Abstract
Background
Following stroke, acute symptomatic seizures (manifestation within seven days) and epilepsy, i.e. occurrence of at least one unprovoked seizure (manifestation after more than seven days), are reported in 3–6% and up to 12% of patients, respectively. Incidence of acute symptomatic seizures is higher in intracranial haemorrhage (10–16%) than in ischaemic stroke (2–4%). Acute symptomatic seizures and unprovoked seizure may be associated with unfavourable functional outcome and increased mortality. In view of the clinical relevance, the European Stroke Organisation has issued evidence-based guidelines on the management of post-stroke seizures and epilepsy.
Method
A writing committee of six clinicians and researchers from five European countries and Israel identified seven questions relating to prevention of (further) post-stroke seizures and epilepsy and to amelioration of functional outcome and prevention of mortality. Recommendations are based on findings in randomised controlled trials and observational studies using the grading of recommendations assessment, development and evaluation approach.
Results
In the absence of adequately powered randomised controlled trials, evidence for all recommendations is very low. Based on findings in observational studies, some weak recommendations have been made. In most instances, we suggest not to administer antiepileptic drugs. Due to high incidence of seizure recurrence after one post-stroke unprovoked seizure, secondary antiepileptic drugs prophylaxis needs to be considered.
Conclusion
Due to very low evidence, these guidelines only give some weak recommendations on prevention of occurrence and recurrence of post-stroke acute symptomatic seizures and unprovoked seizure. Adequately powered randomised controlled trials are required to assess interventions for post-stroke seizure management.
Keywords
Introduction
The relationship between stroke and epileptic seizures or epilepsy is bidirectional. For 1 in 10 adult patients, new-onset epilepsy can be attributed to stroke, and this aetiology is seen in almost every fourth epilepsy patient aged 65 years and above. 1 Interestingly, middle-aged and elderly patients with newly diagnosed epilepsy have a two- to three-fold increased risk to suffer from subsequent stroke within the next couple of years.2,3 The hypothesis behind this finding is that epilepsy in those patients may be caused by subtle microangiopathic alterations predisposing to later overt cerebrovascular events, thus seizures may be an early biomarker for subsequent stroke.
Definitions of terms used in the current guidelines.
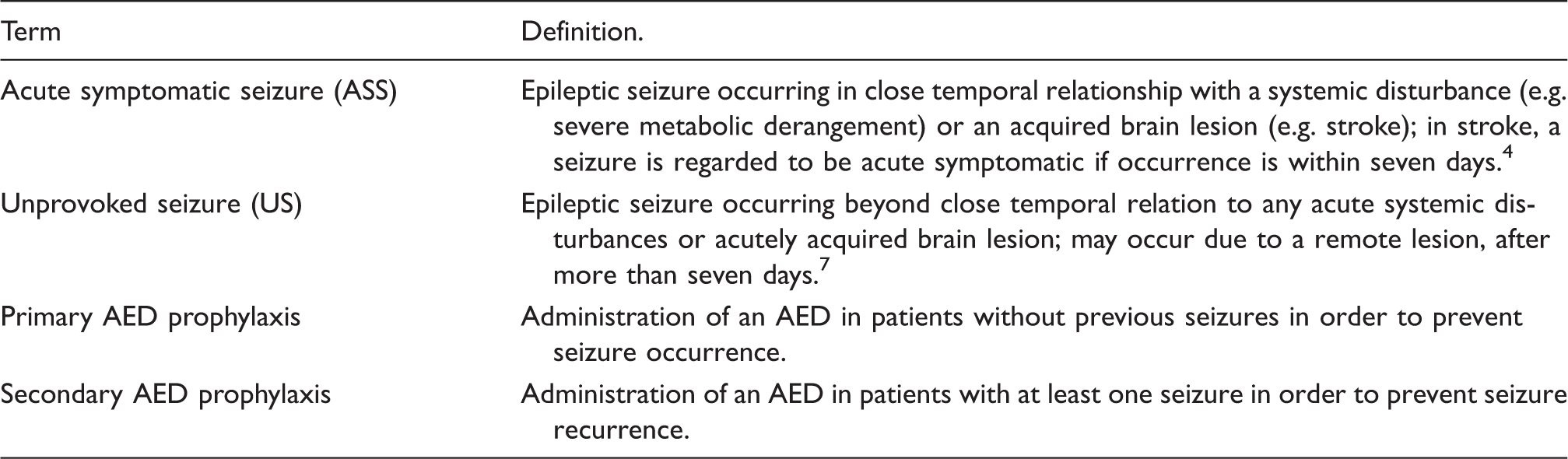
AED: antiepileptic drug.
