Abstract
Keywords
Since the advent of modern antiretroviral therapy (ART), HIV has transformed into a chronic and manageable disease.1–3 Coupled with the ongoing release of new effective treatments,4–6 the goal of therapy is to extend survival to levels near that of the general population while maximizing quality of life. 7 To that end, researchers have used health status instruments to assess HIV’s impact on functioning and quality of life, thus adding useful information to clinical trials.8–13
The Medical Outcomes Study HIV Health Survey (MOS-HIV) is one of the most frequently used instruments in HIV quality-of-life research and is well-established in terms of its reliability, construct validity, and responsiveness.11,14,15 However, MOS-HIV scores are not suited for calculating quality-adjusted life years (QALYs) for use in cost-effectiveness analysis (CEA) as the instrument does not assign preference-based values to health states. The QALY, generated from preference-based values or utilities, provides a generic health outcome comparable across disease areas and is the preferred measure where CEA is used to inform national health care resource allocation. 16 In practice, preference-based measures, such as the Health Utilities Index Mark 3 (HUI3) or EQ-5D-3L, are infrequently included along with other quality-of-life instruments in trials, thus limiting the ability to assess the cost-effectiveness of HIV interventions from such data.
Mapping, sometimes referred to as cross-walking, may help researchers who wish to conduct CEAs, but are lacking utility values. The fundamental assumption underlying this approach is that the impacts on quality of life captured by the disease-specific health status measure overlap with those captured in the target utility measure. Mapping algorithms can be derived by specifying a regression model that uses the “target” utility measure as the dependent variable and includes the disease-specific health status measure as an independent variable. Parameters from this regression can then be used to predict utility values based on the health status measure gathered in a different population. 17 Bansback and others, 18 for example, developed several models in which either the preference-based EQ-5D-3L or Short Form-6D (SF-6D) were regressed on the domain scores from the Health Assessment Questionnaire Disability Index (HAQ-DI) in patients with rheumatoid arthritis (RA). These algorithms were then applied to studies that measured the HAQ-DI but not utility values. Predicted utility values were used to assess the cost-effectiveness of numerous interventions in RA. 19 Similar mappings have been developed using data from patients with Parkinson’s disease, 20 cystic fibrosis, 21 depression, 22 and from other cohorts.17,23
Huang and others 24 mapped the MOS-HIV onto the EQ-5D-3L, but to our knowledge, there are no studies that map the MOS-HIV onto the HUI3.
Our objective was to estimate and validate mapping algorithms to predict HUI3 and EQ-5D-3L utility values from the MOS-HIV in patients with HIV. We built upon the work by Huang and others 24 by comparing a series of ordinary least squares (OLS) models of increasing complexity with two-part, beta regression-based, and finite mixture models 25 and assessing model performance using external validation tests.
Methods
Data
Data from the VA Cooperative Studies Program Options in Management with Antiretrovirals (OPTIMA) trial formed the estimation data sets. The design and main findings have been reported previously.26,27 Briefly, OPTIMA was a 2 × 2 factorial randomized clinical trial that randomized 368 multidrug-resistant HIV-infected patients to an intended 12-wk treatment interruption or no interruption and to ART intensification (five or more antiretroviral drugs) or standard ART (four or fewer antiretroviral drugs). Patients were eligible for enrollment if they had evidence of serial treatment failure or antiretroviral resistance, were receiving ART, had low CD4 counts (≤300 cells/mm3), and had HIV-1 viral load levels ≥5,000 copies/mL. The OPTIMA study was conducted at more than 70 clinical sites throughout the United Kingdom, Canada, and the United States between 2001 and 2006, with a median follow-up time of 3.2 years. We collected health status (MOS-HIV) and health-related quality-of-life (HRQoL) data (HUI3 and EQ-5D-3L) at baseline; weeks 6 and 12; and every 12 weeks thereafter.
