Abstract
Highlights
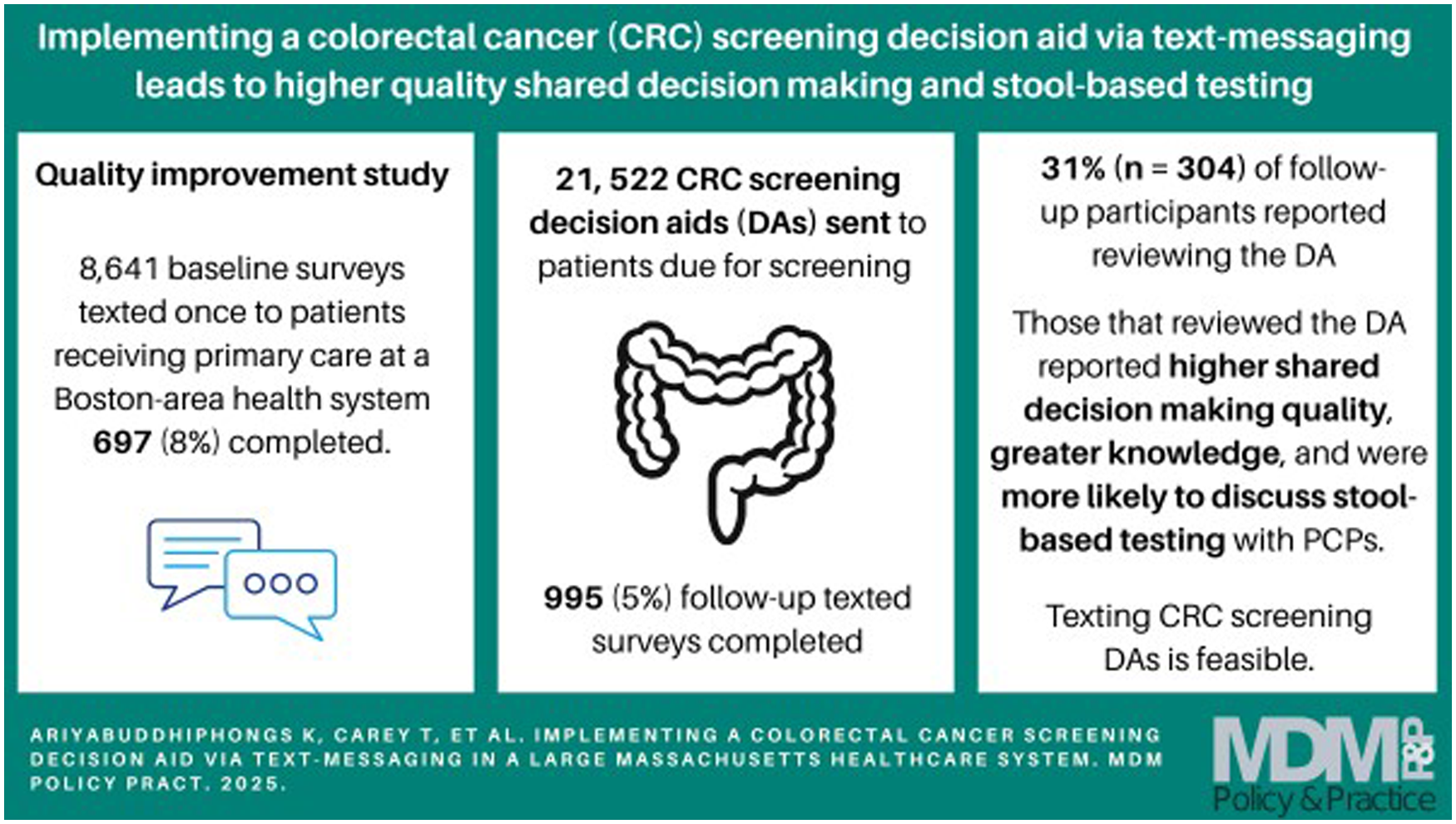
It is feasible for large health systems to automatize text messaging of colorectal cancer (CRC) screening decision aids (DAs) to patients due for CRC screening before a visit with their primary care practitioner.
Patients who review a texted CRC screening DA report higher shared decision-making quality and knowledge about CRC screening.
