Abstract
Introduction:
This descriptive study aimed to evaluate the depth, extent, and perception of pharmacogenomics instruction in schools and colleges of medicine in the United States. Changes in medical pharmacogenomics instruction over the past decade were also assessed by comparing our results with those of a previous study.
Methods:
An electronic survey was emailed to all accredited allopathic and osteopathic medical schools across the US using Qualtrics online survey software. Multiple email reminders were sent to increase the response rate.
Results:
Of 151 targeted eligible medical schools across the United States, 22 responded to the survey. One invalid response was excluded, resulting in a response rate of 13.9%. Of responding schools, 85.7% cover pharmacogenomics in their curriculum, mainly in the second year, however, none teach pharmacogenomics as a stand-alone course. The depth and the extent of pharmacogenomics coverage varied among responding programs. Although 66.7% of respondents believe that neither physicians nor other health care professionals possess appropriate knowledge in pharmacogenomics, only 23.8% plan to increase pharmacogenomics instruction in their curricula in the near future.
Conclusions:
Most medical schools surveyed include some pharmacogenomics instruction in their curricula, although the depth and the extent of the instruction varies. Most respondents believe that physicians and other health care professionals today do not possess an appropriate level of knowledge in pharmacogenomics; however, few institutions report short-term plans to increase pharmacogenomics instruction. Pharmacogenomics plays a significant role in personalized medicine; greater efforts by medical school decision-makers are needed to improve the level of pharmacogenomics instruction in medical curricula.
Introduction
Pharmacogenomics is the study of the impact of genetic variability on drug response. The overarching goals of pharmacogenomics studies are to maximize the therapeutic efficacy of drugs and to minimize their risk of toxicity.1-3 Completion of the Human Genome Project in 2003 greatly influenced the evolving field of pharmacogenomics and opened doors for innovative new drug discovery and development, and for increasingly personalized medicine. 4 Indeed, the increasing number of known clinically significant genetic variants have been recognized across all disciplines of medicine. As a result, the classic one-size-fits-all treatment strategy has been falling out of favor and is being slowly replaced by a personalized medicine approach in which medical therapy is tailored to patient-specific factors including genetic profile.
The growing clinical relevance of pharmacogenomics is reflected in drug labeling; as of March 2019, more than 350 drugs approved by the US Food and Drug Administration (FDA) 5 contain pharmacogenomic-related prescribing information in their labels, including black box warnings, dose adjustments, and other precautions that should or must be taken for patients with certain genetic polymorphisms. For example, the painkiller codeine is a prodrug that requires metabolism by the CYP2D6 enzyme to generate its active metabolite. The drug label for codeine contains a black box warning highlighting the risk of life-threatening respiratory depression in patients who have the CYP2D6 “Ultra-rapid metabolizer” polymorphism and take codeine. 5
Despite the increasing amount of pharmacogenomic information available, fewer than 1 in 8 US primary care physicians report having ordered a pharmacogenomic test or feeling confident about using a pharmacogenomic test result. 6 Several medical centers and health systems have implemented programs encouraging consideration of pharmacogenomics in clinical care, and electronic health records increasingly contain pharmacogenomic decision support tools; however, even among physicians benefiting from these resources, uncertainty about preparedness to receive pharmacogenomic test results exists.6,7
The International Society of Pharmacogenomics (ISP) held a Pharmacogenomics Education Forum during its third annual meeting in October 2004, which resulted in recommendations and a call to action being sent to Deans of medical, pharmaceutical, and other health professional schools around the world.
8
ISP stated that “medical, nursing, pharmacy, medical biology, and health administration students should all be able to comprehend pharmacogenomics foundations and principles by the time they finish their graduate studies.” Several private, governmental, and professional organizations have also promoted pharmacogenomics education. The American Medical Association (AMA) and its partner the National Coalition for Health Professional Education in Genetics (NCHPEG) are dedicated advocates for genetics and personalized medicine implementation in improving health care outcomes.9,10 NCHPEG mission “Is committed to ensuring that our nation’s health care providers have the knowledge, skills, and resources to integrate new genetic knowledge and technologies into prevention, diagnosis, and management of the disease; provide high quality services for patients, families, and communities affected by genetic-related conditions; and understand and address the ethical, legal and social implications of advancing genetic knowledge and technologies.”