Data from the CIHR Canadian HIV Trials Network Micronutrients and Antioxidants in HIV Infection (MAINTAIN) study formed the external validation data sets for the HUI3 and EQ-5D-3L mapping models.28,29 MAINTAIN was a multicenter randomized double-blind clinical trial evaluating micronutrient supplementation that randomized 171 HIV-infected patients to receive either a micronutrients and antioxidants preparation or multivitamins and minerals for 2 years. Participants were eligible if they were asymptomatic adults with baseline CD4 counts ≥375 and ≤750 mm3 and were ART naïve. MAINTAIN, which enrolled patients between 2009 and 2012, was conducted at 16 sites throughout Canada. Follow-up concluded in 2014. Data on HRQoL, including MOS-HIV, HUI3, and EQ-5D-3L, were collected at baseline and at quarterly intervals through study termination (week 96).
While there are several recommendations currently available for use,30,31 we chose to follow the MApping onto Preference-based measures reporting Standards (MAPS).32,33 The US Department of Veterans Affairs, the UK Medical Research Council, and the Canadian Institutes for Health Research approved the design and conduct of the OPTIMA clinical trial. The Canadian Institutes for Health Research approved the use of MAINTAIN data for the purposes of this study. None of the agencies listed had a role in the writing of this article. The authors have no competing interests.
Instruments
The MOS-HIV is a 30-item descriptive disease-specific questionnaire assessing 10 dimensions of health focused on capturing health impairments that are unique to patients with HIV. 15 The subscales for the dimensions range from 0 to 100 with higher scores indicating better health.
The HUI3 includes 17 questions used to calculate 8 attributes each with 5 or 6 levels for a total of 972,000 possible health states. 34 Preference weights were estimated with valuation data from a random sample of the general population in Hamilton, Ontario, Canada, and were used in a multiplicative model to compute utility values ranging from −0.36 to 1. For all preference-based instruments used in the primary and validation data sets, a score of 0 is equivalent to death and a score of 1 represents full health.
The EQ-5D-3L is a five-item questionnaire covering five dimensions of health. 35 Each dimension contains three levels for a total of 245 possible health states. Value sets for the EQ-5D-3L have been developed for many countries, which enable the estimation of an index for each possible health state. 36 We used Shaw and colleagues’ 37 US population–based EQ-5D-3L preference weights to calculate the EQ-5D-3L index (range: −0.11 to 1). All instruments were in English and paper-based.
In order to assess conceptual overlap between the MOS-HIV and either the HUI3 or EQ-5D-3L, we diagrammed the overlap between HUI3 or EQ-5D-3L domains and MOS-HIV dimensions (Figure 1) and analyzed Spearman correlations between HUI3 or EQ-5D-3L domains and MOS-HIV dimensions or summary scores (Appendix Table 1).

Conceptual overlap between the MOS-HIV dimensions and HUI3 or EQ-5D-3L domains.
Statistical Analysis
All surveys with complete responses to the HUI3 or EQ-5D-3L were included. Per developer guidelines, we substituted missing MOS-HIV values with mean values if more than 2 items in a subscale were missing and if the number of missing items was ≤50%. 15 Given that the relationship between the utility measure (either the HUI3 or EQ-5D-3L) and the MOS-HIV is unlikely to change over time, we pooled data from all time points for the estimation or external validation data sets. We calculated robust cluster-corrected standard errors to take into account multiple observations per subject. All analyses were performed using STATA version 13.1 (StataCorp, College Station, TX) and SAS 9.4 (SAS Institute, Cary, NC).
We reported descriptive statistics as means (standard deviation [SD]) for continuous data and percentages for categorical data, unless noted otherwise, by study.
We applied four different statistical techniques in building our mapping models, OLS, two-part regression, beta regression, and finite mixture, to map to either the HUI3 or EQ-5D-3L index scores. OLS is the most common mapping approach17,23 and has been shown to predict mean values with reasonable accuracy. We evaluated several OLS regression models.
Two-part models have also been used in previous mapping studies
38
as a way to accommodate those with EQ-5D-3L (HUI3) scores of one (perfect health) and any differences they have compared to those with less than perfect health.