Use of CRC DAs may decrease screening via colonoscopy but not overall screening rates.
A more intensive intervention than text messaging is likely needed to increase the number of patients who review a CRC screening DA.
The United States Preventive Services Task Force (USPSTF) and other professional organizations recommend that adults aged 45 to 75 y undergo colorectal cancer (CRC) screening via stool-based tests or by direct visualization tests such as colonoscopy. To support shared decision making (SDM) around CRC screening, experts have developed decision aids (DAs; educational tools that provide evidence-based information about the benefits and risks of different medical interventions).1–4 Trials of CRC screening DAs have found that using DAs results in patients having greater knowledge of different CRC screening options, more engagement in decision making, and higher-quality screening decisions. 5
Despite these favorable effects, few health systems have implemented CRC screening DAs. Two primary care–based studies found it effective to mail CRC screening DAs to patients.6,7 However, the postage costs and staff effort to compile printed materials and to identify appropriate patients to receive DAs are barriers to adoption of this implementation strategy. Thus, additional research is needed to inform implementation of CRC screening DAs. Here, we report on a quality improvement (QI) project that aimed to improve SDM quality around CRC screening in a large Boston-area health system via implementation of a CRC screening DA.
Methods
We used the Theory of Change (ToC) QI framework to inform the development of a CRC screening SDM intervention in our health system. The ToC framework describes how a program brings about specific long-term outcomes through a logical sequence of intermediate outcomes. 8 We chose ToC over other QI methodologies because ToC is an intuitive framework for empowering stakeholders to achieve a straightforward goal in a short time period at low cost. As is common when using ToC, we used a backward mapping approach starting with our long-term outcome goal and then mapped the required process of change to reach this goal.
Our long-term goal was to increase SDM around CRC screening within our health system. Based on prior studies, we hypothesized that SDM would increase patient awareness of CRC screening options and their adherence to their screening choice.9,10 Our health system’s associate chief medical officer (K.A.) convened a working group to develop our intervention. The working group included physician leaders in internal medicine (n = 6), gastroenterology (n = 2), and SDM (n = 1); communication experts (n = 2); a Patient Family Advisory Committee leader; and a project manager. After several initial meetings, the group decided to focus on implementing a CRC screening DA to increase SDM. The group thought that implementing the DA prior to a primary care visit would achieve our goal of informed decision making while alleviating burden from primary care practitioners (PCPs). PCPs could then focus their CRC screening discussions on patient preferences rather than detailing the benefits and risks of different CRC screening options during already busy encounters.
At the time of this initiative, the 10 large-group practices in our health system used multiple different electronic medical records (EMRs) with various abilities to access data on CRC screening. Thus, the working group concluded that the most efficient way to implement a CRC screening DA would be via text (SMS) messaging through Arcadia. Arcadia is a cloud-based health care data platform that the health system uses for population health management. Since the health system runs several Arcadia text campaigns every month (e.g., texting patients overdue for mammograms) and uses Arcadia to report performance metrics to payers, the data are high quality. Also, prior studies have found that texting patients CRC screening reminders increases screening rates,11–13 and patients have found it acceptable to receive information about CRC screening via text, including those with low health literacy. 14 In addition, consistent with prior studies of DAs, patient and family advocate committee members (n = 6) reported that they would prefer to receive a DA in advance of a visit.15,16
Next, we aimed to identify a CRC screening DA to implement. We reviewed the literature17–21 and compendiums of DAs 22,23; however, we could not find a concise DA that included adults aged 45 to 49 y and discussed multitargeted stool DNA tests (mt-sDNA), an increasingly recommended CRC screening modality. 24 Therefore, we decided to iteratively develop our own CRC screening DA based on international DA standards, 25 the Ottawa Decision Support Framework, 26 and our experience developing and testing other DAs.27–31 In developing the DA, we iteratively asked our patient and family advocates over 3 different meetings to provide input on the usability and acceptability of the DA. We revised the DA based on their feedback to make the DA concise, clear, and readable on various types of smartphones. The health system’s marketing and compliance departments also reviewed and approved the DA before implementation.
