Abstract
Purpose
The aim of this systematic review and meta-analysis is to determine the diagnostic accuracy of computed tomography brain perfusion in the prediction of haemorrhagic transformation and patient outcome in acute ischaemic stroke.
Method
Electronic databases and grey literature published over the last 10 years related to healthcare and radiology were searched using the key terms: ‘computed tomography perfusion’, ‘haemorrhagic transformation’, ‘acute ischaemic stroke’, ‘functional outcome’ and their synonyms using both UK and American spellings. Inclusion criteria were: sample size at least 30 patients, original research, evaluate ability of computed tomography perfusion to predict haemorrhagic transformation, reports diagnostic accuracy or provide relevant data for a 2 × 2 contingency table, use follow-up non-contrast computed tomography (NCCT) or magnetic resonance imaging as reference standard.
Findings
Twelve studies were included in the review; studies cover a total of 808 patients. Haemorrhagic transformation occurred in 30.2% of patients. Pooled sensitivity and specificity were 85.9% (95% CI; 65–97%), 73.9% (95% CI; 45–92%) and accuracy of 79.1% (95% CI; 57–98%). Pooled NPV was 92.9% with a high false positive rate (19.8%), which could be explained in terms of outcome classification, acquisition artefact and computed tomography perfusion processing algorithms.
Discussion
This review evaluated the importance of using pre-defined threshold measurement for optimal prediction of HT, the relevance of patient pre-treatment clinical parameters to HT occurrence, the CTP parameters and the measurements that are independent predictors of HT, the significance of rtPA rather as an exacerbator of HT and the impact of both minor and major HT/PH on patient 20 functional outcome.
Conclusion
Computed tomography perfusion has a high sensitivity and moderately high specificity for prediction of haemorrhagic transformation in acute ischaemic stroke. Pre-treatment clinical decision making requires consideration of clinical factors in addition to imaging findings. This systematic review and meta-analysis highlights that pre-treatment computed tomography perfusion adds to clinical confidence by predicting potential for haemorrhage, both in thrombolysed and un-thrombolysed patients, and also influences decisions about alternative treatments for acute ischaemic stroke patients.
Keywords
Introduction and literature review
Ischaemic stroke (IS) is associated with multiple risk factors which can be divided into modifiable (e.g. hypertension) and non-modifiable (e.g. race) categories. IS is caused by sudden blockage of the blood supply to a region of brain by a clot or acute stenosis. 1
When a patient is admitted with stroke symptoms, diagnostic imaging is performed to answer four critical questions; 2 (i) is there haemorrhage or a stroke mimic? (ii) can ischaemia or thrombus be identified? (iii) can an irreversibly damaged infarct core be identified? and (iv) can a salvageable tissue (penumbra) be identified?
Recombinant tissue plasminogen activator (rtPA), a thrombolytic drug, gives significant benefits to IS patients by reducing degree of disability. However, its administration is guided by knowing the interval since symptom onset – up to 4.5 h for intravenous administration (for good benefit and good functional outcome, but there are report of iv use up to 9 h) and up to 6–9 h using an intra-arterial route and measurable neuro-deficits, thus limiting the number of patients who can benefit. 3 rtPA also causes fibrinolysis and thus carry the potential to induce or increase haemorrhagic transformation (HT) of ischaemic lesions.4,5 HT occurs in up to 40% of patients and is frequently seen within the first week of stroke 6 causing rapid deterioration and poor functional outcome. 7
Since about 2007, there has been a shift from the traditional ‘time-based’ approach to intervention to a physiology-based approach. This reflects a better understanding of the haemodynamic changes that occur during arterial occlusion and specifically the identification of two distinct regions in the ischaemic hemisphere – irreversibly damaged tissue (infarct core) and potentially salvageable tissue (Penumbra). 8 This was made possible by advancements in neuro-imaging, especially the development of perfusion imaging. Perfusion imaging helps delineate the core-penumbra borders/mismatch. 9 The appearance of this penumbra on imaging differs from individual to individual. 10 The main aim of penumbra imaging is to identify the patients that are likely to improve following successful reperfusion from those that are at risk of complications, e.g. haemorrhage. 9 Penumbra imaging is a useful predictor of HT in acute IS. 11
Under normal circumstances, the blood–brain barrier (BBB) is impermeable to large molecules (e.g. blood cells); however, in the presence of pathology, e.g. neoplasm or ischaemia, BBB permeability increases with associated increased diffusion of large molecules into the extravascular space leading to haemorrhage and oedema. Loss of integrity of the BBB may play a role in the pathogenesis of HT in ischaemic lesions and is thought to occur as early as 3.5 h post onset. 12
HT is associated with poorer clinical outcome independent of thrombolytic therapy; it occurs 10 times more frequently in thrombolysed patients versus placebo. 10 The severity of HT is well correlated with functional outcome at three months, 13 patients with larger haemorrhages being at higher risk of more severe disability and death. 14 The ability to predict the likelihood of HT prior to thrombolytic intervention thus becomes more important as clinicians attempt to extend the potential benefits of therapeutic intervention across a larger number of patients.
Computed tomography (CT) has few contra-indications. It is relatively quick and cost-effective to perform and is an accessible imaging test for stroke patients. Several studies have shown correlation between CT perfusion (CTP) imaging and magnetic resonance imaging (MRI)8,15–19 and correlation of CTP penumbra appearances and permeability surface area measurements with stroke severity and HT.20,21 This paper presents a systematic review and meta-analysis evaluating the diagnostic accuracy of CTP to predict HT of acute ischaemia.
Methods
A broad and comprehensive search of MEDLINE, EMBASE, CINAHL, DARE, AMED, NICE Evidence, OVID, TRIP, COCHRANE AND Greylit electronic sources was performed. Hand searches of key radiology journals did not yield additional articles. A structured search strategy was adopted using the PICOS framework 22 to identify relevant key concepts, search terms and keywords including ‘computed tomography perfusion’, ‘haemorrhagic transformation’, ‘sensitivity’, ‘specificity’, ‘acute ischaemic stroke’, ‘functional outcome’ that were then adapted for each separate database.
