Abstract
Introduction
Lung cancer is the leading cause of cancer-related deaths in the United States, accounting for over 135,720 cancer deaths in 2020. 1 A diagnosis of lung cancer is often considered deadly because most patients are diagnosed at advanced stage at which the survival prospect is poor. As of 2020, the 5-year relative survival for lung cancer remains as low as 21%, compared to 69% survival for all cancers. 1 Historically, cancer control and prevention efforts for lung cancer had focused on primary prevention (e.g., smoking cessation) due to a lack of an effective screening approach. The National Lung Screening Trial (NLST) is a large-scale, multicenter trial sponsored by the US National Cancer Institute to compare the effect of low-dose computed tomography (LDCT) and chest X-ray on lung cancer death rates among a high-risk population of current or former heavy smokers. Findings from the NLST confirmed the efficacy of LDCT, as demonstrated by a 20% reduction in lung cancer death compared to chest X-ray. 2 The NLST offers the very first clinical evidence of an efficacious screening procedure for lung cancer and has led to a paradigm shift in lung cancer screening.
Following the NLST trial, the US Preventive Services Task Force (USPSTF) issued a Grade B recommendation for lung cancer screening with LDCT in adults aged 55 to 80 years who are current or former smokers with a 30 pack-year smoking history. 3 Although the prevention provision of the Affordable Care Act mandates insurance to cover preventive services with Grade A or B recommendations from the USPSTF, 4 the USPSTF does not provide billing codes for reimbursement. The Centers for Medicare & Medicaid Services (CMS) began to cover lung cancer screening with LDCT in 2015 and created specific billing codes (G0297) for reimbursement. 5 Unlike other cancer screening procedures covered by CMS, the coverage for LDCT is limited to beneficiaries who share similar characteristics as the eligibility criteria of the NLST. Specifically, those who are between 55 and 77 years old, asymptomatic of lung cancer, and current smokers or former smokers who have quit smoking within the past 15 years with a smoking history of at least 30 pack-years. 5
The CMS coverage policy for LDCT also requires a written order documenting a shared decision making (SDM) visit with smoking cessation counseling when submitting claims for LDCT with a specific billing code, G0296. This unprecedented mandate of SDM by CMS in its coverage of lung cancer screening can be viewed as a strategy to facilitate better understanding of the pros and cons of LDCT given the high false-positive rate reported in the NLST.6,7 In addition, this mandate is considered a personalized cancer screening practice as SDM has long been considered a key component of patient-centered care. It is not clear whether and how prevalent private insurance plans will adopt CMS policy mandating SDM in the coverage of lung cancer screening with LDCT. This information is critically important because many privately insured individuals meet the age eligibility criterion for lung cancer screening; thus, informed decision making facilitated by SDM could potentially spare thousands of working-age adults from the invasive diagnostic procedures following false-positive findings from LDCT.
This study examined the implementation of SDM in the context of LDCT for lung cancer screening among individuals who were under the age of 65 and have private insurance.
Methods
We used MarketScan Commercial Claims and Encounters (CC&E) database from October 1, 2015, to December 31, 2017. The MarketScan Research Databases contain paid claims, encounter data, and enrollment information collected from a selection of large employers, health plans, and government and public organizations, covering private sector health data from approximately 350 payers. 8 The MarketScan CC&E database collects data from active employees, early retirees, ex-employees with continued coverage through Consolidated Omnibus Budget Reconciliation Act, as well as spouses and dependents covered by the employer-sponsored plans. 8 We limited the study population to individuals under 65 years of age to fully capture the practice pattern governed by private insurance because once a person reaches age 65, Medicare will become the primary payer. We included a 3-month time window to allow for discussions that occurred during an SDM visit to take effect on individuals’ subsequent decisions on whether to undergo LDCT for lung cancer screening. We imposed continuous enrollment requirement to allow complete data capture during this observational window and to apply claims-based algorithm to calculate comorbidity scores. 9 To explore whether the rate would be substantially higher with longer duration of follow-up, we conducted a sensitivity analysis with a 6-month time window.
