Abstract
Purpose:
To develop and implement a comprehensive program to train providers to place subclavian central venous catheters (CVCs) using real-time ultrasound guidance.
Study design:
Simulation-based prospective study at an academic medical center. Of 228 anesthesia providers and intensivists eligible to participate, 106 participants voluntarily enrolled. The training program consisted of a didactic module, hands-on instruction and practice using a CVC simulator and a standardized patient. The success of the program was measured by pre and post knowledge tests and direct observation during the hands-on sessions.
Results:
Of 106 participants who enrolled, 70 successfully completed the program. Out of 20 possible procedure steps, an average of 17.8 ± 2.9 were correctly performed in the simulated environment. The average time to needle insertion, defined by positive aspiration of stained saline, was 3.35 ± 3.02 min and the average time to wire insertion with ultrasound confirmation was 3.85 ± 3.12 min.
Conclusions:
Participants learned how to successfully perform ultrasound-guided catheterization of the subclavian vein. Since ultrasound-guided subclavian CVC placement is a useful clinical skill that many practitioners are unfamiliar with, increasing competence and comfort with this procedure is an important goal. Other centers could consider adopting an approach similar to ours to train their providers to perform this technique.
Introduction
Hospitalized patients frequently require central venous catheters (CVCs), but these are associated with a variety of serious complications. 1 Common approaches to central venous catheterization include the internal jugular, subclavian, and femoral veins. In adults, the subclavian site is recommended by the Centers for Disease Control and Prevention (CDC) because it is associated with fewer infectious and thrombotic complications. 2 However, in some centers the subclavian is considered the approach of last resort, because subclavian CVC insertion is technically more challenging and can be associated with substantially more mechanical complications (ie, pneumothorax).2-4 Furthermore, reducing the incidence of iatrogenic pneumothorax from CVC placement is a critical initiative for many hospitals since this complication is a Department of Health and Human Services Agency for Healthcare Research and Quality “patient safety indicator.” Because fewer subclavian CVCs are being inserted, practitioners and trainees are becoming less experienced and less comfortable with this approach, and may not be able to competently perform it when it is indicated or even absolutely necessary.
Ultrasound guidance is frequently used for the internal jugular approach but still rarely used for the subclavian approach, despite increased cannulation success rates, increased ability to identify vascular anomalies and pathologies, and fewer complications during CVC insertion with ultrasound.2-7 For example, in a large prospective randomized study that evaluated complications of CVCs by insertion site, an anatomical landmark technique was used in 86% of subclavian insertions but in only 33% of internal jugular insertions. 1 This discrepancy is likely due to a lack of familiarity with the use of ultrasound for subclavian CVC insertion and the perception that ultrasound guidance is technically challenging to perform. Consequently, we developed and implemented a comprehensive program to teach providers how to perform ultrasound-guided catheterization of the subclavian vein, more specifically using a lateral subclavian (axillary) vein approach.
Methods
The training program was offered to all 228 providers in our department who place CVCs, including anesthesiologists, anesthesiology residents and fellows, certified registered nurse anesthetists, intensivists, and advanced practice providers working in an intensive care unit. Participation was voluntary. Most providers had little to no prior familiarity with the ultrasound-guided subclavian technique. The program consisted of 3 phases.
Phase 1 included a written assessment and online didactic module. A ten-question multiple-choice pre-test evaluated baseline knowledge of requisite sonographic anatomy, indications, contraindications, and potential complications of CVC placement. No score or feedback was offered to participants at that time. Next, a didactic module consisting of slides describing the aforementioned topics in more detail, and a video demonstrating the technique, was presented (the video can be found at https://www.nejm.org/doi/full/10.1056/nejmvcm1406114). 3
In phase 2, participants completed a hands-on training module. First, a preceptor experienced with CVC placement demonstrated the relevant surface and sono-anatomy using a standardized patient. Next, participants practiced patient positioning, skin preparation and draping, and identification of relevant surface and sono-anatomy on a standardized patient. Using ultrasonography (Sonosite X-PORTE with probe Model HFL38xp, bandwidth 13-6 MHz Linear), subclavian artery and vein positions were identified, and then both structures were interrogated with color flow Doppler. The location of the clavicle and pleural line relative to the vein were noted, and the optimal site and trajectory for needle entry was selected.
