Abstract
Highlights
Adolescents and young adults with newly diagnosed cancer, and their caregivers, endorsed that decision support is important for their understanding of cancer and treatment and for enhanced communication with their health care teams.
Implementation of a decision support intervention shortly after cancer diagnosis is acceptable, feasible, and usable for adolescents and young adults and their caregivers.
Oncology clinicians confirm the usability of decision support interventions for their adolescent and young adult patients at diagnosis.
Access to a decision support intervention at diagnosis may improve the involvement of adolescents and young adults in their treatment-related decision making and decision processes.
Future studies can continue to refine the content and delivery of decision support interventions close to the time of diagnosis, including value of a coach and the role of the caregiver, and targeted to a range of treatment-related decisions.
Keywords
Approximately 85,890 adolescents and young adults (AYA) ages 15 to 39 y were diagnosed with cancer in the United States in 2023, comprising 4.4% of all new cancers across all age groups. 1 Mortality rates are falling for AYA, with a nearly 86% 5-y relative survival rate comparable with that of children. However, the incidence of cancer is increasing, 1 and outcomes vary based on sociodemographic characteristics, access to comprehensive care, 2 and access to clinical trials.3–5 The National Cancer Institute has undertaken initiatives to increase AYA’s access to clinical trials, yet AYA remain less likely to be treated on trials than pediatric patients are, trial enrollment differs by diagnosis, and minoritized groups are less likely to enroll when offered trial entry.2–5 Furthermore, AYA experience reduced well-being during and after cancer treatment due in part to substantial challenges to achieving developmental milestones, including independence and autonomy, and complicated by inconsistent involvement across treatment-related decisions.6–11 Treatment-related decision making begins with initial decisions at diagnosis (e.g., selecting a treatment protocol or enrolling in clinical trial) and evolves to include ongoing decisions throughout treatment (e.g., adjustments to treatment protocols, side effect management, and lifestyle changes). The range of treatment-related decisions that AYA encounter carry both short- and long-term consequences for their health and well-being. 10 As such, AYA may benefit from developmentally consistent decision support interventions that increase their involvement in treatment-related decision making and enhance their decision-making experiences (i.e., decision processes) over the course of their care.12,13
AYA are often overshadowed in treatment-related decision making by their caregivers, despite their developmental ability and legal authority (at ≥18 y).7,14 A systematic review of 14 studies, as well as our research, confirms that AYA involvement in treatment-related decision making at diagnosis and throughout treatment is more limited than AYA prefer.11,15–17 Due to health status, decision-making time frame, and emotional maturity, AYA often rely on caregivers to gather and share information in an understandable format. 11 When AYA believe they lack comprehensive information, are not fully involved in decision making, or their personal values misalign with those of their caregivers, they may experience decisional conflict.16,18 AYA involvement in treatment-related decision making is particularly important when there are multiple options to consider and when decisions involve tradeoffs between AYA values, such as more aggressive treatment protocols that offer higher cure rates alongside considerable side effects that reduce quality of life. 8 Decisions that align with values are associated with increased satisfaction and self-efficacy and decreased conflict and regret, highlighting the importance of actively engaging AYA in decision making with caregivers and clinicians to promote well-being.16,18,19–21
Decision support interventions that increase knowledge and align values with decisions have been shown to be efficacious in improving engagement in decision making and decision processes (or perceptions of the decision-making experience) in adult oncology.13,19,21 These interventions provide information on options, risks/benefits, and values clarification and can be self-administered, practitioner administered, and/or coach assisted. 22 Decision coaching increases knowledge and participation in decision making and enhances engagement in adult decision aids and other AYA digital interventions; however, its positive impact on decision processes remains uncertain, and the benefit of coaching for AYA decision making has not been evaluated.22,23 Decision support interventions have not yet been tested for AYA immediately following cancer diagnosis despite their potential to address AYA cancer knowledge gaps while providing structure and support for AYA participation in treatment-related decision making and communication with their health care team.13,19,20 A pilot study in Australia that assessed a cancer clinical trial decision aid (DELTA) indicated its benefit and relevance, but this study was conducted with parents of adolescents >1 y after trial enrollment. 24 In another pilot study that tested the clinical usefulness of a conjoint analysis–based decision making and communication tool (MyPref) for AYA with advanced cancer, caregivers and clinicians reported the utility and accuracy of values clarification. 25
