Abstract
Introduction
Limited data exist on the outcome of pregnancies and deliveries in women with ischaemic stroke. We investigated the incidence of pregnancy- and delivery-related complications in women with ischaemic stroke before and after pregnancy compared with stroke-free matched controls.
Patients and methods
Of our 1008 consecutive patients aged 15–49 years with first-ever ischaemic stroke, 1994–2007, we included women with pregnancy data before or after stroke recorded in the Medical Birth Register (MBR) (
Results
A total of 124 stroke mothers had 207 singleton pregnancies before and 45 mothers 68 pregnancies after stroke. The incidence rate ratio (IRR) for the composite outcome of pregnancy and delivery complications adjusted for socioeconomic status and maternal smoking was 1.43 (95% confidence interval [CI] 1.00–2.03,
Discussion and conclusions
Compared with stroke-free mothers, we found a higher incidence of pregnancy- and delivery-related complications in mothers with ischaemic stroke. Larger studies are needed to verify our results.
Introduction
Although the total incidence of ischaemic stroke (IS) is declining, the incidence of IS at younger ages has increased during the past decades. 1 Stroke at younger age can be particularly devastating for young women in their best child-bearing ages. However, the reproductive health of IS patients remains understudied. A history of pre-eclampsia, and with weaker evidence also gestational diabetes mellitus (GDM), have been associated with an increased risk of cardiovascular disease (CVD) including stroke in the subsequent years after pregnancy, but type 2 diabetes seems to account for much of the increased risk associated with GDM.2,3 Also, other pregnancy-related events such as a history of low birth weight and preterm birth of an offspring are associated with maternal risk for CVDs later in life.4,5 Interestingly, placental abruption and IS share common risk factors, such as smoking and alcohol abuse. 6 Women with multiple pregnancy loss or at least one stillbirth have an increased risk of IS. 7
Less is known on patients having suffered a stroke and their subsequent reproductive health. Previous studies on this topic are mostly either case series, rely on questionnaire and interview data,8–10 or are limited to strokes occurring during pregnancy and puerperium.11,12 According to two follow-up studies, the outcome of pregnancies after stroke appeared to be similar to that expected in the general population.8,13