A two-stage approach to study selection (initial screening of titles and abstracts against inclusion criteria and then screening of full text to identify relevant articles) 23 was undertaken to reduce subjectivity bias and the selection process was conducted using the PRISMA flowchart. 24 Inclusion criteria were (1) sample size at least 30 IS patients, (2) admission CTP performed, (3) CT or MRI as reference standard, (4) Diagnostic accuracy values of CTP were reported and (5) primary studies published between 2007 and 2017.
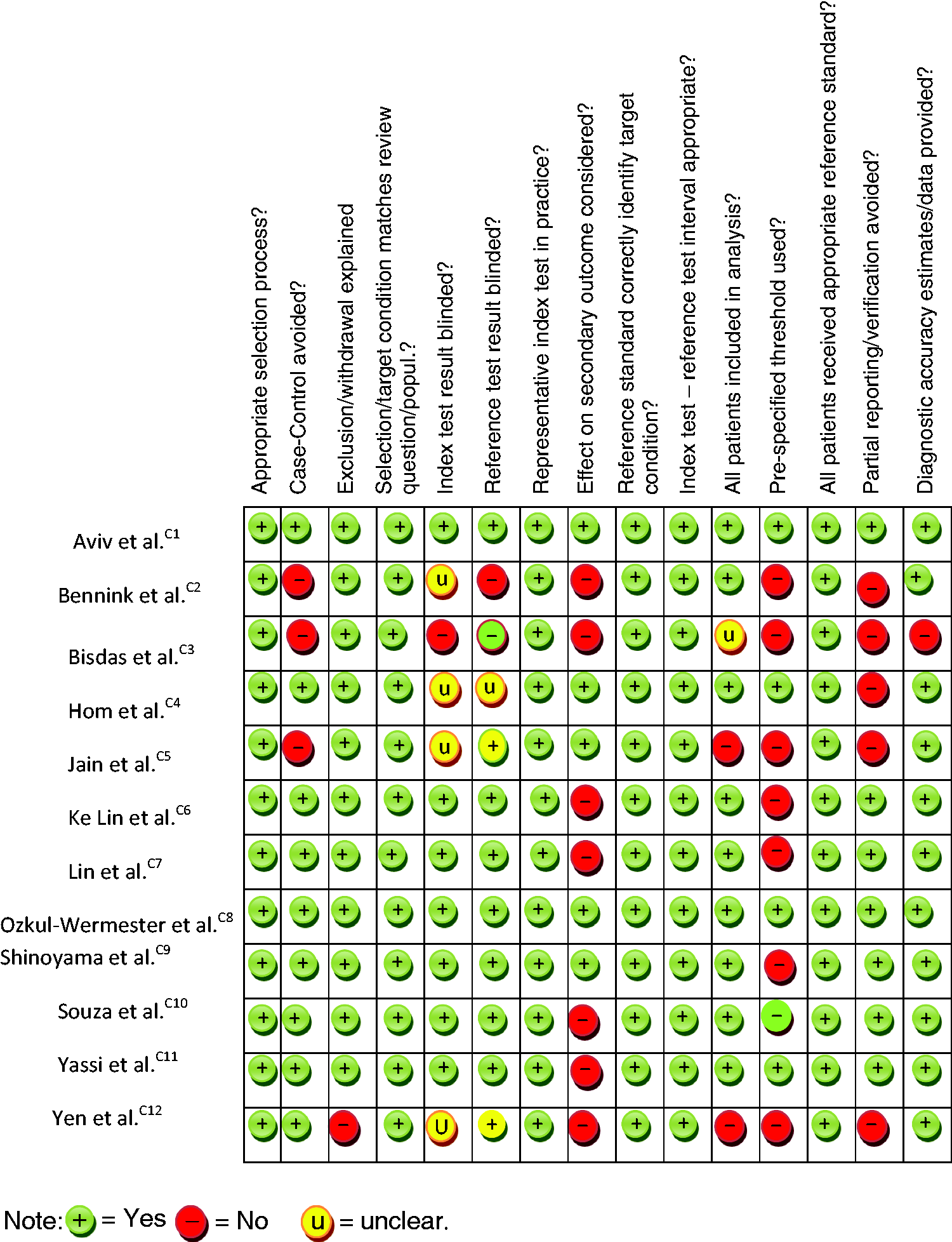
Methodological quality of selected studies was assessed by using CASP diagnostic test checklist 25 for methodological adequacy and the validity of results and the QUADAS 2 diagnostic accuracy checklist 26 was used to address the risk of bias and applicability. The CASP tool required slight modification with addition of few questions but the QUADAS tool was not modified.
The Cochrane diagnostic accuracy data extraction tool 27 was used to extract data in a consistent and standardised manner to eliminate subjectivity bias. Meta-analysis of extracted data was undertaken; combining the results of individual studies in a meta-analysis increases the power and precision in estimating the effects of intervention, narrows the confidence intervals and provide a greater chance of detecting a real effect as statistically significant. 23 RevMan v5.3™ and MedCal v17.5™ software were used to synthesise the collated data, for subgroup analysis and to generate forest plots and a summary receiver operating characteristic curve and the final diagnostic accuracy values.
Results
Search and selection ‘results’ – PRISMA chart
A total of 76,791 articles were initially identified and use of the inclusion/exclusion criteria narrowed this down to 12 studies which met the criteria (Figure 1).
Quality assessment results
Table 1 shows the assessment outcome using QUADAS2 tool. Overall, selected studies were of good methodological quality (See also additional online table i; outcome using CASP tool).
Risk of bias and methodological quality assessment using QUADAS 2 tool.