We constructed two study cohorts: an LDCT cohort using Current Procedural Terminology (CPT) code G0297 or S8032 and an SDM cohort via the CPT code for SDM counseling visits (G0296). Although individuals may have more than one LDCT screening or SDM during our 2+ year study period as annual screening is recommended, we limited our study cohort to the first LDCT or SDM identified from the MarketScan data to capture the initial decision making regarding lung cancer screening. The reasons for constructing two study cohorts were twofold: 1) to better understand the actual implementation of SDM in lung cancer screening setting and 2) to explore whether SDM may have an impact on individuals’ subsequent decision to undergo screening with LDCT among nonelderly individuals with private insurance. Specifically, for the LDCT cohort, we examined the quarterly trend of receiving SDM on or within 3 months prior to the day of LDCT to determine the extent to which CMS mandate of SDM was implemented in private insurance plans. We then conducted multivariable logistic regression to assess factors associated with the receipt of SDM before LDCT. For the SDM cohort, we reported the quarterly trend of receiving LDCT on or within 3 months after the day of SDM visit to examine individuals’ screening behavior subsequently, followed by multivariable logistic regression to explore factors associated with the receipt of LDCT after SDM. Figure 1 depicts the construction of each study cohort and the corresponding study time frame and duration of observation (i.e., 3-month follow-up in base case analysis v. 6-month follow-up in sensitivity analysis).

Study cohorts and corresponding study time frame.
We conducted Cochran-Armitage test to evaluate the statistical significance of the quarterly time trends, including 8 quarters for the LDCT cohort and 7 quarters for the SDM cohort in the base case analysis. 10 Covariates in the logistic regressions included age, gender, comorbidity score, geographic region, insurance plan type, whether the individual was the primary insurance holder, and calendar year. Although we imposed 3-month continuous enrollment prior to the index event (i.e., LDCT or SDM) to maximize sample size, each person’s comorbidity score was computed from the inpatient and outpatient claims in the 6 months prior to the index event using a modified Charlson algorithm developed by Klabunde et al. 9 For the vast majority of the study cohort, the algorithm used ICD10 diagnoses codes from inpatient and outpatient claims. If a person was not continuously enrolled for the 6-month duration, we designated his/her comorbidity as “unknown.” For those whose index dates were in 1Q2016, we used a combination of ICD-9 and ICD-10 codes to calculate comorbidity scores as the ICD-10 codes were implemented on October 1, 2015. Geographic location was characterized by census divisions. Insurance type was classified into four groups: preferred provider organizations (PPOs), health maintenance organization (HMOs), high deductible plans, and others. High deductible plans included two plan types with high deductible features in the MarketScan data: consumer-driven health plans and high deductible health plans. All analyses were performed in 2020 using SAS version 9.4 (SAS Institute, Cary, NC). This study was exempt from the institutional review board at the authors’ institution.
Results
Receipt of SDM Among Privately Insured Individuals Who Underwent LDCT for Lung Cancer Screening
The LDCT cohort consisted of 21,084 individuals who received LDCT for lung cancer screening between January 1, 2016, and December 31, 2017. The uptake of SDM exhibited a significant upward trend, from 3.1% in 1Q2016 to 8.2% in 4Q2017 (P < 0.001; Figure 2A). A similar trend was found in the sensitivity analyses that extended the observational time window to 6 months prior to LDCT, with the uptake of SDM increasing from 4.8% in 2Q16 to 8.5% in 4Q17 (Figure 2A). Although most privately insured individuals who received LDCT were between age 55 and 77, 4.16% (877/21,084) were younger than 55 and did not meet CMS coverage policy criteria for age (Table 1).
