Abstract
Introduction
The role of intracerebral haemorrhage location in haematoma expansion remains unclear. Our objective was to assess the effect of lobar versus non-lobar haemorrhage on haematoma expansion and clinical outcome.
Patients and methods
We analysed data from the prospective PREDICT study where patients with intracerebral haemorrhage presenting to hospital under 6 h of symptom onset received baseline computed tomography (CT), CT angiogram, 24 h follow-up CT, and 90-day mRS. Intracerebral haemorrhage location was categorised as lobar versus non-lobar, and primary outcomes were significant haematoma expansion (>6 ml) and poor clinical outcome (mRS > 3). Multivariable regression was used to adjust for relevant covariates. The primary analysis population was divided by spot sign status and the effect of haemorrhage location was compared to haematoma expansion in exploratory post hoc analysis.
Results
Among 302 patients meeting the inclusion criteria, lobar haemorrhage was associated with increased haematoma expansion >6 ml (p = 0.003), poor clinical outcome (p = 0.011) and mortality (p = 0.017). When adjusted for covariates, lobar haemorrhage independently predicted significant haematoma expansion (aOR 2.2 (95% CI: 1.1–4.3), p = 0.021) and poor clinical outcome (aOR 2.6 (95% CI: 1.2–5.6), p = 0.019). Post hoc analysis showed that patients who were spot sign negative had a higher degree of haematoma expansion with baseline lobar haemorrhage (lobar 26% versus deep 11%; p = 0.01). No significant associations were observed in spot-positive patients (lobar 52% versus deep 47%; p = 0.69).
Discussion and Conclusion
Haematoma expansion is more likely to occur with lobar intracerebral haemorrhage and haemorrhage location is associated with poor clinical outcome. As expansion is a promising therapeutic target, hemorrhage location may be helpful for prognostication and as a selection tool in future ICH clinical trials.
Introduction
With a one-month mortality rate as high as 40%, intracerebral haemorrhage (ICH) is a devastating form of stroke that is difficult to predict, prevent, and treat. 1 Haematoma expansion is common in ICH and significantly worsens mortality and morbidity.2,3 With emerging therapies targeting haematoma expansion, there is significant interest in improving patient selection for clinical trials. Baseline ICH volume, the CT-angiography spot sign, and coagulation status are well-established risk factors for haematoma expansion.1,3–5
Haematoma location is a characteristic whose predictive quality for haematoma expansion remains unclear. While previous studies have shown increased mortality in infratentorial ICH,6,7 the effect of location on haematoma expansion has not been extensively explored. Recent work from Kim et al. 8 did not show a discernable relationship between ICH location and haematoma expansion, but this study assessed spot sign positive patients only and was limited by a small sample size. Brouwers et al. 9 have shown an increased risk of expansion in lobar ICH, possibly related to the ɛ2/ɛ4 variants of the APOE gene and its strong association with cerebral amyloid angiopathy (CAA). However, other studies have shown less expansion in probable/definite CAA populations, 5 and a recent study showed an association between the ɛ4 variant of the APOE gene with non-lobar ICH, 10 a bleed type typically associated with arteriosclerosis and small vessel disease. Furthermore, there are many similarities between the characteristics of presumed CAA and hypertensive ICH, including shared risk factors that are predictive for spot sign. 11
While it is clear that the relationship between haematoma expansion and ICH location is complex, it is possible that the physical dynamics of haemorrhage and anatomical constraints of the brain play a role in expansion and outcome. Our objective was to assess the relationship between ICH location, haematoma expansion, and clinical outcome in a multicentre sample of patients presenting with acute ICH (<6 h). We hypothesised that lobar-located ICH would facilitate haematoma expansion as it provides a larger potential volume for expansion as compared to deep locations. However, due to the close proximity of critical structures and increased risk of ventricular rupture, we also hypothesised that deep ICH would have a paradoxically increased risk of mortality and morbidity.
