Abstract
Although established theoretical models suggest that socioeconomic inequalities in physical health are partially mediated or explained by exposures to environmental toxins, there is little empirical evidence to support these processes. Building on previous research, we analyze data from the National Health and Nutrition Examination Surveys (2007–2008) to formally test whether associations between socioeconomic status and self-rated physical health are mediated by direct biological assessments of environmental toxins. We find that the embodiment of environmental toxins is disproportionate in individuals of lower socioeconomic status. Although toxins in general and lead in particular are unrelated to self-rated health, socioeconomic differences in self-rated physical health are partially mediated by embodied cadmium. Our mediation analyses suggest that education and income may promote physical health by reducing exposures to cadmium. This study contributes to previous work by bridging the fields of social epidemiology and environmental inequality and by formally testing established theoretical models.
Introduction
Over the past four decades, volumes of studies have shown that people of higher socioeconomic status (SES) tend to exhibit better health and longer life expectancies than people of lower SES (Adler and Ostrove 1999; House 2002; Link and Phelan 1995; Marmot and Wilkinson 1999; Mirowsky and Ross 2003a, 2003b; Schnittker and McLeod 2005; Williams and Collins 1995). This general pattern has been documented across a range of health-related outcomes, including mental health, physical health, all-cause mortality, and cause-specific mortality. We have also seen replications across several dimensions of SES, including educational attainment, employment status, income, financial hardship, wealth, and subjective social status.
Although this empirical literature has made significant contributions to our understanding of the link between SES and health, we are still working to establish the precise underlying mechanisms. Previous studies suggest that people of higher SES are healthier because they tend to have greater access to health care, lower levels of stress, safer living conditions and communities, healthier lifestyles, and more psychosocial resources (Adler and Ostrove 1999; Cockerham 2005; Evans and Kantrowitz 2002; Evans and Kim 2010; House 2002; Lantz et al. 2001; Link and Phelan 1995; Marmot and Wilkinson 1999; McEwen and Gianaros 2010; Mirowsky and Ross 2003a, 2003b; Schnittker and McLeod 2005; Williams and Collins 1995). Although well-known theoretical models developed by eminent scholars like David Williams (Williams and Collins 1995), Nancy Adler (Adler and Ostrove 1999), and James House (House, Lantz, and Herd 2005) identify “environmental exposure,” “exposure to carcinogens and pathogens,” and “toxic chemical and physical conditions” as important pathways linking SES and health, direct empirical measurements and assessments of the embodiment of environmental risk are often overlooked (Evans and Kantrowitz 2002; Evans and Kim 2010). Evans and Kantrowitz (2002:325) explain that “Exposure to multiple, suboptimal environmental risk factors is one viable mechanism among several that could be a partial explanation for the gradient between SES and multiple health outcomes.”
In this study, we build on previous research by formally testing whether associations between SES and self-rated physical health are mediated by the embodiment of environmental toxins. In what follows, we discuss (1) socioeconomic variations in environmental risk and (2) the potential physical health consequences of embodied toxins. After describing our data, measures, and statistical procedures, we summarize the results of our analyses. We end with a discussion of our key findings and research limitations. We also describe several avenues for future research.
Background
SES and Environmental Risk
Low SES individuals, families, and communities are regularly confronted with the inequities and hardships of environmental injustice (Adams et al. 2006; Ard 2015; Berkman 2004; Brulle and Pellow 2006; Bullard 2000; Crowder and Downey 2010; Downey and Van Willigen 2005; Elliott and Frickel 2013; Evans and Kantrowitz 2002; Evans and Kim 2010; House and Williams 2003; Järup 2003; Krieger et al. 2005; Lynch et al. 2000; Mohai and Bryant 1992; Tyrrell et al. 2013). Occupational and financial constraints tend to limit opportunities for safety in nutrition, housing, employment, and transportation. For example, low-income housing is often characterized by dilapidation, which exposes residents to toxins, infections, and illness (Evans and Kantrowitz 2002; Evans and Kim 2010). As a result, poorer individuals and families tend to exhibit higher levels of lead poisoning (Berkman 2004; Krieger et al. 2005). Researchers attribute these patterns to lead-based paint and pipes in substandard housing. Low SES individuals and families are often trapped in these structural contexts of environmental inequality because they often lack the knowledge, financial assets, legal resources, and political power to block or evade the siting of pollution-producing facilities in their communities (Adler and Ostrove 1999; Bullard 2000; Evans and Kantrowitz 2002; Evans and Kim 2010; Grant et al. 2010; Mohai, Pellow, and Roberts 2009).
