Abstract
Purpose
Haematoma expansion is a devastating complication of intracerebral haemorrhage (ICH) with no established treatment. Tranexamic acid had been an effective haemostatic agent in reducing post-operative and traumatic bleeding. We review current evidence examining the efficacy of tranexamic acid in improving clinical outcome after ICH.
Method
We searched MEDLINE, EMBASE, CENTRAL and clinical trial registers for studies using search strategies incorporating the terms ‘intracerebral haemorrhage’, ‘tranexamic acid’ and ‘antifibrinolytic’. Authors of ongoing clinical trials were contacted for further details.
Findings
We screened 268 publications and retrieved 17 articles after screening. Unpublished information from three ongoing clinical trials was obtained. We found five completed studies. Of these, two randomised controlled trials (RCTs) comparing intravenous tranexamic acid to placebo (n = 54) reported no significant difference in death or dependency. Three observational studies (n = 281) suggested less haematoma growth with rapid tranexamic acid infusion. There are six ongoing RCTs (n = 3089) with different clinical exclusions, imaging selection criteria (spot sign and haematoma volume), time window for recruitment and dosing of tranexamic acid.
Discussion
Despite their heterogeneity, the ongoing trials will provide key evidence on the effects of tranexamic acid on ICH. There are uncertainties of whether patients with negative spot sign, large haematoma, intraventricular haemorrhage, or poor Glasgow Coma Scale should be recruited. The time window for optimal effect of haemostatic therapy in ICH is yet to be established.
Conclusion
Tranexamic acid is a promising haemostatic agent for ICH. We await the results of the trials before definite conclusions can be drawn.
Keywords
Introduction
Spontaneous intracerebral haemorrhage (ICH) is a devastating condition with a median 30-days mortality rate of 40%. 1 It is also associated with significant morbidity with 60% of patients remaining dependent one year after the event. 1 Haematoma expansion (HE) is a major cause of early neurological deterioration and poor outcome, occurring in 38% of ICH patients within the first few hours from onset of symptoms. 2 Nevertheless, patients with small haematoma volume have an excellent prognosis. 3 Therefore, there is an urgent need to direct treatment goal towards limiting haematoma size.
To date, there is no proven treatment to limit HE. However, two main non-surgical strategies have been postulated to reduce HE: ameliorating the driving force of HE and haemostatic therapy. 4 The former can be achieved through reduction of perfusion pressure by decreasing systemic blood pressure. The INTERACT trial reported reduction in HE (mean haematoma volume difference of 3.15 mL at 24 h, p = 0.004) in patients treated with intensive blood pressure reduction (systolic blood pressure, SBP < 140 mmHg) as compared to standard blood pressure reduction (SBP < 180 mmHg). 5 In addition, The INTERACT 2, ATACH and ATACH-II trials reported non-significant reduction in HE. However, these did not translate into significant reduction in death or dependency.6–8 At the same time, there were concerns that rapid blood pressure reduction may result in cerebral ischaemia. The INTERACT, INTERACT 2, ATACH and ATACH-II reported no significant cerebral ischaemic events in patients treated with intensive blood pressure reduction.5–8 Current guidelines suggest treating blood pressure to a SBP of 140 mmHg is safe and can/may be effective.9,10
Several haemostatic agents have been researched for possible effect in limiting HE. An extensively researched haemostatic agent, recombinant activated factor VIIa, was highly effective in reducing HE but failed to significantly improve clinical outcomes in five trials (total patient number = 1395), possibly due to a trend towards increased thromboembolic complications. 11
Tranexamic acid
Tranexamic acid has been used as an effective haemostatic agent in acute gastrointestinal bleeding, menorrhagia, epistaxis, post-cardiac and post-orthopaedic surgery.12–16 Besides reducing bleeding rate and need for blood transfusion, tranexamic acid also improves healing and reduces inflammation.17,18
Tranexamic acid has been studied in subarachnoid, subdural and traumatic intracranial haemorrhage. In aneurysmal subarachnoid haemorrhage (SAH), tranexamic acid was effective in stopping bleeding when use in conjunction with endovascular coiling or clipping, 19 although an increased risk of cerebral ischaemia offset any benefit. 20 Many of the trials of tranexamic acid in SAH involved giving high doses of up to 6 g per day for a prolonged duration of three to six weeks, and this could have accounted for the increased ischaemic events. In view of this, an ongoing trial is examining the efficacy of short-term tranexamic acid in SAH at lower dose, i.e. 1 g given 8 hourly for a maximum of 24 h. 21
The Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage (CRASH-2), a megatrial involving 20,211 patients found that tranexamic acid reduced mortality in patients with polytrauma (Odds Ratio (OR) = 0.91, 95% confidence interval (CI) = 0.85–0.97). 22 A subgroup analysis of CRASH-2 (n = 270) showed that patients with traumatic ICH had a non-significant reduction in mortality (adjusted OR = 0.47, 95% CI = 0.21–1.04). 23 An ongoing trial, CRASH-3, is examining the efficacy of tranexamic acid in improving outcome after traumatic brain injury. 24 An additional trial is testing tranexamic acid in the non-surgical treatment of chronic subdural haematoma.25,26
Given these promising results from trials of tranexamic acid in various conditions, we examined the efficacy of tranexamic acid in improving clinical outcome in spontaneous ICH.
