Abstract
Highlights
We evaluated patients’ preferred and actual decision-making role and decisional conflict following a decision-making interaction about lung cancer screening (LCS).
Concordant decision-making preference was not associated with decisional conflict.
Actual decision-making role was also not associated with decisional conflict.
Efforts to enhance decision-making interactions may not decrease LCS decisional conflict.
Many organizations recommend that clinicians use shared decision-making (SDM) processes when they discuss annual low-dose computed tomography (LDCT) for lung cancer screening (LCS) with their patients.1–5 Authors of a Cochrane review found that SDM using decision aids in many settings improves patients’ knowledge and may improve other outcomes such as decisional conflict. 6 Despite the lack of trials showing the benefits of an SDM approach using LCS decision aids at the time, 7 the Centers for Medicare and Medicaid Services (CMS) required for the first time that clinicians include multiple formalized components of information exchange and SDM.8,9 In 2023, 8 y after this decision, it is still not clear if formalized SDM improves patient-centered outcomes for patients considering LCS in routine care settings.6,7,10,11
Many patients do not participate in guideline-concordant SDM prior to receiving LCS. While requesting reimbursement for SDM and actually engaging in SDM are not synonymous, 12 among patients who had the CMS code for an LDCT in 2016, approximately 10% also had a CMS code for the SDM interaction, with similar low rates in 2017.13,14 Brenner and colleagues 15 found that the average decision-making discussion lasted 1 minute and clinicians seldom performed recommended SDM behaviors. 15 Previously, we qualitatively found that the LCS decision-making interaction between clinicians and patients seldom fulfilled the components of “true” SDM as defined in theoretical models as sufficient information exchange, elicitations of values and preferences, and sharing the decision.16–19 Several studies have reported that most patients and clinicians valued the concepts that underlie SDM but also found discrepancies from guidelines regarding how these concepts were applied during LCS clinical encounters.20–22
To further explore the decision-making interaction, we used quantitative data from a multisite observational study of patients who had decision-making interactions regarding LCS. We aimed to evaluate the association of concordant (congruence between actual and preferred roles) and shared LCS decision making with patient-centered outcomes such as decisional conflict and satisfaction with their clinician generally and their communication processes specifically. Our primary hypothesis was that discordant decision making would be associated with decisional conflict and lower satisfaction.
We will henceforth avoid the term “shared decision-making visit” and use “decision-making interaction” because patients in our previous work often declined to label it as a discussion 21 and, as noted above, this interaction may not include all the recommended elements of SDM. That said, based on previous work by our team, it is likely that all participants received a decision aid (even if they did not recall this receipt in the survey; see below) and had this decision-making interaction. 21
Methods
Setting and Recruitment
We have previously described our observational, nested, mixed-methods study design. 23 Of note, the entire study used a repeated-measures design, but we report only baseline results in this analysis. Briefly, we recruited patients from 3 medical centers with established LCS programs: VA Portland Health Care System (VAPORHCS), Portland, Oregon; Minneapolis VA Health Care System, Minneapolis, Minnesota; and Duke University Medical Center, Durham, North Carolina. We administered surveys developed based on theoretical models of patient-centered communication and SDM.16–19 We obtained institutional review board approval at each site (VAPORHCS #3482; Minneapolis VA #4645-B; Duke #Pro00073394), and each participant completed the informed consent process prior to enrolling.
From March 2016 to April 2019, we recruited patients for a baseline survey after a clinician considered a patient eligible for, but before the patient received, an LDCT (if the patient decided to undergo screening). We contacted potential participants after documentation or confirmation from the relevant clinician that screening was discussed with the patient. VAPORHCS and Duke had centralized LCS programs that required all patients to have a decision-making interaction with a dedicated coordinator (VAPORHCS via telephone, Duke in-person). At the Minneapolis VA, patients could have a decision-making interaction with either their primary care provider (PCP) or a dedicated coordinator. Each program required that patients who interacted with a coordinator receive a formal decision aid and encouraged PCPs to provide one (Miranda et al., for the aids used). 23 We included patients who were eligible for LCS based on their local institution’s criteria (similar to 2013 USP United States Preventive Services Task Force STF eligibility criteria) without reassessing those criteria, with enrollment limited to English speakers. 23 Experienced research coordinators completed the survey with participants over the phone.
