Abstract
Introduction
Knowledge of the implementation gap would facilitate the use of intravenous thrombolysis in stroke, which is still low in many countries. The study was conducted to identify national implementation targets for the utilisation and logistics of intravenous thrombolysis.
Material and Method
Multicomponent interventions by stakeholders in health care to optimise prehospital and hospital management with the goal of fast and accessible intravenous thrombolysis for every candidate. Implementation results were documented from prospectively collected cases in all 45 stroke centres nationally. The thrombolytic rate was calculated from the total number of all ischemic strokes in the population of the Czech Republic since 2004.
Results
Thrombolytic rates of 1.3 (95%CI 1.1 to 1.4), 5.4 (95%CI 5.1 to 5.7), 13.6 (95%CI 13.1 to 14.0), 23.3 (95%CI 22.8 to 23.9), and 23.5% (95%CI 23.0 to 24.1%) were achieved in 2005, 2009, 2014, 2017, and 2018, respectively. National median door-to-needle times were 60–70 minutes before 2012 and then decreased progressively every year to 25 minutes (IQR 17 to 36) in 2018. In 2018, 33% of both university and non-university hospitals achieved median door-to-needle time ≤20 minutes. In 2018, door-to-needle times ≤20, ≤45, and ≤60 minutes were achieved in 39, 85, and 93% of patients.
Discussion
Thrombolysis can be provided to ≥ 20% of all ischemic strokes nationwide and it is realistic to achieve median door-to-needle time 20 minutes.
Conclusion
Stroke 20–20 could serve as national implementation target for intravenous thrombolysis and country specific implementation policies should be applied to achieve such target.
Introduction
Stroke is the second leading cause of death and lost years of healthy life worldwide. 1
Intravenous thrombolysis (IVT) is the only pharmacological treatment of stroke that can reverse neurological deficit due to stroke and that either is, or could be, readily available in most countries around the world. 2 Despite the relative ease of use of IVT, the low cost of the drug, and the proven cost-effectiveness, the implementation into clinical practice has been challenging. In developing countries, the availability of the drug could be the major problem, as thrombolytic rates were reported to be below four percent. 3 , 4 However, even in highly developed countries, utilisation of thrombolysis varies and some reported hospital-based thrombolytic rates were less than ten percent.5–7
Not only treatment itself, but also sooner treatment from symptom onset with IVT improves outcome. 8 , 9 It has been demonstrated that in experienced stroke centres a door-to-needle time (DNT) of 20 minutes or less is achievable. 10 It is, however, not clear if such a short DNT could be used as a national implementation target.
After 20 years of IVT use in clinical practice, it remains unclear what is the maximum percentage of patients with ischemic stroke that can be treated with IVT and how low a national DNT can be achieved. If such benchmarks were known, it would allow identifying the implementation gap, i.e. the difference between current practice and target level of utilisation. Here, we present the national experience in the Czech Republic including implementation strategy and health policies and propose 20 20 benchmarks, meaning that from all ischemic strokes in a certain population ≥ 20% undergo IVT, with a median door-to-needle time of ≤20 minutes.
Methods
The aim of this study was to document 1) the high levels of utilisation of intravenous thrombolysis and its ultrafast delivery at a national level, and 2) the most important strategies and actions that led to such results. The level of utilisation was documented by prospectively collected data in the Safe Implementation of Treatments in Stroke (SITS) registry (download 11 Nov 2019).
Development of stroke centres in the Czech Republic
In the nineteen nineties, around 100 neurology departments existed around the Czech Republic. IVT started to be used in clinical practice in 1998 11 based on the results of National Institute of Neurological Disorders and Stroke (NINDS) trials but as an off label treatment. In 2004 National regulatory authorities approved alteplase (Actilyse®, Boehringer Ingelheim, Ingelheim am Rhein, Germany) for stroke treatment and since then all patients treated with IVT have been registered in SITS registry till the end of 2018. In 2016 all stroke centres began using the Registry of Stroke Care Quality (RES-Q), which included data on quality of stroke care, and since 2019 also included data on IVT, and mechanical thrombectomy. 12
In 2008 the Czech Ministry of Health initiated a program for standardisation and accreditation of Specialised Care Centres. In 2011, the Committee for Specialized Care in Neurology accredited 10 Comprehensive Stroke Centres (CSC) and 23 Primary Stroke Centres (PSC). Nowadays, there are 13 CSCs and 32 PSCs throughout the country (Figure S1). In Supplemental methods, there is detailed description of the development and organisation of stroke centres in the Czech Republic related to: Pre-hospital care, Stroke care quality control and feedback, Other actions to improve in-hospital logistics and shorten DNT, Use of Informed consent, and System of post-graduate education. The most important strategies for stroke care improvement are summarised in Figure S3.