The current study is highly innovative as it uses best practice, mixed methods to pilot a rigorously developed, theoretically informed, developmentally consistent Web-based decision support intervention (DECIDES) for AYA newly diagnosed with cancer and their caregivers in the context of real-time treatment-related decision making.26–28 DECIDES was primarily designed to increase AYA involvement in treatment-related decision making and to enhance their decision processes while acknowledging the role of caregivers in supporting this process. Components include cancer, treatment, and clinical trials information (to increase knowledge), an interactive fact-or-fiction quiz (to address attitudes to clinical trials), a values clarification exercise vis-à-vis treatment (to identify and align AYA values with treatment choices), guidance on communication between AYA, caregivers, and clinicians (to promote shared decision making), and inclusion of AYA cancer-specific resources (to connect AYA to supplemental support). The primary objectives of this study were to 1) examine the feasibility, acceptability, and usability of DECIDES for treatment-related decision making and 2) to evaluate the utility of coach-assisted support for increasing AYA engagement with DECIDES given evidence for coaching in adult decision aids and for other AYA digital interventions. The secondary objective was to explore differences in knowledge, decision-making involvement, and decision processes among AYA and caregivers who accessed DECIDES (with or without a coach) for treatment-related decision making at diagnosis in contrast to a usual care group. Acceptable, scalable interventions to increase AYA involvement in treatment-related decision making, enhance communication about cancer and treatment (including clinical trials), clarify values, and improve decision-making processes are critical to efforts to address disparities in AYA cancer and psychosocial outcomes.3,18,29
Methods
This pilot randomized controlled trial used a mixed-methods, convergent, quantitative + qualitative study design to test DECIDES for treatment-related decision making. Data were collected between September 2020 and March 2023. This study was approved by the Institutional Review Board (IRB No. 20-017521) and registered on ClinicalTrials.gov (NCT ID No. NCT06191679).
Participants
AYA, aged 15 to 24 y, with a new or relapsed cancer diagnosis (leukemia/lymphoma, extracranial solid tumor, or brain tumor) and their caregivers (parents/legal guardians) were recruited from Children’s Hospital of Philadelphia in the United States. In collaboration with pediatric oncology colleagues, we identified an optimal recruitment window (within 6 wk of the initial diagnostic/treatment planning meeting) to balance feasible enrollment with timely decision support for treatment-related decisions. AYA (and their caregivers) were eligible if they could read/speak English and had no preexisting cognitive deficits. Potential participants were identified through outpatient appointment and inpatient unit lists, and oncology clinicians were consulted for study appropriateness. To enroll, consent and/or assent was obtained during clinic visits or via phone. Parent/legal guardian consent and AYA assent were required for AYA <18 y. AYA ≥18 y and up to 2 caregivers (primary and second) provided consent for themselves.
Of 312 AYA identified, 194 (62%) were ineligible because they were >6 wk after the initial diagnostic/treatment planning meeting (n = 152), did not speak English (n = 27), or had preexisting cognitive deficits (n = 15) (Figure 1). AYA (n = 5) with significant psychosocial distress or a deteriorating medical condition were not approached. The study team attempted to contact 113 (36%) eligible AYA. Of the 86 (76%) AYA reached, 29 (34%) declined participation and 57 (66%) enrolled along with 47 caregivers (45 = primary, 2 = second). Thirty-six oncology clinicians of enrolled AYA were identified to participate in a qualitative interview. Of the 19 (53%) approached for recruitment, 6 (32%) were not reached, 13 (68%) consented to participate, and the remaining 17 (47%) were not approached due to data saturation.