Characteristic of included studies
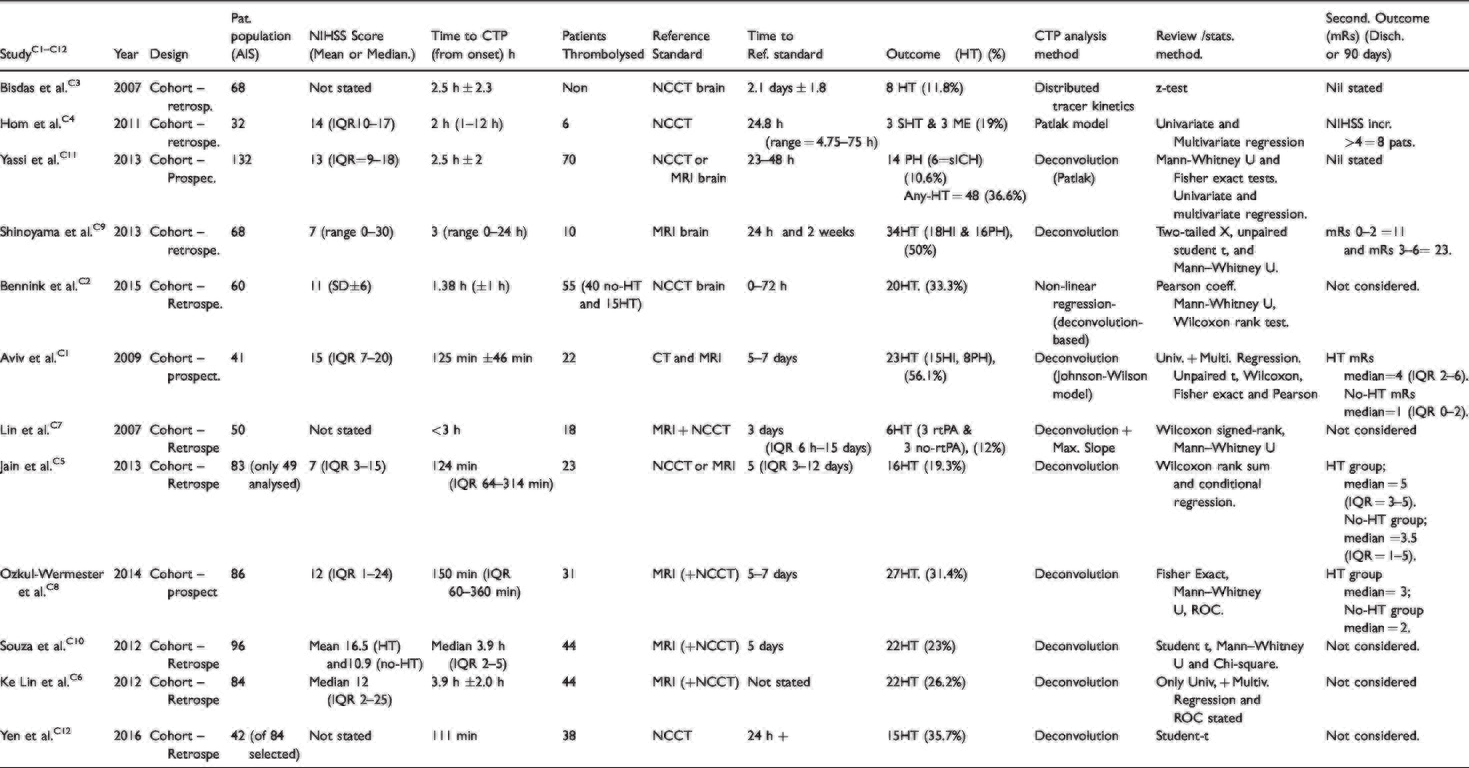
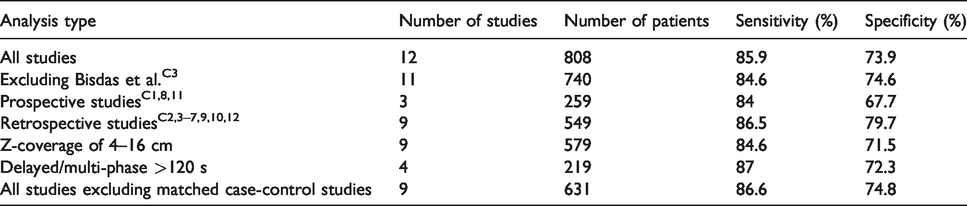
The 12 selected studies were published between 2007 and 2017; the studies were heterogeneous in terms of design, index test parameters, z-coverage, perfusion image analysis method and reported accuracy values. The characteristics of the 12 studies are summarised in Table 5.
Patient selection
Three studies selected patients prospectivelyC1,C8,C11 and nine selected them retrospectively.C2–C7,C9,C10,C12 The 12 studies collectively included a total of 874 patients although only 808 patients were accounted for in analysis; Jain et al. C5 excluded 34 patients because no matched-controls were found and Yen et al. C12 excluded 32 patients who had only MRI as follow-up. Average age of patients was 69.9 years (range33–93 years) and median admission NIHSS score was 11.4 (IQR 1–25).
Imaging protocols
Mean time from symptom onset to CTP was 2.5 h (±2.3 h; range 1–24 h). Four studies used NCCT alone as the reference standard, one study used MRI alone and seven studies used both NCCT and MRI. The mean time to reference standard imaging was 2.9 days (range 1–15 days). Thrombolysed patients tended to receive earlier reference standard/confirmatory test imaging.
Seven of the studies used ‘permeability surface area product’ (PS or relative PS) measurements for HT prediction although image acquisition time and technique varied. Souza et al. C10 suggested that for PS to correlate well with HT acquisition, time must be at least 120 s to allow for sufficient contrast leakage into extravascular space, and Lin et al. C7 argued that increased BBB permeability is possible within 2–4 h post ictus and should be detectable on admission CTP performed within this time. A summary of study imaging parameters is included in Tables 6 & ii (online).
