Abstract
Introduction
Previous studies showed insufficient control of cardiovascular risk factors (CVRF) and high stroke recurrence rates among ischemic stroke patients in Germany. Currently, no structured secondary prevention program exists in clinical routine. We present the trial design and pilot phase results of a complex intervention to improve stroke care after hospital discharge in Germany.
Patients and methods
SANO is a cluster-randomized trial with 30 participating regions across Germany aiming to enrol 2,790 patients (drks.de, DRKS00015322). Study intervention combines both structural and patient-centred elements. Study development was based on the Medical Research Council framework for complex interventions. In 15 intervention regions, a cross-sectoral multidisciplinary network is established to enhance CVRF control as well as detection and treatment of post-stroke complications. Recommendations on CVRF are based on high-quality secondary prevention guidelines. Study physicians use motivational interviewing and agree with patients on therapeutic targets. While hospitalised, patients also receive dietary counselling and a health-passport to track their progress. During regular visits, CVRF management and potential complications are monitored. The intervention is compared to 15 regions providing usual care. The primary endpoint is the combination of recurrent stroke, myocardial infarction and death assessed 12 months after enrolment and adjudicated in a blinded manner.
Results
Eighteen patients were enrolled in a pilot phase that demonstrated feasibility of patient recruitment and study procedures.
Conclusion
SANO is investigating a program to reduce outcome events after ischemic stroke by implementing a complex intervention. If successful, the program may be implemented in routine care on national level in Germany.
Introduction
In Germany, there are currently about 260,000 strokes occurring each year. 1 Acute care is provided on a high qualitative level within the settings of more than 300 stroke units (SUs) with dedicated certification procedures and quality management strategies. 2 However, stroke recurrence rates among German ischemic stroke (IS) patients are high compared to other Western European countries.3,4 An observational study based on health insurance data showed a one-year recurrence rate of almost 10%. 5 In two other studies conducted in Germany, recurrence rates of 3.5% and 5.6%, respectively, were found within 90 days after hospital treatment.6,7 Rehospitalisation rates within 90 days are substantial with up to 10.3%. 7 Nevertheless, only few regional projects offer limited access to cross-sectoral structured post-stroke care for dedicated subgroups of stroke patients. Of note, no standardised screening for post-stroke complications such as depression and cognitive decline is implemented in post-stroke care in Germany, which can delay timely treatment.8,9
According to literature, between 50% and 80% of secondary stroke events might be prevented by optimal treatment of cardiovascular risk factors (CVRFs) with high contribution to the population attributable risk including hypertension, hyperlipidemia, atrial fibrillation, smoking, unhealthy diet and low physical activity.10,11 However, studies from routine clinical care in Germany showed deficits in appropriate CVRF control among stroke patients.6,12 This might be partially attributed to structural properties of the German health-care systems, such as the sectoral division between acute hospital and outpatient care often causing challenges as an insufficient transition process. 13
Trials outside of Germany have investigated the effectiveness of post-stroke care programs to reduce the rate of secondary cardiovascular events with divergent results.14,15 However, such programs need to be adapted according to the specific requirements of national health-care systems. Currently, no standardised large-scale post-stroke care program is implemented in Germany.
We present the trial design and results of the pilot phase from the structured ambulatory post-stroke care program ‘Strukturierte ambulante Nachsorge nach Schlaganfall’ (SANO) currently being tested in Germany and aiming to improve stroke care in first-ever IS after hospital discharge.
Methods
Study design
SANO is a parallel-arm cluster-randomised controlled trial with 30 participating clusters running from January 2019 until July 2021. A cluster is defined as the region, acute stroke care is provided by the participating SU. Clusters are eligible if acute stroke care is provided by a neurological hospital with a dedicated SU treating more than 500 acute stroke patients per year. Catchment areas of participating hospitals should not overlap and no other post-stroke care program should be regularly offered to participating patients to reduce contamination bias. Blinding of patients and physicians is not possible in this study since the intervention includes educational elements offered by the study team.
Recruitment
Consecutive acute stroke patients are screened after admission to hospital and recruited within 14 days after index stroke. Patients are eligible if they have a diagnosis of a first-ever IS (defined as either typical neurological symptoms present independent of their duration and stroke confirmed using computed tomography (CT) or magnetic resonance imaging (MRI) OR typical neurological symptoms present >24 h, for which no other cause than cerebral ischemia was possible, while intracerebral bleeding was excluded using CT or MRI) and at least one of the following pre-existing or newly diagnosed CVRFs: arterial hypertension (systolic values ≥140 mmHg or diastolic values ≥90 mmHg during hospitalisation); hypercholesterinemia (cholesterol levels >200 mg/dl in fasting blood sample); diabetes mellitus (measured glucose levels in venous fasting blood sample ≥126 mg/dl or HbA1c ≥6.5%); being a current smoker (regular consumption of tobacco products); or atrial fibrillation (AF) (AF documented for at least 30 s in a resting or long-term electrocardiogram). Patients are not eligible if they are severely disabled (modified Rankin scale (mRS) >3), suffer from severe cognitive impairment, severe aphasia or severe psychiatric disease or are unable to take part in the follow-up visits (for details, see Supplementary Material Table 1).
