Abstract
Background and aims
The vision of autistic people has been characterised as focused on detail, with a disinclination (or reduced ability) to integrate information into coherent ‘wholes’. In contrast to this view, we recently demonstrated enhanced integration of visual motion signals in autistic children compared to typically developing children. Here, we aimed to investigate the robustness of our finding of increased motion integration in autism with a new sample of children and to determine whether increased integration in autistic children would extend to a static, orientation judgement.
Methods
We presented motion and orientation equivalent noise and coherence tasks to 46 autistic children aged 6 to 14 years and 45 typically developing children matched in age and non-verbal IQ. The equivalent noise tasks consisted of two interleaved conditions: a high-noise condition in which children judged the average direction or orientation of elements whose range of direction or orientations was manipulated, and a no-noise condition in which children judged the direction or orientation of elements sharing the same direction or orientation. Equivalent noise modelling provided estimates of internal noise (the precision with which children can estimate the orientation/direction of one element) and global sampling (how many elements children are effectively using to judge the overall orientation/direction). Children also completed coherence tasks in which the proportion of signal elements sharing the same direction or orientation amidst otherwise random noise elements was manipulated. We assessed group differences using a combination of frequentist and Bayesian statistical approaches.
Results
Analysis of the data in this new sample alone did not provide sufficient evidence either in favour or against the hypothesis of increased integration in autism. However, when combining motion data from this and the original experiment, autistic children exhibited superior integration of direction information in the high-noise condition compared to typically developing children, with similar no-noise and coherence thresholds. Equivalent noise modelling of these data revealed increased sampling in autistic children for motion information but no conclusive evidence for atypical levels of internal noise. There was no evidence of differences between autistic and typically developing children in the orientation equivalent noise and coherence tasks.
Conclusions
Overall, autistic children effectively integrated more direction information than typically developing children. However, the groups overlapped considerably and there was substantial individual variability, so that the effect may be difficult to detect in small groups. There was no indication of atypical integration of orientation in the current study, although larger samples will be required in order to provide conclusive evidence.
Implications
These results help characterise the nature of sensory processing in autism, which is of high import and relevance given the recent inclusion of sensory symptoms in diagnostic criteria. Increased integration of motion information could potentially lead to feelings of sensory overload in autistic children. If such increased integration is specific to motion information, domain-specific accounts of autistic perception will be required.
Introduction
Alongside its effects on social communication and interaction, autism is also characterised by atypical sensory processing (Diagnostic and Statistical Manual [DSM-5], American Psychiatric Association [APA], 2013). In terms of vision, atypical sensory processing can manifest in a variety of ways, including aversion to bright, fluorescent or flickering lights, and/or unusually intense looking at objects or people, including moving objects like fans (Bogdašina, 2003; Leekam, Nieto, Libby, Wing, & Gould, 2007; see also Simmons et al., 2009, for review). Theoretical accounts have suggested that autistic individuals may have either enhanced local processing (of parts of objects; Mottron & Burack, 2001; Mottron, Dawson, Soulières, Hubert, & Burack, 2006), and/or weaker global processing (i.e. poorer combinations of parts into wholes; Frith & Happé, 1994; Happé & Frith, 2006), meaning that they may tend to focus on the details of a visual scene rather than the whole. For example, autistic individuals may focus on individual fish moving within a shoal but be less inclined or able to perceive overall movement of the shoal.
Contrary to this view of compromised visual integration in autism, we recently reported enhanced integration of direction information in autistic children (n = 33) compared to typically developing children (n = 33; Manning, Tibber, Charman, Dakin, & Pellicano, 2015). These findings were obtained using an equivalent noise task (Dakin, 2001; Dakin, Mareschal, & Bex, 2005; Tibber et al., 2015; Tibber, Kelly, Jansari, Dakin, & Shepherd, 2014), in which participants were asked to determine the average direction of moving dots whilst the directional variability (or ‘external noise’) was manipulated. Such tasks probe so-called ‘ensemble’ coding: an observer’s ability to judge the overall or statistical properties of images (see Dakin, 2015, for a recent review). Equivalent noise paradigms are unique in that they allow the quantification of local and global limits on visual integration (or ensemble coding). Specifically, equivalent noise modelling estimates both the imprecision with which individual elements are processed (local internal noise), and the effective number of elements which are averaged over (global sampling). Importantly, these equivalent noise tasks do not require observers to segregate signal from noise, in contrast to more commonly used ‘motion coherence’ tasks, which require observers to report the direction of a set of coherently moving signal dots amidst randomly-moving noise dots (Newsome & Paré, 1988). Reduced sensitivity has previously been reported in autistic individuals in motion coherence tasks (e.g. Milne et al., 2002; Pellicano, Gibson, Maybery, Durkin, & Badcock, 2005), but not consistently (e.g. Jones et al., 2011; Manning, Charman, & Pellicano, 2015).
In our equivalent noise task, we found that autistic children could reliably estimate the average direction of moving dots over a greater range of directions (external noise) than typically developing children for both slow- (1.5°/s) and fast- (6°/s) moving stimuli (Manning, Tibber, et al., 2015). Yet, they showed comparable discrimination thresholds when the dots all moved in the same direction (i.e. no external noise). Equivalent noise modelling of these data suggested that autistic children sampled over more elements and had similar levels of internal noise compared to typically developing children. Interestingly, the autistic children had similar motion coherence thresholds as typically developing individuals. The fact that the autistic children did not have improved motion coherence thresholds despite their superior direction integration in the equivalent noise task suggests that their performance with motion coherence stimuli is limited not by integration but by difficulty in segregating signal from noise. Similarly, a difficulty segregating signal from noise has been proposed as an explanation of why migraine sufferers have elevated motion coherence thresholds yet no performance deficits in equivalent noise tasks (Tibber et al., 2014). We therefore suggested that reduced segregation of signal from noise could explain previous reports of elevated motion coherence thresholds in autism (e.g. Milne et al., 2002; Pellicano et al., 2005).