The 2-page DA (available at https://www.bilhpn.org/patient-resources/patient-education/ and in the appendix) is written at a fifth-grade reading level, includes information on CRC, and presents the lifetime risk of dying from CRC with and without screening. 32 The DA compares procedures and outcomes of stool-only, mt-sDNA, and colonoscopy screening, the 3 most commonly used screening modalities, 33 and includes a values clarification exercise. It also includes 7 question prompts for discussing CRC screening with clinicians since question prompts also increase patient engagement, knowledge, and satisfaction with care.34,35 The DA was professionally translated into Chinese, Italian, French, Portuguese, Russian, Spanish, Creole, and Vietnamese before implementation. 36
Initially, we planned to roll out implementation of the DA slowly starting with our health system’s large academic practice. However, before DA implementation, leadership at a large community group (community-group1) in our health system, consisting of 43 primary care practices in the Greater Boston area, learned about the study and asked to participate. Two months later (after we had initiated the baseline survey but before we had implemented the DA), a second large community group (community-group2), consisting of 7 practices in northwest suburban Boston, also asked to be included in the study. None of the leaders of the other 7 large group practices in our health system asked to implement the DA, and we did not reach out to them to participate.
Baseline Data Collection
Before implementing the DA, we texted patients in our health system’s academic practice and at community-group1 an anonymous English-language survey (available in the appendix) to learn patients’ perceptions of SDM around CRC screening. The survey, which included 18 questions, began by asking patients if they had discussed CRC screening with their health care providers in the past 2 y. If so, patients were then asked the 4-item SDM Process Scale, a measure of SDM quality endorsed by the National Quality Forum (total scores range from 0 [low quality] to 4 [high quality]).37,38 The scale assesses whether patients perceive that their clinician asked them if they 1) wanted to be screened for CRC, 2) how much their clinician talked about the reasons they may want to have CRC screening and 3) may not want to have CRC screening, and 4) whether their clinician asked which type of screening test they wanted. The survey also asked patients their likelihood (5-point scale) of being screened for CRC in the future and 2 knowledge questions (the age at which a person at average risk should start screening and which screening tests [stool blood test, stool DNA test, colonoscopy, or all of the above] are recommended for CRC screening). Finally, the survey assessed participants’ age group, gender, race/ethnicity, educational attainment, and PCP’s name. We used PCP name to map patient responses to a study site.
To test the feasibility of texting patients study surveys, we initially texted the survey only to patients aged 50 to 55 y who were not current with CRC screening at the academic practice (on October 18, 2021). National performance metrics (i.e., HEDIS criteria) at that time focused on adults 50 to 75 y, even though the USPSTF CRC screening guidelines expanded to include adults 45 to 49 y in May 2021. In addition, our Arcadia data showed that adults aged 50 to 55 y were the age group between ages 50 and 75 y least likely to screen for CRC. Patients did not have to opt in for texting, but if they had previously opted out of text messages, they were not included. Patients also had the ability to opt out once texted by this program. We found that >60% of the initial texted study surveys transmitted successfully. Thus, 8 days later, we broadened the sample population to include patients aged 56 to 75 y not current with CRC screening at the academic practice and to patients aged 50 to 75 y not current with CRC screening at community-group1. Three weeks later, we expanded the sample population further and texted the survey to all patients aged 45 to 49 y and to all patients aged 50 to 75 y current with CRC screening at the academic practice and at community-group1. Since Arcadia had the ability to text only in English at that time, surveys were texted in English even though the DA itself could be texted in the patient’s preferred language. We texted patients the survey one time. We provided no incentive to participate.
DA Implementation
In January 2022, through automation, we began texting the DA to patients in the academic practice due for CRC screening, who had an annual or follow-up appointment scheduled with their PCP (physician, nurse practitioner, or physician assistant) in the next month, did not have clinical or claims data indicating CRC history, and had phone numbers in Arcadia. While our health system’s data integration team is constantly doing data control on Arcadia, if there was a gap in receiving claims or clinical data then a patient with CRC or who had been screened prior to establishing care in our health system could have received a DA.
