylaxis. Intracranial haemorrhagic complications were not found to differ when referenced to days with and without heparin prophylaxis. The study did not report the incidence of DVT or PE.
Temperature management

Analysis of current evidence
Active body temperature management was defined as the management of body temperature with predefined temperature thresholds triggering interventions (not limited to feedback-controlled systems) and predefined temperature measurement intervals to regulate temperature in adults with ICH. Where appropriate, the method – pharmacological intervention or physical – was indicated. We graded the following outcomes as critical: death and dependence.
Our literature search identified four RCTs evaluating active body temperature management in stroke or neurointensive care patients (any type of stroke or traumatic brain injury) including assessments of functional outcome (Supplement PICO 5.1.3: description of single studies).170–173 A further RCT was subsequently identified. However, none of the trials evaluated an intervention specifically in ICH patients. Therefore, due to the low number of ICH patients in the trials, and heterogeneity among interventions, we refrained from conducting a meta-analysis.175–178 The INTREPID trial (223 ICH; total evaluation sample 677 patients) compared automated surface temperature management to maintain normothermia with conventional, mainly pharmacological, fever treatment. 179
No significant difference in functional outcome or death was demonstrated by any of the identified active body temperature management trials (Supplement: description of single studies). The management of body temperature through mainly pharmacological interventions was a component of care-bundle trials, elaborated upon in detail in PICO 5.2. It is therefore recommended that this approach be implemented in conjunction with additional elements of a care-bundle.
Additional information
Hyperthermia subsequent to ICH has been associated with worse outcome in observational studies.180,181 Several preclinical studies have demonstrated that therapeutic hypothermia can reduce perihaematomal oedema and blood-brain barrier breakdown as well as improve neurological outcome, but there is a lack of evidence from clinical trials. The fate of the target temperature management (TTM)-ICH trial, started in 2013 (NCT01607151), a phase 1/2 RCT comparing the effect of 72 h of TTM aiming for hypothermia (32–34°C) versus aiming for normothermia (36–37°C) in spontaneous ICH patients is unclear. The CINCH trial, investigating hypothermia in large ICH, has been terminated without available results (ISRCTN28699995). 182
Blood glucose control

Analysis of current evidence
The guideline group graded the following outcomes as critical: death and dependence, and hypoglycaemia. As glucose control was not considered in the 2014 ESO guideline, the search strategy for this PICO was expanded to include studies published before 2013. We identified one RCT including patients with stroke and traumatic brain injury. The single-centre trial randomised 81 mechanically ventilated neurologic ICU patients (18 (22%) ICH patients) to intensive blood glucose control (range of 80–110 mg/dL (4.4–6.1 mmol/L)) or to conventional control (⩽150 mg/dL (⩽8.3 mmol/L)). 183 The trial found a trend towards more deaths in the intensive treatment arm, no difference in functional outcome, and more severe hypoglycaemias with the intensive treatment. 183 We did not identify any trial comparing glucose control versus no glucose control. Importantly, glucose management was also part of care-bundle trials, elaborated in detail in PICO 5.2.
Additional information
Indirect evidence against use of intensive blood glucose management is derived from a RCT in ischaemic stroke. The SHINE trial compared intensive with standard treatment of hyperglycaemia and looked at functional outcome in patients with acute ischaemic stroke. There was no benefit of intensive (target 80–130 mg/dL (4.4–7.2 mmol/L)) versus standard glucose treatment (80–179 mg/dL (4.4–9.9 mmol/L)) over a period of up to 72 h. 184 More hypoglycaemic events occurred in the intensive treatment arm (11.2% vs 3.2%).
Intracranial pressure (ICP) monitoring


Analysis of current evidence
The guideline group graded the following outcomes as critical: death and dependence. Our literature search revealed one RCT investigating the effect of intracranial pressure (ICP) monitoring on functional outcome and death (Supplement GRADE evidence profile for PICO 5.1.5). 185 The single-centre RCT from China included 90 patients with ICH caused by hypertensive microangiopathy. Patients who were randomised to ICP measurement received an intraventricular ICP-sensing probe, which was inserted in the anterior horn of the lateral ventricle. Conversely, a conventional external ventricular drain was inserted contralaterally. ICP elevations were defined as values above 25 mmHg. The primary outcome, a composite of incidence rate of HE and brain herniation, was assessed within 1 month after study onset. While the incidence of HE was similar among groups (33% with ICP-monitoring vs 39% without, p = 0.76), patients with ICP-monitoring developed less frequently brain herniation (11% vs 21%, p = 0.04; patients showing brain herniation received immediate haematoma evacuation). The death rate at 6-months was 6.5% in cases where ICP-monitoring was employed, as compared with 9.1% in cases where it was not (p = 0.04).
Adverse events due to the insertion of the ICP-probe were not observed. It is notable that the trial was conducted in patients that in most circumstances would not have required ventricular drainage, given that the mean ICH volume was modest (~20 mL), patients not necessarily required deep sedation (mean GCS in the intervention group, 10 ± 1) and intraventricular haemorrhage was not present at enrolment in all patients. The study did not explore whether sequential brain imaging or clinical assessments of signs of increasing intracranial pressure could have served as indicators with similar test accuracy compared with the invasive measurements.
Additional information
A large multicentre prospective observational study conducted among 146 sites in 42 countries included 2395 patients with acute brain injury (587 ICH patients). These patients received either invasive ICP monitoring or not, according to the local physician’s indication. 186 The majority of patients with ICH had ICH-volumes > 30 mL (69%), a GCS of ⩽8 (85.7%) but pupils were mostly both reactive (one unreactive 13%, both unreactive 26%). The primary rationale for invasive ICP monitoring in these patients was deterioration in clinical status (64%), with an intraventricular device (148/276, 54%) being most commonly used. The 6-month death rate was lower in the ICP-monitoring cohort (35% (441/1317) vs 49% (517/1049), p < 0.0001), and unfavourable neurological outcome was likewise lower (extended Glasgow Outcome Scale score < 5; 60% (733/1220) vs 65% (633/982), p = 0.039). In the subgroup of patients with intracranial haemorrhage and bilateral reactive pupils, ICP monitoring was associated with reduced hazards of death (HR 0.57, 95% CI 0.38–0.87), with similar findings in those with one unreactive pupil (HR, 0.34, 95% CI 0.22–0.53). However, ICP monitoring was not associated with better neurological outcome (OR 0.83, 95% CI 0.41–1.68; p = 0.6077). 187 ICP-catheter replacement was frequently needed (22%), with misplacements being a frequent reason (1 out of 10).
Anti-inflammatory treatment

Analysis of current evidence
The guideline group graded the following outcomes as critical: Death/dependence, and as important: perihaematomal oedema increase.
We identified RCTs investigating nine substances of eight categories with anti-inflammatory properties in people with acute ICH. However, no RCT comparing one agent versus another could be identified (Supplement GRADE evidence profile for PICO 5.1.6). Therefore, the available evidence is presented according to the main substance category investigated. All trials examined short-term anti-inflammatory treatments after ICH, despite pathophysiological considerations of a ‘delayed’ anti-inflammatory effect on oedema development and recovery.
Anakinra
Anakinra is an interleukin-1 receptor antagonist, licensed for treatment of rheumatological diseases. A trial (BLOC-ICH) assessed the effect of anakinra (100 mg s.c. within 8 h of onset, followed by 5 × 12 h 100 mg s.c.) in mitigating periheamatomal oedema in acute ICH, in addition to its impact on clinical outcomes. 188 The trial was stopped prematurely due to slow recruiting. Enrolling 25 of the initial attempted 80 patients, it remained underpowered. Oedema extension distance, a measure used to describe perihaematomal oedema, did not differ between the anakinra and placebo-arm after 72 h. Numerically more people had a poor outcome in the anakinra group at 3-months (58% (7/12) vs 30% (3/10), OR 3.3, 95% CI 0.6–19.3). 188
Celecoxib
Experimental data have shown a reduction of perihaematomal oedema, inflammation and cell death by celecoxib, a selective inhibitor of cyclo-oxygenase 2 (COX-2). A small clinical trial randomised 44 Korean adults with ICH (<24 h after onset) to either celecoxib (N = 20; 400 mg BD) or standard treatment (N = 24). 189 Primary endpoint was the change in perihaematomal oedema (⩾20%) from day 1 to day 7. While no absolute difference in perihaematomal oedema volume was observed at day 7 (23 mL with celecoxib vs 24 mL with standard treatment), a significant shift to reduced expansion of oedema in the celecoxib group was reported. 189 No differences in functional outcome and death were reported.
Citicoline
Citicoline showed a reduction of ischaemic lesions associated with ICH in experimental studies. 190 In a small RCT, ICH patients within 6 h of onset (mean volume ~ 24 mL) were enrolled to receive citicoline (N = 19; 1g/12 h for 2 weeks) or placebo (N = 19). 191 The primary endpoint was the rate of adverse events, which was not different between the groups. At week 12, five patients in the citicoline group (6.7%) and one patient in the control group (27.8%) achieved a mRS of 0–2, but the baseline ICH severity tended to be lower in the citicoline group (median NIHSS 9 vs 15).