Multiple epidemiological studies have consistently reported that incidence of ASS in all stroke patients lies between 3 and 6%.8–13 Incidence rates are higher in patients with intracranial, i.e. intracerebral or subarachnoid, haemorrhage rising to 10 to 16%.10,11 Independent risk factors identified by multivariate analyses include cortical involvement, total anterior circulation infarct, severe stroke, and haemorrhagic transformation of ischaemic stroke.8–13 Continuous EEG monitoring in the intensive care setting has demonstrated that a substantial portion of stroke patients has electrographic seizures without a clinical correlate. In 102 patients with intracerebral haemorrhage, 18% had unequivocal seizure patterns in the EEG, while only one patient exhibited behavioural seizures. 14 As by now the clinical significance of pure electrophysiological seizures is undetermined, these paroxysmal EEG phenomena are not considered in the current guideline. For recommendations on the use of continuous EEG in critically ill stroke patients, we refer to the consensus statement from the Neurointensive Care Section of the European Society of Intensive Care Medicine. 15
Unprovoked seizures, i.e. post-stroke epilepsy, have been reported in 10 to 12% of patients with a follow-up of 5 to 10 years16,17 when the new practical definition of epilepsy is applied. 6 Interestingly, the risk is similar considering ischaemic infarction and different forms of intracranial bleeding such as intracerebral and subarachnoid haemorrhage. Independent risk factors comprise cortical involvement and stroke size and severity.16–20 In patients with subarachnoid haemorrhage, independent predictors include accompanying intracerebral haemorrhage of more than 15 cm3 volume, Hunt & Hess grade III-V, and ASS. 21
Some epidemiological studies have addressed the question if ASS or US determines unfavourable functional outcome or increased mortality, but findings are contradictory.9,10,17,19,22–28
So far, no clear consensus exists on indications for primary or secondary prophylaxis with antiepileptic drugs (AED) in regard of post-stroke ASS and US.29,30 Therefore, the European Stroke Organisation (ESO) decided to issue guidelines incorporating recommendations on how to prevent or manage post-stroke seizures and epilepsy. These recommendations are based on findings in randomised controlled trials (RCTs) and observational studies. They were agreed on in consensus by the involved authors using the grading of recommendations assessment, development and evaluation (GRADE) approach and the ESO standard operating procedure (SOP) for guidelines development 31 and have the approval of the ESO Executive Committee.
The aim of this Guideline document is to assist physicians treating patients with ischaemic or haemorrhagic stroke with or without additional seizures or epilepsy, both in the acute hospital-based setting (emergency physicians, neurologists) and on a long-term outpatient basis (general practitioners, internists, neurologists), in their clinical decisions with regard to primary or secondary prophylactic management with antiepileptic drugs.
Methods
A group of six clinical researchers with expertise in epilepsy and/or stroke from five European countries and Israel was proposed by the Guidelines Committee of the ESO and confirmed by the ESO Executive Committee. The leader of this group (MH) and four other group members (EB, FB, RK, RR) have a long-standing clinical and scientific expertise in epileptology. The co-leader of this group (HC) has a long-standing clinical and scientific expertise in stroke, she was involved in the development of previous ESO guidelines and of the ESO guideline SOP.
31
Following the ESO guideline SOP, selection of leaders and members of the group were based on scientific integrity, professionalism, self-motivation, clinical expertise, availability, and conflicts of interest.
31
Standardised steps which were undertaken by the working group are summarised in the following:
The group discussed and decided by consensus on specific and clinically relevant PICO (patient, intervention, comparator, outcome) therapeutic questions. The entire process of creating these guidelines was guided by the GRADE working group’s recommendations
32
and the ESO SOP.
31
The group identified all available publications related to the PICO questions in a broad single search. These were guided by the 2011 Centre for Evidence Based Medicine’s levels of evidence.
33
We searched the Cochrane database of systematic reviews (CDSR), the Cochrane central register of controlled trials (CENTRAL) as well as MEDLINE (1990 through March 2016). Furthermore, we searched the reference lists of review articles and clinical trials on post-stroke seizures and epilepsy for further appropriate studies (for details, see online Appendix 2). The group selected eligible studies. Due to the rather small number of PICO questions (n = 7) and authors (n = 6), all members of the working group independently screened the relevant articles identified by the electronic search. As we identified only few RCTs and no systematic reviews or meta-analyses of RCTs, we also included observational and epidemiological studies that may allow creating recommendations or proposals. The group graded quality of evidence of RCTs and strength of recommendations by use of the GRADE approach. The final summaries of the quality and strength of evidence and recommendations for each PICO question were discussed by the whole group, recommendations were agreed on by the majority of authors.