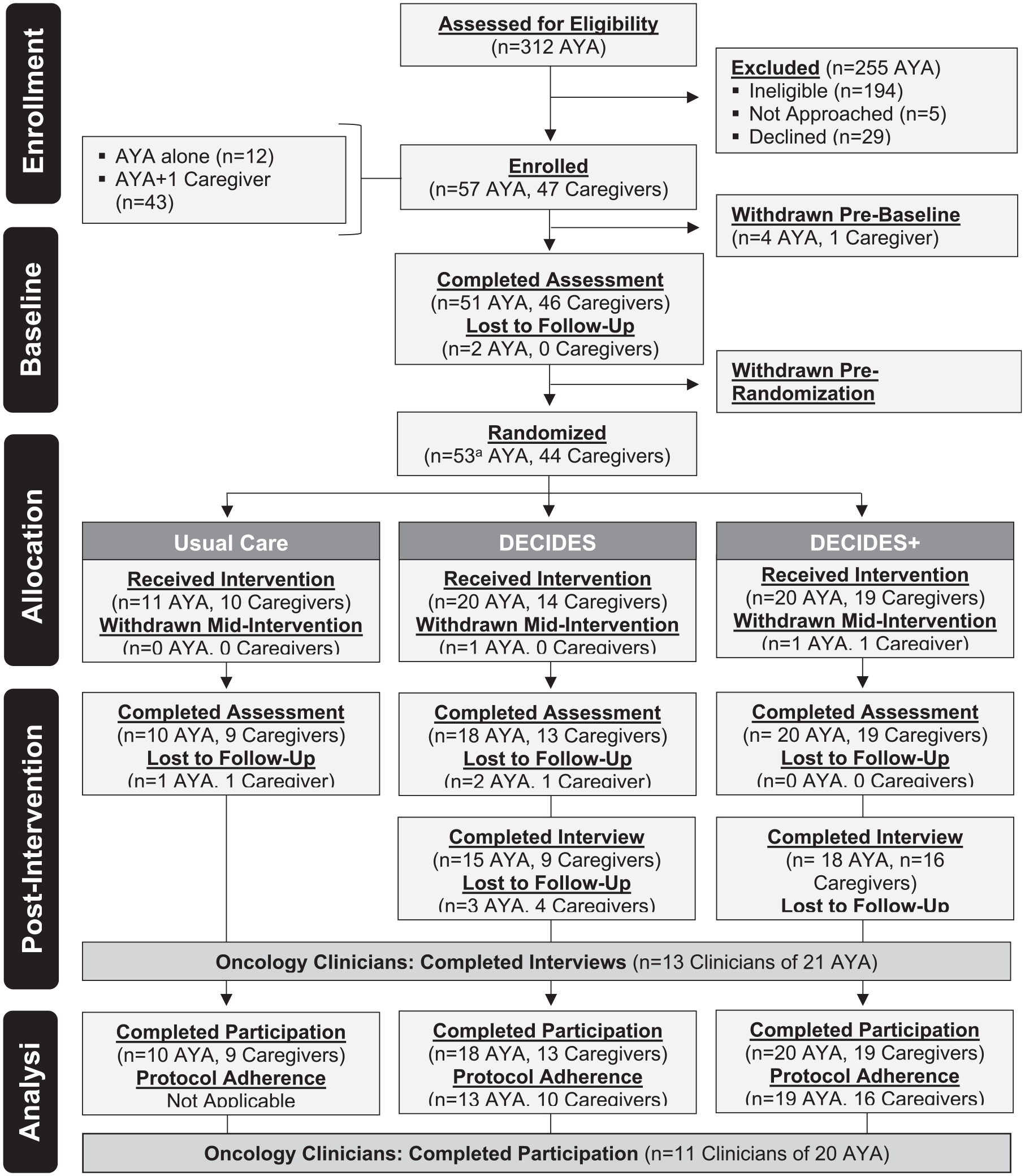
Consort.
Procedures
Fifty-one AYA (89%) and 46 (98%) caregivers completed a baseline assessment. Fifty-three AYA (including 2 with missing baseline data) and 44 caregivers were then randomized in a 1:2:2 ratio (stratified by AYA age <18 and ≥18 y) to usual care (n = 11 AYA, n = 10 caregivers), DECIDES (n = 21 AYA, n = 14 caregivers), or DECIDES+ (n = 21 AYA, n = 20 caregivers). The usual care group continued to access their health care team for treatment-related decision-making support, whereas AYA and caregivers in both DECIDES groups received a unique login ID to access DECIDES together or separately in addition to usual care. The DECIDES group also received a handout with an overview of DECIDES and instructions to complete the values clarification exercise. The DECIDES+ group had access to a coach (i.e., study team member) who reviewed DECIDES, facilitated the values clarification exercise with AYA, and provided ongoing support as requested. Values clarification exercise results were provided to AYAs’ oncology clinicians. Four weeks after randomization, participants in both the DECIDES and DECIDES+ groups received a text/e-mail reminder to prompt AYA access to the website for engagement with DECIDES. Eight weeks after randomization, 48 (84%) AYA and 41 (87%) caregivers completed a postintervention assessment. Qualitative interviews were conducted with 33 (79%) AYA and 25 (74%) caregivers in the DECIDES groups and 13 (100%) oncology clinicians. AYA and caregivers received $50 to $70 for completed assessments and interviews; oncology clinicians were not compensated. Assessment data were collected via Research Electronic Data Capture (REDCap). 30 Interviews were recorded in Microsoft Teams and transcribed by research assistants (A.A. and N.R.).
