Abstract
Highlights
We translated, adapted, and tested the French version of Low Literacy Decisional Conflict Scale in adolescents.
The French version of the Low Literacy Decisional Conflict Scale is adapted to people with limited literacy skills.
We hope that it might facilitate the participation of vulnerable populations in clinical research projects.
Patient-reported outcome measures (PROMs) are powerful tools that capture patients’ perspectives on their health, driving personalized and patient-centered care by directly informing clinical decisions and improving health care outcomes. The Decisional Conflict Scale (DCS) is a PROM designed to measure the decisional conflict, which is defined as a state of uncertainty about a course of action, and considered a core component of the quality of the decision-making process. 1 The original 16-item DCS was developed in the 1990s by O’Connor 1 and includes 5 subscales: Uncertainty, Feeling Informed, Values Clarity, Feeling Supported, and Effective Decision Making. An abbreviated 10-item version of the DCS was later designed by the same team for individuals with limited literacy skills (Low Literacy Decisional Conflict Scale[ LL-DCS). 2 This modified version differs from the original DCS in several aspects. While the standard DCS uses item statements with a 5-point Likert-type agreement scale, the LL-DCS reformulates items as questions with simplified response options (yes, no, unsure). Furthermore, the LL-DCS omits the Effective Decision Making subscale and reduces the number of items in both the Uncertainty and Values Clarity subscales by 1 each.
Although the original 16-item DCS has been translated and validated in many languages, 3 the LL-DCS has only been validated in English 4 and Spanish.5,6 There is currently no French version of the LL-DCS, despite French being the fifth most widely spoken language globally, with more than 310 million speakers and official status in 29 countries across multiple continents. 7 There is a need to make PROMs available in French to obtain quality research data from French-speaking populations and to capture views and perspectives from diverse populations with various cultural, linguistic, and ethnic backgrounds.
The DCS has been used in research involving children and adolescents8–11 and has demonstrated strong psychometric properties, outperforming other instruments in pediatric contexts. 12 Adolescents face unique decision-making vulnerabilities compared with both children and adults, driven by the ongoing maturation of the prefrontal cortex until around age 25, as well as their progressive individuation and growing autonomy from their parents. 13 Therefore, validating assessment tools for this age group is essential to address their distinct decision-making challenges and to improve support interventions.
Choosing hormonal treatments for contraception or menstrual management can be particularly challenging for adolescents. 14 This difficulty arises from the variety of available options and their specific characteristics, the frequent desire for confidentiality from parents, financial barriers (in Switzerland, as in many countries, contraceptive methods are not reimbursed by health insurances even when used for menstrual management), and the need for consistent adherence, especially with short-acting reversible contraceptives.
In this study, we aimed 1) to translate and adapt the English version of LL-DCS into French and 2) to assess its psychometric properties among adolescents considering hormonal medications for contraception and menstrual management.
Methods
A translation, cross-cultural adaptation and validation study was conducted at the Pediatric and Adolescent Gynecology Consultation at a university hospital in Switzerland from June 2024 to January 2025, in 2 phases. Phase 1 involved translation and cross-cultural adaptation through forward and backward translations by professional translators, expert consensus to resolve discrepancies, and pilot testing with target users. Phase 2 consisted of scale validation through psychometric testing, evaluating its reliability and validity in adolescents using internal consistency, factor analysis, and tests of discriminant validity before and after clinical encounters.
Study Design and Setting
The Pediatric and Adolescent Gynecology (PAG) Consultation is a statewide pediatric tertiary clinic for children and adolescents up to the age of 20 years and is part of the Adolescent and Young Adult Outpatient Health Clinic. The PAG consultation consists of a multidisciplinary team with 2 gynecologists and 1 pediatrician, all of whom hold a specialty in PAG, and specialized nurses. Care is provided using a low-threshold approach, with the possibility of confidential consultations without parental knowledge, access to translators, and inclusivity for vulnerable populations, including migrants, individuals with disability, and those with gender diversity.
Participants
Adolescents aged 12 to 20 years who were postmenarchal and presented to the PAG Consultation for discussion regarding contraception and/or menstrual management were invited to participate. Exclusion criteria were inability to read and understand French.