All studies used low-osmolar non-ionic contrast media with similar molecular weights (800 mg/mol ± 22) giving relatively similar diffusion performance (permeability) across studies. 28
CTP software and analysis
All studies used deconvolution-based software except Bisdas et al. C3 which used custom-written software. Lin et al. C7 initially used Maximum Slope method to calculate parametric maps but subsequently used deconvolution method to calculate PS for HT prediction. In general, software algorithms were either tracer delay-sensitive, delay-insensitive or delay-corrected. A detailed discussion about these principles could be found in Kontas et al.2,29 and Abels et al. 30
The range of statistical tests used by each study is shown in Table 5 and Table 6 displays the regression analysis used to determine the parameters that are independent predictors of HT with a level of significance set at p < 0.05 and the diagnostic accuracy values (see also online table ii for review methods used by each study).
Eleven (92%) of the included studies used a combination of visual assessment and a pre-defined threshold to determine the optimal measurement(s) for predicting HT.
These thresholds are quantitative measurements (numbers) representing the point at which HT is deemed certain by the authors, and the value of which depends on the parameter being used for measurement, e.g. relative permeability surface area, infarct size, etc.
Primary and secondary outcome measures
All studies used HT as primary outcome measure except Yassi et al. C11 which used parenchymal haemorrhage (PH). Hom et al., C4 Ke Lin et al. C6 and Yen et al. C12 also considered whether HT (or PH) is symptomatic. Five studies (Hom, C4 Shinoyama, C9 Aviv, C1 Jain C5 and Ozkul-Wermester C8 ) considered the effect of HT on patient’s functional (2°) outcome, whether HT was minor, asymptomatic or symptomatic; overall concluding that patients with HT were worse off at discharge or at 90-day follow-up. Example of 2° outcome measures stated in selected studies includes disability (e.g. hemiplegia), dependence and death, and severity of 1° outcome correlates well with degree of 2° outcome. See also Table 2 for pooled numerical results of each 1° outcome. As shown, 30 patients (12.3%) had symptomatic HT/PH, with sICH being defined as a CT evidence of a bleed correlating with patient symptoms and neurological deterioration >4 on NIHSS. Table 3 shows the overall accuracy of CTP to predict symptomatic HT.
Number of HT, PH and symptomatic HT in included studies.
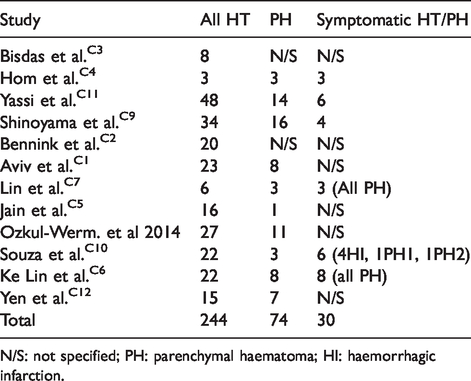
N/S: not specified; PH: parenchymal haematoma; HI: haemorrhagic infarction.
Accuracy of CTP to predict symptomatic HT (from included studies).

Thrombolysis/thrombectomy and HT occurrence
Across the 12 studies, 361 patients (44.7%) were thrombolysed (± thrombectomy), 134 (37.1%) of these developing HT and 227 shows no HT. On the other hand, 447 patients were not thrombolysed but 110 (24.6%) of these developed HT. Thus, HT occurred in 244 patients (30.2%) in total (Table 5).
Diagnostic accuracy analysis
Diagnostic accuracy of CTP to predict HT was given in 11 of the 12 studies. Bisdas et al. C3 stated only odds ratio and p-values; 2 × 2 contingency tables were constructed for each study with estimated values calculated from the ‘Mean (+/− SD)’ data in Bisdas et al. C3 This is an acceptable approach/practice according to CRD 28 and pooled values are further displayed via subgroup analysis – see Table 7.
The lowest sensitivity reported was 71.4% (Yen et al. C12 ), and the highest being 100% (Hom et al., C4 Shinoyama et al. C9 and Bisdas et al. C3 ). The lowest specificity was 41%, C8 and (Ozkul-Wermester et al.C8)), the highest was 100% (Lin et al. C7 ).
Figure 2 shows the study sensitivity and specificity with 95% confidence intervals and paired forest plots (see also online fig. i MedCal v17.5 software display of diagnostic accuracy). Figure 3 shows pooled estimates of sensitivity and specificity in the receiver operating characteristic curve (ROC) space.
