Abstract
Background
Considering a patient’s full risk factor profile can promote personalized shared decision making (SDM). One way to accomplish this is through encounter tools that incorporate prediction models, but little is known about clinicians’ perceptions of the feasibility of using these tools in practice. We examined how clinicians react to using one such encounter tool for personalizing SDM about lung cancer screening (LCS).
Design
We conducted a qualitative study based on field notes from academic detailing visits during a multisite quality improvement program. The detailer engaged one-on-one with 96 primary care clinicians across multiple Veterans Affairs sites (7 medical centers and 6 outlying clinics) to get feedback on 1) the rationale for prediction-based LCS and 2) how to use the DecisionPrecision (DP) encounter tool with eligible patients to personalize LCS discussions.
Results
Thematic content analysis from detailing visit data identified 6 categories of clinician willingness to use the DP tool to personalize SDM for LCS (adoption potential), varying from “Enthusiastic Potential Adopter” (n = 18) to “Definite Non-Adopter” (n = 16). Many clinicians (n = 52) articulated how they found the concept of prediction-based SDM highly appealing. However, to varying degrees, nearly all clinicians identified challenges to incorporating such an approach in routine practice.
Limitations
The results are based on the clinician’s initial reactions rather than longitudinal experience.
Conclusions
While many primary care clinicians saw real value in using prediction to personalize LCS decisions, more support is needed to overcome barriers to using encounter tools in practice. Based on these findings, we propose several strategies that may facilitate the adoption of prediction-based SDM in contexts such as LCS.
Highlights
Encounter tools that incorporate prediction models promote personalized shared decision making (SDM), but little is known about clinicians’ perceptions of the feasibility of using these tools in practice.
We examined how clinicians react to using one such encounter tool for personalizing SDM about lung cancer screening (LCS).
While many clinicians found the concept of prediction-based SDM highly appealing, nearly all clinicians identified challenges to incorporating such an approach in routine practice.
We propose several strategies to overcome adoption barriers and facilitate the use of prediction-based SDM in contexts such as LCS.
Introduction
Prediction-based approaches to clinical care can often facilitate more equitable, patient-centered care based on the principle of “equal management of equal risks.” 1 This is because using a well-validated risk prediction model, by incorporating a patient’s overall combination of risk factors, can provide much more accurate information about patient-specific benefit and support more personalized decision making. For decisions about lung cancer screening (LCS) with low-dose computed tomography, using prediction is particularly promising because of the documented wide variation in screening benefits across individuals in the target population that results from substantial heterogeneity in lung cancer risk and life expectancy across patients.2–6 For example, a 60-year-old man with a 30-pack-year smoking history has a 0.083% risk of dying from lung cancer in 6 y. In comparison, a 70-year-old woman with a 75-pack-year smoking history has a much higher 5.52% risk of dying of lung cancer in 6 y. While both patients are eligible for LCS according to the US Preventive Task Force (USPSTF) guideline recommendations, they have vastly different lung cancer risks and thus very different chances of benefitting from screening. 7
National LCS guidelines have also emphasized the importance of shared decision making (SDM) before initiating LCS, due to the preference-sensitive nature of the LCS decision for many patients (i.e., the appropriateness of screening depends on patient preferences).4,5,8 Clinical encounter–based decision tools are a promising approach to supporting SDM in practice.9–11 In the context of LCS, encounter-based tools that incorporate prediction models are particularly exciting because they can provide individualized benefit-harm information and potentially support the routine implementation of highly personalized SDM for LCS in practice. Further, statin use guidelines and other cardiovascular prevention guidelines, which have already successfully moved toward recommending using risk prediction for guiding certain decisions, may have created new norms among clinicians to use predictive approaches to support personalized decision making about prevention and screening.12–15
Nonetheless, practical aspects of carrying out personalized, prediction-based SDM using an encounter-based decision tool, such as the need to input risk factor information, may pose implementation challenges that have not yet been addressed in the cancer screening context. 16 While there have been significant strides in understanding the advantages of prediction-based LCS decisions, little is known about clinicians’ perceptions of the feasibility of implementing such an approach in practice.4,5,17–19
As part of a quality improvement (QI) study in Veteran Health Affairs, we developed and studied the implementation of a Web-based encounter decision tool (the DecisionPrecision [DP] tool). 20 DP provides individualized information based on risk prediction and is designed to support personalized SDM for LCS to facilitate the delivery of more equitable and patient-centered screening for USPSTF-eligible patients based on age and smoking history. 21 One of the implementation methods used in this QI study was academic detailing, an educational outreach approach focusing on one-on-one clinical education and behavior change. 22 Detailing numerous primary care clinicians across multiple medical facilities allowed us to explore in-depth clinician views on routinely implementing personalized, prediction-based SDM for cancer screening in the primary care setting. Thus, we conducted a qualitative study that used data from these academic detailing visits to examine 1 key component of these academic detailing discussions: how clinicians react to using our encounter-based decision tool (DP) to support personalized SDM.