Sample Size
In phase 1 (translation and cross-cultural adaptation), participants were recruited until saturation was reached, indicating that no further information could be obtained from conducting additional pilot testing.
There are currently no clear guidelines regarding sample size calculation for validation studies or factorial analysis. 15 We used the subject to item ratio approach and sample sizes from previous studies to determine our sample size.16,17 In phase 2 (validation), the aim was to recruit 8 to 9 participants per item (for a total of 10 items), considering that the English version had been tested among sample sizes ranging from 63 individuals 2 to 149 individuals 4 for preintervention testing and from 63 individuals 2 to 89 individuals 4 for postintervention testing. We recruited 85 participants, fulfilling our criteria.
Outcome Measures and Procedures
Phase 1
Translation and cross-cultural adaptation
Translation and cross-cultural adaptation were performed following the recommendations from the scale developers 3 and published guidelines. 18
The original LL-DCS was translated from English into French by 2 independent professional translators, whose mother language is French. One of the translators had a background in healthcare.
A multidisciplinary team of 5 health care professionals compared the 2 versions and resolved discrepancies through consensus during an in-person meeting. This team included 4 bilingual experts, with mother language being either French or English, with 3 of them being pediatric and adolescent gynecologists and 1 of them being an adolescent medicine specialist.
This reconciled version was then back-translated into English by 2 other independent professional translators, whose mother language is English. One of them had a background in health care. Both were completely blind to the original version of the LL-DCS.
A multidisciplinary team of health care professionals, including bilingual experts (as described above in step 2), compared the 2 back-translated versions with the original to ensure accuracy and proposed corrections to the French version during an in-person meeting.
A sample of patients from the target setting completed the translated LL-DCS and evaluated the clarity of the instructions, items, and response options using a checklist (“I find the instruction/question/response option easy to understand” or “I have trouble understanding the instruction/question/response option”). If an item was deemed unclear, participants were asked to suggest specific modifications to improve its clarity and comprehensibility. A minimum of 80% interrater agreement was required for validation.
Phase 2
Validation
We conducted psychometric testing of the final version in a sample of 85 adolescents participating in a pilot study evaluating an encounter decision aid for contraception and menstrual management. The encounter decision aid, developed by our team, was initially designed for adolescents with developmental disabilities (“Changeons les Règles!” 19 ) and later expanded to include all adolescents. Of the participants, 41 engaged in a clinical encounter in which the health care provider used the decision aid to discuss treatment options for contraception and menstrual management, whereas 44 participants had a consultation in which the health care provider discussed treatment options without the decision aid (“standard counseling”). Participants completed the French version of LL-DCS before (T0) and immediately after (T1) their clinical encounter. In addition, they completed a questionnaire collecting demographic (age, gender identity, education level, birth country, native language, and language most spoken at home) and clinical data (comorbidities and disabilities, reason for consultation, previous or current use of medication for contraception and/or menstrual management, initial and final preference of treatment). The question regarding treatment preference (asked at T0 and T1) was: “Which treatment would you like to start?” The multiple-choice options included hormonal and nonhormonal treatments for contraception and menstrual management as well as the responses “none” and “I am not sure.” No standardized assessment of participants’ health literacy was conducted.
Internal consistency was measured using Cronbach’s alpha, and values ≥0.70 were considered acceptable. Confirmatory factorial analyses (CFA) were performed to assess model fit using a χ2 test of model fit, root mean square error of approximation (RMSEA), the comparative fit index (CFI), the Tucker–Lewis Index (TLI), and the squared root mean square residual (SRMR). A combination of a statistically nonsignificant χ2 value (P > 0.05), CFI > 0.95, TLI > 0.95, RMSEA < (0.05–0.08), and SRMR < 0.08 indicated adequate model fit. Discriminant validity was evaluated by comparing scores at T0 and T1 using Brunner–Munzel tests (a robust, nonparametric method used to consider heteroscedasticity at T1), based on participants’ responses regarding treatment preference (“I am not sure” v. other options). “Participants undecided about treatment” were defined as those who answered “I am not sure” to the question, “Which treatment would you like to start?”“Participants decided about treatment” were those who selected one of the hormonal or nonhormonal options for contraception and menstrual management or the answer “none.”
