Abstract
Introduction:
Evaluating the antiviral potential of commercially available mouthrinses on SARS-CoV-2 holds potential for reducing transmission, particularly as novel variants emerge. Because SARS-CoV-2 is transmitted primarily through salivary and respiratory secretions and aerosols, strategies to reduce salivary viral burden in an antigen-agnostic manner are attractive for mitigating spread in dental, otolaryngology, and orofacial surgery clinics where patients may need to unmask.
Methods:
Patients (
Results:
The rinse containing CPC significantly reduced salivary SARS-CoV-2 viral load 30 min postrinse relative to baseline (
Conclusions:
Our findings suggest that preprocedural oral rinsing could be implemented as a feasible, inexpensive approach to mitigate spread of SARS-CoV-2 and potentially other enveloped viruses for short periods, which is relevant to clinical procedures involving the nasal and oropharyngeal region.
Knowledge Transfer Statement:
Rinsing with a cetylpyridinium chloride–containing mouthrinse can significantly reduce salivary SARS-CoV-2 viral load for up to 30 min; patients are willing to use mouthrinses in medical and dental settings to limit transmission risk in clinics.
Introduction
More than 676 million cases of SARS-CoV-2 have been reported globally since its emergence in December 2019 (Johns Hopkins University & Medicine 2023). SARS-CoV-2 is a positive-sense single-stranded RNA virus, transmitted primarily through respiratory droplets and aerosolized secretions. Salivary glands are a site of early SARS-CoV-2 viral replication; the oral cavity, pharynx, and saliva act as major viral reservoirs (Zou et al. 2020), with salivary loads as high as 1.2 × 108 copies/mL (To et al. 2020). This stems from the fact that salivary glands and oral mucosa highly expressing the angiotensin-converting enzyme 2 receptor, required for viral envelope fusion with host cells (Chen et al. 2020).
Without proper infection control precautions, orofacial health care delivery can increase transmission risk among patients and providers, including the use of aerosol-generating tools and procedures, proximity to the oropharyngeal region in the absence of patient masking, high patient volumes, and performing work in open-bay clinics (Harrel and Molinari 2004; Suri et al. 2020; Araujo et al. 2021). Furthermore, the SARS virus remains viable in aerosols for at least 3 h (Doremalen et al. 2020). As a result, several groups proposed preprocedural oral rinsing with various active ingredients—ethanol (EtOH), povidone iodine (PI), hydrogen peroxide (H2O2), chlorhexidine gluconate (CHX), and cetylpyridinium chloride (CPC), among others—as a method for mitigating SARS-CoV-2 transmission early in the pandemic. These groups recommended the conduct of human trials to evaluate in vivo efficacy, which we and others pursued (Bidra et al. 2020; Meister et al. 2020; O’Donnell et al. 2020; Carrouel, Gonçalves, et al. 2021; Seneviratne et al. 2021; Martínez Lamas et al. 2022). Guiding these recommendations were several in vitro studies and in vivo human trials, which indicated that these active ingredients could be effective in reducing SARS-CoV-2 viral load and infectivity (Meister et al. 2020; Elzein et al. 2021; Seneviratne et al. 2021). Other trials have demonstrated that a rinse protocol with phthalocyanine derivatives could improve symptoms and reduce the length of hospital stays (Silva Santos et al. 2021), while a β-cyclodextrin and Citrox (bioflavonoids) rinse reduced salivary viral loads over several hours in adults with asymptomatic to mild disease as a transmission barrier method (Carrouel, Valette, et al. 2021; Sbricoli et al. 2023; Zhang et al. 2023)
EtOH, PI, H2O2, CHX, and CPC appear to function by disrupting the viral lipid envelope, preventing viral fusion with host cells (O’Donnell et al. 2020; Muñoz-Basagoiti et al. 2021; Alemany et al. 2022; Alzahrani et al. 2023). Specifically, EtOH has been shown to destroy the viral envelope at high concentrations and increase viral susceptibility to antibody neutralization by destabilizing envelope constituents at lower levels (Bidra et al. 2020; Meister et al. 2020; O’Donnell et al. 2020; Meyers et al. 2021), while H2O2 has been shown to produce oxygen-free radicals that disrupt the viral envelope (O’Donnell et al. 2020). PI’s metabolites include free molecular iodine, which oxidizes membranes and proteins, including host cell receptors, preventing viral attachment (Naqvi et al. 2020). The positive charge of CHX reacts with the negatively charged viral membrane, leading to penetration and leakage. Finally, CPC, a quaternary ammonium compound, has a hydrophobic membrane anchor group effective at viral membrane disruption via surfactant action; CPC also can inhibit interaction between the viral S protein and angiotensin-converting enzyme 2 receptors (Koch-Heier et al. 2021; Alemany et al. 2022; Bañó-Polo et al. 2022; Okamoto et al. 2022). Results regarding the efficacy of these oral rinses are conflicting, with some earlier studies being underpowered (samples size of 2 to 20 participants per group, some without controls).