Decision Support Intervention Description
We followed the Informed Medical Decisions Foundation procedures20,31,32 to develop DECIDES. First, we developed content based on 1) a review of AYA cancer literature on recommendations for AYA decision support 33 and decision aid development guidelines in adult oncology,20,31,32 (2) results of our studies of AYA decision making and the Pediatric Research Participation Questionnaire of attitudes to clinical trials,11,34–36 and 3) central concepts for individual factors that promote health behavior change proposed by the Health Belief Model (perceived severity, perceived barriers, perceived benefits, cue to action, self-efficacy) and Theory of Reasoned Action and Planned Behavior (attitudes toward behavior, subjective norms, behavioral intention, perceived behavioral control).37,38 Next, we outlined content in storyboard format and obtained input from an AYA patient (n = 9, 15–19 y at diagnosis, 16–24 y at interview, most with leukemia/lymphoma diagnoses) and caregiver (n = 8) Steering Committee, and a Scientific Advisory Committee (n = 6 multidisciplinary experts in pediatric oncology, and cancer decision making). Informed by these steps, DECIDES was revised in an iterative process to produce multimodal, graphically interesting content written for low health literacy. To promote accessibility and appeal to AYA preferences for digital tools, 39 DECIDES was packaged as a Web-based tool compatible with both desktop and mobile devices. Components include cancer, treatment, and clinical trials information; an interactive fact-or-fiction quiz; a values clarification exercise vis-à-vis treatment; guidance on communication between AYA, caregivers, and clinicians; and AYA cancer-specific resources (Supplemental Figure 1, Supplemental Table 3).
Measures
Participant characteristics
Demographics were collected from AYA and caregivers at baseline. AYA medical information was extracted from the electronic health record (EHR).
Feasibility, acceptability, and usability
Feasibility was evaluated with participant retention and attrition, protocol adherence, and intervention/coaching engagement. Participant retention percentage study completion) and attrition (percentage withdrawn or lost to follow-up) were calculated based on total enrollment. Participants were considered “protocol adherent” if they accessed DECIDES (DECIDES, DECIDES+) and met with the coach (DECIDES+). The usual care group had no intervention protocol to follow and were not classified as adherent. Intervention engagement was assessed by the rate of DECIDES access (recorded in Google Analytics using unique login IDs) and completion of the values clarification exercise. Coaching engagement was measured by the frequency and duration of coaching sessions attended. Acceptability and usability were evaluated postintervention using the Acceptability of Decision Aid (ADA) 40 and Systems Usability Scale (SUS), 41 respectively. Interviews guided by the Consolidated Framework for Implementation Research (CFIR) 42 were conducted to learn about treatment-related decision making (Individuals Domain – Characteristics Subdomain) and to obtain feedback about DECIDES 1) components (Innovation Domain), 2) modalities (Inner Setting Domain), 3) deliverers (Individual Domain), and 4) implementation success (Implementation Process Domain). AYA and caregivers (DECIDES, DECIDES+) completed the ADA, SUS, and interviews; oncology clinicians completed the SUS and interviews only.
Knowledge, decision-making involvement, and decision processes
Baseline and postintervention clinical trial knowledge (Knowledge of Clinical Trials [KCT]) 43 and postintervention measures of decision-making involvement (Shared Decision-Making Questionnaire [SDM-Q-9/Doc] 44 and Physician Decision-Making Scale [PDEMS]) 45 and decision processes (Decision Conflict Scale [DCS], 46 Decision Regret Scale [DRS], 47 Decision Self-Efficacy Scale [DSES], 48 Satisfaction with Decision Scale [SWD]) 49 were collected from AYA and caregivers across groups. For decision-making involvement and decision processes, AYA and caregivers were directed to respond based on their perceptions of AYA involvement and their experiences in cancer treatment decision making. All measures demonstrated adequate reliability for this sample (Table 1 for measure information; Supplementary Table 1 for additional measure details).