Therefore, in this study, we aimed at investigating the incidence of pregnancy- and delivery-related complications (1) before IS, and (2) after IS in patients from the Helsinki Young Stroke Registry (HYSR) compared with matched stroke-free controls.
Patients and methods
Study population
HYSR includes 1008 consecutive patients, 15–49 years, with a first-ever IS from January 1994 to May 2007 treated at the Department of Neurology, Helsinki University Hospital (HUH). 14 The World Health Organization definition of stroke was used with the exception that imaging positive cases were regarded as an IS even if the symptoms lasted <24 h.14,15 Baseline tests done and the modified TOAST classification used to determine stroke aetiology have been previously described.14,16
We included those women from HYSR ( Flow chart of patients from HYSR and stroke-free matched control mothers included in the analyses.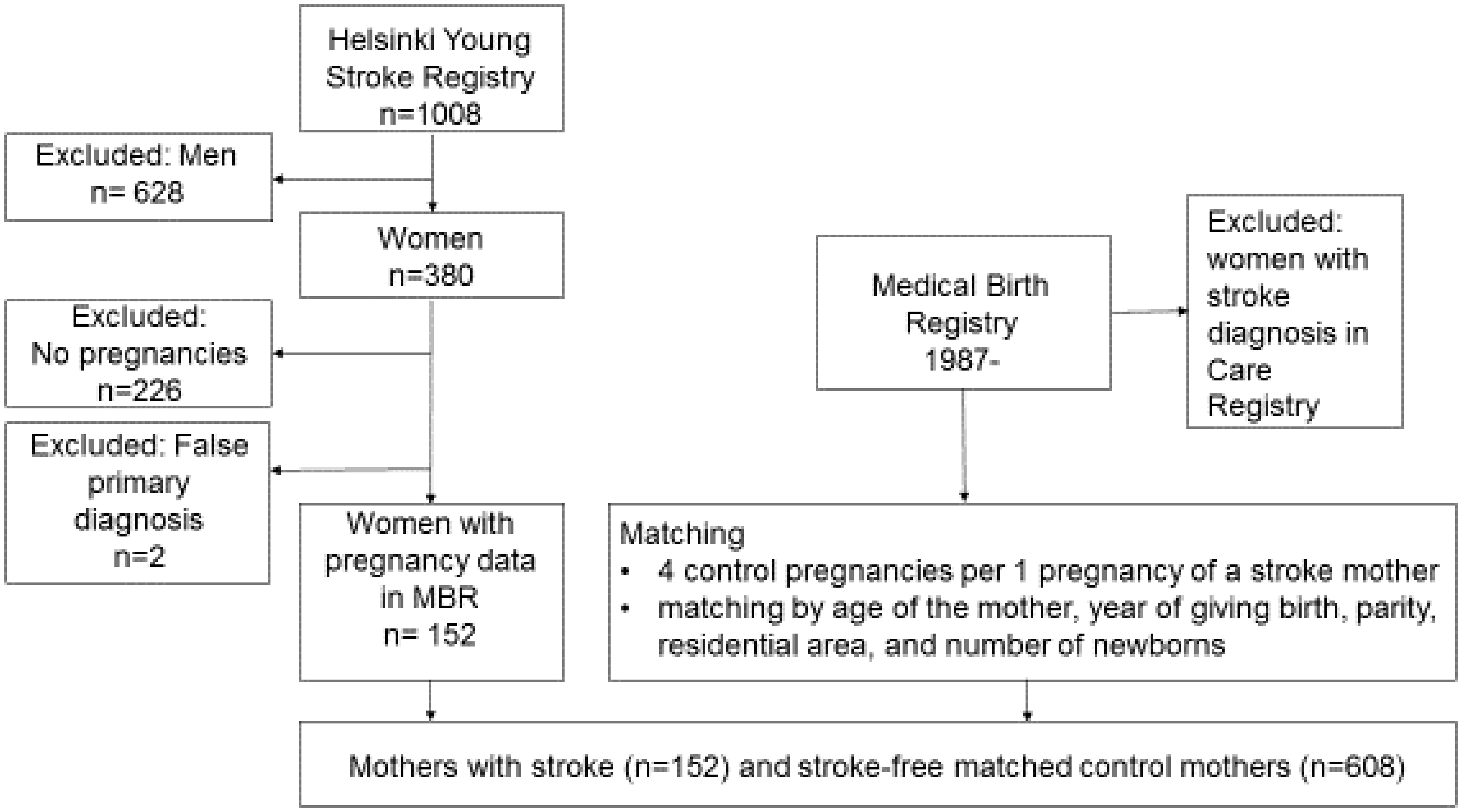
A total of 152 women with IS and their four matched controls were included, resulting in 608 control mothers matched by mother’s age, parity, year of delivery, residential area and number of newborns in the last pregnancy recorded. THL verified that stroke-free controls did not have hospital admissions between 1969 and 2014 with a discharge diagnosis code 430-438 by ICD-8 and ICD-9, or with codes I60-I69, G45 by ICD-10 in the Hospital Discharge Register (HDR). A total of 434 matched control’s last pregnancy met all the matching criteria. In 112 controls, the residential area was extended from the same municipality to the same subregion, in 29 controls the residential area was the same county, in 19 controls the residential area was the same subregion and mother’s age was ±two years different, and in 11 controls the residential area was unknown and mother’s age was ±three years different from HYSR mothers. We obtained data for all mothers on all-cause hospital admissions 1969–2014 from HDR and the causes and dates of death from Statistics Finland until the end of 2014.
Maternal characteristics
We classified socioeconomic status according to mother’s occupation as upper white and lower white collar worker, blue collar worker, other and unknown or missing.18,19 We divided marital status as follows: (1) married or cohabiting; (2) single, divorced or widow; and (3) unknown. We obtained data on smoking, use of assisted reproductive technologies (ART), previous induced abortions, spontaneous abortions, ectopic pregnancies, caesarean sections (CS) and stillbirths as recorded in MBR. Pre-pregnancy body mass index was available from 2004 onwards and was calculated by dividing mother’s weight in kilograms by the square of mother’s height in metres.