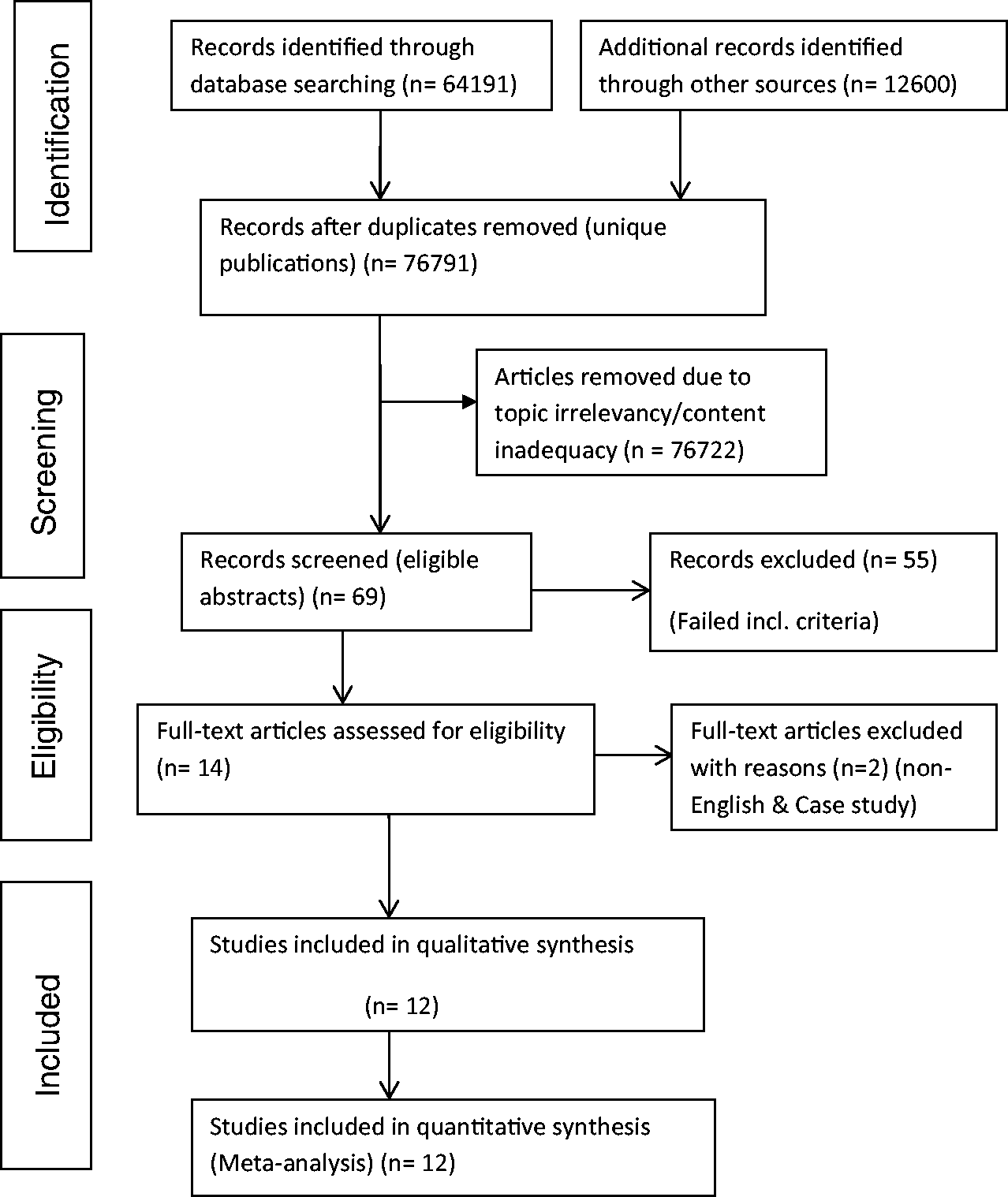
PRISMA (2009) search flow diagram for eligible articles.33
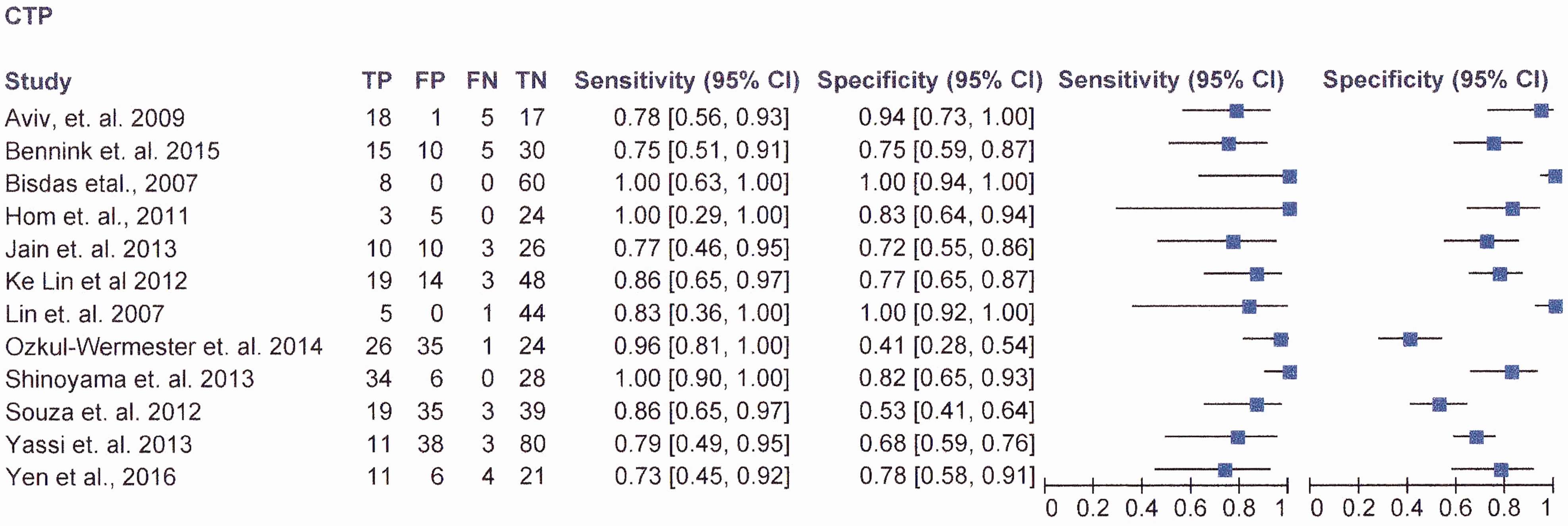
RevMan v5.3 software data.