One month later, in February 2022, we began texting the DA to patients at community-group1, and in March 2022, we began texting the DA to patients at community-group2. We considered patients due for CRC screening if they had not had a colonoscopy in the past 10 y, an mt-sDNA test in the past 3 y, or a stool test in the past year. Overall, the DA was texted to 21,522 patients not current with CRC screening. We also made the DA available on the health system’s Web site. We informed PCPs via e-mail and during faculty meetings about the DA. In a grand rounds presentation at the academic practice, we described SDM around CRC screening and showed PCPs the DA. PCPs who attended the presentation received continuing medical education credit. We also emailed PCPs at the community groups a video link to this grand rounds presentation. No data were collected on how many PCPs reviewed the training. The Beth Israel Deaconess Medical Center’s Institutional Review Board determined this QI project to be human subjects exempt.
Data Collection
To learn patients’ perceptions of SDM around CRC screening after implementing the DA, in August 2022 and in May 2023 (follow-up), we texted a study survey to patients who had been texted the DA in prior months. The follow-up surveys included the same questions as the baseline survey but also asked patients if they reviewed the DA. Patients were texted this survey in English, once and no incentive was provided.
In addition, we obtained data on CRC screening rates seen at all practices in the health system in 2021 and 2022. These summary data were available only for adults aged 50 to 75 y since the health system pulled data relevant to national quality measurements and quality measures for CRC screening included only adults aged 50 to 75 y during the study period. 39 In addition, we obtained data on which screening test patients in the health system received between January 2022 and May 2023. No other CRC screening interventions were initiated across the health system during this study; however, the year before the DA was implemented, many practices began sending patients due for CRC screening a mt-sDNA test (the academic practice) or a fecal immunohistochemical test (FIT; sent by most of the community practices).
Analyses
All analyses were completed using SAS v9.4. We used chi-square statistics to compare the characteristics of patients who completed baseline versus follow-up. We used multivariable linear regression to examine the mean differences in SDM-process scores between baseline and follow-up surveys, adjusting for patient age, gender, race/ethnicity, educational attainment, and site. In the post hoc analyses, we used a t test to examine the effect of having a gender-concordant PCP on SDM process scores. We used 2-by-2 contingency tables to compare follow-up participants’ likelihood of being screened for CRC in the future (likely v. neutral/unlikely), knowledge about age of CRC screening initiation and which tests are recommended for screening with baseline, and to examine which test patients reported receiving most recently. We used chi-square statistics to compare sociodemographic characteristics between participants missing data on these outcomes versus those with complete data. We repeated our analyses, limiting the follow-up participants to only those who reported reviewing the DA. In sensitivity analyses, we limited our sample to patients from the academic practice and community-group1 since no community-group2 patients completed baseline. Finally, we used chi-square tests to compare CRC screening rates and type of test received by patients in the 3 participating large-group practices compared with the 7 nonparticipating group practices (a mix of small and large primary care practices in eastern Massachusetts). We followed SQUIRE guidelines in reporting this QI study.
Results
Sample Characteristics
eTable 1 displays participant sample flow. We sent the baseline survey to 15,616 patients, of which 55% (n = 8,641) were received by a smartphone that accepted texts (i.e., no kickback error message received). Overall, 91.5% (21,523/23,517) of follow-up survey texts were transmitted successfully; we did not retext patients if the survey 1 text failed. Overall, of the 30,163 survey texts transmitted successfully, 600 (2.0%) patients sent a “STOP” response to stop receiving texts. Of the remaining 29,563 patients, 1,692 (5.7%) completed at least part of a survey (n = 697 baseline, n = 995 follow-up). Of these 1,692 participants, 45.3% were ages 60 to 75 y; 8.5% were non-Hispanic Black, 5.0% were non-Hispanic Asian, and 4.9% were Hispanic; 57.2% were female; 16.0% had a high-school degree or lower educational attainment; and 75.3% had previously received CRC screening (Table 1). Of the 87.9% of participants who reported their PCP’s name (allowing mapping of a participant to a practice), 84.2% were seen at a community practice. Women were more likely to have a gender-concordant PCP than men were (78.0% v. 64.5%, P < 0.001).
Participant Characteristics for Baseline and Follow-up Surveys

CRC, colorectal cancer; DA, decision aid.
Site was determined by mapping the patient’s response for primary care provider (PCP) name. If the PCP’s name was not reported, we could not identify the site where the patient was seen.
P < 0.05 for comparison with baseline survey; **P < 0.001 for comparison with baseline survey.
