Abstract
Highlights
People with cervical spinal cord injury prioritize gaining upper extremity function after injury, but few individuals receive information about treatment options.
A newly created patient decision aid (PtDA) provides information about recovery after spinal cord injury and the role of traditional tendon and newer nerve transfer surgery to improve upper extremity upper extremity function.
The PtDA improved knowledge and decreased decisional conflict in this pilot study.
Future work should focus on studying dissemination and implementation of the ptDA into clinical practice.
Keywords
Introduction
Making decisions about elective surgical treatment can be challenging and should consider biological, psychosocial, and other factors.1–3 Thoughtful consideration of all factors is particularly important when considering surgery to restore function in people living with cervical spinal cord injury. Due to paralysis involving the upper extremity, individuals with cervical-level spinal cord injury require varying levels of assistance with many activities of daily living (ADLs), including feeding, bowel/bladder function, locomotion (with the assistance of a wheelchair), and transfers (from bed to chair/vehicle). As such, restoration of upper extremity function is a priority,4,5 and small improvements can result in substantial gains in independence with ADLs, self-esteem, and autonomy6–8 and may reduce the $3 to $5 million US dollars in lifetime care costs for a single person with spinal cord injury. 9
Most spontaneous recovery of movement and muscle strength occurs within 6 to 12 mo of injury. 10 Rehabilitation, including occupational and physical therapy, are vital to gaining strength and incorporating new movement into the performance of ADLs. Surgery can also be used to gain movement. Both tendon and nerve transfer surgeries use expendable functioning donor muscles and nerves to restore absent recipient function such as elbow extension, wrist extension, and hand opening and closing. Traditional tendon transfers are well-established,11–15 reliable, and restore strong movement but require immobilization and non–weight bearing for weeks to months after surgery.6,16 Newer nerve transfer surgeries have efficacy across settings17–21 and do not require immobilization or non–weight bearing, but there are higher rates of failure to restore strength.22,23 Importantly, nerve transfers are time sensitive, meaning that some individuals may lose the opportunity to undergo this surgery as time passes.17,24–26
Few eligible people with spinal cord injury undergo these surgeries.27,28 There are multiple barriers to care including a lack of 1) information about treatment options,29–31 2) cross-specialty collaboration/referrals,32,33 and 3) provider availability.5,34,35 To overcome this first barrier, detailed information is needed to support informed, preference-consistent choices about appropriate treatment. Patient decision aids (PtDA) effectively convey information about treatment options and assist users in considering the advantages and disadvantages in the context of their own personal circumstances.36–38
Using information from the literature and previous studies, we developed a PtDA that communicates information about surgical and nonsurgical treatment of the upper extremity after spinal cord injury. The purpose of this pilot study was to carefully evaluate this new PtDA in a research setting. We hypothesized that use of the PtDA would improve user knowledge and decrease decisional conflict about upper extremity management for individuals living with cervical spinal cord injury.
Methods
Decision Aid Development
A Web-based mobile-friendly PtDA was developed by our multidisciplinary research team consisting of clinical spinal cord injury experts, patient advocates, PtDA developers, clinicians, and researchers with expertise in patient-centered care. The determination to create an online tool was informed by our previous research, which showed that people with spinal cord injury prefer the use of computers, phones, and tablets for accessing health information. 29 The content of the PtDA was informed by the current literature, results from the European Multicenter Study about Spinal Cord Injury (EMSCI), 39 and a prospective mixed-methods cohort study of people living with cervical spinal cord injury (who did and did not undergo surgery) and their caregivers.40–42 Information was summarized and incorporated into the PtDA. The Ottawa Decision Support Framework criteria, 43 International Patient Decision Aid Standard Collaboration (IPDAS),44–47 and other best practice recommendations48,49 were followed. The PtDA can be found at https://healthliteracymedia.wixsite.com/mysite.