Environmental Risk and Physical Health
Toxins are pervasive in our environments and generally refer to a range of potential hazards like lead, arsenic, polychlorinated byphenyls, and other heavy metals. In this study, we focus on toxic heavy metals found throughout the built environment, including arsenic, barium, cadmium, cobalt, cesium, lead, molybdenum, thallium, and tungsten. Exposure to these toxins can occur through multiple pathways, including, for example, air pollution, water contamination, industrial fumes, and substandard housing.
The primary threats to humans from heavy metals are exposures to cadmium and lead (Järup 2003). These toxins are worthy of investigation due to their association with numerous adverse health outcomes (Agency for Toxic Substances and Disease Registry [ATSDR] 2005). Emissions of these metals occur through exposures to contaminated air, water, soil, and food. Cadmium is consistently associated with higher rates of inflammation, diabetes, liver disease, renal failure, skeletal damage, cardiovascular disease, obstructive lung disease, breast cancer, stroke, early onset osteoporosis, developmental disabilities, all-cause mortality, and mortality from specific causes (Ciesielski et al. 2012; Gallagher, Kovach, and Meliker 2008; Hecht et al. 2013; Järup 2003; Larsson, Orsini, and Wolk 2015; Lin et al. 2009; Mendy, Gasana, and Vieira 2012; Tellez-Plaza et al. 2013; Wallia et al. 2014). Lead is also harmful to human health. Lead poisoning has been linked with higher rates of headaches, irritability, abdominal pain, nerve damage, kidney ailments, ventricular hypertrophy, hypertension, and all-cause mortality (ATSDR 2007; Järup 2003; Navas-Acien et al. 2005; Schober et al. 2006; Schwartz 1991).
While cadmium and lead are among the most dangerous and pervasive environmental toxins, research has identified other harmful heavy metals. For example, arsenic is a heavy metal found in groundwater intended for human consumption. About 90 percent of all arsenic is used in preserving wood for fences, decks, and picnic tables. Arsenic is also found in pesticides and lead-acid batteries (ATSDR 2007). Arsenic can induce vomiting, diarrhea, and fatigue in the short term. Long-term effects include skin changes, circulatory and peripheral nervous disorders, abnormal fetal development, and lung cancer (ATSDR 2007).
The embodiment of toxins like antimony, cobalt, molybdenum, tungsten, and uranium can also occur through occupational hazards (e.g., heavy industrial work). These toxins are associated with a range of adverse health consequences, including higher rates of asthma, fibrosis, cardiovascular disease, cerebrovascular disease, liver damage, thyroid disorders, and vision impairments (Agarwal et al. 2011; Christensen 2013; De Palma et al. 2010; Mendy et al. 2012; Navas-Acien et al. 2005; Shiue 2013). There is also some evidence linking related exposures to elevations in mortality risk (Lasfargues et al. 1994; Moulin et al. 1998).
To our knowledge, only one study has tested the connection between heavy metal toxins and self-rated health, which is the primary outcome of this study. Shiue (2015) found that people who exhibited higher levels of urinary arsenic and heavy metals, including cadmium, cobalt, manganese, molybdenum, lead, antimony, strontium, tungsten, and uranium, were more likely to describe their general health status as poor, fair, or good than very good or excellent.
Hypotheses
In accordance with previous research, we developed three hypotheses to guide subsequent analyses:
Hypothesis 1: People of lower SES will tend to embody higher levels of environmental toxins.
Hypothesis 2: People who embody higher levels of environmental toxins will tend to exhibit poorer overall self-rated physical health.
Hypothesis 3: People with lower levels of SES will tend to exhibit poorer overall self-rated physical health, and this association will be at least partially mediated or explained by the embodiment of environmental toxins.
Data
The data for this study come from the 2007–2008 cross-sectional National Health and Nutrition Examination Surveys (NHANES). The NHANES employs a complex, multistage, probability sample of the noninstitutionalized civilian U.S. population (Curtin et al. 2013). Starting in 2007, a new sampling methodology was implemented. Several groups were oversampled, including adults at or below 130 percent of federal poverty level and adults aged 80 and over. The survey consists of questionnaires administered in the home, followed by a standardized health examination in specially equipped mobile examination centers. Blood and urine samples were taken from random subgroups (about one-third) of the full sample. Our final analytic sample included 1,792 adults aged 18 to 80 who were tested for toxins in their urine. Table 1 provides descriptive statistics for all variables included in the analyses.
