Abstract
Introduction
Determining the cause of spontaneous (non-traumatic) intracerebral haemorrhage (ICH) is critical to guide treatment and prognosis. We investigated whether small vessel disease (SVD) in addition to clinical and other radiological findings on acute neuroimaging predicts a low risk of a macrovascular cause (e.g. an arterio-venous malformation, aneurysm or dural arteriovenous fistula).
Patients and methods
We identified patients with acute spontaneous ICH who underwent acute non-contrast CT, CT angiography (CTA) and intra-arterial digital subtraction angiography (IADSA) at our institution from January 2010 to April 2014. Logistic regression including CTA result, SVD, age, pre-ICH hypertension and ICH location was used to derive a prediction model, validated using bootstrapping.
Results
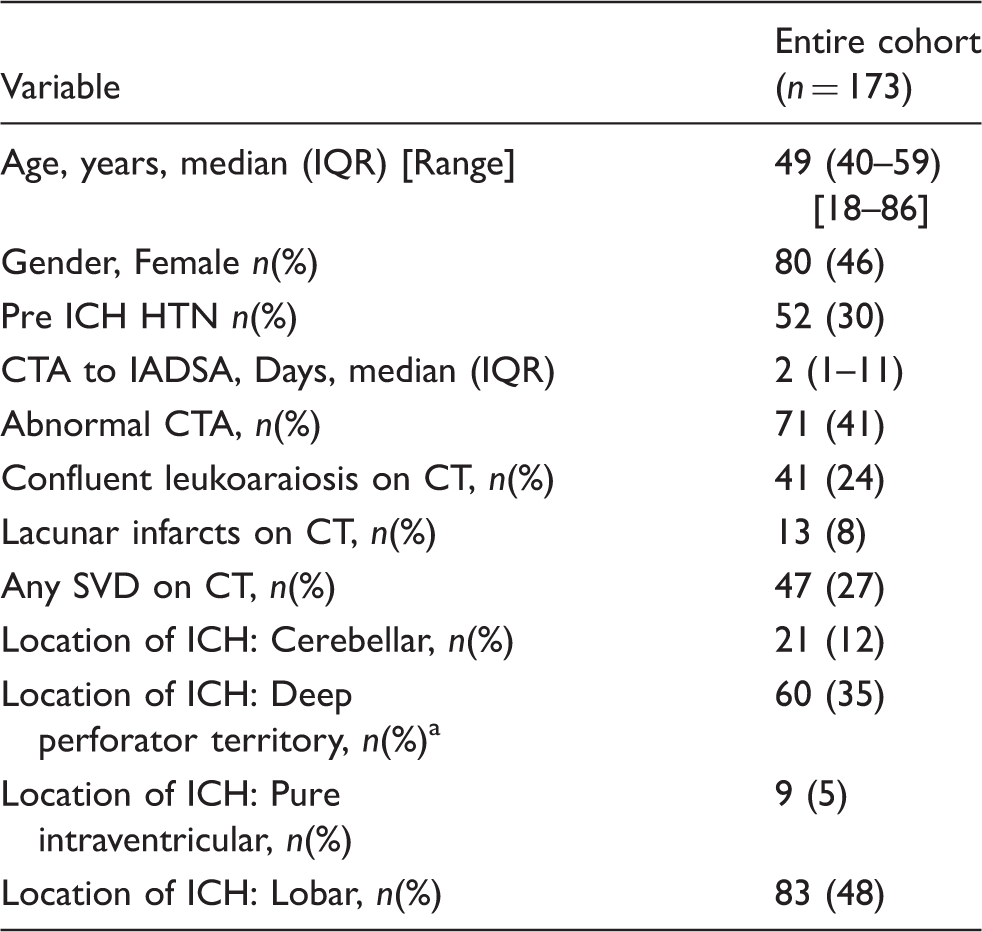
173 patients (46% female, median age 49) of whom 78 had a macrovascular cause on IADSA were included. Predictors of a macrovascular cause were: abnormal CTA (OR 67.4; p < 0.001); absence of SVD (OR 5.0; p = 0.019); and absence of pre-ICH hypertension (OR 3.4; p = 0.05). In our internally derived prediction model, the combination of CTA, SVD and pre-ICH hypertension predicted the likelihood of an underlying macrovascular cause (optimism-adjusted ROC area 0.919). Patients with negative CTA, SVD and pre-ICH hypertension have a low likelihood of an underlying macrovascular cause (1.8%).
Discussion and conclusion
A combination of CTA, SVD and pre-ICH hypertension predict the likelihood of finding a macrovascular cause in patients with acute spontaneous ICH, allowing informed decisions regarding the likely benefit and risk of IADSA.