This study had two aims. First, it aimed to test the robustness of our finding of increased direction integration in autism by presenting the same equivalent noise task to a new sample of autistic and typically developing children. We considered this replication effort important given how inconsistent findings in the literature on visual processing in autism (e.g. motion coherence, see Simmons et al., 2009 for review), and in the field of psychology more generally (Open Science Collaboration, 2015) can be.
Second, the study aimed to assess whether the increased integration reported for motion direction extends to other tasks. To this end, participants were also presented with a static equivalent noise task in which they were required to judge the overall orientation (or tilt) of elements (Dakin, 2001; Tibber et al., 2015). Understanding whether increased integration is domain-specific or domain-general will help to refine theories of autistic perception and clarify their relationship with underlying physiology. Moreover, cross-task comparisons may help to characterise the profile of visual processing atypicalities in different disorders, and as a result, help determine whether reported perceptual abnormalities reflect specific etiological processes or general markers of vulnerability in the system. For example, Tibber et al. (2015) recently presented motion, orientation and size equivalent noise tasks to schizophrenic patients and reported reduced integration specifically for orientation tasks, but not motion and size. Previous studies in autism have suggested that orientation discrimination is either indistinguishable (Freyberg, Robertson, & Baron-Cohen, 2016; Schwarzkopf, Anderson, de Haas, White, & Rees, 2014; Shafai, Armstrong, Iarocci, & Oruc, 2015) or enhanced (Dickinson, Bruyns-Haylett, Smith, Jones, & Milne, 2016) in autistic adults compared to neurotypical individuals, with enhancements restricted to discriminations made from oblique rather than vertical reference stimuli (Dickinson et al., 2016). Yet, these studies have all measured the ability to discriminate between two stimuli of different orientations, rather than assessing the ability to integrate orientation information over space.
In this study, autistic children and typically developing children were presented with closely-matched equivalent noise tasks to quantify their integration of both direction and orientation information. In addition, children completed coherence tasks for both direction and orientation information. Together these tasks allowed for comparison of performance in ‘pure’ integration tasks with those that involve segregation of signal from noise. Following our previous results (Manning, Tibber, et al., 2015), we hypothesised that autistic children would show enhanced integration for direction information, yet typical performance both in a direction discrimination task with no external noise and a motion coherence task, with these findings possibly extending to orientation information. The use of equivalent noise tasks provided a further test of accounts of increased (Simmons et al., 2009) and reduced (Davis & Plaisted-Grant, 2015) levels of internal noise in autism. Based on our previous results, we predicted no differences in levels of internal noise between the autistic children and typically developing children.
Methods
Participants
A power analysis was conducted using G*Power 3 software (Faul, Erdfelder, Lang, & Buchner, 2007) in order to predict the sample size needed to detect group differences a priori. The effect size corresponding to the group means of maximum tolerable noise for the slow condition (1.5°/s) reported by and Manning, Tibber, et al. (2015) (autism: M = 1.65; SD = .13; typically developing: M = 1.49, SD = .23, in log units) was d = .85. Based on this effect size and a conservative, two-tailed test with 95% power and an alpha level of .05, it was deemed that at least 38 participants per group were required to obtain sufficient power to detect group differences.
Forty-six autistic children (three female) and forty-five typically developing children (18 female) completed both direction-processing and orientation-processing tasks in this study. The autistic children all had a clinical diagnosis of an autism spectrum condition, and typically developing children had no (parent-reported) diagnoses of developmental conditions. Parents completed the Social Communication Questionnaire (SCQ; Rutter, Bailey, & Lord, 2003) and autistic children were administered Module 3 or 4 of the Autism Diagnostic Observation Schedule (ADOS-2; Lord et al., 2012). We used calibrated severity scores from the ADOS to ensure comparability across modules (Lord et al., 2012), using the revised algorithm for Module 4 (Hus & Lord, 2014). All autistic children scored above threshold for an autism spectrum condition on one or both measures, and no typically developing children scored above the threshold of 15 on the SCQ (Rutter et al., 2003). All children were cognitively able (IQ ≥ 70), as assessed by the Wechsler Abbreviated Scales of Intelligence, Second Edition (WASI-II; Wechsler, 2011). Normal or corrected-to-normal vision was confirmed using a Snellen acuity chart. ANOVAs with group (autism, typically developing) and study (original, replication) as between-participants factors confirmed that this group of participants were of comparable age, non-verbal and verbal ability as the 33 autistic and 33 typically developing children tested by Manning, Tibber, et al. (2015) [ps ≥ .27 and ηp2 < .01 for all main effects of study and group by study interactions]. Characteristics of participants in the original study are provided in Manning, Tibber, et al. (2015).
Participant characteristics.

