Abstract
Highlights
Most lung cancer screening (LCS) interventions evaluated to date have been educational in nature and focused primarily on shared decision making or the initial uptake of screening, with some interventions demonstrating statistically significant improvements in patient knowledge and initial LCS order/uptake.
A critical gap in knowledge remains regarding how to effectively support LCS eligibility assessment as well as adherence to annual screening and appropriate diagnostic testing.
Findings underscore the need for the field to expand beyond education-focused interventions and incorporate multilevel targets when designing interventions to support high-quality LCS in practice.
Keywords
Background and Purpose
In 2011, the US-based National Lung Screening Trial (NLST) 1 and again in 2020 the Nederlands–Leuvens Longkanker Screenings Onderzoek (NELSON) Trial 2 found that annual lung cancer screening with low-dose computed tomography (LDCT) substantially reduces mortality from lung cancer. 1 In 2013, the US Preventive Services Task Force (USPSTF) released its first recommendation for offering lung cancer screening (LCS) to asymptomatic adults aged 55 to 80 y who have a 30-pack-year history of cigarette smoking and who either currently smoke or quit smoking within the past 15 y. Yet, because of the inherent risks associated with LDCT screening for LCS, including the risks associated with radiation exposure, false-positive results, overdiagnosis, and invasive follow-up procedures, LDCT screening is considered a preference-sensitive decision. 3 As recommended by the USPSTF guidelines, LDCT screening should therefore be offered in the context of shared decision making (SDM) and include counseling regarding either smoking cessation or continued abstinence. Based on new scientific evidence and in an attempt to mitigate known lung cancer disparities, the 2021 update to the USPSTF guidelines expanded eligibility to those aged 50 to 80 y who have a 20 pack-year smoking history and currently smoke or have quit within the past 15 y. 4 Consistent with the preference-sensitive nature of the LDCT screening decision, per benefit requirements, Medicare beneficiaries need to review a decision aid and undergo SDM counseling regarding LDCT screening risks and benefits and the importance of adherence to annual screening prior to an initial LDCT screening. 4 Counseling on either tobacco cessation or maintaining tobacco abstinence are also prerequisites for Medicare reimbursement among screening-eligible people.
As such, the implementation of LCS is complex, requiring organizations to consider a number of steps along the screening process, including how they will identify and recruit people for screening, assess screening eligibility, engage eligible people in SDM, provide appropriate tobacco cessation counseling, ensure an order/referral for screening is made for those deciding to screen, support those electing to undergo screening with the completion of testing via LDCT, and prompt and monitor appropriate annual screening or diagnostic testing based on test results.5,6 The complexity of LCS means that organizations wanting to implement comprehensive LCS programs must consider and decide how to achieve multiple micro-processes simultaneously.
To our knowledge, the evidence upon which to make such implementation-related decisions has not been summarized previously. The overarching goal of this systematic review is to inform strategies for supporting the continuum of activities needed for health care organizations to implement LDCT screening for lung cancer. We sought to identify interventions designed to facilitate the initiation, adoption, or improvement of the LCS process within health care organizations regardless of the intervention’s target population (e.g., patient, provider, or otherwise). Evaluations of interventions were included if they focused on any step in the LCS process prior to a diagnosis of lung cancer, including risk or eligibility assessment, SDM, adherence to initial or annual screening, or abnormal follow-up testing. The specific research questions of interest included the following:
What are the key components of interventions that have been used to initiate, adopt, or improve LCS processes, and where along the LCS continuum are these interventions focused?
What behavioral frameworks, models, and theories have been used in the design and evaluation of these interventions?
What are the types and levels of outcomes that have been evaluated within the context of these LCS interventions?
Which patient populations have been targeted by these interventions, and how have interventions addressed (or not addressed) issues of equity in LCS?
What is the emerging evidence regarding the effects of interventions designed to facilitate the initiation, adoption, or improvement of the LCS process on patient- and other-level outcomes?