Statistical analysis was performed using R software. 20
Ethics
Approval from the competent local ethics committee was waived because the study was deemed to fall outside of the scope of Swiss legislation regulating research on human subjects (Req-2024-01055). We obtained written informed consent from all participants.
Results
Phase 1: Translation and Cross-Cultural Adaptation
Steps 1 to 4 were carried out as described. Several differences were identified in step 2 (comparison of the 2 translated versions), affecting both the questions and the answer options. Ambiguities and discrepancies related to conceptual and semantic equivalence in 2 items from the Supported subscale were discussed and resolved by members of the multidisciplinary team. The response option je ne suis pas sûr-e (“I am not sure”) was selected to more accurately capture the meaning of the original term “unsure” than the alternative c’est dur à dire (“it is hard to tell”), which had been proposed by one of the translators. As a result of step 4 (comparison of the 2 back-translated versions), changes were made to questions 4 and 5 (Values Clarity subscale) so that the French translated version would best reflect the meaning of the original LL-DCS (supplementary material). Pilot testing of the prefinal version of the French LL-DCS was conducted among 14 participants. The mean age of the participants was 16.1 y (SD = 2.1 y), and their sociodemographic characteristics are detailed in the supplementary material. Several changes were suggested by participants, including partial reformulation of sentences to shorten and simplify them as well as word changes to improve comprehension (suffisamment [“sufficiently”] was replaced with assez [“enough”]). Based on feedback, changes were made to 3 (30%) of the questions (supplementary material). There were no changes to the answer options, as they were deemed clear to all participants.
Phase 2: Validation
Sample characteristics
The sociodemographics of the sample can be found in Table 1. One-third of the participants were born outside Switzerland, and nearly half reported a native language or a language most commonly spoken at home other than French. Most participants were enrolled in school (with approximately half attending upper secondary school), while about 10% had discontinued their education. The mean time to complete the French LL-DCS was 2.5 (±0.9) min.
Demographics of the 85 Adolescents Participating in Psychometric Testing of the Final Version of the French Low Literacy Decisional Conflict Scale (LL-DCS)

COCP, combined oral contraceptive pill; IM, intramuscular; LNG IUD, levonorgestrel intrauterine device.
The comorbidities section reports the number of conditions rather than the number of participants, as some participants had multiple comorbidities. Not all participants had comorbidities, which is why the total count of comorbidities (n = 41) does not equal the total number of participants (N = 85).
Before the clinical encounter (T0), more than half of the participants (n = 48/85, 56.5%) did not know which medication they would like to start (Table 1). After the clinical encounter (T1), only a minority was still unsure (n = 6/85, 7.1%). Combined oral contraceptive pill (n = 28/85, 32.9%), contraceptive patch (n = 11/85, 12.9%), and levonorgestrel intrauterine device (LNG-IUD; n = 11/85, 12.9%) were most commonly chosen following the consultation.
Psychometric validation of the French version of the LL-DCS
Internal consistency of the total scale was acceptable, with a Cronbach’s alpha of 0.72 at both T0 and T1. At the subscale level, the Uncertainty subscale showed good reliability (0.83 at T0 and 0.91 at T1), while the Informed, Values Clarity, and Supported subscales demonstrated lower reliability (Table 2).
Median (P25–P50) for the French Low-Literacy Decisional Conflict Scale (LL-DCS) Total and Subscales and Cronbach’s Alphas at T0 and T1

Model fit was adequate at T0, indicating good factor validity (Table 3). There was evidence for the original 4-factor model at T0, but exploratory analysis suggested a 3-factor solution with Informed and Values Clarity as one factor (Table 4 and Figure 1). The 4-factor model could not be identified at T1. Discriminant validity was strong, with the scale effectively distinguishing between adolescents who had made a choice about treatment options and those who remained undecided (Table 5).
Fit Indices from the Confirmatory Factor Analysis (4 Factors) a

CFI, comparative fit index; df, degrees of freedom; RMSEA, root mean square error of approximation; SRMR, squared root mean square residual; TLI, Tucker–Lewis index.
Interpretation: A combination of a statistically nonsignificant χ2 value (P > 0.05), CFI > 0.95, TLI > 0.95, RMSEA < (0.05–0.08), and SRMR < 0.08 indicates adequate model fit.