VIQ: Verbal IQ; PIQ: Performance IQ; FSIQ: Full-scale IQ. IQ scores were assessed using the Wechsler Abbreviated Scales of Intelligence (WASI-II; Wechsler, 2011). SCQ: Social Communication Questionnaire (Rutter et al., 2003); ADOS: Autism Diagnostic Observation Schedule-2 (Lord et al., 2012); Conners 3AI: Conners 3 ADHD Index (Conners, 2008); SP: Short Sensory Profile-2 (Dunn, 2014).
Note: Data are presented as mean (SD) range.
One autistic child was excluded from the motion-processing analysis and four autistic and two typically developing children were excluded from the orientation-processing analysis because they did not perform significantly above chance in the catch trials in the equivalent noise and/or coherence task (see ‘Data screening and transformation’). These exclusions resulted in 45 autistic and 45 typically developing children in the motion dataset, and 42 autistic and 43 typically developing children in the orientation dataset. These groups did not differ significantly in age [motion: t(88) = 1.70, p = .09, d = 0.36; orientation: t(83) = 1.59, p = .12, d = 0.35] or non-verbal ability [motion: t(88) = 1.22, p = .23, d = 0.26; orientation: t(83) = .96, p = .34, d = 0.21], although the autistic children had lower verbal abilities than the typically developing children overall [motion: t(88) = 1.97, p = .05, d = 0.42; orientation: t(83) = 2.12, p = .04, d = 0.47], consistent with their clinical profile.
Apparatus and stimuli
Experiments were run on a Dell Precision M3800 laptop (2048 × 1152 pixels, 60 Hz) using MATLAB (MathWorks, Cambridge, MA, USA) and elements of the Psychophysics Toolbox (Brainard, 1997; Kleiner, Brainard, & Pelli, 2007; Pelli, 1997). A yellow-bordered hard-edged circular aperture (15° diameter) and an anchor-shaped fixation point (0.57° × 0.57°) were presented centrally on a grey screen (Figure 1). Two smaller yellow-bordered circular apertures (6.12° diameter) were presented to the left and right of the aperture and contained red and green images, respectively, that served as reference points. Stimuli in the motion tasks were 100 white dots (0.44° diameter) displaced by 0.075° every three frames, yielding a dot speed of 1.5°/s. Stimuli in the orientation tasks were 100 static Gabor patches with a spatial frequency of 3.4 c/°, presented with random phases and at 50% contrast, each presented within a circular hard-edged aperture (diameter 0.44°). All stimuli were presented for 400 ms.
Schematic representation of stimuli presented in the motion and orientation tasks. Arrows represent the direction of motion in the direction tasks. The upper panel represents trials from the high-noise condition in the equivalent noise tasks, with the direction and orientation of elements taken from Gaussian distributions with a standard deviation of 10°. The lower panel represents trials from the coherence tasks with 40% coherence in the direction and orientation of elements. The coherent (‘signal’) elements are outlined in red for illustration purposes only.
General procedure
Outline of tasks and measures.

Performance measures obtained directly from tasks.
Measures estimated from equivalent noise modelling of performance measures.
Motion tasks
The motion tasks were those used by Manning, Dakin, Tibber, and Pellicano (2014), and Manning, Tibber, et al. (2015). Whereas slow (1.5°/s) and fast (6°/s) conditions were presented in Manning, Tibber, et al. (2015), the fast condition was omitted here because, a) it was necessary to reduce the testing time in order to also present orientation tasks and b) we found no interacting effects between group and speed on performance in these tasks (Manning, Tibber, et al., 2015). The equivalent noise task consisted of both no-noise and high-noise conditions. In the no-noise condition, the standard deviation of directions was fixed at 0° whilst the mean directional offset was varied. In the high-noise condition, the mean directional offset was fixed at ±45° whilst the standard deviation of directions was varied. In the motion coherence task, the proportion of ‘signal’ dots moving in a coherent direction of ±90° was manipulated, whilst the remainder of dots (‘noise’ dots) moved in random directions. Children were told to judge whether the ‘fish’ swam overall to the red (left) or green (right) reef or rocks in the equivalent noise and motion coherence tasks, respectively. Further details of these tasks have been reported previously (Manning, Tibber, et al., 2015; Manning et al., 2014).
Orientation tasks
The orientation equivalent noise task was modified from that used by Tibber et al. (2015) and presented within a ‘game’ in order to maximise children’s engagement. In the no-noise condition, the standard deviation of orientations was fixed at 0° whilst the mean orientation was varied. In the high-noise condition, the mean orientation was fixed at ±22.5° whilst the standard deviation of orientations was varied. A new orientation coherence task was created to provide a counterpart to the motion coherence task. Here, a proportion of elements were designated to be ‘signal’ elements, with a coherent orientation of ±45°, whilst the remainder were ‘noise’ elements with random orientations. The proportion of signal elements was manipulated throughout the task. The children were told to work out whether the ‘jellyfish with stripes on their back’ overall faced the red (left) or green (red) camera or islands, in the equivalent noise and coherence tasks, respectively.
Task phases
All tasks were introduced to children as ‘games’ using under-the-sea themed animations. Children first completed a demonstration and criterion phase (‘level 1’), in which they were presented with four demonstration trials and were then required to respond correctly to four consecutive trials within 20 trials in order to progress to the next phase. These criterion trials were designed to be ‘easy’, with no external noise (SD = 0°) and a mean direction of ± 45° or a mean orientation of ±22.5° in the motion and orientation equivalent noise tasks, respectively, and 100% coherence in the motion and orientation coherence tasks. Visual and verbal feedback was provided. Next, children completed a practice phase (‘level 2’), in which they completed eight trials of increasing difficulty. Visual and verbal feedback was provided, although there was no criterion for proceeding to the next phase.