Keywords
Introduction
Intracerebral haemorrhage (ICH) is devastating: mortality is 54% at 1 year, 1 while only 12–39% of survivors recover to independence. 2 Most cases (77–88%) of spontaneous (non-traumatic) intracerebral haemorrhage are termed ‘primary,’ 3 with a presumption that they are caused by small vessel disease (SVD). 4 However, it is critical to exclude ‘secondary’ structural causes of ICH, including macrovascular causes (e.g. arterio-venous malformations, aneurysms and dural arteriovenous fistulae), which can be treated.
The ‘reference standard’ for detection of a macrovascular cause is intra-arterial digital subtraction angiography (IADSA) or neurosurgery. IADSA is invasive, requires skilled operators and is associated with a small but appreciable mortality and morbidity, especially in acute ICH. 5 Selecting which ICH patients have a sufficiently high likelihood of a macrovascular cause to recommend IADSA is a common and important clinical question. Current practice varies widely: 6 typically, the presence of pre-ICH hypertension, deep location of ICH and age are used as indicators of SVD to select patients unlikely to require IADSA, 6 but the evidence supporting this is scant and conflicting.7–9 A scoring system incorporating pre-ICH hypertension and age (among other factors) to identify patients at risk of a macrovascular cause has been previously validated, but did not include imaging markers of SVD and only had moderate discrimination outside of the United States.10,11 SVD can be directly identified on brain imaging by leukoaraoisis and lacunar infarction, even on plain CT. We therefore hypothesised that, in acute ICH patients, visualisation of SVD will predict a low yield of a macrovascular cause; furthermore, we aimed to develop and internally validate an algorithm to help clinicians identify patients at high risk of having a macrovascular cause underlying acute spontaneous ICH.
Patient and methods
Patient selection
We retrospectively reviewed all consecutive patients who underwent IADSA (the reference standard for detecting a macrovascular cause of ICH) for the investigation of ICH at the National Hospital for Neurology and Neurosurgery, Queen Square, from January 2010 to April 2014. Inclusion criteria were: acute, symptomatic, non-traumatic (spontaneous) ICH, with availability of diagnostic quality CTA, non-contrast CT and an IADSA. Patients with a primary diagnosis of subarachnoid haemorrhage (SAH) or subdural haemorrhage were excluded. In our centre, all patients presenting to our stroke unit with acute ICH have acute non-contrast CT and CTA unless there is a contra-indication. The need for an IADSA is decided during a weekly multidisciplinary vascular neuroradiological meeting where the patient’s age, ICH location and history (including hypertension) are taken into account, as per standard clinical practice. We did not exclude patients on the basis of age. We recorded whether the patient had a suspicion of a macrovascular cause on the non-invasive imaging prior to the IADSA to explore partial verification bias.
Data collection
Variables collected were: age (dichotimised into aged 45 years or over vs. under 45, in keeping with current clinical practice and previous studies8,12); pre ICH hypertension (defined by previously documented HTN for which either lifestyle advice or antihypertensive medication had been provided); and location of the ICH (cerebellar, intraventricular (pure), lobar and deep perforator territory (brainstem and basal ganglia)).
Image analysis
A trained vascular neuroradiology fellow (AO) reviewed the acute CTA, blinded to the IADSA result, for the presence of any structural macrovascular cause. This CTA evaluation was then compared to the final clinical report (by an accredited consultant vascular neuroradiologist). Disagreement was reviewed by a professor of vascular neuroradiology (HRJ) and a consensus decision was reached. A ‘negative’ CTA was defined as showing no indication of a macrovascular cause. A ‘positive’ CTA was defined by the final neuroradiology assessment as being suspicious for a macrovascular cause (graded as either having a ‘possible’ or ‘definite’ underlying macrovascular cause). A clinical research fellow (DW) trained in SVD rating rated the non-contrast CT for SVD using the simplified Fazekas scale 13 and the presence of lacunae, blinded to the IADSA result. SVD on CT was dichotomised into moderate-severe (Fazekas grade ≥2 in either periventricular or deep white matter distribution) and/or the presence of any lacunar infarction vs. mild/none SVD and no lacunar infarctions. SVD was rated in the hemisphere contralateral to the symptomatic ICH to avoid misclassification of peri-haematomal oedema. Haematoma location was classified using a recently published rating instrument. 14 The IADSA was reported by a certified consultant vascular neuroradiologist. The routine IADSA protocol for investigation of intracerebral haemorrhage at our institution includes selective catheterisation of the internal carotid, external carotid and vertebral arteries, with angiographic runs of each of these vessels in at least two projections and followed through to the venous phase.