Corticosteroids
We identified six RCTs (n = 429) investigating the effects of corticosteroids in people with acute ICH with regard to clinical and radiological endpoints.192–197 Five of the RCTs had been previously published and were included in a Cochrane analysis in 2005. Building upon this analysis, the meta-analysis was updated to incorporate results from a sixth trial, published in 2008.
197
With the exception of the trial by Poungvarin published in 1987, all trials contain moderate to severe biases. Death rate could be assessed in all trials, however with varying assessment points (ranging from discharge to 6 months). The summary effect indicates a higher
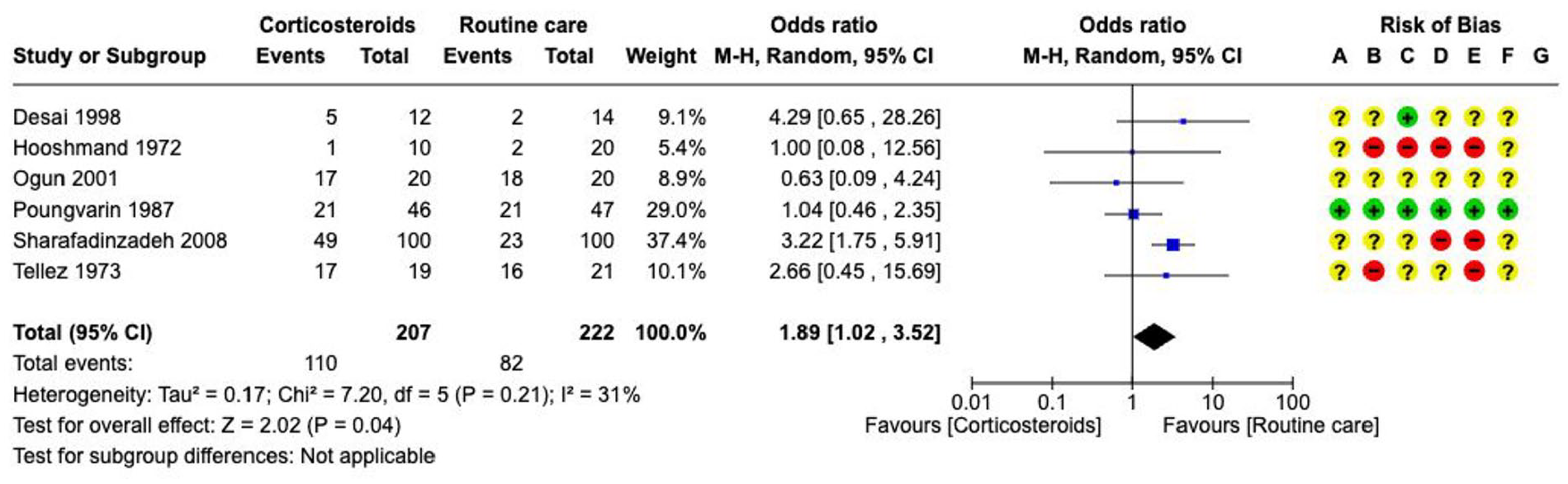
Effect on
Deferoxamine
Deferoxamine, a drug capable of binding iron or aluminium, has shown some neuro-protective and anti-inflammatory effects in preclinical research. We identified two trials investigating deferoxamine infusion in spontaneous ICH. A small (N = 42), underpowered trial with regard to clinical outcomes, reported smaller oedema volumes in the experimental group, but methodological concerns were raised by the reported mean haematoma volume declines of >10 mL within only 14 days after onset.
198
The i-DEF trial was a larger, multicentre placebo-controlled double-blind randomised trial, assigning 144 patients with supratentorial ICH to the deferoxamine mesylate infusion (32 mg/kg/day for 3 days) group and 147 to placebo.
199
Primary outcome was mRS 0–2 at day 90. It was achieved by 34% of patients in the deferoxamine group, and 33% in the placebo group, a non-significant difference. There was also no difference in
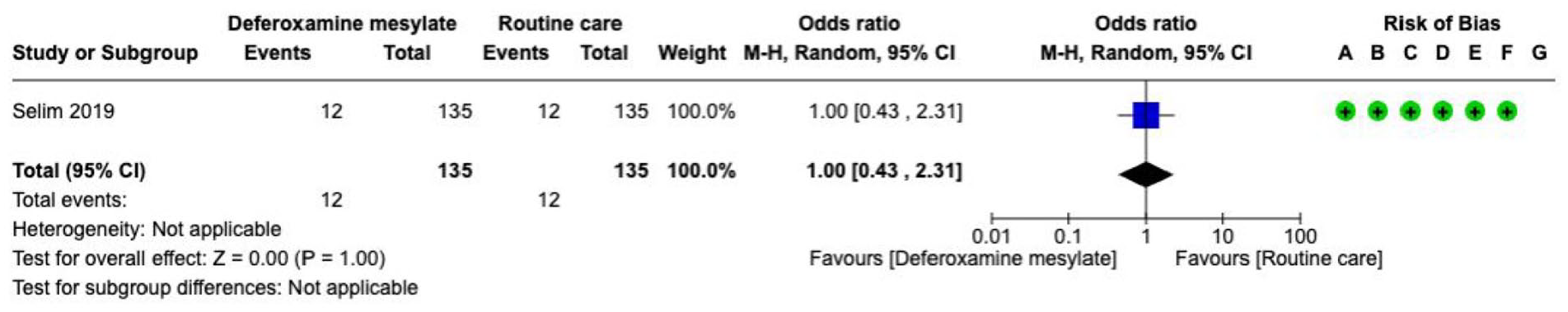
Effects on
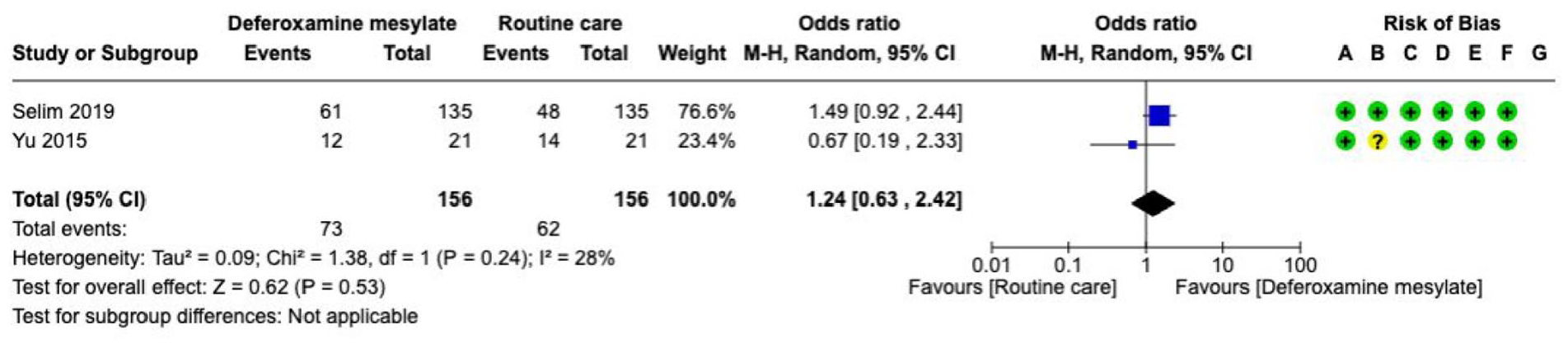
Effects on
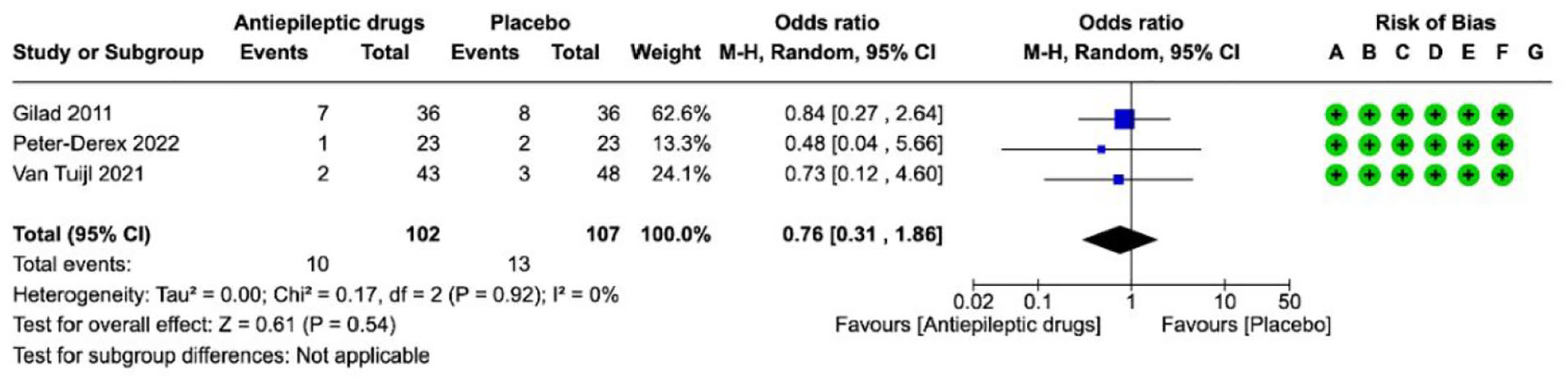
Effects on the occurrence of
Edaravone
A number of small RCTs have been conducted in China to evalute the antioxidant edaravone (sample sizes 58–190). A recent systematic-review and meta-analysis identified 38 RCTs published until 2021 including 3454 patients with ICH in whom edaravone treatment (30–60 mg/d) was started within 7 days of onset (continued until day 14–30). 200 No further trials were identified, that had not already been included in the aforementioned meta-analysis. The meta-analysis demonstrated that edaravone treatment was not associated with reduced mortality (RR 0.51, 95% CI 0.11–2.32). 200 Long-term functional outcome was not assessed. The validity of reported beneficial effects on neurological deficits, activities of daily living, and haematoma volume is constrained by the partly severe biases inherent in the RCTs.