Patients and methods
The design and results of the PREDICT study are published. 4 Briefly, PREDICT was a prospective multicentre observational study of patients presenting with spontaneous ICH under 6 h, who had baseline CT and CT-angiogram and follow-up imaging at 24 h post event. Participants provided informed written consent and were enrolled in centres from Canada, USA, Germany, Poland, India, and Spain. ICH volume was measured via ABC/2 methods. Exclusion criteria were ICH >100 ml, known renal impairment precluding CTA, baseline mRS >3, or terminal illness. The project as a whole received ethics board approval, with additional approval at each recruiting site as required by local ethics boards. The results of the original PREDICT study were based on data collected from 2006 to 2010; PREDICT continued to enrol patients until it officially closed on 27 February 2012. For the current study, we excluded patients that did not have baseline CT or CTA, follow-up CT, were treated with recombinant Factor VIIa, or had craniotomy for haematoma evacuation or ventricular drainage prior to repeat CT. Patients who could not be classified into lobar versus non-lobar ICH were also excluded.
Primary outcomes were significant haematoma expansion and poor clinical outcome. Significant haematoma expansion was defined as absolute ICH growth of 6 ml on follow-up imaging; this threshold predicts poor clinical outcome, falls within the minimal detectable difference (MDD) of the measurement software, and is used in prior haematoma expansion studies.3,12,13 Poor clinical outcome was defined as mRS > 3 at 90 days. 12 Secondary outcomes included spot sign prevalence, early neurological worsening defined as a ≥4-point increase in NIHSS over 24 h, and 90-day mortality. The primary independent variable of interest was haemorrhage location, which was categorised as either lobar or non-lobar. Non-lobar haemorrhages included subcortical, thalamic, basal ganglionic, brainstem, and cerebellar haemorrhages. Mixed haemorrhage: those that extended into both cortex and subcortical regions were excluded from the study.
We assessed the modes of distribution prior to the start of statistical analysis in order to select the appropriate test. We performed exploratory univariate analyses for associations between patient characteristics and the primary outcomes. Co-linearity was assessed with Spearman’s correlation. Multivariable logistic regression was used to determine the independent predictors of haematoma expansion and poor clinical outcome. Candidate variables were those identified during exploratory analysis with p < 0.10. Non-significant variables were eliminated. We assessed the effect of haemorrhage location on our secondary outcome variables using Chi-Square, Fisher’s exact test, ANOVA, and Mann–Whitney U tests, as appropriate.
In exploratory post hoc analysis, the association of location and expansion was further assessed using significant expansion defined as >33% relative to baseline ICH volume. The primary analysis population was also divided by spot sign status and the effect of haemorrhage location was compared to the primary outcomes. SPSS version 23 (Armonk, New York) was used for all statistical analyses.
Results
Baseline characteristics for primary analysis. Stratified by bleed location.