Recruitment is anticipated to take place over 12 months and each patient will be followed up for 1 year.
Randomisation
Randomisation was stratified by rural and urban regions according to the classification of the German Federal Institute for Research on Building, Urban Affairs and Spatial Development. 16 Large urban cities (>100,000 inhabitants) were classified as urban, all other participating centres as rural region. Each stratum was block-randomised in blocks of six using SAS Version 9.4.
Primary and secondary outcomes
We defined the combined outcome events of any recurrent stroke (both ischemic and haemorrhagic), myocardial infarction (MI) and death from any cause within the first year after stroke as primary endpoint. The outcome events will be adjudicated by an independent endpoint committee blinded to the allocation of individual patients.
Secondary endpoints include CVRF control according to predefined targets based on clinical guidelines (hypertension, diabetes, hypercholesterolemia, smoking, diet, physical activity), prescription of antiplatelets or oral anticoagulants, rate of hospital re-admissions, rate of stroke-related complications (cognitive decline, depression, anxiety and falls) and their management according to guidelines, appropriate provision of assistive devices, quality of life after 12 months, and event rates of any transient ischemic attack (TIA) (Supplementary Material Table 2). In addition, a cost–benefit analysis will be performed (Supplementary Material Document 1).
Development of the study intervention
Development of the intervention followed the recommendations of the Medical Research Council (MRC) Framework for complex interventions. 17
First, existing literature on multimodal secondary prevention interventions was summarised to guide the development of the complex intervention. Second, a systematic literature search was performed to identify high-quality guidelines on CVRF control. For this purpose, the Guidelines International Network (GIN), Medline and Tripdatabase were searched on 2 March 2018 (for more details, see Supplementary Material Tables 3 and 4). For each eligible guideline, quality assessment was performed by FAE and one medical student using the ‘German instrument to assess guidelines methodologically, domain 3’. 18 Whenever differences between the raters occurred, a third researcher with experience in guideline assessment was consulted.
In total, 325 guidelines were identified (GIN = 18, MEDLINE = 81 and Tripdatabase = 226). Twenty-one full-text papers were screened for eligibility, and 10 were included in the quality assessment. Seven guidelines achieved a quality score above 50% (Supplementary Material Table 5). Based on the recommendations of these guidelines, a team of neurologists developed a guidance book including chapters on all modifiable risk factors as well as antiplatelets, anticoagulants, carotid stenosis, post-stroke depression, cognitive impairment, fall prevention and assistive devices. Each chapter contains a summary of the current evidence-based guidelines on the topic, the therapeutic targets for the study, measures to achieve the targets, and when applicable, a description of routine assessment and evidence-based management of post-stroke complications.
Intervention program
Based on existing scientific literature, it was hypothesised that a combination of organisational and behavioural interventions is the most promising approach to improve stroke care after hospital discharge. Thus, the intervention consists of two main elements, the establishment of a multidisciplinary network and the delivery of an individual patient-centred intervention.
A cross-sectoral multidisciplinary network is established in each intervention region prior to study start (Figure 1). General practitioners (GPs), specialists and therapists are notified about the study with an information letter and invited to a local information event. Key to the network is the intensified exchange between local study physicians and the GPs. The respective GP is informed about his patient’s interest in taking part in the study and asked for his consent to participate. Regular contact between the study physician and the GP is maintained during the trial to exchange essential information. If a patient’s GP is not willing to participate, another treating physician such as the patient’s cardiologist is contacted instead. Furthermore, outpatient physicians, therapists, support groups, sports groups, social workers and providers of smoking cessation programs shall be included in the network.