Upon review of the literature, we proposed to assay salivary viral load as an indicator of potential transmission risk after rinsing with Food and Drug Administration–approved mouthwashes in a large patient population spanning all variant waves to date (in 2023). To this end, we conducted double-blinded randomized controlled trials (RCTs) of commercially available rinses to determine their effect on salivary viral load and participants’ willingness to use oral rinses in a variety of settings. These studies rigorously address the in vivo utility of widely available oral rinses in limiting SARS-CoV-2 viral load. Though COVID-19 immunization is well underway, high transmission rates, virus mutability, and continued emergence of novel variants, as well as a high rate of vaccination and booster declination (Mathieu et al. 2021), make the discovery and implementation of antigen-agnostic antiviral strategies against SARS-CoV-2 an area of scientific, clinical, and public health interest (Thanh Le et al. 2020; Lazarus et al. 2021; NCIRD 2023). Identifying antiseptic mouthrinses with effective antiviral effects on SARS-CoV-2 holds potential for mitigating transmission, particularly in health care settings.
Methods
Human Participants and Study Design
We conducted RCTs investigating the efficacy of commercially available mouthrinses containing 27% EtOH (Listerine Original; Johnson & Johnson Consumer Inc.), 0.1% CPC (Crest Pro-Health; Procter & Gamble), 1.5% H2O2 (Oral-B Mouth Sore; Procter & Gamble), 1% PI (Betadine Antiseptic; Purdue Pharma), and 0.12% CHX (Peridex; Procter & Gamble) as compared with a placebo (0.9% NaCl saline, sterile water) in reducing salivary viral load in participants who were SARS-CoV-2 positive. All studies were reviewed and approved by the Institutional Review Board of the University of North Carolina (UNC) at Chapel Hill. Informed consent was obtained from all participants prior to participation (20-2040, 21-3063, 21-3019). The 2 applicable RCTs were registered with ClinicalTrials.gov (NCT04584684, posted October 14, 2020, enrolling December 18, 2020; NCT05178173, posted January 5, 2022, enrolling January 20, 2022) in accordance with the Food and Drug Administration Amendments Act and National Institutes of Health policy. The first trial enrolled when Delta was the most common variant, while the second trial enrolled when Omicron was most prevalent. The study design followed the Consolidated Standards of Reporting Trials.
Enrollment Criteria and Randomization
Adults 18 to 65 y of age (n = 322) with physician- or laboratory-confirmed SARS-CoV-2–positive status, as indicated by polymerase chain reaction or antigen testing within 10 d of study participation, who met all inclusion and exclusion criteria (Appendix Table 1) were consented and enrolled in 1 of 2 RCTs (Table 1, Appendix Tables 2–4). Participants were randomized by a block randomization schema generated by a statistician, who had no interaction with participants. A staff member, with no patient interaction and no involvement in study visits, performed randomization for group assignment. Trained and blinded study coordinators enrolled participants and conducted visits.
Participant Demographic and Clinical Data from Trials 1 and 2.
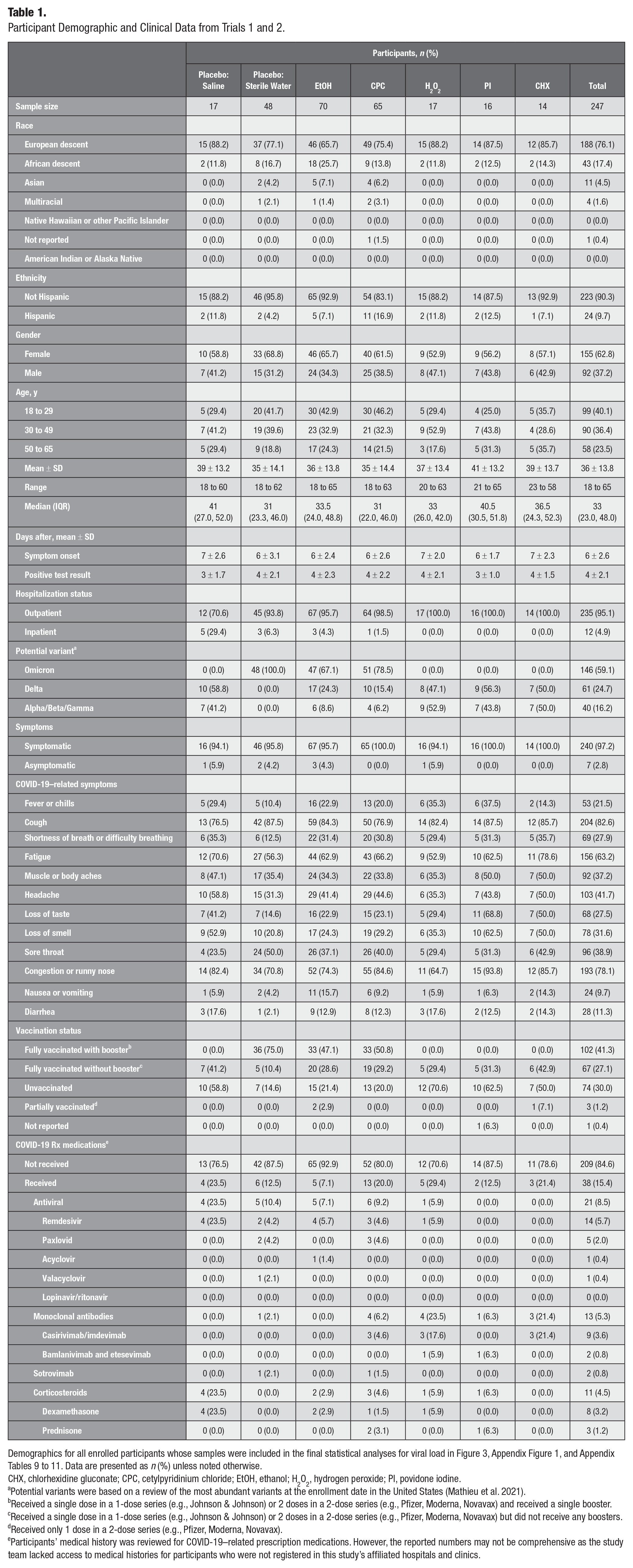
Demographics for all enrolled participants whose samples were included in the final statistical analyses for viral load in Figure 3, Appendix Figure 1, and Appendix Tables 9 to 11. Data are presented as n (%) unless noted otherwise.