Statistical analysis
Univariable odds ratios and the sensitivity and specificity (presented as ROC area under the curve) were undertaken for the following variables: age, ICH location, HTN, CTA result and SVD identification on CT against the reference standard of IADSA detected macrovascular causes.
We fitted a logistic regression model to generate risk coefficients for macrovascular causes. We checked the fit of the model by comparing the models prediction to the observed outcomes. This model was then internally validated using bootstrap validation with 1000 samples. Discrimination was quantified using the ROC area and calibration was assessed using the Cox–Miller calibration slope.
All analyses were performed using STATA 12.0 (StataCorp LP, TX).
The study was conducted and reported according to reference standards described in the TRIPOD guidelines (https://www.equator-network.org/reporting-guidelines/tripod-statement/)
Ethical approval
The study was approved by the Clinical Governance Committee of the National Hospital and the UCL Institute of Neurology and National Hospital Joint Research Ethics Committee.
Results
We identified 204 patients with acute spontaneous ICH who had an IADSA. The median age was 49 (range 18–86, IQR 40–59)) and 54% were males. After quality assurance, we included 173 patients with diagnostic quality CTA in the study (Figure 1). Within this group there were 78 IADSA-defined macrovascular causes (68 AVMs, 7 dural fistulas, 2 aneurysms and 1 carotid-cavernous fistula). The median CTA to IADSA time was 2 days (IQR 1–11). Three vascular malformations were only detected on repeat IADSA; of these patients the median CT-repeat DSA was 264 days (IQR 78–314). ICH locations in the final cohort were: lobar 83, deep perforator territory 60 (basal ganglia, thalamus, caudate, brainstem), Cerebellar 21, pure intraventricular: 9 (Table 1).