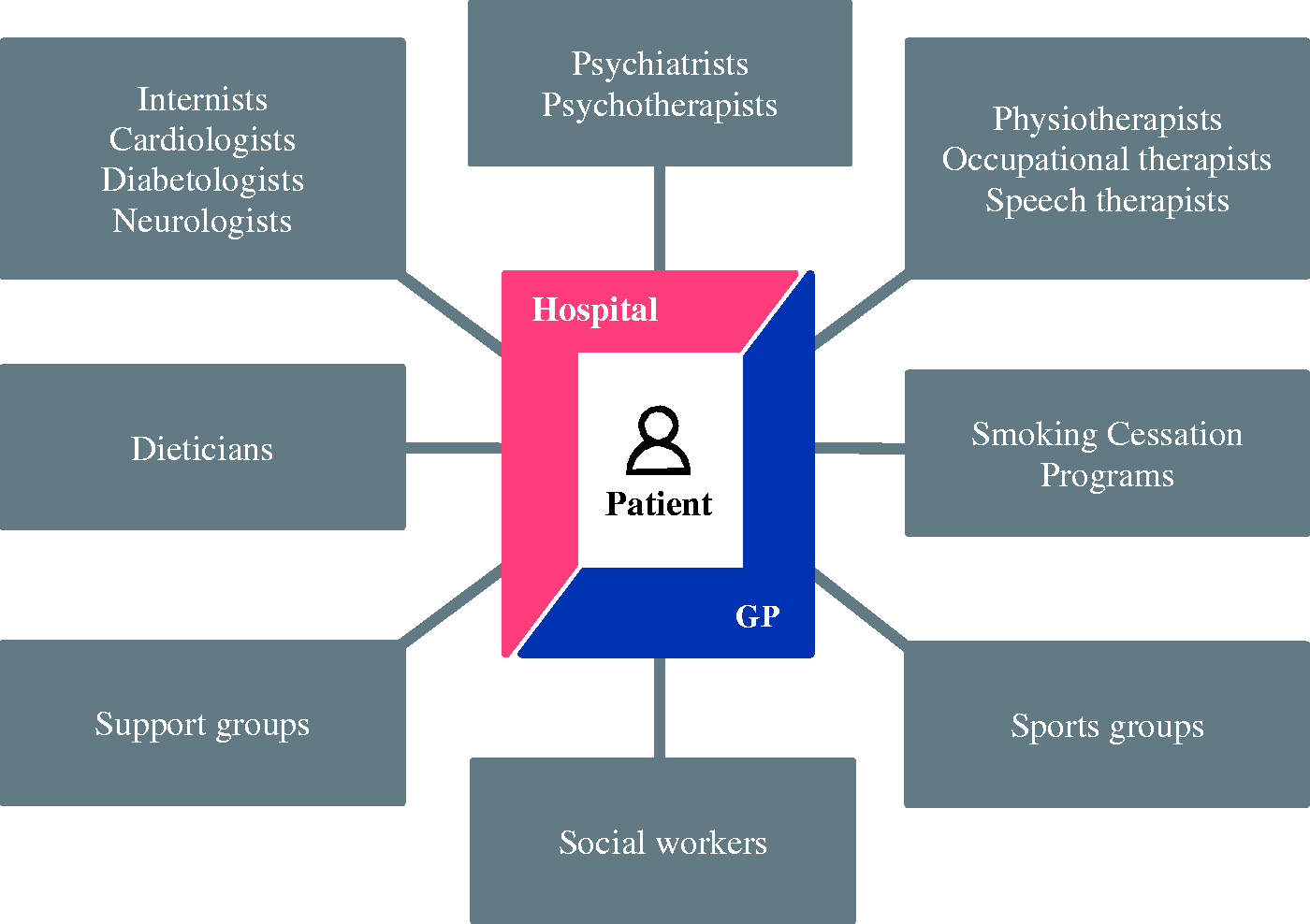
Graphical illustration of the cross-sectoral network supporting recovery and CVRF control. Local composition of the networks can differ and also include other professional groups; regular contact between the clinic and GP to coordinate treatment and monitor the patient’s progress.