Measures: Short Description
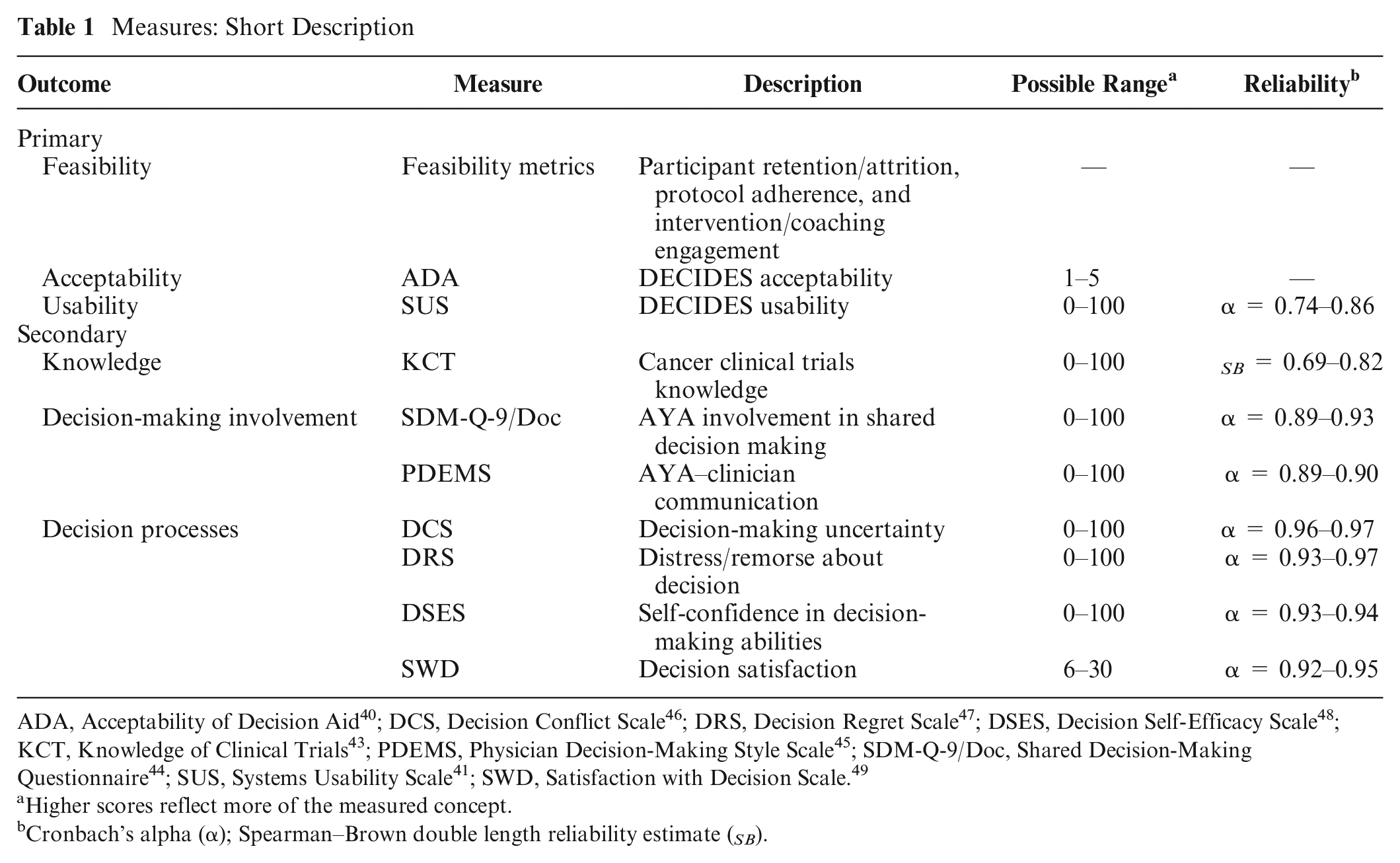
ADA, Acceptability of Decision Aid 40 ; DCS, Decision Conflict Scale 46 ; DRS, Decision Regret Scale 47 ; DSES, Decision Self-Efficacy Scale 48 ; KCT, Knowledge of Clinical Trials 43 ; PDEMS, Physician Decision-Making Style Scale 45 ; SDM-Q-9/Doc, Shared Decision-Making Questionnaire 44 ; SUS, Systems Usability Scale 41 ; SWD, Satisfaction with Decision Scale. 49
Higher scores reflect more of the measured concept.
Cronbach’s alpha (α); Spearman–Brown double length reliability estimate ( SB ).
Data Analytic Plan
Participant characteristics and feasibility metrics were described as was the level of engagement of each of the DECIDES groups in accessing DECIDES and time spent on the site. Protocol-adherent AYA and caregivers were included in descriptive analyses (acceptability, usability, postintervention knowledge, decision-making involvement, decision processes) and directed content analyses (interviews). Exploratory mean differences (MDs) were computed (postintervention knowledge, decision-making involvement, decision processes) to compare DECIDES and usual care groups, and DECIDES+ and usual care groups. These descriptive statistics were intended to identify potential trends, generate future hypotheses, and inform future study design, with the understanding that this pilot trial was not powered for statistical hypothesis testing across groups. An initial codebook (7 categories, 20 codes) was developed to code interview transcripts. Two trained researchers independently coded 3 transcripts. The primary coder (S.N.H.) merged coding, resolved discrepancies, and expanded the 20 codes into 64 subcodes. Remaining transcripts were coded by S.N.H. and a second coder (S.H.), with discrepancies resolved. Joint displays were created to link acceptability ratings to pertinent qualitative codes. Oncology clinician SUS data were included in analyses; their interviews were not analyzed for this report. Quantitative data were analyzed with SPSS (v29) 50 and qualitative data with Atlas.ti.23.51,52
The funding source had no role in this study.