Flow chart for study. Characteristics of the patients in the study. HTN: hypertension; ICH: intracerebral haemorrhage; CTA: computed tomography angiography. Brainstem, basal ganglia, thalamus and caudate nucleus.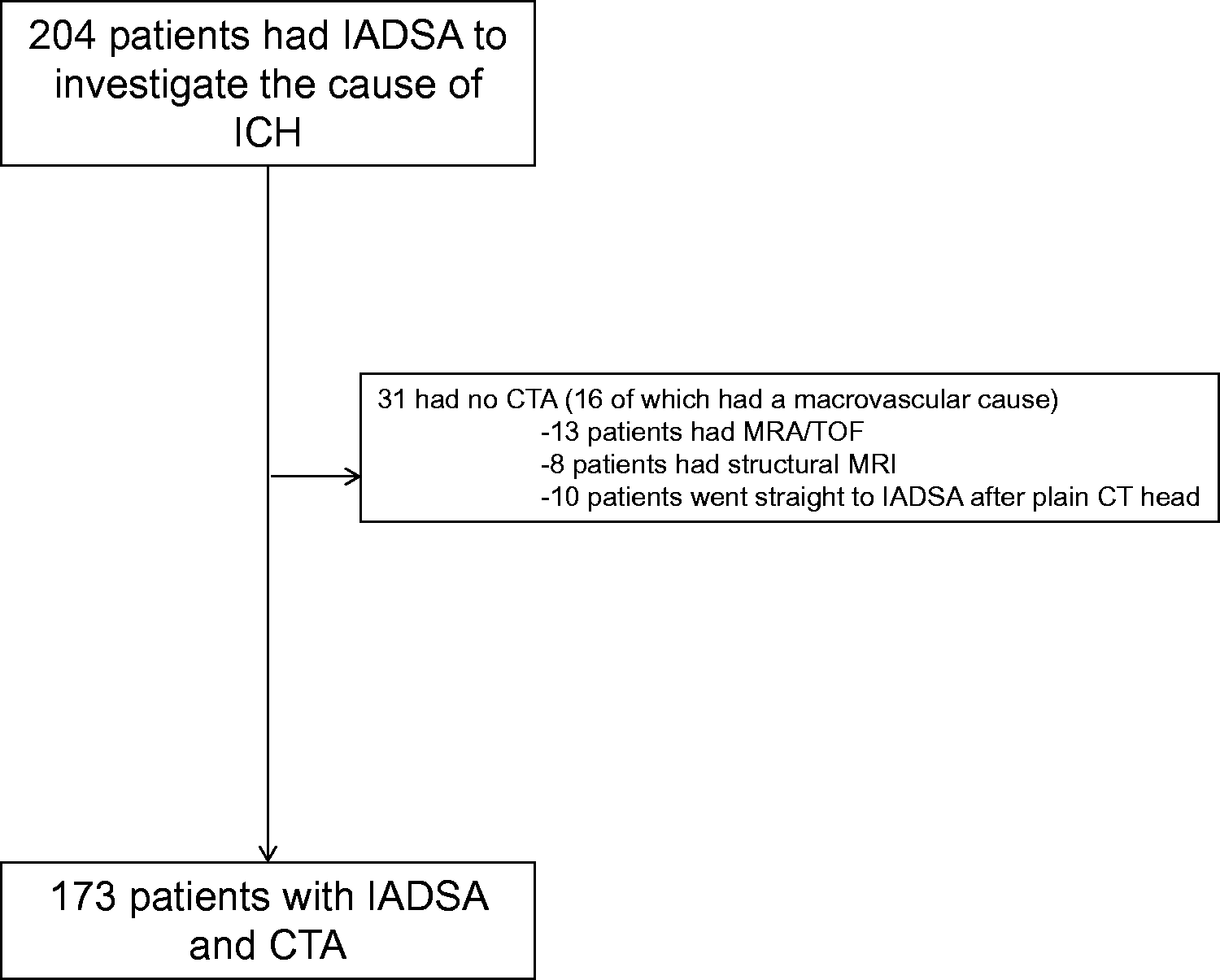
CTA identified 85 cases with a definite or possible macrovascular cause. 67/67 cases identified as ‘definite’ macro vascular abnormalities on CTA were identified as true macrovascular causes on IADSA. 11/18 cases identified as ‘possible’ macrovascular causes on CTA were identified as true macrovascular causes on IADSA. In 17 instances, there was discordance between the trained vascular neuroradiology fellow and the clinical report; 11 of these were judged to have a possible macrovascular cause by consensus adjudication with the professor of vascular neuroradiology (HRJ).
Univariable predictors of presence of a macrovascular cause on IADSA.
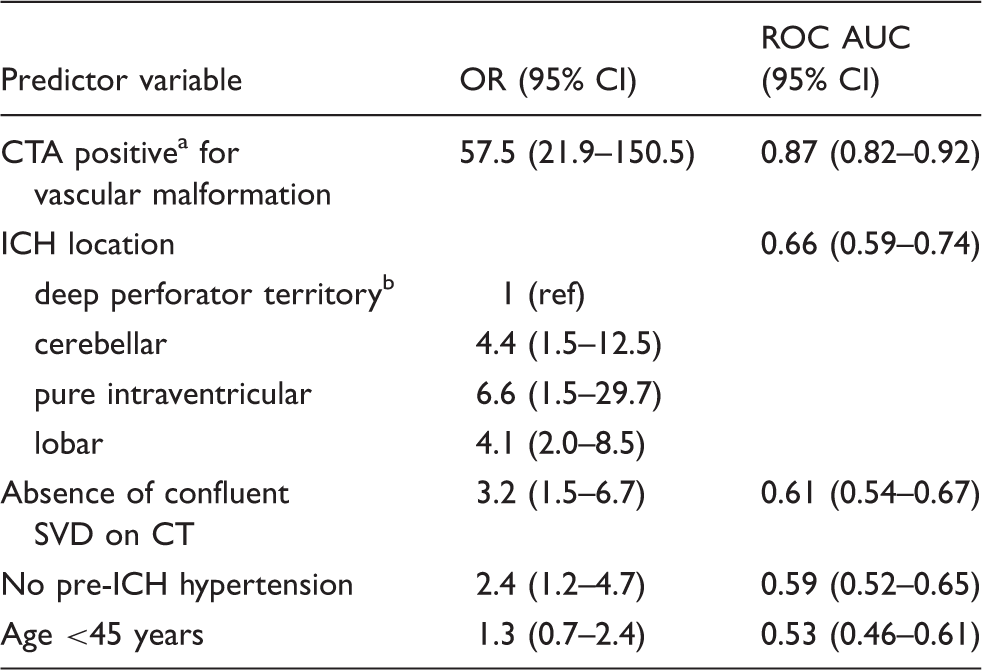
IADSA: intra-arterial digital subtraction angiography; ICH: intracerebral haemorrhage; CTA: computed tomography angiography; SVD: small vessel disease; OR: odds ratio; ROC: receiver operator curve; AUC: area under the curve.
Positive includes both ‘definite or possible’ suspicion for a macrovascular cause by neuroradiological expert consensus.
Brainstem, basal ganglia, thalamus and caudate nucleus.
Yield of intracranial macrovascular causes from the optimised model compared to the actual yield.