Loadings from the Exploratory Factor Analysis (with Oblique Rotation)
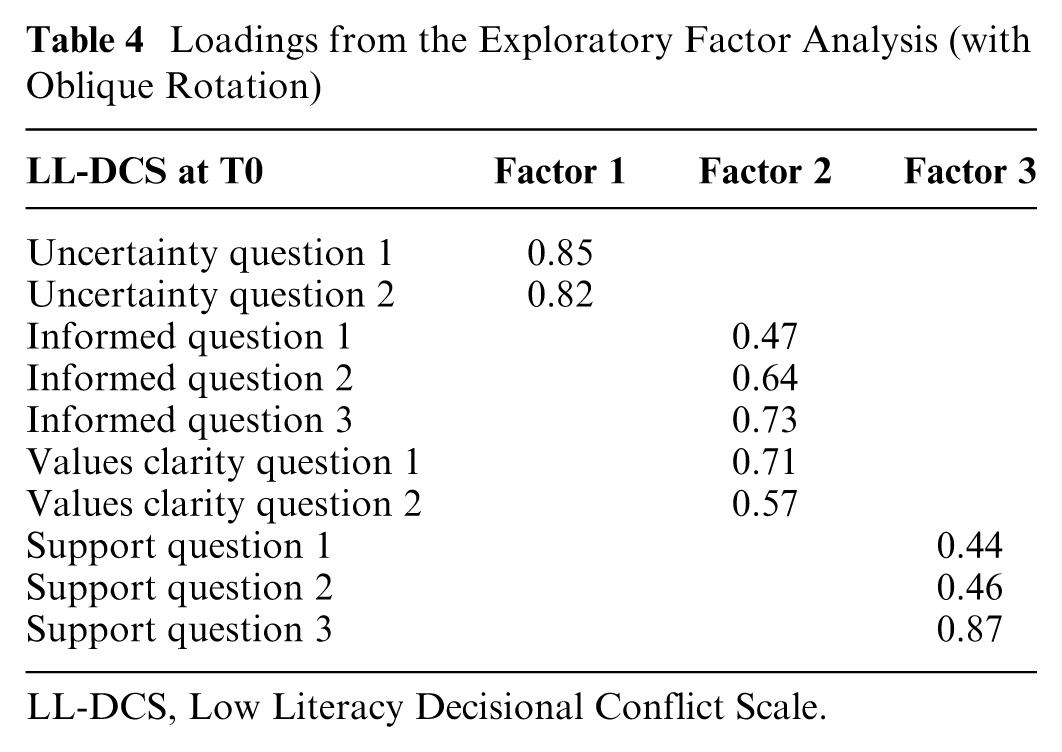
LL-DCS, Low Literacy Decisional Conflict Scale.

Exploratory factorial analysis (only T0).
Descriptive Statistics of Total DCS at T0 and T1: Median (P25–75) a
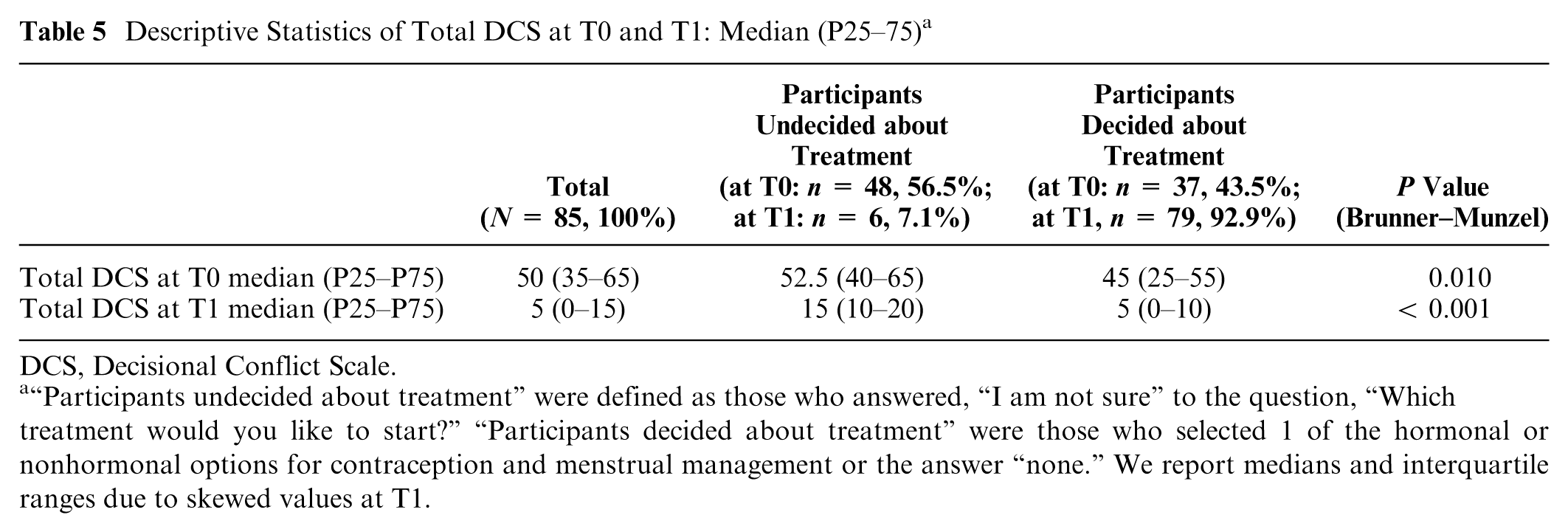
DCS, Decisional Conflict Scale.