The PtDA incorporated salient information about spontaneous recovery after spinal cord injury and functional outcomes and experiences after surgical and nonsurgical (rehabilitation alone) interventions. An implicit values clarification section encourages users to reflect on the various benefits, risks, and uncertainties about the complex and potentially time-sensitive decision to proceed with or forgo an intervention to improve upper extremity function. Health literacy experts ensured the PtDA met presentation, comprehension, and readability goals. An independent multidisciplinary advisory panel reviewed materials. This panel included people living with spinal cord injury, their caregivers, a diverse group of clinicians (including nonsurgeon physicians, spinal cord injury specialists, physical/occupational therapists, social workers, and psychologists who specialize in the treatment of individuals with spinal cord injury), and researchers with expertise in patient-centered care. Further iterative refinement of the PtDA was based on their review and input. This advisory panel was organized to provide a diversity of opinions and included individuals who received upper extremity reconstruction for their spinal cord injury and those who did not. The current iteration includes information on the natural history after spinal cord injury and expected recovery without surgical intervention, descriptions of nerve and tendon transfer surgeries, and a discussion of factors that could affect choice (financial situation, caregiver availability, expected gains of function, risks of surgery, and tolerance of immobilization). The values clarification section contains a downloadable file that users can share with their treating providers.
Testing the Decision Aid
Adults between 18 and 80 y of age with midcervical spinal cord injury were recruited for study participation between August 2021 and September 2022. Due to the COVID-19 pandemic, these individuals were recruited online, through spinal cord injury newsletters, mailing lists, support groups, blogs, and social media. Recruitment partners included organizations/Web sites such as Paralyzed Veterans of America, North American Spinal Cord Injury Consortium, and Northern California Spinal Cord Injury Foundation. Eligibility was determined through a screening survey that interested participants completed online, via the secure Research Electronic Data Capture (REDCap) system.50,51 Participants with cervical-level spinal cord injury were included if they endorsed limited hand function, wheelchair dependency, and English language fluency. Individuals were excluded if they had previously undergone nerve or tendon transfer surgery. Those eligible provided their contact information for study team follow-up. After an informed consent process and study enrollment, participants were provided a unique link to the testing instruments in REDCap. They completed surveys about demographic and clinical characteristics.
A de novo 10-item knowledge survey was created and revised by the research team with input from the advisory board and health literacy consultants. Questions were used to assess participant understanding of natural recovery after spinal cord injury, surgical and nonsurgical means to improve upper extremity function, and other information included in the PtDA (Supplementary Table 1); answers were true, false, or unsure, and participants were provided the answers at the end of study participation. Correct responses were added, and a score was created to indicate the number of correct responses out of the total possible, yielding a percentage correct; unsure responses were counted as incorrect. Decisional conflict (a state of uncertainty when asked to make a decision related to care) was assessed and scored using the validated 4-item SURE measure, yielding responses of either decisional conflict or no decisional conflict. 52 After the baseline assessment, participants were provided a link to interact with and review the PtDA online.
After the PtDA interaction, participants were directed back to REDCap and completed the postintervention procedures. They were readministered the knowledge and decisional conflict measures. They were also asked to complete a validated single-item health literacy screening question 53 (Supplementary Table 2) and answer closed and open-ended questions to assist in future refinement and improvement of the PtDA. The Acceptability Tool 54 was modified and used to query participants about the length, content, style, presence of bias toward one treatment option over another, and overall usefulness of the PtDA (Supplementary Table 2). Users were also asked about how they reviewed the PtDA (on computer and/or phone, alone or with others, and in one setting or over a period of time). Finally, participants were asked an open-ended question about suggestions for how to improve the PtDA. This information was collected within the online study procedure survey as well as verbally during a final follow-up phone call with a research team member. Information provided over the phone was summarized qualitatively.
This study was approved by the Washington University Institutional Review Board (IRB No. 202106018) and the Human Research Protection Office of the US Army Medical Research and Development Command. Informed consent was obtained from all participants; they received a $30 gift card upon completion of all study activities.