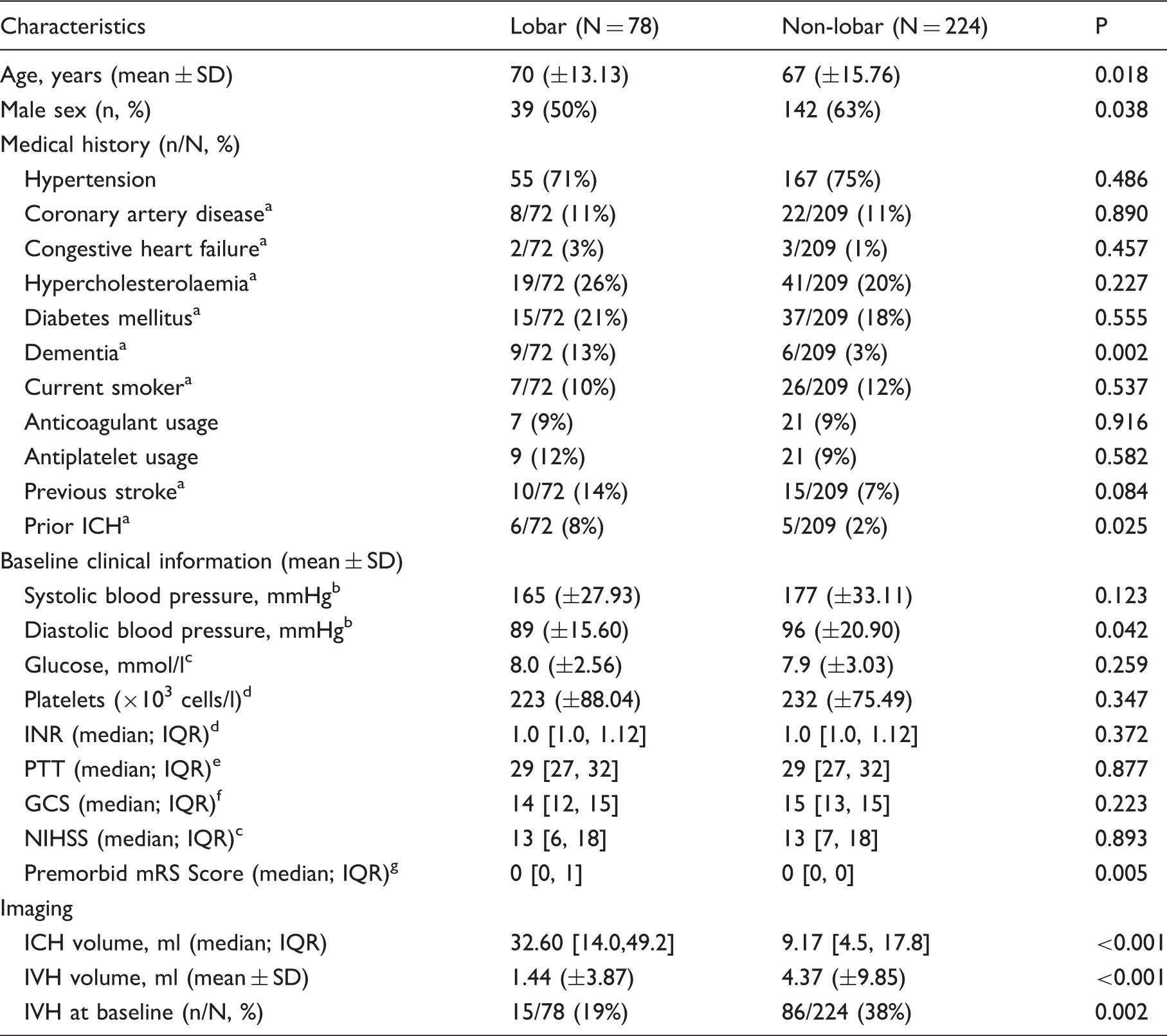
GCS: Glasgow Coma Scale; ICH: intracerebral haemorrhage; INR: international normalized ratio; IQR: interquartile range; IVH: intraventricular haemorrhage; mRS: modified Rankin scale; NIHSS: NIH Stroke Scale; PTT: partial Thromboplastin Time; SD: standard deviation.
Missing 21 values.
Missing one value.
Missing three values.
Missing five values.
Missing 11 values.
Missing 24 values.
Missing 25 values.
Univariate analysis of expansion and clinical outcome in relation to location.