Fingolimod
Fingolimod, a sphingosine-1-phosphate receptor modulator, inhibits lymphocytic infiltration into the central nervous system. In the EU, it has been approved for relapsing-remitting multiple sclerosis. In a small RCT in 23 patients with acute supratentorial ICH, fingolimod was tested to reduce the inflammatory reaction associated with ICH and consequently reduce perihaematomal oedema (volume 5–30 mL). 201 Eleven patients were randomised to fingolimod (0.5 mg orally OD for 3 days, starting 1 h after the baseline CT scan). Despite similar baseline characteristics, GCS scores were better in the fingolimod group at day 14 (GCS > 15, 100%; control-group 58%, p = 0.04). Likewise, more patients achieved a mRS 0–1 in the fingolimod-group at day 90 (63% vs 0%, p = 0.001). Perihaematomal oedema was smaller in the fingolimod group. 196 The replication of these findings is currently pending.
Minocycline
Minocycline, a tetracycline antibiotic, iron chelator and inhibitor of matrix metalloproteinases has been investigated as a neuroprotectant in acute ischaemic stroke and in experimental ICH models. Two small RCTs investigated minocycline in people with acute ICH. The MACH trial randomly assigned eight patients to receive minocycline (single-dose 400 mg i.v., followed by 400 mg orally for 4 days), and eight patients to receiving standard treatment. 202 Expected serum concentrations were reached, but no differences in inflammatory biomarkers, radiological makers, or functional outcome (mRS at day 90) were observed. A second trial (N = 20) randomised ICH patients to intravenous minocycline (10 mg/kg body weight, max 700 mg/d) for 5 days, or placebo. 203 In 1/10 patients the drug had to be stopped due to adverse events, and no differences in radiological and functional outcome parameters were observed. 203
Panax notoginseng
Screening of references revealed studies investigating panax notoginseng (a Chinese herb with attributed anti-inflammatory properties), exclusively conducted in China. A meta-analysis including 20 studies encompassing 1891 patients with intracranial haemorrhages was identified. The aetiology of bleedings was found to be heterogenous, including subarachnoid haemorrhages. All studies were of small sample sizes, with severe bias and heterogenous outcome assessments. With regard to functional outcome, the ‘neurological deficit score’ was reported to be lower in the panax notoginseng group than in the standard treatment group. Death rate (overall < 10%) was reported to be lower with panax notoginseng, but data presented in other parts of the analyses were conflicting, indicating instead a higher death rate. 204
Reactive oxygen species scavengers
Reactive oxygen species (ROS) are hypothesised to play a role in the development of perihaematomal oedema. A randomised pilot study from Korea examined the impact of administering two ROS scavengers simultaneously (N-acetylcysteine 2000 mg/d and selenium 1600 µg/d i.v.) on the change of perihaematomal oedema volume in people with ICH. 205 The trial which compared ROS scavenger (N = 57) with placebo (N = 66) revealed several limitations, including the heterogeneity of disease entities reflected by the inclusion of patients with haemorrhage due to intracranial malignancies (N = 24) and haemorrhagic infarctions (N = 5), the allowance of surgery, and the exclusion of patients from the active treatment group, who did not reach serum efficacy levels. While the haemorrhage volumes were comparable between groups, perihaematomal oedema volume was significantly different after 14 days (ROS, 21.9 ± 17.6 mL vs placebo, 30.7 ± 32.4, p < 0.01). No difference in 30-day functional outcome was reported. 205
Additional information
Ongoing clinical trials and further substances:
Anti-seizure medications


Analysis of current evidence
This PICO constitutes an update to the 2017 ESO guideline for the management of post-stroke seizures and epilepsy. The 2017 ESO guideline referred to post-stroke seizure occurrence, or recurrence, as well as to functional outcome, and mortality as critical or important outcomes.
207
An updated literature search was performed and a total of 3 RCTs (217 participants) were identified which compared primary preventive treatment with anti-seizure medications (valproate, diazepam and levetiracetam) to placebo after spontaneous ICH (Supplement PICO 5.1.7: description of single studies, Supplement GRADE evidence profile for PICO 5.1.7).208–210 No difference in the
There was no difference in
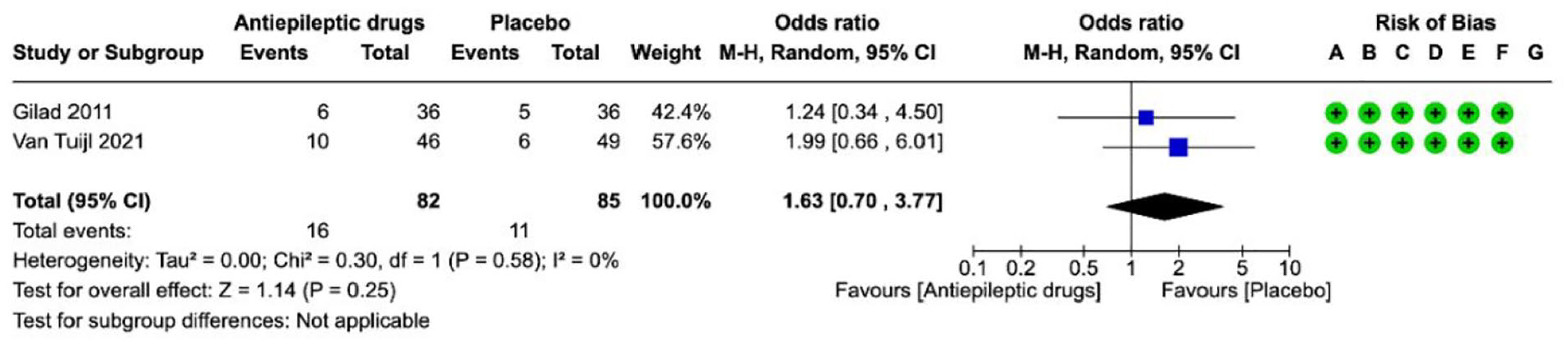
Effect on
Additional information
A meta-analysis of mostly observational cohort studies including 3241 participants failed to identify an association between the use of preventive anti-seizure medications and poor outcome (OR 0.99, 95% CI 0.66–1.49). 211 Observational trials have reported acute symptomatic seizures in 8%–16% of patients following ICH, with an increased risk observed in more severe strokes and cortical involvement.212–214 Continuous EEG studies have demonstrated the occurrence of clinical and electrographic seizures combined in up to 31% of critically ill ICH patients,215,216 though this rate may not be applicable to a general ICH population. Observational studies have shown conflicting results regarding the association between acute symptomatic seizures and functional outcome/mortality after ICH.212,213,217 Importantly, acute symptomatic seizures may be a marker of more severe ICH and not an independent risk factor for worse outcome. The recurrence rate of clinical seizures in the acute phase of ICH has been shown to be low (13%–15%).212,218 Nevertheless, secondary prevention in such cases is common in clinical practice, likely to reduce the risk of clinical worsening in the acute setting. 207
The underlying concept of this approach likely is based on pathophysiological considerations such as increased neuronal excitotoxicity, peri-infarct depolarisations, and inflammatory response. 219 These are considered to be risk factors for acute recurrence of epileptic seizures, and therefore clinicians may tend to administer anti-seizure medication. 207 There is currently no evidence to support this practice, and we encourage withdrawing anti-seizure medication after the acute phase.
Ongoing studies
Presently, two RCTs are ongoing to evaluate the antiepileptogenic effect of early antiseizure medications following ischaemic stroke or ICH. One phase II, multicentre, randomised, double-blind, placebo-controlled study is evaluating whether treatment with eslicarbazepine acetate for one month after stroke can prevent unprovoked post-stroke seizures (EudraCT; number 2018-002747). Recruitment has now ceased and follow-up is in progress. 220 Another phase II, randomised, double-blind, placebo-controlled trial (ACTRN12618001984280) is under way to evaluate the antiepileptogenic efficacy with perampanel compared with placebo in preventing late post-stroke seizures.