32
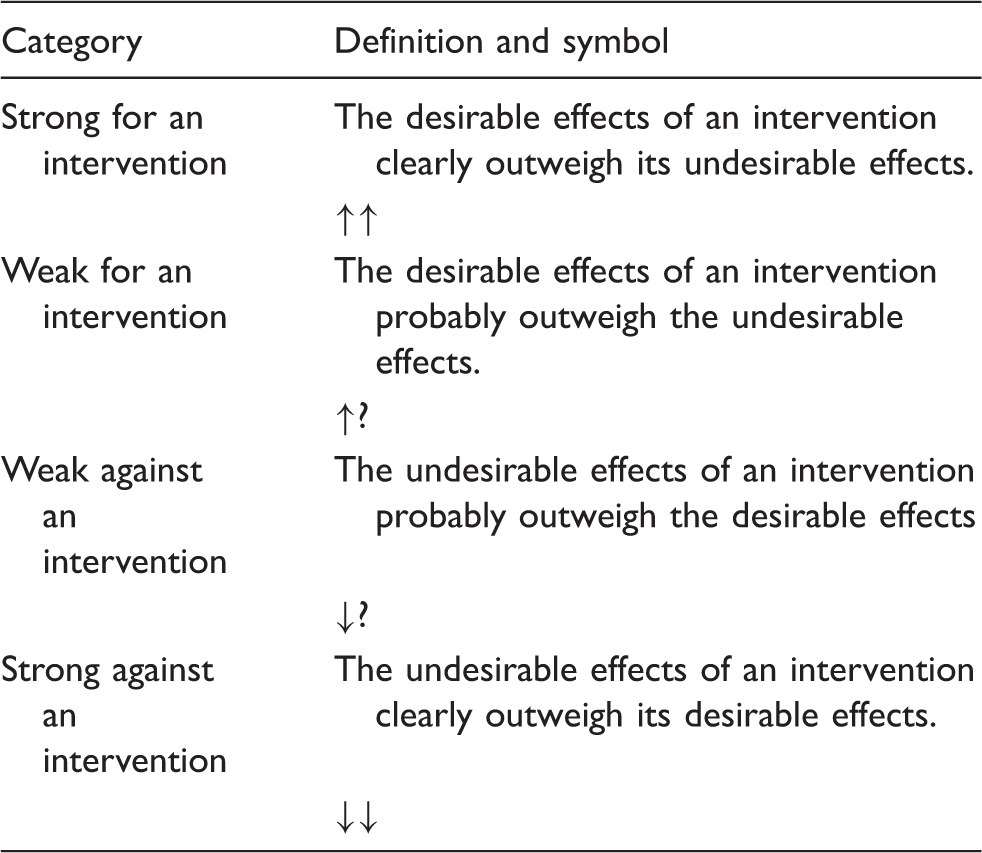
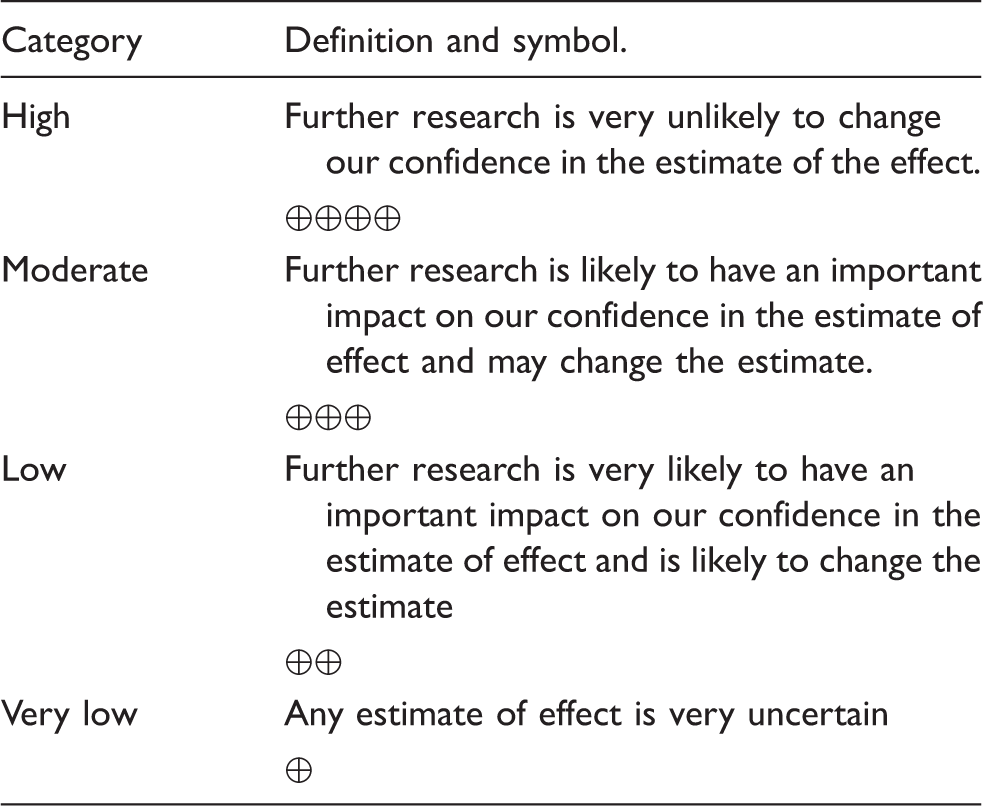
Quality of evidence was graded into high, moderate, low, and very low (for definitions, see Box 2). Strength of recommendation was assessed according to specific features and levels of strength were dichotomised to be either strong or weak (Box 3). Weak recommendations are also termed suggestions. Wording of treatment recommendations referred to published guidance.
34
The group generated ‘additional information’ mostly based on observational and epidemiological studies or common clinical practice which were not used for creation of the present recommendations34,36 but for proposals on how to manage the patients. This guideline document has been discussed during a plenary session during the ESO-Karolinska Stroke Update Conference in November 2016. It was approved by consensus by the members of the working group for the preparation of the ESO Guidelines about management of post-stroke seizures and epilepsy (online Appendix 1A). It was reviewed by two external reviewers (online Appendix 1D online), who did not carry any responsibility for its integrity. It was submitted to and approved for publication by the ESO Guidelines Committee (online Appendix 1B) and the ESO Executive Committee (online Appendix 1C). Grades of quality of evidence.