11
The AMA 10 has also developed various Continuing Medical Education (CME) resources, including an online certificate program through the American College of Clinical Pharmacology (ACCP), 12 and a web-based training program to further enhance clinicians’ understanding of pharmacogenomics application in clinical practice. The Association of American Medical Colleges (AAMC) 13 and the Association of Professors of Human and Medical Genetics (APHMG) 14 have also requested that schools of medicine should have the proper training for their students so that the new graduates are competent in the field of medical genetics and genomics. The National Institutes of Health National Human Genome Research Institute (NHGRI) and the Genetics and Genomics Competency Center (G2C2) also provide educational and practice-based resources for various medical professionals including physicians about the current pharmacogenomics competencies.15,16 The FDA has used several means such as educational workshops to enhance the knowledge and the use of pharmacogenomics.
A 2007 survey of US and Canadian medical schools reported that 77% of respondents taught medical genetics; 62% of those schools devoted between 20 and 40 hours to medical genetic instruction. 17 In 2010, a survey study was used to assess the depth of pharmacogenomics instruction in the schools of pharmacy, and the results showed that most pharmacy schools across the United States have increased pharmacogenomics education. 18 A similar study done in the same year to assess the depth of pharmacogenomics education in allopathic and osteopathic medical schools across the United States and Canada revealed that unlike the schools of pharmacy, the medical schools’ curricula incorporated an inadequate level of pharmacogenomics education, and this inadequacy has led to future physicians not being fully prepared for the implementation of pharmacogenomics into their future clinical settings. 19
A study published in 2012 assessed the adoption of pharmacogenomics testing by US physicians. Of 10 303 physicians who completed the questionnaire, only 29.0% had received any education in pharmacogenomics, and only 10.3% felt adequately informed about pharmacogenomic testing. 20 These data were consistent with a 2008 US Department of Health and Human Services report, which stated that many clinicians do not possess the knowledge to interpret pharmacogenomic information and that pharmacogenomic education needs to be better incorporated in medical school curricula. 17
This study examines the current status of pharmacogenomics education in the US allopathic and osteopathic schools of medicine, current perceptions of pharmacogenomics’ importance in those schools, and plans they have for changing the amount of pharmacogenomics content in their curricula. Our results are compared with those of Green et al 19 in 2010 to explore how medical schools have changed over the past decade in their perception of pharmacogenomics and incorporation of this field in their curricula.
Methods
Following the precedent of previously published studies in this area, a web-based survey consisting of 26 questions was created to (a) evaluate the extent and the depth of pharmacogenomics instructions in the US allopathic and osteopathic schools and colleges of medicine, and to (b) capture respondents’ perceptions of and (c) future plans for pharmacogenomics instruction in US medical curricula.18,19 The survey was sent using the Qualtrics web-based survey tool to all US medical school administrators with oversight of the school’s curriculum. The survey was active for almost 3 months and multiple email reminders were sent to maximize the response rate. Data received were analyzed using descriptive statistical analysis and compared with a similar survey published by Green et al 19 in 2010.