Care bundles


Analysis of current evidence
The guideline group graded the following outcomes as critical: death and death and dependence. The systematic literature search revealed three RCTs investigating pre-defined care bundles, that is, combination of specific treatment elements, in adults with ICH.80,221,222 Two of the RCTs (PRECIOUS, QASC) enrolled people with ischaemic stroke or ICH and data did not allow further differentiation (Supplement PICO 5.2: description of single studies).80,221 Therefore, details of the PRECIOUS and QASC trial are found in the additional information.80,221 Details of the studies including a discussion of their limitations are summarised in the Supplement.
INTERACT-3, a stepped-wedge cluster randomised trial, focused on the treatment of abnormal variables for 7 days in adults with acute ICH (admission within 6 h of onset, Supplement GRADE evidence profile for PICO 5.2). The care-bundle specified early intensive blood pressure treatment (target < 140 mmHg systolic), glucose control (target 110–141 mg/dl (6.1–7.8 mmol/L), non-diabetic; 141–180 mg/dl (7.8–10.0 mmol/L), diabetic), treatment of elevated body temperature (target ⩽ 37.5°C) and reversal of anticoagulation (vitamin K-antagonists, target-INR < 1.5 within 1 h). The common odds ratio of achieving a poor functional outcome (mRS 3–6) was lower with care-bundle treatment (cOR 0.86; 95% CI 0.76–0.97; p = 0.015). 80
The INTERACT-3 trial showed a higher chance of a

Effects on
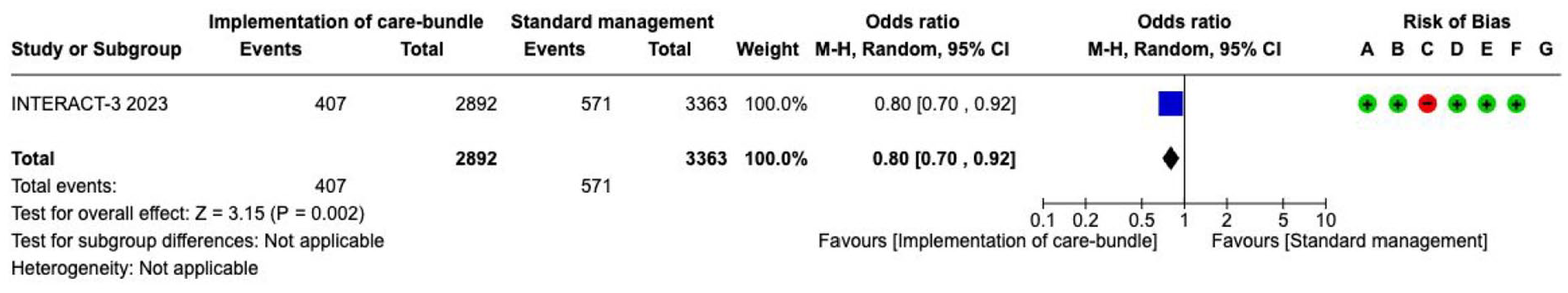
Effects on
Additional information
The QASC trial studied a combined approach of prevention of complications and treatment of abnormal physiological values and was published in 2011. In this cluster-randomised trial conducted in Australia, ischaemic stroke and ICH patients were enrolled. No separate analysis of the small subgroup of ICH patients (N = 51) is available. In the overall study population (N = 1009 in outcome analysis), death and dependence (mRS ⩾ 2) were more frequently observed at 3-months in the control group compared with the intervention group (259/449, 58% vs 236/558, 42%, p = 0.002). Death occurred in 5.3% in the control group versus 3.8% in the intervention group. 220
The ABC-ICH study was a large non-randomised interventional observational study, which measured effects before (N = 353 patients), during (N = 266) and after implementation (N = 241) of a care-bundle which consisted of three elements: (1) Reversal of orally anticoagulated patients (vitamin K+PCC for VKA, idarucizumab for dabigatran-associated ICH, both within 90 min after arrival); (2) invasive blood pressure lowering (SBP target 130–140 mmHg within 6 h after onset, if SBP > 150 mmHg); (3) immediate neurosurgical referral in patients with premorbid mRS ⩽ 2 if GCS < 9, posterior fossa ICH, haematoma volume > 30 mL or obstructed third/fourth ventricle. 51 In the intervention period, the adjusted OR of 30-day mortality was lower compared with the pre-intervention period (0.62, 95% CI 0.38–0.98, p = 0.003). The observed benefits were sustained in the post-intervention period. As a ‘side-effect’, less patients received an early do-not-resuscitate order, compared with the pre-intervention period. This non-pre-specified study effect contributed to more than 50% of the associations between before and after the introduction of the care bundle and reduced mortality.
Briefly, the PRECIOUS trial, which was prematurely terminated in 2022, focused on the prevention rather than treatment of certain conditions (fever and infection). The study included 1471 patients (ischaemic stroke: N = 1250, ICH N = 201, other N = 20) in the intention-to-treat analysis, who were randomly assigned in a multifactorial design to receive metoclopramide (or not), ceftriaxone (or not) and paracetamol (or not). The primary outcome (mRS at day 90) did not differ between the prophylactic medication groups and the controls. 222 The PRECIOUS trial was not included in the meta-analysis due to its study design.
Current studies investigating the application of care-bundles in ICH are: MAX-ICH Pilot Trial (NCT06648369, planned sample size N = 50), and I-CATCHER (NCT06429332, N = 3500).
Secondary prevention
Blood pressure


Analysis of current evidence
The guideline group graded the following outcomes as critical for blood pressure control after ICH: recurrence of any stroke, ICH recurrence and major adverse cardiovascular events (MACE). In ICH management, studies have evaluated the treatment of blood pressure (BP) in the acute phase (⩽7 days of onset). However, our literature search did not identify any RCTs that compared targeted BP control or the use of a specific antihypertensive agent with standard care after the acute phase (i.e. >7 days) following ICH.
For the
In a post-hoc analysis of ICH patients (n = 189),224,225 there was an overall low rate of recurrent stroke, with lower absolute numbers in the intensive blood pressure control group compared with standard care (intensive 4/101 (4.0%) vs standard 9/88 (10.2%)). Overall, in the meta-analysis, a significant between-group difference in the