Exposures
We assessed concordance between participants’ preferred and actual decision-making roles regarding the LCS decision as the primary exposure and whether participants reported the decision was shared as a secondary exposure. Preferred and actual decision-making roles were assessed for each encounter using the Control Preferences Scale (CPS). 24 The CPS has been used extensively in decision-making research, including when preferred and actual roles were measured simultaneously, and measures the construct defined as “the degree of control an individual wants to assume when decisions are being made about medical treatment.” The CPS is a valid and reliable measure of roles in health care decision making.25,26
Roles are measured with a 5-point Likert scale that ranges from the individual making the treatment decisions, “patient-controlled”; to the individual making the decisions jointly with the clinician, “shared”; to the clinician making the decisions, “provider-controlled.” Participants were asked how they wanted the LCS decision to be made (“preferred decision-making role”) and how the final decision actually was made (“actual decision-making role”) for the decision-making interaction. We assigned 3 categories of decision making (patient controlled, shared, or provider controlled) on the basis of the CPS.27,28 Role concordance was defined as exact agreement (using 3 categories) between participants’ preferred and actual decision-making roles.
Outcomes
We included 4 patient-centered outcome measures. Participants were asked to answer questions about the clinician (PCP or LCS coordinator) who spent the most time discussing LCS with them. If participants did not recall a decision-making interaction about LCS, these communication, experiences, and clinician rating measures were not administered as part of the survey and were counted as missing.
First, to measure our primary outcome, decisional conflict, we used the Decisional Conflict Scale (DCS), a multidimensional tool of 16 items divided into 5 subscores: personal uncertainty, deficits of feeling uninformed, unclear values, inadequate support, and perception that an ineffective choice has been made. 29 Items were scored using a 5-point Likert scale (0 = strongly agree to 4 = strongly disagree) and converted to a 100-point scale. Scores < 25 indicated no decision-making difficulty, ≥25 to ≤37.5 were indeterminate, and scores >37.5 suggested delayed decision making or uncertainty about decision making. 30 As the DCS user manual does not specify how to interpret or analyze missing values, similar to previous research, we excluded responses that were missing more than 6 items from the total score analysis. 31 For participants missing 1 to 5 items, we used adjusted DCS scores in accordance with recommendations. 31 When converting the 100-point scale, we used the total number of completed items to calculate an average (by summing the scores and dividing by the total number of completed answers) and multiplied the resulting number by 25. We did not calculate subscores for respondents who did not answer any of the individual items in the subscore.
Second, we used the Consultation Care Measure (CCM), which is based on the patient-centered communication model, to measure participant-reported communication quality.32,33 The primary measure was the statement, “The overall quality of communication with your provider is excellent,” which was rated on a 7-point Likert scale ranging from very strongly disagree = 1 to very strongly agree = 7. For the analysis, and in keeping with previous work using the CCM, we dichotomized this variable as “high quality” if participants indicated they agreed, strongly agreed, or very strongly agreed (score 5–7) and “low quality” if the response was neutral, disagreed, strongly disagreed, or very strongly disagreed (score 1–4). 34 Missing responses were coded as missing and not included in the analyses.