Basu and Manca
25
explored several regression models based on the beta distribution. The authors sought to address the characteristics typical of HRQoL data, including negative skew, upper and lower bounds to observed values (truncated supports), and spikes at 1 (perfect health). They found that one- and two-part beta regression models are more robust in estimating covariate effects than OLS. The HUI3 and the EQ-5D-3L data possess many of these typical characteristics including long left tails, an upper bound at 1, and a lower bound determined by tariffs. As such, we evaluated beta regression based on quasi likelihood estimation (Beta QMLE) using the STATA program
Finite mixture models have been used to address the often multimodal distribution of utilities. These types of models allow for unobserved heterogeneity for different classes of individuals by assuming a combination of two or more distributions mixed with unknown probabilities. Previous studies have used finite mixture models to map the Roland Morris Questionnaire and Parkinson’s Disease Questionnaire to the EQ-5D-3L20,39 and estimate the HUI3.
40
Coca Perraillon and others mapped the SF-12 to the EQ-5D-3L and found that finite mixture models outperformed OLS and two-part models. The authors noted that finite mixture model predictions were best at the tails of the distribution.
41
We used the authors’
We fit our models using the OPTIMA data set as our estimation cohort. The models were also applied to the MAINTAIN data set to explore the external validity of the algorithms. Parameters from these models were then applied to the MOS-HIV values to predict the utility values that would have been estimated by either HUI3 or EQ-5D-3L. We compared the predicted values to the actual values obtained in the MAINTAIN trial. We explored the models’ goodness of fit by examining the mean and range of predicted values. We also reviewed the difference between predicted values and the values observed in the OPTIMA and MAINTAIN trials by reporting the mean squared error (MSE), which is the mean of squared differences between observed and predicted utility value scores, and mean absolute error (MAE), which is the mean of the absolute differences between observed and predicted utility value scores. A priori, we used MAE to choose the preferred model. We also reviewed the pattern of MAE across the range of utility values for HUI3 and EQ-5D-3L by reporting predicted values for subsets of the indices.
Results
Study Cohort
OPTIMA collected 4,783 HUI3 surveys, 4,852 EQ-5D-3L surveys, and 4,787 MOS-HIV surveys over a maximum of 6.25 years (median, 3.2 years) for 367 patients. One patient did not complete any HRQoL surveys. Of those surveys, 4,610 included data on both the HUI3 and all MOS-HIV dimensions of health, while 4,662 had data on both the EQ-5D-3L and all MOS-HIV dimensions. MAINTAIN collected 1,163 HUI3 surveys, 1,182 EQ-5D-3L surveys, and 1,163 MOS-HIV surveys over 30.7 months (median, 10.9 months) for 168 patients. Of those, 1,135 included data on both the HUI3 and all 10 MOS-HIV dimensions of health; 1,152 had data on both the EQ-5D-3L and all MOS-HIV dimensions. Three patients failed to complete any HRQoL surveys.
OPTIMA subjects, who made up the estimation sample, were on average older (48 years v. 38 years), included more men (98% v. 83%), were more racially diverse (51% non-White v. 29%), and had a lower median CD4+ cell count (108 v. 494 cells/mm3) than MAINTAIN subjects (external validation sample).
There were also differences in HRQoL data recorded in the estimation and validation data sets (Table 1). For example, the MOS-HIV score for physical functioning was 64.8 (SD 28.3) in the OPTIMA cohort versus 88.2 (SD 23.2) in the MAINTAIN cohort. Preference-based scores also varied, with a mean HUI3 index score of 0.62 (SD 0.31) in OPTIMA and 0.84 (SD 0.21) in the MAINTAIN data set. The mean EQ-5D-3L index score was 0.77 (0.19) in the OPTIMA data set and 0.90 (SD 0.12) in the MAINTAIN data set. As expected, EQ-5D-3L scores were skewed at the upper bound; approximately 22% of OPTIMA surveys and 51% of MAINTAIN surveys had scores of 1.
Health-Related Quality-of-Life Scores for the OPTIMA Estimation Sample and MAINTAIN External Validation Sample

Note: IQR = interquartile range.