Outcome measures
We looked for all end-points with corresponding ICD codes given in Table e-1 from the mothers’ and the child’s diagnoses from MBR, and from all-cause hospital admissions. Pre-eclampsia, eclampsia and venous events were only screened by the ICD codes. In MBR, data on a pathological two-hour oral glucose tolerance test or whether insulin was started during pregnancy were available from 2004 onwards. We defined GDM as a pathological glucose tolerance test, insulin started during pregnancy or a diagnosis of GDM registered at MBR and/or HDR. We obtained data on hospital care during pregnancy excluding those that were due to delivery only from the MBR. Gestational age (GA) in MBR is calculated from the last menstrual period and confirmed with the first trimester ultrasound examination. We defined preterm birth as GA < 37 weeks, low birth weight as birth weight <2500 g, stillbirth as death of the foetus before or during delivery from 22 gestational weeks onwards, and early neonatal death as a neonate dying within a week after birth, or diagnoses of these registered at MBR and/or HDR. Perinatal deaths included stillbirths and early neonatal deaths. We defined small-for-gestational age (SGA) as birthweight ≤−2 standard deviations (SD) of the national sex-specific standard. 20 Apgar scores at 1 min after birth were evaluated by giving each neonate zero to two points for the following characteristics: (1) pulse rate; (2) breathing; (3) reflexes; (4) muscle tone; and (5) skin color.
The endpoints were: (1) pre-eclampsia and/or eclampsia; (2) placental abruption; (3) GDM; (4) hospital admission during pregnancy recorded in MBR; (5) preterm birth and/or low birth weight; (6) SGA; (7) Apgar at 1 min ≤ 6; (8) the child not at home one week after the delivery; (9) perinatal death; (10) and a composite outcome including outcomes 1 to 9 as a count outcome.
Statistical analyses
We analysed data using SPSS 22.0 for Windows (SPSS Inc., IBM, Armonk, NY, USA) and STATA 14.1 for Mac (StataCorp, Texas USA). We compared the outcomes of pregnancies and deliveries separately before stroke (research question 1) and after stroke (research question 2) using Poisson regression mixed models analysis (random intercept models) on three levels to account for the clustering of the data: (1) delivery; (2) mother; (3) matched cases and controls. We included only singleton pregnancies into the main analyses and analysed the outcomes separately after adjusting for possible confounders; socioeconomic status and smoking of the mother. We excluded cases with missing data. In sensitivity analyses, we analysed the data (a) with both singleton and multiple pregnancies, (b) excluding hospital admissions of the mother form the composite outcome, and (c) excluding those mothers with strokes during pregnancy or puerperium (
Results
A total of 1432 pregnancies were included, of which 1380 were singleton pregnancies. Of these pregnancies, 124 mothers from HYSR had 207 pregnancies before the impending IS, 45 mothers from HYSR had 68 pregnancies after suffering IS, and there were 17 mothers with both pre-stroke and post-stroke pregnancies. The stroke characteristics of the 152 stroke mothers are shown in Table e-2. The median modified Rankin scale score at three months from IS was 2.0 (interquartile range 0.0–2.0). There were four mothers from HYSR with IS during pregnancy and four mothers with IS during the puerperium, until six weeks after delivery. No mothers died during pregnancy or puerperium. The median time from the last birth of a child to stroke was six years (interquartile range 0.02–11).