Methods
This qualitative QI study evaluated the implementation of the DP encounter decision tool. The DP tool, and our overall QI evaluation, have been described previously. 21 Briefly, DP provides a combination of prediction-based information on the benefits and harms of LCS and prompts for improved patient-clinician communication and SDM. Individualized estimates of lung cancer risk and net benefit are based on the well-validated Bach et al. 23 prediction model, which estimates individualized lung cancer incidence based on the 6 inputs shown in Figure 1 (imputing no asbestos history if unknown) and which has been found to be well calibrated across multiple modern cohorts including US-representative cohorts. 24 The SDM component of the tool provides 30-s SDM scripts, electronic health record (her) documentation templates, and information explaining risk to patients in plain language. The prediction component of the tool provided risk and benefit information that an individual patient can expect with undergoing computed tomography (CT) screening based on their specific set of health factors. This includes a prediction-based, individualized assessment of the net benefit of CT screening—and presentation of how preference sensitive the screening decision is for a patient (i.e., the extent to which the best course of action is a matter of individual judgment about the balance of pros and cons), based on prior published work. 5 Figure 1 and Appendix 1 present screenshots of the key features of the DP encounter decision tool. The DP decision tool was designed to support “everyday shared decision making,” which has been described previously.4,25,26 Briefly, this approach to SDM is designed to be very brief and feasible within the intense time constraints of primary care practice, where there is typically only 1 to 2 min for SDM on LCS. It emphasizes providing the patient a personalized recommendation, qualitative information about the important tradeoffs, and support for the patient’s need for more information and autonomy to make the final decision, which includes the power to veto the initial recommendation. This last aspect is key and emphasizes there is always an essential role for patient preference to guide the ultimate screening decision.
DP was implemented in a 2-phase process at 7 Veterans Affairs medical centers (VAMCs), each with a well-established LCS program. 21 Phase 1 implementation engaged primary care team leadership, LCS coordinators, primary care clinicians, and specialist clinicians (pulmonologists, radiologists, and oncologists) in a QI training program and user-centered design process. 27 Phase 2 implementation discussions, which these results focus on, sought to more directly engage practicing primary care providers (PCPs) in using the DP tool to personalize SDM for LCS in their daily practice, through the use of academic detailing (see Appendix 2 for the detailing visit key discussion points). The phase 2 outcomes measured included qualitative field notes of the feedback provided from PCPs who participated in the academic detailing meetings. None of the academic detailing meetings were audio recorded. This project was categorized as QI and not formally reviewed by the institutional review board. Written informed consent was not required.