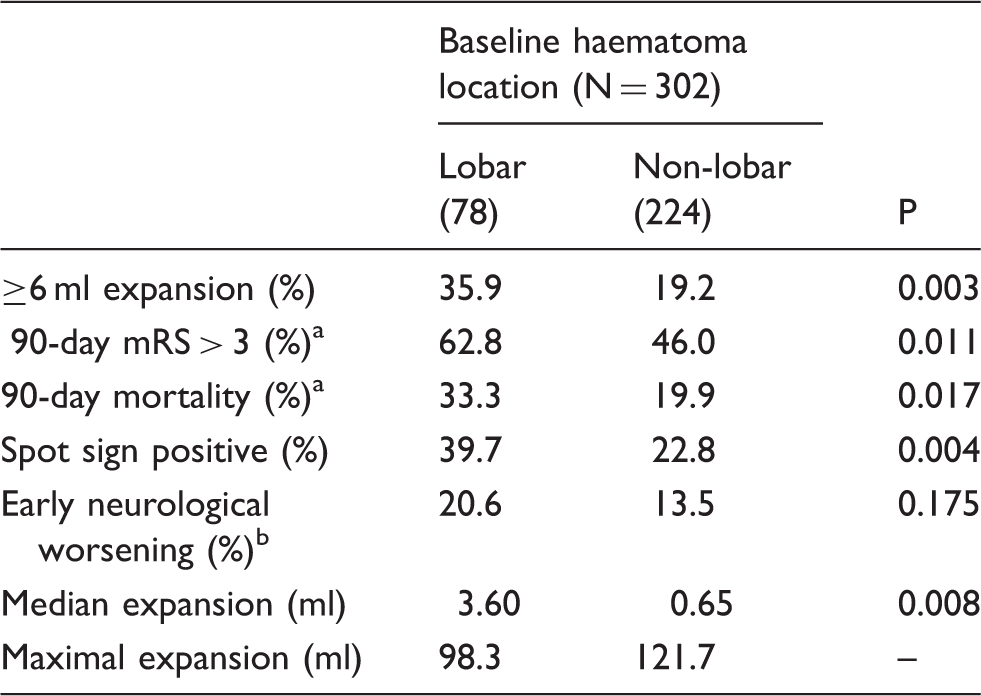
mRS: modified Rankin scale.
Missing 13 values.
Missing 47 values.
Multivariable-adjusted independent predictors of expansion and clinical outcome.
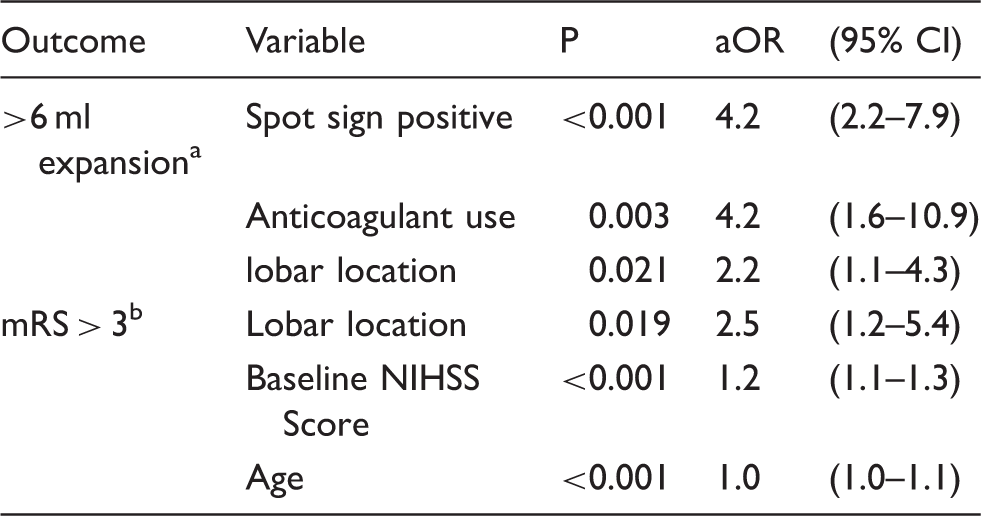
aOR: adjusted odds ratio; CI: confidence interval; mRS: modified Rankin scale; NIHSS: NIH Stroke Scale.
Variables included in analysis: anticoagulant use, baseline GCS and NIHSS, time to CT scan, lobar location, spot sign, prior stroke.