Third, we used the Consumer Assessment of Healthcare Providers and Systems (CAHPS). We adapted 1 item from the CAHPS survey 3.0 to assess respondents’ perception of the primary clinician who discussed LCS. 35 Participants received the question, “Using any number from 0 to 10, where 0 is the worst provider possible and 10 is the best provider possible, what number would you use to rate this provider?” 36 These ratings were dichotomized in our analysis, with 0 to 8 defined as moderate and 9 to 10 defined as best. 37
Fourth, based on previous work, we asked patients to rate their satisfaction with the experience of the LCS discussion on a 11-point Likert scale. 34 The item was, “Please rate your experience with your discussion around LCS with your provider on a scale of 0–10.” We anchored zero as “I had a very poor experience with my discussion around lung cancer screening” and 10 as “I had a very good experience with my discussion around lung cancer screening.” As in our previous work, we dichotomized the results in our analysis with ratings of 0 to 8 defined as poor experience and ratings of 9 to 10 defined as a very good experience. 34
Other Variables
Self-reported demographic information included age, gender, race, marital status, education, income, and employment status. We also assessed self-reported mental and physical comorbidities, such as history of depression, post-traumatic stress disorder (PTSD), and chronic lung disease. We collected self-reported details of the decision-making interaction and data abstracted from the medical record. We used the Single Item Literacy Screener to assess reading difficulty. 38
Analysis
Descriptive statistics were used to characterize patient variables by preferred LCS decision-making role. The primary outcome was decisional conflict examined as both a continuous and dichotomous variable in separate models. Scores on the DCS were dichotomized 2 different ways in modeling, representing 1) indeterminate decisional conflict and 2) delayed decision making or uncertainty about decision making (i.e., DCS scores >25 and scores >37.5, respectively). Secondary outcomes included communication quality, perception of the clinician who discussed LCS, and patient satisfaction, and these outcomes were dichotomized according to prespecified categories. Separate models were analyzed for each outcome. Exposures included role concordance and actual SDM role; reference groups for these exposures in modeling were role discordance and patient- or provider-controlled decision-making role, respectively. Multivariable linear (for continuous outcomes) and logistic (for dichotomous outcomes) regression models were used, adjusting for age, race/ethnicity, education, marital status, income, and study site. Patients with missing data were excluded from the analyses. Sensitivity analyses included additional adjustments for depression/PTSD as a comorbidity in modeling. Data analysis was performed using Stata, version 16.0 (StataCorp LLC), and 2-sided statistical significance was defined as a resultant P value of <0.05.
Results
Overall, 409 patients undergoing LCS participated from the 3 study sites, and the mean age was 65 y (s = 5.6), 354 (87%) identified as male, 342 (86%) reported White race, 165 (46%) reported a history of lung disease, 203 (56%) reported a history of depression and/or PTSD, and 138 (35%) recalled using a decision aid (Table 1).
Cohort Characteristics Stratified by LCS Preferred Role in Decision Making

COPD, chronic obstructive pulmonary disease; CPS, Control Preferences Scale; DCS, Decisional Conflict Scale; EHR, electronic record; PTSD, post-traumatic stress disorder; VA, Veterans Affairs.
All percentages are from nonmissing data. The column shading serves to highlight the base cohort characteristics stand out from those broken down by preferred decision making role.
Excludes those with unknown CPS preference (n = 18).
Evaluated using the Single Item Literacy Screener (SILS).
COPD, asbestos exposure, or other lung disease.
Participants were asked separate questions regarding if their primary care provider and a screening coordinator discussed lung cancer screening with them.
LCS Decision-Making Role
Among the 391 participants who completed the CPS, 168 (43%), 175 (45%), and 48 (12%) preferred patient-controlled, shared, or provider-controlled decision making, respectively (Table 1). While we did not perform comparative statistics, there were no large differences in preferred LCS decision-making role for most characteristics. Notably, 12% of veterans and 14% of nonveterans preferred a provider-controlled role.
According to participants, 176 (46%), 148 (39%), and 57 (15%) indicated their actual decision making was patient controlled, shared, or provider controlled, respectively. When stratified by preferred LCS decision-making role (data not shown), participant characteristics were similar to those shown in Table 1 stratified by actual role. LCS decision-making role concordance (i.e., agreement between preferred and actual role) occurred for 315 (83%) of patients. Role concordance differed by site as 174 (88%), 97 (80%), and 44 (71%) were concordant at VAPORHCS, VA-Minneapolis, and Duke University, respectively.
Decisional Conflict
We did not observe large differences in DCS scores when stratified by the participant’s reported actual role in LCS decision making (Figure 1). The mean DCS score was 24.0 (s = 14.1) and was slightly higher among patients with a preferred (

Mean Decisional Conflict Scale (DCS) score by (a) preferred and (b) actual lung cancer screening (LCS) decision-making role. Box plot of decisional conflict as measured using the DCS, stratified by participant-reported preferred and actual LCS decision-making role. The midline of each box is the median, the box represents the 25th to 75th percentile, whiskers represent the greatest values excluding outliers, and dots indicate outliers. Horizontal lines (red dashed) for indeterminate decisional conflict (DCS > 25) and delayed decision making (DCS ≥ 37.5).