31
Results
Summary of recommendations and suggestions.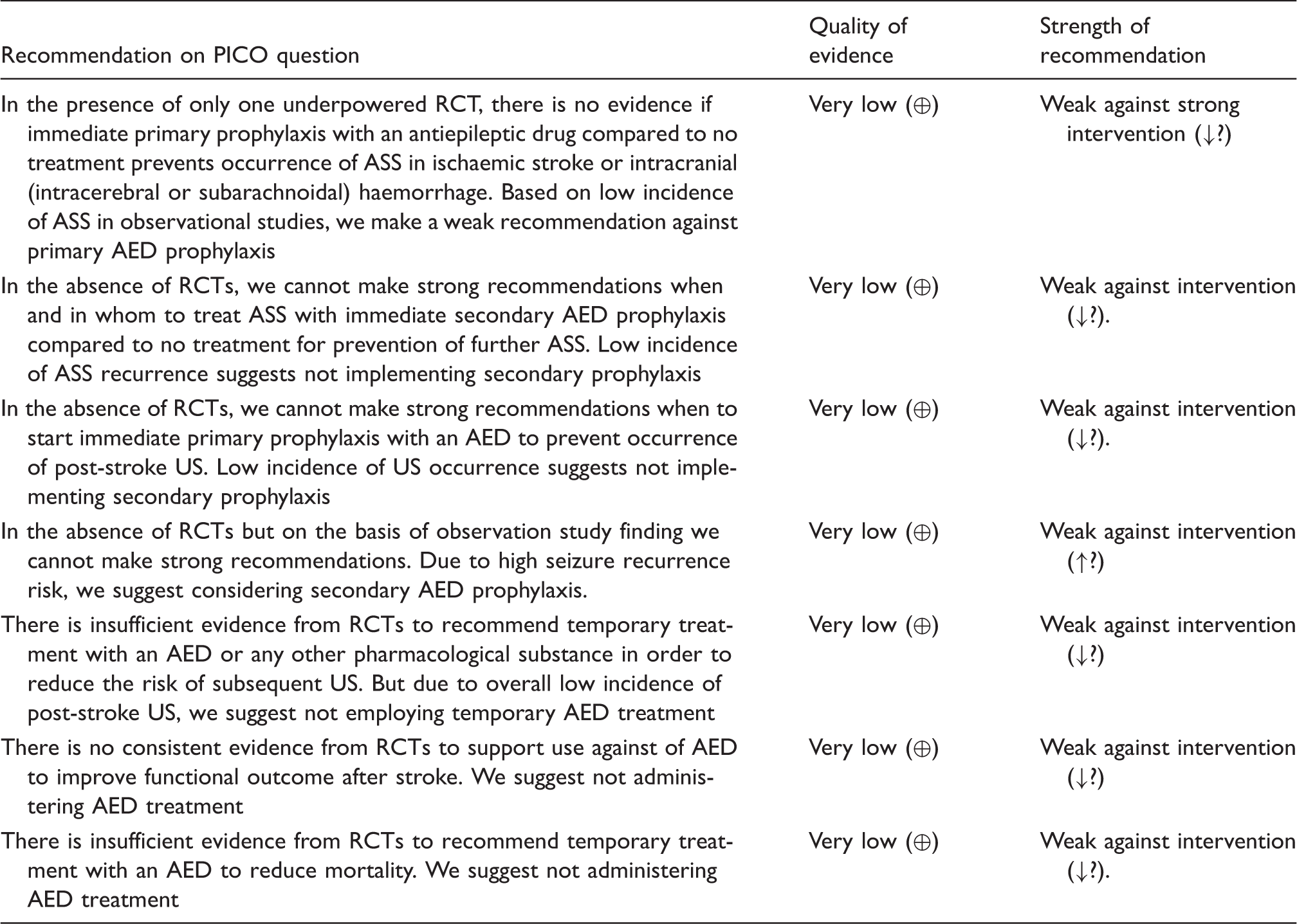
PICO: patient, intervention, comparator, outcome; RCT: randomised controlled trial; ASS: acute symptomatic seizure; AED: antiepileptic drug; US: unprovoked seizure.
Summary of findings table and GRADE evidence profile for PICO (patient, intervention, comparator, outcome) question 1.

Note: The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
CI: confidence interval; RR: risk ratio.
GRADE: Working Group grades of evidence
High quality: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect.
Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect.
Only one randomised controlled trial available.
The number of events and the sample size were very low.

CI: confidence interval; RR: risk ratio.
Only one randomised controlled trial available.
The number of events and the sample size were very low. p = 0.16.
No RCTs are available on the question if – in patients who have suffered at least one ASS – immediate secondary prophylaxis with an AED compared to no treatment prevents occurrence of further ASS. Risk of acute recurrence of ASS (within seven days of the same stroke) is 10–20%.25,38