CHX, chlorhexidine gluconate; CPC, cetylpyridinium chloride; EtOH, ethanol; H2O2, hydrogen peroxide; PI, povidone iodine.
Potential variants were based on a review of the most abundant variants at the enrollment date in the United States (Mathieu et al. 2021).
Received a single dose in a 1-dose series (e.g., Johnson & Johnson) or 2 doses in a 2-dose series (e.g., Pfizer, Moderna, Novavax) and received a single booster.
Received a single dose in a 1-dose series (e.g., Johnson & Johnson) or 2 doses in a 2-dose series (e.g., Pfizer, Moderna, Novavax) but did not receive any boosters.
Received only 1 dose in a 2-dose series (e.g., Pfizer, Moderna, Novavax).
Participants’ medical history was reviewed for COVID-19–related prescription medications. However, the reported numbers may not be comprehensive as the study team lacked access to medical histories for participants who were not registered in this study’s affiliated hospitals and clinics.
In the first RCT (NCT04584684, enrolling December 18, 2020, to October 25, 2021) participants (n = 128) were randomly assigned to receive 1 of 6 mouthrinses per block randomization as follows: group 1 (EtOH, n = 27), experimental; group 2 (CPC, n = 22), experimental; group 3 (H2O2, n = 19), experimental; group 4 (PI, n = 20), experimental; group 5 (CHX, n = 19), experimental; and group 6 (saline, n = 21), placebo (Appendix Table 2). All mouthrinses are commercially available and used clinically (Appendix Table 5). Participants’ data were excluded from analysis if there was insufficient saliva or the baseline salivary viral load was lower than the assay limit of detection (<1 copy of N1 and N2), resulting in a final sample size of 101: group 1 (EtOH, n = 23), group 2 (CPC, n = 14), group 3 (H2O2, n = 17), group 4 (PI, n = 16), group 5 (CHX, n = 14), and group 6 (saline, n = 17) (Fig. 1A, Table 1, Appendix Table 2).
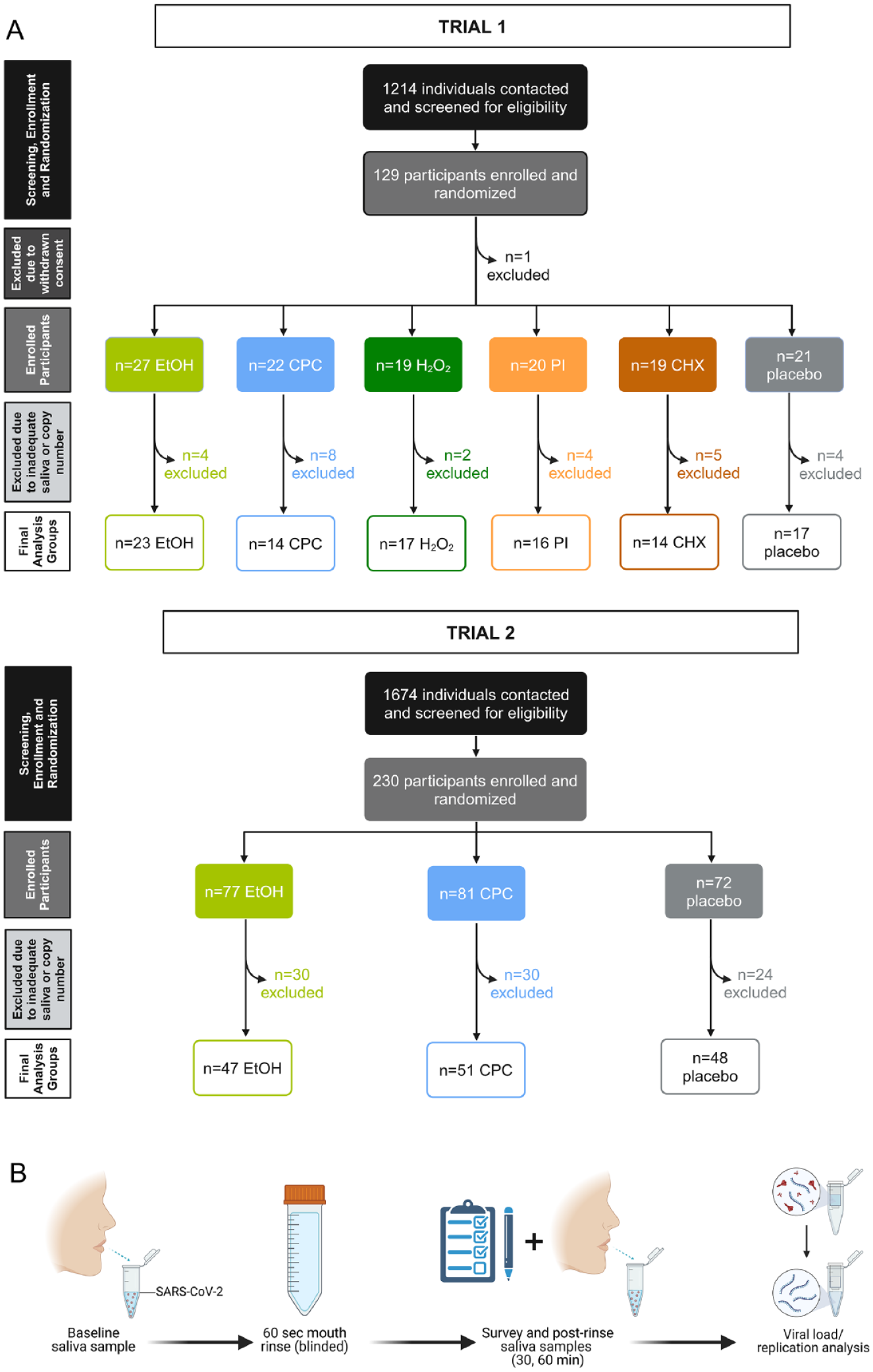
Trial designs and study visit summary. (
The first RCT’s data were analyzed to guide a second RCT, where the 2 most efficacious rinses, in terms of viral load reduction at 30 min postrinse, were included with a placebo control (sterile water). In the second RCT (NCT05178173, enrolling January 19, 2022, to July 12, 2023), 230 participants were enrolled and randomly assigned to receive 1 of 3 mouthrinses based on block randomization as follows: group 1 (EtOH, n = 77), experimental; group 2 (CPC, n = 81), experimental; group 3 (sterile water, n = 72), placebo (Fig. 1A, Appendix Table 3). Participants’ data were excluded from analysis according to the same criteria as in the first RCT, resulting in a final sample size of 146: group 1 (EtOH, n = 47), group 2 (CPC, n = 51), group 3 (sterile water, n = 48) (Fig. 1A, Table 1, Appendix Tables 3 and 4). The second RCT was concluded upon enrolling the planned sample size. Sample size calculations are described in the Appendix.