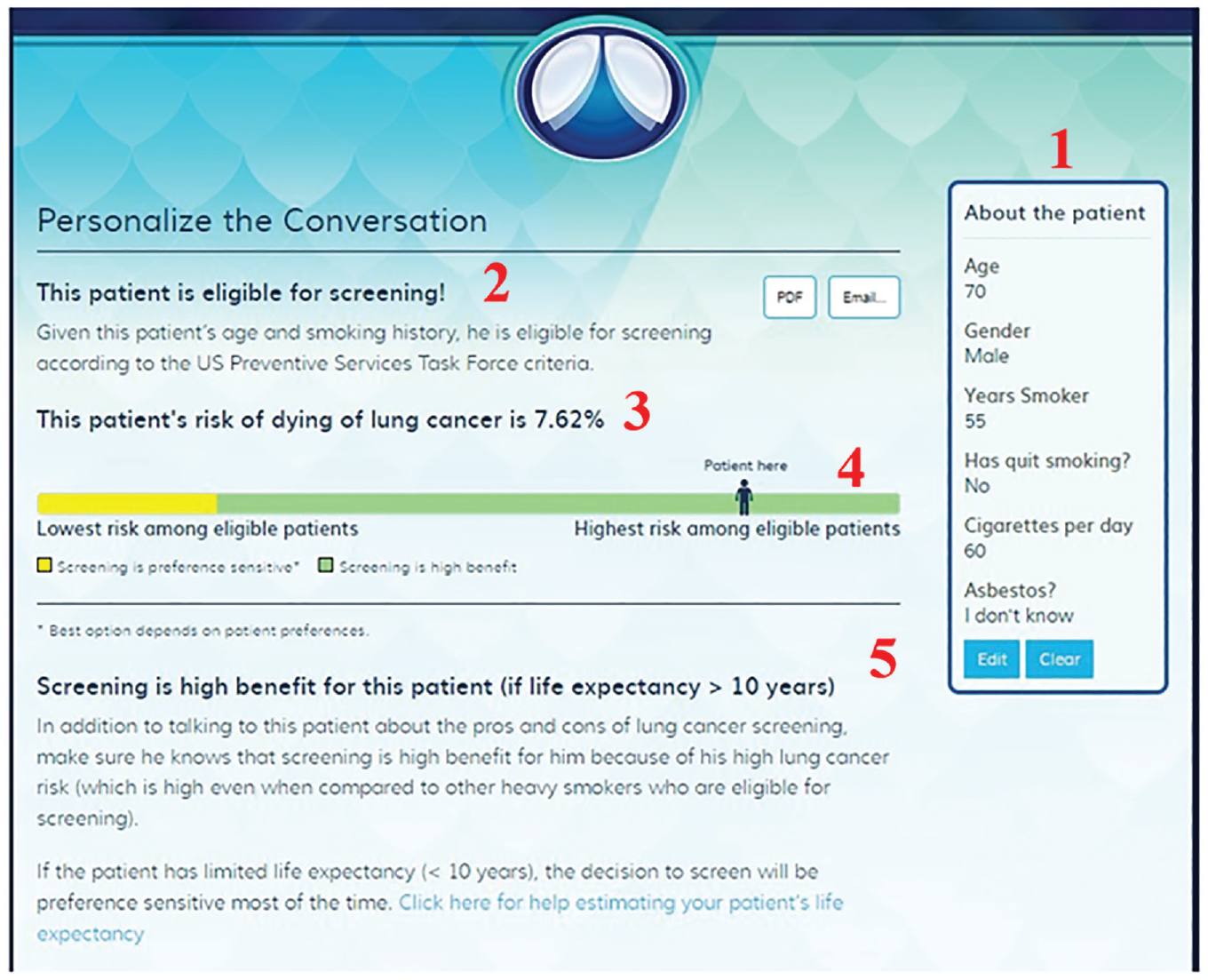
Output from the DecisionPrecision encounter-based decision tool (example for a high-benefit patient. (1) Risk factor inputs. (2) Patient’s screening eligibility status. (3) Patient’s risk of dying from lung cancer in the next 6 y. (4) Visual representation of where the patient falls on the risk spectrum for the target population of screening-eligible patients. (5) Notes on how to discuss screening options with patient.
Academic Detailing
An academic detailer on the study team (N.J.L.) visited 7 VAMCs and 6 community-based outpatient clinics between June 2018 and January 2019. Each site visit lasted approximately 2 to 3 d, and approval from each facility’s ambulatory care leadership was received before the visit. The study did not financially or otherwise incentivize individual participants and health care systems.
The academic detailing visits’ goal was to engage one-on-one with clinicians and clinical leaders to discuss 2 main points: 1) the rationale for prediction-based SDM and 2) how to use the DP encounter decision tool with eligible patients to personalize LCS recommendations. 20 These meetings were generally arranged ad hoc with the clinicians in clinic that day. All meetings were one-on-one, except 3 detailing meetings with 2 to 3 clinicians. At 5 of these facilities, the head of ambulatory care scheduled an initial larger group presentation, typically during a routine staff meeting, to provide clinicians with a general overview of the detailer, brief background on LCS and prediction-based decision making, and an introduction to the DP tool.
Immediately after every group and individual detailing meeting, the detailer took extensive field notes, documenting clinician perceptions about the barriers and benefits of using DP and prediction-based SDM for LCS. The field notes also included questions or concerns raised by each clinician and the detailer’s overall perceptions of the clinicians’ receptiveness to the information presented.
Analysis
QSRI’s NVivo version 11 data analysis software and Microsoft Excel were used to organize the detailer’s field notes. At the completion of data collection, the study team conducted thematic content analysis to systematically search for and examine themes and patterns that emerged from the qualitative data. A blended approach was used to code the field notes to examine predetermined codes that fit the academic detailing manual and emerging codes that appeared through the conversations. A qualitative analyst and the academic detailer independently summarized the field notes from the detailing sessions, fitting content into each predetermined code and new codes. At the completion of independent review, full team discussion and summarization of the analyses occurred, resulting in a shared understanding and acceptance of content themes.