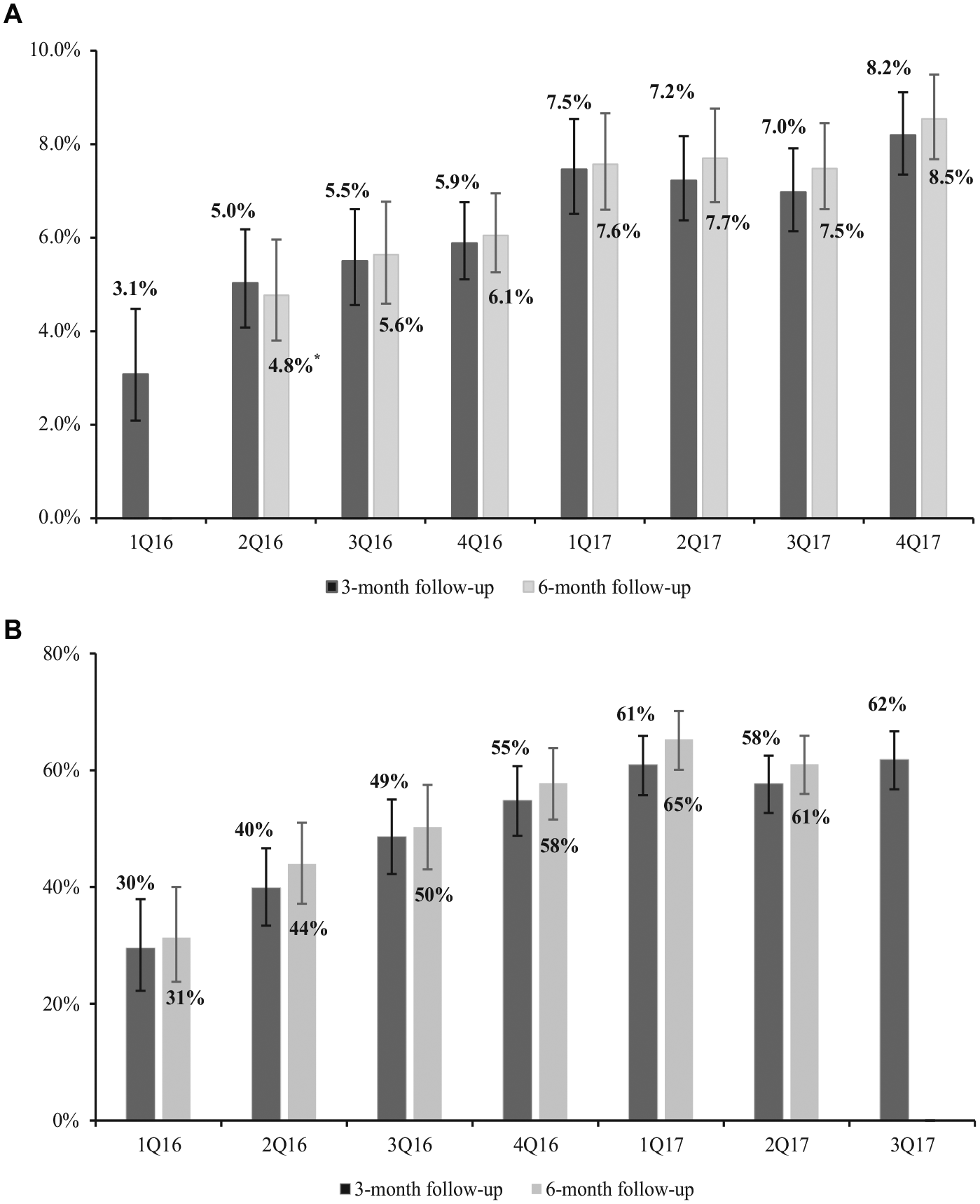
(A) Percent individuals in the LDCT cohort who had an SDM visit on or within 3 months versus 6 months prior to the day of LDCT. (B) Percent individuals in the SDM cohort who underwent LDCT on or within 3 months versus 6 months after the day of SDM visit.
Factors Associated With Having an SDM Visit Prior to LDCT (LDCT Cohort)

CI, confidence interval; LDCT, low-dose computed tomography; OR, odds ratio; SDM, shared decision making.
Source: Authors’ analysis of MarketScan data for 2016-2017.
Results from the logistic regression showed that three factors were significantly associated with having an SDM visit prior to the receipt of LDCT: age, geographic region, and time (Table 1). Individuals who were age 55 years and over (odd ratio [OR] 1.89 for age 55-59 v. <55; OR 1.79 for age ≥60 v. <55) and received LDCT in 2017 (OR 1.44 v. 2016) were significantly more likely to have received SDM within the 3 months prior to LDCT. Compared with the privately insured who underwent LDCT and resided in New England region, those who lived in East North Central and West North Central region were more likely to receive SDM whereas those in Mid Atlantic, West South Central, and Pacific region were less likely to receive SDM. Results from the logistic regression in the sensitivity analysis (i.e., with 6-month observational time window) reached similar conclusions with slight difference in the pattern of geographic variation (results not shown).
Receipt of LDCT Among Privately Insured Individuals Who Had an SDM Consultation for Lung Cancer Screening
The SDM cohort included 2048 individuals with an SDM visit between January 1, 2016, and September 30, 2017. The percentage of individuals who received LDCT within 3 months following the SDM visit increased from 29.5% in 1Q2016 to 61.8% in 3Q2017 (P < 0.0001, Figure 2B). Figure 2B also shows a similar trend but slightly higher rates of LDCT when extending the observation time window to 6 months after SDM. Among the privately insured with a claim indicative of an SDM visit, approximately 6% (122/2,048) were younger than 55 years old.