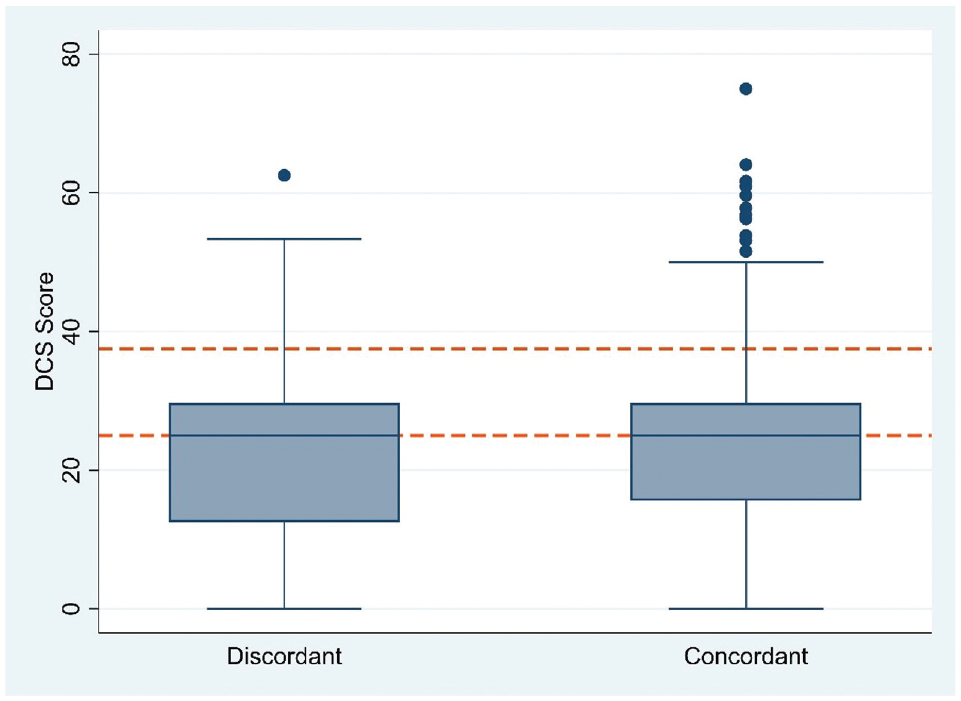
Mean Decisional Conflict Scale (DCS) score by lung cancer screening (LCS) decision-making role concordance. Box plot of decisional conflict as measured using the DCS, stratified by participant-reported LCS decision-making role concordance. The midline of each box is the median, the box represents the 25th to 75th percentile, whiskers represent the greatest values excluding outliers, and dots indicate outliers. Horizontal lines (red dashed) for indeterminate decisional conflict (DCS > 25) and delayed decision making (DCS ≥ 37.5).
DCS Scores by Actual LCS Decision-Making Role and Preferred versus Actual LCS Decision-Making Role (Concordant v. Discordant)

DCS, Decisional Conflict Scale; LCS, lung cancer screening.
DCS subscale scores were similar based on role concordance (Table 3). However, actual provider-controlled decision making had consistently higher scores across all subscales (informed, values clarity, support, uncertainty, effective decision).
Mean DCS Subscale Scores by Actual and Concordant LCS Decision-Making Roles

DCS, Decisional Conflict Scale; LCS, lung cancer screening.
Primary Patient-Centered Outcomes
LCS decision-making role concordance was not associated with DCS score (coefficient = 0.34, 95% confidence interval [CI[:−3.95 to 4.64, P = 0.88) compared with role discordance. Role concordance was also not associated with indeterminate DCS score (adjusted odds ratio [AOR] = 0.64, 95% CI: 0.39–1.05, P = 0.08) or delayed decision-making DCS score (AOR = 0.86, 95% CI: 0.38–1.94, P = 0.71) compared with role discordance. Actual shared LCS decision-making role was not associated with DCS score (coefficient = −0.113, 95% CI: −3.09 to 2.87, P = 0.94) compared with patient- or provider-controlled role (Table 4). Actual SDM role was also not associated with indeterminate DCS score (AOR = 0.80, 95% CI: 0.51–1.24, P = 0.32) or delayed decision-making DCS score (AOR = 0.99, 95% CI: 0.51–1.93, P = 0.98) compared with patient- or provider-controlled role. Sensitivity analyses adjusted for self-reported depression and/or PTSD and were similar to primary results (data not shown).