Finally, children completed a threshold estimation phase (‘level 3’) in which the stimuli presented were controlled by QUEST (Watson & Pelli, 1983). In the no-noise conditions, QUEST controlled the angular offset of the direction or orientation of elements, and in the high-noise conditions, QUEST controlled the standard deviation of the direction or orientation of elements to find the maximum tolerable noise for completing the discrimination. The no-noise and high-noise conditions each consisted of 75 trials which were randomly interleaved in the equivalent noise tasks. In the coherence tasks, QUEST controlled the percentage of signal elements across 75 trials. An additional 15 catch trials were randomly interleaved into each task. These trials presented the same ‘easy’ stimuli presented in the criterion trials. This yielded a total of 165 trials in each equivalent noise task and 90 trials in each coherence task, for both motion and orientation stimuli. No feedback was given during this phase, although general encouragement was presented throughout. To aid motivation, children were told that they were competing against computer characters. In each task, trials were divided into four blocks, after which participants were shown a simulated graph of the ‘points’ they and their ‘opponent’ had attained. These values were randomly jittered around a fixed set of values, ensuring that participants always received slightly more points than their opponent. Thresholds were computed as the stimulus intensities (angular offset; standard deviation; or percentage coherence) leading to 84% correct performance. We refer to these thresholds as no-noise, maximum tolerable noise and coherence thresholds (Table 2).
Equivalent noise analysis
Equivalent noise analysis provides estimates of two independent limits on integration performance: internal noise (σ2int), corresponding to the imprecision with which individual elements are processed, and sampling (nsamp), which is the effective number of elements that are averaged. These parameters are estimated by relating discrimination thresholds (σ2obs) to the external noise (σ2ext) in the stimulus, which is manipulated in our tasks by varying the standard deviation of the distribution from which the direction or orientation of elements are drawn (Figure 2):
Equivalent noise function constrained by data from no-noise (blue) and high-noise (red) conditions. The mean direction or orientation offset (left or right of vertical) that is needed to discriminate at a certain level of accuracy (i.e. threshold) increases as a function of the standard deviation of the distribution of directions or orientations from which the elements are drawn (i.e. external noise). Thresholds are relatively unaffected by low levels of external noise where instead, internal noise dominates. However, as external noise is increased further, the internal noise is swamped and thresholds start to increase. In the no-noise condition, the standard deviation is fixed at 0° in order to determine the finest mean offset that can be discriminated in the absence of external noise. In the high-noise condition, the mean offset is fixed at ± 45° in the motion task and ±22.5° in the orientation task, and the standard deviation is varied to find the maximum level of tolerable noise. Increased sampling shifts the entire function downwards, yielding improved discrimination performance at all levels of external noise. Increased levels of internal noise, however, lead to higher thresholds at low levels of external noise, and a rightwards shift of the elbow of the function, so that more external noise is required before thresholds begin to rise.

Here, we used the efficient version of the equivalent noise model used previously with children and clinical populations (Manning, Tibber, et al., 2015; Tibber et al., 2014, 2015), which constrains the fit of the equivalent noise function using two highly informative points on the function with orthogonal confidence intervals. These points correspond to the no-noise and high-noise conditions of the motion and orientation tasks (Figure 2). Sampling (nsamp) was estimated by transforming the maximum level of tolerable noise (MTN) in the high-noise condition as follows:
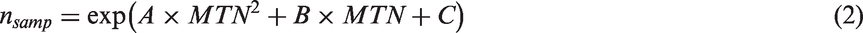

Data screening and transformation
The data were screened and transformed as described by Manning, Tibber, et al. (2015). Participants who did not perform significantly above chance in the catch trials in the equivalent noise and/or coherence tasks were removed from the analysis (see ‘Participants’). Two additional autistic children had orientation coherence thresholds estimated to be above 100% and one autistic child had a motion coherence threshold estimate above 100%, indicating an inability to perform the task. These participants were removed from the analysis of the coherence task but retained in the equivalent noise analysis, following Manning, Tibber, et al. (2015). All threshold measures and internal noise and sampling estimates were log-transformed to minimise the effects of skewness and kurtosis. Measures were screened for outliers, defined as data points lying more than 3 SD above or below the group mean. An outlier was identified in each of the motion maximum tolerable noise and coherence values, belonging to two typically developing children. Outliers were identified in the orientation no-noise threshold, maximum tolerable noise and internal noise values, belonging to one autistic child. Outliers were replaced with values corresponding to ±2.5 SDs from the group mean (Tabachnick & Fidell, 2007).
Results
Motion processing
First we assessed differences in performance between autistic and typically developing children in the motion tasks in our new sample. Based on the findings of Manning, Tibber et al., (2015) we did not predict group differences in no-noise thresholds or motion coherence thresholds. Accordingly, in our new sample, there were no significant group differences in either no-noise thresholds, t(72.04) = .54, p = .59, d = 0.11 (autism: M = .84; SD = .20; typical: M = .81; SD = .33, in log units), or coherence thresholds, t(87) = .47, p = .64, d = 0.10 (autism: M = −.41; SD = .17; typical: M = −.43; SD = .15, in log units). However, we did predict group differences in maximum tolerable noise values. Whilst the autistic group had higher maximum tolerable noise levels than the typically developing children overall (autism: M = 1.63; SD = .19; typical: M = 1.56; SD = .24, in log units; see Figure 3), this was not statistically significant, t(88) = 1.47, p = .15, d = 0.31, in contrast to our previous study. Next, we assessed the parameters obtained from equivalent noise modelling. The autistic children did not differ significantly from the typically developing children in either internal noise, t(88) = 1.46, p = .15, d = 0.31 (autism: M = .86; SD = .23; typical: M = .78; SD = .29, in log units), or sampling levels, t(88) = 1.30, p = .20, d = 0.28 (autism: M = .04; SD = .37; typical: M = −.06; SD = .35, in log units).