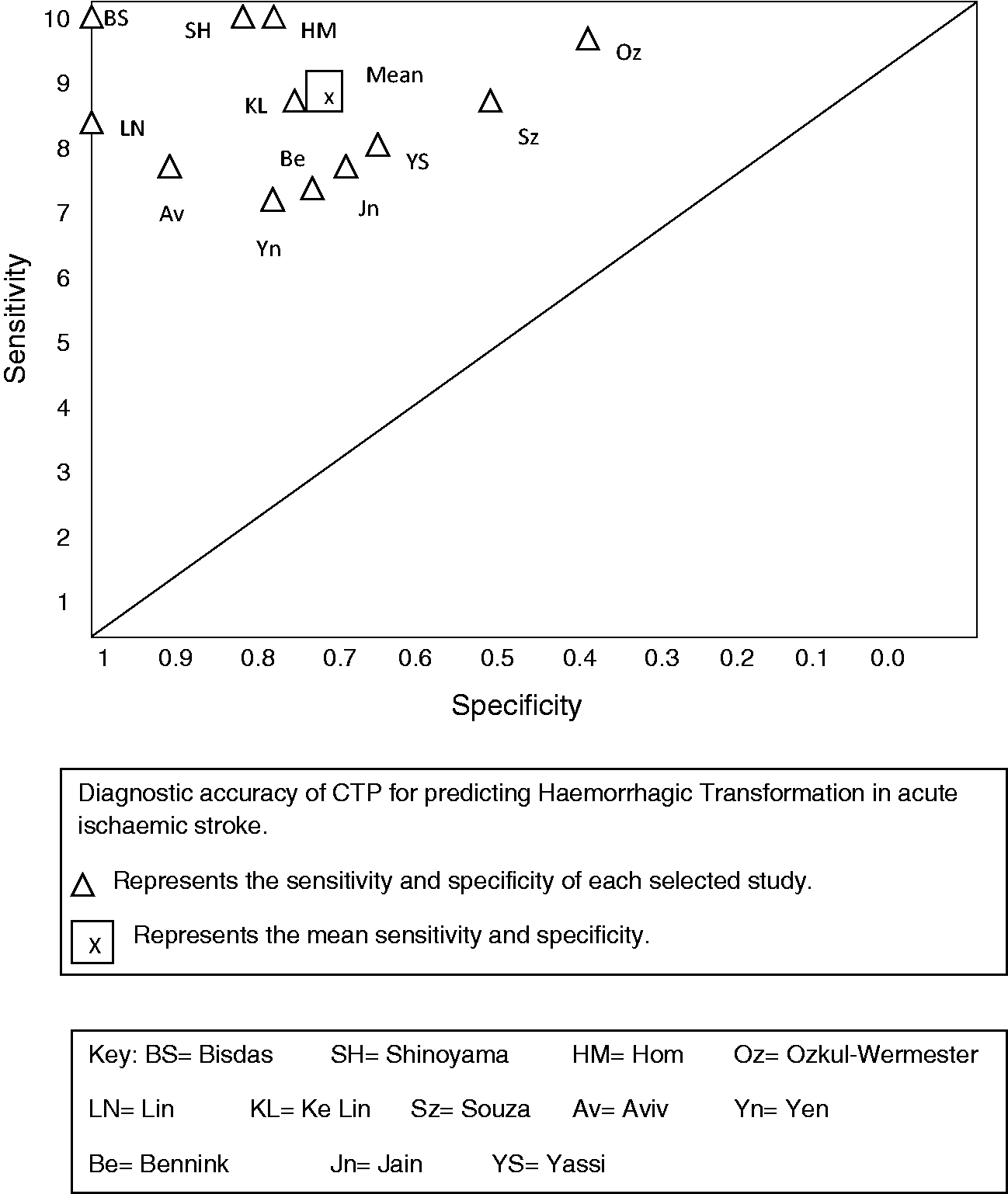
Summary sensitivity and specificity in ROC space.
Table 4 shows the result of Multivariate regression analysis performed by authors to determine independent predictors of HT (or PH) (and their threshold).
Multivariate analysis result.
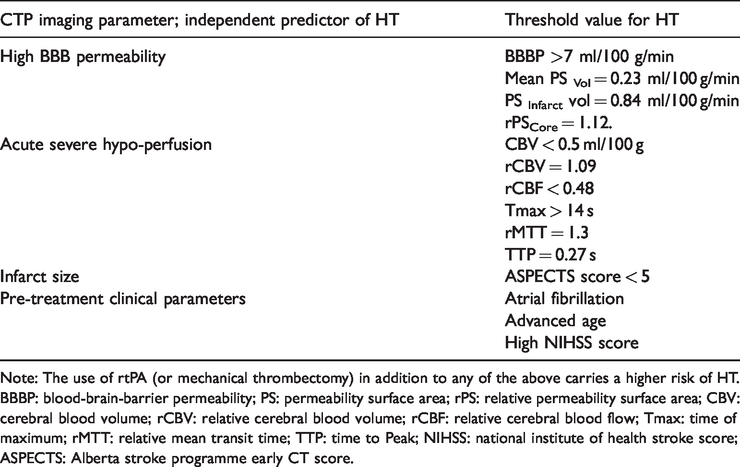
Note: The use of rtPA (or mechanical thrombectomy) in addition to any of the above carries a higher risk of HT.
BBBP: blood-brain-barrier permeability; PS: permeability surface area; rPS: relative permeability surface area; CBV: cerebral blood volume; rCBV: relative cerebral blood volume; rCBF: relative cerebral blood flow; Tmax: time of maximum; rMTT: relative mean transit time; TTP: time to Peak; NIHSS: national institute of health stroke score; ASPECTS: Alberta stroke programme early CT score.
Characteristics of included studies.
CTP outcome ± statistical analysis.