Results
Participant Characteristics
Table 2 presents AYA and caregiver characteristics. There were no observable differences between groups, although the usual care group had higher baseline KCT scores than the DECIDES groups did. Oncology clinicians included attendings (n = 6), fellows (n = 4), and a nurse practitioner (n = 1) with a wide range in years of experience in pediatric oncology (Myears = 11, range = 2–25).
Participant Characteristics

AYA, adolescents and young adults.
Feasibility, Acceptability, and Usability
Feasibility
High retention was achieved, with 48 (84%) AYA and 41 (87%) caregivers completing the study. Nine (16%) AYA and 6 (13%) caregivers withdrew or were lost to follow-up at postintervention. Data from 2 second caregivers in the DECIDES+ group were excluded due to the limited sample size. Eleven (85%) oncology clinicians completed participation.
AYA and caregivers received access to DECIDES 6 wk (
Qualitative interviews revealed that few AYAs received access to DECIDES before initial treatment consent, and most AYA and their caregivers wished they received access to the decision support intervention sooner (Table 3 and Supplemental Table 2). Most AYA perceived DECIDES as helpful in understanding the initial treatment decision and described its utility for future treatment-related decisions that arise. One AYA indicated, “Having access to DECIDES gave me a better understanding of my diagnosis and treatment. If I need to make future decisions about cancer treatment, it will be helpful for me to have access to DECIDES to help me think about things in a different perspective” (16 y old, Hodgkin lymphoma), and another AYA indicated, “I didn’t really have a decision when it came to the first treatment of chemo. But then I found out later that I needed radiation, and I already had access to DECIDES—so I went on to see if I had any other options besides the radiation and I was reading all about that and it helped me” (18 y old, Hodgkin lymphoma).
DECIDES Acceptability: AYA

ADA, Acceptability of Decision Aid; AYA, adolescent and young adult.
Joint display of protocol-adherent AYA ADA ratings (n = 32 [13 DECIDES, 19 DECIDES+]) and interviews (n = 29 [12 DECIDES, 17 DECIDES+). 1–3 = low-neutral acceptability; 4–5 = high acceptability.
Acceptability
Table 3 and Supplemental Table 2 present the joint display of acceptability ratings (ADA) and matched interview quotes for protocol-adherent AYA and caregivers, respectively. The average total ADA score for AYA was 4.10 for DECIDES and 4.06 for DECIDES+, with both groups indicating lower ratings for items related to timing of DECIDES delivery. Average total ADA scores for caregivers in the DECIDES (
Usability
DECIDES usability (SUS) was rated as primarily good to excellent, with clinician ratings higher than protocol-adherent AYA and caregivers (Table 4).
DECIDES Usability a

Systems Usability Scale (SUS) data for protocol-adherent adolescents and young adults (AYA) and caregivers, and oncology clinicians.
Knowledge, Decision-Making Involvement, and Decision Processes
AYA
Compared with usual care, protocol-adherent AYA in the DECIDES and DECIDES+ groups reported higher scores on the PDEMS (MD = 19.31, 9.78) and SDM-Q-9 (MD = 12.58, 11.93) and lower scores on the DRS (MD = −13.19, −16.55). Both DECIDES groups had lower scores on the KCT questionnaire at postintervention than those in the usual care group did (MD = −5.75, −7.13), but their baseline scores were also lower. Observed scores on the DCS, DSES, and SWD were similar across groups (Table 5).
Knowledge, Decision-Making Involvement, and Decision Processes

Descriptive statistics and mean differences (MD) between values for protocol-adherent participants in the DECIDES groups and usual care.
Caregivers
Compared with usual care, protocol-adherent caregivers in DECIDES and DECIDES+ groups reported higher scores on the PDEMS (MD = 19.33, 18.33), SDM-Q-Doc (MD = 8.15, 3.43), and DSES (MD = 13.71, 5.87) and lower scores on the DCS (MD = −11.87, −4.98). The DECIDES+ group also reported lower scores on the Decision Regret Scale than the usual care group did, while the DECIDES group reported higher scores in the unexpected direction (DRS; MD = −4.65, 10.22). The observed scores on the KCT questionnaire at postintervention and SWD were similar across groups (Table 5).