We did not collect sociodemographic data on nonresponders. However, study participants were of similar gender to patients overall in these practices but were more likely to be White and less likely to be Hispanic and aged 60 to 75 y (eTable2). Compared with baseline participants, follow-up survey participants were more likely to be women, aged 45 to 49 y, and less likely to be non-Hispanic White, highly educated, or to have ever been screened (Table 1). eTable3 demonstrates the characteristics of participants who completed a study survey but were missing data on an outcome of interest.
At follow-up, 30.6% of participants reported reviewing the DA. Compared with baseline participants, follow-up participants who reviewed the DA were older, less likely to be non-Hispanic White, and less likely to have completed graduate school. Compared with follow-up participants who did not report reviewing the DA, patients who reported reviewing the DA were more likely to be aged 70 to 75 y (eTable 4).
Survey Results
Compared with baseline participants, follow-up participants were more likely to know that CRC screening is recommended for adults aged 45 to 75 y and to have had a stool test for their most recent screening test, to report that their health care professional discussed mt-sDNA tests, and to have lower intentions to screen in the future (Table 2). Follow-up participants who reported reviewing the DA reported higher SDM process scores than baseline participants did. They were also more likely to know that guidelines recommend CRC screening for adults aged 45 to 75 y and that multiple CRC screening tests are recommended. Participants who reviewed the DA were more likely to have discussed CRC screening with their health care professional in the past 2 y, to have had a stool test for their most recent screening test, to report that their health care professional discussed mt-sDNA tests, and to have lower screening intentions (Table 2). Regardless of whether or not follow-up participants reported reviewing the DA, women had lower SDM-process scores than men did; SDM-process scores were not associated with having a gender-concordant PCP. When we excluded patients from community-group2 from the analyses, findings were similar (eTables 5/6).
CRC Screening across the Health System
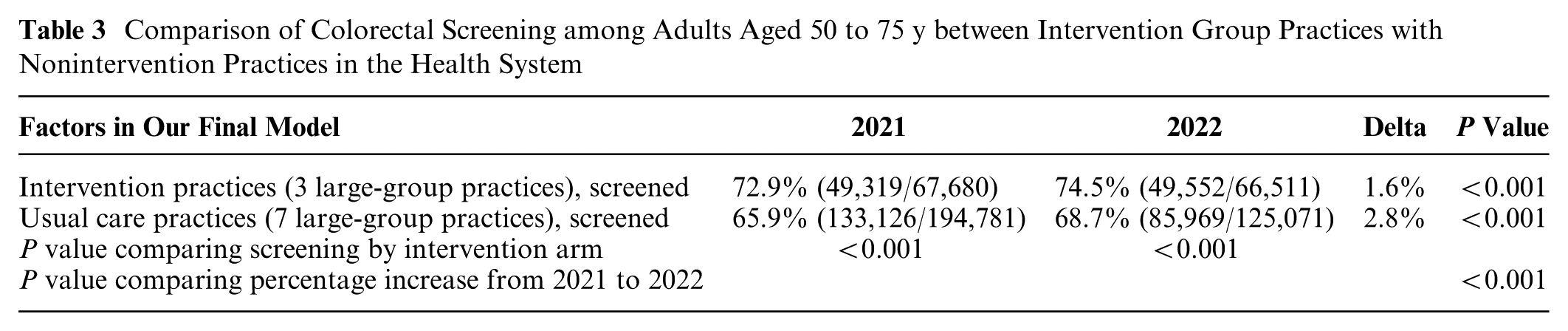
In 2021, before DA implementation, 72.9% (49,319/67,680) of adults aged 50 to 75 y seen at implementation sites were current with CRC screening compared with 65.9% (133,126/194,781) of patients at nonintervention sites (P < 0.001). After DA implementation, screening increased across the health system but less so in intervention sites (Table 3). Patients seen at intervention sites (60.1%) were also less likely to receive a colonoscopy (62.3%) than those seen at nonintervention sites (P < 0.001; Table 4).
Comparison of the Effects of the Colorectal Cancer Screening Decision Aid between Follow-up Participants and Baseline Participants

CI, confidence interval; CRC, colorectal cancer; DA, decision aid; RR, relative risk; SDM, shared decision making.