Sources of information on thrombolysis
Patients’ characteristics, number of treatments, and logistic data were obtained from prospectively collected data in the SITS registry. Since 2011, also twelve key performance indicators, including the number of thrombolytic treatment and DNTs are provided to the Ministry of Health annually. Data on the volume of thrombolytic and other treatments were generated from hospital statistics.
Accuracy of data from SITS was checked against these official annual reports for the purpose of this analysis and any inconsistencies found are also reported here. Further, information on the number of units of alteplase sold in the Czech Republic in 2018 was obtained from Boehringer Ingelheim to obtain additional complementary confirmation.
Calculation of the thrombolytic rate
Population based thrombolytic rates are provided using two methods of calculation. The numerator is the number of thrombolytic treatments as reported in the SITS registry, provided that this number is consistent with other sources of data, including official hospital reports submitted to the Ministry of Health, and for data collection since 2016, also the international quality registry, RES-Q. 12 The denominator for the first method is the number of admissions of ischemic strokes as calculated based on the validated data from the National Registry of Hospitalized Patients. 13 For the second method, the total number of ischemic strokes recorded yearly until 2017 is based on published Global Burden Disease data (GBD). 14
Temporal trends of association between factors associated with DNT
We have previously demonstrated that younger age, National Institutes of Health Stroke Scale score (NIHSS), and onset-to-door time (ODT) independently predicted DNT ≤60 minutes. 15 Here we present how the strength of the association between ODT and DNT has changed over years.
Statistics
Descriptive statistics were used with mean, medians, 95% confidence intervals (CI), and interquartile ranges (IQR). All hospitals with any number of cases reported were included. Missing data are reported but there was no imputation for missing data. Trends in DNT are presented in figures. The relationship between ODT and DNT are demonstrated as scatterplots, and correlations. All statistical analyses were performed using IBM SPSS software version 22.0 (IBM, Armonk, NY, USA). Data have been collected as part of routine clinical practice and approval of Ethics Committees was not required. The Ethical Committee of St. Anne’s University Hospital in Brno approved the analysis of the data as presented here.
Results
Altogether 66 hospitals reported 31,524 patients treated with intravenous thrombolysis by alteplase throughout 2004 – 2018. The overall number of patients treated with IVT in a single hospital ranged from 537 to 2314 in 13 CSCs (median 1137, IQR 801 to 1377), from 178 to 912 in 32 PSCs (median 412, IQR 325 to 648), and from one to 236 cases in 21 not certified stroke centres (median 34, IQR 16 to 47). Annual use of intravenous thrombolysis per hospital in 2005, 2010, 2015, and 2018 was median 7 (IQR 5), 17 (IQR 17), 94 (112), and 102 (IQR 118) cases, respectively. Of all 31,524 patients, 15,327 (49%) were treated in 13 CSCs, 15,305 (49%) in 32 PSCs, and 892 (3%) outside certified comprehensive, or primary stroke centres. Altogether 10,886 (35%) patients were treated in nine university hospitals (eight of them are CSCs and one is PSC). Baseline and demographic patients’ data are shown in Table 1.
Demographic and baseline characteristics of all patients treated with intravenous thrombolysis.
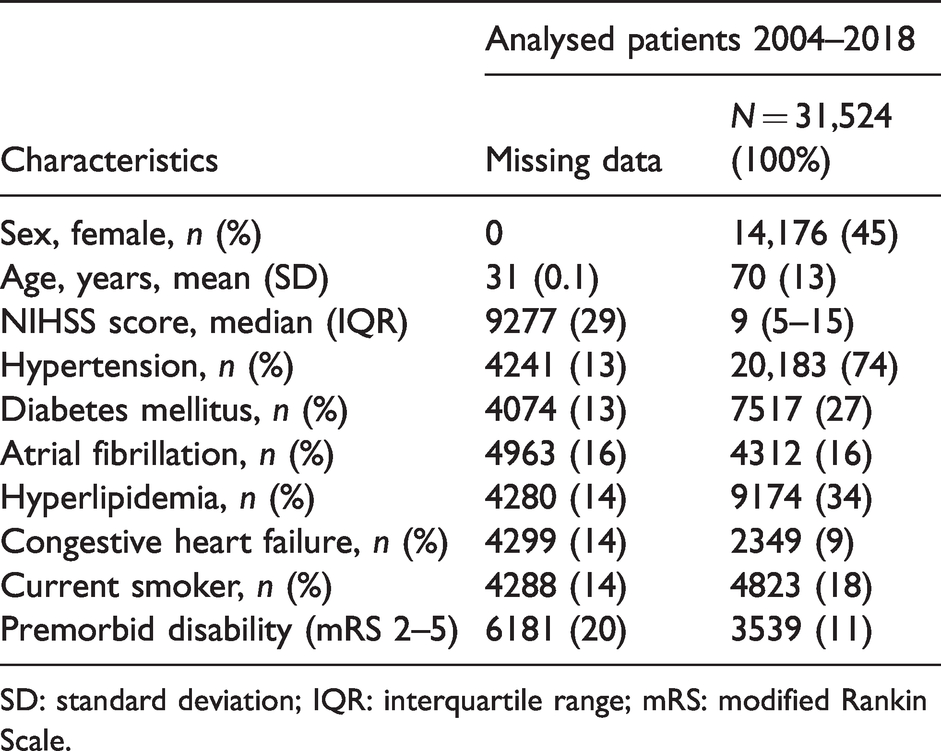
SD: standard deviation; IQR: interquartile range; mRS: modified Rankin Scale.