A process evaluation designed on the basis of MRC guidance and an assessment of implementation fidelity of the intervention and its influence on outcome variables as well as obstacles identified by the study teams will be conducted (Supplementary Material Table 6). 19
In addition, the following patient-centred elements are part of the intervention (Figure 2):
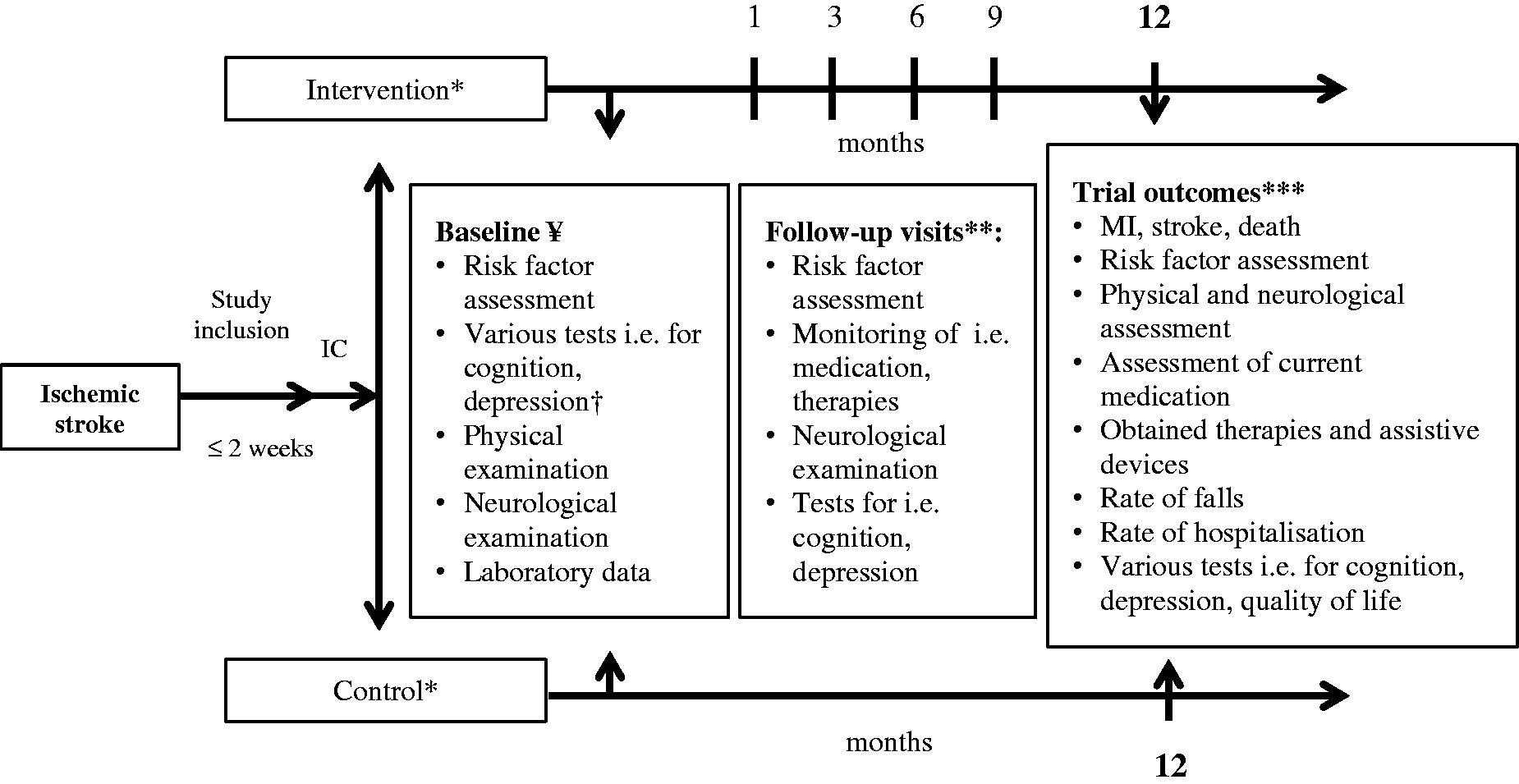
Trial course of control and intervention group in the SANO trial. IC: informed consent; *cluster-randomisation; **intervention group only; *** outcome assessment, rate of falls and hospitalisation refer to the first 12 months after the index event; † screening for risk of falls in intervention group only. ¥ the following questionnaires and scales are used during the baseline assessment and regularly during follow-up: National Institutes of Health Stroke Scale, modified Rankin Scale, Barthel-Index, The Montreal Cognitive Assessment, PHQ9, PHQ-GAD7, EQ-5D-5L, International Physical Activity Questionnaire, Food frequency questionnaire.
Patient education on present CVRFs and their best management according to current guidelines.
Defining therapeutic targets for each CVRF together with the patient and, whenever possible, the patient’s next of kin.
Use of the ‘motivational interviewing’ approach to support self-motivated decision-making on therapeutic targets.
Dietary counselling prior to hospital discharge.
A ‘health-passport’ for the patient containing essential information about the patient-specific stroke aetiology, a list of individual CVRFs and therapeutic targets.
Follow-up visits at the hospital 1, 3, 6 and 9 months after stroke to provide additional support to achieve adjustment of CVRFs.
Data collection
All patients receive a baseline examination by the study team while still hospitalised (Figure 2), during which the following data are collected: sociodemographic factors, past medical history, alcohol and tobacco consumption, quality of life, physical activity, dietary habits, anthropometry (weight, height, waist and hip circumference), standardised pulse and blood pressure measurement, and neurological status. Furthermore, structured screening by instrumental scales is performed for the following potential complications: cognitive impairment, depression and anxiety. Lastly, medical data about the index stroke (stroke aetiology according to the TOAST criteria, diagnostics, therapeutic interventions) and existing comorbidities are collected. During the follow-up visits, screening for post-stroke complications is performed in a standardised manner (Figure 2) and treatment options are discussed with the patient.