For “knew screening is recommended for adults 45–75,” 97% of baseline participants responded and 97% of follow-up participants (99% of participants who reviewed the DA responded); “most recent screening test,” 88% of baseline participants responded and 64% of follow-up participants overall (68% of participants who reviewed the DA responded); for “discussed CRC screening in the past 2 years,” 89% of baseline participants responded and 87% of follow-up participants overall (98% of participants who reviewed the DA responded); for “Shared Decision-Making Process Scale,” 95% of baseline participants responded and 94% of follow-up participants overall (95% of participants who reviewed the DA responded); for likelihood of being “screened in the future,” 99% of baseline participants participated and 99% of follow-up participants (99% of participants who reviewed the DA responded).
The Shared Decision-Making (SDM) Process Scale and screening intentions were asked only of patients who said they discussed colorectal cancer screening in the past 2 y with their health care professional.
Comparison of Colorectal Screening among Adults Aged 50 to 75 y between Intervention Group Practices with Nonintervention Practices in the Health System
Comparison of Colorectal Cancer Screening Modality Received from January 2022 to May 2023 among Adults Aged 50 to 75 y Seen at Intervention Group Practices and Nonintervention Practices in the Health System a

These data were available only for adults aged 50 to 75 y since our health system pulls only data relevant to national quality measurements and during the study quality measures for colorectal screening of only included adults aged 50 to 75 y.
Discussion
In this QI study, 21,522 patients not current with CRC screening were successfully texted a novel CRC screening DA via automation. Five percent of these patients completed a study survey, of whom 31% reported reviewing the DA. Although a 5% response rate is low, it is consistent with other studies that texted surveys and did not provide incentives to participate. For example, an Australian study sent a text message to 8,333 patients asking them if they would agree to complete a study survey; 8.2% (702) texted yes, but only 431 (5.2%) commenced the survey. 40 Participants in our study who reported reviewing the DA reported higher SDM quality and greater knowledge of CRC screening test options. Health system data suggest that patients seen at intervention sites were less likely to choose colonoscopy screening. However, the DA’s effects on the overall uptake of CRC screening is uncertain since screening went up across the health system between 2021 and 2022 but more so in nonintervention sites. In summary, we found that it was feasible to implement a CRC screening DA in a large health system via text messaging; however, a more intensive intervention may be needed to have greater numbers of patients read the DA.
Prior studies have found that automatic delivery of CRC screening DAs through postal mailings or patient portals results in high numbers of patients receiving DAs.15,41 However, automated delivery often leads to the low use of DAs; 8% to 40% of patients mailed a DA in prior studies reported reading the DA, which is similar to the 31% of survey respondents who reported reviewing the DA in our study.7,14,15,41 Studies have also found that CRC screening DAs have little effect on CRC screening rates,6,19–21,42–44 unless they are combined with patient navigators or methods for patients to order a stool-based screening test immediately after reading a DA.6,20,42,43,45,46 Although follow-up participants were less likely to intend to be screened in the future than baseline participants were, the DA and thus follow-up surveys were sent only to patients due for CRC screening. Baseline participants included those current with screening who may have been more accepting of screening in general. However, prior DA studies have shown that once patients become more aware of the harms of medical interventions through a DA, they tend to choose less invasive and aggressive care. 10
Participants who reviewed the DA were more likely to report that their most recent screening test was a stool-based test. These findings suggest that DA use may have shifted some patients from colonoscopy-based screening to stool-based screening. On a population level, no CRC screening strategy has been shown to be more cost-effective,47,48 since individuals who choose stool-based testing may not adhere to follow-up colonoscopy after a positive test. However, from a health system perspective, mailing FIT tests results in a lower 10-y average per-person cost of screening but with lower screening compliance and fewer cancers detected than colonoscopy screening. 49 In a recent study accounting for real-world adherence to different stool-based screening strategies, mt-sDNA screening was found to be more cost-effective than FIT or fecal occult blood testing screening. 48 Since no cancer screening strategy is consistently more cost-effective than another, an approach to screening that focuses on SDM may be the most patient centered.