The number of cases reported in the SITS registry versus in official hospital reports to the Ministry of Health were nearly the same, e.g. in 2017: 4859 versus 4853 and in 2018: 4958 versus 4862 cases. SITS includes the same patients as the registry of the Ministry of Health, plus additional cases from non-certified hospitals. Of all 50 mg (4833) and 20 mg (15,203) units of alteplase sold in the Czech Republic in 2018, altogether 3141 (65%) of 50 mg, and 11376 (75%) of 20 mg were used for stroke treatment. The national number of IVT´s performed in each year is documented in Table S1.
Initial thrombolytic rates in 2005 was 1.3 (95%CI 1.1 to 1.4) and final in 2018 was 23.5% (95%CI 23.0 to 24.1%). As demonstrated in Table S1, the proportion of patients treated with thrombolysis over 80 years of age and with mild strokes substantially increased over time. Overall proportion of patients treated with intravenous thrombolysis over 80 years of age and with mild strokes was in comprehensive versus primary stroke centres 21.5% (95% CI 20.8 to 22.1%) versus 20.4% (95% CI 19.7 to 21.0%) and 24.8% (95% CI 24.0 to 25.7%) versus 26.5% (95% CI 25.7 to 27.3%), respectively.
Data on DNT were provided for 26,899 (85%) patients and data on ODT on 27,546 (87%) of cases. Patients with missing data on DNT had nearly the same baseline characteristics as patients with available data, e.g. age 70 ± 13 vs. 70 ± 13, age >80 20% vs. 19%, NIHSS 10 (IQR10) vs. 9 (IQR 9), hypertension 74% vs. 73%, diabetes 27% vs. 27%, treaded in certified stroke centres 97% vs. 99%, and treated in CSCs 46% vs. 53%. Graphical presentation of median DNTs from 2004 till 2018 and in CSCs and PSCs separately demonstrates Figure 1 ((a) and (b), respectively). Although DNT was shorter in CSCs as compared to PSCs in several previous years, it became almost the same in 2017 and 2018. DNT accounted for 53% of total time delay (i.e. onset-to-needle time) in 2005 but only 26% in 2018.