Individual threshold values (upper panel) and equivalent noise modelling estimates (lower panel) in the direction tasks for autistic (blue) and typically developing (purple) children in the original study by Manning, Tibber, et al. (2015) (circles) and in the current replication study (crosses). N.b. data are shown with outliers replaced (Tabachnick & Fidell, 2007).
To allow comparison between the data obtained by Manning, Tibber, et al. (2015) with those collected in the current study, we conducted ANOVAs with two between-participants factors: group (autistic, typically developing) and study (original, replication; see Figure 3). There was no significant effect of group on no-noise thresholds, F(1,152) = .03, p = .86, ηp2 < .01, motion coherence thresholds, F(1,150) = .02, p = .90, ηp2 < .01, or internal noise values, F(1,152) = 3.16, p = .08, ηp2 = .02. For these measures, there were also no significant effects of study [no-noise thresholds: F(1,152) = 3.04, p = .08, ηp2 = .02; motion coherence thresholds: F(1,150) = .01, p = .95, ηp2 < .01; internal noise: F(1,152) = 1.00, p = .32, ηp2 = .01] nor group by study interactions [no-noise thresholds: F(1,152) = .88, p = .35, ηp2 = .01; motion coherence thresholds: F(1,150) = .58, p = .45, ηp2 < .01; internal noise: F(1,152) = .05, p = .83, ηp2 < .01].
However, we were particularly interested in the maximum tolerable noise and sampling values, given the apparently discrepant results between the current sample and the sample used in the original study by Manning, Tibber, et al. (2015). We found a main effect of group on maximum tolerable noise values, with the autistic children having higher maximum tolerable noise thresholds than typically developing children, F(1,152) = 11.71, p = .001, ηp2 = .07, with no interaction between group and study, F(1,152) = 1.97, p = .16, ηp2 = .01. Additionally, there was no main effect of study, F(1,152) = .55, p = .46, ηp2 < .01. The autistic children also had significantly higher levels of sampling than the typically developing children overall, F(1,152) = 8.66, p = .004, ηp2 = .05, and this effect did not interact with study, F(1,152) = 1.20, p = .28, ηp2 = .01. There was also no main effect of study on sampling estimates, F(1,152) = 1.34, p = .25, ηp2 = .01. The lack of interaction effects suggests that increased maximum tolerable noise and sampling in the autistic children is not restricted to the original study, with the difference in the size of the between-group effect across studies not being statistically significant.
Orientation processing
In addition to providing a replication of our previous study assessing direction integration, we also assessed whether these findings generalised to orientation integration (Figure 4). The autistic children showed no significant differences in performance compared to typically developing children: in no-noise thresholds, t(83) = .17, p = .86, d = 0.04 (autism: M = .48, SD = .33; typical: .48, SD = .22, in log units), maximum tolerable noise, t(83) = 1.40, p = .17, d = 0.31 (autism: M = 1.30, SD = .32; typical: M = 1.38, SD = .13, in log units) and coherence thresholds, t(82) = .28, p = .78, d = 0.06 (autism: M = − .26, SD = .14; typical: M = −.27, SD = .13, in log units). Likewise, the groups did not differ in the resulting equivalent noise modelling parameters, with no significant group differences in either internal noise, t(83) = .37, p = .72, d = 0.08 (autism: M = .53, SD = .27; typical: M = .54, SD = .19, in log units) or sampling, t(83) = 1.01, p = .32, d = .22 (autism M = .08, SD = .32; typical: M = .15, SD = .30, in log units).
Individual threshold values and equivalent noise parameters in the orientation tasks for autistic (blue) and typically developing (purple) children. N.b. data are shown with outliers replaced (Tabachnick & Fidell, 2007).
Bayesian hypothesis-testing
Our use of frequentist statistics (null hypothesis significance testing), suggests a failure to reject the null hypothesis of no difference between autistic and typically developing children in any orientation measure, but suggests that autistic children have higher levels of maximum tolerable noise and sampling than typically developing children in the motion tasks when combined across studies. Two outstanding issues remain. First, we need to solve the discrepancy whereby there was a main effect of group and no group by study interaction in maximum tolerable noise values and sampling in the motion task, yet no group difference when analysing the replication sample in isolation. Second, we need to consider the possibility that the lack of significant group differences in the orientation task could be attributed to the fact that the sample is too small to detect effects in this task.
These issues arise in frequentist statistical approaches because the use of the p-value does not allow the relative evidence for the null hypothesis (that there are no group differences) and the alternative hypothesis (that the groups differ) to be quantified (Wagenmakers, 2007). Rather than simply stating that we failed to reject the null hypothesis in the motion task in the replication sample using the conventional cut-off of p < .05, it would be more informative to quantify the relative evidence for the null and alternative hypotheses within this sample. Bayesian statistics allow us to do this, whilst using the previously collected data (Manning, Tibber, et al., 2015) to inform the prior distribution, and hence assessing whether we should update our relative belief in favour of the null or alternative hypothesis in light of the newly collected data. Furthermore, the use of Bayesian statistics can quantify the strength of the evidence for the null hypothesis in the orientation tasks, given our sample size.