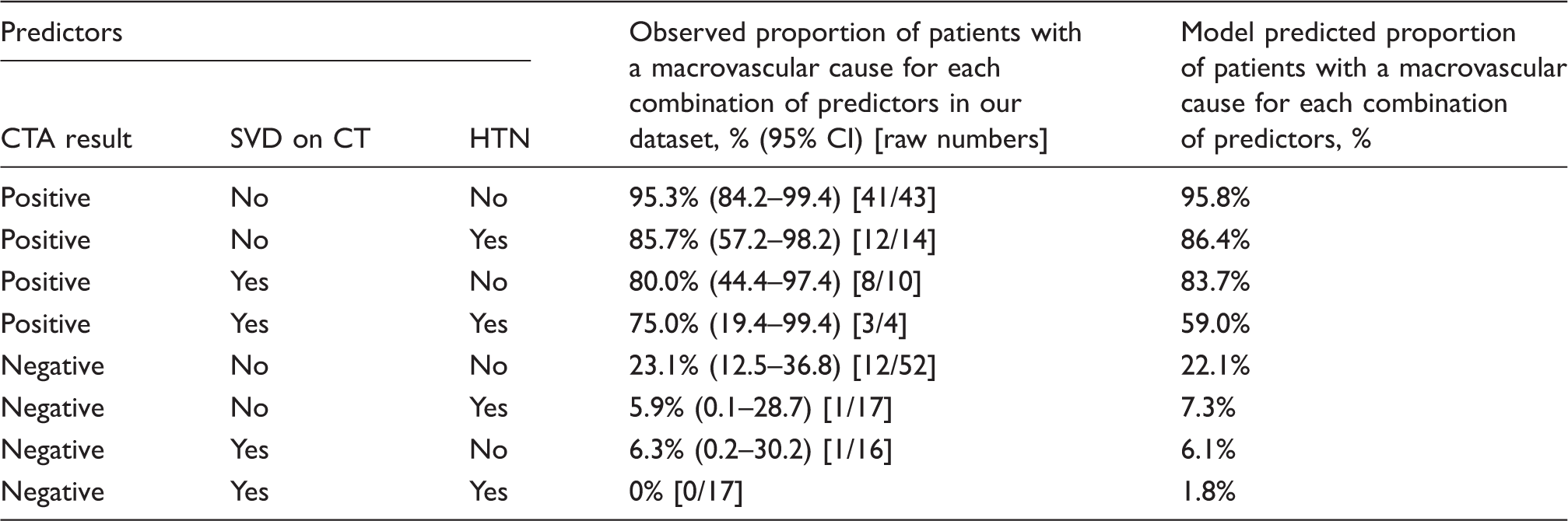
CTA: computed tomography angiography; HTN: hypertension; SVD: small vessel disease.
Positive’ includes CTA showing a ‘definite’ or ‘possible’ macrovascular cause; ‘negative’ denotes CTA showing no suspicion of a vascular malformation.
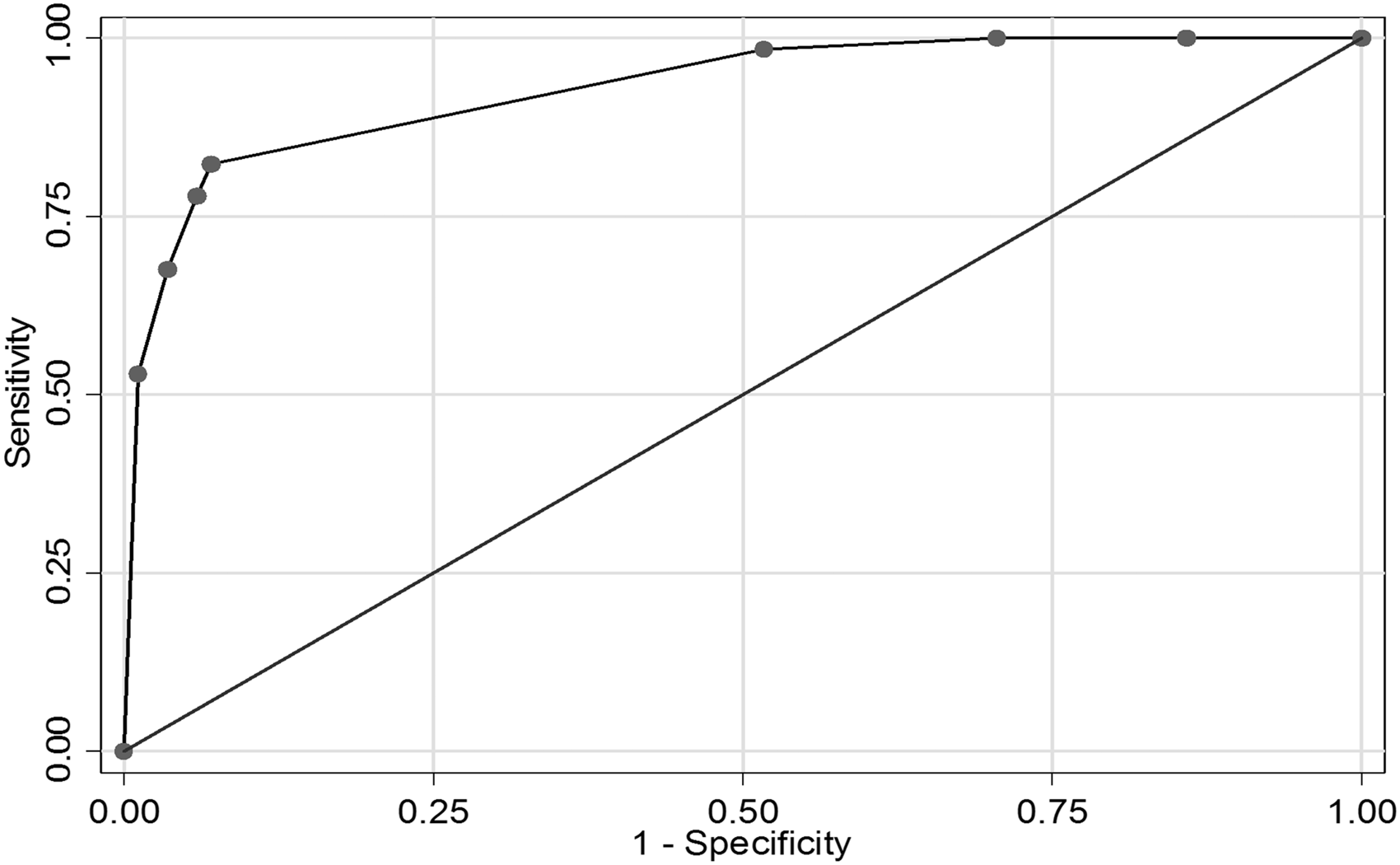
Optimised model ROC curve with abnormal CTA, no pre-stroke hypertension and no moderate to severe SVD on CT used as predictors of macrovascular cause.
The final model allowed us to estimate the risk of a macrovascular cause for any patient based on the combination of CTA result, pre-ICH hypertension and SVD on CT (Table 3). The predicted yield of a macrovascular cause for each combination of predictors is also presented in an algorithm (Figure 3).
Suggested diagnostic algorithm.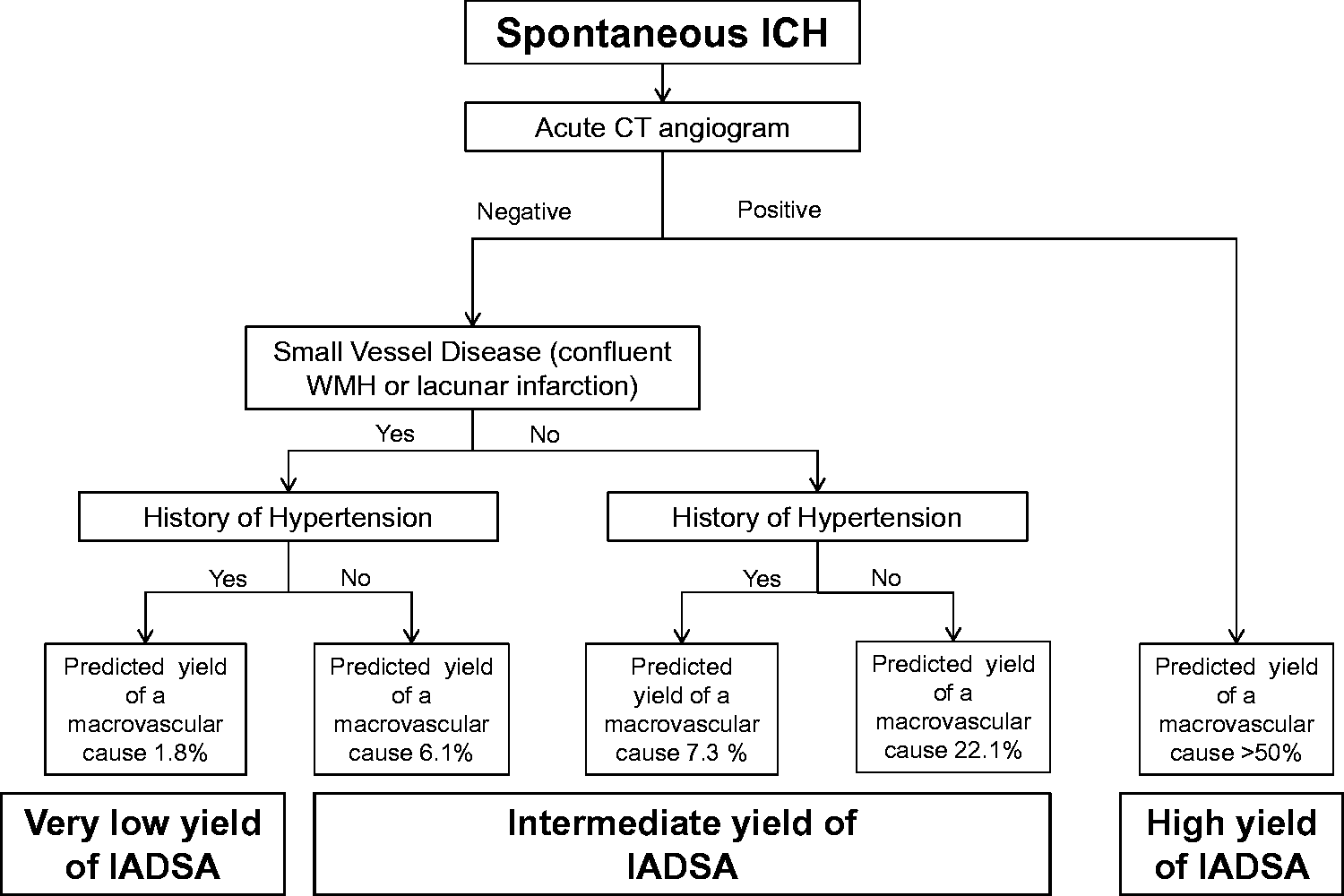
Discussion
In a population of acute ICH patients at risk of an underlying macrovascular cause (having had IADSA based on age, ICH location and vascular risk factors), the presence of small vessel disease (confluent leukoaraiosis and/or one or more lacunes) on non-contrast CT is a useful predictor of the likelihood of finding an underlying macrovascular cause. In our internally validated model, an abnormal CTA, or the combination of a normal CTA with no evidence of SVD (confluent leukoaraiosis on CT) indicate an intermediate to high yield of an underlying macrovascular cause, which might justify undertaking IADSA.