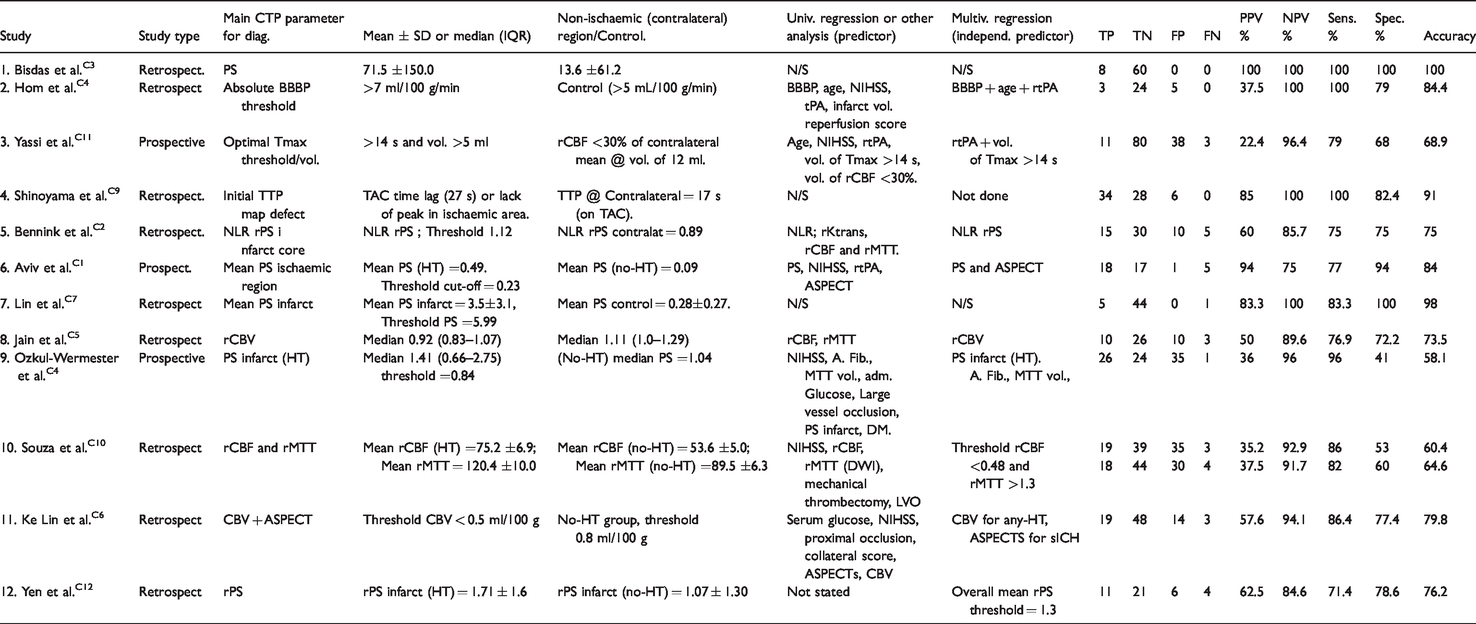
Pooled sub-group analysis.
From selected studies, overall odds ratio for HT is 2.319 (95%CI; 1.497–3.592; p = 0.0002) and relative risk ratio of HT is 1.938 (95%CI 1.348–2.788; p = 0.0004) (using MedCal v17.5) (OR = Bad outcome TP, FN vs. Good outcome TN, FP and RR = Experimental bad outcome TP, good outcome TN vs. Control bad outcome FN, good outcome FP).
There was no statistically significant difference in patients who had HT whether they received rtPA or did not (37.1% thrombolysed (p = 0.0019) vs. 24.6% un-thrombolysed (p = 0.0104)). However, there was a statistically significant difference between patients who received rtPA and then developed HT compared to those who received rtPA but did not develop HT (37.1% (p = 0.0019) vs. 62.9% (p = 0.998) respectively).
Discussion
This systematic review and meta-analysis demonstrated high sensitivity (85.9%, 95% CI; 65–97%) and moderately high specificity (73.9%, 95% CI; 45–92%) for CTP in predicting HT (overall accuracy of 79.1%). The high NPV (92.9%) and lower PPV (60.3%) suggested that despite high sensitivity, factors such as patient clinical status were also important consideration at thrombolysis decision point. This is reflected by the high false positive rate (19.8%; 160 of the 808 patients). On the other hand, the false negative rate is very low (3.5%; only 28 patients). Thus, the power of this index test lies in its high negative predictive value.
However, the review demonstrated methodological heterogeneity in selected studies including CTP data acquisition and analysis technique, index test measurement, outcome classification and some inappropriate exclusion. CTP parameters varied amongst the studies included in this review but the most commonly used diagnostic measurement was the permeability surface area product (PS or rPS). Eleven of the 12 included studies used a combination of visual assessment and a pre-defined threshold to determine the optimal measurement(s) for predicting HT. In all cases whatever CTP parameter was used, the location of measurement which produced the optimal threshold corresponded to the region of the most acute BBB injury, the highest permeability value or region of severe hypo-perfusion, and thus the region most likely to undergo HT. Another important advantage of the optimal threshold approach was that they represented the point at which HT was deemed certain (by the investigators) irrespective of whether or not the patient received thrombolysis, and thus are important markers for clinicians. There was also heterogeneity in study population, and these include age (range 33–93 years), gender (51% male), pre-treatment clinical factors (HTN, AF, Diabetes, use of antiplatelets, high admission glucose, etc.) and stroke severity measured by admission NIHSS score (Median was 11.4). These factors were given consideration in many of the selected studies because of their impact on risk of HT after rtPA and/or thrombectomy.
The parameters and thresholds used by these selected studies using regression analysis to determine the independent predictors of HT (or PH) include; CTP imaging appearance suggestive of high BBB permeability (i.e. BBBP > 7 ml/100 g/min; mean PSvol. 0.23 ml/100 g/min; PSinfarct vol. 0.84 ml/100 g/min; rPScore = 1.12); acute severe hypoperfusion (i.e. CBV < 0.5 ml/100 g; rCBV = 1.09; rCBF < 0.48; Tmax > 14 s; rMTT.1.3; TTP 0.27 s); infarct size (ASPECTS < 5) and pre-treatment clinical parameters; Atrial Fib., advanced age and high NHISS score. Any of the above plus rtPA or mechanical thrombectomy carries a higher risk for HT occurrence.
In comparison, there is literature evidence of the potential use of MRI to predict HT. MR perfusion maps showing large diffusion-weighted imaging (DWI) lesion or low apparent diffusion coefficient (ADC), 15 extreme hypo-perfusion (low CBF, low CBV, high MTT, prolonged Tmax) showing as low signal intensity on T2*W (PWI) 31 imaging, and features of microbleeds on T2W–GRE or early contrast enhancement on T1W imaging are useful radiological biomarkers. However, there is lack of consensus on the specific imaging parameter that best predict HT but current trend favours parameters with perfusion imaging and setting a critical threshold beyond which prediction of HT has a high specificity and sensitivity (e.g. CBV > 2 mL/100 g, CBF > 4.9 mL/100 g/min or MTT > 145% of contra-lateral side).