Based on these resulting themes, we categorized each clinician into “adoption potential” categories based on the perceived benefits and barriers expressed during the detailing discussion. Each adoption potential category describes, along a spectrum, the clinician’s willingness to engage with and use the DP encounter decision tool to take a prediction-based approach to LCS. We also examined the relationship between the adoption potential categories and clinician age group (as roughly perceived by the detailer) and job description (i.e., resident, leadership, attending).
Results
The study team contacted the 7 VA sites participating in our QI project, and 6 agreed to a detailing visit. One facility reported being overwhelmed with staffing and leadership changes and declined. All sites have an academic affiliation and had a well-established LC screening program prior to our study’s engagement, half (n = 3) with a centralized program in which primary care clinicians referred patients to a screening coordinator to discuss enrolling in screening and to manage repeat screens and follow-up evaluation for those enrolled. Within the 6 participating sites, the detailer visited 7 VAMCs and 6 community-based outlying clinics. Across all sites, 96 clinicians engaged in a detailing session, and 15 were physicians in a leadership role (i.e., chief of primary care, clinic lead, chief quality improvement; Table 1). All participating clinicians engaged in direct patient care except 8 of the clinical leaders who were influential in making site-level LCS decisions and, therefore, whose input was deemed important for this project. The sessions ranged from 4 to 40 min, lasting an average of 13 min. Five of the 7 VAMCs requested a predetailing introductory presentation with attendance and duration ranging from 14 to 45 clinicians and 15 to 45 min. Meeting details can be found in Table 1. The thematic content analysis identified 6 categories describing the varying degree of clinician adoption potential, as described below (Table 2).
Characteristics of the Academic Detailing Meetings

Range of Primary Care Clinician Views about Adopting a Prediction-Based Approach to Lung Cancer Screening, Using an Encounter-Based Decision Tool (N = 96)
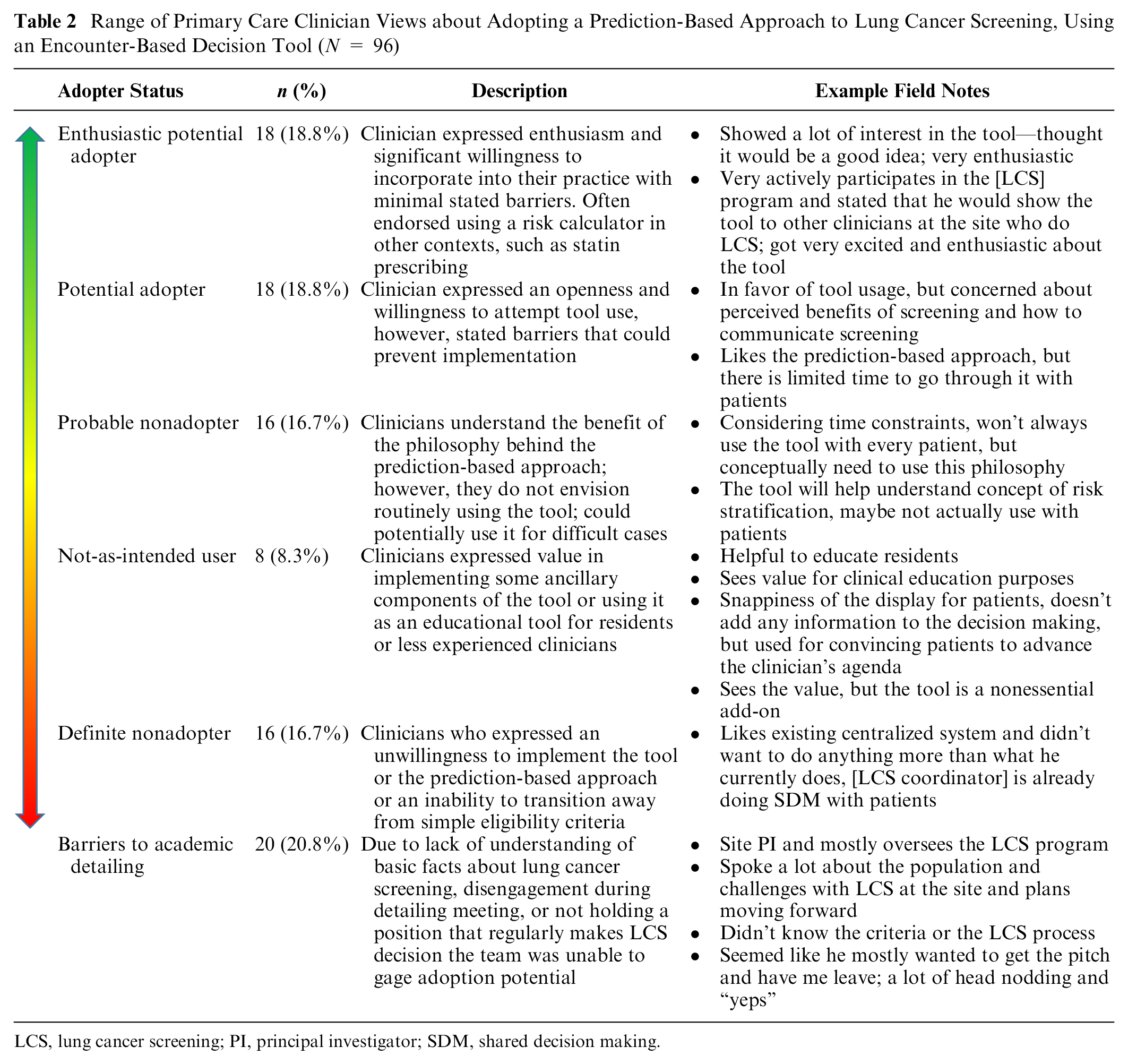
LCS, lung cancer screening; PI, principal investigator; SDM, shared decision making.