Model Estimation
Results from all models are shown in Appendix Tables 3 and 4; results from our preferred models are shown in Table 2. Across the OLS (1a-1c), two-part (1d), and beta QMLE (1e) HUI3 models, improvements in almost all MOS-HIV dimension scores were significantly associated with improvements in preference-based quality of life. Squared terms for pain and social functioning in Models 1b and 1c are always negative and significant (
Parameter Estimates for the Preferred Models Used to Estimate HUI3 Index (Model 1b) and EQ-5D-3L Index (Model 2f)
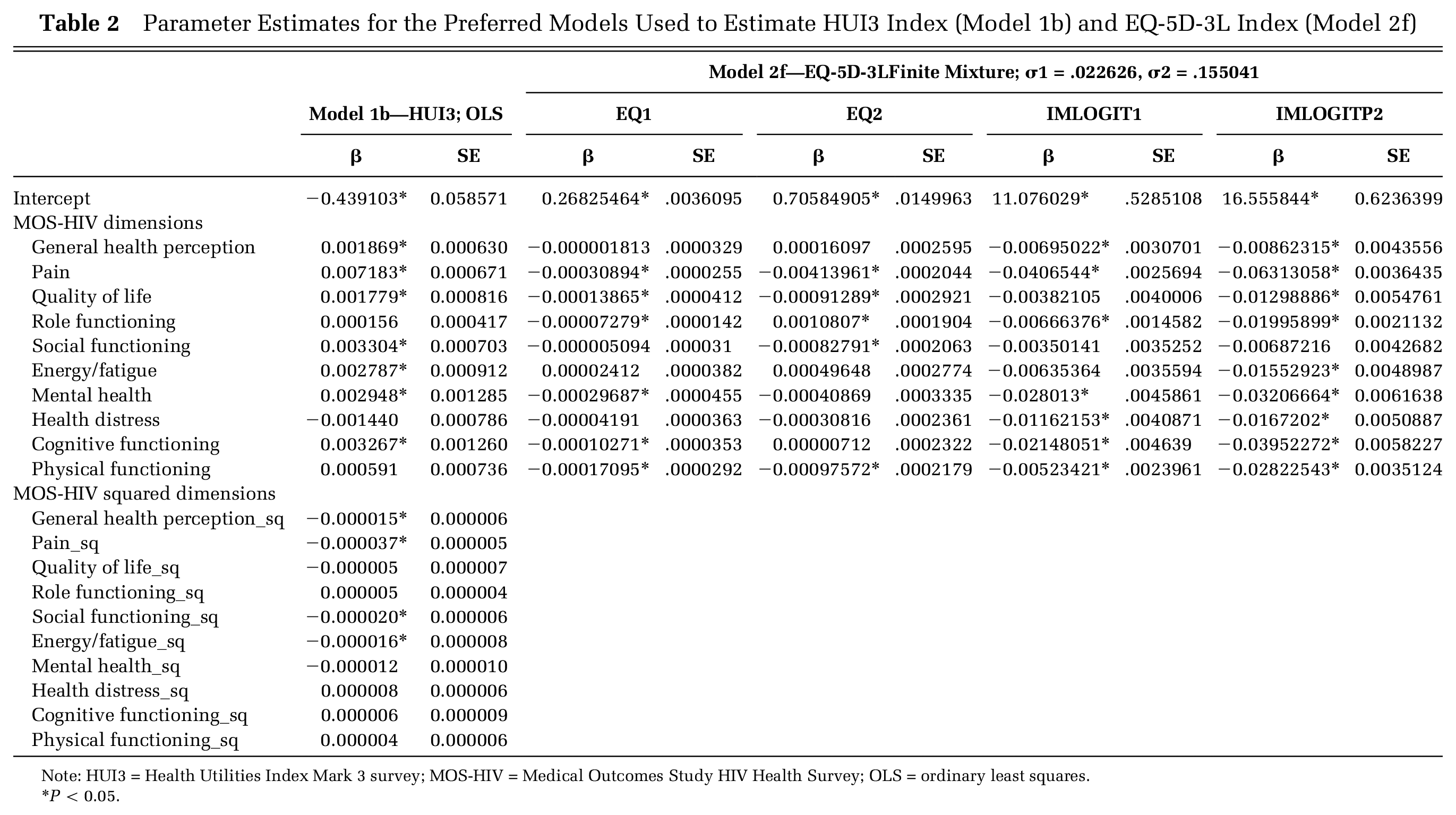
Note: HUI3 = Health Utilities Index Mark 3 survey; MOS-HIV = Medical Outcomes Study HIV Health Survey; OLS = ordinary least squares.