Methods
The aim was to assess whether tranexamic acid, as compared to placebo or no additional treatment, improved outcome in patients with intracerebral haemorrhage. Randomised and quasi-randomised controlled clinical trials, and before-after studies, were included and assessed by systematic review and meta-analysis.
Trials were included if they involved adult patients with primary non-traumatic intracerebral haemorrhage, i.e. likely to be due to hypertension or cerebral amyloid angiopathy. Trials of patients with secondary ICH due to arteriovenous malformation, intracranial aneurysm, cavernoma, dural arteriovenous fistula, haemorrhagic brain metastasis, trauma or thrombolysis were excluded. Trials could use tranexamic acid at any dose, route of administration and time window for administration. The main functional outcomes of interest were modified Rankin Scale (mRS), Barthel Index and Glasgow Outcome Scale. Meta-analysis of the primary outcome was performed if at least two RCTs reported relevant data.
Trials were identified in MEDLINE, EMBASE and CENTRAL using search strategies incorporating the terms ‘tranexamic acid’, ‘antifibrinolytic’ and ‘intracerebral haemorrhage’. No language restrictions were imposed in searches. Searches of www.clinicaltrials.gov, the European registry of clinical trials and WHO international clinical trial registry were also performed, which covered both ongoing and unpublished trials. Where relevant, experts in the field were contacted for further information regarding unpublished data or trials. A complete list of search strategies and results is included in the online supplementary material.