Enthusiastic Potential Adopter (n = 18, 18.8%)
Many “enthusiastic potential adopters” strongly supported using the DP encounter decision tool to take a prediction-based approach to SDM for LCS. Some commented that decisions around LCS could be complex, and knowing when to recommend screening can be challenging. Specifically, clinicians expressed uncertainty about whether screening is always the right decision, even when a patient meets the guideline eligibility criteria. These clinicians expressed that access to prediction-based, individualized information and information on how preference-sensitive the decision is for a patient helped navigate these challenges. The tool also allowed these clinicians to articulate scientific uncertainties to their patients. While some enthusiastic potential adopters had initial concerns basing a recommendation on prediction rather than simple age and smoking history cutoffs, by the end of the detailing session, clinicians in this category began to endorse the value of more explicitly including all of a patient’s lung cancer risk factors and consideration of life expectancy into the recommendation. Clinicians asserted that this approach is beneficial for being more proactive and thoughtful, focusing on patients at higher risk, in reasonable health, and thus with more potential to benefit from screening.
The enthusiastic potential adopter category also included many younger clinicians currently in training or recently out of training. The detailer observed that these younger clinicians, especially resident trainees, were often more engaged in the detailing sessions, appeared to value the educational aspects of detailing, and were more open to absorbing new information on practice improvement. In addition, most stated familiarity with using risk prediction and calculators for other aspects of care. Thus, the younger physicians were able to grasp the importance of personalization and move beyond using conventional eligibility criteria toward prediction-based SDM more quickly. These enthusiastic potential adopter clinicians expressed eagerness to use the DP tool to personalize SDM in their routine practice, voicing few to no barriers to implementation.
Potential Adopter (n = 18, 18.8%)
“Potential adopters” also expressed willingness to use DP to personalize SDM for LCS. These clinicians felt the tool allowed them to understand patient-specific predicted risk and net benefit better, guiding them in how strongly to encourage screening for different patients. Several in this category felt the DP tool would help them better identify patients who would be high benefit.
However, unlike enthusiastic potential adopters, these clinicians expressed concerns about the feasibility of using the DP tool to implement personalized, prediction-based SDM in practice. The primary concern was the limited clinic time available to calculate and discuss risk and carry out personalized SDM. With many competing demands and time constraints on clinicians during appointment times, they worried about not having time to include routine tool use in the clinic. A second concern for some was uncertainty about the overall net benefit of LCS. A few potential adopters expressed general skepticism toward the utility of conducting any LCS and worried about overestimating the benefits of screening. While the general idea of adopting a more refined prediction-based approach was appealing, these clinicians were unsure if LCS was the most beneficial clinical context for such an approach.