Methods
Data Sources
We searched CINAHL, Cochrane Library, Embase, Ovid Medline, PsycINFO, and Scopus to identify relevant studies. Database searches were originally performed in 2020 and updated in the first quarter of 2024, enabling the inclusion of papers published January 1, 2011, through December 31, 2023. In addition to published articles, we searched conference proceedings and abstracts to identify relevant items. We also manually reviewed the reference lists of any systematic reviews identified during the search process to identify additional relevant studies. Databases were searched by combining the MeSH terms and other search terms related to lung cancer (“Lung Neoplasms” OR “lung cancer” OR “lung tumor”) with those specific to cancer screening (e.g., “Cancer Screening,” “Early Detection of Cancer”) and behavioral and other interventions (e.g., “Health Behavior,” “Health Promotion,” “Intervention Trials”). We worked with a health sciences librarian to tailor terms to be appropriate to each specific database searched, but the terms covered similar group-level concepts: 1 = lung cancer, 2 = cancer screening, 3 = low-dose CT, and 4 = behavior and intervention terms. Full search strategies are available in the appendix.
For the search update, we used an artificial intelligence (AI) tool to expedite the updating process. There is substantial evidence on the application of AI, specifically machine learning (ML) tools, to systematic reviews and other literature-based research across the health sciences.7–11 This ML tool, the Document Classification and Topic Extraction Resource or DoCTER, 12 is a predictive application in which articles are classified using algorithms such as naïve Bayes and support vector machine. While AI tools typically require a large amount of training data, applying ML to a systematic review update allows the original screening decisions and included papers to be used as the training dataset.13,14
For the update, a health sciences librarian (R.B.C.) used the studies found relevant and irrelevant during the first level of the initial review process conducted in 2020 as training data. All citations in the search update were run through the ML tool and then put in priority order from most likely to least likely to be relevant according to the algorithms. Using the same 2-level review process outlined below, the team then manually screened all the newly identified studies in priority order based on probability score. The studies screened in probability order were graphed against the screening decisions to ensure that screening precision was declining precipitously as expected and that the recall threshold would be met. 15 Once the team perceived the remaining identified studies were no longer relevant, an additional 500 citations were screened to ensure no relevant citations would be lost. While AI was used to augment the search update, it did not replace the screening decisions and expertise of the research team.
Study Selection
We included studies that explicitly evaluated an intervention or specific component of an intervention to initiate, adapt, or improve LCS and its relationship to at least 1 quantitatively measured outcome. Only studies that reported the use of a comparator group were included regardless of whether an experimental or quasi-experimental design was used. Comparators could include usual care (within which an organized LCS intervention may or may not have been available), a different type of LCS screening intervention, or historical controls, including single-arm pre–post designs. Because of the limited number of such studies thought to be available when we initiated our review, we elected to include any English-language study, regardless of country context. We excluded studies targeting people who did not meet currently accepted LCS eligibility criteria (e.g., eligibility as endorsed by Medicare 16 or the US Preventive Services Task Force 17 ) unless the study was attempting to identify people who were LCS eligible. We also excluded studies for which the only outcome mentioned within the published abstract was “smoking cessation” as well as studies that did not report a measured relationship between an intervention or specific intervention component(s) and a patient- or other-level outcome. We limited searches to items published in print or online in English between January 1, 2011, and December 31, 2023, and included only those studies for which a full text was available. We did so to ensure the inclusion of a broad array of interventions that collectively would be of interest to those attempting to implement LCS within health care organizations, particularly those wanting to target adults for whom LCS was compatible with current US-based reimbursement policies.
We used a 2-level review process followed by data extraction among eligible studies. We used DistillerSR 18 and Covidence 19 for literature management. For the first-level review, 2 members of the study team independently reviewed each identified title and abstract. Any study marked for inclusion by at least 1 team member was included in the second-level review. For the second-level review, 2 members of the study team independently reviewed full texts of all studies marked for inclusion in the level 1 review. Disagreements during the level 2 review were resolved by consensus among study team members during regular research team meetings.
Data Extraction
Once a study was determined as eligible for inclusion, 2 reviewers independently conducted data extraction using the configurable forms available within the literature management software. The abstraction form contained items for study characteristics (e.g., author, publication year, country of study, funding source, and study design), participant characteristics (e.g., sample size and demographic characteristics), a description of the intervention, and study outcomes (e.g., primary and other outcomes, level of measurement, and data source). Each identified intervention was characterized by the steps along the LCS process being targeted. These steps were categorized as risk and eligibility assessment, SDM, initial screening uptake, annual screening adherence, or abnormal LCS screening follow-up. Study forms also contained fields to capture any behavioral framework, model, or theory used to guide the design of the intervention or its implementation. Conflicts were resolved by discussion and consensus among investigators during research team meetings.