Two authors (NS, ZL) independently screened the search results for potential studies. The full text of shortlisted articles was then obtained for review.
Results
Identified studies
Our search identified 268 articles; after deduplication, 163 articles were available for screening. Of these, 146 articles were excluded for various reasons (Figure 1). The full text of 17 articles were extracted and reviewed. A search of clinical trial registers revealed nine relevant records of trials, which corresponded to four clinical trials. Some clinical trials were registered in more than one trial register. Unpublished information on three ongoing studies was obtained from authors. These articles and unpublished information described five completed and six ongoing studies that are described in the following text.
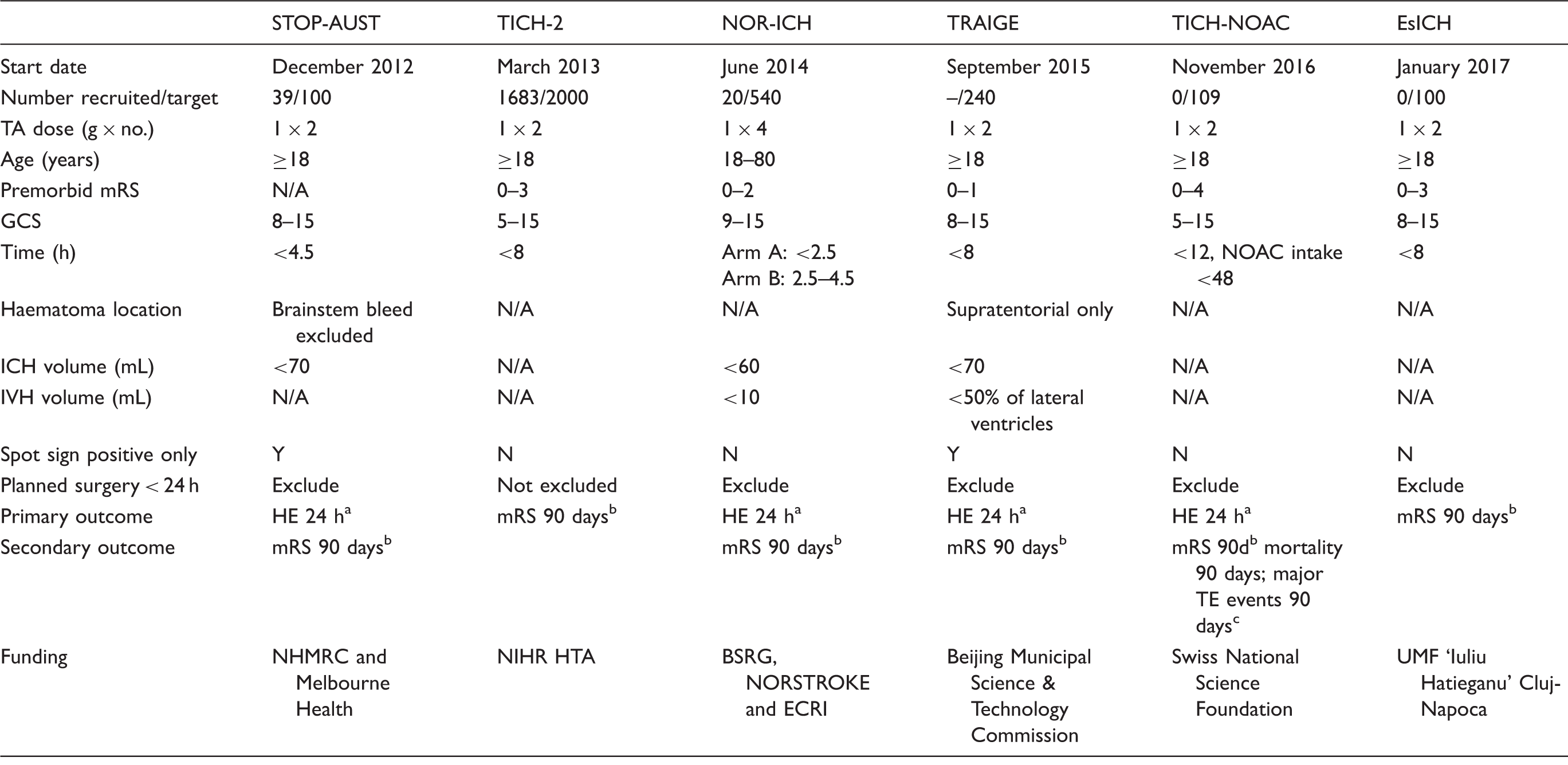
PRISMA flow chart of systematic search. ICH: intracerebral haemorrhage; ICTRP: International Clinical Trials Registry Platform; RCT: randomised controlled trial. Completed studies and clinical trials of tranexamic acid in ICH. GOS: Glasgow Outcome Scale; HE: haematoma expansion; ICH: intracerebral haemorrhage; IV: intravenous; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; NR: not reported; RCT: randomised controlled trial; TA: tranexamic acid. Paper additionally includes all rapid infusion patients of the 2005 study, total n = 188. Comparison of ongoing trials of tranexamic acid in ICH. BSRG: Bergen Stroke Research