Probable Nonadopter (n = 16, 16.7%)
“Probable nonadopters” voiced an understanding of the benefits of a personalized, prediction-based approach to LCS but expressed strong perceived barriers to implementation that must be addressed before considering adoption. Many of these clinicians understood the philosophy behind using prediction to personalize decisions. They appreciated the idea of including risk stratification, mentioning that it is particularly beneficial for certain “difficult” or “fuzzy” cases. However, there was a genuine concern about routinely implementing this approach for a single decision amidst the daily work of a busy clinic. Like the potential adopters described above, they emphasized issues with time and competing clinical demands. However, these concerns were stronger for clinicians in this category. Some stated that this type of tool does not fit within their workflow and is too different from how they currently practice. Others voiced concerns that LCS was too low of a priority and that having this tool for a low-priority isolated decision was too granular. A tool informing multiple preventive care decisions would be more appealing, as a more comprehensive tool can help make every similar decision easier. Clinicians in this group also suggested that if the risk factor information were fully automated or prefilled in the tool, it would increase the possibility of engagement. In addition, concerns regarding health system policies were voiced. One clinician voiced frustration over the potential future development of performance measures. At the same time, another expressed concern that if prediction-based referrals for LCS are not the local practice norm, clinician referrals for LCS could be blocked by other specialists, exacerbating the workflow problems.
Not-as-Intended User (n = 8, 8.3%)
“Not-as-intended” users were neutral about prediction-based SDM personalizing how strongly to recommend screening for patients. Instead, they emphasized using the tool for facilitating the education of others (patients or other clinicians and trainees) or for research purposes. These clinicians voiced that the tool and the prediction-based approach seem beneficial for educating clinicians on the nuances of LCS, praising the value of continued medical education. Rather than routinely using this tool with patients during their own clinic, this tool would be better suited as an educational tool for residents and other clinicians who are less confident handling LCS.
Other clinicians in this category discussed how some aspects of the DP encounter-based decision tool, such as the visually appealing display and the individualized information, could be useful patient-facing information. As the detailer recorded in his field notes, these clinicians did not explicitly endorse the idea that LCS could be preference sensitive. Instead, they focused on using the tool to support a clinician-established goal (e.g., to convince a patient to quit smoking by showing them their lung cancer risk) and to encourage what the clinician already determined was the best course of action. The detailer noted these clinicians seemed, therefore, to have such positive underlying beliefs about the value of screening that the tool’s information about individualized risks/benefits and how preference-sensitive the decision is for a patient would not necessarily change their LCS recommendation. They were using the individualized information presented in the DP tool, rather, to help convince the patient to screen or to promote smoking cessation. The tool’s presentation of how preference sensitive the decision is for a patient was thus not perceived as important when making LCS recommendation decisions.
Definite Nonadopter (n = 16, 16.7%)
“Definite nonadopters” expressed disinterest in engaging with or adopting a prediction-based approach for LCS. These clinicians expressed more concerns related to the SDM methods and spoke less directly about concerns related directly to the use of the DP tool. Many clinicians (n = 9) in this category expressed satisfaction and comfort with the current screening process and no desire to change their current practices. There were 2 main reasons expressed for a desire to maintain current status quo. First, some clinicians stated they had been making cancer screening recommendations a certain way for years and did not see a compelling need for adopting a new prediction-based approach. Field-note reflections observed that older clinicians, who had been practicing for many years or endorsed being close to retirement, tended to fall into this category. A second reason is that some clinicians relied on screening coordinators to personalize decision making. Thus, they did not see substantial value in using a prediction-based tool to complete personalized SDM with the patient prior to their referral to a screening coordinator. It was not clearly stated whether these clinicians used any version of SDM in their practice. Rather, they made it clear they were happy with their current practices basing screening decisions on eligibility and when the clinical reminder is triggered and did not want to change.
In addition to those wanting to maintain the status quo, others in this category (n = 5) were unwilling to consider prediction-based SDM because they believed it might conflict with current guideline eligibility criteria. The detailer noted that these clinicians did not like the conceptual complexity of prediction calculators and felt the decision to recommend screening should be based on simple cutoffs derived from trials and endorsed in national guidelines, what they saw as the gold-standard approach. These clinicians voiced that the evidence for personalized risk was not strong enough for incorporating into their practice.
Finally, 2 clinicians who fell into this category stated they would not implement the prediction-based approach for LCS due to strong concerns about the merits of adopting any LCS. They believed the financial cost and burdens of LCS clearly outweigh the, as they perceived, small benefits. Therefore, such clinicians gave low priority to any type of LCS in their clinical practice.
Barriers to Academic Detailing (n = 20, 20.8%)
For various reasons, we were unable to gauge some clinicians’ adoption potential. Specifically, 6 clinicians did not sufficiently participate in the academic detailing visit to provide enough feedback, 3 did not have an LCS-appropriate patient panel, and 3 lacked sufficient LCS knowledge to thoroughly discuss prediction-based SDM for LCS. Finally, 8 clinicians, who held various leadership roles, did not regularly make patient-level LCS decisions and were unable to provide feedback from an individual patient decision-making perspective. Rather, they provided feedback from a clinic management or health system–level perspective.