We assessed the quality and risk of bias among included studies using the Cochrane Collaboration’s risk of bias tools, ROB 2.0 20 or ROBINS-I, 21 respectively, for randomized trials and nonrandomized studies. Each of these tools assesses the risk of bias across a set of domains that reflect aspects of study design. The ROB 2.0 assesses quality across the 5 domains of randomization process, intervention fidelity, missing outcome data, outcome measurement, and selection of the reported results. 20 The ROBINS-I tool assesses quality across the 7 domains of confounding, selection of participants, classification of interventions, fidelity of interventions, missing data, measurement of outcomes, and selection of reported results. 21 In general, results from studies assessed to have high risk of bias (ROB-2) or serious or critical risk of bias (ROBINS-I) should be interpreted with caution. Two team members independently completed these assessments for each included study. Discrepancies were resolved by consensus at research team meetings. The protocol for this systematic review was registered with PROSPERO. 22 We follow the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for reporting results. 23
Results and Data Synthesis
Study Selection and Characteristics
Following PRISMA, 24 the flow diagram of study disposition and inclusion is depicted in Figure 1. Sixty-four published articles (Table 1) were identified as study eligible.25–88 As depicted in the table, 19 of the included studies were randomized trials. The remainder used a quasi-experimental approach, most frequently a single-arm pre–post design. Fifty-five studies (15 of the 19 randomized trials and 40 of the 45 quasi-experiments) were conducted within the United States. The remaining studies were conducted within the United Kingdom (n = 4), Australia (n = 1), Austria (n = 1), Canada (n = 1), South Korea (n = 1), or Turkey (n = 1).
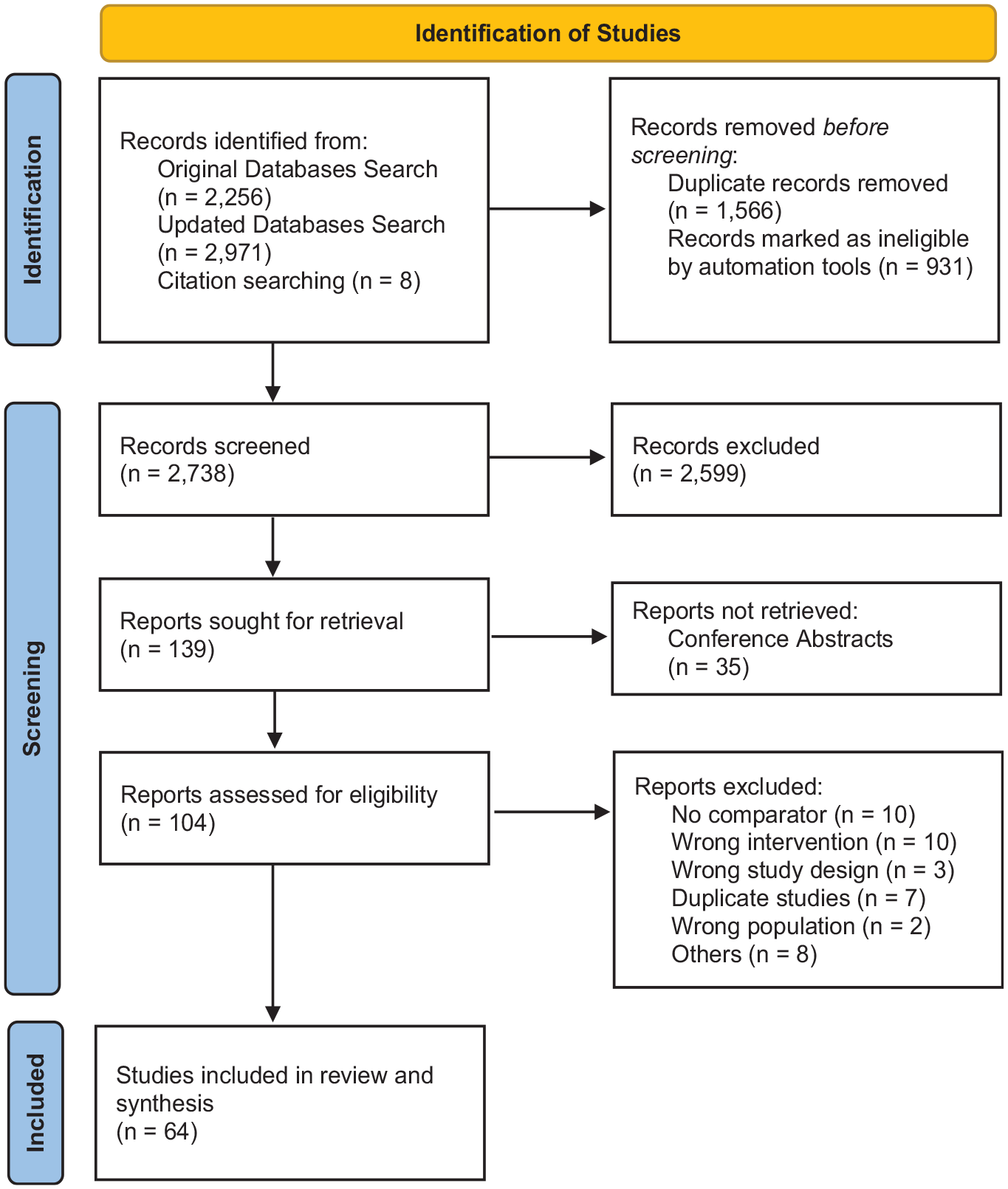
PRISMA 2020 flow diagram for systematic reviews.
Included Studies.

Characteristics of Study Participants
Included studies ranged in size from 16 to more than 48,000 participants, with almost a third (n = 21) enrolling fewer than 100 people. As illustrated in Table 2, all but 9 studies used patients as the only unit of observation. Those using the patient as the unit of observation tended to include people who currently smoked and previously smoked, but 1 of the randomized trials included only people who previously smoked 55 and 2 included only people who