Data Analysis
An a priori power analysis was performed based on the literature and expected findings using the validated decisional conflict measure as the primary outcome meaure.55,56 Recommendations for prospective randomized clinical trials anticipate an effect size of 0.3 to 0.4; we assumed an effect size of 0.4 and sought to recruit 52 participants.
The normality of continuous variables was assessed with a Shapiro-Wilk test. Knowledge survey results before and after viewing the PtDA were compared using a paired-sample t test. Decisional conflict results were compared using a chi-squared test. Feedback on the PtDA was reported with descriptive statistics. Normally distributed variables were reported with mean and standard deviation (s). Nonnormally distributed variables were reported with median and interquartile range (IQR). Subgroup analysis was also performed as numbers permitted. Statistical analysis was performed using Prism (Graphpad, San Diego, CA). Only individuals who completed the knowledge and decisional conflict measures before and after viewing the PtDA were included in the final analysis.
Funding
This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs, through the Spinal Cord injury Research Program under Award No. W81XWH-17-1-0285. Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the Department of Defense. Grant funding supported research staff salaries and participant reimbursement. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing this report.
Results
Study Participation and Demographics
The screening questionnaire was completed by 170 individuals (Figure 1); of these, 80 were eligible to participate. Of the 67 successfully contacted, 11 were deemed ineligible on further questioning by study personnel as they did not meet the inclusion criteria. Of these 11 ineligible individuals, 4 were diagnosed with additional neuromuscular diseases; 3 were no longer interested in participating; 2 had ineligible injury patterns; and 1 had already received upper extremity reconstruction. An additional individual was excluded as their health data could not be collected confidentially. The remaining 56 individuals were enrolled, and 42 of these (75%) completed the knowledge and decisional conflict measure before and after viewing the PtDA. The median time each participant used to view the PtDA was 30 min (IQR = 22.5). Of these 42 enrollees, all but 1 provided feedback on PtDA acceptability and provided suggestions for improvement.

Study enrollment and participation.
Participant demographics are described in Table 1. Most were male (79%) with a mean age of 54.2 y. Most participants had their spinal cord injury many years ago (mean of 21.8 y). Most identified as non-Hispanic White (67%); 14% identified as Black. Most participants received care through the VA Healthcare system; few had received care through a Spinal Cord Injury Model System center.
Participant Demographics (N = 42)

IQR, interquartile range; PtDA, patient decision aid; SCI, spinal cord injury; VA, s, standard deviation; Veterans Affairs y, year.
States grouped according to the 4 defined US Census Bureau geographic regions (https://www.census.gov/).
Improved Knowledge and Decreased Decisional Conflict
Before viewing the PtDA, participants exhibited a median knowledge score of 60% (IQR = 30). After viewing the PtDA, the median knowledge score increased to 80% (IQR = 20, P < 0.001; Figure 2A). Participants exhibited a median decisional conflict score of 2 (IQR = 2.5) before viewing the PtDA and 4 (IQR = 1) after viewing it. The percentage of participants experiencing decisional conflict (score <4) decreased after viewing the PtDA (33/42, 79% v. 18/42, 43%, P = 0.001; Figure 2B). Viewing the PtDA improved median knowledge scores in both White (20%, P < 0.001, n = 28) and non-White participants (20%, P = 0.011, n = 14) as well as in male (20%, P < 0.001, n = 33) and female participants (40%, P = 0.008, n = 9). The percentage of individuals experiencing decisional conflict decreased in White (22/28, 79% v. 12/28, 43%, P = 0.013) and male (25/33, 76% v. 12/33, 36%, P = 0.003) participants. The percentage of non-White (11/14, 79% v. 6/14, 43%, P = 0.1201) and female participants (8/9, 89% v. 6/9, 67%, P = 0.58) experiencing decisional conflict trended downward after viewing the PtDA; however, these changes were not statistically significant.