“Participants undecided about treatment” were defined as those who answered, “I am not sure” to the question, “Which treatment would you like to start?”“Participants decided about treatment” were those who selected 1 of the hormonal or nonhormonal options for contraception and menstrual management or the answer “none.” We report medians and interquartile ranges due to skewed values at T1.
Discussion
In this study, we translated and adapted the English version of the LL-DCS into French and assessed the psychometric properties of the French LL-DCS in an adolescent sample.
The translation and cross-cultural adaptation process were carried out by key stakeholders, including professional translators, clinical experts, and adolescents who presented for consultation to discuss treatment options for menstrual management and contraception. Pilot testing of the prefinal version among adolescents identified 3 instances of unclear wording and sentence formulation (involving 3 items of the scale) and led to suggestions for changes to ensure clear and accurate understanding of the scale.
Regarding psychometric testing, we found that the French LL-DCS demonstrated overall acceptable internal consistency, although there was variability in reliability at the subscale level. The Supported subscale showed low internal consistency (a finding also observed in the English LL-DCS 4 ), suggesting that perceived support may not significantly contribute to the uncertainty about treatment options. Overall, the internal consistency of the individual subscales was lower than the original English version of the LL-DCS (0.83–0.86)2,4 but similar to the Spanish translated version (0.72). 2 This suggests that the total French LL-DCS score should be used, rather than individual subscales.
Factor validity was good. Similarly to the English version of the LL-DCS, 4 there was evidence for the original 4-factor model at T0, but exploratory analysis suggested a 3-factor solution, with Informed and Values Clarity combining into a single factor. This suggests that perceptions of being informed about available options and the values an individual attributes to those options may capture the same underlying concept. We could not identify the 4-factor model at T1, a finding that was also observed for the English version of the LL-DCS. 4 The observed differences in factorial structure between T0 and T1 may reflect both a meaningful clinical process (greater certainty after counseling) and reduced variance in decisional conflict following the encounter, particularly among participants who used the decision aid. At T0, greater variability allows the original 4-factor model to emerge. However, by T1, following the clinical encounter and counseling, participants tended to become more certain, reducing the variability in decisional conflict and limiting the ability to adequately investigate the factor structure. This temporal shift in factor structure likely reflects changes in how decisional conflict is expressed before versus after the clinical encounter. Clinically, this highlights the importance of considering the timing of scale administration when interpreting scores and emphasizes the value of repeated measurement for tracking changes in decisional conflict over time, while relying primarily on baseline data to evaluate structural properties of the instrument. The French LL-DCS also demonstrated strong discriminant validity by clearly distinguishing adolescents who had chosen a treatment option from those who had not yet decided.
The use of an encounter decision aid likely influenced both the level and variability of decisional conflict at T1. Decision aids are expected to reduce decisional conflict and restrict score variability, 21 making postencounter data less suitable for evaluating psychometric properties that rely on a broad distribution of responses. Accordingly, the most valid assessment of internal consistency, factor structure, and discriminant validity stems from baseline (T0), when decisional conflict was more variable and no participant had yet completed their clinical encounter. T1 findings are best interpreted as evidence of responsiveness rather than as indicators of structural validity. This interpretation is consistent with the validation of the original English version of the LL-DCS by Linder et al., 4 who similarly reported lower and less variable decisional conflict following clinical encounters.