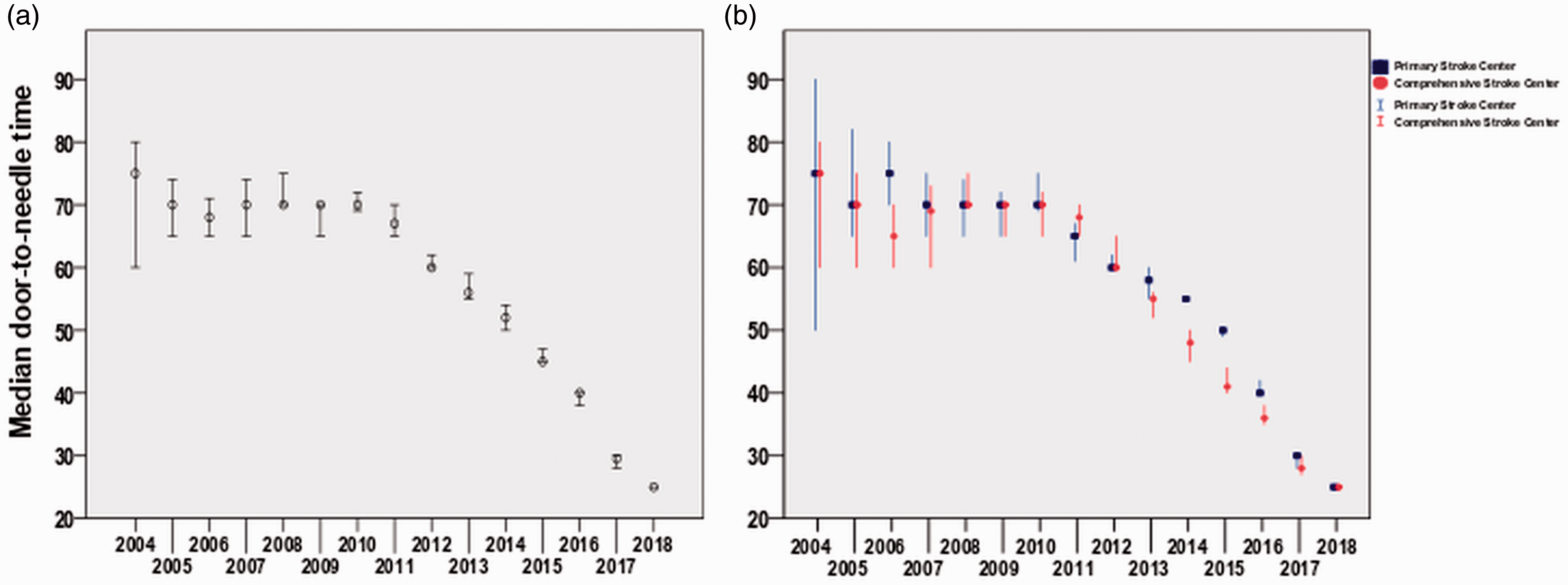
Door-to-needle time (DNT) during the period 2004–2018. (a) Median DNT in minutes and 95% confidence intervals from 2004 till 2018 in all stroke centers. (b) Median DNTs in minutes and 95% confidence intervals in Comprehensive Stroke Centres (boxes) as compared to Primary Stroke Centres (dots) from 2004 till 2018. Median DNT was significantly shorter in Comprehensive as compared to Primary Stroke Centres between 2013 and 2016 but become the same in 2017–2018.
In 2018, median DNT was 25 minutes (IQR 17 to 36), ODT 87 minutes (IQR 60 to 133), and onset-to-needle time 117 minutes (IQR 88 to 164). In 2018, fifteen (33%) hospitals achieved median DNT ≤20 minutes (three [33%] from university and 12 [33%] from non-university hospitals). Another 23 hospitals achieved median DNT 21–30 minutes and nine hospitals median DNT 41–60 minutes. No hospital had median DNT above 60 minutes with the worst median DNT 44 minutes. Also in 2018, a DNT ≤20, ≤45, and ≤60 minutes was achieved for 39, 85, and 93% of patients. Occurrence of parenchymal haemorrhage in 2005, 2010, 2015, and 2018 was 6.8% (95% CI 3.8 to 9.8), 7.7% (95% CI 6.3 to 9.1), 4.0% (95% CI 3.4 to 4.7), and 5.0% (95% CI 4.3 to 5.7), respectively.
Figure 2 demonstrates that patients were treated faster (i.e., with shorter DNT) if physicians had less time (i.e., patients arrived later and thus had longer ODT) toward the end of the thrombolytic window in previous years. However, in the most recent years, hospital logistics became the same for patients with different time intervals left till the end of the thrombolytic window.