No RCTs are available on prevention of unprovoked post-stroke seizures with vs. without sustained primary AED prophylaxis. Some observational studies focus on the incidence of US (10–12%, increasing with time after stroke) and risk factors for US (cortical involvement, ASS, severe stroke). Patients with intracerebral haemorrhage and cortical involvement, age <65 years, volume >10 ml and ASS had an US risk of 46.2%. 17 Patients with subarachnoid haemorrhage and accompanying intracerebral haemorrhage of more than 15 cm3 volume had an US risk of 33% after five years. 21 In 80 patients with malignant middle cerebral artery (MCA) infarction and craniectomy, US occurred in 44.8% after a median of seven months. Independent predictor for US was delayed surgery (> 42 h). There was no influence of primary AED prophylaxis on seizure occurrence. 40
No RCTs are available on prevention of further unprovoked post-stroke seizures after one index US with vs. without secondary AED prophylaxis. US recurrence risk is reported to be higher than 70% in 10 years. 7 Two RCTs compared efficacy of each two different AED after stroke. In these underpowered trials, seizure freedom rates after 12 months did not differ comparing levetiracetam and carbamazepine 41 and comparing lamotrigine and carbamazepine. 42
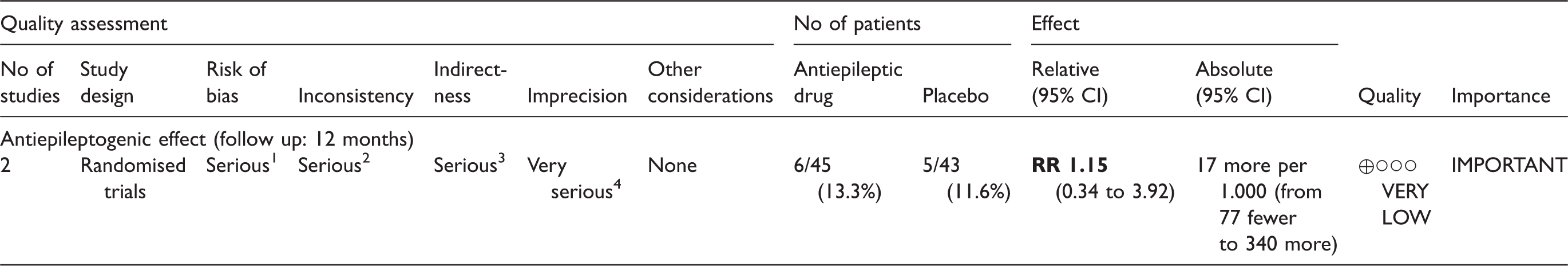
Summary of findings table and GRADE evidence profile for PICO (patient, intervention, comparator, outcome) question 5.