Discussion
Our qualitative study of 96 primary care clinicians across 6 VA health systems identified significant variations in clinicians’ willingness to use a Web-based clinical encounter decision tool (the DP tool) to personalize SDM for LCS. We identified clinicians along a spectrum of “enthusiastic potential adopters” to “definite nonadopters,” akin to the innovators-to-laggards spectrum in Rogers’ theory of diffusion of innovations. 28 Across these clinician clusters, we found varying degrees of hesitancy around using encounter aids to carry out prediction-based SDM for LCS. Some concerns seemed more focused specifically on SDM or implementing LCS overall rather than risk prediction, but we could not clearly differentiate these elements because this qualitative study was conducted in the context of academic detailing visits that discussed all of these elements as a package (prediction-based SDM for LCS and its practical application using the DP tool). Nonetheless, these findings point to the future steps that might be necessary to apply this approach in routine practice.
The concept of prediction-based SDM was appealing to many of the clinicians detailed in our study (n = 52, 54.2%; “enthusiastic adopters,” “potential adopters,” and “probable nonadopters”). However, to varying degrees, these clinicians voiced concerns about carrying out such an approach in practice. Uptake for these clinicians may be more feasible if predictions are automated and decision support is well integrated into existing EHRs. 16 For instance, top-level information about patient-specific net benefit and preference sensitivity could be automated and presented directly in the clinician’s EHR workflow without any extra clicks, while access to the full encounter-based decision tool is just 1 click away. 25 To make automation more feasible, it will be important to make sure that all of a prediction model’s inputs are available within the health record (or can be imputed in a way that maintains adequate predictive performance in the population18,24,29,30).
Those highly amenable to adopting a prediction-based approach to personalizing SDM may simply need to be made aware that an encounter-based decision tool such as DP exists. 20 For others open to adoption but who have questions or concerns about the practical aspects of carrying out personalized SDM in practice (i.e., “potential adopters,” “probable nonadopters,” and “not-as-intended users”), additional detailing on how to apply predictions to inform very brief, personalized LCS discussions may be necessary.25,26 Return detailing visits, reenforcing that LCS is highly preference sensitive for some eligible patients at intermediate risk, may also be needed for clinicians such as those in the “not-as-intended” group, who see lung cancer risk estimates as a way to strongly encourage all patients meeting simple eligibility cutoffs to be screened.
For a substantial portion of clinicians, a well-integrated decision tool and targeted academic detailing alone may not be enough to facilitate the adoption of prediction-based SDM for LCS. In our study, this could include up to 22 of 96 clinicians detailed (30% if including all “definite nonadopters” and assuming that the 6 clinicians in the “barriers to AD” category who were not sufficiently engaged in the detailing visit were also “hidden” definite nonadopters). These clinicians were often older and less receptive to adopting a different approach to well-established practices around selecting patients for prevention and screening. They would likely need additional incentives to change their SDM approach. This encouragement could come from local champions, leadership at the health-system level, or national guideline recommendations supporting a prediction-based approach to personalizing SDM.4,31 In addition, dedicated screening coordinators trained in implementing personalized, prediction-based SDM for LCS can prevent the need for all clinicians to routinely adopt this approach. Indeed, many clinicians in the “definite nonadopter” category were happy to refer patients to a screening coordinator trained in using DP to tailor the SDM conversation for individual patients. This approach to involving nonclinicians could be an important potential solution for overcoming many clinician-related barriers.
There are several study limitations to consider. These results reflect the clinician’s initial reactions to using the DP tool and taking a prediction-based approach to SDM rather than their beliefs about the topic after longitudinal education and training. However, gauging initial clinician reactions was the goal of this analysis as they are highly informative for guiding future implementation efforts. The context of the QI project could potentially influence how clinicians responded in our detailing discussions. This is unlikely in our study, however, because few if any of the clinicians had heard of the QI effort or engaged with the DP tool prior to the detailing visit. In fact, we observed in this study that detailing enabled us to obtain the candid opinions of front-line clinicians who are often hard to engage otherwise. This ability to get open feedback from a diverse sample of front-line clinicians may be an underappreciated strength of the detailing approach. An inherent limitation of our study design is that the detailer as a person (i.e., age, gender, and educational background) can affect the study participant’s engagement and the credibility of the messages conveyed.32–34 Audio recording of these detailing visits would have likely provided richer qualitative data but was not possible due to the barrier that would have posed on opportunistically engaging busy clinicians during our short detailing visits to participating facilities. Still, our detailing approach and use of field-note data resulted in capturing in-depth feedback from many front-line clinicians and allowed us to learn about a wide range of clinician concerns and perspectives that are likely to affect front-line use of encounter-based tools to personalize SDM. Finally, while we engaged clinicians across multiple sites and the sample size was large for a qualitative study, our study focused only on VA clinicians. The views expressed may not represent the full range of views within other health care settings.