Results
Of 151 eligible medical schools, 22 responded to our survey. One invalid survey was excluded from our study resulting in a 13.9% response rate. Table 1 provides information about the amount and level of pharmacogenomics education in respondents’ curricula, respondents’ perceptions of physician and other health care professionals’ pharmacogenomics knowledge, as well as details on future plans regarding the incorporation of pharmacogenomics in respondents’ curricula. The results of the current study are compared with the results of a similar study published in 2010, where applicable. 19 As shown in Table 1, 85.7% of the respondents include pharmacogenomics education in their curricula; this is a small increase from 82% reported by Green et. al. in 2010. Surprisingly, none of the respondents taught pharmacogenomics as a stand-alone course; this is a decrease from 2010 when 2.7% of respondents had stand-alone required courses in pharmacogenomics, and 9.5% offered stand-alone pharmacogenomics electives. Instead, 80.9% of responding schools currently teach the topic as part of another course and spend less than 10 didactic hours on the subject per term. Most pharmacogenomics coursework (61.9%) resides in the second year of the responding schools’ curricula and the coursework is mainly taught by faculty members from the basic sciences department (80.9%). Although 71.4% of programs responded to our survey offer pharmacogenomics education to medical students, 28.6% of programs offer the subject to their PhD candidates. Pharmacogenomics education is also offered to other health care professionals such as physicians, nurses, or pharmacists at 23.8% of responding institutions.
General characteristics, perception and plans of respondents on pharmacogenomics instruction.

Abbreviations: FY, fiscal year; MD, medical doctor; PA, physician assistant.
% for each question was calculated based on total number, an = 74, dn =71 gn = 77, hn = 75 in Green et al 19
Elective didactic course (stand-alone or mixed). 19
MD/DO. 19
Adequate. 19
Not at all adequate. 19