Associations of Actual Shared and Concordant LCS Decision-Making Roles with Decisional Conflict
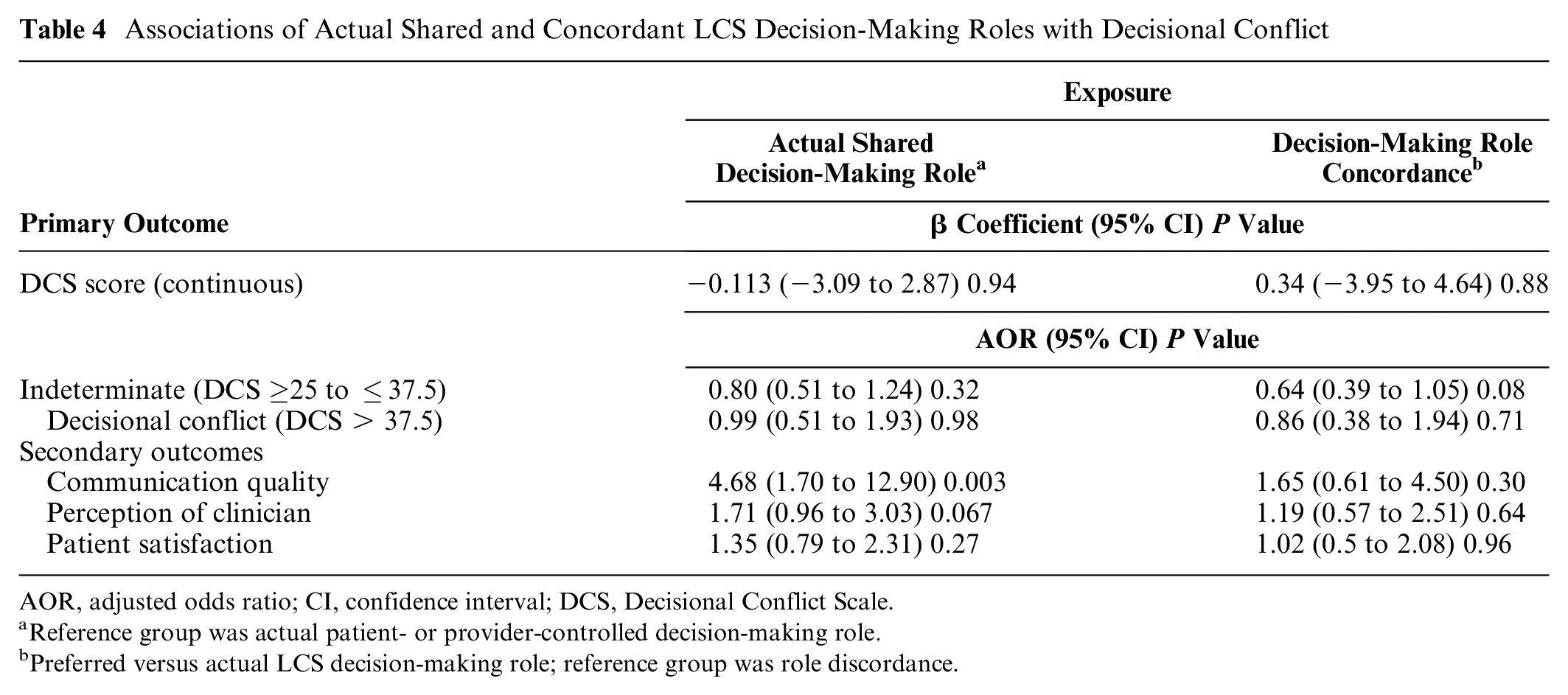
AOR, adjusted odds ratio; CI, confidence interval; DCS, Decisional Conflict Scale.
Reference group was actual patient- or provider-controlled decision-making role.
Preferred versus actual LCS decision-making role; reference group was role discordance.
Secondary Patient-Centered Outcomes
Role concordance was not associated with communication quality (AOR = 1.65, 95% CI: 0.61–4.50, P = 0.30), perception of the clinician who discussed LCS (AOR = 1.19, 95% CI: 0.57–2.51, P = 0.64), or patient satisfaction (AOR = 1.02, 95% CI: 0.5–2.08, P = 0.96) compared with role discordance. Actual SDM role was associated with high communication quality (AOR = 4.68, 95% CI: 1.70–12.90, P = 0.003) compared with patient- or provider-controlled role (Table 4). Actual SDM role was not associated with perception of the clinician who discussed LCS (AOR = 1.71, 95% CI: 0.96–3.03, P = 0.067) or patient satisfaction (AOR = 1.35, 95% CI: 0.79–2.31, P = 0.27) compared with patient- or provider-controlled role.