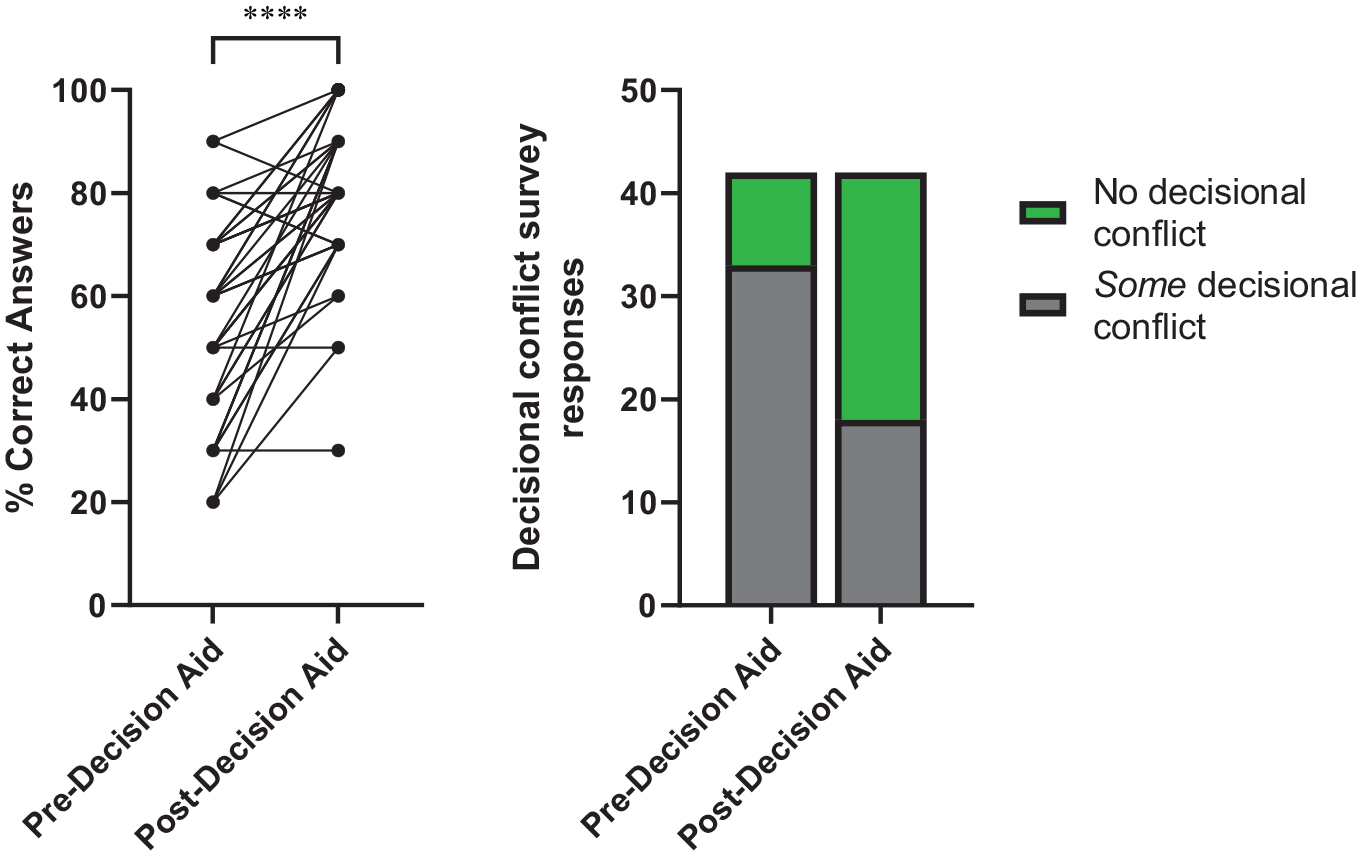
Review of a de novo Patient Decision Aid improved study participant knowledge (Left) and reduced decisional conflict (Right) about treatment options.