Therefore, we examined the data by calculating Bayes Factors for each dependent variable. A Bayes Factor provides an index of the fit of the data under the alternative hypothesis, relative to that under the null hypothesis. Bayes Factors above 1 represent more evidence in favour of the alternative hypothesis than the null hypothesis, whilst Bayes Factors below 1 suggest more evidence in favour of the null hypothesis compared to the alternative hypothesis. Whilst Bayes Factors represent a continuum of evidence favouring the null and alternative hypotheses, cut-offs have been proposed to facilitate their interpretation. Here, we use the commonly used convention that Bayes Factors under 1/3 or above 3 provide substantial evidence for the null hypothesis and alternative hypothesis, respectively, and that Bayes Factors between 1/3 and 3 provide only anecdotal evidence in support of either hypothesis (Jeffreys, 1961) suggesting that the data are insensitive.
Results of Bayesian analyses on the motion-processing task measures.

BF: Bayes Factor.
Inverse Bayes Factors (i.e. 1/BF) are reported to quantify the amount of support for the null hypothesis that the effect sizes are the same across experiments, and thus in support of the replication attempt. Default tests were conducted with a Cauchy prior of (0,1). Replication test refers to the method of Verhagen and Wagenmakers (2014). Effect size test refers to the equality-of-effect size test of Bayarri and Mayoral (2002). Meta-analysis test refers to the method of Rouder and Morey (2011).
Next, we used the Bayesian replication test proposed by Verhagen and Wagenmakers (2014) which uses the posterior distribution from the original study as the prior for the replication test (Figure 5) to compare the evidence for two competing hypotheses: the null hypothesis (H0) proposing that the effect is absent in the replication data (effect size = 0), and the alternative hypothesis (Hr) proposing that the replication data is consistent with the effect reported in the original study (effect size = posterior distribution from original study). Here, we found insufficient support for either the alternative or null hypothesis (Table 3; column 4) for maximum tolerable noise and sampling. These results suggest that the data in the replication sample are insensitive for discriminating between the null and alternative hypotheses, and that more data are required in order to update belief in the hypothesis that maximum tolerable noise and sampling for motion are enhanced in autistic children. In order to determine whether the effect size from the original experiment was equal to that in the replication study, we conducted an equality-of-effect size test (Bayarri & Mayoral, 2002). We report inverse Bayes Factors in Table 3 (i.e. 1/BF) to quantify the amount of evidence in support of the null hypothesis that the effect size is the same in the two experiments, and thus the success of the replication attempt (see also Verhagen & Wagenmakers, 2014). There is inconclusive evidence for determining whether the effect sizes for maximum tolerable noise and sampling differ or not across the experiments (Table 3; column 5).
Posterior distributions from the original experiment (dotted line) and replication experiment (solid line) for motion maximum tolerable noise (left panel) and sampling (right panel). The posterior distribution of the original experiment was used as the prior distribution for the replication test (Verhagen & Wagenmakers, 2014). BFr0 refers to the relative amount of evidence in favour of the alternative hypothesis (Hr) that the effect in the replication experiment is consistent with that in the original experiment, compared to the null hypothesis (H0) that there is no effect.
We have established that the data from the replication study were insensitive to group differences in motion maximum tolerable noise and sampling, but that there was strong evidence for group differences in the original sample. These results complement well our frequentist statistics. As the experiments could be considered exchangeable, we pooled the data from both experiments and assessed whether the effect was present or absent using a fixed-effect meta-analytic Bayes Factor test (Rouder & Morey, 2011) using a Cauchy distribution (0,1) as the prior for each experiment. This analysis revealed that the maximum tolerable noise values were 20 times more likely to occur under the alternative hypothesis than the null hypothesis, and that the sampling values were 7.77 times more likely to occur under the alternative hypothesis than the null hypothesis. These Bayes Factors represent strong and substantial support (Jeffreys, 1961) for the alternative hypothesis that there are differences between groups in maximum tolerable noise and sampling, respectively, and that, therefore, motion integration is enhanced in autistic children.
We then analysed the other dependent variables in the motion tasks, for which we found no group differences in the original study. We omitted Verhagen and Wagenmakers’ (2014) replication test, as there was no need to distinguish between the null hypothesis of no group effect in the replication dataset and the competing alternative hypothesis that the data from the replication dataset are consistent with the original dataset. Using default tests with a Cauchy (0,1) prior, there was substantial support for the null hypothesis (no group differences) for no-noise thresholds and coherence thresholds in both original and replication experiments. The equality-of-effect size test revealed that the effect sizes in the two experiments were comparable. Moreover, the fixed-effect meta-analysis test revealed considerable evidence for the null hypothesis when the no-noise and coherence thresholds were pooled across experiments, with the data being 16.67 (1/0.06) times more likely under the null hypothesis than the alternative hypothesis. Thus, we can be confident that there are no group differences between autistic and typically developing children in either no-noise motion thresholds or motion coherence thresholds. The evidence for atypical levels of internal noise in autism in the motion tasks is less conclusive. The default tests and meta-analysis test provide only weak/anecdotal evidence in support of the null hypothesis, but the effect sizes in both experiments are comparable.