In phase 3, participants practiced the CVC placement procedure on a task trainer (Blue Phantom®, Gen II ultrasound central line training model, CAE Healthcare, Sarasota, FL). A preceptor (EAD) was present to provide coaching and feedback to the participants. They had to successfully verbalize and demonstrate the pre-procedural steps and successfully acquire ultrasound images on the standardized patient. Participants then were observed while performing the procedure on the task trainer. Key portions of the procedure were recorded, including needle stick, venous access, guide wire insertion and confirmation, insertion of the catheter, and removal of the guide wire. Any observed errors were recorded. During phase 3, feedback was not provided until the procedure was completed. Finally, a post-test questionnaire was administered and graded.
Results
Of 228 invited participants, 106 (47%) accessed the online didactic module, and 70 (31%) completed the entire training program. The majority of participants (94%) reported having placed at least 1 internal jugular CVC during the prior 12 months, and only a minority (28%) had placed at least 1 subclavian CVC during the prior 12 months (Table 1).
Respondents’ characteristics and experience with central venous catheter placement.*
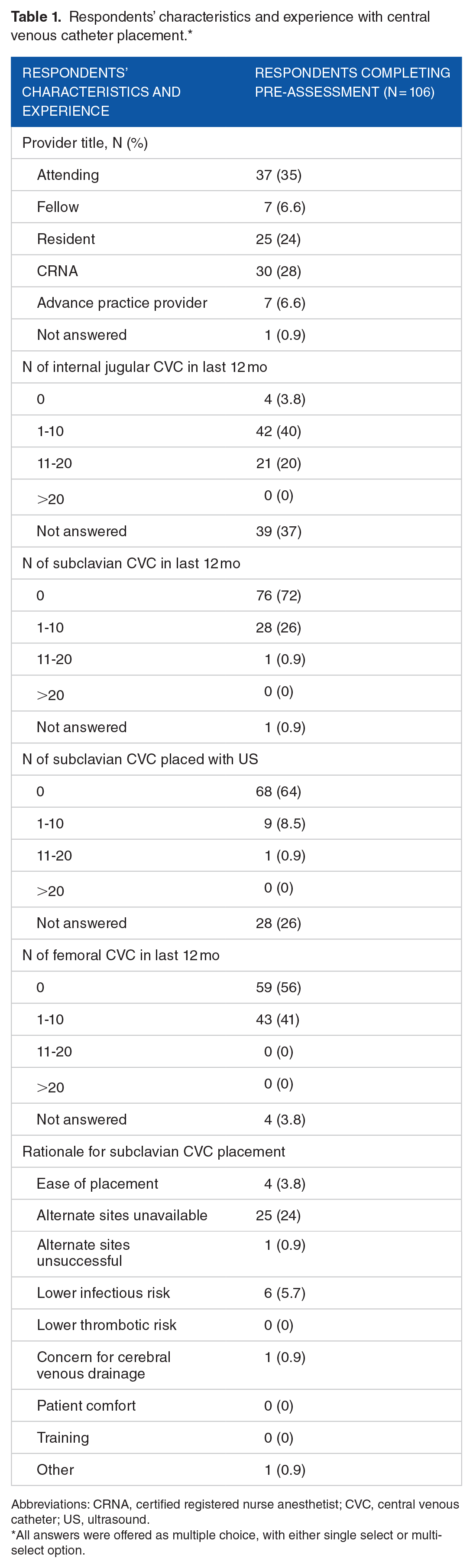
Abbreviations: CRNA, certified registered nurse anesthetist; CVC, central venous catheter; US, ultrasound.
All answers were offered as multiple choice, with either single select or multi-select option.
The post-test revealed that participants learned key information about subclavian CVC placement (Table 2).
Pre-training and post-training assessment. Values shown are N (%).

Abbreviations: CLABSI, central line associated bloodstream infection; IJ, internal jugular; US, ultrasound.
P values comparing discordant pairs for the correct responses in the pre- and post-test assessment questionnaires using McNemar test for pair binary data. Only participants completing both tests are included in this analysis (n = 70).
This indicates the correct answer. Our institutional policy requires US-guidance for placement of internal jugular CVC.
Specifically, post-training assessment revealed significant improvement in key learning points including incidence of central line associated bloodstream infection (CLASBI) and thromboembolism with the subclavian as compared with alternate sites of CVC placement, appropriateness of subclavian CVC placement in coagulopathic patients, and institutional policy details regarding the placement of subclavian CVCs. The majority of participants correctly performed all a priori defined 20 steps of the procedure checklist in the standardized patient and in the task trainer, with an average score of 17.8 ± 2.9 (SD) out of 20 (Table 3). The average time to needle insertion in the subclavian vein, defined by the positive aspiration of blue stained saline, was 201 ± 181 seconds (range 25-827 seconds), and the average time to wire insertion was 231 ± 187 seconds (range 45-909 seconds). The average number of attempts defined as each time the needle was completely removed and reinserted, was 2.3 ± 1.6 (range 1-8). Complications included arterial puncture (n = 4), wire advanced through the vein (n = 1), and inability to successfully cannulate during the assessment period (n = 1). No pleural puncture was observed (Table 3).