Descriptive Statistics (n = 1,792).

Source: National Health and Nutrition Examination Surveys (2007–2008).
Measures
Environmental Toxins
The NHANES 2007–2008 included urine samples for arsenic, barium, cadmium, cobalt, cesium, molybdenum, lead, thallium, and tungsten. Urinary tests of heavy metals have been shown to accurately reflect the amount of heavy metals in the body (ATSDR 2012). The amount of heavy metals present in urine shows both recent and past exposure (ATSDR 2012). The urine collection procedure consisted of urine specimen collection and processing. Urine specimens were processed, stored, and shipped to the Division of Environmental Health Laboratory Sciences, National Center for Environmental Health, and Centers for Disease Control and Prevention for analysis.
Inductively coupled plasma-mass spectrometry (ICP-MS) was employed to quantify heavy-metal concentrations in urine (Date and Gray 1989). This multi-element analytical technique measures the following 12 elements in urine: beryllium (Be), cobalt (Co), molybdenum (Mo), cadmium (Cd), antimony (Sb), cesium (Cs), barium (Ba), tungsten (W), platinum (Pt), thallium (TI), lead (Pb), and uranium (U) (Mulligan, Davidson, and Caruso 1990). Most elements were measured in micrograms per liter (ug/L) (Centers for Disease Control and Prevention 2008).
In this study, toxins are measured in two ways. We first examine a mean index of the nine toxins (α = .804). To account for variations in response metrics, all toxin measures were standardized before indexing. We also examine exposures to lead and cadmium separately. Lead and cadmium are studied independently because, as noted in the previous discussion, they are often found to be the most pervasive and dangerous of the metal toxins (ATSDR 2005; Krueger and Wade 2016; Weidemann et al. 2015).
Self-rated Physical Health
Following recent work in environmental inequality (Ard et al. 2016), we measure physical health status with a standard overall self-rated health item. Self-rated health is a common subjective indicator of general health status that is consistently associated with more objective health measures, including physician diagnoses and mortality risk (Burström and Fredlund 2001; Frankenberg and Jones 2004; Idler and Benyamini 1997; Kennedy et al. 1998). According to Idler and Benyamini (1997), self-rated health is a robust predictor of mortality risk over and above numerous specific medical, behavioral, and psychosocial risk factors. In this study, we focus on self-rated physical health precisely because it is a subjective indicator of general health status. As a global subjective indicator, self-rated health captures symptoms and conditions that are experienced, not simply diagnosed or undiagnosed. This is important because our toxin measures may indicate embodiment in the distant past or more recently. While recent toxin exposures are unlikely to generate chronic conditions like liver disease, measures of self-rated health may capture the subjective experience of subclinical symptoms (e.g., short-term pain and discomfort). Thus, overall self-rated physical health may be especially sensitive to the potential mediating influence of toxin exposures. Respondents were asked to rate their physical health on a scale ranging from poor (1) to excellent (5).
Socioeconomic Status
We employ three common indicators of SES, including educational attainment, employment status, and personal income. Education is an ordinal measure of educational attainment. Response categories include (1) less than 9th grade, (2) Grades 9–11 (including Grade 12 without completion), (3) high school grad or GED equivalent, (4) some college or AA degree, and (5) college graduate or higher. Employed is a dichotomous variable that asks if a respondent is (1) currently employed or (0) not. Income is measured in US dollars earned in the previous calendar year. The index is split into increments of $5,000, with $0 to $4,999 being the lowest increment and $100,000 or more being the largest increment. Income is logged to normalize its positively skewed distribution. All missing values on income were replaced via multiple imputation. The multiple imputation model employed all independent variables from the full model to predict income. Our final income measure reflects average estimates from 10 iterations.
SES measures were not indexed for two primary reasons. First, it is customary to examine indicators of SES separately because they are causally related to each other. According to standard life course models, these factors build on each other (Mirowsky and Ross 2003a). Second, different indicators of SES tend to have different theoretical paths to toxin exposure. For example, education is often tied to occupational exposures because credentials shape employment opportunities, whereas income may be more relevant for residential exposures because financial resources are necessary for housing.