If externally validated on a prospective cohort, the use of our algorithm in clinical practice could lead to more macrovascular causes being detected (higher sensitivity) with an acceptable level of non-diagnostic IADSAs. For instance, if we used our algorithm on our own sample, choosing a predicted yield for a macrovascular cause of >5% as an indication for IADSA, our algorithm would yield 12 more macrovascular causes compared to using CTA alone, at the expense of 85 more negative IADSAs (i.e. an extra 7 patients undertaking an IADSA per additional macrovascular cause identified). In practice, clinicians must balance the expected yield of the IADSA against the procedural risk.
The sensitivity of acutely performed CTA for identifying a macrovascular cause in our study (82%) is lower than reported in a recent meta-analysis of 95% (CI 90–97%), 16 likely due to our use of repeat or delayed IADSA, which revealed a macrovascular cause not seen on the acute IADSA in three instances. Delayed IADSAs were not undertaken in studies involved in the above meta-analysis. Previous studies compared only acute CTA to acute IADSA. However, a recent multicentre prospective study also found a lower sensitivity of acute CTA, in line with our findings. 17 The difference is consistent with an additional yield of macrovascular causes from delayed IADSA, a well-recognised observation in clinical practice, which could be in part related to acute haemorrhage and perihaematomal oedema obscuring small macrovascular lesions. Our study was not able to investigate whether delayed CTA has similar diagnostic accuracy to delayed IADSA, which is a topic for further research.