Study Visit and Clinical Procedures
Participants were seen in the private Clinical Research Space of the Jacox Lab at UNC Adams School of Dentistry and were asked to first provide unstimulated saliva samples (1 to 5 mL) over a 5-min interval, as described by Graves et al. (2024). Following baseline sample collection, participants rinsed for 60 s with an aseptic mouthwash (experimental) or placebo (control), administered in an on-label fashion (Fig. 1B, Appendix Table 5). Unstimulated saliva samples (1 to 5 mL) were then collected for analysis at 30 and 60 min postrinse (Fig. 1B). Between saliva collections, participants were asked to complete study questionnaires.
Clinical and demographic data were collected from questionnaires and charts, including age, self-reported race, ethnicity, medical history, COVID-19–related symptomatology, vaccination history, and treatment course (e.g., diagnosis, medications) (Table 1, Appendix Tables 2–4). Survey questions were developed to query participants’ rinsing experiences and willingness to use mouthrinses in various health care, employment-related, and social settings (Fig. 2). These questions were developed and revised in consultation with a survey specialist at the UNC Odum Institute for Research in Social Science, pretested with 10 laypersons, and iteratively revised until a final draft was approved by the survey expert and study team. The final survey consisted of 24 questions: 4 on rinsing experience, 6 on rinsing sensations, and 14 on willingness to rinse. Survey questions are provided as Appendix Table 6.
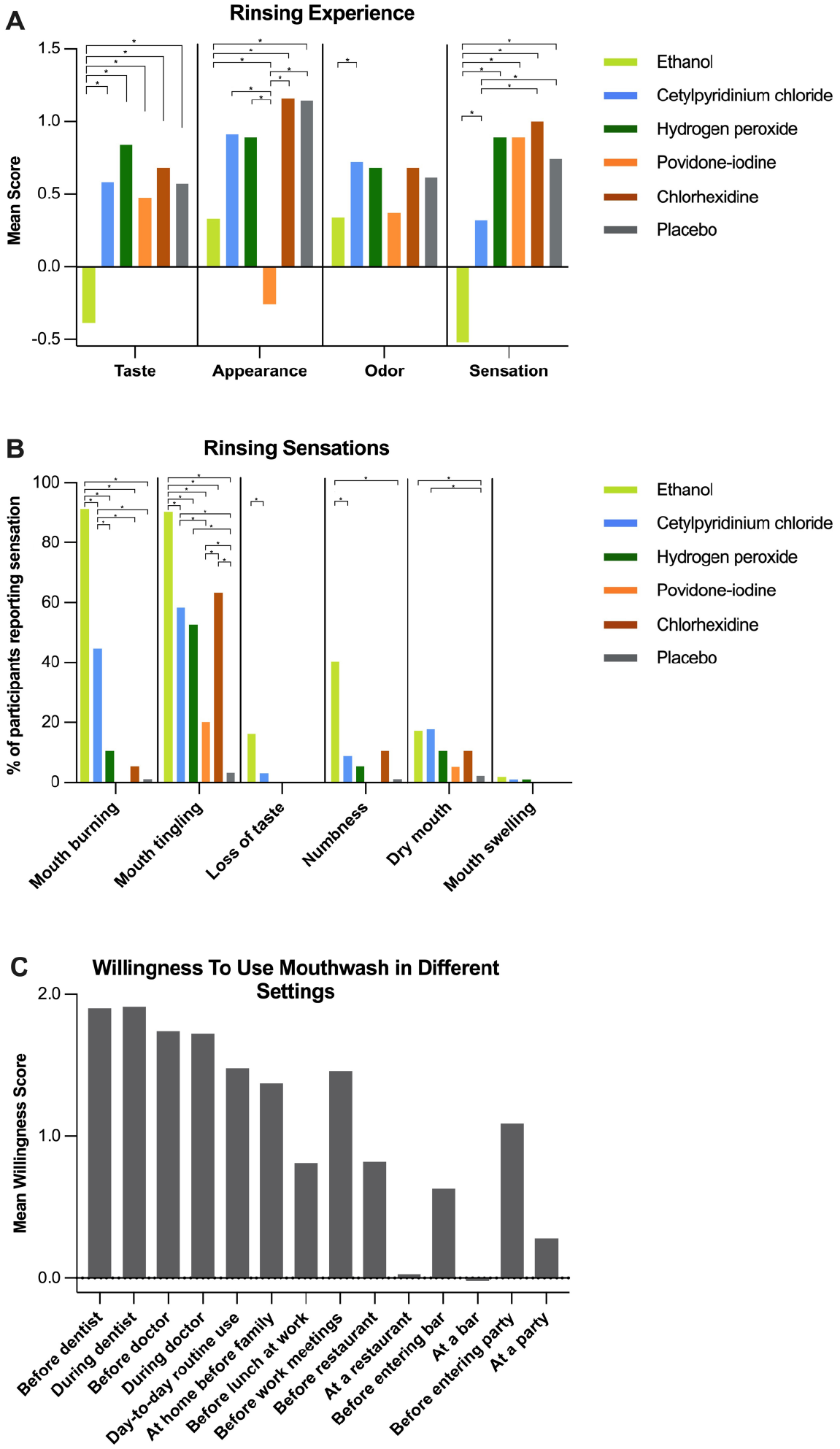
Willingness to use mouthwashes and rinsing experiences. (
Details regarding the survey are included in the Appendix Methods, Appendix Tables 6 to 8, and Figure 2.
SARS-CoV-2 Viral RNA Extraction and Quantitative Polymerase Chain Reaction
Whole saliva samples were transferred on ice to the UNC Respiratory TRACTS Core and aliquoted in prelabeled barcoded tubes for storage at −80 °C. The samples were thawed on ice, cleared by centrifugation (1,000g, 5 min, 20 °C), arrayed into 96-well plates, and stored at −80 °C until extraction. Viral RNA was extracted by a MagMAX Viral/Pathogen Nucleic Acid Isolation Kit according to manufacturer protocols (Applied Biosystems) and including proteinase K treatment on a Kingfisher Flex platform (BindIt 4.0). SARS-CoV-2 RUO qPCR Primer and