Next, we analysed the data from the orientation tasks. In the absence of previous data to inform the priors for the orientation tasks, we used a default Cauchy prior (0,1) as above. The Bayes Factor indicated substantial evidence for the null hypothesis for no-noise thresholds (BF = 0.17), sampling (BF = 0.27), internal noise (BF = 0.18) and coherence thresholds (BF = 0.17), yet only anecdotal evidence in support of the null hypothesis for maximum tolerable noise (BF = .41). Thus, more data must be collected on the orientation tasks in order to distinguish between the null and alternative hypotheses for this variable.
Understanding variability
Figures 3 and 4 show considerable individual variability in both autistic and typically developing children. In an attempt to understand this variability, we conducted correlations between participant characteristics (e.g. age, IQ) and no-noise thresholds, maximum tolerable noise thresholds and coherence thresholds, across all participants pooled together, using a conservative value of p < .01 to correct for multiple comparisons. In the motion tasks, there were age-related reductions in no-noise thresholds, r(154) = −.40, p < .001, increases in maximum tolerable noise values, r(154) = .31, p < .001, and reductions in coherence thresholds, r(152) = −.33, p < .001. In the orientation tasks, there were age-related reductions in no-noise thresholds, r(83) = −.35, p < .001, and coherence thresholds, r(82) = −.35, p < .001, but no age-related changes in maximum tolerable noise values, p = .06. Performance IQ was negatively related to no-noise motion thresholds, r(154) = −.29, p < .001, motion coherence thresholds, r(152) = −.31, p < .001 and no-noise orientation thresholds, r(83) = −.40, p < .001, with higher IQ scores being associated with lower thresholds (i.e. higher sensitivity). Verbal IQ was negatively related to orientation no-noise thresholds, r(83) = −.29, p = .007. All other relationships between IQ and thresholds were non-significant (ps ≥ .01). There were no significant relationships between these motion and orientation threshold measures and levels of autism symptoms (SCQ scores, ADOS severity scores) nor scores on the Conners or Sensory Profile subscales (ps ≥ .02).
Response bias and inter-trial effects
In order to ensure that the superior performance of autistic children in the motion task could not be attributed to non-sensory factors, such as being less prone to bias or inter-trial effects, we estimated levels of bias and inter-trial response-dependencies in each participant for both motion and orientation tasks. The groups did not differ significantly in these non-sensory factors in either the motion or orientation tasks (see Supplementary Material).
Discussion
In this study, we set out to replicate a previous finding of increased integration of motion information in autistic children, whilst also assessing whether this perceptual advantage extended to processing of static, orientation information. We used equivalent noise and coherence tasks, for motion and orientation stimuli, with a new sample of autistic and typically developing children matched in age and non-verbal IQ.
Enhanced motion integration in autistic children
When pooling across participants tested in both the original and replication studies (n = 78 in each group), the results of frequentist and Bayesian statistics showed that autistic children have enhanced integration of direction information. Specifically, the autistic children were able to compute the average direction over greater levels of directional variability compared to typically developing children, which was reflected in greater levels of sampling in the equivalent noise model. Yet, the data collected in our replication sample alone (n = 45 in each group) do not provide sufficient evidence either for this expected group difference or for the null hypothesis. Our power analysis based on the effect size from the original study suggested that we would be sufficiently powered in this replication study. However, the original study may have overestimated the true effect size, as a result of sampling error. Whilst there appears to be a real effect of increased integration of motion information in autistic children, it is relatively small, with considerable between-participant variability and much overlap between the groups (see Figure 3). As a result, large samples are required to reliably detect the effect.
Meanwhile, we found substantial evidence in favour of the null hypothesis of no group differences for no-noise thresholds and coherence thresholds in the motion tasks across both the original and replication experiments. This pattern of results reinforces our view that the equivalent noise paradigm is an informative task in identifying the processing components that are affected in autism. Specifically, autistic children appear to have a selective enhancement in motion integration, despite typical fine direction discrimination in the absence of noise, and typical motion coherence thresholds. We suggest that autistic children may not benefit from their increased integration capacities in the motion coherence task due to the additional requirements of segregating signal from noise. The data for internal noise are less conclusive, suggesting that even with a combined sample (n = 78 participants in each group), the data are not sensitive to distinguish between the null and alternative hypotheses. Thus, in terms of whether internal noise levels are altered in autism, it appears that the jury is still out. Yet, it should be noted that mean levels of internal noise are slightly higher in autistic children than typically developing children, suggesting that if a group difference in internal noise levels does indeed exist, it is likely to be in the direction of increased internal noise in autism (Simmons et al., 2009, cf. Davis & Plaisted-Grant, 2015).
No evidence for altered orientation processing in autistic children
The second aim of this study was to assess whether the motion findings generalised to a static, orientation task. We found no evidence for differences between autistic and typically developing children in orientation processing in any measure. However, whilst there was substantial evidence in favour of the null hypothesis for no-noise thresholds, coherence thresholds, internal noise and sampling values, there was only weak/anecdotal evidence in favour of the null hypothesis for maximum tolerable noise. Thus, whereas we can be confident that autistic children perform similarly to typically developing children in most measures in the orientation task (see also Freyberg et al., 2016; Schwarzkopf et al., 2014; Shafai et al., 2015), larger samples will be required to provide conclusive evidence as to whether the integration of orientation information is atypical in autism. Yet, the values presented here suggest that orientation integration may be slightly weaker in autistic children compared to typically developing children, as opposed to enhanced as in the case of direction perception.