Maternal characteristics at first pregnancy in MBR for mothers from HYSR and matching pregnancies for control mothers. a
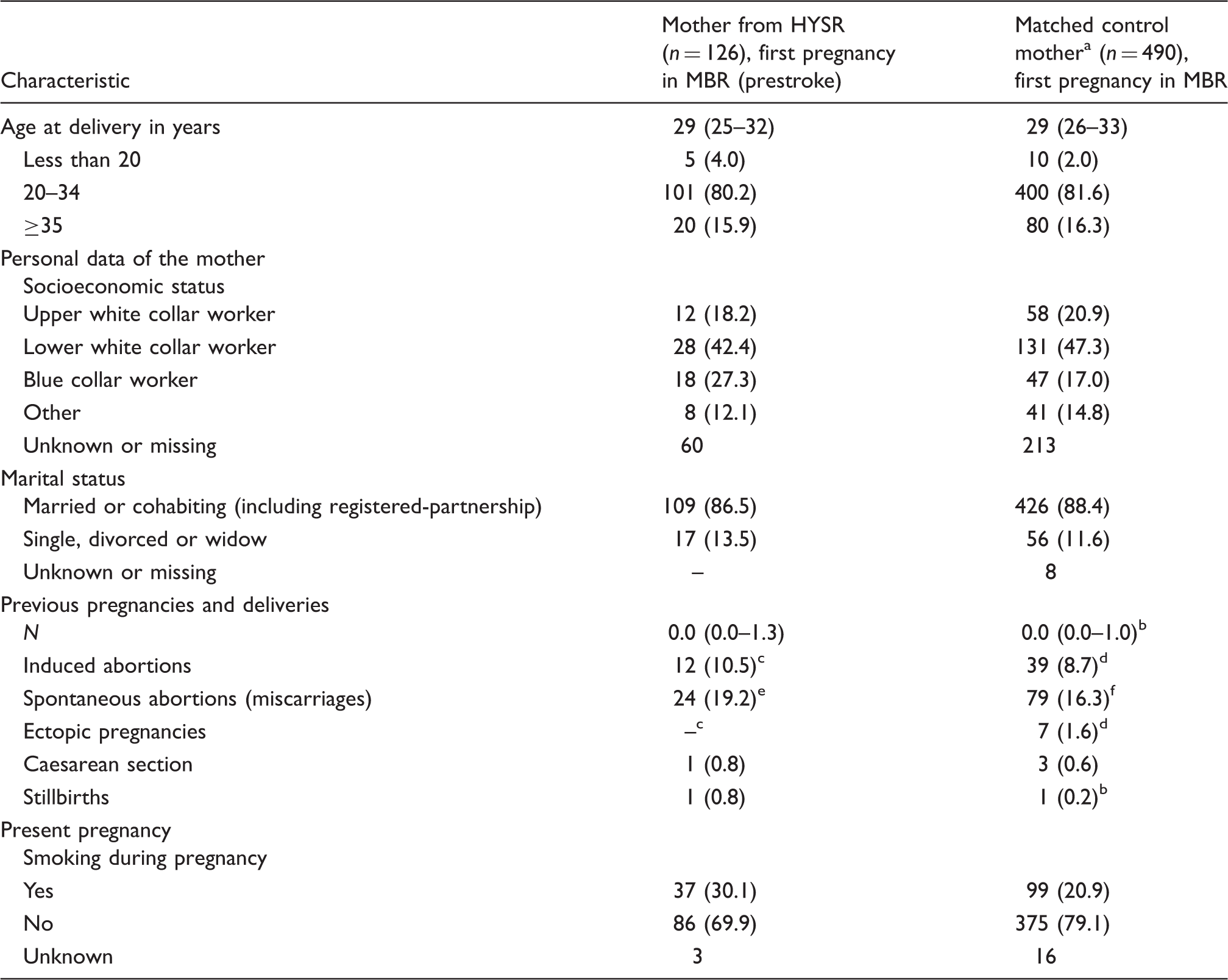
MBR: Medical Birth Registry; HYSR: Helsinki Young Stroke Registry; BMI: body mass index.
Data are
Matched by age of the mother, year of birth, residential area, parity and number of newborns.
Missing data for 4 mothers.
Missing data for 12 mothers.
Missing data for 44 mothers.
Missing data for 1 mother.
Missing data for 6 mothers.