Probe Kit containing N1-, N2-, and RNase P–specific primers and FAM probes (Integrated DNA Technologies) was used in a standard 20-µL reaction containing 5-µL of nucleic acid extract per sample. Samples were run in technical duplicate. Viral copy numbers were determined through an 8-point standard curve via a commercially available heat-inactivated SARS-CoV-2 Centers for Disease Control and Prevention standard (ATCC VR-1986HK). Resulting copy numbers for each gene and each sample were normalized by log-normal transformation prior to downstream analyses.
Statistical Analyses
Viral load was compared between baseline (i.e., before) and postrinsing at 30 and 60 min separately with a linear mixed-effects model (R software version 4.2.2; Lucent Technologies) including the group, time effect, and potential interactions (random effect was participant effect), adjusted for age and gender (Fig. 3, Appendix Fig. 1). The model was employed separately for 2 viral transcripts (N1, N2). Two hypotheses were tested: first, whether an antiseptic mouthrinse significantly reduced the salivary viral load of SARS-CoV-2 at either 30 or 60 min after rinsing as compared with baseline; second, whether there was a significant difference in viral load between baseline and 30 or 60 min among groups as compared with placebo. RCTs were regarded as batch variables in testing the second hypothesis. After different contrasts were tested by F tests, model coefficients and P values were reported.
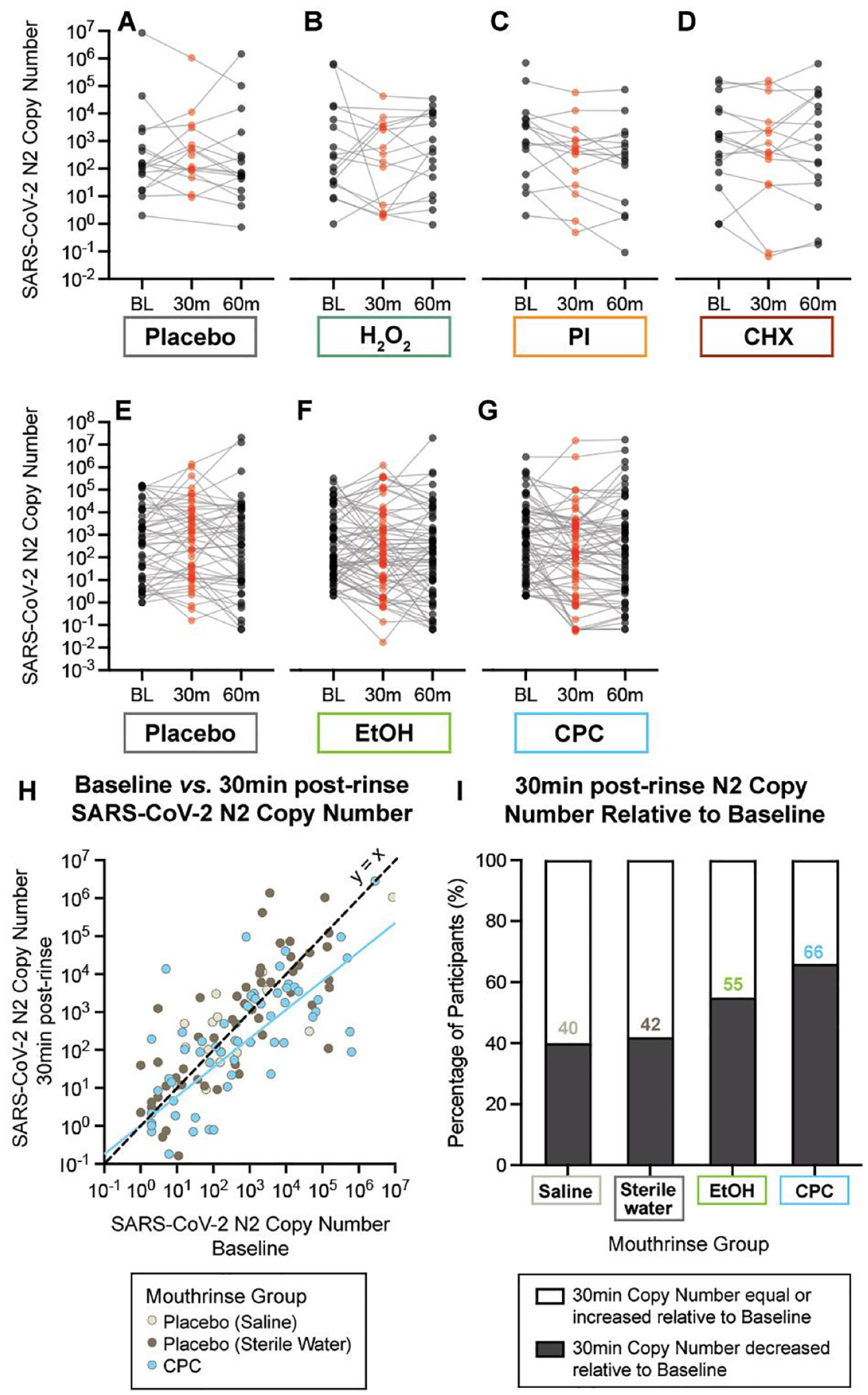
Salivary viral load at baseline (prerinse) and 30 and 60 min postrinse. (
Survey responses are reported using descriptive statistics in Appendix Tables 7 and 8. Ordered logistic regression (SAS version 9; SAS Institute) was used to model the response distribution for each ordered categorical outcome variable (e.g., rinsing experience and willingness values) as a function of group membership, and the resulting Wald statistic was used to test the null hypothesis of no mean differences among groups, overall and between pairs, with a significance threshold of P < 0.05.
Statistical analyses were conducted with R version 4.2.2 and SAS version 9 and analyzed on an intention-to-treat basis. P values are reported with false discovery rate adjustments. Graphs were generated using Prism version 9 (GraphPad Software Inc.), and figures were created with Adobe Suite (Adobe Inc.) and BioRender (BioRender Inc.).
Results
