Abstract
Abstract
Introduction
About one-fourth of ischaemic strokes are classified as embolic strokes of undetermined source. Lambl’s excrescences are commonly seen on cardiac valves, and data are limited on whether they may be a source of embolization. We examined the relationship between Lambl’s excrescences and embolic stroke of undetermined source.
Patients and Methods
We performed a case-control study of patients in the Cornell AcutE Stroke Academic Registry. Stroke aetiologies were adjudicated using the Trial of Org 10172 in Acute Stroke Treatment and embolic stroke of undetermined source criteria. We included patients with acute ischaemic stroke between 2011 and 2016 who underwent transthoracic or transoesophageal echocardiography within six months of hospitalisation. Cases were embolic stroke of undetermined source patients and controls were patients with an identified, non-cardioembolic stroke aetiology (i.e. small- or large-vessel strokes). Multiple logistic regression was used to evaluate the association between Lambl’s excrescences and embolic stroke of undetermined source after adjustment for demographics, comorbidities and mode of echocardiography.
Results
A total of 923 patients met the criteria for this analysis, including 530 with embolic stroke of undetermined source and 393 with small- or large-vessel strokes. Lambl’s excrescences were identified in 47 (8.9%) patients with embolic stroke of undetermined source and 11 (2.8%) patients with small- or large-artery strokes, but the majority (54/58) of Lambl’s excrescences were visualised on transoesophageal echocardiogram and embolic stroke of undetermined source patients were more likely to undergo transoesophageal echocardiogram. After adjustment for demographics, comorbidities and mode of echocardiography, we found no association between the presence of Lambl’s excrescences and embolic stroke of undetermined source (odds ratio 0.9; 95% confidence interval 0.4–2.3).
Conclusion
We found no association between Lambl’s excrescences and embolic stroke of undetermined source. These results do not support the hypothesis that Lambl’s excrescences are an occult cause of embolic stroke of undetermined source.
Introduction
Embolic strokes of undetermined source (ESUS) are non-lacunar, embolic-appearing cryptogenic strokes that comprise approximately 25% of all ischaemic strokes.1,2 ESUS is more common in younger patients and has a substantial rate of stroke recurrence. 3 As a result, there is increasing investigation to better understand embolic sources underlying ESUS with the hope of further tailoring preventive treatments to reduce stroke recurrence.
Lambl’s excrescences are filiform projections of fibrous tissue found along the line of valve closure and are thought to result from endothelial degeneration.4–6 Estimates of their presence on cardiac valves ranges from 5% to 40%.6–8 While many cases of Lambl’s excrescences are asymptomatic, some studies have suggested an association between these valvular strands and stroke.5,7,9–16 It is hypothesised that stroke from Lambl’s excrescences is secondary to thrombi formation on the valve in the setting of an underlying hypercoagulable condition or embolization of the strand itself. 4 However, it is unclear whether Lambl’s excrescences are the cause of the stroke or an incidental finding, as patients with Lambl’s excrescences frequently have accompanying cardiac and atherosclerotic risk factors.6–8,10,17,18 If Lambl’s excrescences are a currently underrecognised cause of stroke, Lambl’s excrescences should be more common in ESUS cases than in strokes of non-cardioembolic aetiology. Current data are conflicting regarding whether Lambl’s excrescences are more frequent in ESUS compared to other stroke subtypes.7–10,12,19 We therefore examined this association in a large, prospective ischaemic stroke registry.
Methods
Study design
We performed a case-control study of patients in the Cornell AcutE Stroke Academic Registry (CAESAR), a prospective registry of patients diagnosed with acute stroke at a large teaching hospital and comprehensive stroke centre in New York City. Details on the registry have been previously published. 20 For this analysis, we included all patients 18 years or older with acute ischaemic stroke who were admitted to our institution between 1 January 2011 and 31 December 2016 and who underwent a transthoracic echocardiogram (TTE) or transoesophageal echocardiogram (TEE) during the index hospitalisation or within six months of presentation. Since Lambl’s excrescences persist unchanged over time, we chose a six-month cut-off to allow adequate time for obtaining basic and advanced outpatient cardiac imaging. Stroke aetiologies were ascertained per the Trial of Org 10172 in Acute Stroke Treatment classification system and ESUS definition by a panel of neurologists. 21 For this analysis, all patients with cardioembolic stroke or stroke from other determined aetiologies were excluded. Patients classified as cryptogenic stroke because of multiple stroke aetiologies or incomplete workups were excluded from the ESUS group. Our final cohort consisted of all patients with ESUS and, as controls, all patients with small- or large-artery strokes. This study was approved by the Weill Cornell Medicine Institutional Review Board with a waiver of the requirement for informed consent (protocol #1309014352). Deidentified data supporting this analysis will be shared by the corresponding author upon reasonable request.