Most main coefficients in the EQ-5D-3L Models 2a (OLS), 2d (two-part), 2e (Beta QMLE) had the expected sign and were significant, indicating that improvements in MOS-HIV dimensions were associated with higher EQ-5D-3L utility value scores. Across all EQ-5D-3L models, improvements in pain and physical functioning were significantly associated with an improvement in EQ-5D-3L index score. As with the HUI3 models, squared terms for pain and social functioning in Models 2b and 2c are negative and significant, as is quality of life (
Model Validation and Performance
All models performed similarly in their ability to predict the mean observed HUI3 utility value score in the validation sample 0.84 (SD 0.21) with means of 0.80 or 0.81 (SD 0.18–0.21) (Table 3). Most models predicted negative values with Model 1c predicting the value closest to the observed minimum value (−0.05 v. −0.14). Ranked by MSE and MAE, the OLS model with squared terms (1b) gave the best predictions (MSE 0.0197 and MAE 0.0961).
Performance of Mapping Models to Predict HUI3 Utilities From MOS-HIV Data
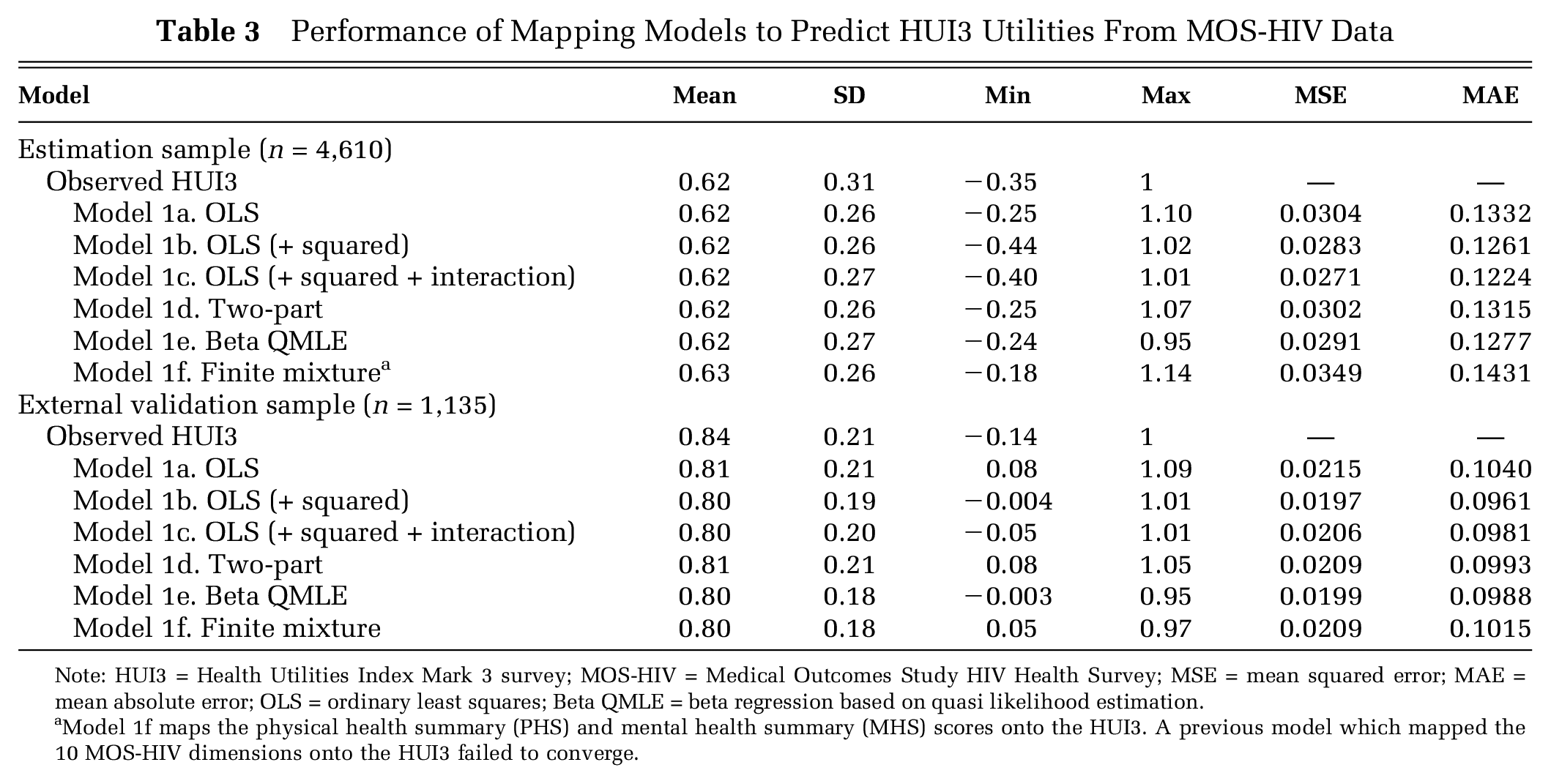
Note: HUI3 = Health Utilities Index Mark 3 survey; MOS-HIV = Medical Outcomes Study HIV Health Survey; MSE = mean squared error; MAE = mean absolute error; OLS = ordinary least squares; Beta QMLE = beta regression based on quasi likelihood estimation.
Model 1f maps the physical health summary (PHS) and mental health summary (MHS) scores onto the HUI3. A previous model which mapped the 10 MOS-HIV dimensions onto the HUI3 failed to converge.
Also, all models were similar in their ability to predict mean EQ-5D-3L in the external validation data set (0.90, SD 0.12) with means of 0.87 or 0.88 (SD 0.09–0.13) (Table 4). The finite mixture model (2f) came closest to predicting the lowest observed EQ-5D-3L utility value score of 0.20 with a lowest predicted score of 0.31. The two-part model (2d), along with the finite mixture model (2f), came closest to predicting the maximum EQ-5D-3L utility value score without exceeding 1. The model with the lowest MSE (0.0083) was the OLS model with squared and interaction terms (2c). If ranked by MAE, however, the finite mixture model (2f) gave the best predictions (0.0567).