A comprehensive presentation of study participant characteristics is available in Table 1 and Appendix Tables 2 to 4. Most participants were outpatients who tested positive for SARS-CoV-2 at UNC and local clinics across several variant waves (Fig. 1). Five percent of participants were conscious, stable hospital inpatients. Most participants (97%) were symptomatic, with common complaints including cough (83%), congestion (78%), and fatigue (63%). A minority reported severe symptoms, such as difficulty breathing (28%), fever (22%), diarrhea (11%), and nausea (10%). Symptom distribution varied, with 55% of participants experiencing 4 to 7 symptoms, 32% reported 0 to 3 symptoms, and 13% experiencing >7 symptoms. Most (76%) were of European descent, with smaller representations of African (17%) and Asian (5%) descent, while 10% reported being of Hispanic ethnicity. Participants’ mean age was 36 y (median, 33; range, 18 to 65). Participants were seen on average 6 d after symptom onset and 4 d after a positive test result; 70% were vaccinated and 85% were unmedicated.
An Oral Rinse Containing CPC Significantly Reduces Salivary SARS-CoV-2 Viral Load
The oral rinse containing the active ingredient CPC was the only one that resulted in a significant reduction in SARS-CoV-2 viral load at 30 min postrinse relative to the prerinse baseline (P = 0.015) and to relative placebo (P = 0.017) (Fig. 3, Appendix Tables 9–11). No other rinse in either RCT significantly affected viral load at 30 min after rinsing. At 60 min postrinsing, no group had a significant reduction in SARS-CoV-2 copy number relative to baseline, indicating a rebound in salivary viral load over a 1-hour window.
Mouthrinse Acceptability and Willingness to Use
Participants were asked to assess their rinsing experience by rating sensation, appearance, taste, and odor. They were also asked to report experiences of burning, tingling, loss of taste, loss of sensation, dryness, and swelling following the mouthrinse. A positive score is indicative of a more favorable rinsing experience with the mouthrinse. Statistical analysis showed that overall, the taste, appearance, odor, and sensation of most mouthrinses were judged as acceptable, with average ratings in the fair-to-good range (0 or 1), except EtOH’s taste and sensation and PI’s appearance (Fig. 2 A, B; Appendix Table 8). EtOH was judged to cause oral burning or tingling by >90% of respondents, in addition to having the least favorable taste with a negative mean score. For CPC, more than half of respondents rated its taste (56%), odor (77%), and appearance (66%) as excellent or good. CPC’s odor was rated more positively than some of the mouthrinses, while CPC’s oral sensation was reported as significantly less favorable than CHX, with 35% of all respondents evaluating the sensation as poor or very poor.