Measurements
Our exposure variable was the presence of Lambl’s excrescences on either TTE or TEE, defined based on documentation in the official echocardiogram report. Patients that had both TTE and TEE done were classified according to their TEE results. Strands were defined as thin mobile filamentous projections attached to either the mitral or aortic valvular leaflets. We excluded cases of Lambl’s excrescences that were associated with torn chordae or flail leaflets to avoid misclassification. 9 This information was abstracted from echocardiography reports by two investigators (SSO and SC) blinded to patients’ stroke subtype. Our main outcome of interest was ESUS. To account for confounding factors in the association between Lambl’s excrescences and stroke subtype, we identified the following vascular risk factors: age, sex, race/ethnicity, prior stroke, hypertension, dyslipidaemia, diabetes mellitus, coronary artery disease, peripheral vascular disease, valvular heart disease, congestive heart failure, chronic kidney disease, tobacco use and alcohol use.
Statistical analysis
Descriptive statistics with exact confidence intervals (CIs) was used to characterise the cohort. The chi-square test and the t-test were used to evaluate for differences in patient and clinical characteristics between groups. Multiple logistic regression was used to evaluate the association between Lambl’s excrescences and ESUS after adjustment for demographics, vascular risk factors and mode of echocardiography. While Lambl’s excrescences can be detected on TTE and TEE, TEE has been shown to have a higher sensitivity and specificity. 17 We also performed analyses stratified by mode of echocardiography. Statistical significance was set at a two-sided α of 0.05. All statistical analyses were performed by SSO and HK using Stata/MP, Version 14 (College Station, TX).
Results
Of the 2116 patients registered in CAESAR with acute ischaemic stroke from 2011 to 2016, 923 (mean age, 67.9 years; 48.2% women) met eligibility criteria and were included in this analysis (Figure 1). Among the final cohort, 530 (57.4%) were classified as having ESUS. The control group consisted of 393 patients (161 with small-vessel occlusion and 232 with large-artery atherosclerosis). Patients with ESUS were more often female, had fewer stroke risk factors and were more likely to undergo TEE (Table 1). Lambl’s excrescences were identified in 58 patients. Patients with Lambl’s excrescences were younger and had fewer stroke risk factors such as hypertension (Table 2). Of the 207 patients who underwent TEE, Lambl’s excrescences were seen in 54 (26.1%) patients. Among the 887 patients who had a TTE, Lambl’s excrescences were observed in only five (0.6%) patients. One patient had Lambl’s excrescences demonstrated on both TTE and TEE.
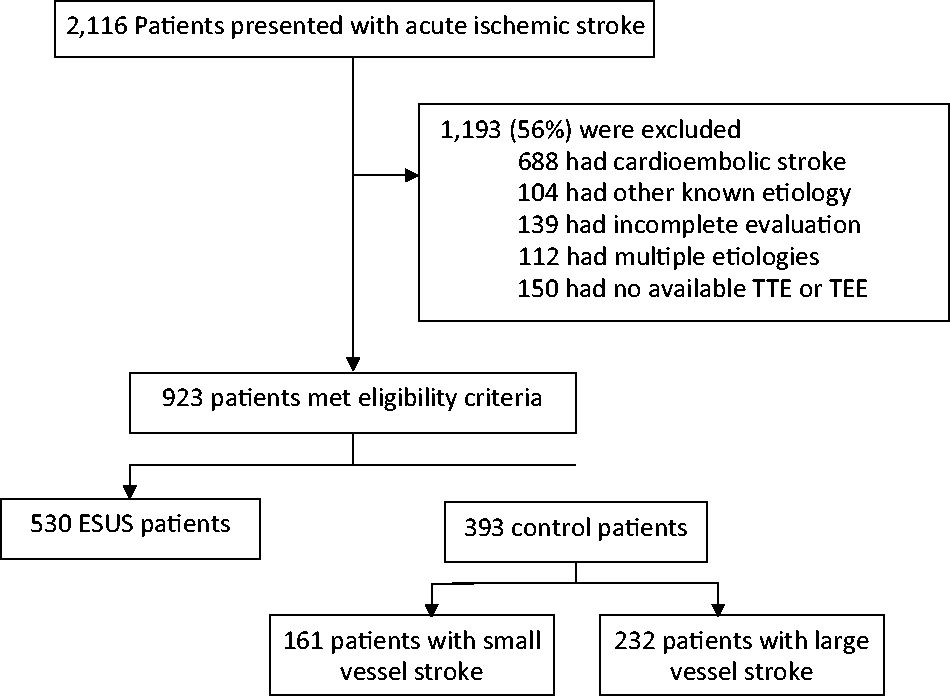
Flow diagram depicting the flow of all the patients within the Cornell AcutE Stroke Academic Registry.