currently smoked.45,78 Most studies reported information on participant sex or gender (n = 50), and each of the 41 studies that reported information on participant race included participants from multiple racial groups, albeit always with substantially more White than other race participants. Notably, the patient samples used among the more recently published studies were relatively more racially and ethnically diverse. Also of note was the fact that barely one-third of the studies reported information regarding participants’ health insurance coverage.
Participant Characteristics.

AI/AN, American Indian/Alaskan Native; Con, control group; IQR, interquartile range; Inter, intervention group; LCS, lung cancer screening; NA, not applicable; NR, not reported; NHS, National Health Service; NHI, national health insurance; NH/PI, Native Hawaiian/Pacific Islander; NR, not reported; VA, Veterans Affairs.
Characteristics of the Interventions Evaluated
Of note, all but 15 studies evaluated interventions that targeted the patient alone (Table 3). Among those 15 studies, 11 targeted providers alone,* 2 targeted both patients and providers,37,54 and 2 targeted communities as defined by a geographical area in the case of one and the social networks of community health workers as defined by the other.28,84 Except for 2 studies,50,68 none of these latter interventions were studied in the context of a randomized trial.
Characteristics of Lung Cancer Screening (LCS) Interventions Evaluated.

EHR, electronic health record; LCS, lung cancer screening; LDCT, low-dose computed tomography; NSCLC, non-small-cell lung cancer; SDM, shared decision making.
Almost 60% of studies (n = 38) targeted 1 specific step of the LCS process, while the remainder targeted 2 or more steps (Table 3). SDM was most often targeted (n = 36), with most of these studies (n = 21) focused solely on that 1 step of the LCS process. Twenty-seven studies focused on interventions to enhance initial screening uptake, again with most focused on that 1 step alone (n = 14). Nineteen studies evaluated interventions focused on screening risk or eligibility assessment (n = 19). Those doing so typically included a focus on at least 1 additional step of the LCS process (n = 13). Only 5 studies38,60,63,65,73 included a focus on follow-up testing, most often using an outcome defined as “recommended follow-up,” regardless of what that follow-up entailed. Only 1 study 73 focused on follow-up after a normal scan alone (i.e., repeat screening).