Maternal characteristics at first pregnancy after IS for mothers from HYSR and matching pregnancies for control mothers. a
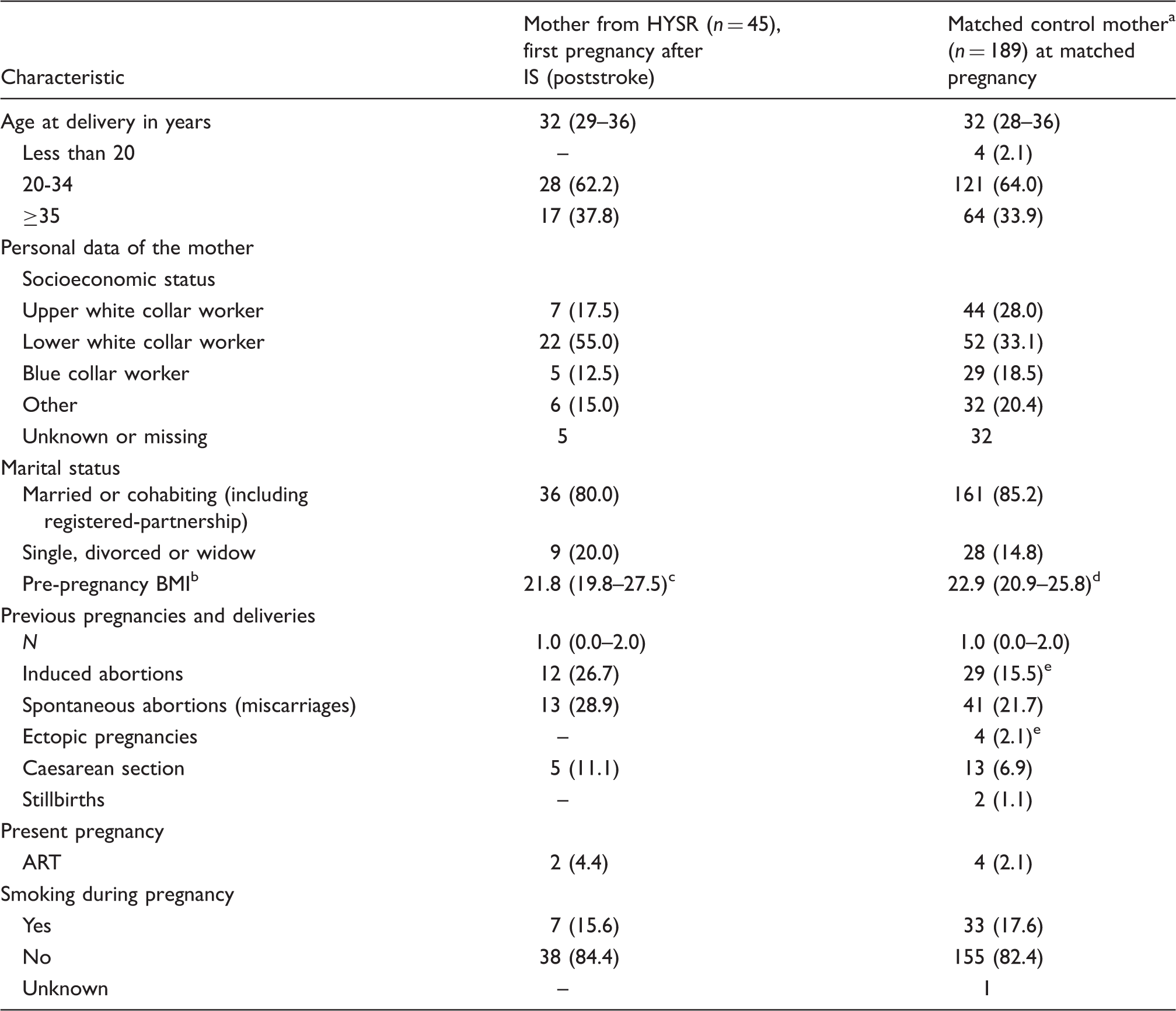
IS: ischaemic stroke; HYSR: Helsinki Young Stroke Registry; BMI: body mass index; ART: assisted reproductive technologies.
Data are
Matched by age of the mother, year of birth, residential area, parity and number of newborns.
Available from 2004 onwards.
Missing data for 25 mothers.
Missing data for 116 mothers.
Missing data for 2 mothers.
Numbers and relative risks of complications during pregnancy, delivery and puerperium in singleton births according to Poisson regression mixed models analysis before IS in patients from HYSR compared with matched controls.