High Acceptability
Overall, participants found the PtDA useful (Table 2). They felt the PtDA was an appropriate length (36/42, 86%), provided enough information without being overwhelming (29/42, 69%), and gave a balanced perspective that did not favor either tendon or nerve transfer surgery (37/42, 91%). Participants largely felt the PtDA would be helpful when considering surgery to improve upper extremity function in spinal cord injury (37/42 responded that it would be helpful, 91%).
PtDA Acceptability

IQR, interquartile range; PtDA, patient decision aid; SCI, spinal cord injury.
One participant did not complete all feedback questions.
Suggestions for Improvement
Forty participants provided suggestions for improvement through free response questions within the survey and at the end-study follow-up phone call (Table 3). Participants wanted the PtDA to contain more information on the available nerve and tendon transfer surgeries, outcomes, advantages and disadvantages to surgery and links to additional resources. Seven (17%) study participants reported interest in learning more about upper extremity reconstruction from a health provider. One study participant reported scheduling an appointment with a hand surgeon after study participation and had upcoming plans for a tendon transfer surgery.
Free Response Feedback on the PtDA

PtDA, patient decision aid; SCI, spinal cord injury; UE, upper extremity.
Discussion
This newly developed Internet-based, mobile device–accessible PtDA improved knowledge and decreased decisional conflict in a heterogeneous and diverse population of individuals with cervical spinal cord injury. Although regaining upper extremity function is a priority for these individuals,4,34,57,58 there is a paucity of information describing these treatment options on the internet.5,30,31,34 People with spinal cord injury use the Internet but also rely on getting information from their trusted healthcare providers—physical medicine and rehabilitation, spinal cord injury specialists, and occupational and physical therapists, among others. 29 These nonsurgeon clinicians often lack access to information and knowledge about these treatment options and may have biases against some or all of these surgical treatments.33,58,59
A recent United States study found that only 8% of eligible individuals underwent this restorative surgery. 35 Information dissemination is a critical component of improving access to care; our PtDA successfully improved participant knowledge by 20%. This is similar to results seen in the use of PtDAs for making informed choices about other elective reconstructive procedures. For example, a randomized controlled trial observed that individuals receiving breast reconstruction answered 84.6% of questions correctly on their 11-question knowledge survey after viewing a PtDA; those who did not answered 58.2% of questions correctly, comparable to the observations reported in this study. 60 Our PtDA also substantially improved decisional conflict, which is encouraging; however, this needs further testing in the clinical setting.
People living with spinal cord injury want information about surgical and nonsurgical treatment options to gain upper extremity function, and they want the information early after injury and often. With modification, this newly developed PtDA could be adapted to fill the information void around upper extremity function in cervical spinal cord injury.30,31
Prior to use in routine care, however, study participants suggested modifications to the existing PtDA. To be a more useful informational tool, the PtDA could contain additional information about outcomes, surgery types, and timing of surgery. Respondents had mixed opinions on the title of the PtDA, indicating that it could be changed to improve clarity. Iterative testing and modification is an important component of PtDA development. The suggested modifications will assist in the creation of a more effective clinical tool.
While this study presents promising results, it has limitations. Study participants were recruited with the assistance of several national and regional organizations serving the spinal cord injury population. However, our study population may not reflect the US spinal cord injury population at large, which may limit generalizability. Our study group was 79% male and 67% White, consistent with the demographics of new spinal cord injury cases in the United States since 2015. 61 However, our participants were older and may have different injury causes than that of an ideal surgical candidate. Sub-group analysis among non-White and female participants showed an improvement in knowledge but no change in decisional conflict before and after viewing the PtDA; this deserves future investigation. The knowledge survey has not been previously validated, may be biased toward assessing knowledge of surgery, and should be refined before future research study use. For example, questions about nonsurgical treatments and outcomes could be added. Furthermore, while our results indicate the PtDA could be useful for providing information and reducing decisional conflict, it is not yet clear if access to this tool will lead to substantive changes in clinical practice or clinical outcomes.