Background Variables
Subsequent multivariate analyses adjust for three background variables that are known correlates of toxin exposure and self-rated health. These variables include age, gender, and race/ethnicity. Age is measured in continuous years (18–80). Gender is a dichotomous variable (1 = female; 0 = male). Race is assessed using a categorical measure of racial identification, converted into a series of dummy variables. The sample includes (1) whites, (2) blacks, (3) Hispanics, and (4) other races and ethnicities. In multivariate analyses, whites serve as the common reference category.
Statistical Procedures
Our primary statistical analyses proceed in two steps. In the first step, we employ ordinary least squares (OLS) regression to model the embodiment of environmental toxins (Table 2) and self-rated physical health (Table 3) by SES and background variables. We acknowledge that toxin exposure is continuous and self-rated physical health is ordinal. DeMaris (2002:259) notes that in large samples, ordinal variables “with at least five levels” can be treated as “approximately interval” in OLS regression. In supplemental analyses (not shown), we replicated our regressions of self-rated physical health using ordered logistic regression. These results were substantively identical to our reported OLS models.
Ordinary Least Squares Regression of Toxin Exposure (n = 1,792).
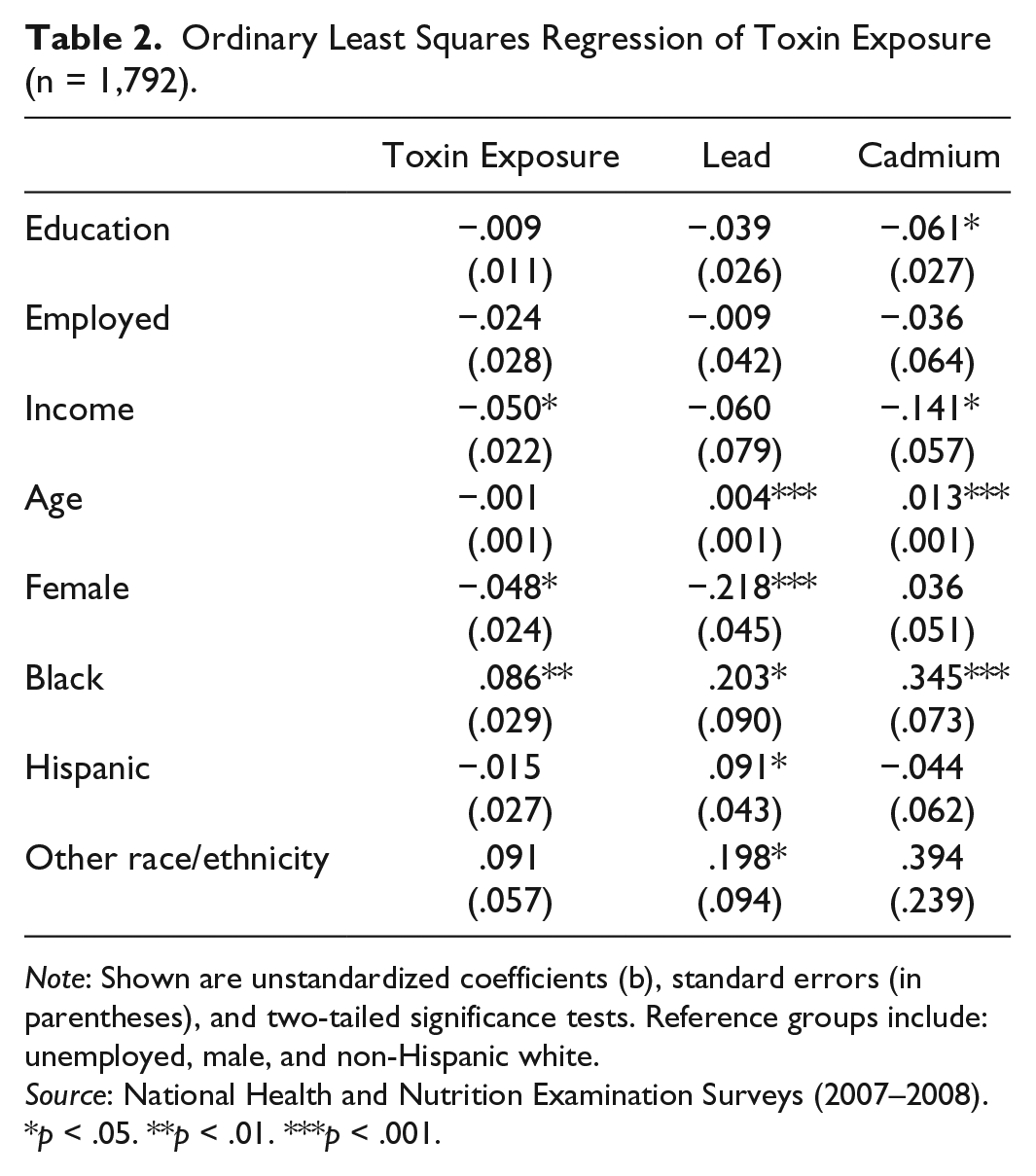
Note: Shown are unstandardized coefficients (b), standard errors (in parentheses), and two-tailed significance tests. Reference groups include: unemployed, male, and non-Hispanic white.