Performance of Mapping Models to Predict EQ-5D-3L Utilities From MOS-HIV Data

Note: EQ-5D-3L = EuroQol Five-Dimension Three-Response Levels survey; MOS-HIV = Medical Outcomes Study HIV Health Survey; MSE = mean squared error; MAE = mean absolute error; OLS = ordinary least squares; Beta QMLE = beta regression based on quasi likelihood estimation.
Mean absolute error, regardless of model, was much greater at the lower ends of the utility measure ranges, which indicate poorer health, than at the upper ends (Figure 2a and b). For example, in the Beta QMLE model, for HUI3 utility value scores below 0.40, the mean absolute errors in the external validation sample ranged from 0.21 to 0.31. An observed HUI3 score of, for example, 0.40, might have a complementary predicted score of anywhere between 0.19 and 0.61. In contrast, for HUI3 utility scores above 0.40, the MAE ranged from 0.17 to 0.08. The Beta QMLE HUI3 mapping model performed best (lowest MAE) at the lowest end of the score ranges (<0); the finite mixture EQ-5D-3L model performed best for those with EQ-5D-3L scores <0.40.

(a) Mean absolute errors (MAE) by observed HUI3 score in external validation sample. (b) Mean absolute errors (MAE) by observed EQ-5D-3L score in external validation sample.
Applying the Mapping Algorithm
We have illustrated how to calculate utility value scores from the MOS-HIV by providing an example below as well as examples in an online supplement. Here we have calculated HUI3 scores based on the parameter estimates from the HUI3 OLS+squared terms model (Table 2, Model 1b); the online supplement calculates EQ-5D-3L scores from the finite mixture model estimates (Table 2, Model 2f).

Discussion
In accordance with the MAPS statement,32,33 we developed and compared the performance of several regression models mapping MOS-HIV dimension scores onto HUI3 or EQ-5D-3L utility values. The OLS model with squared terms (1b) is the preferred model with the best goodness of fit (MAE 0.0961) among the HUI3 mapping models; the finite mixture model (2f) is the preferred model among the EQ-5D-3L mapping models with a MAE of 0.0567. To our knowledge, this study is the first to map the MOS-HIV onto the HUI3.