Discussion
In this large, multisite, observational study of patients considering LCS, more than 80% reported concordance between their preferred and actual decision-making role. Only 14% of participants reported decisional conflict, although almost 60% had an indeterminate level. We did not observe large differences in DCS scores or in validated cut points for indeterminate and decisional conflict levels based on LCS decision concordance or actual LCS decision-making role. We also observed similar DCS subscale scores when stratified by LCS role concordance though somewhat higher scores across all subscales for provider-controlled actual LCS decisions. However, the multivariable analyses were mostly nonsignificant. Concordant LCS decision-making processes were not associated with decisional conflict or satisfaction with clinicians’ communication processes and decision-making interaction experience. Only actual shared LCS decision-making role was associated with increased participant-reported quality of communication with their LCS discussant.
We have conducted several qualitative and quantitative analyses regarding LCS processes in the same settings, so the current analysis should be viewed in light of our previous findings. Our previous qualitative analyses have found discrepancies between many recommended SDM processes and actual practice (e.g., elicitation of values and preferences) when patients make decisions about undergoing LCS.20,21 In line with other research, 15 PCPs reported brief interactions, rarely provided detailed information, and often omitted complicated information, such as overdiagnosis and the occurrence of false-positive results, mostly based on their belief that they implicitly understood the patient’s values and preferences (based on previously established therapeutic relationships).20,21 Dedicated LCS coordinators provided detailed LCS information but reported that patients often did not understand or appreciate this information. 21 Neither PCPs nor screening coordinators elicited patients’ values and preferences, a principle component of SDM. However, PCPs felt that patients implicitly understood them based on prior experiences, and coordinators expressed that many patients already preferred to undergo screening by the time the interaction occurred. Our current results support that these “implicit” assessments described in previous qualitative work may result in decision roles that are concordant with patient preferences most of the time.20,21
In qualitative and quantitative analyses, patients were satisfied with the decision-making interaction overall, although they had little recollection about the harms and benefits of screening, did not consider the decision had been a “discussion,” and seldom found formal decision aids to be useful. 21 Despite this lack of reliance on core SDM processes, most patients expressed positive perceptions of their LCS decision-making interaction and of their clinician although they received or retained little knowledge about LCS.21,39 Basic LCS knowledge was not associated with decisional conflict. 39 Nishi and colleagues 40 also reported patients had low LCS knowledge, but most reported they were involved with the decision as much as they wanted, and 34% reported decisional conflict. Eberth et al. 41 recently reported that patients’ LCS knowledge was low and only weakly related to decisional conflict. As previously mentioned, we have used the phrase “decision-making interaction” throughout as it is possible not all patients in the study underwent SDM with their clinician. Still, consistent with patient-centered communication models and previous work in this area, our findings suggest that LCS information exchange and “true” SDM may be important but may not meaningfully influence other patient-centered outcomes or care experiences. Instead, implicit communication about patients’ values and preferences may be sufficient to achieve decision role concordance.
Among patients undergoing cancer screening in general and LCS in particular, participants in randomized trials who received formalized SDM processes, such as using decision aids, had improved knowledge and often less decisional conflict.7,11,42,43 Similarly, we identified a significant association between an actual SDM role and high communication quality (both as perceived by patients), suggesting that SDM may improve patients’ communication experiences with their physicians or that high-quality communication between patients and physicians is conducive to SDM. However, there is more uncertainty about the benefits for SDM for other patient-centered outcomes and if these associations persist in routine care settings. There is also a paucity of evidence about the association of communication strategies in general and SDM specifically with other outcomes that may be meaningful to patients, such as decision regret after the LDCT, distress (especially if a pulmonary nodule is found), smoking behaviors, and adherence to follow-up recommendations.44,45 We plan to evaluate these outcomes in future work.