Source: National Health and Nutrition Examination Surveys (2007–2008).
p < .05. **p < .01. ***p < .001.
Ordinary Least Squares Regression of Self-Rated Physical Health (n = 1,792).

Note: Shown are unstandardized coefficients (b), standard errors (in parentheses), and two-tailed significance tests. Reference groups include: unemployed, male, and non-Hispanic white.
Source: National Health and Nutrition Examination Surveys (2007–2008).
p < .05. **p < .01. ***p < .001.
In the second step, we test whether the association between SES and health is mediated by embodied environmental toxins. There are two general approaches to testing mediation models. The first is the “coefficient change” approach, which assesses the change in the coefficient for the focal predictor across nested models (i.e., before and after the mediator variable is added to the regression equation). The second is the “indirect effect” approach, which assesses the statistical significance of an indirect effect (i.e., the product of two coefficients, the coefficient for the effect of the focal predictor on the mediator and the coefficient for the effect of the mediator on the outcome). In this study, we employ the indirect effect approach and the Sobel test (Sobel 1982) of statistical significance because this approach is more commonly used and requires information for each link in the proposed mediation process (X → M and M → Y). In supplemental analyses (not shown), we replicated our mediation analyses using the coefficient change approach and the statistical test developed by Clogg, Petkova, and Haritou (1995). These results were substantively identical to our reported indirect effect tests.
Results
Toxin Exposures
According to the first column of Table 2, the embodiment of toxins overall is unrelated to educational attainment and employment status. However, there is some evidence to suggest an inverse association with income. In other words, respondents who report higher incomes also tend to exhibit lower levels of heavy metals in their urine specimens. In the second column of Table 2, we see that the embodiment of lead is unrelated to educational attainment, employment status, and income. The last column shows that education and income are inversely associated with embodied cadmium. These results suggest that respondents who report higher levels of education and income also tend to exhibit lower levels of cadmium in their urine. The embodiment of cadmium did not vary according to employment status.
Self-rated Physical Health
Table 3 reveals predictable associations between SES and overall self-rated physical health. Education and income are positively associated with self-rated physical health. Employed respondents also tend to report better self-rated physical health than their unemployed counterparts. These patterns are consistent across models with adjustments for environmental toxins. The embodiment of toxins overall and lead in particular are both unrelated to self-rated health. However, cadmium is inversely associated with self-rated health. This suggests that respondents who exhibit higher levels of cadmium in their urine also tend to report poorer overall self-rated physical health.
Mediation Analyses
Given that the Sobel test of indirect effects requires that each link in the proposed process be statistically significant, we can rule out toxins overall and lead as potential mediators of the association between socioeconomic status and self-rated physical health. Income is predictably associated with the embodiment of toxins overall, which is unrelated to self-rated health. Lead is unrelated to both socioeconomic status and self-rated health. This leaves us with cadmium. Figure 1 presents our mediation analyses, including unstandardized path coefficients, standard errors, and formal Sobel tests (z). We observe statistically significant indirect effects for education (z = 2.043, p < .05) and income (z = 2.197, p < .05) through the embodiment of cadmium. Taken together, our mediation analyses suggest that education and income may promote physical health by reducing exposures to cadmium. There is no evidence to support toxins overall or lead as mechanisms of socioeconomic status.