Potential side effects and adverse events such as hypotensive crisis are monitored in the intervention group during follow-up and assessed in both groups after 12 months.
After 12 months, primary and secondary endpoints are assessed during a final visit primarily by an independent, but not a blinded physician. If personal participation is not possible, a telephone interview can be carried out instead. If patients are not reachable, the relatives or the residents’ registration office will be contacted if consent for this was given.
Sample size calculation
From an aggregated data set of a follow-up examination of the Rhineland-Palatinate stroke registry, an event rate of 11% in the control group after one year was estimated as well as an intra-class correlation coefficient (ICC) of 0.0001. 7 Based on the aforementioned data and the work of Leistner et al., 20 it is assumed that a structured follow-up program can reduce the rate of a secondary vascular event (MI, stroke or death) by 30%, representing an absolute risk difference of 3.3%. Using a two-sided unpooled z-test for cluster-randomised studies, the null hypothesis of equal rates shall be tested.
For 15 clusters per group with an ICC of 0.0001, 83 stroke patients per cluster achieve a power of 80% to detect a significant absolute risk difference of 3.3% between control- and intervention regions with a significance level of 5%. Assuming a dropout rate of 10%, the total number of patients needed across 30 clusters is therefore 2790 patients (93 per cluster).
Statistical analysis
For the primary outcome, a univariable mixed-effect model will be used modelling the correlation within clusters using random effects. The effect of ‘therapy group’ will be included as a fixed effect, the effect of ‘clinics’ as a random effect. All secondary analyses are carried out exploratively using a significance level of 5%. The analyses of secondary endpoints are based on further univariable and multivariable mixed-effect models. A possible difference in follow-up costs between the two groups is examined with the two-sample t-test for independent samples. All analyses are performed based on intention-to-treat. For the pilot phase, descriptive analyses of the results were performed using R version 3.5.1.
Pilot phase
A pilot study was conducted in two regions (Ludwigshafen, Würzburg) prior to study start between July and December 2018. The aim of the pilot phase was the assessment of recruitment rates, testing of case report forms (CRFs) and questionnaires especially regarding length and comprehensibility for the patient, estimating the duration of the baseline assessment and the one-month follow-up visit, and receiving feedback from GPs regarding their willingness to participate in the study.
Sub-study
In four participating centres (Ludwigshafen, Schweinfurt, Würzburg, Bad Neustadt), a cross-sectional sub-study is conducted investigating to what extent stroke patients understand the full context of the informed consent procedure and whether understanding differs with regard to age, sex and stroke severity (‘Querschnittsstudie zur Abfrage von Inhalten eines Aufklärungsgespräches zu einer Sekundärpräventionsstudie nach ischämischem Schlaganfall’ (INA)). Results of the sub-study will be published independently.