The coverage of basic genetics-related topics and competencies in responding medical schools’ curricula is detailed in Table 2. More than half of schools reported offering instruction in 6 out of 9 items; the 3 items that fewest schools reported coverage of were as follows: (1) “Identification of the appropriate information resources on pharmacogenomics,” confirmed by 28.5% of respondents; (2) “Family genetic history as an assessment tool of disease predisposition and prevention,” confirmed by 33.3%; and (3) “Understanding genotyping versus phenotyping methods of applications in genetic-associated diseases,” confirmed by 47.6%. In terms of contact hours, coverage varied but in general, 5 or fewer hours were devoted to most basic genetics topics and competencies. However, nearly a quarter of respondents (23.8%) spent 6 or more hours on “Basic genetics, gene regulation, and related terminologies.”
Basic genetic concept competencies and topics taught (n = 21).

Clinical pharmacogenomics results are detailed in Table 3. “Pharmacogenomics of cardiovascular diseases and antiplatelets/anticoagulants drugs” and “Genetic variability that affects hypersensitivity and adverse drug reactions” are the 2 most frequently covered items; both are covered by 71.4% of schools. On the other hand, the least-covered topic was “Pharmacogenomics of respiratory diseases and related drugs,” with confirmed coverage in only 9.5% of schools. The number of contact hours offered varied among the items listed, with no schools reporting more than 6 hours of coverage.
Clinical competencies and topics taught (n = 21).
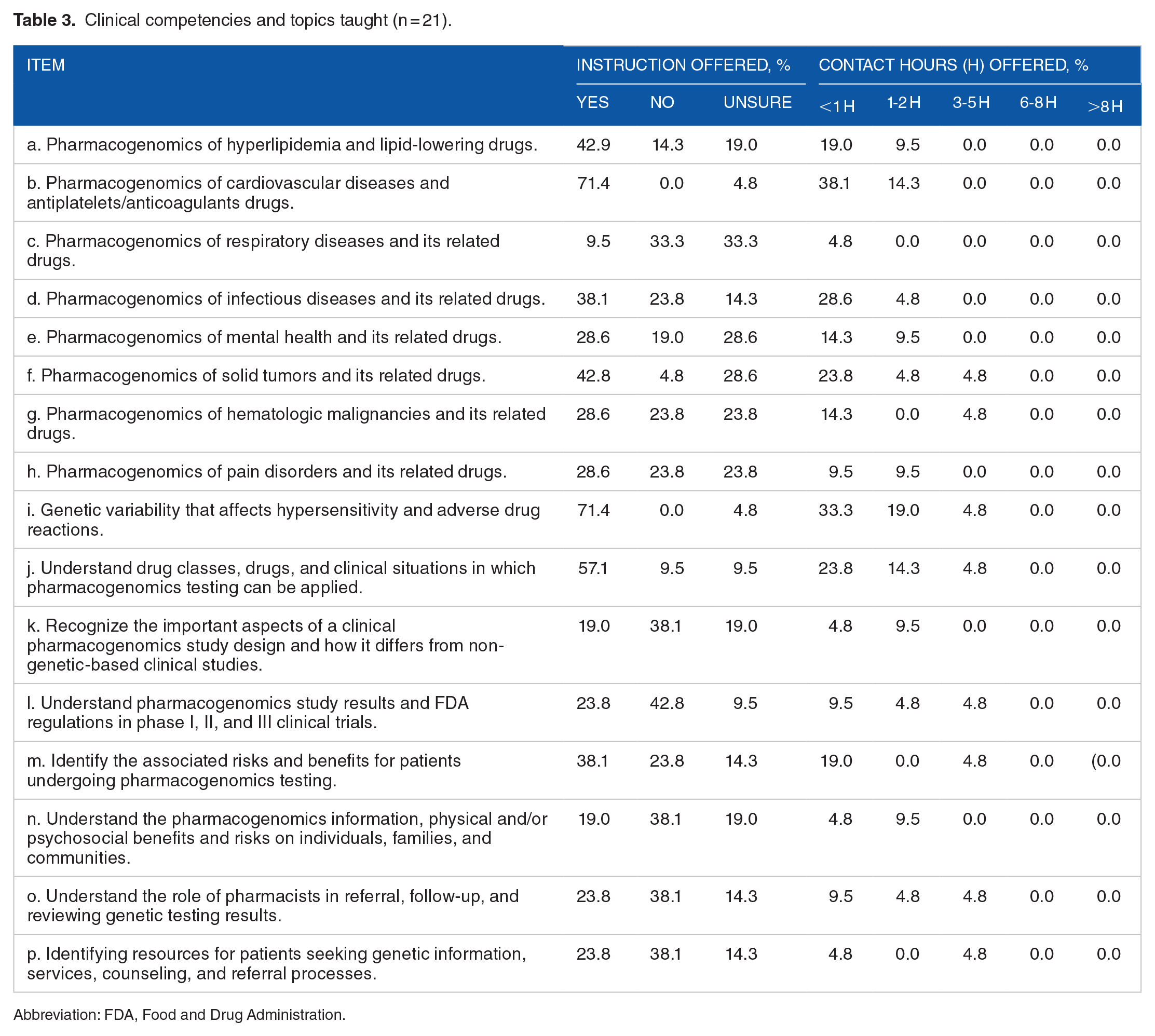
Abbreviation: FDA, Food and Drug Administration.
Ethical, legal, social, and economic core competencies and topics results are shown in Table 4. These competencies are covered by a much smaller proportion of schools when compared with most of the basic genetics and clinical competencies described in Tables 2 and 3. Less than one third of responding institutions reported offering instruction in any of the 8 ethical, legal, social, and economic topics. The item with the highest coverage (28.6%) was “Maintain the confidentiality and security of patient health records.” Two or fewer contact hours were typically devoted to these topics; however, 1 school did report spending 6 to 8 contact hours on “Fairness and impartiality in the delivery of patient education.”
Ethical, legal, social, and economic competency and topics taught (n = 21).