Group; ECRI: European Cerebrovascular Research Infrastructure; GCS: Glasgow coma scale; HE: haematoma expansion; ICH: intracerebral haemorrhage; IVH: intraventricular haemorrhage; mRS: modified Rankin Scale; N/A: not available; NHMRC: National Health and Medical Research Council, Australia; NOAC: novel oral anticoagulants; NORSTROKE: Norwegian Stroke Research Registry; TA: tranexamic acid; UMF: University of Medicine & Pharmacy. Haematoma expansion measured at 24 h post-ictus. Modified Rankin Scale at 90 days. Myocardial infarction, ischemic stroke, pulmonary embolism.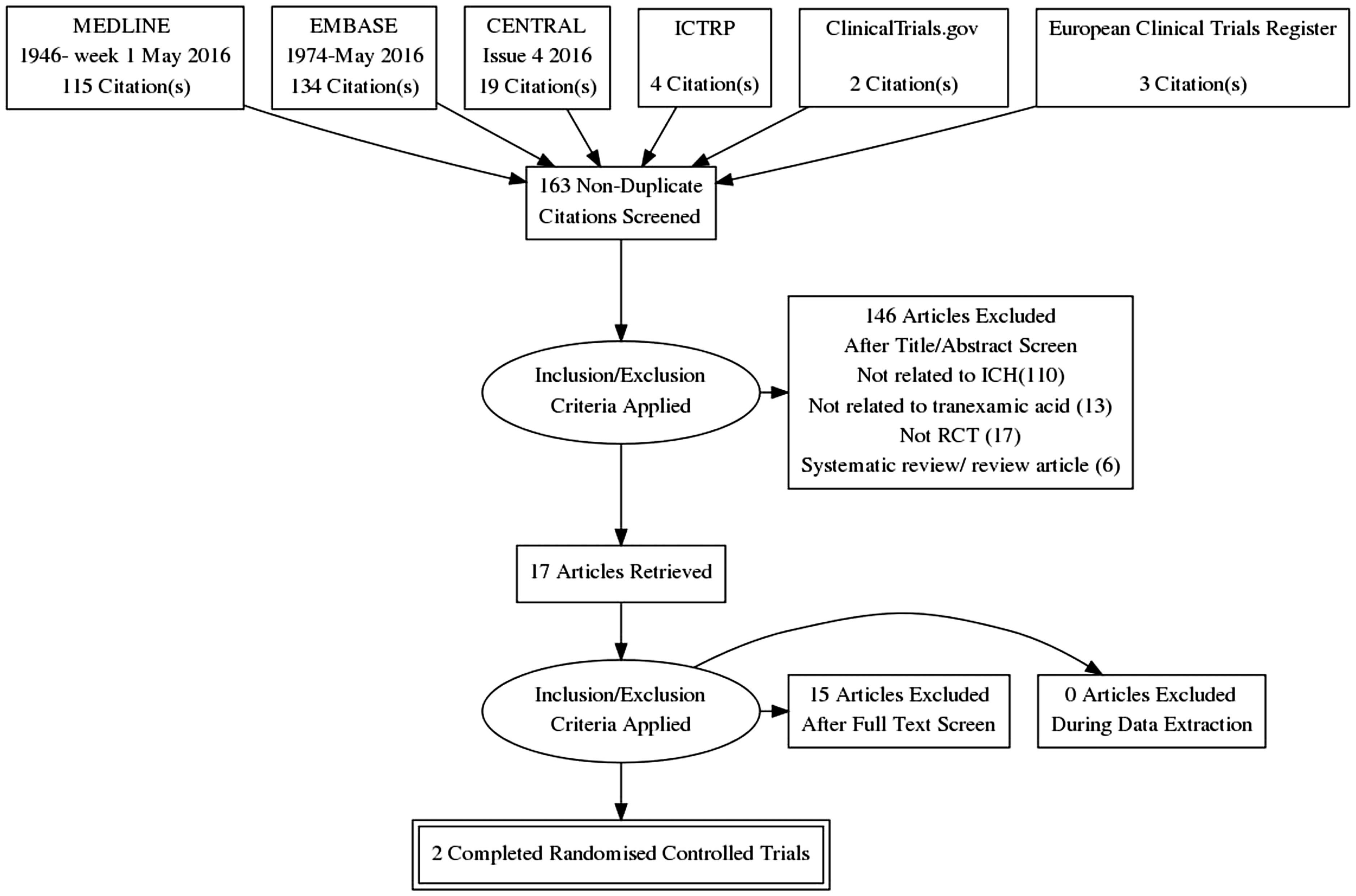
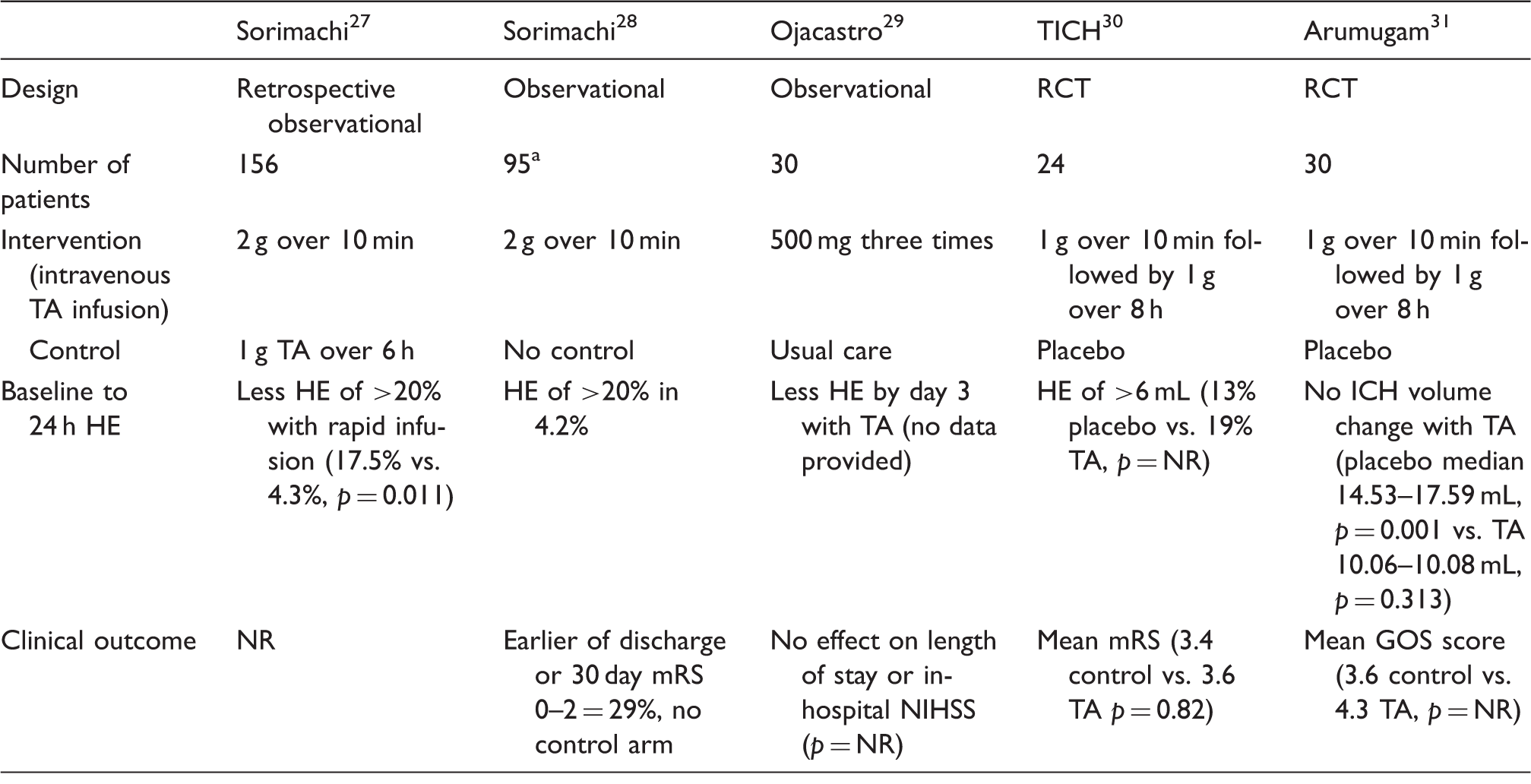
The five completed studies including two small RCTs involving 54 patients (Table 1). Since these trials did not report sufficient information on clinical outcomes, meta-analysis could not be performed.
Completed observational studies and clinical trials
Observational studies
Sorimachi et al. reported a retrospective observational before–after study comparing the efficacy of rapid infusion of tranexamic acid 2 g over 10 min to prolonged infusion of 1 g over 6 h in patients presenting within 24 h of ICH. The study recruited 156 patients (>80% presented within 3 h of onset) and showed that rapid infusion was associated with less haematoma enlargement of >20% than prolonged infusion (17.5% vs. 4.3%, p = 0.011). 27 The authors subsequently reported a study of additional 95 patients treated with rapid infusion of tranexamic acid with HE occurring in 4.2% of these patients. 28 However, there was no control arm in either study.