Variables included in analysis: anticoagulant use, baseline GCS, mRS, and NIHSS, lobar location, spot sign, age, baseline IVH volume.
Exploratory univariate analysis revealed associations between poor clinical outcome and anticoagulant use, baseline GCS, mRS, and NIHSS scores, haemorrhage location, spot sign presence, prior stroke, age, and baseline ICH and IVH volumes (all p < 0.1; data not shown). In the univariate model, lobar haemorrhages were associated with 90-day mRS > 3, and mortality. Co-linearity was found between premorbid mRS and prior stroke. Hence, prior stroke was removed from our multivariable analysis. When included into a multivariable model, age, haemorrhage location, and baseline NIHSS independently predicted 90-day mRS scores > 3. Again, baseline ICH volume was not included in the multivariable analysis due to the co-linear relationship with haemorrhage location.
Post hoc analysis: Assessing the influence of spot sign on haematoma expansion.
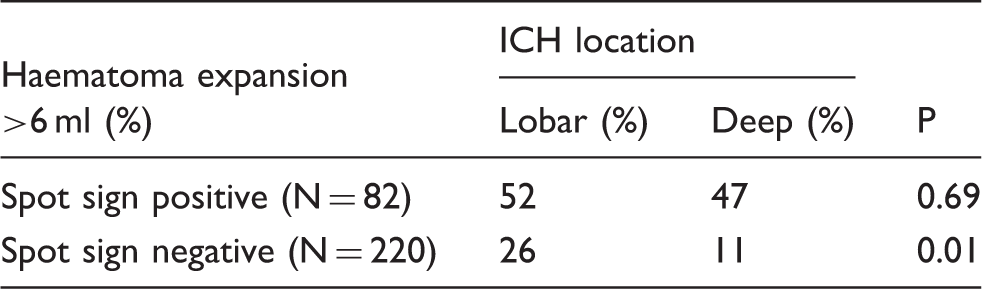
ICH: intracerebral haemorrhage.
Discussion
In our study, lobar ICH was independently associated with haematoma expansion. These findings are consistent with the published literature.5,14 Additionally, baseline lobar haematoma volumes were larger than those of deep ICH. This is also consistent with prior studies.5,15
Lobar haemorrhages, baseline NIHSS, and age were also associated with poor clinical outcome. While the findings of baseline deficit and age are consistent with previous studies, 4 the association of lobar haemorrhages with poor outcome was contrary to our initial hypothesis, and to previous studies that have shown increased mortality with non-lobar bleeds.6,7 Our study excluded patients that did not have follow-up imaging, or whose follow-up CTs were potentially affected by treatments such as recombinant factor VIIa or craniotomy. The exclusion of these patients from the primary analysis may have biased the sample towards less severe outcomes and reduced our ability to detect an association between non-lobar haemorrhage and clinical outcome.
The increased haematoma expansion with lobar ICH seen in this study is likely multifactorial. One proposed mechanism is related to the underlying pathology associated with amyloid angiopathy.16,17 Amyloid angiopathy may increase the likelihood of secondary vessel rupture during the transection of adjacent tissue and previous investigations have suggested that the presence of cerebral microbleeds, the primary imaging finding of amyloid angiopathy, is associated with a higher risk of haemorrhage. 18 Such amyloid haemorrhages are more common in the lobes and in the elderly; our lobar cohort consisted of patients with advanced age and a higher proportion of dementia, which may have enriched the sample with patients at higher risk of amyloid angiopathy. However, the relationship between amyloid angiopathy and haematoma expansion is unclear, as at least one study did not show a discernable association.5,18,19 Recent work by Boulouis et al. 20 has even shown that the absence of cerebral microbleeds correlated with larger baseline haemorrhage volume and increased risk of haematoma expansion in lobar haemorrhages.