Most studies (n = 50) evaluated educational interventions, whether one-on-one education, group education, or mass media, and whether alone (n = 40) or in combination with something else (n = 10). Twenty-two interventions addressed structural barriers to LCS, often using or in combination with educational interventions. Such interventions provided patient navigators,26,45,65 electronic forms to determine and/or identify LCS eligibility,31,43,66,74 and community health workers to connect eligible persons to LCS services.78,83
Use of Behavioral Theories, Frameworks, or Models
The use of a specific behavioral theory, model, or framework was relatively rare among published studies. Exceptions were 2 studies that reported using the Precaution Adoption Process Model alone 57 or linked with the Health Belief Model 30 and those that reported using the Health Belief Model,83,84 the Integrated Model of Health Behavior, 36 or motivational interviewing techniques. 45
In addition, 3 studies56,60,68 reported using guidance from implementation frameworks, and one evaluated different screening messages grounded in communication theories. 79 Some studies testing decision aids reported using the International Patient Decision Aid Standards41,53 or the Option Grid 35 to guide decision aid content or the Ottawa Decision Support Framework to combine decision aids with personal coaching or online decision support to guide their work,39,56 and a few educational intervention studies referenced specific educational approaches (i.e., repetitive review methods, 58 small-group interactive learning, 44 and teachable moments).77,78
Evaluation of Intervention Effects
Two-thirds of the studies (n = 43) reported statistically significant findings (Table 4). Approximately one-third of those (n = 16) focused on improving knowledge as the study’s primary outcome, primarily patient knowledge. Most of these studies did not use a randomized design (n = 13), and while none of the randomized trials were assessed to have a high risk of bias, 5 of the quasi-experimental design studies were determined to face serious risk of bias. Ten studies with statistically significant findings focused on initial LCS uptake, † 3 of which used a randomized design.45,65,78 While 1 of those studies was determined to be of high risk of bias, 78 all but one 66 of the quasi-experimental design studies were classified as serious risk of bias. Other studies with statistically significant findings that used a randomized design focused on the patient40,53,81 and provider 68 decision making or patient behavioral intent. 79 Among studies employing a quasi-experimental design that were assessed as not having a serious risk of bias, outcomes significantly affected by the tested interventions included provider confidence, 85 patient eligibility, 62 perceived risk of cancer, 35 decisional conflict,41,64 behavioral (LCS) intent, 86 adherence to annual LCS, 73 and medical record completeness. 74 Overall, 14 of the 19 randomized trials and 18 of the 45 quasi experimental design studies (a total of half of the identified studies) were assessed to be at low or moderate/some risk of bias, and among these, most (n = 27) reported statistically significant findings.
Evaluation of Outcomes and Risk of Bias.

LCS, lung cancer screening; LDCT, low-dose computed tomography; NR, not reported.
Discussion
In this systematic review, we sought to summarize the evidence for interventions designed to initiate, adopt, or improve LCS and identify where in the LCS continuum interventions have been focused. We also sought to characterize this literature in terms of the target populations of the interventions and the outcomes that have been assessed. We found 64 English-language published articles, not surprisingly with most from the United States. Less than one-third were randomized trials, with the remainder using a quasi-experimental design with the risk of bias variable within both types of design. Most interventions regardless of country context targeted only patients, with some targeting providers. Only a few targeted communities or a combination of levels, each of these among US-based populations. The most common category of intervention was education, typically provided to individuals one-on-one. Some studies targeted structural barriers, at times alongside educational interventions, and although the use of navigators and community health workers was most prevalent, there was not one common intervention used to address structural barriers. Our analysis was unique in examining the nature of interventions along steps in the LCS continuum from risk and eligibility assessment to SDM, initial screening uptake, annual screening adherence, and abnormal follow-up testing. However, rather than finding interventions targeted across this continuum, the majority, regardless of country context, targeted the early steps in this process, with only 2 studies (1 randomized trial 65 and 1 quasi-experimental design, 73 both of which were US based) found not to be of serious/high risk of bias focused on follow-up testing 65 or adherence to annual LCS. 73