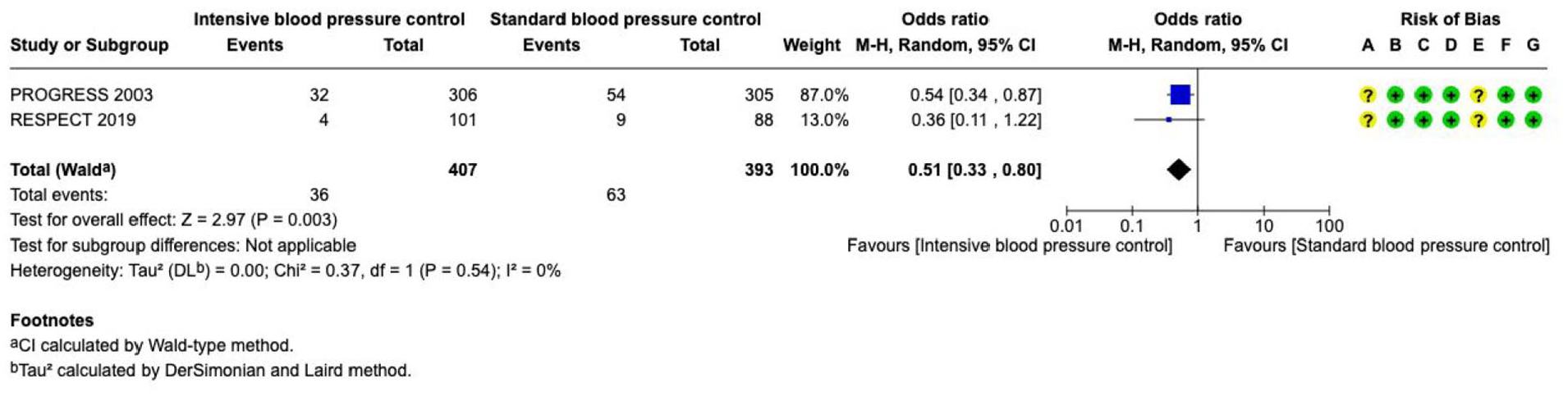
Effect on
Regarding the outcome
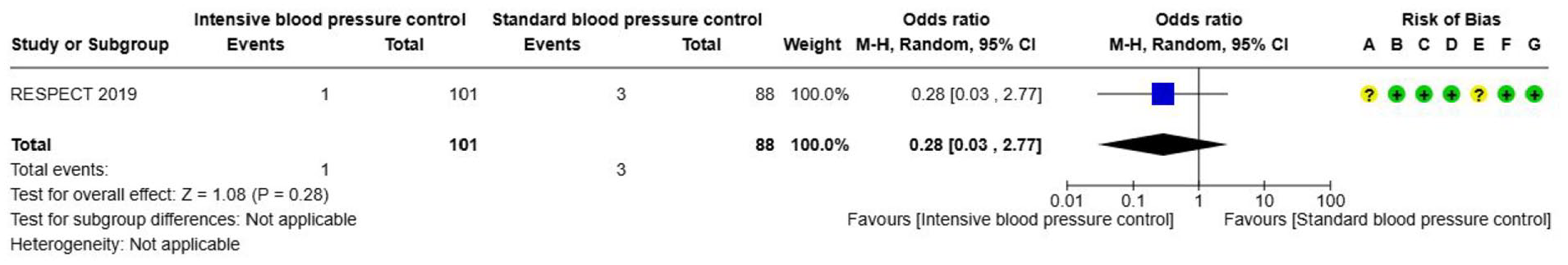
We identified two observational studies comparing individuals with prior intracerebral hemorrhage (ICH) who received or did not receive antihypertensive treatment. The first study, a retrospective cohort analysis from Denmark, examined ICH recurrence in 15,270 patients with first-ever ICH, using data from the Danish national health registry with a minimum follow-up of 6 months. 226 Among patients receiving antihypertensive treatment (n = 5854), the recurrence rate was 10.8% (636 cases), compared to 15.0% (1417 cases) in untreated individuals (n = 9416). This corresponded to an adjusted risk ratio of 0.82 (95% CI 0.74–0.91), favouring treatment. The second study, a prospective South Korean cohort study, followed 1978 adults with supratentorial ICH for a mean of 44.9 months (SD ± 31.5). 227 The recurrence rate was 9.8% (129/1317) among those receiving antihypertensive treatment targeting <140/90 mmHg or <130/90 mmHg in patients with relevant comorbidities, compared to 18.2% (120/661) in those without specific treatment. These studies were not included in the meta-analysis due to their observational design, heterogeneity in treatment and follow-up, and the availability of post-hoc data from RESPECT.224,225
Additional information
Based on current European and American general hypertension management guidelines, a target BP value ⩽130/80 mmHg is recommended by using a combination of antihypertensive drugs for patients with prior cerebrovascular disease.228,229 In adults with prior ICH, this approach is supported by retrospective data from the ERICH study, which found a higher rate of recurrent ICH in patients unable to achieve guideline-coherent blood pressure control (lobar ICH: HR 3.53, 95% CI 1.65–7.54; non-lobar ICH: HR 4.23, 95% CI 1.02–17.52). 230 According to the European Society of Cardiology (ESC) Guidelines, hypertension is defined by a BP threshold of ⩾140/90 mmHg for office measurements and ⩾135/85 mmHg for home measurements. 231 In adults with elevated BP and high cardiovascular risk, treatment is recommended for those with BP levels of ⩾130/80 mmHg to help reduce cardiovascular risk. The SPS3 showed a reduced risk of ICH with SBP target <130 mmHg versus 130–149 mmHg in lacunar stroke patients (who share same vascular pathological substrate as most ICH; arteriolosclerosis). 232 Data on the outcome death and dependence related to antihypertensive treatment in the post-acute phase following ICH were not identified.
Oral anticoagulation (OAC)


Analysis of current evidence
The guideline group graded the following outcomes as critical for reinstating OAC treatment after ICH: death, major adverse cardiovascular events, and ICH recurrence. While untreated NVAF strongly increases the risk of systemic embolism and death, resuming long-term OAC therapy in adults with prior ICH and NVAF is still challenging. We searched databases from January 2013 to May 2024. This was updated in line with a recent RCT that was included in the guideline and had searched the literature up to January 2025.
8
We identified three RCTs (n = 619) addressing the use of long-term oral anticoagulant (OAC) therapy after spontaneous ICH in patients with AF (SoSTART n = 203; APACHE-AF n = 101; PRESTIGE-AF n = 319; Supplement for PICO 6: description of single studies, Supplement for PICO 6.2: GRADE evidence profile).8,233,234 There was no difference in the risk of
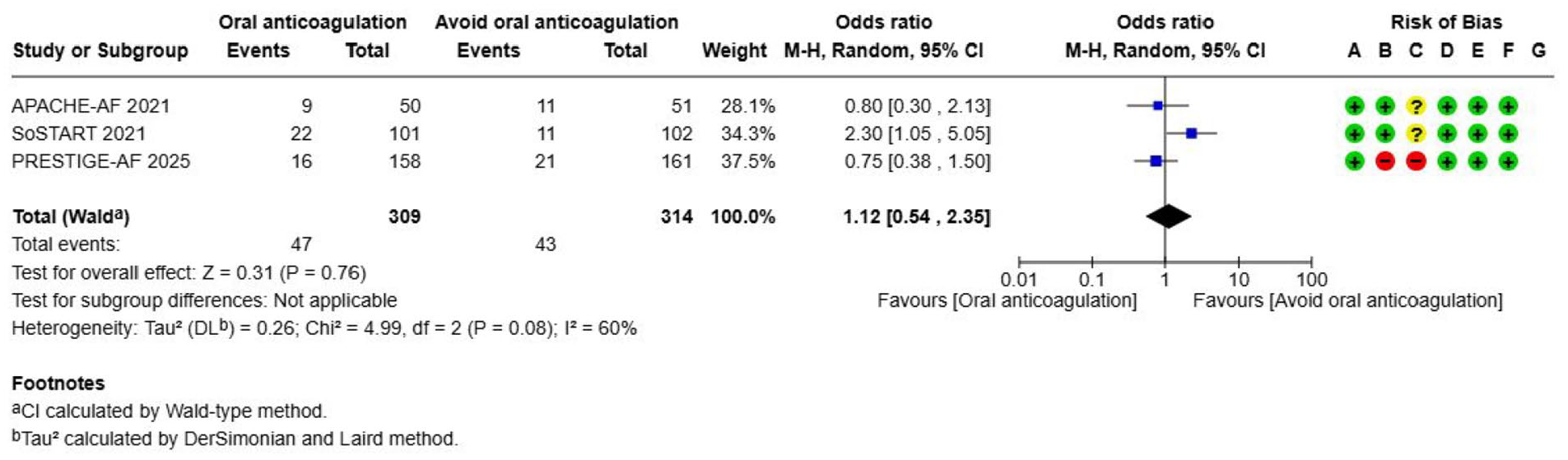
Effects on
Overall, there was a reduction in the occurrence of
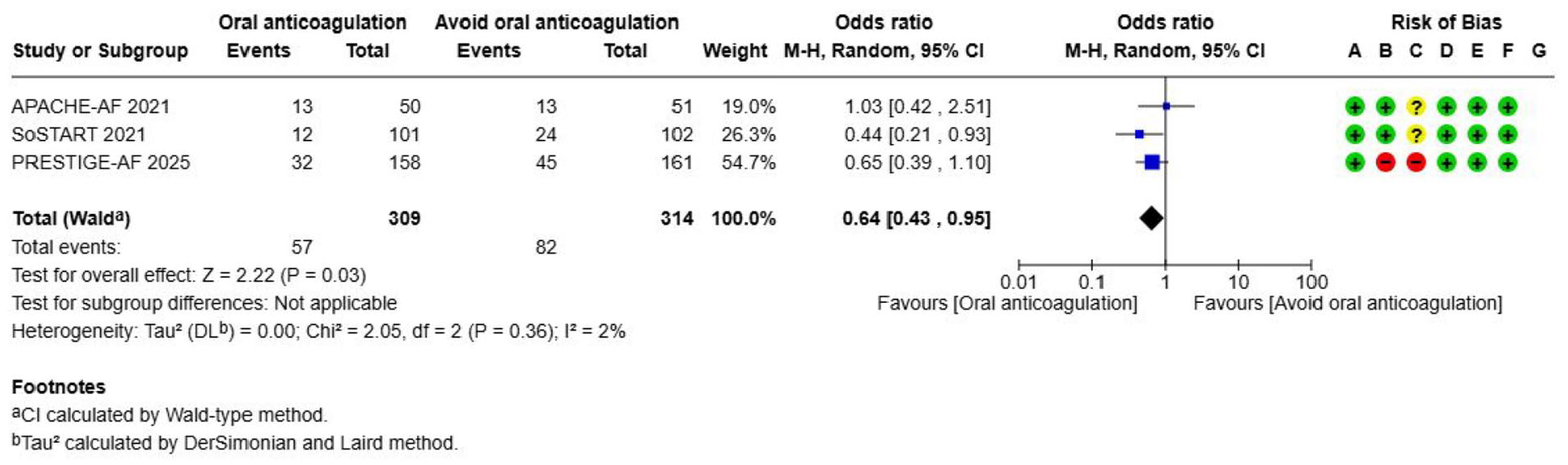
Effect of
There is a higher risk of
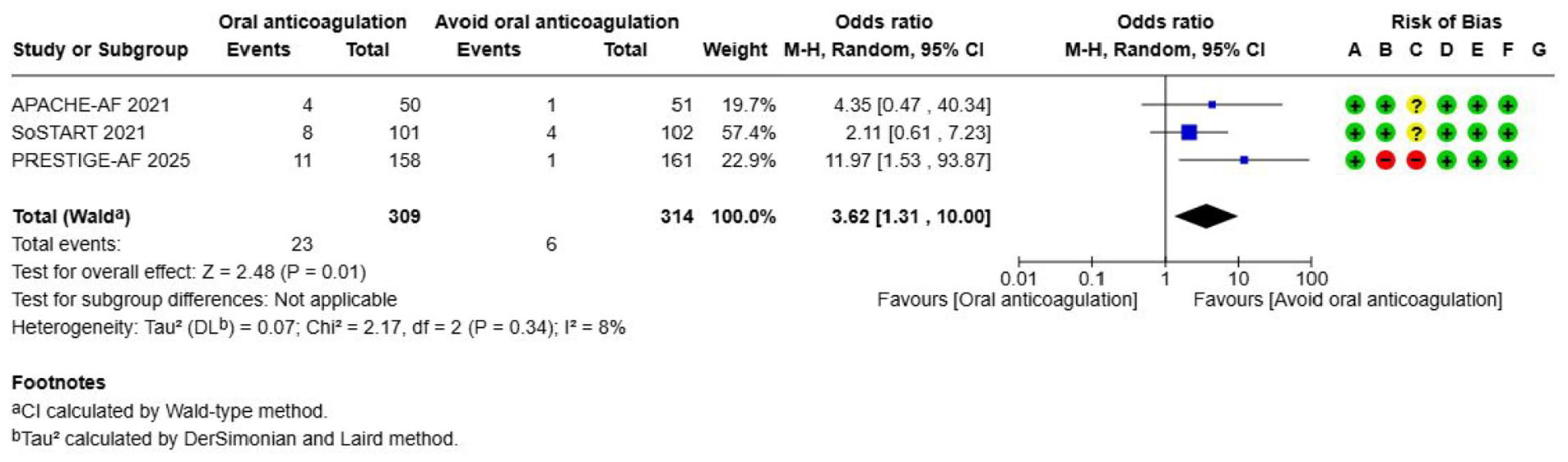
Effects on
Additional information
Our literature search identified a Cochrane review (2023) that included the NASPAF-ICH trial (NCT02998905) that randomised adults with prior ICH and atrial fibrillation (CHADS2 ⩾ 2) to DOAC versus acetylsalicylic acid (ASA), however this paper was unpublished and patient data was shared through the individual patient data meta-analysis (IPDMA). 174 Meta-analyses from the Cochrane review demonstrated a reduction of all major adverse cardiovascular events (MACE) in patients receiving OAC treatment compared with avoiding OAC (RR 0.61, 95% CI 0.40–0.94). Our literature search also identified a recent meta-analysis that studied the effects of starting versus avoiding anticoagulation in people with spontaneous ICH and NVAF. 235 This meta-analysis included a subgroup of ICH patients from the ELDERCARE-AF trial (n = 80) whose data was retrieved through the IPDMA. ELDERCARE-AF studied off-label low-dose edoxaban (15 mg/day) versus placebo. 236 Results from the meta-analysis demonstrated a reduction in ischaemic stroke in patients receiving OAC treatment compared with avoiding OAC (HR 0.27, 95% CI 0.13–0.56). Considering that the NASPAF-ICH trial was unpublished, and that the ELDERCARE-AF trial randomised patients to an off-label (15 mg) dose of edoxaban (standard dose 60 mg, adjusted dose 30 mg) for NVAF, these two studies were not included in the current meta-analysis.