Previous mapping models have reported MAEs at the individual level ranging from 0.0011 to 0.19 17 ; the models presented here performed well within that range with external validation MAEs ranging from 0.0567 (EQ-5D-3L, Model 2f) to 0.1040 (HUI3, Model 1a). We also found that, similar to other mapping studies, there was considerable mean absolute error variation across the range of either HUI3 or EQ-5D-3L index scores. This phenomenon has been found in previous studies.16,17 Hawton and others, 16 for example, mapped the Multiple Sclerosis Impact Scale onto the EQ-5D-3L and found that MAEs for predicted utility value scores increased as EQ-5D-3L scores decreased, with MAE values greater than 0.25 for EQ-5D-3L utility values less than 0.249. It is important to note that variation in MAE at the lower end of the range may be related to smaller sample sizes. Only 5.4% of all of the EQ-5D-3L values collected in the validation sample fell below 0.40.
To date, only Huang and others 24 have mapped the MOS-HIV onto the EQ-5D-3L. Their preferred models included a latent class model and a two-part model with a log-transformed dependent variable. We were unable to compare model performance since the authors did not report estimates of mean absolute error or mean standard error and did not report the parameters for the intercept needed to apply the mapping algorithms to our external validation data set. However, in comparing parameter estimates from Huang’s basic OLS model and our basic OLS model (2a), we found that physical functioning, pain, health distress, and mental health were all statistically significant predictors for both models. Differences between our mapping algorithms may be explained by the fact that Huang’s cohort was younger, had a slightly higher mean baseline CD4 cell count, and was less racially diverse than the OPTIMA sample used to derive our mapping algorithm. The percentage of EQ-5D-3L scores at the ceiling may also play a part; over 40% of patients had the maximum EQ-5D-3L score of 1 in Huang’s cohort compared to just 22% in OPTIMA.
Our study had several limitations. First, we did not include sociodemographic or clinical variables into any of our models. Including these variables would control for more factors that would influence quality of life; however, it would also restrict the application of our mapping algorithms to only those trials that recorded the same information along with the MOS-HIV. However, adding basic sociodemographic characteristics such as age may allow for the fact that measures of HRQoL often do not perform equally across different age groups. Second, we applied US tariffs when calculating EQ-5D-3L scores regardless of the country of origin for the individual quality of life surveys in the estimation and validation data sets. Most scores (78%) in our estimation sample came from US study participants. Choosing a single tariff allowed us to focus on internal validity, despite the loss of some generalizability. We expect the mappings to behave similarly for other tariffs, but we leave this topic to future research. Finally, we found that predictions were less accurate at the lower range of EQ-5D-3L utility values. These problems may limit the applicability of this mapping algorithm for studies with cohorts in very poor health.
Regardless of the HIV cohort, prospective direct or indirect elicitation of utility values is important and is preferred to mapped utility values as it avoids introducing additional uncertainty into the estimates. 30 However, when preference-based methods are not feasible or not available, our externally validated mapping algorithms allow clinicians and researchers to obtain reliable estimates of mean HUI3 and EQ-5D-3L scores from studies that only collected the MOS-HIV. These predicted utility values can be useful in populating health states in economic models.
Footnotes
Acknowledgements
We thank Adam Chow for his assistance with data cleaning and Manisha Desai, PhD, for her assistance with data analysis.
This material is based on work supported by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Cooperative Studies Program; the UK Medical Research Council; and the Canadian Institutes for Health Research. Options in Management with Antiretrovirals is the initial trial of the trinational program involving the aforementioned three agencies. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the US government. The primary funding sources were the Department of Veterans Affairs Cooperative Studies Program, the United Kingdom Medical Research Council, and the Canadian Institutes for Health Research. Supported by the Cooperative Studies Program of the US Department of Veterans Affairs Office of Research and Development, the Canadian Institutes for Health Research, and the UK Medical Research Council.