Our systematic review found support for the premise that English-language published interventions designed to improve specific steps early in the process of LCS have been successful. Six randomized controlled trials assessed to have only a low or some risk of bias found significant improvements in knowledge or preparation for decision making.30,34,40,48,53,81 Quasi-experimental studies had similar results, with multiple studies with low or moderate risk of bias reporting that interventions increased patients’ knowledge,39,42,49,52,67 improved their perceptions of lung cancer risk, 35 or reduced decisional conflict.41,64 Similarly, multiple studies reported increased provider confidence, knowledge, or both.58,71,85 However, evidence was less robust regarding the outcomes of LCS uptake: only 2 randomized trials with low or moderate risk of bias reported an intervention that increased LCS uptake 45 or compliance with follow-up testing. 65 In contrast, 3 randomized control trials with low or moderate risk of bias failed to have an impact on either attendance at a lung health check appointment in the United Kingdom 57 or receipt of LCS in the United States.55,80 Similarly, 2 quasi-experimental studies with low or moderate risk of bias had an effect on LCS receipt in the United States 66 or adherence to annual LCS in the United States, 73 while 2 failed to have an impact on LCS receipt or intention to have LCS, 1 in the United States and 1 in South Korea.28,86
Our study highlights the relative lack of English-language published interventions that target follow-up testing or repeated annual LCS. Prior studies have found rates of follow-up testing to be suboptimal, particularly among marginalized populations. 89 As has now been well established, the benefits of cancer screening accrue from the receipt of high-quality care across the cancer screening continuum from eligible patient identification and test uptake through to adherence to guideline-concordant interval testing frequency to the follow-up of abnormal results.90–92 For LCS to incur both individual- and population-level benefits, interventions targeted to increase adherence to follow-up and annual screening are needed.
Most studies evaluated patient-level interventions. Among these, the findings related to knowledge of LCS and preparation for decision making are consistent with the broader literature that finds patient decision aids and other types of SDM increase patient knowledge about interventions.93,94 We identified only 3 randomized trials with low risk of bias that specifically targeted structural barriers to LCS, all based in the United States. In 2 studies,45,65 patient navigator interventions significantly increased the receipt of LCS. This finding is consistent with evidence that patient navigation is an effective way to address structural barriers to other types of cancer screening. 95 In contrast, a third trial 55 leveraged patient portal messaging to address gaps in tobacco history within EHRs and found no significant change in LCS receipt. Given that LCS is one of the most underutilized cancer screening interventions, the reliance on one-on-one targeted interventions is unlikely to result in substantively meaningful uptake at a population level.
Our findings are noteworthy for the relative absence of commonly used interventions in the context of cancer screening such as text messaging, mHealth, the use of prompt and reminders, assessment and feedback, or the use of mass media or small media for outreach, all of which have been evaluated and found to be effective within the context of other types of cancer screening.96–100 Nor did we find many studies that targeted multiple levels of intervention, despite the wide-held belief of their relative advantages.90–92 In addition, we did not find studies that explicitly considered the costs associated with LCS implementation strategies. Each of these topics likely warrants further study.
We abstracted sociodemographic characteristics of participants including all categories of race and ethnicity reported in the study groups. Barely two-thirds of the identified studies reported some amount of race and ethnicity descriptors of study participants with varied levels of race and ethnicity categories. Furthermore, barely one-third reported information regarding participants’ health insurance coverage. Given historic and emerging disparities by race and ethnicity in LCS, at a minimum, it is necessary to report race, ethnicity, and socioeconomic factors among study populations to assess the generalizability of outcomes across diverse populations and to advance equity. However, to achieve equity, more than inclusive study populations and stratified reporting are necessary; rather, we will need to design studies to target determinants of disparities and achieve equity of outcomes. Designing interventions that will be both effective and equitable remains an important objective for the field.