A safety review of the ENRICH-AF trial (NCT03950076) has resulted in the decision to cease patient recruitment to the trial based on observations of unacceptably high risk of ICH recurrence among patients with lobar ICH and/or non-aneurysmal convexity subarachnoid haemorrhage (SAH), but these data remain unpublished. 237
Altogether, these findings suggest that there is an expected reduction in ischaemic events and increase in haemorrhagic events with OAC for NVAF after ICH, but the overall effects – and in particular the effect on death or dependence – are uncertain.
Prior to reinstating OAC treatment after ICH, the individual risk for bleeding and thromboembolic events should be carefully evaluated. This evaluation should consider several factors including the underlying ICH cause. Validated risk assessment tools, such as the HAS-BLED score for bleeding risk and the CHA2DS2-VASc score for thromboembolic risk, are useful in guiding clinical decisions. However, the HAS-BLED score has several limitations including its applicability to VKA-ICH, limited predictive power, and its inability to account for certain non-modifiable risk factors. Therefore, reinstating OAC treatment after ICH should not be based solely on these tools but should involve a comprehensive clinical evaluation. Selection criteria for adults with prior ICH and NVAF that may not benefit from the resumption or the initiation of OAC include the following:
patients with CAA-unrelated ICH and ⩾10 CMBs, 238
patients with CAA-related ICH with superficial siderosis or non-aneurysmal convexity SAH,233–235,237,239,240
patients with CAA-related ICH with the presence of ⩾5 lobar CMBs,
uncontrolled hypertension, 238
chronic alcohol abuse. 241
In considering anticoagulation in patients with prior ICH, the use of DOAC is recommended over VKA given the lower risk profile of intracranial bleeding associated with DOAC. 242
Ongoing trials
There are ongoing trials that address the resumption of OAC following ICH in people with AF: NCT03243175 (A3ICH, planned sample size N = 300), NCT03907046 (ASPIRE, N = 700), NCT03950076 (ENRICH-AF, N = 948). The STATICH trial (NCT03186729) has been terminated due to a lack of resources, with no results available yet. Further information about overall effects and net benefit on death or dependence will be available from the inclusion of these RCTs in the COCROACH IPDMA. 243
Left atrial appendage occlusion (LAAO) for atrial fibrillation


Analysis of current evidence
Adults with ICH and previous NVAF pose a unique challenge due to the relative contraindication of long-term anticoagulation. Left atrial appendage occlusion (LAAO) has emerged as an alternative for NVAF patients who are unsuitable for long-term oral anticoagulation. Our search found no published RCTs addressing this specific population. The concept of LAAO application in adults with contraindication for long-term OAC is supported by the PREVAIL and PROTECT-AF trials showing non-inferiority to vitamin K-antagonists (VKA) in preventing ischaemic stroke, with lower rates of ICH and mortality.244,245 A description of observational studies on LAAO for patients with ICH and NVAF who are unsuitable for long-term anticoagulation is provided in the Supplemental material. All studies either lack control groups or rely on propensity score-matched retrospective designs, using registry data to form control groups.
Additional information
There are no randomised trials evaluating periprocedural antithrombotic treatment in the context of LAAO. The European Society of Cardiology has made recommendations addressing periprocedural antithrombotic therapy as well as alternative procedures for patients unable to tolerate antiplatelet therapy. 246 More information on smaller, single-centre and multicentre prospective, observational studies is provided in description of single studies for PICO 6.3 in the Supplement.
Two phase III clinical trials addressing LAAO versus OAC in patients with prior ICH and AF are stated as ongoing in ClinicalTrials.gov: NCT03243175 (A3ICH), NCT04298723 (CLEARANCE; end expected in 2027), while two trials are finished: NCT03463317 (CLOSURE-AF, completed), and NCT02830152 (STROKECLOSE; status suspended).
Antiplatelet therapy


Analysis of current evidence
The guideline group graded the following outcomes as critical for reinstating antiplatelet treatment after ICH: ICH recurrence, major occlusive vascular events, and death. Our literature search identified one RCT comparing a strategy of restarting antiplatelet therapy to avoiding restarting antiplatelet therapy in patients who survived an ICH at least 24 h and had their therapy discontinued: The RESTART trial enrolled 537 patients with ICH recruited at a median of 76 days after the event (Supplement for PICO 6.4: GRADE evidence profile). 247 Patients were randomly assigned in a 1:1 ratio to start or avoid antiplatelet therapy and were followed for a median of 2.0 years (IQR 1.0–3.0). It is of noteworthy that the RESTART study included only patients who had been on antithrombotic therapy prior to the onset of ICH.