Ojacastro et al. reported an observational study of 30 patients aged 40–70 years with hypertensive ICH who received either intravenous tranexamic acid 500 mg for three doses or standard care. The study is reported as an abstract only, with no data other than a general statement of significant haematoma volume reduction with tranexamic acid without effect on length of stay or in-hospital National Institute of Health Stroke Scale (NIHSS). 29 We were unsuccessful in contacting the authors for further information.
RCTs
The Tranexamic acid in IntraCerebral Haemorrhage (TICH) trial recruited 24 patients within 24 h (mean 12.6 h) of ICH to receive either 1 g tranexamic acid or placebo over 10 min followed by 1 g over 8 h. There was no 90-day outcome difference (mean mRS 3.6 in treatment arm and 3.4 in control, p = 0.82) with non-significantly more HE (>6 mL growth) in the treatment arm (19% vs. 13%). 30 However, this trial was designed as a feasibility trial and not to demonstrate efficacy.
In addition, Arumugam et al. conducted a single-centre, single-blind RCT that allocated 30 patients to receive within 8 h (mean 6 h) of ICH either intravenous tranexamic acid 1 g over 10 min followed by 1 g over 8 h or placebo. The control group had significant HE (median growth from 14.53 to 17.59 mL, p = 0.001) while the treatment group did not (10.06–10.08 mL, p = 0.313). 31 The mean Glasgow Outcome Scale score was 3.6 in the control group and 4.4 in the treatment group, but no statistical tests comparing the groups were provided.