Discussion
This study presents findings of a first-of-its-kind trial of a decision support intervention for treatment-related decision making among AYA newly diagnosed with cancer. Overall, results regarding feasibility, acceptability, and usability across both DECIDES groups are encouraging, with exceptions for timing of delivery of the intervention and preferences regarding how information was presented. Participants with access to a coach appeared to spend more time on DECIDES and completed the values clarification exercise more frequently. Regarding knowledge, decision-making involvement, and decision processes, AYA in the DECIDES groups generally appeared to report higher involvement in decision making and lower decision regret than AYA in the usual care group did. Caregivers in the DECIDES groups appeared to report higher AYA involvement in decision making, higher decision self-efficacy, and lower decision conflict than caregivers in the usual care group did, with larger MDs observed for the DECIDES group. Knowledge scores differed across groups at baseline, with very small changes observed at postintervention. Qualitative findings confirmed the premise of the study—decision support interventions are acceptable, and there is a need for trusted cancer information and decision support from the moment of diagnosis and to inform ongoing treatment-related decision making.
In contrast to a recent review suggesting limited value of coaches in decision support interventions 23 —an observation aligned with our descriptive quantitative findings—these results indicated that having a coach contributed to more time on DECIDES and a higher likelihood of completing the values clarification exercise. Qualitative interview data underscored that AYA and caregivers in the DECIDES+ group appreciated access to the coach. Factors that may have limited the potential impact of a coach in this study were variability in the number of meetings beyond initial accessing of the site, inconsistent coach guidance in completing the values clarification exercise, and variability in family requests for additional coaching sessions. As such, systematizing the coach in decision support interventions, introducing the coach at diagnosis, and evaluation of the cost-effectiveness of adding coaching to decision support interventions are needed to weigh the benefit of a coach to ensure engagement on the Web site.
Values clarification exercises are intended to explicitly identify AYA values related to cancer and treatment53,54; however, the DECIDES values clarification exercise was not consistently completed by AYA, either prior to the treatment planning meeting or, in some cases, at all. This raises the question of whether results might have been stronger for decision processes if AYA completed the exercise prior to making an initial treatment decision and if the results were delivered to oncology clinicians, as intended. Studies have outlined AYA priorities in decision making of mental mindset, self-advocacy, self-care practices, and navigating relationships as well as their preferences for reduced length of treatment, minimization of side effects, and return to “normal life.”55,56 Responses of AYA who completed the values clarification exercise confirm these values as they prioritized support for their treatment decision by family and friends, minimizing length of treatment and side effects, and regular contact with their health care team. Oncology clinicians’ ability to assess, acknowledge, and accommodate AYA values may influence their relationship with their AYA patients and in turn AYA engagement in decision making and treatment. 57 Future studies require attention to implementation of values clarification during treatment-related decision making for improving decision processes.58,59
Implementation of decision support interventions will require careful evaluation using established implementation frameworks. 42 Although AYA and caregivers expressed a strong desire for access to DECIDES at the time of diagnosis during initial treatment decision making, barriers remain to even earlier intervention. We set out to recruit AYA ≤6 wk postdiagnosis, and oncology clinicians viewed DECIDES at diagnosis as acceptable and usable. However, the quick turnaround between the diagnostic and treatment-planning meetings often meant that initial treatment decisions were made with or for AYA before DECIDES could be offered. Similar to those noted in the literature, 11 additional challenges, such as oncology clinician concerns about AYA physical condition, family distress, and time demands for consultations and procedures, further complicated timely access to the tool as early as intended. Even though AYA and caregivers endorsed the benefits of access to decision support after the initial treatment decision, for understanding their cancer and treatment and for ongoing decision making throughout treatment, optimizing timing is key. Potential implementation strategies for earlier intervention, gleaned from the qualitative interviews, included delivery of DECIDES via the EHR or during new diagnosis education and reviewing DECIDES during clinic visits. These suggestions from our sample map onto a review of barriers (e.g., time, skill, knowledge, oncology clinician attitudes) and facilitators (e.g., usefulness and patient preference) to the implementation of decision support aids in adult practice.60,61
DECIDES was developed using a rigorous, iterative, participatory approach with input from AYA, caregivers, and oncology clinicians. Intervention components were based on decision support guidelines and data from our prior studies of AYA decision making.11,34–36 This approach to development is time-consuming, while new technology and AYA digital preferences evolve quickly. As such, AYA perceived some graphics and Web site organization to be outdated and provided recommendations for improved usability even in the context of high acceptability and SUS scores. Specifically, AYA and their caregivers suggested improved cross-device compatibility, updated universal graphics, additional resources related to all cancer diagnoses, and opportunities for connection to other AYA.