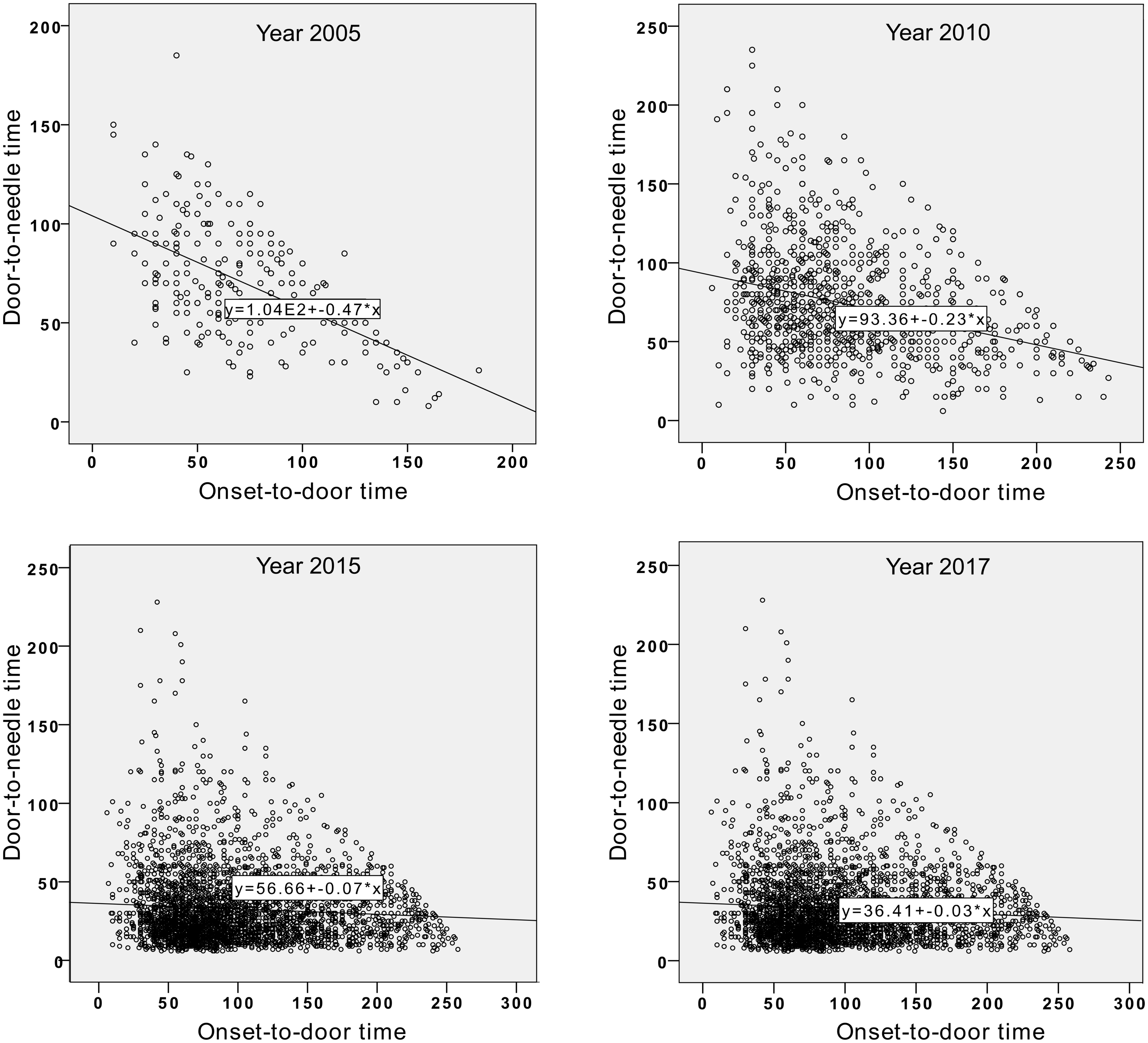
Comparison of treatment startup within the thrombolytic window. The graphs show that patients arriving to hospital towards the end of thrombolytic window used to be treated with thrombolysis faster (in 2005 and 2010) but in recent years (2015 and 2017), patients door-to-needle time (DNT) was the same regardless of onset-to-door time (ODT).
Implementation and quality improvement components are conceptualised as puzzle in Figure 3.
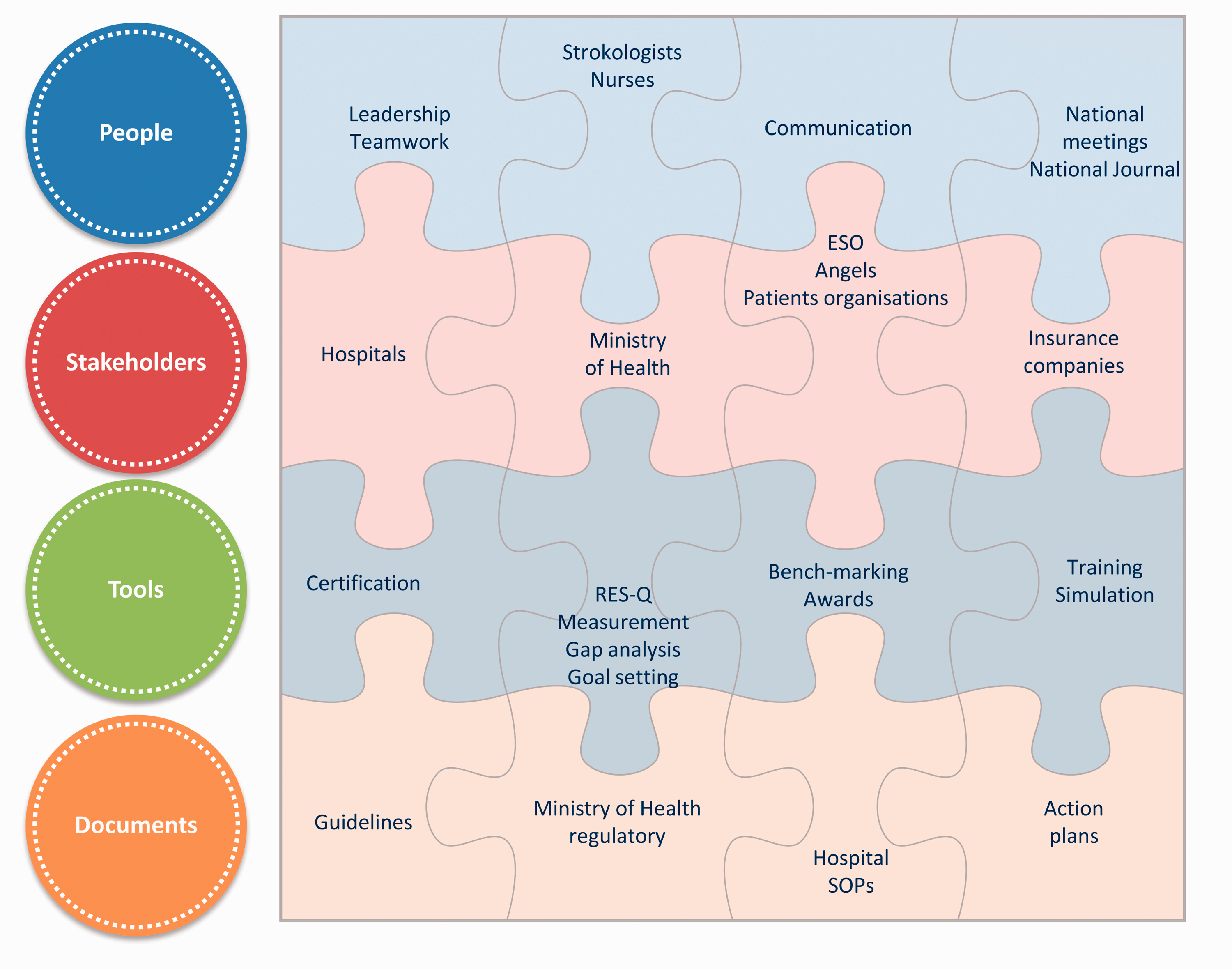
The implementation and quality improvement puzzle. All components support each other and fall into four categories such as people, stakeholders, tools, and documents. Leadership by stroke society, communication with healthcare professional using emails, journal (in local language), and national conferences is the first line followed by stakeholders involvement such as hospitals and the Ministry of Health. Tools such as quality registry allow for data driven certification, monitoring of quality of health care, identification of gaps and setting the targets. Documents supporting the overall strategies such as national guidelines, regulations and hospital standard operating procedures are the final line.
Discussion
The use of IVT in clinical practice could be facilitated if benchmarks for utilisation and treatment logistics are defined. In our study we propose such benchmarks based on evidence that over 20% of patients with acute ischemic stroke were treated with IVT. We also propose that a median DNT ≤20 minutes is realistic given the fact that one third of hospitals have already achieved similar results in 2018 and the overall national median DNT dropped to 25 minutes.
In our study we also described health policy implementation process and although the generalisability to other countries might be limited due to differences in organisation of health services, the principles remain. First of all, we managed that all strokes are mandatorily triaged to certified and quality-controlled stroke centres with sufficient capacity. As a consequence, an