Results from the logistic regression show a statistically significant association between the receipt of LDCT after an SDM visit and five variables: age, gender, geographic region, plan type, and calendar year. Among individuals in the SDM cohort, those who were older than age 55 (OR 6.27 for age 55-59 and 7.07 for age ≥55 v. <55), enrolled in an HMO plan (OR = 1.45 v. PPO) and had the SDM visit in 2017 (OR = 1.72 v. 2016) were significantly more likely to undergo LDCT. Privately insured individuals who had an SDM visit from the West North Central and East South Central regions were more likely to have undergone LDCT, compared to those from the New England region. Females were significantly less likely to undergo LDCT after an SDM visit (Table 2). Findings from the logistic regression in the sensitivity analysis were largely similar to those in the base case analysis, with small discrepancies in geographic variations (results not shown).
Factors Associated With LDCT Screening Among the Individuals With an SDM Visit (SDM Cohort)

CI, confidence interval; LDCT, low-dose computed tomography; OR, odds ratio; SDM, shared decision making.
Source: Authors’ analysis of MarketScan data for 2016-2017.
Discussion
Private insurance plans tend to adopt reimbursement policies established by CMS. In the case of LDCT, private insurance plans are required to cover this screening service for eligible individuals as it received a Grade B recommendation from the USPSTF. However, the USPSTF recommendation does not mandate insurance coverage for SDM, which was required by CMS. We are interested in understanding the implementation of SDM among privately insured individuals who are not regulated by CMS. Our analysis showed an upward trend in adopting CMS requirement for an SDM visit before LDCT, although the increase has been slow. Nearly 3 years after CMS coverage policy took effect, only 8.2% of privately insured individuals who underwent LDCT for lung cancer screening had a billing record indicating SDM visits. We also found that approximately 40% of the privately insured individuals who had an SDM visit did not undergo LDCT subsequently, suggesting that SDM may have affected screening behaviors of the privately insured by educating individuals about the harm-benefit tradeoff of LCS.
Findings from the NLST were released in 2011 and CMS coverage of LDCT for lung cancer screening did not begin until early 2015. The initial hesitation for covering lung cancer screening largely reflected the concern over an overall 23.3% false positive rate and approximately 9% complication rate from invasive diagnostic procedures reported in the NLST. 11 CMS mandated SDM as a personalized approach to assist health care consumers making informed decision regarding whether to undergo lung cancer screening. Uptake of this well-intended strategy has been low (although increasing) not only among the privately insured reported in our study but also among Medicare beneficiaries. A recent analysis of Medicare claims data found as of December 2016 approximately 10% of beneficiaries who received LDCT had an SDM visit on or in the 3 months prior to the day of screening. 12 Predictors of SDM found in that study were race, sex, geographic regions, and educational attainment at neighborhood level. It is worth noting that although the reimbursement rate of LDCT was similar between Medicare and private insurance (around $242), the median payment of SDM visits (calculated from MarketScan CC&E) was higher than CMS reimbursement rate ($37 v. $30); therefore, the lower rate of SDM reported in our study than that in Medicare (8.2% v. 10%) was unlikely driven by a lower reimbursement rate of SDM in private insurance.
The slow uptake of SDM among individuals who are interested in lung cancer screening may be caused by confusions over who is responsible to deliver this consultation and which decision aid to use as CMS does not offer specific guidance on either.13–15 Despite the availability of decision aids or templates to facilitate SDM in lung cancer screening,16,17 it remains unclear whether SDM should be provided by primary care physicians (PCPs) or imaging facilities.14,15 Although several practice models have surfaced to meet the SDM requirement mandated by CMS, such as the centralized approach proposed by pulmonologists in the Cleveland Clinic, 18 the responsibility of delivering SDM most likely rests upon PCPs, with support from members of their clinical team such as navigators. Therefore, organizations such as HMOs that already integrate PCPs in their care delivery system may be better positioned to implement this CMS mandate. This might explain the slightly higher rate of SDM prior to the receipt of LDCT and also the higher rate of LDCT within 3 months of an SDM visit among HMO enrollees compared to those enrolled in other plans.
Approximately 60% of the privately insured decided to undergo LDCT after an SDM visit, a rate similar to that observed among primary care patients after receiving a decision aid on lung cancer screening. 19 This rate is considerably higher than the rates in the 6% to 15% range estimated among screening eligible individuals from national databases.20,21 Since CMS mandate of SDM does not apply to the privately insured, the higher screening rate observed in the SDM cohort likely reflects the possibility that individuals who had an SDM visit were those more interested in LDCT, thus biasing the rate of lung cancer screening upward in our analysis. This finding suggests that the overall low screening rate observed among the screening-eligible population may be in part driven by a lack of awareness of LDCT as an effective screening modality. Indeed, a recent survey showed that knowledge of lung cancer screening was positively associated with willingness to screen. 22 To improve the screening rate, policy initiatives and interventions should focus on raising the awareness of the use of LDCT for lung cancer screening among patients as well as health care providers.