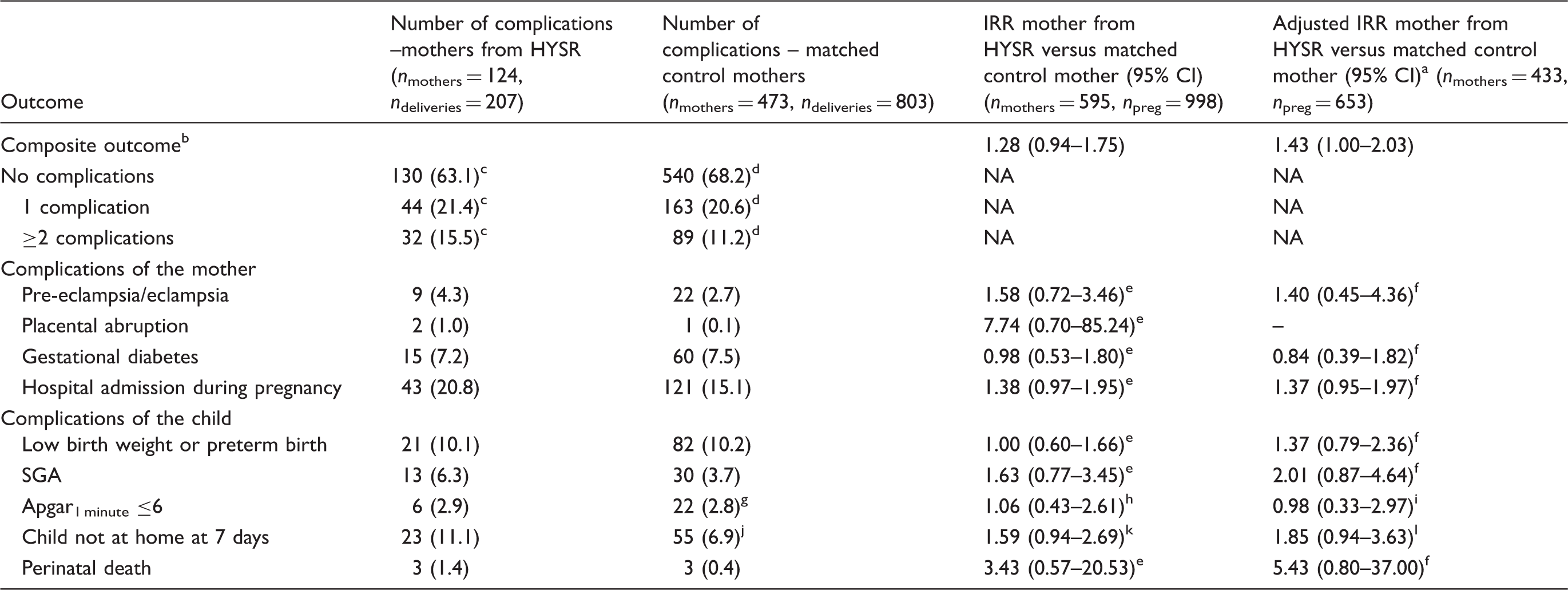
IS: ischaemic stroke; HYSR: Helsinki Young Stroke Registry; IRR: incidence rate ratio; CI: confidence interval; SGA: small-for-gestational age.
Adjusted for smoking and socioeconomic status.
Including pre-eclampsia and/or eclampsia, small for gestational age, preterm birth and/or low birth weight, placental abruption, gestational diabetes, hospital care during pregnancy, Apgar1 minute ≤6, the child not at home one week after delivery and perinatal death.
Missing data in one pregnancy.
Missing data in 11 pregnancies.
Missing data in 4 pregnancies.
Missing data in 11 pregnancies.
Numbers and relative risks of complications during pregnancy, delivery and puerperium in singleton births according to Poisson regression mixed models analysis after IS in patients from HYSR compared with matched controls.