Ongoing clinical trials (including those in preparation, Table 2)
STOP-AUST. The Spot sign and Tranexamic acid On Preventing ICH growth – AUStralasia Trial (STOP-AUST, NCT01702636) is a phase-II multicentre randomised, double-blind, placebo-controlled trial exploring the efficacy of tranexamic acid in preventing HE in ICH patients, recruiting patients from Australia, Finland and Taiwan. 32 The trial selects patients with a positive spot sign on Computed Tomography (CT) angiography to receive either placebo or 1 g tranexamic acid over 10 min followed by 1 g over 8 h. The primary outcome measure is HE (>33% or >6 mL) by 24 h, with mRS and thromboembolic complications at 90 days as secondary outcomes. The trial aims to recruit 100 patients and has recruited 39 patients by August 2016.
TICH-2
Tranexamic acid for Intracerebral Haemorrhage-2 (TICH-2, EudraCT number 2012-004108-37) is a phase-III, pragmatic double-blind, randomised, placebo-controlled trial aiming to recruit 2000 patients in the United Kingdom and internationally. The study commenced in March 2013 and is due to be completed in December 2017. It has recruited 1683 patients as of August 2016. The TICH-2 trial is recruiting ICH patients presenting within 8 h of onset regardless of whether they had CT angiography to detect positive spot sign, a predictor of HE. Of the patients recruited, only 7% (n = 103) had CT angiography and of that, 17.5% (n = 18) were spot-sign positive. The patients are randomised to receive intravenous tranexamic acid 1 g bolus (given within 10 min) followed by a 1 -g infusion given over 8 h. The primary outcome assessed is death or dependency using mRS at 90 days. Secondary outcomes include neurological impairment (NIHSS) at day 7 or sooner, functional outcome (Barthel index), quality of life (EuroQol, EQ-5D, and EQ-VAS), cognition (Telephone Interview Cognition Score-Modified), mood (Zung Depression Scale) at days 90 and 365, cost, radiological efficacy and safety outcomes up to day 90. Selected centres are invited to participate in an MRI sub-study looking at diffusion weighted imaging hyperintense lesions (DWIHLs), peri-haematomal oedema volume and diffusion restriction at five days as well as haematoma cavity volume and surrounding gliosis at 90 days. 33
PATCH-iT is a Danish sub-study of TICH-2 aiming to recruit 72 patients with spot sign or post-contrast extravasation. Primary endpoint in analysis of this sub-group is haematoma volume in spot-sign positive patients; secondary endpoints include difference in proportion of neurological deterioration within 24 h of symptom onset; recruitment started in July 2016.
NOR-ICH
The Norwegian Intracerebral Haemorrhage Trial (NOR-ICH, EudraCT number 2012-005594-30) is a multicentre prospective randomised, open-label, blinded endpoint (PROBE) trial. There are two arms in the study: arm A treat patients presenting within 2.5 h from onset and arm B treat patients presenting between 2.5 and 4.5 h. The trial recruits spot positive and negative patients. The participants are randomised to receive IV tranexamic acid 1 g bolus followed by 1 g at 2 h, 4 h and 8 h from the first dose compared to no treatment. The primary endpoint is HE at 24 h while secondary endpoints include increase in NIHSS score ≥ 4 within 36 h, NIHSS at day 7, mRS at 90 days and symptomatic thromboembolic events within seven days. The investigators originally planned to recruit 540 patients by early 2017. However, the recruitment of patients has been slow, as many participating centres have not started recruiting patients yet.
TRAIGE
Tranexamic Acid for Acute ICH Growth prEdicted by Spot Sign (TRAIGE, NCT02625948) is a multicentre phase-II randomised, double blind, placebo-controlled trial recruiting patients from China who have a positive spot sign on CT angiography and can be treated within 8 h from onset. The trial compares the efficacy of intravenous tranexamic acid followed by intravenous tranexamic acid 1 g infusion over 8 h versus placebo. The primary outcome measure is HE (>33% or >6 mL) by 24 h, with mRS and thromboembolic complications at 90 days as secondary outcomes. The trial aims to recruit 240 patients by December 2018.