absolute majority of recanalisation treatments were provided within certified and quality controlled network (with about half in comprehensive and half in primary stroke centres), both previously documented to improve stroke care and outcome.16–20 Although hospitals have different providers and owners, we succeeded in coordinating them to act as one functional unit, which is governed by the same rules and principles. The applied tools, such as audit and feedback, have had an impact on system performance as a whole.
We systematically and prospectively measured implementation outcome and demonstrated that the thrombolytic rate was increasing every year through 2017 when exceeded the median 23%, and then plateaued in 2018. Nationally, this level of utilisation of IVT has not been reported in any country.5–7 So far, the highest IVT utilisation was reported in Austria (16.8%) in 2013, Catalonia (12%), and Australia (12%) in 2014.21–23 In Germany documented a 13.6% thrombolytic rate, however, it is not clear how this hospital-based data differs from population-based. 24 Our results, therefore, support the conclusion that a thrombolytic rate over 15% before 2030 as defined by Stroke Action Plan for Stroke in Europe is not only realistic, but could be potentially overly conservative, at least for high-income European countries. 25
We also documented a dramatic decrease in national DNT with 50% and 39% of patients treated with DNT ≤25 minutes and ≤20 minutes, respectively in 2018. These results greatly exceed the 2018 recommendation by American Heart Association stating that ≥50% of patients should achieve DNT ≤45 minutes. 26 Recent data from the Netherlands reports a median national DNT also 25 minutes, thereby confirming generalisability outside of a single health-care system. 27 While in many countries DNT is still around 60 minutes, 28 this experience, both from the Netherlands and the Czech Republic, establishes a new benchmark for DNT. It also raises the question of whether guidelines should provide even stricter target goals for DNT.