To our knowledge, this is the first study assessing clinician views on using a clinical encounter-based decision tool to take a prediction-based approach to SDM for cancer screening. The limited number of prior studies assessing clinician views on using prediction in decision making have focused on atherosclerotic cardiovascular disease prevention, where guidelines have already been encouraging the approach for years.12,35–37 The LCS context is different. Throughout this study, all guidelines explicitly endorsed SDM for LCS, and the Centers for Medicare & Medicaid Services mandated SDM prior to an initial screen. 32 Our tool and implementation efforts focused on using prediction to inform more personalized SDM for guideline-eligible persons, with the particular emphasis on identifying high-benefit–eligible persons for stronger encouragement. This approach to personalizing SDM has been endorsed in the most recent CHEST guideline on LCS. 4 There is also growing interest in using prediction to identify high-benefit persons missed by current guidelines, particularly high-benefit minorities. 18 This context may help motivate the need for personalized SDM and help facilitate dissemination and implementation of tools such as DecisionPrecision. Nonetheless, our findings are consistent with this prior research with regard to 3 aspects: 1) most clinicians accept the value of prediction for preventive care, 2) some clinicians find it challenging to convert to decision making based on a multivariable risk score (as opposed to using 1 to 2 simple risk factors), and 3) clinicians strongly emphasize the importance of well-integrated and automated tools that limit the effect on clinical workflow.
Conclusions
While many primary care clinicians see great value in considering individualized information when making LCS decisions, more support is needed to implement these tools into routine practice. Beyond automating predictions and integrating decision support within the EHR, 38 important facilitators to implementation likely include having clinical practice guidelines that encourage using prediction 4 and investing in targeted education efforts that provide clinicians with guidance on how to quickly use predictions to personalize screening recommendations and carry out brief SDM. 25 Ultimately, identifying ways to feasibly implement prediction-based decision making is critical for preventive interventions such as LCS, where the clinical benefit can vary dramatically across the target population. Routine use of prediction in this context can enable screening programs to support more equitable screening and realize the principle of “equal treatment for equal benefit.”5,38–40
Supplemental Material
sj-pdf-1-mpp-10.1177_23814683241252786 – Supplemental material for Clinician Perceptions on Using Decision Tools to Support Prediction-Based Shared Decision Making for Lung Cancer Screening
Supplemental material, sj-pdf-1-mpp-10.1177_23814683241252786 for Clinician Perceptions on Using Decision Tools to Support Prediction-Based Shared Decision Making for Lung Cancer Screening by Sarah E. Skurla, N. Joseph Leishman, Angela Fagerlin, Renda Soylemez Wiener, Julie Lowery and Tanner J. Caverly in MDM Policy & Practice
Supplemental Material
sj-pdf-2-mpp-10.1177_23814683241252786 – Supplemental material for Clinician Perceptions on Using Decision Tools to Support Prediction-Based Shared Decision Making for Lung Cancer Screening
Supplemental material, sj-pdf-2-mpp-10.1177_23814683241252786 for Clinician Perceptions on Using Decision Tools to Support Prediction-Based Shared Decision Making for Lung Cancer Screening by Sarah E. Skurla, N. Joseph Leishman, Angela Fagerlin, Renda Soylemez Wiener, Julie Lowery and Tanner J. Caverly in MDM Policy & Practice
Footnotes
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors report grants from VA QUERI during the conduct of the study. Drs. Caverly and Lowery and Ms. Skurla reports grants from VA HSR&D during the conduct of the study. Dr. Wiener serves as deputy chief consultant for the VA National Center for Lung Cancer Screening. In addition, Dr. Caverly has a patent Apache 2.0 issued.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Department of Veterans Affairs Quality Enhancement Research Initiative QUE 15-286 and Department of Veterans Affairs Career Development Award CDA 16-151. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The following authors are employed by the sponsor: Sarah Skurla, Angela Fagerlin, Renda Wiener, Julie Lowery, and Tanner Caverly.
Prior Presentations
Parts of these data were previously presented at the 2019 Society of General Internal Medicine Annual Research Meeting.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