The use of antiseptic rinses in social, clinical, and workplace situations depends on people’s willingness to rinse. Surveys revealed high willingness to use oral rinses before and during health care appointments, including the dentist (94% reported being very willing) and doctor (83% very willing), and before interacting with family at home (60.5% very willing) and coworkers in meetings (65% very willing), with no significant difference among rinses (Fig. 2C, Appendix Table 7). Respondents were less willing to use oral rinses before and during social gatherings at restaurants, bars, parties, or lunch with coworkers.
Discussion
Strategies to reduce salivary viral load can be effective community health tools considering the emerging SARS-CoV-2 variants and other membrane-bound viruses, such as influenza (Tegally et al. 2021; NCIRD 2023). Implementing preprocedural antiseptic mouthrinses is a promising strategy to reduce viral transmission in an antigen-agnostic manner, particularly in clinical settings. Our findings revealed a significant decrease in salivary SARS-CoV-2 viral load 30 min after rinsing with a CPC-containing product, across several variant waves, including Delta during the first trial and Omicron during the second. As antiseptic mouthrinses exert their antiviral properties through antigen-agnostic mechanisms (i.e., damage to the viral envelope), the specific variant is unlikely to affect the response to mouthrinse active ingredients, highlighting the broad clinical applicability of oral rinsing (Carrouel, Gonçalves, et al. 2021).
CPC is a quaternary ammonium compound and surfactant that is broadly effective against bacteria, fungi, and enveloped viruses and is widely used in commercially available antiseptic mouthwashes and sprays. Recent studies show that CPC can exert anti–SARS-CoV-2 effects at concentrations both high (250 μg/mL; Bañó-Polo et al. 2022; Takeda et al. 2022) and low (10 to 50 μg/mL, 0.001% to 0.005%) through lipid membrane disruption and viral particle inactivation, effectively reducing viral load and suppressing SARS-CoV-2 infectivity in vitro (Koch-Heier et al. 2021; Muñoz-Basagoiti et al. 2021; Anderson et al. 2022). Human trials evaluating CPC-based rinses identified a reduction in salivary SARS-CoV-2 infectivity and increased levels of nucleocapsid protein as a marker of viral particle disruption in patients postrinse (Alemany et al. 2022; Bonn et al. 2023; Tarragó-Gil et al. 2023). Although in vitro infectivity and nucleocapsid protein levels were not assessed in this study, which represents a limitation, these data are broadly in agreement with existing literature suggesting that CPC is an effective anti–SARS-CoV-2 strategy with relevance to dental and oral health care practices (Cieplik and Jakubovics 2022).
In addition to the general antiviral and specific anti–SARS-CoV-2 properties described here, other advantages exist. CPC is an odorless, mild-tasting, and neutral-colored product and is easy to formulate as compared with CHX, PI, and EtOH, with broad applicability. Furthermore, CPC is accepted for use in pediatrics, unlike EtOH, so it can be widely employed with pediatric and adult clinical populations (D’Amico et al. 2023). These advantages make CPC feasible in a variety of clinical settings, though other trials have indicated efficacy of oral rinses containing CHX, PI, EtOH, and H2O2 against SARS-CoV-2 (Ziaeefar et al. 2022; Farmaha et al. 2023). Variations across RCT results likely stem from differences in sample sizes, participants’ disease acuity, rinsing protocols, time points measured, and rinse formulations of active and inactive ingredients. As more trials are published, additional meta-analyses will be important in guiding best practice recommendations and protocols for oral rinsing.
In this study, no oral rinse was associated with a significant reduction in copy number at 60 min postrinse as compared with baseline, indicating a rebound in salivary viral load. This is not surprising as virus is shed from the oral epithelium and salivary glands produce 0.3 to 0.4 mL/min of saliva (Iorgulescu 2009; Huang et al. 2021). Altogether, our findings support the implementation of oral rinsing with the active ingredient CPC. Although some studies have found that CPC may be effective at reducing SARS-CoV-2 viral load for longer periods, our data indicate that CPC may be most effective within 30 min (Alemany et al. 2022; Bezinelli et al. 2023).