Unstandardized coefficients and Sobel tests (z) for statistically significant indirect effects.
Supplemental Analyses
In supplemental analyses (not shown), we replicated our mediation analyses by replacing overall self-rated physical health with several diagnosed chronic disease conditions, including liver disease, heart disease, heart attack, congestive heart failure, stroke, cancer, and arthritis. There was no evidence of mediation for any measure of socioeconomic status through any of our three toxin measures. These supplemental analyses confirm that overall self-rated physical health may be especially sensitive to the potential mediating influence of environmental toxins.
Discussion
Although established theoretical models suggest that socioeconomic inequalities in physical health are at least partially explained by exposures to environmental toxins, there is little empirical evidence to support these processes. We aimed to extend previous work by formally testing whether the association between socioeconomic status and self-rated physical health is at least partially mediated by the embodiment of environmental toxins.
Our first hypothesis stated that people of lower SES would tend to embody higher levels of environmental toxins. This hypothesis received mixed support. Although educational attainment and employment status were unrelated to toxins overall, income was inversely associated with toxin exposure. SES was unrelated to lead exposure. Education and income were inversely associated with the embodiment of cadmium, but employment status was not. Our observed associations tend to support previous research (Adams et al. 2006; Berkman 2004; Bullard 2000; Evans and Kantrowitz 2002; Evans and Kim 2010; House and Williams 2003; Järup 2003; Krieger et al. 2005; Lynch et al. 2000; Mohai and Bryant 1992).
Our second hypothesis stated that people who embody higher levels of environmental toxins would tend to exhibit poorer overall self-rated physical health. Evidence for this hypothesis was inconsistent. Overall toxin exposure and lead exposure were unrelated to self-rated physical health. However, cadmium was inversely associated with self-rated health. Our results for cadmium are generally consistent with previous work (Ciesielski et al. 2012; Gallagher et al. 2008; Hecht et al. 2013; Järup 2003; Larsson et al. 2015; Lin et al. 2009; Mendy et al. 2012; Rokadia and Agarwal 2013; Tellez-Plaza et al. 2013; Wallia et al. 2014). However, our results for lead exposure fail to support previous research (ATSDR 2007; Järup 2003; Navas-Acien et al. 2005; Schober et al. 2006; Schwartz 1991).
Our third hypothesis stated that people with lower levels of SES would tend to exhibit poorer overall self-rated physical health and this association would be at least partially mediated or explained by the embodiment of environmental toxins. This hypothesis received moderate support. Education, employment, and income were associated with better self-rated physical health (Hill and Needham 2006; Mirowsky and Ross 2003a). Although we were able to rule out toxins overall and lead as potential mediators of the association between SES and self-rated physical health, we observed statistically significant indirect effects for education and income through the embodiment of cadmium. This evidence suggests that education and income may promote physical health by reducing exposures to cadmium. To the best of our knowledge, this is the first study to directly test the mediating influence of embodied environment toxins.
Our analyses are limited in several respects. First, the cross-sectional nature of the data does not allow us to assess long-term changes in toxin exposure and health. Longitudinal data are required to make stronger causal inferences. Second, our measurement of SES omits important measures like wealth and alternative indicators of income like the income-to-poverty ratio. It would be important for future research to replicate our results across additional indicators of financial status. Third, our measurement of employment status is relatively crude. More detailed assessments of occupational sectors and work-related environments are clearly required. For example, the Agency for Toxic Substances and Disease Registry (2012) notes that cadmium is often found in the manufacturing of pesticides, batteries, glass, and PVC.
Conclusion
Despite these noted limitations, our results suggest that the embodiment of cadmium may help explain broader socioeconomic inequalities in physical health, especially those according to education and income. Although this study contributes to previous work by bridging the fields of social epidemiology and environmental inequality and by formally testing established theoretical models, the veracity of our results is contingent on replication with longitudinal data and more comprehensive assessments of socioeconomic status. Additional research is also needed to explore socioeconomic variations in toxin exposure and physical health in different national contexts and at different levels of analysis (Ard 2015, 2016; Ard et al. 2016). Research along these lines would advance our collective understanding of the environmental processes linking socioeconomic status and health. Even broader extensions of our work could assess the role of environmental inequality in other established health disparities, including those tied to race/ethnicity or area income and wealth inequality (Ard et al. 2016; Hill and Jorgenson 2018).