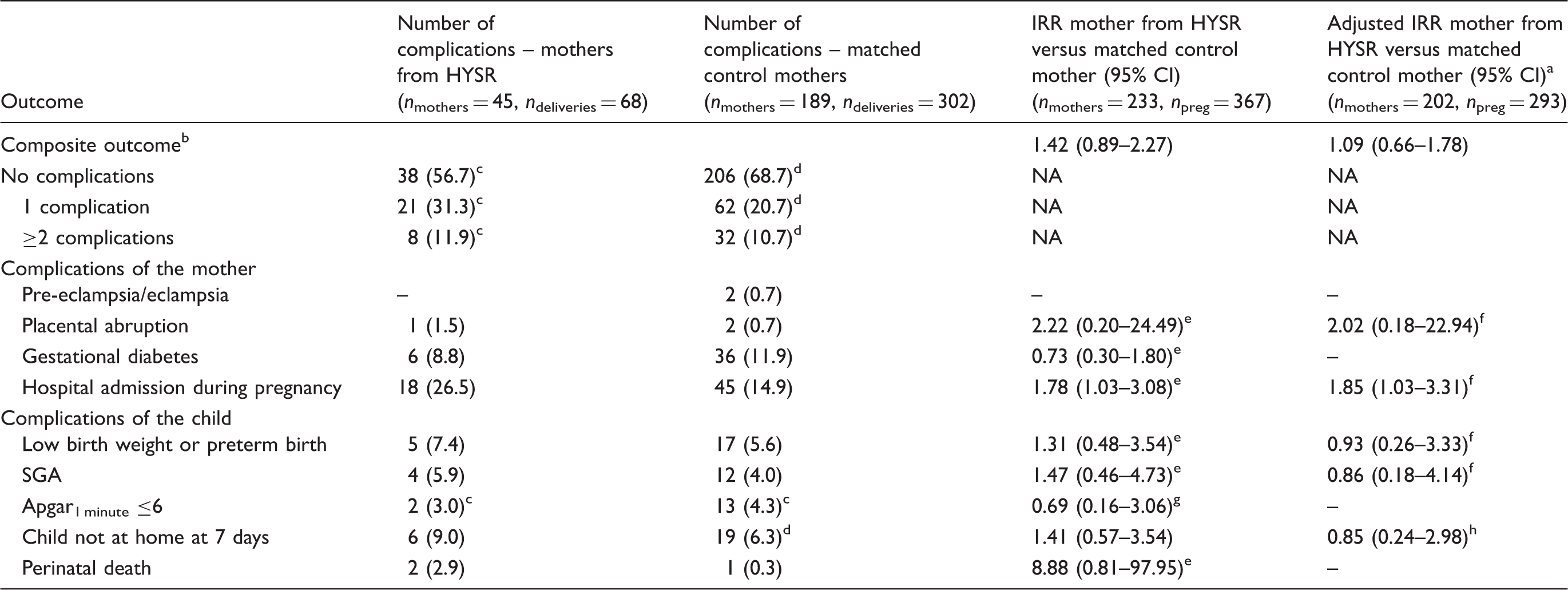
IS: ischaemic stroke; HYSR: Helsinki Young Stroke Registry; IRR: incidence rate ratio; CI: confidence interval; SGA: small-for-gestational age.
Adjusted for smoking and socioeconomic status.
Including pre-eclampsia and/or eclampsia, small for gestational age, preterm birth and/or low birth weight, placental abruption, gestational diabetes, hospital care during pregnancy, Apgar1 minute ≤6, the child not at home one week after delivery and perinatal death.
Missing data in one pregnancy.
Missing data in two pregnancies.
In the sensitivity analyses, when including both multiple and singleton pregnancies, the IRR before stroke was 1.18 (95% CI 0.88–1.57) (
Discussion
In our retrospective matched cohort study, we found a slightly higher incidence of pregnancy- and delivery-related complications for women with IS, especially before stroke. Women with IS had more hospital admissions during pregnancies after stroke than their matched controls, suggesting higher morbidity in this group of mothers. The difference in the incidence of perinatal deaths seemed to be higher in mothers from HYSR compared with control mothers of the same age, parity, year of birth, residential area and multiplicity, although not reaching statistical significance.
Perinatal death rate in Finland was 4.3 per 1000 births in 2008, as was found for the control mothers in our study. 21 Similarly to the increased perinatal death rate found in our study, a Dutch case control study indicated an increased risk of IS after stillbirth, although not being statistically significant, with an adjusted odds ratio of 2.06 (95% CI 0.81–5.23). 7 Risk factors for stillbirths in high-income countries include overweight and obesity, maternal age >35 years, maternal pre-existing hypertension and diabetes, pre-eclampsia, smoking and post-term pregnancy, whereas infections cause less stillbirths than in low- and middle-income countries. 22 Many of these risk factors are also risk factors for stroke, 14 and this could be one of the reasons why our patients from HYSR had a higher risk for perinatal death. Also, a higher socioeconomic status is inversely associated with stillbirths. 23 However, when we adjusted for socioeconomic status and smoking of the mother, the IRR of perinatal deaths before stroke was even more pronounced, the IRR being nearly five, although again lacking statistical significance. Still, because of the low number of cases and wide confidence intervals, these results should be interpreted with caution.