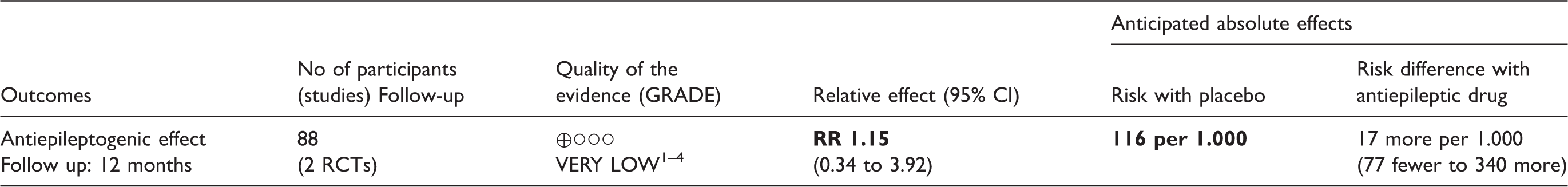
Note: The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
CI: confidence interval; RR: risk ratio.
GRADE Working Group grades of evidence
High quality: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect.
Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect.
Incomplete accounting of outcome events.
Unexplained heterogeneity.
Differences in intervention (different antiepileptic drugs).
The number of events and the sample size were very low.
CI: confidence interval; RR: risk ratio.
Incomplete accounting of outcome events.
Unexplained heterogeneity.
Differences in intervention (different antiepileptic drugs).
The number of events and the sample size were very low. p = 0.81.
Three RCTs have assessed functional outcome with temporary post-stroke AED treatment vs. placebo. One trial (with 849 patients) on diazepam administered for three days did not demonstrate increased rate of patients with independence (RS <3) at three months (the primary endpoint), considering all patients. 47 More patients with independence were only demonstrated in the subgroup with cardioembolic infarction, but the trial was not powered for this analysis. Patients with intracerebral haemorrhage had significantly more often pneumonia with diazepam compared to those in the placebo group. The second trial (with 72 patients) demonstrated that valproate compared to placebo for 1 month after intracerebral haemorrhage was associated with significantly lower NIHSS at 12 months. 37 The third trial compared levetiracetam administered for 12 weeks post-stroke to placebo, but was stopped due to poor recruitment of 16 patients, only. 46 As the available trials use different outcome scales and do not define cut-offs allowing for dichotomised analysis of data, risk ratios cannot be calculated. There are no trials on the effect of continuous post-stroke AED treatment vs. placebo on functional outcome. All other available studies focus on the question if ASS or US are independent risk factors for unfavourable functional outcome, but results are ambiguous.