Many complimentary factors contributed to the Czech Republic achieving a high thrombolytic rate and short DNT including: stroke centres carry enough bed capacity, geographically accessible stroke units, stroke suspects are transported by Emergency Medical Services (EMS) only to certified stroke centres (after pre-notification), stroke treatments are covered by mandatory health insurance, national guidelines support acute stroke treatments (e.g. eliminating age or disabling deficits with low NIHSS as contraindications), and very importantly, additional motivation to promote evidence-based stroke treatments using a regular national benchmarking (reports are not anonymised, see Figure S2). In other countries many of components, such as accreditation and certification, reporting performance measures, presence of feedback, and leadership; have all been documented to increase compliance with guidelines such as the use of thrombolytic therapy. 5 ,29–31 Online benchmarking increased the number of IVT in Austria, and similarly, monthly reporting of logistic results may have contributed to a decrease in DNT in Czech stroke centres. 32 Many components and principles may not be, however, directly transferable to other health-care systems although using “audit and feedback,” or “online benchmarking” is an example of one principle that could be transferable quite easily.
Broadening inclusion criteria for thrombolysis led to an increase in patients treated by IVT (see Table S1). Patients of older age and milder neurological deficit are two notable subgroups that now receive thrombolytic therapy much more often than in the past. And the proportion of such patients is not less in primary than in comprehensive stroke centres, which was the case in a previous study from Germany. 33 Also, the extension of the time window from 3 to 4.5 hours in 2008, and decreasing DNT in recent years allowed for thrombolytic treatment of patients arriving to hospital later, i.e. closer to the end of thrombolytic time window.
For the improvement of stroke logistics across stroke centres two training tools were created to support ultra-fast delivery of thrombolysis based on the Helsinki model. 10 These tools included video records of thrombolytic treatment from admission until treatment, with very short DNT to be presented at national stroke conferences. The other tool was training of stroke teams in a simulation centre available in one of the stroke centres. A final, non-training related tool, the monthly reporting of DNTs, was already described. Interestingly, all of these tools reduced the propensity for physicians to use more time in cases where they had more time for thrombolytic treatment i.e. the patient arrived earlier in the thrombolytic treatment window (Figure 2), which was also reported from other experienced stroke centres. 34
There are several limitations to our study. Calculation of the thrombolytic rate based on the total number of ischemic strokes in the population may not be accurate. Estimation is possible however, based on either data from GBD (last available year is now 2017) or national epidemiological data. National data reflects stroke admissions while GBD stroke incidence. Because of high availability of stroke services in the Czech Republic, thrombolytic rates are comparable if calculated of either stroke admission or stroke incidence but this might not apply to other countries. Also, our calculation may somewhat overestimate the thrombolytic rate because only the first ever stroke is considered, but this concern is not relevant for benchmarking.
We intentionally calculated population-level thrombolytic rates rather than hospital-level rates because they can differ substantially depending on the disparity in stroke care quality. Hospital-based thrombolytic rate in 2018 in the Czech Republic based on national quality registry RES-Q was in average 30% (minimum 7%, maximum 53%), compared to a 23% population-level rate. 11 Having a high hospital-level thrombolytic rates but low population based rate would mean that high thrombolytic rate at one hospital must be counterbalanced by low thrombolytic rate in other hospitals. Thus high hospital-level thrombolytic rate does not necessarily mean good stroke care quality nationally.
Another limitation is unavailability of DNT value in 15% of patients and that information of thrombolysis use comes from a registry without validation of its accuracy. However, we do not expect inconsistences because; registry data used for this analysis are nearly the same as data provided from hospital statistics (mandatory reports to the Ministry of Health), the data reflect clinical practices which are frequently discussed at meetings such as national conferences (the annual Czech and Slovak Stroke Congress), the data entered into registries come from official hospital records as a source, the registry results are shared with officials such as the Ministry of Health, units of sold alteplase are not less than expected, and patients with missing data do not differ from those that have data available.