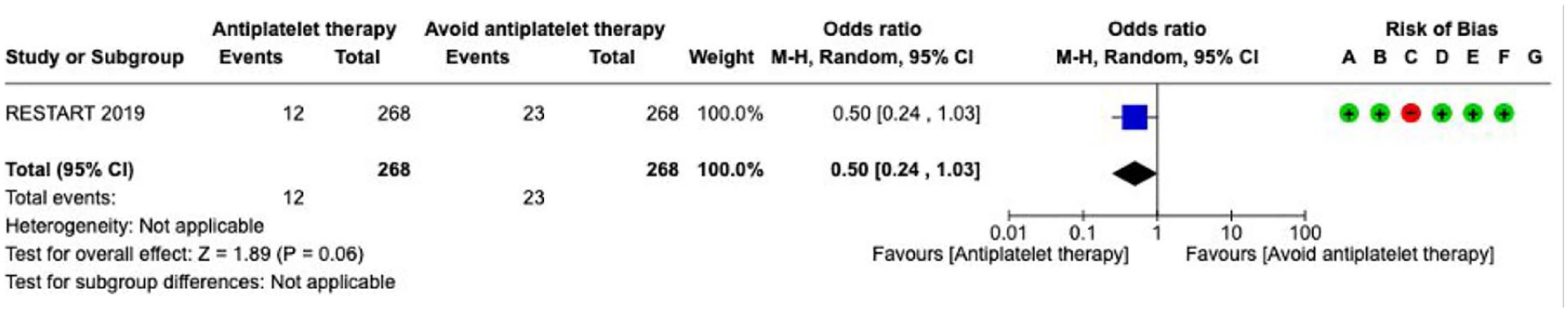
Effects on
Thirty-nine (15%) participants allocated to antiplatelet therapy had
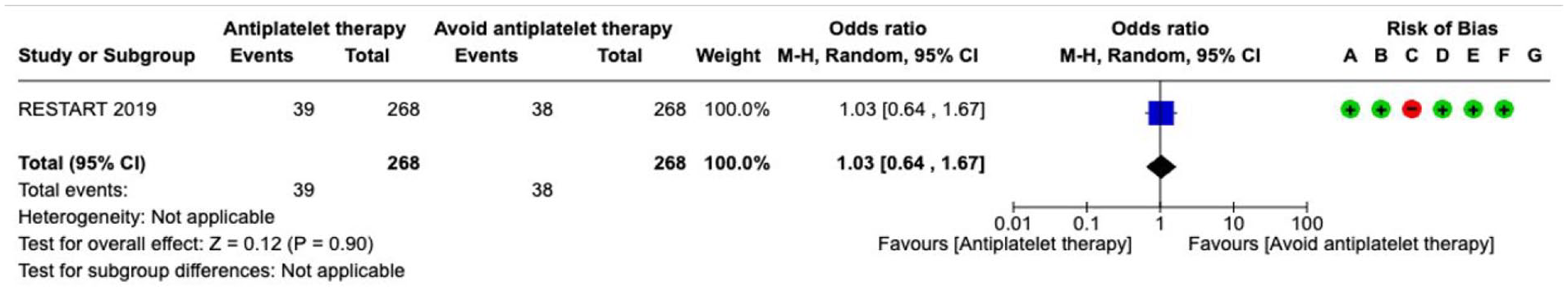
Effects of
No difference in the
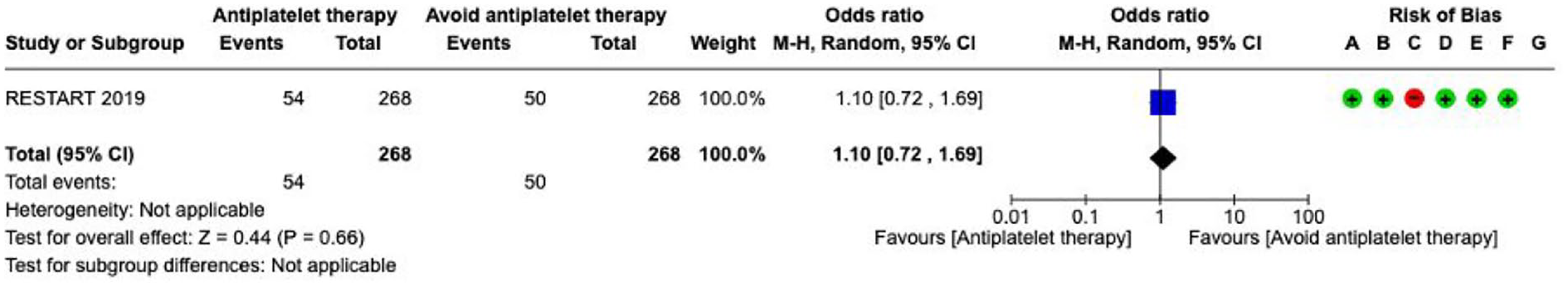
Effects on
Additional information
Recent observational studies have identified patients with ICH as having an increased risk of arterial ischaemic events compared with population controls.141,242 The risk of ischaemic stroke is high in people with lobar and with non-lobar ICH.248,249 The short-term risk of ischaemic stroke appears to be as high in people with deep ICH as with lacunar ischaemic stroke, both conditions thought to be caused by the same underlying hypertension-related small vessel disease.249,250 Thus, patients with ICH due to an underlying hypertensive microangiopathy may also be at an increased risk of major ischaemic vascular events. Consequently, antiplatelet therapy could prove beneficial, regardless of prior antiplatelet use before ICH onset. However, there is currently no evidence to support the initiation of antiplatelet therapy solely based on a history of ICH.
The ongoing ASPIRING trial (ISRCTN16705062) seeks to determine the safety and efficacy of antiplatelet agents in ICH survivors, regardless of prior antiplatelet use, and aims to recruit 4148 participants.
Statins


Analysis of current evidence
The guideline group graded the following outcomes as critical or important for statin treatment after ICH: death, major ischaemic vascular events, and ICH recurrence. While there is high level evidence for the preventive benefits of statins in cardiovascular disease, controversies exist regarding their effect on the risk of ICH.251,252 In one large RCT, the SPARCL trial revealed a higher risk of ICH among patients receiving statin therapy compared with placebo. 253 This effect did not offset the significant reduction in cardiovascular mortality in people who were on statins and the absolute number of ICH occurrence was low. Nevertheless, taken together with observational studies indicating an inverse correlation between elevated cholesterol levels and ICH risk, 254 these findings have led to question the advisability of statin use in all patients with a history of ICH. 45 Currently, there are no RCTs addressing the effects of statin use on specific endpoints following ICH.
As there were no RCTs, we performed a meta-analysis including a total of nine observational studies comparing the resumption or continuation of statin treatment versus no statin treatment after ICH, with a follow-up period of at least 1 year (Supplement for PICO 6.5: GRADE evidence profile). Additionally, a sub-study from the SPARCL trial, which involved a small ICH population (n = 93), was incorporated into the analysis. The analysis revealed a considerable heterogeneity among the included studies, attributable to variations in the timing of statin intervention and the duration of follow-up periods. Furthermore, due to the observational nature of the studies included in the meta-analysis, inherent bias cannot be overlooked. Individuals identified in the patient registers who fulfilled inclusion criteria for statin use most likely had a cardiovascular risk factor necessitating treatment in comparison to patients who avoided statins. An indication for statin therapy may have been overlooked in the patients identified as control.
A total of seven studies were included in mortality analysis.