TICH-NOAC
Tranexamic acid for IntraCerebral Haemorrhage secondary to Novel Oral AntiCoagulants (TICH-NOAC, NCT02866838) is an investigator-initiated, multicentre double-blind placebo-controlled trial, investigating the safety and efficacy of tranexamic acid in ICH related to non–vitamin K (direct) oral anticoagulants (NOAC). 34 The trial will include patients from Switzerland who had ICH and taken an NOAC (dabigatran, apixaban, rivaroxaban, edoxaban) within the prior 48 h, or proven NOAC activity by relevant coagulation assays. The time window for recruitment is up to 12 h after the onset of ICH. Patients are randomised to receive tranexamic acid 1 g bolus followed by 1 g infusion over 8 h or placebo. All patients received standard treatment including specific antidote where available. TICH-NOAC aims to demonstrate that treatment with tranexamic acid reduces rate of HE as measured at 24 h compared to the best medical treatment. The study implementation process has started. The first patient is expected to be included in Fall 2016.
EsICH
Tranexamic acid and biomarkers in Emergency management of Spontaneous IntraCerebral Hemorrhage (EsICH) is a Romanian multicentre double-blind RCT recruiting spot-sign positive and negative ICH patients within 8 h of onset. Patients will be given a total of 2 g tranexamic acid or placebo (normal saline, 10 mL in total) during a course of 8 h. A series of biomarkers will be tested upon arrival in the emergency department (including point-of-care biomarkers for troponin, D-dimer and C-reactive protein) to evaluate their role in early risk stratification. The primary endpoint is a favourable functional outcome (mRS 0–3) at 90 days, assessed by telephone survey. The study is awaiting ethical approval. EsICH aims to recruit 100 patients between January 2017 and December 2018.
Discussion
Tranexamic acid is a promising haemostatic agent given its efficacy in reducing bleeding rates in surgical and trauma patients. However, there is currently insufficient evidence from completed trials to draw conclusion on the effect of tranexamic acid on clinical outcome and HE in ICH. The completed trials recruited only a small number of patients and did not provide sufficient data for meta-analysis.
Apart from recruiting patients with primary ICH while excluding secondary ICH, the six ongoing trials vary in terms of selection criteria, dosing regime and outcome assessments. This raises several questions about the design of clinical trials. The first is whether tranexamic acid should only be given to patients with positive spot sign. Some trials only include patients who had positive spot sign on CT angiography (STOP-AUST, PATCH-it and TRAIGE) while others do not (TICH-2, NOR-ICH, TICH-NOAC and EsICH). A positive spot sign is an indication of ongoing haemorrhage in ICH, with a 61% chance of significant HE (>33% or >6 mL). 35 However, approximately 22% of spot-sign negative patients also have HE.35,36 Therefore, excluding spot-sign negative patients may unnecessarily preclude them from possible benefits. 36 On the other hand, targeting haemostatic therapy to patients with ongoing haemorrhage only will reduce the number of patients unnecessarily exposed to potential thromboembolic complications. As CT angiography is not done routinely in ICH in many centres, translation of clinical trial results into practice would be easier if spot-sign evaluation was not necessary.
The second clinical question is: what is the optimal time window that tranexamic acid might be beneficial? Whilst there is no doubt that treatment benefit is time dependent, the exact cut-off time is uncertain. STOP-AUST and NOR-ICH exclude patients presenting later than 4.5 h, where presumably HE would have occurred. However, a few considerations should be taken into account here. In a study based on serial measurements of haematoma volumes by B-mode ultrasound, HE was observed up to 7–8 h after symptom onset but most frequently in the first hours. 37 Brott et al. 2 reported that 26% of HE occurred by 4 h and an additional 12% HE between 4 and 20 h. By recruiting patients presenting later than 4 h, a larger pool of patients who may suffer subsequent HE may be included. In addition, there may be other causes of neurological deterioration besides HE such as peri-haematomal oedema and neuroinflammation. 38 Tranexamic acid may potentially reduce peri-haematomal oedema and attenuate inflammatory response, a process that starts within the first few hours and last for days to weeks after ICH. On the other hand, the mega-trial CRASH-2 demonstrated in trauma patients a strongly time-dependent treatment effect with tranexamic acid, with reduced mortality when given within 3 h and strongly increased mortality when given beyond 5 h, when compared to placebo. 39 A clearer time window in ICH may be established in the future pending results of ongoing studies.
The third question is whether patients with poor prognostic factors should be excluded from trials. Such factors include advanced age, poor pre-morbid mRS, poor Glasgow Coma Scale (GCS), infratentorial haematoma, high haematoma volume (>60 mL or 70 m), intraventricular extension and patient undergoing surgery should be excluded from trials. A key point in anti-fibrinolytic therapy is that it aims to prevent HE but not treat HE after it occurred. It would be logical to exclude patients in whom the HE has presumably already occurred (high haematoma volume, intraventricular extension) or has resulted or would have resulted in poor clinical outcome (poor GCS, brainstem haematoma, need for surgery). However, the initial haematoma volume itself is a predictor of HE. 40 The risk of HE is greater in large haematoma and as there is no definite way to determine whether a rebleeding has stopped, tranexamic acid may still benefit those with poor prognostic factors. 40 As customary, we noted that of the ongoing trials, the phase-III trial was more inclusive.