According to our survey, 66.7% of respondents believe that neither physicians nor the other health care professionals such as pharmacists, nurses, or physician assistants possess the appropriate knowledge of pharmacogenomics, as shown in Table 1. In addition, 42.9% of respondents feel that the present state of pharmacogenomics education in schools of medicine is “average,” whereas none feel that it is above average (“good” or “very good”). A few respondents were more positive about the state of pharmacogenomics education at their own institutions; 52.4% felt that it was “average,” and 1 school felt that it was “good.” Although almost one quarter of the respondents plan to increase the incorporation of pharmacogenomics in their curriculum (23.8%), none of the respondents plan to hire new faculty to teach the subject. Finally, 61.9% of the respondents are unsure whether their program is planning to access a shared pharmacogenomics curriculum, an online educational program developed by the University of California, San Diego Skaggs School of Pharmacy and funded by the Centers for Disease Control and Prevention (CDC) grant for a component of didactic instruction.21,22
Discussion
Medical students should know the essentials of pharmacogenomics to better ensure that medication use is effective and safe. Despite recent attention to pharmacogenomics in the medical community, it appears that there is still a lack of emphasis on the topic among most of the schools of medicine.
According to our study, 85.7% of the US responding schools included pharmacogenomics within their curricula, a small increase from the 82% of schools reported pharmacogenomics coverage in a 2010 study that was conducted in medical schools across United States and Canada. 19 Increasing pharmacogenomics coverage in medical curricula supports the importance of pharmacogenomics in medical education and aligns with several educational and professional organizations initiatives and recommendations.8,13,14,19,23 US schools of medicine are behind schools of pharmacy in this regard.18,24 In 2005, only 78% of pharmacy schools offered pharmacogenomics education in their curricula, but this increased to 92% in 2010, then to 100% in 2018.18,24,25 In addition, more schools of pharmacy are teaching pharmacogenomics as a stand-alone subject, whereas all responding programs in our survey offer pharmacogenomics as a part of another course. These findings suggest that schools of pharmacy are putting more emphasis on delivering pharmacogenomics instruction to their students and stricter adherence to the Accreditation Council for Pharmacy Education (ACPE) 26 2016 educational standards compared with medical schools. Most pharmacogenomics coursework resides in the second year of the responding schools’ curricula and little exposure to pharmacogenomics during the third and the fourth years in which the students are doing their clinical rotations. Indeed, the clinical applications of pharmacogenomics can be better taught if offered while students are doing their clinical rotations. A possible explanation of less emphasis on pharmacogenomics instruction during the third and fourth years is that the subject may be viewed by the curriculum decision-makers as a basic science topic and not valued as clinically relevant to students’ clinical years.
Approximately 81% of respondents to our survey met the recommendation and included up to 10 didactic hours per term on the subject. 17 Although the number of didactic hours offered is satisfying, yet we believe it is inadequate. One possible reason why medical schools are not incorporating more pharmacogenomics in their curricula is the lack of emphasis of pharmacogenomics on the medical licensing exam.10,27,28 Our results also showed that more PhD students in the medical programs are receiving instruction on the topic compared with previous study and such improvement might be explained by the increased number of graduate programs within schools of medicine. Although pharmacogenomics instruction was reported to be mostly delivered by single instructors, our study revealed that basic science faculty members are more involved in teaching the subject. The less contribution of the clinical practice faculty members in teaching pharmacogenomics may suggest that further emphasis on the clinical applications, social, and administrative topics is needed to enhance their involvement.
Regarding the 3 major competencies and related topics, we have found that the depth and the breadth were variable among the schools. Although the basic genetics scored the highest average of 54.5% for all of the 9 items listed in Table 2, followed by the clinical competencies with an average of 35.4% to the 16 items listed in Table 3, the ethical, legal, and social competency and topic taught scored the lowest with an average of 16.1% to the 8 items as listed in Table 4. There are many possible explanations for the low coverage of ethical, legal, and social competencies: the inadequate data on cost benefits of pharmacogenomics testing, the limited availability of ethical cases, and the faculty members are lacking the required experience and knowledge on the topic and the possibility that our survey questions did not include other areas related to ethical, legal, and social competency taught at the responding schools.