Most pregnancies, meaning 207 of 275 singleton pregnancies for mothers from HYSR, occurred before IS, which is in part explained by the high median age of mothers at stroke. Nonetheless, the impact of stroke on family planning should not be underestimated. A French study reported that although 34% of stroke mothers aspired to have more children, the reasons not getting pregnant after IS or cerebral vein thrombosis were concern of recurrent stroke, medical advice against pregnancy and residual handicap. 8 However, the median number of deliveries before stroke was two in our study. As the total fertility rate in Finland was 1.78 during the study period, 24 the small number of pregnancies after IS was rather as expected. Post-stroke pregnancy rate for stroke mothers, 45.7 pregnancies/1000 follow-up years, was slightly lower than found among women aged 15–49 years in the same hospital district, 47.7/1000 women years. It is worth noting, that our study was not designed to analyse this question and it included women with IS and at least one pregnancy, leading possibly to a selection bias which should be considered when interpreting these results. There were more pregnancy complications even given the fact that perhaps only the less affected stroke patients were included in our study. Also, our women from HYSR had less risk factors for stroke than the total HYSR cohort had, 14 suggesting that these women were overall healthier than young stroke patients in general are.
Although with small numbers and without a clear statistically significant difference in the incidence of placental abruption, the pre-stroke IRR of almost eight in women from HYSR compared with control mothers, was interesting. This again, could be linked to a common risk factor, such as smoking, 6 or it could be related to other common pathologies, such as uteroplacental ischaemia. 25
The strengths of our study include quantitative data analysis on the outcomes of pregnancies, adjustment for socioeconomic status and smoking, rigorous and well-established matching criteria, many outcomes explored, and searching for the majority of endpoints from the MBR and HDR.
Limitations of our study include that it is registry-based, possibly leading to detection bias, with varying levels of data accuracy during the years in MBR, although in 99% of cases even reproductive history were found to be correct. 26 The positive predictive value for common diagnoses in HDR has been 75–99%. 27 Second, control mothers failed to fulfill all matching criteria, potentially leading to residual bias, but over 70% of the primary matching criteria were fulfilled well. Third, the sample size was too small to detect differences in most outcomes, addressing the need for larger sample sizes in future studies. Fourth, including hospital admissions into the composite outcome could have included a single outcome twice or resulted in admission bias, but was considered important as this could represent a more severe outcome. The sensitivity analyses still showed similar results when excluding these hospital admissions. Also, data on miscarriages and antithrombotic therapy were lacking, the former possibly influencing our results if acting as a competing outcome. Older age after stroke could be a confounder, but age was one of the matching criteria and still we found a slightly higher incidence of complications for women with IS before stroke, when women were younger. Finally, strokes occurring during pregnancy or puerperium could have influenced the results. However, again, our sensitivity analyses excluding these cases showed similar results than when including those.
Conclusion
In summary, there was a slightly higher incidence of pregnancy- and delivery-related complications for mothers with IS, compared with stroke-free mothers, only reaching statistical significance in the pre-stoke setting. These results may suggest that mothers with IS have a higher morbidity during pregnancy and experience more perinatal deaths than their stroke-free controls. However, more studies with an emphasis on the mothers and their functional outcome, with larger sample sizes, and studies among other ethnic groups are warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Doctoral Programme in Clinical Research, University of Helsinki, Orion Research Foundation, Maire Taponen Foundation, CSB Virchow Scholarship, and Biomedicum Helsinki Foundation (KA), and Helsinki and Uusimaa Hospital District and Academy of Finland (JP).
Ethical approval
Helsinki University Hospital's (HUH's) Ethics Committee approved the protocol of this study, and the National Institute for Health and Welfare (THL) and Statistics Finland gave permission to use their register data in this study.
Informed consent
Not applicable.
Guarantor
JP.
Contributorship
KA, JP and UG analyzed the data. KA and JP drafted the first version of the manuscript. MG, BS, MK, TT, MT and JP gave administrative, technical, and material support. All authors reviewed, edited, and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