Although a small case series has previously shown the added diagnostic value of MRI when IADSA is inconclusive, 18 we are only aware of one other study that specifically tests direct visualisation of small vessel disease to improve the prediction of the likelihood of an underlying vascular malformation. 17 In keeping with this study, 17 , our data suggest the presence of SVD on acute CT (when CTA is not suspicious for a macrovascular cause) is a useful predictor of a low yield of an underlying macrovascular cause in the investigation of younger patients with spontaneous ICH (mean ages 53 years in the previous study 17 and 50 years in the current study).
Importantly, in our study three patients with an underlying macrovascular cause were older than 45 years with known hypertension and a cerebellar ICH. This combination of variables has previously been associated with a 0% yield of macrovascular causes, 8 but our data indicate that further investigation looking for a macrovascular cause should be considered in this clinical situation.
Our study has important strengths. All patients identified with a suspected macrovascular cause for ICH at our centre undergo IADSA, so our initial search captured all patients diagnosed with a known macrovascular cause over the study period. Most patients had acute CTA on admission within a standardised care pathway, reducing the potential for selection bias. Our centre has a low threshold for requesting IADSA in suspected macrovascular causes and we were therefore able to include a large cohort of patients with CTA, CT and IADSA (n = 173), which compares favourably in size to previous studies.12,19–22 The time interval between CTA and the initial DSA was very short, and we included independent blinded CTA ratings by vascular neuroradiologists. Furthermore, we performed delayed and multiple IADSAs in selected patients, which increased the detection of macrovascular causes.