Ten of the 12 studies recorded false positive (FP) and false negative (FN) cases. The remaining two studiesC3,C7 used first pass acquisition data at which time it is possible that significant BBB injury has not occurred. In some studies, FP and FN cases were modified by choice of primary outcome measure. For example, Hom et al. C4 selected symptomatic HT plus ECASS III benchmark of clinical deterioration and NIHSS increase of > 4. Five of the patients in their study who met these criteria had malignant oedema, pneumonia or septicaemia, rather than HT. However, in other studies, it was not possible to determine the reason for classifying cases as FP or FN.
Similarly, in the area of the effect of chosen outcome on patient prognosis, 8 of the 12 studies considered that any HT at all was an important contributor to patient morbidity and mortality and included all patients with HT. Two studies, Ke Lin C6 and Yen C12 considered only symptomatic or catastrophic HT as important but still included patients with minor HT in their analysis. Two studies, Yassi C11 and Hom C4 considered only PH2 or symptomatic HT as important and eliminated patients with minor HT from their analysis. The relevance for this review relates to the fact that minor/asymptomatic HT if not predicted in advance has the potential to progress to severe and symptomatic HT if thrombolytic therapy is administered, and the consequent worsening of these patients’ secondary functional outcome. Similarly, as argued by Aviv et al., C1 Lin et al. C7 and Shinoyama et al., C9 even asymptomatic HT (without rtPA administration) requires close monitoring of blood pressure and discontinuation of anticoagulant/antiplatelet therapies to prevent development of catastrophic HT.
Although previous studies4,5 have suggested that rtPA increases the risk of HT, in this review and meta-analysis we found no statistically significant difference in patients who had HT whether they received rtPA or not (37.1% thrombolysed vs 24.6% un-thrombolysed had HT). However, there was a statistically significant difference between patients who received rtPA and then developed HT and those who received rtPA but did not develop HT (37.1% vs. 62.9%). This suggests that rather than having a direct causative effect, rtPA is more likely to exacerbate HT – the patients who had HT after rtPA will already have a condition that predispose them to HT (e.g. high BBB permeability) and were either tipped-over the threshold or their condition exacerbated by rtPA administration. CTP imaging is thus useful to determine the pre-treatment perfusion characteristics of an individual patient’s ischaemic lesion. 32
This review has generated evidence that CT brain perfusion imaging can augment clinical confidence in stoke intervention decision making because of its ability to predict HT with high sensitivity and high negative predictive value.
We know that the extent of brain perfusion after an ischaemic event is individualistic in nature, that ischaemia induces injury to the BBB and that there is a potential for HT to develop in the most ischaemia-injured portion of the brain. In clinical practice therefore using CTP to both judge the extent of an individual’s perfusion status and predict the possibility of HT is a clinically reasonable approach to inform treatment option(s).
This review shows that both minor and major HT are significant for patients as both impact on functional outcome, and patients experiencing HT being worse-off at discharge or 90-day follow-up. Predicting even minor HT is important to clinicians because it influences their decision to either administer rtPA, or choose an alternative intervention and consider treatment choices for patients on other (co-morbid) medications before the stroke event.
Conclusion, recommendation and implication for practice
In conclusion, CTP is an accurate predictor of HT in acute IS patients and a useful prognostic tool for clinicians at the point of intervention decision making. It is therefore recommended that CTP be included in the initial (admission) imaging protocol of acute stroke patients. The technique could be readily incorporated into existing emergency CT protocols, and can be performed by most multi-slice scanners and adds minimal time (usually less than 10 min) to patient treatment for a clinically significant contribution to prognosis. CTP requires minimal additional resource by way of imaging staff training and/or scanner system upgrade.
Limitations and proposal for future research
This systematic review has some important limitations. First, the potential for a reviewer to erroneously interpret or report studies and for methodological failures. However, measures were taken to minimise these errors and bias by re-reading and double-checking every step of the review process and by conducting minor pilots where appropriate. Also, publication bias cannot be excluded as only English language articles were included. Similarly, majority of the selected studies were retrospective with the well-known inherent bias in such studies which may influence accuracy values. Future research may also consider the cost effectiveness of CTP against other imaging modalities.
Supplemental Material
ESO883461 Supplemetal Figure1 - Supplemental material for Diagnostic accuracy of computed tomography perfusion in the prediction of haemorrhagic transformation and patient outcome in acute ischaemic stroke: A systematic review and meta-analysis
Supplemental material, ESO883461 Supplemetal Figure1 for Diagnostic accuracy of computed tomography perfusion in the prediction of haemorrhagic transformation and patient outcome in acute ischaemic stroke: A systematic review and meta-analysis by Olushola D Adebayo and Gary Culpan in European Stroke Journal
Supplemental Material
ESO883461 Supplemental Tables - Supplemental material for Diagnostic accuracy of computed tomography perfusion in the prediction of haemorrhagic transformation and patient outcome in acute ischaemic stroke: A systematic review and meta-analysis
Supplemental material, ESO883461 Supplemental Tables for Diagnostic accuracy of computed tomography perfusion in the prediction of haemorrhagic transformation and patient outcome in acute ischaemic stroke: A systematic review and meta-analysis by Olushola D Adebayo and Gary Culpan in European Stroke Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for this article because it is a systematic review and meta-analysis of published research and does not involve the use of human or in vitro subject.
Informed consent
Informed consent was not sought for this article because it is a systematic review and meta-analysis of published research and does not involve the use of human or in vitro subject.
Guarantor
ODA.
Contributorship
ODA and GC conceived the study and researched literature. GC and ODA involved in protocol development, QA tools and data analysis. ODA involved in data extraction and quality assessment. ODA wrote the first draft of the manuscript. Both authors reviewed and edited the manuscript and approved the final version of the manuscript.
Acknowledgements
The reviewers wish to thank Dr David Ryan, Dr Cathy Dewhurst and Dr Marie Staunton for reviewing the piloting and extraction/QA tools and checking the accuracy of CTP information. And Dr Lee Crush for clinical mentoring and Mr Andrew Owen for facilitating the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.