Our study reported a small percentage of the LDCT cohort were younger than 55 years old, although CMS limits the age eligibility criterion for lung cancer screening to age 55 to 77. The observation of LDCT use in this young age group likely reflects the broader screening eligibility criteria set forth by the National Comprehensive Cancer Network (NCCN). 23 Specifically, the NCCN lung cancer screening guideline lowered the starting age to 50 years for smokers with one additional risk factor for lung cancer. The USPSTF updated its lung cancer screening guideline in July 2020, the draft recommendation widens the eligibility criteria from age 55 to 80 years and a smoking history of 30 pack-years to age 50 to 80 and 20 pack-years. 24 Therefore, the rate of LDCT screening is likely to increase both overall and especially among individuals between the age of 50 and 54 years. An interesting topic for future research is to examine the demand for lung cancer screening among smokers from the theoretical framework of human capital investment, following Kenkel’s work on the demand for breast examinations for breast cancer screening and Pap tests for cervical cancer screening. 25
A recent analysis of the 2017 Behavioral Risk Factor Surveillance System data documented a low rate of lung cancer screening among eligible individuals nationwide (∼14.4%). 21 The study also reported wide variations in lung cancer screening utilization across states, ranging from 6.5% in Nevada to 18.1% in Florida. 21 Our findings of geographic variations both in the rate of SDM before LDCT and the rate of LDCT after an SDM visit suggested that in addition to the geographic variations in screening utilization reported in prior research, the actual screening practice can also differ substantially across geographic regions. Future research should examine whether such practice variation is associated with geographic variations in the supply of PCPs as well as lung cancer screening facilities.
Our analysis had several limitations. First, we were not able to determine the smoking status of individuals because behavior variables were grossly underreported in claims data. 26 Therefore, although we were able to determine whether an individual who received LDCT met the age eligibility criterion, we were not able to ascertain whether the individual met the criteria related to smoking history. Second, MarketScan database shares inherent limitations of commercial claims data. For example, findings from the MarketScan data cannot be generalized to other nonelderly adults without private insurance, such as those enrolled in Medicaid. Neither do findings from this analysis inform whether there is discrepancy in LDCT or SDM by race and ethnicity as such information is not available in MarketScan. Third, the rate of SDM may be underreported in our study because SDM visits were identified from claims data and anecdotal evidence suggested some providers either did not bother to bill for this service or were not aware that a unique billing code existed for the SDM visit—although we did notice that the rate of LDCT screening after SDM appears to be increasing among the privately insured. Last, while it is known that the quality of SDM conversations between patients and providers varies, 27 we were not able to examine the impact of the quality of the SDM conversations on subsequent screening decisions.
In summary, our study provides the first empirical evidence about the real-world implementation of shared decision making for the purpose of advising the use of LDCT for lung cancer screening among nonelderly individuals with private insurance. This information is clinically and policy relevant because many heavy smokers who are under the age of 65 and enrolled in private insurance plans will meet the age eligibility criteria (i.e., 55–77) for lung cancer screening. Claims data from private insurance plans show a growing but low uptake of SDM among privately insured individuals who underwent LDCT. The higher rate of LDCT among individuals who had an SDM visit than the screening rate reported in national studies highlights the importance of raising the knowledge and awareness of the use of LDCT for lung cancer screening among patients and providers.
Footnotes
Acknowledgements
YCTS had full access to all the data in the study and takes full responsibility for the integrity and accuracy of the data analysis. YCTS and YX contributed substantially to the study design and data analysis. YCTS, LMW, and RJV contributed to the writing and interpretation of the manuscript.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ya-Chen Tina Shih received consulting fees, travel, and accommodations for serving on a grants review panel for Pfizer Inc. and an advisory board for AstraZeneca in 2019. All other authors have no conflict of interest to disclose.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was in part supported in part by the Boone Pickens Distinguished Chair for Early Prevention of Cancer to E. Hawk and The University of Texas MD Anderson Cancer Center Duncan Family Institute for Cancer Prevention and Risk Assessment and the NIH/NCI under award number P30CA016672. The authors also acknowledge funding from the National Cancer Institute (R01 CA207216.