The optimal dosing of tranexamic acid needs to be determined as well. After intravenous administration, tranexamic acid reaches peak concentration almost immediately and has an onset of action within a few minutes. It has a short plasma elimination half-life of 3 h. The tissue half-life is 17 h due to low plasma protein binding (3%) and wider distribution in the tissue compartment. 41 Repeated dosing may result in higher concentration and accumulation in the brain. TICH-2, STOP-AUST, TRIAGE, TICH-NOAC and EsICH trials administer intravenous tranexamic acid at the same dose similar to the CRASH-2 trial (1 g bolus, 1 g infusion over 8 h), while NOR-ICH involves giving 1 g tranexamic acid boluses four times in the first 24 h. It is not known whether more frequent dosing will result in longer accumulation in the target organ or whether this will result in better outcome or more adverse events.
The effects of tranexamic acid beyond anti-fibrinolysis remain to be explored. DWIHL indicative of acute or subacute ischaemic infarcts are known to co-exist in ICH. 42 Given the potential of tranexamic acid to precipitate ischaemic events, the TICH-2 MRI sub-study explores whether the incidence of DWIHL will be higher in those receiving the drug. Second, tranexamic acid may have anti-inflammatory and neuroprotective effects. This may reduce peri-haematomal oedema and improve healing process manifesting as a reduction in residual haematoma cavity and surrounding gliosis.
We plan to resolve the uncertainties concerning the use of tranexamic acid by conducting an individual patient data meta-analysis (IPDMA). IPDMA had been shown to improve data quality and reliability of effect estimates of several stroke interventions.43–45 In addition, a broader meta-analysis of clinical trials encompassing the use of tranexamic acid in polytrauma, traumatic brain injury, SAH, subdural haemorrhage and spontaneous ICH may provide answers regarding the optimal time window and dosage of tranexamic acid. Such a meta-analysis may provide a more accurate estimation of thromboembolic risk with use of tranexamic acid as well.
Conclusion
In summary, the results of ongoing trials of tranexamic acid for ICH are awaited. Future trials should also examine the effect of tranexamic acid on plasma biomarkers and pathological changes to determine the effects of tranexamic acid. Prospective meta-analysis of individual patient data from these ongoing trials will provide the most reliable method of assessing the efficacy and safety of tranexamic acid.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AM has received speaker honoraria and consulting fees from Boehringer Ingelheim, Stryker, Phagenesis, and Nestle, and is the principal investigator of the STOP-AUST trial. STE has received funding for travel or speaker honoraria from Bayer and Boehringer Ingelheim. He has served on scientific advisory boards for Bayer, Boehringer Ingelheim, BMS/Pfizer and Covidien, and on the editorial board of Stroke. He has received an educational grant from Pfizer and research support from the Science Funds (Wissenschaftsfonds) of the University Hospital Basel, the University of Basel, the Swiss Heart Foundation and the Swiss National Science Foundation. He is the co-principal investigator of TICH-NOAC. PB is Stroke Association professor of Stroke Medicine. He has received consulting fees from Athersys, Nestle, Phagenesis and ReNeuron. He is an unpaid advisor to Platelet Solutions. EMM has been granted a 2016 ESO fellowship through the ‘Department-to-Department Visit’ program for a one-week study visit at TICH-2 Coordinating Centre from Nottingham, UK.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The STOP-AUST study is funded by the National Health and Medical Research Council (GNT1081718) and Melbourne Health (PG-016-2014). TICH-2 is funded by NIHR HTA grant (project code 11_129_109). NOR-ICH is supported by BSRG, NORSTROKE and ECRI. TRAIGE is supported by Beijing Municipal Science & Technology Commission. TICH-NOAC is funded by Swiss National Science Foundation and EsICH by UMF ‘Iuliu Hatieganu’ Cluj-Napoca.
Ethical approval
Not applicable
Informed consent
Not applicable
Guarantor
NS.
Contributorship
NS and ZKL researched literature and conceptualised the review. ZKL wrote the first draft of the manuscript. All authors reviewed, edited and approved the final version of the manuscript.
Acknowledgements
None.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