Characteristics of patients stratified by embolic stroke of undetermined source.
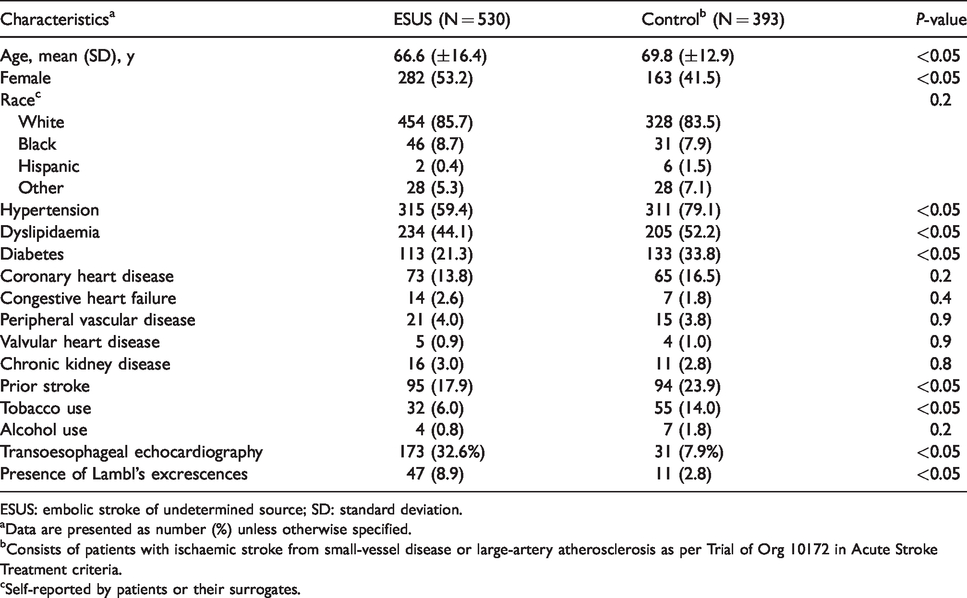
ESUS: embolic stroke of undetermined source; SD: standard deviation.
aData are presented as number (%) unless otherwise specified.
bConsists of patients with ischaemic stroke from small-vessel disease or large-artery atherosclerosis as per Trial of Org 10172 in Acute Stroke Treatment criteria.
cSelf-reported by patients or their surrogates.
Characteristics of patients stratified by the presence of Lambl’s excrescences.
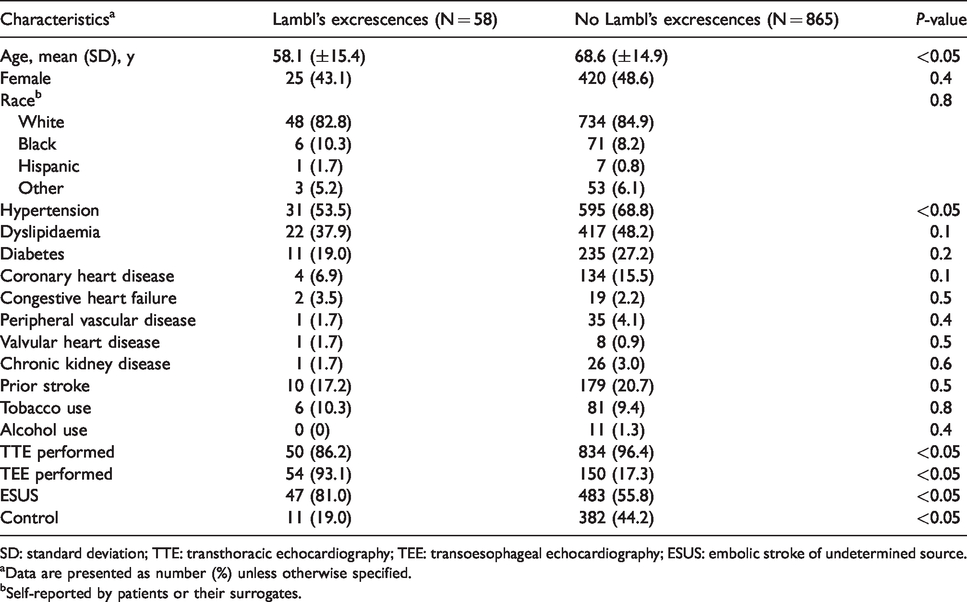
SD: standard deviation; TTE: transthoracic echocardiography; TEE: transoesophageal echocardiography; ESUS: embolic stroke of undetermined source.
aData are presented as number (%) unless otherwise specified.
bSelf-reported by patients or their surrogates.