Consistent with systematic reviews of DA trials, 5 we found that patients who reported reviewing our DA reported higher SDM quality, which we measured using the SDM process scale. The version of the SDM process scale that we used included a question on whether clinicians had talked about reasons not to be screened. 50 However, some investigators specifically ask how much clinicians talked about the reasons a patient might not want to have a colonoscopy to screen for CRC. 51 Since we found no difference between baseline and follow-up participants in their response to the question that we used, we would consider using the colonoscopy-specific version of the question in the future. We also found that men reported higher SDM-process scores than women did, regardless of PCP–patient gender concordance or use of the DA. It is possible that since women have a lower CRC incidence and mortality than men do, PCPs may be less likely to engage women in SDM around CRC screening. 52 However, in a systematic review of characteristics associated with SDM, most studies found no effect of sex. 53 Consistent with our findings, in a meta-analysis of randomized trials, DAs demonstrated similar efficacy regardless of patient–clinician gender concordance. 54
We initially planned to implement an existing CRC screening DA; however, we could not find a DA that included information for adults aged 45 to 49 y. This is likely because the American Cancer Society began recommending CRC screening for adults aged 45 to 49 y at average risk only in 2018 and the US Preventive Services Task Force in 2021. However, as of 2021, only 19.7% of US adults aged 45 to 49 y reported being up to date with CRC screening. 55 It is important to find ways to inform adults aged 45 to 49 y about CRC screening. However, in our study, younger adults were less likely to review our texted DA, suggesting additional interventions are needed.
This study has limitations. We texted our survey only in English; however, 95% of patients aged 45 to 75 y at the intervention sites reported that English was their preferred language. Only 5% of patients completed a study survey, and participants were younger and more likely to be non-Hispanic White than patients aged 45 to 75 y seen in these practices, which may reflect the fact that our DA was texted only to patients with smartphones. Because study surveys were anonymous, it is possible that some patients completed more than 1 survey. While we texted DAs to patients with an upcoming visit with their PCP, the visit may not have been scheduled to address preventive health. We did not collect information on whether patients had recently changed PCPs; however, 88% were able to report their PCP’s name, suggesting a relationship. Patients may have received and completed a study survey months after they received the DA, which could affect recall and participation rates. Despite these limitations, our findings are consistent with tightly controlled randomized trials that show that DAs improve patient knowledge and SDM quality. 5 In addition, we did not interview PCPs, and we do not have data on how many PCPs watched the grand-rounds talk on SDM around CRC screening. Finally, our study was conducted during the COVID-19 pandemic, and CRC screening increased nationally during the COVID-19 pandemic mainly because of increased use of stool-based testing.56,57 However, by 2022, colonoscopies were increasingly available.
We considered our intervention successful in that it led to the development of a brief CRC screening DA that increased patient knowledge about CRC screening and was acceptable to patient and family advocates and health system leadership. As a result, we made the DA widely available on our health system’s patient educational resources page. We also found that text messaging was a useful low-resource method of delivering DAs to a large number of patients. However, our health system now uses 1 electronic medical record systemwide, and in the future, we would use this electronic medical record to deliver DAs, possibly by linking the DA to a CRC screening prompt or to an annual wellness visit for patients due or overdue for CRC screening.
Supplemental Material
sj-pdf-1-mpp-10.1177_23814683261425607 – Supplemental material for Implementing a Colorectal Cancer Screening Decision Aid via Text Messaging in a Large Massachusetts Health Care System
Supplemental material, sj-pdf-1-mpp-10.1177_23814683261425607 for Implementing a Colorectal Cancer Screening Decision Aid via Text Messaging in a Large Massachusetts Health Care System by Kim Ariyabuddhiphongs, Timothy Carey, Emily A. Wolfson, Spencer Rittner, Jonathan Li, Maëlys Amat and Mara A. Schonberg in MDM Policy & Practice
Footnotes
Acknowledgements
None.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Schonberg receives royalty payments from UpToDate for reviewing 2 pages on prevention. Dr. Jonathan Li receives funding for Eli Lilly unrelated to colon cancer screening for quality improvement projects. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided NIH/NIA K24 AG071906. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Ethical Considerations
This study was deemed quality improvement by the Beth Israel Deaconess Medical Center’s Committee on Clinical Investigations.
Consent to Participate
The requirement for informed consent was waved by the Beth Israel Deaconess Medical Center’s Committee on Clinical Investigations.
Consent for Publication
Not applicable.
Data Availability
Data were entered into a REDCap database. To maintain participant confidentiality, the data are not publicly available but are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