Limitations
We focused our review on English-language studies published since 2011, when results from the National Lung Screening Trial were published, 1 through December 31, 2023, targeting studies identifiable via a variety of search engines. As such, our results exclude findings from studies that were not published and those most recently published as well as those not published in English. As evident from the volume of studies identified via the updated search, the field is rapidly expanding, and our study resources did not enable an additional rapid update through the end of 2024. As such, as with all systematic reviews for which the evidence base is evolving, care should be taken when generalizing our findings as the field expands. It should be noted, however, that while adding these newer studies diversified the sociodemographic characteristics of study populations used, it did not substantially alter the conclusions drawn from the current body of evidence. Similarly, because of our perception of the paucity of relevant studies as we began the review, we elected to include English-language studies, regardless of country context. While we did not detect notable differences between US-based and other country-based studies or their findings, care should be taken when extracting non–US-based study findings to the United States. As most non–US-based studies (like most of all the studies identified) focused on educational interventions, such a caution pertains mostly to the 3 studies that addressed structural barriers.27,43,62 We also excluded studies focused solely on interventions targeting smoking cessation due to a separate robust literature focused on this topic. 101
In addition, the risk of bias among the identified studies was highly variable. For example, although most randomized controlled trials had a low or some risk of bias as assessed by ROB 2.0, among studies using a quasi-experimental design, more than 40% were assessed as having a serious risk of bias using ROBINS-I criteria. Collectively, this suggests important gaps in study design, and therefore, the interpretation and future implementation are questionable. In addition, we found interventions often lack grounding within behavioral theory, model, or framework. In addition, our search criteria required a form of the term LDCT scan in the title, abstract, or keywords, a strategy that could miss studies referencing LCS with other terms. In the case of 1 study known to our team, 102 this search criteria failed to identify the study despite its relevance.
Conclusions
The body of literature evaluating interventions to improve the LCS process is strongest for interventions targeting improved knowledge about LCS and SDM. As such, the current body of literature is limited in its ability to help health care delivery organizations identify how to improve LCS uptake and the more distal steps in the LCS continuum. Rigorous studies of interventions targeting points across the LCS continuum are needed to support the translation of the NLST and NELSON trials into practice. Further, socioeconomic status has been found to be a mediator in racial disparities of annual LCS adherence. 89 With regard to the category of interventions developed, most have been one-on-one education, with some targeting different types of structural barriers to LCS faced by patients and, in 1 case, providers. Less well explored are interventions targeting the health system level, use of reminders or incentives, and outreach through the media. Equity remains an important issue across cancer screening interventions requiring data collection and reporting on patient- and population-level characteristics. This systematic review identifies the strengths and gaps in the published literature that can direct future efforts to help health care organizations wanting to implement effective LCS programs.
Footnotes
Appendix: Study Search Strategies by Database
The search ID was linked with a specific MESH/search term and/or conceptual term. Search terms were tailored to the specific database but covered similar group-level concepts: 1 = lung cancer, 2 = cancer screening, 3 = low-dose computed tomography (LDCT), and 4 = behavior and intervention terms. The final search combined groups (linked by “AND”). Included in the Appendix are search terms used in each of the included databases: CINAHL, Cochrane Library, Embase, Ovid Medline, PsycINFO, and Scopus.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JEL and CND reported receiving a grant to their institutions from Genentech. KR reported receiving grants to her institution from Pfizer and AstraZeneca and consulting fees from Merck and the American Journal of Managed Care. MEC reported receiving grants from the National Comprehensive Cancer Network/Astra Zeneca and the American Association for Cancer Research/Johnson and Johnson. AV reported receiving grants to his institution from MagArray Inc, Precyte Inc, Optellum Ltd, Median Technologies, Lungevity Foundation, the Gordon and Betty Moore Foundation, and the National Comprehensive Cancer Network and personal fees from Johnson and Johnson and Intuitive Surgical, outside the submitted work. CS, GLA, JW, LA, MMS, RC, and MO report having nothing to disclose. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award number UM1CA221939 (MPI: Ritzwoller/Vachani). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author’s Note
Claire Staresinic is now affiliated with Case Western Reserve University School of Medicine, Cleveland, OH, USA.
Ethical Considerations
We conducted a systematic review without meta-analyses. As such, our study did not contain private information. Only publicly accessible documents were used, and searches were designed to capture published studies regardless of publication venue, sample size, or the like. The methodological rigor of the included studies was independently assessed and reported using established risk-of-bias tools, and the presentation of results followed standards of quality and rigor in reporting.
Consent to Participate
The study did not use private information.
Consent for Publication
N/A
ORCID iDs
Data Availability
All data were obtained from publicly available sources that are referenced in the article’s bibliography. Each study is readily available to any interested party from online and/or library sources.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