Lambl’s excrescences were present in 47 (8.9%) ESUS patients and 11 (2.8%) control patients (P < 0.001) (Table 2). Unadjusted analysis found an association between Lambl’s excrescences and ESUS (odds ratio (OR), 3.8; 95% CI, 1.8–8.3). After adjustment for demographics, vascular risk factors and mode of echocardiography, there was no association between Lambl’s excrescences and ESUS (OR, 0.9; 95% CI, 0.4–2.3). We also found no association in stratified analyses of patients who only underwent TTE (OR, 3.1; 95% CI, 0.3–28.1) or TEE (OR, 0.7; 95% CI, 0.3–1.7).
Discussion
In an analysis of patients at a large academic centre, we found that Lambl’s excrescences were not more prevalent among ESUS cases compared to small- or large-vessel strokes. Our findings build on previous analyses on this topic. Several studies have reported an association between Lambl’s excrescences and stroke; however, the association between Lambl’s excrescences and ESUS is less clear. A few studies have reported that Lambl’s excrescences are more common in patients with cardioembolic or other established embolic causes of stroke,8,10 while others have reported similar rates of Lambl’s excrescences among cryptogenic and non-cryptogenic strokes.7,12 Potential reasons for these conflicting findings include different definitions for cryptogenic stroke and small sample sizes that might have limited the ability to perform robust statistical analyses to evaluate associations. A large study by Homma et al. examined prospectively collected data on stroke patients assigned to either warfarin or aspirin and found similar rates of Lambl’s excrescences in patients with cryptogenic and known causes of stroke. 19 Our study builds on these findings by only including the subset of cryptogenic stroke patients with ESUS, hence increasing the likelihood that the embolic events could potentially be related to Lambl’s excrescences. In this context, our study indicates that this common echocardiographic finding is unlikely to be an underappreciated cause of strokes that currently lack a defined cause.
Our study must be considered in light of its limitations. First, given the single-centre academic setting of our cohort, the results may not generalise to other stroke populations. Second, we did not have centralised review of echocardiograms nor systematic adjudication of Lambl’s excrescences, which may have led to misclassification. Third, as indicated by the somewhat wide CIs, we lacked statistical power to detect small associations between Lambl’s excrescences and ESUS.
Conclusion
In a large cohort of patients presenting to a comprehensive stroke centre, we found no association between Lambl’s excrescences and ESUS when compared to patients with small- or large-artery ischaemic stroke. Our results suggest that Lambl’s excrescences are not a common source of embolic stroke in ESUS patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kamel serves on the Steering Committee for Medtronic’s Stroke AF trial, has served on an advisory board for Roivant Sciences and receives in-kind study support for the ARCADIA trial from the BMS-Pfizer Alliance and Roche Diagnostics. The remaining authors have no conflicts of interest. All authors abide by the ICMJE guidelines regarding the declaration of conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Salehi Omran is supported by the NIH Stroke Net grant U24NS107237. Navi is supported by the NIH grant K23NS091395 and the Florence Gould Endowment for Discovery in Stroke. Merkler is supported by the AHA grant 18CDA34110419 and the Leon Levy Fellowship in Neuroscience. Kamel is supported by the NIH/NINDS grants R01NS097443 and U01NS095869 and the Michael Goldberg Research Fund.
Ethical approval
This study was approved by the Weill Cornell Medicine Institutional Review Board with a waiver of the requirement for informed consent (protocol #1309014352).
Informed consent
The requirement for informed consent was waived.
Guarantor
HK.
Contributorship
SSO researched literature and conceived the study. SSO and SC were involved in data abstraction and analysis. HK was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. MPL, AEM and BBN were involved in acquisition, analysis and interpretation of data for the work. SSO wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Acknowledgements
The authors are grateful to Monica Chen for her copyediting and clerical assistance. No funding source was involved in the writing of this article. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