Implications for theories and future directions
Our finding of increased direction integration in autistic children stands in contrast to theories proposing reduced global processing in autism (e.g. Frith & Happé, 1994), but in line with the increased perceptual capacity account (Remington, Swettenham, Campbell, & Coleman, 2009; Remington, Swettenham, & Lavie, 2012). Rather than a simplistic ‘local’/‘global’ distinction, we suggest that the component processes of tasks need to be disentangled. For example, a distinction between integration and segregation of signal from noise demands may be informative in understanding autistic perception. Indeed, increased integration of motion information, along with reduced segregation of signal from noise, could be accounted for by contemporary theories of autistic perception using Bayesian and predictive coding models (Lawson, Rees, & Friston, 2014; Pellicano & Burr, 2012; Van de Cruys et al., 2014), with autistic children perhaps not distinguishing between what motion information is relevant and irrelevant in the same way as typically developing children. Finally, our results do not support suggestions of altered levels of internal noise in autism (Davis & Plaisted-Grant, 2015; Simmons et al., 2009), although we note that more data must be collected from the motion task in order to provide conclusive evidence.
Sensory symptoms have only recently been incorporated into the diagnostic criteria for autism (DSM-5; APA, 2013) and we still have a relatively limited understanding of the nature and causes of sensory symptoms. Therefore studies that aim to characterise better autistic perception, like this one, are valuable. Increased integration of motion information in autistic children could potentially be linked to feelings of sensory overload in autistic children. In the current study, task performance was not linked to parent-reported sensory processing, but to fully assess the link between task performance and everyday sensory processing, it will be important to collect more extensive parent-report measures and to supplement these with child-report measures, whilst bridging the gap between laboratory studies and everyday life by designing more real-world tasks.
Whilst more data are required for the orientation tasks, our results raise the intriguing possibility that enhanced integration is specific to motion, and does not extend to static, orientation information. This pattern of results would be particularly informative for guiding theories of autistic perception, by suggesting that prevailing domain-general theories of autistic perception, like atypical levels of internal noise (e.g. Simmons et al., 2009), or reduced global processing (e.g. Frith & Happé, 1994) need to be superseded by domain-specific accounts. More studies comparing performance across different tasks within the same sample of autistic and typically developing individuals are required. Indeed, if the pattern of increased integration for motion but not static information is borne out in these future studies, we must ask the question of what makes motion information special for autistic individuals in terms of integration.
The use of equivalent noise tasks may also allow us to distinguish patterns of perceptual performance between different disorders. The same equivalent noise tasks have already been used with people with schizophrenia and migraine (Tibber et al., 2014, 2015). Schizophrenic patients demonstrate selective difficulties in integrating orientation information, but no differences in their perception of motion and size information (Tibber et al., 2015), whilst migraineurs show no differences in orientation, motion or size equivalent noise tasks, yet have elevated motion coherence thresholds (Tibber et al., 2014). Thus, it seems that enhanced integration of motion information may be a distinguishing feature of autistic perception. However, difficulties segregating signal from noise could be common to both autistic individuals and migraine patients. The combined use of equivalent noise and coherence tasks with different stimuli therefore allow more fine-grained comparisons between disorders, in comparison to use of the motion coherence task alone, which has revealed elevated thresholds in a range of disorders (e.g. dyslexia: Hansen, Stein, Orde, Winter, & Talcott, 2001; Fragile X syndrome: Kogan et al., 2004; Williams Syndrome: Atkinson et al., 1997).
Finally, it is important to emphasise the individual variability in perceptual performance in both autistic children and typically developing children in our sample. This variability in perception may explain inconsistent results in the literature (e.g. motion coherence; Simmons et al., 2009), particularly when combined with the use of small sample sizes. We therefore stress the need for large sample sizes when assessing group differences in autistic perception, with replications and ultimately meta-analyses to establish the robustness of effects. Achieving large sample sizes is undoubtedly difficult when testing clinical populations, and so collaboration between research teams may be necessary. The issue of small sample sizes is compounded by the use of frequentist statistics which cannot quantify the amount of evidence in support of the null hypothesis, thus in the case of a non-significant (p > .05) result, it is not possible to determine whether the groups really do not differ, or whether the data are just too insensitive to detect differences (Wagenmakers, 2007). Therefore, the use of Bayesian statistics will allow researchers to consider the relative evidence for the null and alternative hypotheses when faced with non-significant p-values. Indeed, rather than just assessing group differences between autistic and typically developing children, future studies are needed to try and explain the between-participant variability in perceptual performance. Here, we suggest that age and ability are related to performance in some tasks, although future investigations are needed to determine what might account for the remaining unexplained variability.
Conclusions
Autistic children show enhanced integration of direction information compared to typically developing children, although there is considerable between-participants variability and thus large samples are required to detect the effect. There is no indication of atypical integration of orientation information in the current study, although more data will be required in order to provide conclusive evidence.
Footnotes
Acknowledgements
Thanks to all the participants, families, schools and parent support organisations that participated in and facilitated this research; to Dorothy Bishop for advice on the analysis and interpretation of results, and to Eric-Jan Wagenmakers for advice on Bayesian analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by a Scott Family Junior Research Fellowship at University College and an Oxford University Press (OUP) John Fell Fund grant awarded to Catherine Manning. Steven C. Dakin was supported in part by Cure Kids (New Zealand).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