There is increasing pathological evidence that haematoma expansion occurs due to mechanical damage to vessels peripheral to the original haematoma. In light of this, the lobar location may be more prone to haematoma expansion due to the extensive network of pial vessels found in the lobes of the brain. A combined effect of extensive vessel networks that can be exposed to mechanical shearing and increased vessel fragility brought on by age or pathologies such as cortical superficial siderosis may predispose lobar bleeds to haematoma expansion. 20 Finally, we propose that the anatomical capacity of the brain may be important in influencing haematoma expansion: perhaps there is simply more room to allow expansion in the lobes as compared to deeper structures where there is a high density of axonal fibres.
We performed exploratory ad hoc analyses to assess the relationship between haemorrhage location and expansion in the context of the spot sign. Multiple spot signs were observed in lobar haemorrhages and as observed in previous studies were associated with higher rates of haematoma expansion. 21 We did not observe an association between haemorrhage location and expansion in spot sign positive patients, suggesting location is less important in this population. However, given the relatively low sensitivity of the spot sign, 4 and the recruitment challenges of recent treatment trials, it is possible that haemorrhage location may be helpful to predict haematoma expansion. In particular, combining haematoma location with other non-contrast CT findings such as hypodensities and heterogeneity of the baseline haemorrhage may lead to better predictive scores for haematoma expansion in both clinical prognostication and in future treatment trials.22,23
Our study has important limitations. Although the data were prospectively collected, the observational nature of the analysis cannot exclude the presence of unknown confounders. Unmeasured baseline differences in clinical function between the lobar and non-lobar group (e.g. cognition) can lead to biased outcomes; this can be minimised in future studies using propensity score matching. The associations between haematoma expansion and location were also non-significant when expansion was defined as >33% of baseline ICH volume. Additionally, larger haematomas, a feature of the lobar haemorrhage cohort, are more often able to expand beyond 6 ml thereby potentially biasing HE towards lobar haemorrhage. However, as previous studies have indicated, an absolute definition of >6 ml falls within the MDD for total haematoma volume. 24 Furthermore, the requirement for follow-up imaging to assess for haematoma expansion likely excluded patients at highest risk of early mortality, which precluded our ability to fully determine the effect of haemorrhage location on clinical outcome. Our choice to exclude patients who received a ventricular drain prior to repeat imaging may have biased our sample towards better outcomes. Finally, these results would benefit from replication in a larger external cohort. This is of importance, as several recent studies have shown that particular locations within the lobes of the brain and thalamus have differing influence on clinical outcome.25,26
In conclusion, we showed that haemorrhage location can predict haematoma expansion and clinical outcome in spontaneous ICH. In particular, we found that lobar ICH was associated with significant haematoma expansion and poor outcome. As haematoma expansion is a promising therapeutic target, haemorrhage location may be helpful for prognostication and as an additional selection tool in ICH clinical trials.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VY, CL, ID, CAM, AMD, RIA, DRL, AK, JMB, VP, JR, CSK, RB, MDH, DD declare that there is no conflict of interest. GG is the Chair of the DSMB for SPOTLIGHT.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DD was supported by a Heart & Stroke Foundation of Canada New Investigator Award and a University of Ottawa Department of Medicine Clinician-Scientist Chair Award. MDH was supported by the Heart and Stroke Foundation of Alberta, the Hotchkiss Brain Institute, and by Alberta Innovates Health Solutions. AMD was supported by the Heart and Stroke Foundation, the Hotchkiss Brain Institute, and by Alberta Innovates Health Solutions. The PREDICT study was supported by an unrestricted grant from NovoNordisk Canada
Informed consent
Written informed consent was obtained from all subjects before the study.
Ethical approval
Ethical approval for this study was obtained from all PREDICT enrolling sites.
Guarantor
VY.
Contributorship
VY and DD conceived the study and wrote the manuscript. VY, DD, MDH performed and interpreted the data analysis. All authors reviewed and edited the manuscript and approved the final version of this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