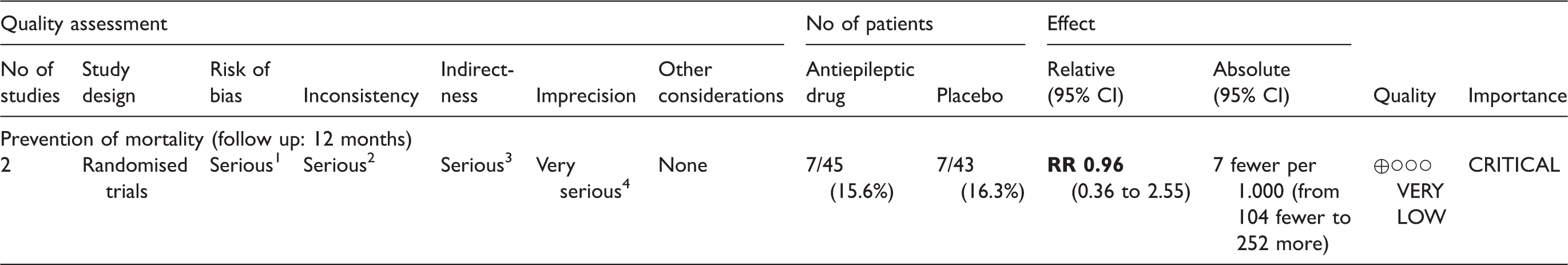
Summary of findings table and GRADE evidence profile for PICO (patient, intervention, comparator, outcome) question 7.
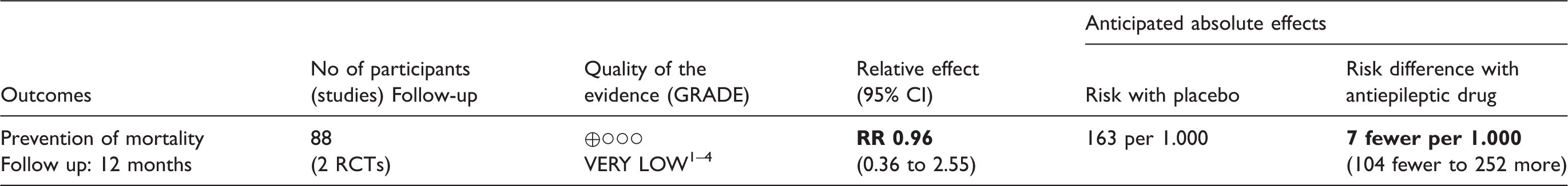
Note: The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
CI: confidence interval; RR: risk ratio.
GRADE Working Group grades of evidence
High quality: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect.
Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect.
Incomplete accounting of outcome events.
Unexplained heterogeneity.
Differences in intervention (different antiepileptic drugs).
The number of events and the sample size were very low.
CI: confidence interval; RR: risk ratio.
Incomplete accounting of outcome events.
Unexplained heterogeneity.
Differences in intervention (different antiepileptic drugs).
The number of events and the sample size were very low. p = 0.93.
Discussion
The very low quality of evidence of the few RCTs assessing interventions for post-stroke seizures and epilepsy does not allow making strong recommendations. But based on some reliable observational studies, we were able to make some weak recommendations and suggest on how to prevent occurrence and recurrence of post-stroke ASS and US as additional information. This offers at least some guidance for the treating physicians.
The only weak recommendation for consideration of AED treatment was made for secondary prophylaxis after one post-stroke US (PICO4). RCTs assessing AED treatment vs. no treatment are lacking, but observational studies have shown seizure recurrence rates within the next 10 years to be higher than 70%. 7 In a randomised open-label but underpowered study, levetiracetam and sustained-release carbamazepine were compared in patients with post-stroke US. 41 Though the excellent efficacy of the two compounds was not different, subjective tolerability and results of neuropsychological tests were significantly better in the levetiracetam group. In epileptology, underlying aetiology of focal epilepsy usually does not influence the choice of the AED. The decision on the suitable substance has to be individualised considering patients’ age, sex, comorbidities, and comedication. The question of AED withdrawal at some time point is difficult to address, this decision as well needs to be tailored for every single patient.
For all other six PICO questions, we suggest not to administer AED treatment. These comprise primary and secondary prophylaxis to prevent ASS occurrence or recurrence (PICO1 & 2), continuous primary prophylaxis for US (PICO3), temporary treatment to prevent or mitigate epileptogenesis (PICO5), and continuous or temporary treatment to improve functional outcome (PICO6) or to prevent mortality (PICO7).