Limitations include that the study took place at a single site, in an academic pediatric cancer program where enrollment on clinical trials is generally higher than in adult treatment settings. With the limited sample size, this pilot trial was not powered to detect statistical comparisons, and therefore, MDs cannot be fully interpreted. Efficacy testing with a larger, more diverse sample of AYAs at diagnosis and further elucidating how decision support interventions inform different types of treatment-related decisions, including in adult treatment settings, is needed as there may be differences in acceptability, feasibility, and effects on decision making. Results related to knowledge were perplexing and require further evaluation because despite randomization, knowledge varied by almost 10 points across groups at baseline. Finally, the 2 DECIDES groups reflected the diversity of our Cancer Center populations, but the usual care group identified as White, and caregiver education levels were high. The evaluation of decision support interventions across AYA that vary in terms of race, ethnicity, socioeconomic status, and health literacy is a priority.
In conclusion, results from this pilot trial endorse the feasibility, acceptability, and usability of a decision support intervention for AYA newly diagnosed with cancer. AYA reports of decision-making involvement and decision processes suggest that exposure to decision support may increase perceived involvement in decisions and reduce decision regret as they reflect on and understand treatment decisions already made and anticipate future decisions. These findings justify and inform further development and testing of decision support interventions that incorporate components to address the broad range of treatment-related decisions for AYA and offer structure for increased engagement in health care and health communications (including making treatment-related decisions). AYA and caregiver feedback underscore the importance of attention to barriers and facilitators during real-time treatment-related decisions, including for clinical trials, for future testing and implementation. Ultimately, increasing AYA involvement in treatment-related decision making remains a critical component of efforts to address disparate AYA cancer outcomes and improve well-being.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683251344624 – Supplemental material for Evaluation of a Decision Support Intervention for Adolescents and Young Adults Newly Diagnosed with Cancer: A Pilot Randomized Trial
Supplemental material, sj-docx-1-mpp-10.1177_23814683251344624 for Evaluation of a Decision Support Intervention for Adolescents and Young Adults Newly Diagnosed with Cancer: A Pilot Randomized Trial by Lamia P. Barakat, Shannon N. Hammer, Yansong Wen, Ashley Anil, Lisa A. Schwartz, Anne Reilly, Rochelle Bagatell, Marilyn M. Schapira, Yimei Li and Janet A. Deatrick in MDM Policy & Practice
Footnotes
Acknowledgements
We thank the AYA patients and their caregivers who participated in this study and contributed to the development of the DECIDES decision support intervention. Damien Leri, MEd, of Big Yellow Star contributed to Web site development. We also thank the DECIDES coaches, the clinical research staff, and those who participated in coding of the qualitative interviews, specifically Saarah Hussain, Rebecca Madden, and Nithyasri Ramaswamy. Study materials are available by request to Lamia P. Barakat.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a B+ Foundation Grant to Lamia P. Barakat (principal investigator), and formative research was funded by a Children’s Hospital of Philadelphia Foerderer Grant and Department of Pediatrics Academic Enrichment Grant to Lamia P. Barakat (principal investigator). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Authors’ Note
Preliminary findings of this study were presented at the Annual Meeting of the Society of Pediatric Psychology, April 2022.
Ethical Considerations
Informed consent was obtained from adult participants and assent from AYA <18 y at enrollment. This study was approved by the Institutional Review Board at Children’s Hospital of Philadelphia (IRB No. 20-017521) and registered on ClinicalTrials.gov (NCT ID No. NCT06191679).
Consent to Participate
Informed consent was obtained from adult participants and assent from AYA <18 y at enrollment. This study was approved by the Institutional Review Board at Children’s Hospital of Philadelphia (IRB No. 20-017521) and registered on ClinicalTrials.gov (NCT ID No. NCT06191679).
Consent for Publication
To enroll in the study, consent/assent were obtained during clinic visits or via phone. Parent/legal guardian consent and AYA assent were required for AYA <18 y to participate. AYA ≥18 y and up to 2 caregivers (primary and second) provided consent for themselves.
Data Availability
Study materials are available by request to Lamia P. Barakat.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