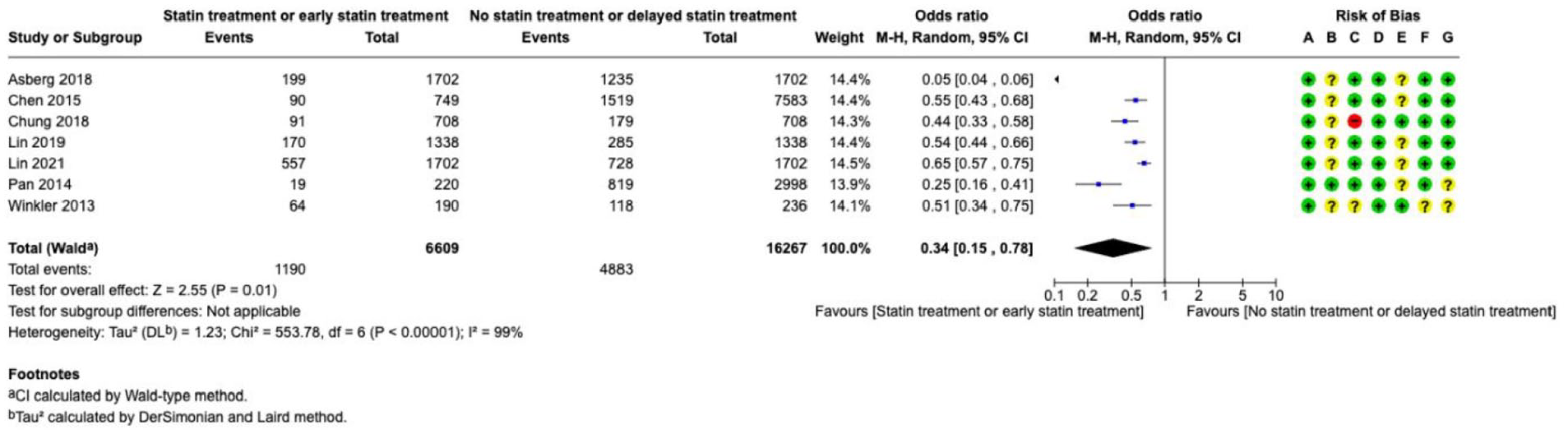
Effect on
In a meta-analysis of two observational studies and a SPARCL sub-study, there was no difference in the occurrence of
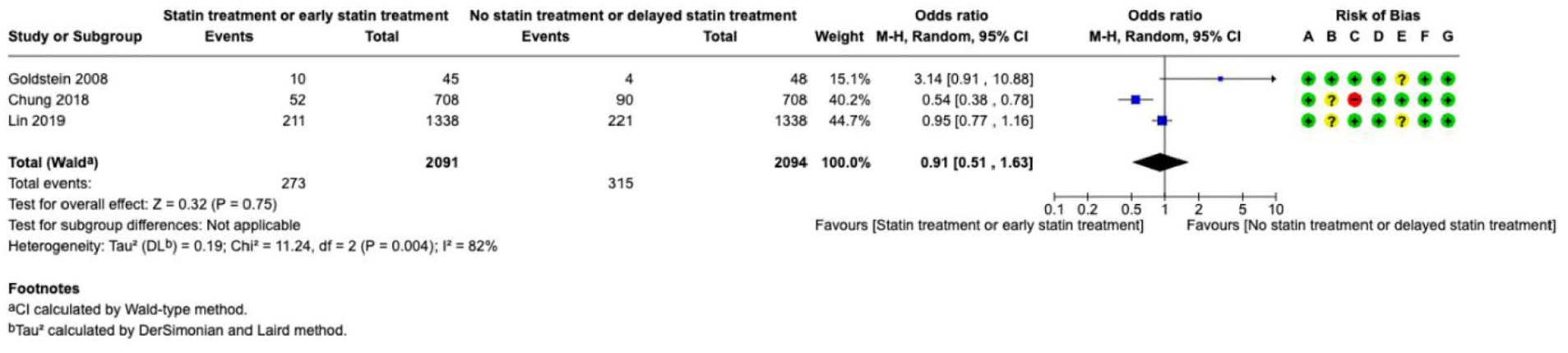
Effect on
In a total of seven studies, a reduction in
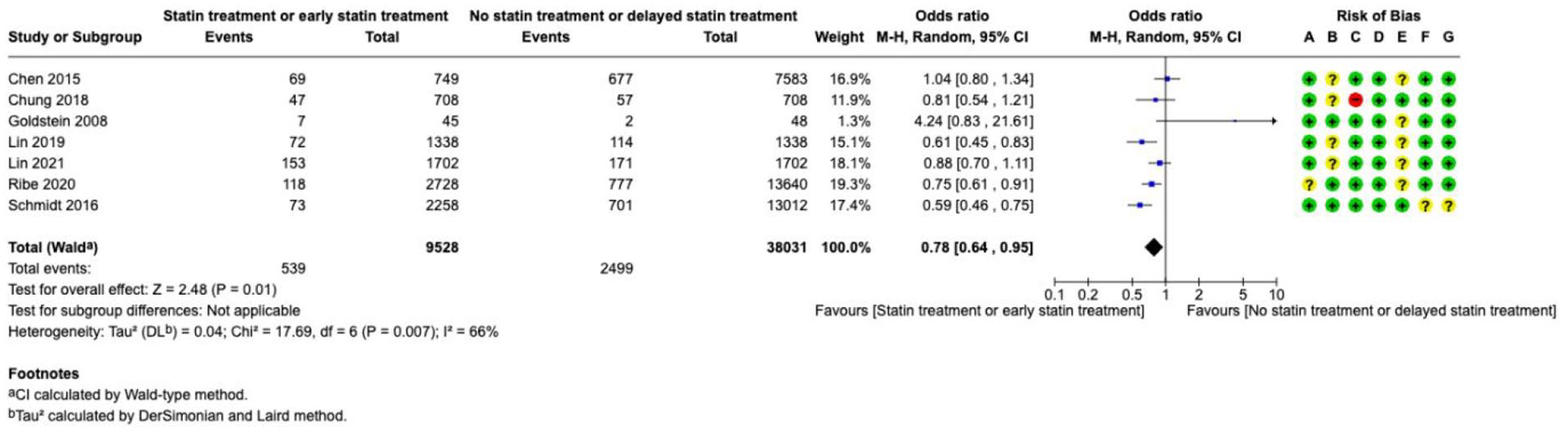
Effects on
Additional information
Statins and other cholesterol-lowering medications have been proven to be effective in preventing of cardiovascular disease. Given that many adults with a history of ICH also possess an elevated risk of cardiovascular disease, it is important to determine whether statin treatment after ICH is associated with negative outcomes. However, due to the absence of strong evidence, there is some reluctance in unequivocally recommending the use of statins in all adults with a history of ICH. 155
There is an ongoing trial addressing statin use in people with lobar ICH already on statins randomised to discontinuation or continuation of statin therapy (SATURN NCT03936361).
Discussion
We present the ESO/EANS guidelines for the management of intracerebral haemorrhage (ICH). Since the last ESO guidelines in 2014, there has been a significant increase in RCTs of interventions for ICH.5,55,256 As a result, our primary focus was on RCTs, incorporating observational studies of high quality only where RCTs were unavailable. Additionally, we shifted our perspective on recommendations, prioritising clinical benefit as the primary criterion for both diagnostic and therapeutic interventions. This change explains why certain recommendations are now accompanied by consensus statements and, in some cases, differ from previous guidelines (Table 2).
Synopsis of all recommendations and expert consensus statements.

We found strong evidence supporting the
Other important management aspects need to be considered, which are a treatment threshold or range, timing, volume of ICH, variability and duration of blood pressure treatment. The evidence concerning these aspects, however, does not allow for a recommendation according to the GRADE approach, which is why we present an expert consensus statement. First, with respect to
None of the
Patients with acute ICH are at high
Meta-analyses of RCTs of
Over the past decade, significant advancements have been made in the field of ICH management. These developments have primarily occurred in three areas: the use of specific haemostatic agents in patients with iatrogenic coagulopathies, neurosurgical interventions, and the implementation of care bundles. However, due to the current body of evidence, we can provide only weak recommendations for certain PICO questions or must rely on expert consensus statements. In many cases, this approach is necessary because the sample sizes of individual studies are too small to establish strong evidence. The primary reasons for this include epidemiological constraints and financial limitations. 270 Therefore, the authors of this guideline advocate for increased efforts to conduct large-scale international studies.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251340815 – Supplemental material for European Stroke Organisation (ESO) and European Association of Neurosurgical Societies (EANS) guideline on stroke due to spontaneous intracerebral haemorrhage
Supplemental material, sj-docx-1-eso-10.1177_23969873251340815 for European Stroke Organisation (ESO) and European Association of Neurosurgical Societies (EANS) guideline on stroke due to spontaneous intracerebral haemorrhage by Thorsten Steiner, Jan C Purrucker, Diana Aguiar de Sousa, Trine Apostolaki-Hansson, Jürgen Beck, Hanne Christensen, Charlotte Cordonnier, Matthew B Downer, Helle Eilertsen, Rachael Gartly, Stefan T Gerner, Leonard Ho, Silje Holt Jahr, Catharina JM Klijn, Nicolas Martinez-Majander, Kateriine Orav, Jesper Petersson, Andreas Raabe, Else Charlotte Sandset, Floris H Schreuder, David Seiffge and Rustam Al-Shahi Salman in European Stroke Journal
Supplemental Material
sj-pdf-2-eso-10.1177_23969873251340815 – Supplemental material for European Stroke Organisation (ESO) and European Association of Neurosurgical Societies (EANS) guideline on stroke due to spontaneous intracerebral haemorrhage
Supplemental material, sj-pdf-2-eso-10.1177_23969873251340815 for European Stroke Organisation (ESO) and European Association of Neurosurgical Societies (EANS) guideline on stroke due to spontaneous intracerebral haemorrhage by Thorsten Steiner, Jan C Purrucker, Diana Aguiar de Sousa, Trine Apostolaki-Hansson, Jürgen Beck, Hanne Christensen, Charlotte Cordonnier, Matthew B Downer, Helle Eilertsen, Rachael Gartly, Stefan T Gerner, Leonard Ho, Silje Holt Jahr, Catharina JM Klijn, Nicolas Martinez-Majander, Kateriine Orav, Jesper Petersson, Andreas Raabe, Else Charlotte Sandset, Floris H Schreuder, David Seiffge and Rustam Al-Shahi Salman in European Stroke Journal
Footnotes
Acknowledgements
We thank Yvonne Brüchert for her extensive support in the preparation of this guideline. We would also like to acknowledge Sabrina Lémeret, Josh Cheyne and Salman Hussain for their support in programming the literature search.
Declaration of conflicting interests
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
TS
Contributorship
Thorsten Steiner, Rustam Al-Shahi Salman, Diana de Aguiar Sousa, Matthew Downer, Rachael Gartly, Hanne Christensen, Silje Holt Jahr, Helle Eilertsen, Jan Purrucker, Stefan Gerner, Trine Apostolaki-Hansson wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript. Leonard Ho conducted the data extraction and statistical analyses.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
