Abstract
Background:
The Colchicine for prevention of vascular inflammation in Non-CardioEmbolic stroke (CONVINCE) trial evaluated long-term treatment with colchicine for the prevention of major adverse cardiovascular events (MACE) in a stroke population. Although the intention-to-treat analysis did not demonstrate a significant reduction in the primary endpoint, fewer outcome events were observed in the colchicine-treated group. It is unknown if a potential treatment effect is modified by ischemic stroke etiology.
Aims:
In this pre-specified secondary analysis, we aimed to evaluate the efficacy of colchicine for prevention of MACE in patients with minor stroke or high-risk transient ischemic attack (TIA) according to index event stroke etiology.
Methods:
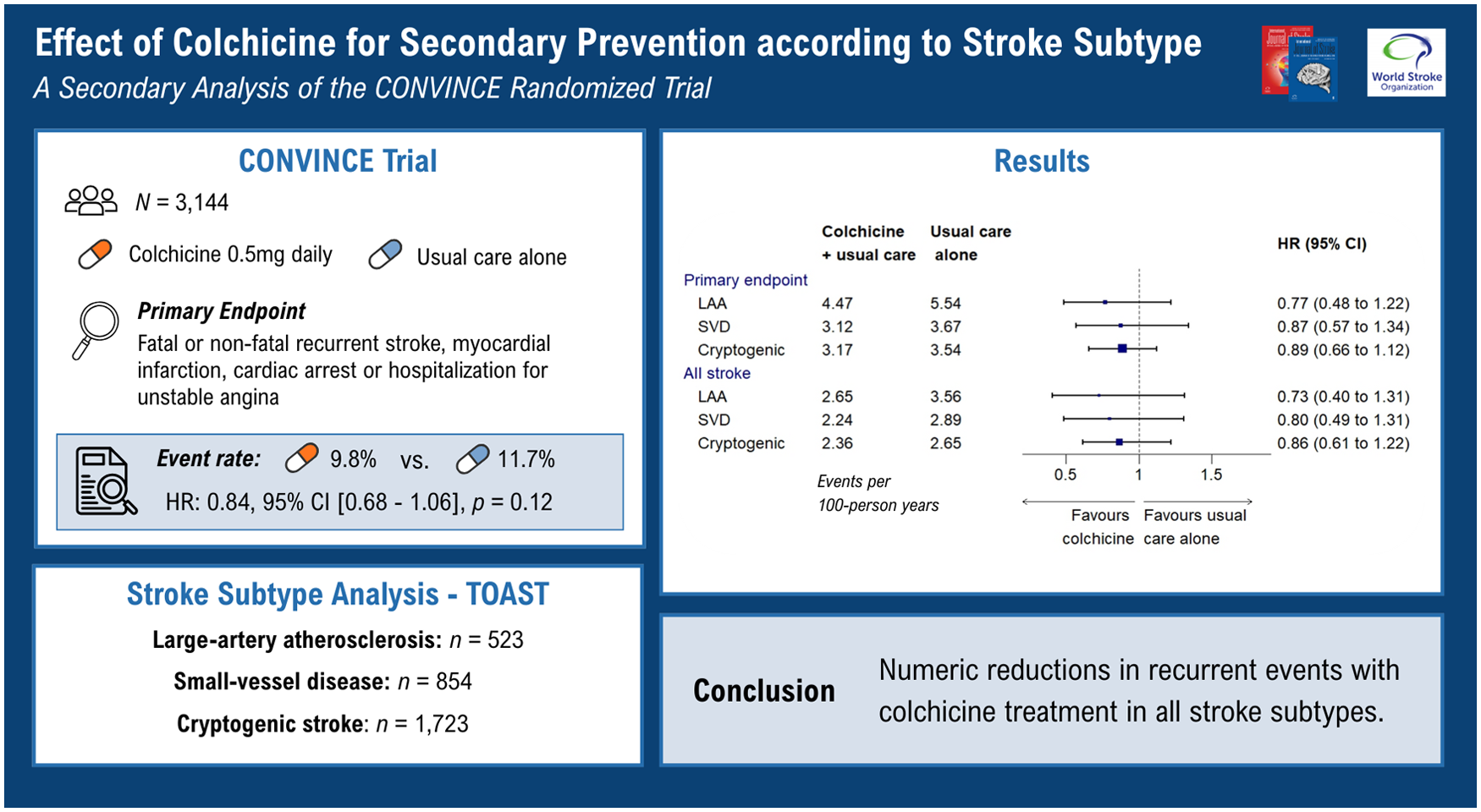
A total of 3154 patients with recent non-cardioembolic stroke or TIA were randomly assigned to receive colchicine, 0.5 mg daily in addition to guideline-based usual care or usual care alone. The primary endpoint was a composite of first fatal or non-fatal recurrent ischemic stroke, myocardial infarction, cardiac arrest, or hospitalization for unstable angina. Subgroups of patients with large-artery atherosclerosis, small-vessel disease, and cryptogenic stroke were evaluated.
Results:
A total of 3100 patients were included in the current analysis. The treatment effect did not vary across stroke subtype subgroups (p = 0.64 for interaction). In patients allocated to colchicine versus usual care alone, the primary endpoint occurred in 32 of 260 (12.3%) versus 42 of 263 (16%) patients with large-artery atherosclerosis (hazard ratio (HR), 0.77 (95% CI, 0.48–1.22)); 39 of 419 (9.3%) versus 47 of 435 (10.8%) patients with small-vessel occlusion (HR, 0.87 (95% CI, 0.57–1.34)); and 82 of 877 (9.4%) versus 92 of 846 (10.5%) patients with cryptogenic stroke (HR, 0.89 (95% CI, 0.66–1.12)).
Conclusions:
The direction of effect for prevention of recurrent MACE favored colchicine, consistent with randomized trials in coronary disease, regardless of stroke subtype. Future stroke trials should consider selecting patients with evidence of atherosclerosis irrespective of stroke subtype.
Trial Registration:
ClinicalTrials.gov Identifier: NCT02898610
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