Coordinating Centers
Coordination of the trial was carried out by each country’s coordinating center. The merged databases were held and maintained at the CTN, Vancouver, British Columbia, Canada; the MRC CTU, London, UK; and the CSPCC, West Haven, CT, USA.
+Optima Study Group
Sandro Cinti, MD (Ann Arbor VAMC); David Rimland, MD (Atlanta VAMC); Anthony Amoroso, MD, Kris Ann Oursler, MD, SCM (Baltimore VAMC); David Johnson, MD, Tiffany Surles, PharmD (Bay Pines VAMC); David Thornton, MD, Judith Strymish, MD, Catherine Fleming, MD (Boston VAMC); Juan Bandres, MD, Catherine Martyn, MS, FNP (Bronx VAMC); Alan George Smulian, MD (Cincinnati VAMC); Robert Bonomo, MD, Gopal Yadavalli, MD, Janet M. Briggs, RN, MSN (Cleveland VAMC); Stephan Hawes, MD (Columbia VAMC); Roger Bedimo, MD, David Margolis, MD (Dallas VAMC); Kenneth Wilson, MD (Durham VAMC); Lisa Dever (East Orange VAMC); Bradley S. Bender, MD (Gainesville VAMC); Constance Pachucki, MD (Hines VAMC); Maria Rodriguez-Barradas, MD (Houston VAMC); Matthew B. Goetz, MD (Los Angeles VAMC); Rodney Wishnow, MD, Stephan Berman, MD, PhD (Long Beach VAMC); Nancy Klimas, MD, Gordon Dickinson, MD (Miami VAMC); Mark Holodniy, MD, Victoria Faria, RN (Palo Alto VAMC); Joel Maslow, MD (Philadelphia VAMC); Chris Reust, MD (Phoenix VAMC); Thomas T. Ward, MD, Melissa Murphy, MD (Portland VAMC); Gregory Anstead, MD, Raymond Chung, MD (San Antonio VAMC); David Looney (San Diego VAMC); Sonia Saavedra, MD, Carlos R. Rivera-Vazquez, MD (San Juan VAMC); Cynthia Gibert, MD (Washington, DC VAMC); Michael Kozal, MD, Michael O. Rigsby, MD (West Haven VAMC).
Professor Brian Gazzard, Chris Higgins, Charlotte Mahuma (Chelsea & Westminster Hospital, London); Professor Margaret Johnson, Dr. Mike Youle, Zoe Cuthbertson (Royal Free Hospital, London); Dr. Alan Winston, Dr. John Walsh, Norman Gariwa (St Mary’s Hospital, London); Dr. Ranjababu Kulasegaram, Isabelle Jendrulek (St Thomas’ Hospital, London); Dr. Brian Angus (Churchill Hospital, Oxford); Dr. Jean Tobin, Linda Tucker (St Mary’s Hospital, Portsmouth); Dr. George Kinghorn, Dr. David Dockrell, Chris Care (Royal Hallamshire Hospital, Sheffield); Dr. Ian Williams, David Cornforth (University College London, Mortimer Market Centre); Dr. Chloe Orkin, James Hand (St Bartholomew’s Hospital, London).
Alain Piché (Centre Hospitalier Universitaire de Sherbrooke, Sherbrooke, Quebec), Sylvie Trottier (Le Centre Hospitalier de l’Université Laval, Quebec, Quebec), Robert O’Brien (Clinique Me′dicale l’Actuel, Montreal, Quebec), Alain Martel (Le Centre Hospitalier de l’Université Laval, Quebec, Quebec), D. William Cameron (The Ottawa Hospital, Ottawa, Ontario), Bill Thompson, Edward Ralph (St. Joseph’s Hospital, London, Ontario), Sharon Walmsley (University Health Network, Toronto, Ontario), Peter Ford, Wendy Wobeser (Kingston General Hospital, Kingston, Ontario), Don Kilby (University of Ottawa Heath Services, Toronto, Ontario), Walter Schlech (Victoria General Hospital, Halifax, Nova Scotia), Brian Conway (Downtown Immunodeficiency Clinic, UBC, Vancouver, British Columbia), John Gill (Southern Alberta Clinic, Calgary, Alberta), Stephen Shafran (University of Alberta Hospital, Edmonton, Alberta).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