Providers’ performance for hands on training, based on the catheter insertion checklist. Values shown are n (%), unless otherwise specified.

Abbreviation: SD, standard deviation.
Discussion
To our knowledge, we are the first to describe a comprehensive training program for ultrasound-guided subclavian CVC placement. Compared to traditional apprentice-model modes of education, simulation and competency-based approaches to procedural instruction for invasive bedside procedures have demonstrated improvements in testing scenarios, and procedural success in practice.8,9 Therefore, for novel or rarely performed procedures, a simulation-based module with competency evaluation in CVC placement should be a standard component of training and continuing education.
For didactic training, we used a PowerPoint with embedded video that the participant viewed independently at their own pace, which allowed more time dedicated to hands-on learning during the simulation component of training.2-4 For hands-on assessment, we chose a hybrid approach with both a standardized patient and task trainer for assessing different skills. The standardized patient provided high fidelity simulation of anatomy and practice in guiding the participant through expected steps of the procedure. The task trainer provided practice with needle guidance, wire advancement, and confirmation of wire placement with ultrasound. To our knowledge, no other training programs have reported this unique approach to aid learners in CVC placement.
To best evaluate competency among participants, checklists and global rating scales are most frequently used, and are well-validated measures of success for educational intervention in procedural skills. While global rating scales appear to have better validity than checklist assessments, they have worse interrater reliability and are less objective. 9 Since the goal of our training program was to familiarize participants with a wide range of clinical experience using a new technique, we believe our checklist provides the best assessment of competency for participants to proceed to supervised clinical practice. Our checklist was formatted similarly to another validated checklist used for measuring procedural success in ultrasound-guided internal jugular CVC placement. 10
Limitations of our approach include lack of evaluation for posterior-wall puncture (as the task trainer could not be assessed physically after each participant’s attempt) which is a common technical error of this procedure. Other measures of procedural competency include hand motion-tracking, eye gaze pattern-tracking, and robotic arm haptic feedback that correlate well with learner expertise, but still require study for threshold validation for learner success in practice. 11
Another potential limitation includes participant dropout (36 of 106 participants who completed phase 1 did not go on to complete the in-person training, a potential source of attrition bias), and lack of follow-up for long-term knowledge and skill retention among participants. We also did not directly evaluate the success and proficiency of internal jugular CVC placement relative to subclavian, primarily because all participants were familiar with internal jugular placement.
Finally, the role of feedback in our protocol was neither assessed nor formalized. While we provided verbal feedback at the discretion of the preceptor, it is possible that “error exposure training” (errors incorporated into simulation with challenge and resultant feedback) may have improved knowledge/skill retention and adaptive transfer of skills as demonstrated in other studies of CVC education. 12
Conclusion
Overall, our study participants demonstrated successful simulated line placement and improved comprehension of knowledge goals, which suggests that our simulation-based subclavian CVC placement training is an effective educational intervention that may be easily reproduced outside our department. In addition, our training intervention provides the basis for future studies to directly compare the success and complication rates of ultrasound-guided internal jugular and subclavian CVCs. Future studies will evaluate clinical skills performing the procedure in clinical practice.
Our training program provides a comprehensive didactic and hands-on approach to training in placement of ultrasound-guided subclavian CVCs made more robust with the addition of standardized patients and checklist assessment of learner competency. Because this program was offered to providers with a wide range of experience, it is potentially generalizable to other institutions that wish to increase competence and comfort with this procedure.
Footnotes
Acknowledgements
The authors wish to thank their colleagues who have provided feedback, support, and participated in the program.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported with departmental funds and by the Foundation for Anesthesia Education and Research (FAER) Medical Student Anesthesia Research Fellowship to Esi Dickson. The authors alone are responsible for the content and writing of the article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Note
The work was performed at Oregon Health and Science University, Portland, Oregon.
Contributors
JDD contributed to data acquisition, drafted and revised the manuscript, and approved the final version; MMT designed the study, analyzed the data, edited and revised the manuscript and approved the final version; EAD contributed to study design, data acquisition, revised the manuscript and approved the final version; PMS designed the study, edited and revised the manuscript and approved the final version.