This study has several limitations. First, the ability for mouthrinse use to reduce salivary SARS-CoV-2 infectivity in vitro was not assessed. Other studies have evaluated infectivity by assessing reductions in plaque-forming units in viral culture or with a modified enzyme-linked immunosorbent assay measuring disintegration of the viral envelope (Alemany et al. 2022; Bonn et al. 2023). Although this does not detract from the validity of our findings with respect to changes in intraoral viral load postrinse, the assessment of RNA copy number is methodologically insufficient for assessing the efficacy of antiseptic rinses on the infective potential of enveloped viral particles, an issue that has been recently reviewed (Cieplik and Jakubovics 2022). Recent studies show, however, that other protective factors abundant in saliva, including lactoferrin, limit in vitro infectivity, thus complicating interpretation of data when saliva is used as the primary biofluid (Yazawa et al. 2023). Although neither viral load nor in vitro culture infectivity can predict infectiousness at an individual level, recent studies have shown that SARS-CoV-2 cycle threshold values, as a proxy for viral load, inversely correlate with transmission rates and patient infectiousness (Singanayagam et al. 2020; Jones et al. 2021; Platten et al. 2021; Savela et al. 2022; Puhach et al. 2023; Panico et al. 2024); this is in alignment with our recently reported findings that salivary SARS-CoV-2 N1/N2 cycle threshold values were negatively correlated with days postinfection (Graves et al. 2024). Taken together, our data suggest that some mouthrinses, including CPC, may reduce the infectious potential of saliva by limiting SARS-CoV-2 viral load; they also highlight the fact that further work is needed to establish robust saliva-specific protocols for the assessment of viral infectivity and to assess the link between reduced viral load and transmissibility. Second, the specific mechanisms mediating observed reductions in salivary viral load were not explored, and we are therefore unable to separate mechanical effects of rinsing from antiseptic action, an issue raised in similar trials (Cieplik and Jakubovics 2022; Alzahrani et al. 2023; Perussolo et al. 2023). Finally, the number of active ingredients evaluated here was limited to those that were commonly used by regional dental providers in clinical settings or mouthrinses that showed promising antiviral activity at the time of study launch (Bidra et al. 2020; Meister et al. 2020; O’Donnell et al. 2020). To overcome the sample size limitations faced by similar studies, several rinses containing promising ingredients with anti–SARS-CoV-2 potential were excluded, such as those containing β-cyclodextrins and Citrox (Carrouel, Gonçalves, et al. 2021; Carrouel, Valette, et al. 2021).
Survey data demonstrate a high level of willingness to rinse before and during doctor and dental visits, indicating potential for mouthwash adoption in health care settings to potentially reduce risk of spread. Our data are consistent with broad adoption of oral rinsing in dental offices seen following recommendations from the American Dental Association (Mohd-Said et al. 2021; Liu et al. 2022). Importantly, this study is one of the few investigations that evaluated participants’ willingness to rinse to prevent COVID-19 transmission, with a potential impact on other infectious diseases.
As expected, lower willingness for mouth washing was reported for social settings. In general, rinsing experiences were perceived positively for nearly all rinses, apart from PI’s appearance and EtOH’s taste and sensation. In summary, findings suggest that health care providers could incorporate mouth rinsing into their clinical workflow with wide patient acceptance.
Conclusions
A CPC-containing mouthrinse provided a significant reduction in SARS-CoV-2 viral load in human saliva 30 min postrinse. This study suggests that using a mouthrinse containing CPC could be an easily and rapidly implementable intervention that is low risk, inexpensive, and feasible in clinical settings, while being antigen agnostic as new variants arise. In addition to providing support for health care workers during unavoidable close contact, these potential benefits could extend to social and educational settings. Affordable and efficient strategies to reduce SARS-CoV-2 viral transmission in antigen-nonspecific manners, such as oral rinsing, meet an ongoing public health need as new variants emerge, with relevance for other membrane-bound respiratory viruses, such as influenza.
Author Contributions
C. Graves, L.A. Jacox, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; N. Ghaltakhchyan, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; T.Q. Ngo, E. Babikow, C. Bocklage, contributed to data acquisition, analysis, and interpretation, and critically revised the manuscript; C. Liu, A. Shoji, M. Freire, K. Divaris, D. Wu, contributed to data analysis and interpretation, critically revised the manuscript; and critically revised Y. Sang, contributed to data acquisition, and critically revised the manuscript; S.T. Phillips, contributed to data acquisition and analysis, and critically revised the manuscript; N. Bowman, S. Wallet, contributed to conception, design, and critically revised the manuscript; S. Frazier-Bowers, contributed to data interpretation, and critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jct-10.1177_23800844241296840 – Supplemental material for A Cetylpyridinium Chloride Oral Rinse Reduces Salivary Viral Load in Randomized Controlled Trials
Supplemental material, sj-docx-1-jct-10.1177_23800844241296840 for A Cetylpyridinium Chloride Oral Rinse Reduces Salivary Viral Load in Randomized Controlled Trials by C. Graves, N. Ghaltakhchyan, T.Q. Ngo, C. Liu, E. Babikow, A. Shoji, C. Bocklage, Y. Sang, S.T. Phillips, N. Bowman, S. Frazier-Bowers, M. Freire, S. Wallet, K. Divaris, D. Wu and L.A. Jacox in JDR Clinical & Translational Research
Footnotes
Acknowledgements
We thank the Go Health Clinic for its guidance and support hosting this study. Carol Culver, Wendy Lamm, and Tammy McGonagle have been instrumental in offering guidance, ordering supplies, setting up facilities, establishing calendars, and assisting with study planning and execution. We thank the UNC Respiratory Diagnostic Centers as well as Jamie Roberts at Duke University and Emily Olsson at NC TraCS. We also thank Will Lovell, Mandy Bush, and the previous and current staff of the UNC Respiratory TRACTS Core. We appreciate the quantitative guidance of Chris Wiesen at the UNC Odum Institute for Research and Ilan Moyer. Finally, we thank all members of the Jacox, Wallet, and Graves laboratories for thoughtful discussion.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Southern Association of Orthodontists Research Award (E.B.), the American Association of Orthodontists Foundation Resident Research Aid Awards (E.B.), and the Robert L. Boyd Biomedical Research Award (L.J. and S.F.-B.). The project was funded by the National Institute of Dental and Craniofacial Research, National Institutes of Health, through a R03 award (grant 1R03DE031301-01 to L.J. and C.G.) and a K08 award (grant 1K08DE030235-01A1 to L.J.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
A supplemental appendix to this article is available online.
Data Availability Statement
The data supporting the findings of this study are available within the article and its supplementary materials.
For the remaining list of references, see Appendix.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