Limitations
We enrolled participants from 3 geographically distinct medical facilities after their clinicians confirmed they had undergone a decision-making interaction, and our results may not be generalizable to other settings and populations. Patients who choose to undergo LCS may be more likely to participate in LCS research, reducing the generalizability of the results. We did not directly measure decision-making interactions and therefore cannot be certain that CMS guidelines for SDM were adhered to. However, each site used standard operating procedures for dedicated screening coordinators, and the results of our qualitative work suggest that screening coordinators adhered to CMS guidelines.20,21 In addition, our mixed-methods design, including evaluating LCS clinician communication strategies, moderates concerns about participants’ recall of the interaction. While frequently included as an outcome in communication studies, it is unclear if decisional conflict is truly a patient-centered outcome, and experts ranked it among the least important measures to assess in gauging effectiveness and outcomes of SDM. 45 Decisional conflict is associated with decision regret 46 but can also decrease over time without interventions, and higher levels can be associated with higher-quality communication strategies. 47 Thus, it will be important to study the trajectory of additional patient-centered outcomes after the LDCT for LCS and their associations with clinician communication processes. As multiple aspects of the decision-making interaction were measured at the same timepoint, including decisional conflict and preferred and actual decision-making role, participants may have responded differently than if they had been asked about these at separate visits. Finally, while the quantitative findings presented are intelligible within the context of the article, they are part of a larger mixed-methods study, and it is important to consider them in light of the related qualitative analyses.
Conclusions
CMS mandates that clinicians use core components of SDM models when making LCS decisions with patients. 8 There is some concern that the use of attestation forms for clinicians to “check a box” that they performed SDM when requesting reimbursement for the LDCT for LCS may trivialize the SDM process.48,49 While many clinicians support SDM concepts,20,22,50,51 they report barriers to performing it in routine care settings and infrequently seek reimbursement or document it.13,14,52 It is clear that in real-world settings, the process of LCS decision making rarely adheres to the theoretical tenets of SDM.15,20–22,40,41 However, several studies now suggest that most patients seem satisfied with LCS decision making and often report sharing in the decision as much as they prefer. Furthermore, improving concordant LCS decision-making interactions may not improve decisional conflict, which is not elevated for most patients. It remains to be seen if improved clinician adherence to the core tenets of SDM models or use of new decision aids will improve other patient-centered outcomes in routine care settings.
Footnotes
Acknowledgements
We would like to acknowledge the assistance of support of all the research assistants, project managers, and site principal investigators at each site as well as all the patients who participated in this study.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors declare no financial conflicts of interest with the work presented in this article. RSW and CGS have local and national roles in VA lung cancer screening programs but do not receive additional compensation for these roles. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by an award from the American Cancer Society 128737-RSG-155-01-CPPB, Lung Cancer Screening Implementation: Evaluation of Patient-Centered Care (Dr. Slatore). Dr. Sullivan was supported by a Research Scholar Grant (RSG-20-127-01-CPPB) from the American Cancer Society. VA Health Services Research and Development declined to fund the study. It was supported by resources from the VA Portland Health Care System, Portland, Oregon, and from the VA Boston Health Care System.
Author’s Note
We presented an earlier version of this manuscript as a poster at the Society for Medical Decision Making Annual Meeting, Portland, Oregon, October 20, 2019. The Department of Veterans Affairs did not have a role in the conduct of the study; collection, management, analysis, interpretation of data; or preparation of the manuscript. VA Health Services Research & Development declined to fund this study on multiple occasions. The views expressed in this article are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs or the US government. Lead site investigators: VA Portland Health Care System: Christopher G. Slatore, MD, MS; VA Minneapolis: Anne C. Melzer, MD, MS; Duke University Medical Center: Santanu Datta, PhD, and James M. Davis, MD. Research and clinical teams: VA Portland Health Care System: Sara Golden, PhD, Sarah Ono, PhD, Leah Miranda, MPH, Tara Thomas, BS, Philip Tostado, MA, Molly Davis, RN, and Cynthia Sadak, RN; VA Minneapolis: Angela Fabbrini, MPH, Megan Campbell, MPH, Ruth Balk, BA, and Miranda Deconcini, BA; Duke University Medical Center: Jillian Dirkes, MSW, LCSW, Leah Thomas, MS, and Betty Tong, MD.
IRB Statement
Institutional Review Board approval was obtained at each study site (VAPORHCS #3482; Minneapolis VA #4645-B; Duke #Pro00073394).