A possible limitation to the broad implementation of results could be that the safety of the Helsinki model of ultra-fast tPA delivery was demonstrated in highly experienced stroke centres but not across different types of hospitals nationwide. 10 , 35 These stroke units can differ in their volume and length of experience with IVT. In the Czech Republic, however, an improvement of logistics has occurred as even smaller hospitals have treated at least few hundreds of patients. Implementing ultra-fast delivery of thrombolysis might not be straightforward and even desirable in countries and centres with less experience (i.e. low utilisation) with thrombolysis. And next, information on stroke mimics is missing in existing registries and it is not clear if faster tPA delivery would result in treating more stroke mimics, although it was documented to be generally safe in these patients. 36 Finally, we did not provide benchmarks for mechanical thrombectomy, for which utilisation in clinical practice is still increasing.
Conclusion
Utilisation and speed of delivery of thrombolytic treatment are the most important performance and quality metrics of acute stroke services in all types of hospitals admitting acute stroke patients. Our study demonstrated it is achievable to treat as many as over 20% of all ischemic stroke patients with thrombolysis. Thrombolytic treatment can also be initiated within 20 minutes after patients’ arrival, not only in specialised university-based stroke centres but also other types of hospitals. It should be demonstrated that dramatic shortening of DNT does not compromise safety and does not lead to unacceptable treatment rate of stroke mimics. Appropriate health policy implementation steps should be taken to achieve such results. Stroke 20 20 is a realistic implementation target.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211007684 - Supplemental material for Stroke 20 20: Implementation goals for intravenous thrombolysis
Supplemental material, sj-pdf-1-eso-10.1177_23969873211007684 for Stroke 20 20: Implementation goals for intravenous thrombolysis by Robert Mikulik, Michal Bar, David Cernik, Roman Herzig, Rene Jura, Lubomir Jurak, Jiri Neumann, Daniel Sanak, Svatopluk Ostry, Petr Sevcik, Ondrej Skoda, David Skoloudik, Daniel Vaclavik and Ales Tomek in European Stroke Journal
Supplemental Material
sj-pdf-2-eso-10.1177_23969873211007684 - Supplemental material for Stroke 20 20: Implementation goals for intravenous thrombolysis
Supplemental material, sj-pdf-2-eso-10.1177_23969873211007684 for Stroke 20 20: Implementation goals for intravenous thrombolysis by Robert Mikulik, Michal Bar, David Cernik, Roman Herzig, Rene Jura, Lubomir Jurak, Jiri Neumann, Daniel Sanak, Svatopluk Ostry, Petr Sevcik, Ondrej Skoda, David Skoloudik, Daniel Vaclavik and Ales Tomek in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R Mikulik, M Bar, J Neumann, and D Sanak received honoraria payments and travel support from Boehringer-Ingelheim. R Herzig received honoraria payments from Bayer, Boehringer Ingelheim, and Gedeon Richter; and travel support from Biogen, Merck, and Teva Pharmaceuticals. A Tomek reports personal fees from Boehringer Ingelheim, Pfizer, Amgen, MSD, Astra Zeneca, Medtronic, outside the submitted work. D Cernik, R Jura, L Jurak, S Ostry, P Sevcik, O Skoda, D Skoloudik, and D Vaclavik report no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: R Mikulik was supported from the project no. LQ1605 from the National Program of Sustainability II (MEYS CR) and from the project FNUSA-ICRC no. CZ.1.05/1.1.00/02.0123 (OP VaVpI); R Herzig from the project no. DRO – UH HK 00,179,906 from the Ministry of Health of the Czech Republic and by the grant project PROGRES Q40 from Charles University, Czech Republic. J Neumann was supported from grant no. 16-30965A from the Czech Health Research Council.
Informed consent
Data have been collected as part of routine clinical practice; therefore there was no need for specific informed consent. Also approval of Ethics Committees for data collection was not required. The Ethical Committee of St. Anne’s University Hospital in Brno however approved the analysis of the data.
Ethical approval
Because the data have been collected as part of routine clinical practice, therefore approval of Ethics Committees for data collection was not required. The Ethical Committee of St. Anne’s University Hospital in Brno, however, approved the analysis of the data.
Guarantor
RM.
Contributorship
RM had full access to all the data in the study and took responsibility for the integrity of the data; the accuracy of the data analysis and he also drafted the manuscript, and designed the figures. RM, MB, DC, RH, RJ, LJ, JN, DS, SO, PS, OS, DS, DV, AT implemented the study and constantly collected data, critically revised the manuscript for intellectual content. RM, MB, DC, RH, RJ, LJ, JN, DS, SO, PS, OS, DS, DV, AT also approved and discussed the results and commented on the manuscript.
Acknowledgements
The authors would like to thank all stroke centres, physicians, nurses, CT technicians, coordinators, and administrators for contributing to the effort to improve stroke care quality as well as for provision of data from stroke centres. We would like to thank Steven Simsic for grammar correction.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