In patients with ischaemic stroke and intracranial haemorrhage, the very low evidence from the only RCT does not allow making any strong recommendation for or against primary AED prophylaxis for ASS (PICO1). As incidence of ASS proves to be very low, we suggest not administering primary prophylaxis in all patients. In some subgroups, e.g. in those with intracerebral haemorrhage with cortical involvement, one out of three patients develops ASS. 10 In these cases, clinicians may decide individually to temporarily administer primary AED prophylaxis (we suggest for not longer than the acute phase). There are no studies on which AED should be preferred. The need for rapid titration of the drug within one day and – where appropriate – for intravenous administration limits the choice of monotherapy-licensed AED to lacosamide, levetiracetam, phenytoin, and valproate. However, the latter may be suboptimal in patients with haemorrhages due to the coagulation-inhibiting properties of valproate 50 though findings are conflicting. 51
No RCTs are available on secondary AED prophylaxis after one index ASS, but observational data indicate acute recurrence rates of less than 20%.25,38 Therefore, we suggest not administering secondary AED prophylaxis. Interestingly, administration of an AED to prevent further seizures in the acute phase of stroke does not seem to be uncommon in clinical practise. But as with primary post-stroke AED prophylaxis against occurrence of ASS, there is at least no need for long-term treatment as risk for later occurrence of US is rather low. 7 We suggest terminating secondary prophylaxis after the acute phase.
There seems to be no general need to administer primary AED prophylaxis to prevent US (PICO3). Some clinical conditions bear a significantly increased risk for US including subarachnoid haemorrhage with accompanying intracerebral haemorrhage of more than 15 cm3 volume 21 and intracerebral haemorrhage with cortical involvement, age <65 years, volume >10 ml, and prior ASS. 17 These may allow for administering AED on a long-term basis following a primary prophylactic approach.
So far, the concept of antiepileptogenic treatment approaches is just theoretical. No clinical trial has ever demonstrated that temporary AED treatment after any acquired brain injury including stroke prevented or mitigated epilepsy. 52 A Chinese group has published the protocol of a randomised controlled trial on patients with intracerebral haemorrhage which are administered valproate of placebo for seven days post-stroke; seizure outcome will be assessed after one year. 53 Results of this trial are pending. Animal models of post-stroke epilepsy or other acquired brain lesions also have never demonstrated antiepileptogenic effects, and we need a better understanding of the pathophysiological mechanisms underlying epileptogenesis in order to identify targets for antiepileptogenic treatment approaches. 54 Therefore, we suggest not administering AEDs (PICO5).
Available data from RCTs on the question if post-stroke AED treatment has a favourable impact on functional outcome are controversial.37,46,47 In this context, we suggest not administering AED treatment (PICO6). Some observational studies show that ASS and/or US are associated with unfavourable functional outcome, but up to now, it remains open if seizures are just a marker for poor outcome. Two non-randomised studies suggest that phenytoin as compared to levetiracetam is associated with unfavourable functional outcome but evidence is low.48,49 Due to its rather poor tolerability profile and the narrow therapeutic window, phenytoin nowadays generally is not a first-choice AED in focal epilepsy.
Two RCTs have addressed the question if temporary AED treatment prevents or reduces mortality37,47(PICO7). In both trials, accordant findings were negative but overall quality of evidence was very low. Therefore, we cannot make a strong recommendation, but suggest not administering AEDs.
The strengths of this guideline include its systematic approach to literature search and guidance by the GRADE recommendations and the ESO SOP. The limitations of our approach reflect the extreme paucity of RCTs on prevention and pharmacological management of acute symptomatic and unprovoked post-stroke seizures. At the same time, of course, this could be also considered as strength of this Guideline Document given that it highlights the areas which need further research. The working group was rather small consisting of six neurologists, but the risk of too strong influences by single members of the group was countered by extensive internal and external review processes. As suggested in the ESO guideline SOP, future updates of these Guidelines may consider physicians with specialities beyond neurology (e.g. emergency physicians), other occupational groups (e.g. nurses), and representatives of patients’ associations as members of the working group. 31
In summary, current evidence for management of post-stroke seizures and epilepsy is very low. There is an urgent need for RCTs addressing this clinically relevant complication of stroke to hopefully improve outcome and quality of life in affected patients.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to authorship, and/or publication of this article: MH received speaker’s honoraria and/or consultancy fees from Cyberonics, Desitin, Eisai, GlaxoSmithKline, Janssen-Cilag, Novartis, UCB Pharma and Viropharma. EB received grants from the Italian Ministry of Health, Italian Drug Agency, UCB Pharma, EISAI, Shire, American ALS Association, Borgonovo, Foundation, consultancy fees from Viropharma. RK received Speaker’s honoraria and/or consultancy fees from Eisai, Fennomedical, GW Pharmaceuticals, Orion, Sage, Sandoz and UCB Pharma. The other authors declare that there is no conflict of interest.
Funding
This guideline development received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
MH.
Contributorship
Every step in the development and writing of these guidelines was a joint work of all co-authors.
Acknowledgements
None.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.