Results
Randomisation and training of centres
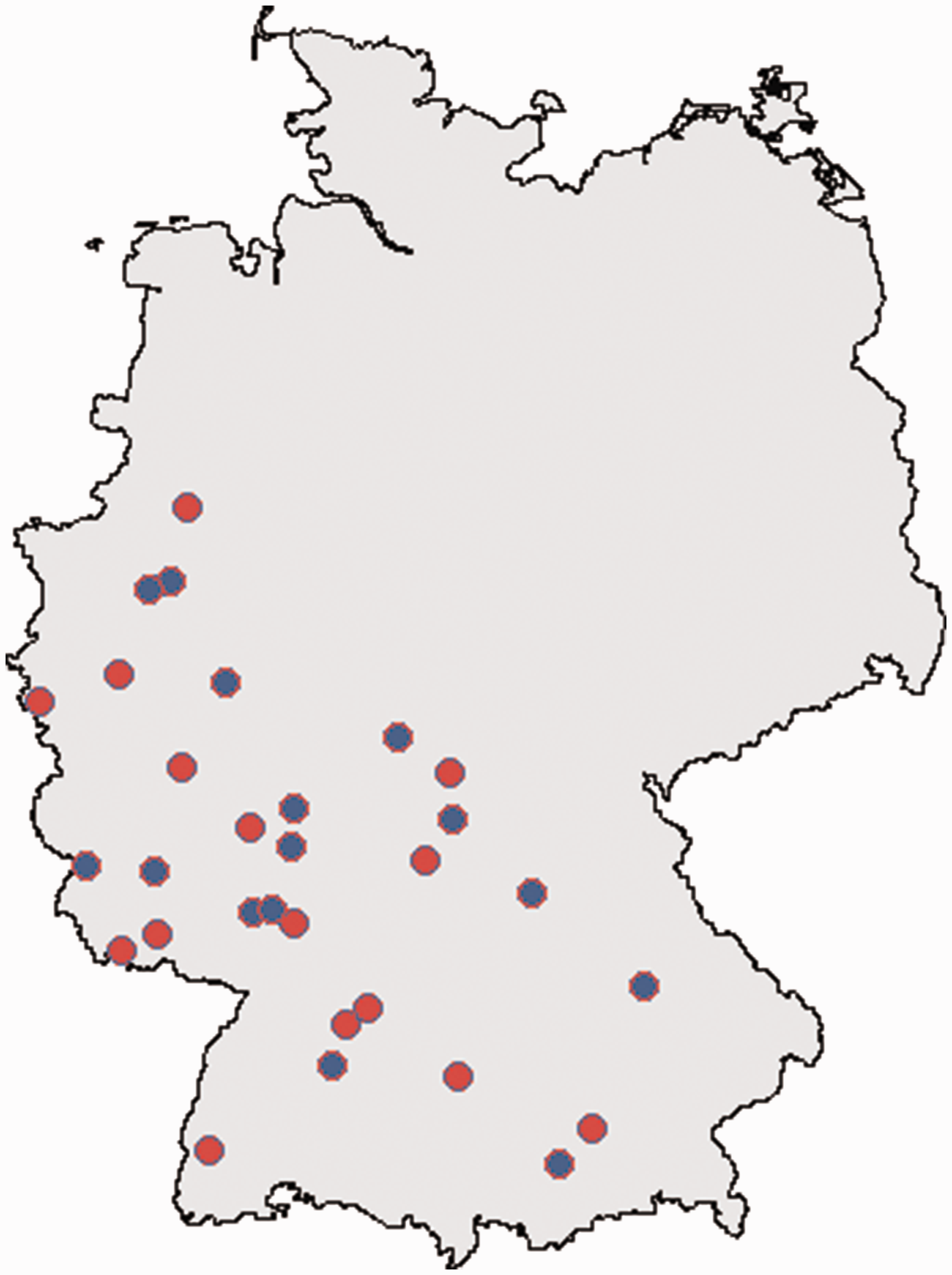
Thirty clusters have been successfully recruited to the study (Figure 3, Supplementary Material Document 2). Group allocation into 15 intervention and 15 control centres was finalised in October 2018. All centres randomised to the intervention received a two-day training covering all aspects of the study intervention. In another two-day meeting, the study teams were trained in the technique of motivational interviewing. Centres of the control regions received an one-day training only for the electronical CRFs and questionnaires used.
Pilot study
Eighteen patients were included in the pilot study (mean age 66 ± 12 years, 72% men) (Supplementary Material Table 7). Recruitment rate based on N = 154 patients screened was 12%. Of all eligible patients, 85% agreed to participate.
Of all patients, 13 had the duration documented for each baseline assessment module. Mean duration of the baseline assessment was 131 ± 47 min and mean duration of the IC procedure was 15 ± 7 min. Among all patients, 94% stated to be satisfied or very satisfied with the organisation of the assessment. Furthermore, 95% of the patients found the comprehensibility of the questionnaires good or very good. The feedback of the GPs contacted was overall positive.
Ten patients (Ludwigshafen only) were offered an additional follow-up after one month. Of these patients, 90% participated (mean age 67 ± 11 years, 78% men). Mean duration of the follow-up assessment was 64 ± 13 min with about half of the time being used to discuss individual CVRF targets.
Discussion
This manuscript describes the development and feasibility testing of a structured cross-sectoral post-stroke care program in Germany. The intervention was developed by an interdisciplinary team and is based on high qualitative secondary prevention guidelines identified by a systematic search.
A successful pilot phase was conducted to test feasibility of patient recruitment and study procedures. It showed that the conduct of the main trial is feasible while timely recruitment of the planned sample size will have to be closely monitored.
High stroke recurrence rates on an international level accompanied by comparably high rates of post-stroke complications within Germany substantiate the urgent need to improve post-stroke care.5,7 The SANO program is designed to overcome existing barriers of in- and outpatient health-care provision in Germany. 13 As ambulatory stroke care is mainly driven by GPs, this trial puts the collaboration between comprehensive stroke centres and GPs in the centre of its approach. Thereby, a high grade of expertise in acute stroke diagnosis and treatment is combined with the irreplaceable individual expertise in ambulatory treatment. The capabilities of ambulatory neurological care will have to be considered for the future implementation into standardised post-stroke care. Transitional models will be continuously evaluated and implementation fidelity monitored during trial course in order to design a realistic model for the implementation into standard care.