In a previous study, 42.9% of medical schools thought their pharmacogenomics education was adequate or better (“adequate,” “good,” or “very good”); however, our findings reveal that 57.1% of medical schools think their pharmacogenomics education is average and above (average, good, and very good), this further increase stipulates an advancement in pharmacogenomics education among medical schools in the United States. 19 Although most medical schools believe their pharmacogenomics education is better than other schools, this perception is also noted in the schools of pharmacy, where most schools of pharmacy also believe their pharmacogenomics education is better than other schools.18,19,24 A possible explanation for the apparent gap in pharmacogenomics instruction between other schools and the respondent’s institution might be due to the responder’s familiarity and knowledge of their own program, their institution efforts, and future plans much more than other schools or programs.
There is an agreement on the importance of pharmacogenomics and its vital role in clinical settings. Also, most respondents agree that today’s physicians and other health care professionals still do not possess adequate knowledge of pharmacogenomics. However, only 23.8% of the respondents express their interest in increasing pharmacogenomics instruction in their schools during the next 3 years with little interest in hiring new faculty members to teach the subject. Possible explanation for not hiring additional faculty members can be attributed to the institution’s financial situation and the availability of well-trained existing faculty members who are qualified to teach pharmacogenomics.
The very low interest in accessing the shared curriculum, the well-developed online program which was created by the University of California San Diego Skaggs School of Pharmacy might be attributed to either a lack of knowledge about the program or the rapidly evolving nature of pharmacogenomics clinical application which may make much of the 2009 program outdated.
Limitations
One of the major limitations of our study is the low response rate. Despite our efforts to encourage the study subjects to complete the survey by sending multiple email reminders, using an easy online survey method, and considering the number of survey questions, few schools responded to our survey. There are many reasons contributed to such low response rate including survey fatigue, lack of easy access to information for answers to in-depth survey questions (eg, coverage hours), and time constraints. Our survey may also be subject to bias if responding programs have a greater emphasis on pharmacogenomics in their curricula than non-responding programs. Finally, responding schools may be teaching aspects of pharmacogenomics that we failed to cover in our survey, as well as the opposite: the questions asked might pertain to topics of pharmacogenomics not being taught or not included in the curricula of medical schools. However, in previous studies, it has been shown that a low response rate did not appear to affect the survey results and study conclusion nor necessarily associated with non-response bias.29,30 Our study gives valuable information on the current status of pharmacogenomics education in medical schools’ curricula across the United States.
Conclusions
Based on our findings, there has been some advancement in pharmacogenomics education and its incorporation in medical schools’ curricula throughout the past decade. However, despite improvement in the field of pharmacogenomics, not all medical schools are incorporating pharmacogenomics instruction in their curricula. Compared with schools of pharmacy, medical schools are behind in this regard. Medical schools should call for further inclusion of the subject into their curricula, to improve and upgrade the depth and extent of pharmacogenomics education, which will eventually lead to future physicians being adequately prepared and confident enough to apply pharmacogenomics in their everyday practice.
Footnotes
Acknowledgements
We would like to thank Dr Monica Shear (The Medical Center of Aurora, Aurora, CO) for preparing the medical schools’ contact list and Dr Alice Ghabauer (University of Charleston School of Pharmacy, Charleston, WV) for manuscript editing and constructive comments.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Note
Pharmacogenomics education is crucial to achieving better therapeutic efficacy and lower drug toxicity in clinical practice. This study investigates the depth, breadth and perception of pharmacogenomics instruction in US medical curricula. Although there may be a small increase in the inclusion of pharmacogenomics education in medical curricula, additional efforts are needed to advance the education on this subject.
Author Contributions
DB, contributed to data analysis, interpretation, literature review and drafted the manuscript. AS, contributed to the study conception and design, survey distribution, data collection, analysis and manuscript editing. DB and AS reviewed the manuscript and approved it for publication.
Ethical Statement
The study was approved and granted exempt status by the University of Charleston Institutional Review Board (IRB), protocol # 17-0011. The study was conducted according to the university’s ethical principle guidelines. Study participants were emailed descriptive information about the study and a link to complete the survey. Participation in the study was voluntary and all respondents’ information was kept confidential.