Nevertheless our study also has limitations. The retrospective nature of the study is likely to cause selection and partial verification bias. Although 63% of our cohort had a normal initial CTA (suggesting that there is little partial verification or ‘work up’ bias), our sample is younger than an unselected ICH cohort (median age 48, IQR 40–48), suggesting selection bias, likely due to the fact that IADSA was an inclusion criterion. This reflects current clinical practice with an inherent hesitancy for requesting IADSA in older individuals (e.g. >70 years) in whom that the yield of a macrovascular cause is perceived to be very low. 8 This selection bias toward younger individuals also explains why our proportion of vascular malformations is much higher than one would expect from an unselected population. Our results can thus only be generalised to younger patients with ICH. This is a difficult bias to overcome in any study, because undertaking IADSA in all unselected consecutive patients with ICH is unlikely to be considered ethical because of the procedural risk, which may be as high as 3% in ICH. 5 Nevertheless, this acknowledged selection bias is unlikely to weaken our main finding that SVD is predictive of a low yield of a macrovascular abnormality.
We did not capture ICH volume in our cohort, which would have been useful to better characterise our population. In particular the proportion of patients with larger ICH might be relevant, because such patients are less likely to undergo acute IADSA, and can be difficult to classify according to anatomical ICH location. 14 We did not include any patients with multiple acute haemorrhages, presumably because they were considered unlikely to have multiple macrovascular causes and thus did not undergo an IADSA at our centre. Despite these limitations, the demographic features of our study suggest that it is likely to be generalisable to the ICH population that in clinical practice is considered for investigation for an underlying macrovascular cause. A further limitation is that we did not routinely undertake MRI in all included patients, and were therefore unable to investigate additional markers of cerebral SVD underlying ICH, such as cerebral microbleeds, dilated perivascular spaces or cerebral atrophy.23,24 The use of standardised MRI prior to IADSA would have strengthened our study as it is better able to differentiate between the type of SVD (i.e. cerebral amyloid angiopathy or hypertensive arteriopathy), and is superior for the detection of other underlying causes for ICH (e.g cavernomas or mass lesions). However, the lack of MRI is unlikely to have compromised our ability to detect leukoaraiosis, the main SVD feature analysed in our study: a previous study 25 demonstrated that although MRI is more sensitive than CT in detecting subtle (Fazekas grade 1) white matter lesions, MRI and CT have a high concordance in detecting clinically more relevant Fazekas grade 2 and 3 lesions, which we used as predictor in our analysis. Our choice of CT (which was available in all patients acutely at the time of haemorrhage) for assessment of white matter lesions is therefore very unlikely to have led to a significant underestimation of moderate-severe (Fazekas grade ≥2) small vessel disease. Furthermore, our (unpublished) internal inter-rater reliability is moderate to good for the identification of Fazekas 2/3 in ICH populations (κ = 0.60), and we were careful to exclude perihaematomal oedema as a potential mimic of small vessel disease by assessing hemisphere contralateral to the ICH. We did not encounter any cases of transependymal oedema due to acute hydrocephalus, likely due to the selection bias of our patients where we only included those with IADSA, an intervention unlikely to be undertaken in such patients. By contrast, the poor sensitivity and specificity (especially with regards to differentiating enlarged perivascular spaces) of identifying lacunar infarction on CT remains a limitation.
Conclusion
In summary, we show, in a younger acute ICH population (median age 49 years) at risk of having an underlying macrovascular cause, the identification of SVD (moderately severe leukoaraiosis or lacunar infarction on CT), in combination with CTA and pre-ICH hypertension ICH, can predict which patients have a low yield of an intracranial macrovascular cause. If externally validated, our algorithm has potential to more effectively target invasive IADSA towards patients with the highest potential diagnostic yield, avoiding unnecessary invasive tests and improving the diagnostic pathway for patients with spontaneous ICH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DW and DJW received finding from the stroke association and the British heart foundation. GA received funding from the National Institute for Health Research University College London Hospitals Biomedical Research Centre.
Ethical approval
Institutional Review Board approval was obtained.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Guarantor
DJW.
Contributorship
DJW and HRJ contributed equally to this work. DW, DJW, IJ and HRJ were involved with study concept, DW, DJW and HRJ were involved with data retrieval and data interpretation, DW and GA were involved with statistical analysis. AO was involved with data interpretation. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