In a review summarising trials on secondary prevention interventions, only nine studies included patients with an exclusive diagnosis of IS, while six studies included both ischemic and haemorrhagic stroke patients and 19 studies included both all stroke and TIA patients. 14 However, the effect of a post-stroke intervention is likely to be associated with the stroke subtype. For example, TIA patients are per definition not disabled after the event and, thus, these patients have fewer difficulties to understand and follow study interventions such as increase of physical activity compared to stroke patients with physical and mental constraints. 21 Developing a standardised intervention for a heterogeneous group of stroke patients therefore poses several difficulties. Thus, the study team of SANO decided to focus on IS patients only.
Furthermore, many studies that assessed interventions for CVRF control had only short follow-up periods of up to six months. 14 It is, however, known that the uptake and maintenance of behavioural changes often take longer than a few months, hence the assessed effect of lifestyle changes on study outcomes in these trials should be treated with caution. 22 Behavioural changes are likely even more difficult for stroke patients due to specific neurological symptoms or stroke-related sequelae such as fatigue, depression or cognitive disturbances.8,23
To implement comprehensive secondary prevention strategies after stroke, physicians and study nurses often receive study-specific training for the intervention prior to study start. However, present knowledge among study personnel of the control group will likely cause contamination bias. 24 A cluster-randomised approach is likely to reduce this contamination bias, which is, thus, applied in the SANO trial. Previous trials often used a parallel group trial design with individually randomised patients, which could have led to underestimated effect estimates. 14 Furthermore, implementation fidelity to the intervention as initially intended including adherence to the content, coverage, frequency and duration of the provided intervention as well as the quality of delivery might have significant effect on the success of program. Therefore, implementation fidelity will be closely monitored during the trial. In conclusion, the SANO trial is conceptionally and methodologically capable to overcome limitations previously discussed for another RCT on secondary prevention after stroke, the INSPiRE-TMS, which has recently been published. 25
Several other aspects identified as beneficial in the review from Bridgwood et al. have been integrated into the SANO trial. First, a multidisciplinary team approach in form of a network building was applied. Second, both elements of patient education and regular monitoring of CVRF targets were included. Third, clinical staff at the intervention centres was trained in motivational interviewing which can lead to improved medication adherence.14,26
As advised for the development of a complex intervention, we used the MRC framework approach, 17 which allowed for a systematic development of the intervention. Besides, as the project is funded by the ‘Innovationsfonds’, quick implementation into routine care is to be expected if study results are positive.
Some limitations exist regarding our study. First, whilst our follow-up period is longer than in several similar studies, it might be too short to assess long-term effects. Therefore, optional consent to an extended follow-up is obtained from all participants and follow-up will be prolonged if subsequent funding becomes available. Second, due to the large number of post-stroke complications and to keep the duration of study visits at an acceptable level, data on frequent complications such as pain and fatigue were not collected during all visits in a standardised manner. However, these topics could still be personally discussed with the patients. Third, the international generalisability of our program is limited due to differences in health-care systems between countries. However, the results of the study can be generalised to other areas in Germany and other countries may get valuable insights from the methodological development of the intervention. Fourth, minor overlaps between catchment areas of the participating SUs might occur. However, the SUs are regionally grown structures and eligible centres were made aware of this criterion early on to discuss possible obstacles. Fifth, for methodological reasons, the program is limited to a subgroup of patients not too severely disabled. If the intervention proves to be effective, the applicability of the concept to a rather heterogeneous cohort of stroke patients and other subtypes should be tested in future trials. Lastly, only SUs treating more than 500 stroke patients per year could participate in this trial. Thus, the results might only partly be generalisable to smaller SUs.
Conclusion
A structured and evidence-based program seems to be promising to improve stroke care after hospital discharge in Germany. SANO is a new approach in this field and may have the potential to reduce secondary outcome events after IS by combining multiple elements and using recommendations of evidence-based guidelines.
Depending on the study results, elements of the post-stroke care program may be implemented on national level.