Participants took an average of 2.5 (±0.9) min to complete the French version of the LL-DCS, making it a brief and easy-to-implement tool in clinical research. The mean completion time for the English and Spanish versions of the LL-DCS has not been reported, so we cannot make a direct comparison.
One of the limitations of this study is the relatively small sample size, which may limit the generalizability of the findings. Another limitation worth mentioning is the lack of assessment of individual health literacy. Nevertheless, although we cannot affirm that these adolescents had low health literacy, the sample population was very diverse: a third of them had an immigrant background, and just under half had a native language or spoke a language at home other than French. While most participants attended upper secondary institutions (in Switzerland, upper secondary education includes academic baccalaureate schools, specialized secondary schools, vocational schools, and commercial schools), nearly 10% had dropped out of school, and 3% attended a special needs school. This diversity in the linguistic and educational profiles of participants, which could be considered a strength, may also introduce response variance unrelated to the underlying construct of the scale. Finally, given that the validation was undertaken in an adolescent sample, the extent to which these findings can be generalized to other populations remains limited and should be interpreted with caution. Adolescents possess distinct cognitive and psychosocial characteristics that may not be representative of adult populations.
Future research should involve larger samples, incorporate standardized health literacy assessments, and undertake validation in diverse age and cultural groups to strengthen external validity.
The strengths of this study include adherence to the recommendations of the scale developers 3 and published guidelines 18 concerning the translation, cross-cultural adaptation, and validation of the French LL-DCS as well as the engagement of key stakeholders throughout these processes.
Ensuring the availability of validated scales adapted for individuals with limited literacy is crucial for fostering equitable participation in clinical research and for generating evidence that can better inform policy and practice. In the absence of such tools, vulnerable populations are at risk of being systematically excluded from research, contributing to the limited generalizability of the findings and the perpetuation of health disparities. 22 Adapting instruments to diverse literacy levels not only strengthens the quality and representativeness of research findings but also reflects an ethical commitment to inclusivity and justice in health research. 23
Conclusion
The French version of the LL-DCS demonstrates satisfactory psychometric properties, including internal consistency, factor validity, and discriminant validity. However, caution should be exercised when interpreting individual subscales, as their internal consistency was somewhat lower than the original English version of the LL-DCS. Further research is needed to assess how health literacy affects the factor structure of the French LL-DCS.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683261438738 – Supplemental material for Translation, Adaptation, and Validation of the French Version of the Low Literacy Decisional Conflict Scale in Adolescents
Supplemental material, sj-docx-1-mpp-10.1177_23814683261438738 for Translation, Adaptation, and Validation of the French Version of the Low Literacy Decisional Conflict Scale in Adolescents by Dehlia Moussaoui, Victoria L. Crofts, Nicolas Silvestrini, Thomas Agoritsas, Arnaud Merglen and Michal Yaron in MDM Policy & Practice
Footnotes
Acknowledgements
We acknowledge the “Plateforme de recherche PGO” at HUG, in particular Carole Grasset-Salomon. We acknowledge Mathilde Fontanet and Susan Pickford from the Faculty of Translation and Interpretating at the University of Geneva and Emeline Jamoul and Claire Hamer for their work on translating the scale.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided in part by a grant “cheffe de clinique scientifique” (Dehlia Moussaoui) from the University of Geneva, Geneva, Switzerland. Dehlia Moussaoui and Michal Yaron are supported by a grant from the “Fondation privée,” Geneva University Hospitals, Geneva, Switzerland. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Ethical Considerations
This study was considered as falling outside of the scope of the Swiss legislation regulating research on human subjects, so that the need for competent local ethics committee (CCER) approval was waived (Req-2024-01055).
Consent to Participate
Written informed consent was obtained from all participants. In the event of a participant capable of judgment but not of consent, their assent was collected in addition to the consent of their legal representative. In the event of a participant incapable of judgment, the consent of their legal representative was collected.
Consent for Publication
Not applicable.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