Our future work will focus on how best to deliver the PtDA. There are barriers to PtDA implementation within the community. Surgeons who perform these procedures seldom see people with spinal cord injury in a timely fashion. Partnership with nonsurgeon providers, finding opportunities to distribute information and targeted creative methods (such as use of telehealth), should be considered to improve access and care in this population.
Conclusions
This PtDA may improve knowledge of treatment options and reduce decisional conflict about upper extremity reconstructive surgery for people living with cervical spinal cord injury. The findings of this study may be generalizable to improving information dissemination about surgical treatment in other medically and psychosocially complex populations with limited access to specialty care.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683231199721 – Supplemental material for Assessing an Online Patient Decision Aid about Upper Extremity Reconstructive Surgery for Cervical Spinal Cord Injury: Pilot Testing Knowledge, Decisional Conflict, and Acceptability
Supplemental material, sj-docx-1-mpp-10.1177_23814683231199721 for Assessing an Online Patient Decision Aid about Upper Extremity Reconstructive Surgery for Cervical Spinal Cord Injury: Pilot Testing Knowledge, Decisional Conflict, and Acceptability by William Moritz, Amanda M. Westman, Mary C. Politi, DOD Working Group and Ida K. Fox in MDM Policy & Practice
Footnotes
Acknowledgements
The members of the Department of Defense (DOD) Working Group are Catherine Curtin, MD1 (
We thank Health Literacy Media (St. Louis, MO, USA) for PtDA development and the stakeholders who served on our advisory board for assistance with refining the PtDA. Data, analytic methods, and study materials are available to other researchers upon request to the corresponding author.
1Palo Alto Veterans Healthcare System, Department of Veterans Affairs, Palo Alto, CA; 2Division of Plastic and Reconstructive Surgery, Department of Surgery, Washington University School of Medicine, St. Louis, MO; 3St. Louis Veterans Healthcare System, Department of Veterans Affairs, St. Louis, MO.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mary Politi, PhD, is a consultant for UCB Biopharma (2022). Ida K. Fox, MD, received grant funding as PI for “Supporting Patient Decisions About Upper-Extremity Surgery in Cervical Spinal Cord Injury,” Department of Defense, Office of the Congressionally Directed Medical Research Programs (CDMRP), Fiscal Year 2016, Spinal Cord Injury Research Program (SCIRP) Investigator-Initiated Research Award, SC160046: W81XWH-17-1-0285, September 1, 2017–August 31, 2022 (includes two year NCE). Ida K. Fox, MD, has grant funding as the principal investigator for “Expanding Knowledge and Information Delivery around Improving Upper Extremity Function after Cervical Spinal Cord Injury,” Department of Defense, Office of the Congressionally Directed Medical Research Programs (CDMRP), Fiscal Year 2021, Spinal Cord Injury Research Program (SCIRP) Expansion Award—Early-Career Partnering PI Option, W81XWH-22-1-0909, 2022-2025. Ida K. Fox, MD, has received honoraria for lectures on restoration of upper extremity function in cervical spinal cord injury from the following organizations: Department of Rehabilitation Medicine, as part of the Licht Visiting Physician Lectureship, University of Minnesota, MN; Shirley Ryan AbilityLab, as part of the Research Seminar Series, Chicago, IL; Kessler Institute for Rehabilitation, as part of a Grand Rounds lecture and workshop, West Orange, NJ. Ida K. Fox, MD, has served in a leadership position (unpaid) for the following organizations: the American Association for Surgery of the Hand and the American Society for Peripheral Nerve. All other authors have no conflict of interest. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided entirely as part of a W81XWH-17-1-0285 grant from the United States Department of Defense. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The contents do not represent the views of the U.S. Department of Veterans Affairs or the United States Government. This study can be found on Clinicaltrials.gov (“Testing the Decision Aid: Supporting Patient Decisions about Upper Extremity Surgery in Cervical SCI”). Findings from this work were presented at the annual meeting of the Midwestern Association of Plastic Surgeons (May 20–21, 2022, Chicago, IL, USA).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