Supplemental Material
ESO910596 Supplemental Material1 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material1 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material2 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material2 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material3 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material3 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material4 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material4 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material5 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material5 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material6 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material6 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material7 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material7 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material8 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material8 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Supplemental Material
ESO910596 Supplemental Material9 - Supplemental material for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO)
Supplemental material, ESO910596 Supplemental Material9 for Trial design and pilot phase results of a cluster-randomised intervention trial to improve stroke care after hospital discharge – The structured ambulatory post-stroke care program (SANO) by Felizitas A Eichner, Christopher J Schwarzbach, Moritz Keller, Karl Georg Haeusler, Gerhard F Hamann, Dirk Sander, Heinrich J Audebert, Klaus Gröschel, Dieter Geis, Stephan von Bandemer, Viktoria Rücker, Martha Schutzmeier, Peter Ulrich Heuschmann, Armin Grau and The SANO Study Group in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FAE, CJS, MK, GFH, DS, DG, SvB, VR, MS and AG declare that they have no conflict of interest. KG reports personal fees and non-financial support from Bayer AG, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo and Pfizer outside the submitted work. HJA reports speaker fees and consultancy honoraria received from Bayer Vital, Boehringer Ingelheim Pharma, Bristol-Myers-Squibb, Pfizer and Takeda. KGH reports study grants by Bayer and Sanofi-Aventis, lecture fees/advisory board fees from Bayer, Sanofi-Aventis, Pfizer, Bristol-Myers-Squibb, Boehringer Ingelheim, Daiichi Sankyo, Biotronik, Medtronic, W. L. Gore & Associates, EIP Pharma and Edwards Lifesciences. PUH reports grants from German Ministry of Research and Education, German Research Foundation, European Union, Federal Joint Committee (G-BA) within the Innovationfonds, Charité -Universitätsmedizin Berlin, Berlin Chamber of Physicians, German Parkinson Society, University Hospital Würzburg, Robert Koch Institute, German Heart Foundation, University Göttingen (within FIND-AF randomized, supported by an unrestricted research grant to the University Göttingen from Boehringer-Ingelheim), University Hospital Heidelberg (within RASUNOA-prime, supported by an unrestricted research grant to the University Hospital Heidelberg from Bayer, BMS, Boehringer-Ingelheim, Daiichi Sankyo), and grants from Charité -Universitätsmedizin Berlin (within Mondafis, supported by an unrestricted research grant to the Charité from Bayer).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Innovationsfonds des Gemeinsamen Bundesausschusses (grant number 01NVF17032).
Ethical approval
The study was approved in May 2018 by the central ethics committee in Mainz (registry number 2018–13184). Local ethical approval from the University Hospital Würzburg was granted in August 2018 (registry number 149/18). All recruiting centres have obtained approval by the local ethics committees before study initiation.
Informed consent
Written informed consent was obtained from all subjects from the pilot phase and the main trial before the study.
Guarantor
AG.
Contributorship
All authors conceived the study. FAE, CJS, MK, MS, PUH and AG researched literature and were involved in protocol development and conceiving of the pilot phase. VR was involved in protocol development. FAE, CJS and MK were involved in coordination of the study. FAE wrote the first draft of the manuscript with support of CJS. All authors reviewed and edited the manuscript and approved its final version.
Acknowledgements
The authors would like to thank all 30 participating study centres and their dedicated teams for their excellent work (Supplementary Material Document 2). The authors also thank Prof. Otto Busse and Prof. Peter Schlattmann to operate the Data Scientific and Monitoring Board, the sections Rheinland-Pfalz, Baden-Württemberg and Bayern of the German Association of General Practitioners for their support, Stephanie Stangl and the medical students Andreas Berlin, Tassilo Dege, Rebecca Geiser, Tobias Gulde, Tobias Haas, Marcel Raub and Anika Quilitzsch for their support to assess the prevention guidelines and Susanne Streib, Martina Haase, Udo Selig and Timo Ludwig for their administrative support. Lastly, the authors thank all patients willing to participate in the pilot and the main phase of the SANO trial. This study was presented at the 5th European Stroke Organisation Conference (ESOC) in Milan, Italy, 22–24 May 2019.
Supplemental Material
Supplemental material for this article is available online.
Appendix: Members of the SANO Study Group
Prof. Hansjörg Bäzner, PD Dr. Andreas Binder, PD Dr. Martin Eicke, Prof. Klaus Fassbender, Prof. Gereon Fink, Prof. Martin Grond, Prof. Karl Georg Häusler, Prof. Gerhard Hamann, Dr. Fabian Hillenbrand, PD Dr. Bernd Kallmünzer, Dr. Moritz Keller/Dr. Marina Oberrauner, Dr. Martin Köhrmann, Prof. Rainer Kollmar, Prof. Matthias Maschke, Prof. Jens Minnerup, Dr. Johannes Mühler, Dr. Mariacristina Musso, Prof. Tobias Neumann-Haefelin, Dr. Sven Poli, Gernot Reimann, Prof. Peter Ringleb, Prof. Dirk Sander, Prof. Felix Schlachetzki, Prof. Jörg Schulz, Dr. Hassan Soda, PD Dr. Annette Spreer, Prof. Thorsten Steiner, Prof. Kristina Szabo, Prof. Helge Topka, Prof. Johannes Wöhrle.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
