Abstract
Introduction
Blood pressure control is critical in acute ischemic stroke patients undergoing thrombectomy. Fluctuations in blood pressure can compromise cerebral reperfusion and influence 90-day functional prognosis. However, the relationship between blood pressure and prognosis remains unclear.
Objective
To examine the impact of blood pressure control strategies on 90-day functional prognosis, intracranial hemorrhage, mortality, and neurological deterioration in patients with acute ischemic stroke following thrombectomy.
Method
We systematically searched major Chinese/English databases up to April 2025 for interventional studies on post-thrombectomy blood pressure management in acute ischemic stroke. Dual-reviewer screening with predefined criteria identified eligible studies. Cochrane Risk of Bias Tool assessed methodological quality. Data extraction and random-effects meta-analysis (RevMan 5.1) evaluated pooled risk ratios (95% confidence intervals), with heterogeneity quantified via I2 statistics.
Results
This meta-analysis of 12 studies (n = 4,794) found no significant benefit of intensive over standard blood pressure lowering in functional recovery, hemorrhagic risk, mortality, or early neurological outcomes post-thrombectomy. However, subgroup analyses revealed critical associations: lower systolic blood pressure correlated with functional independence (−4.46 mmHg), while elevated levels were linked to intracranial hemorrhage (+6.55 mmHg) and mortality (+3.12 mmHg).
Conclusion
The meta-analysis findings suggest that individualized blood pressure thresholds, rather than uniform intensive control, may optimize postintervention outcomes, warranting further prospective validation.
Keywords
Introduction
Acute ischemic stroke, characterized by cerebral artery occlusion-induced tissue infarction, remains a leading cause of global disability and mortality (Team, 2023). While endovascular thrombectomy (EVT) achieves vascular recanalization in 40%–50% of eligible patients (Zhou, 2022), nearly half of these survivors experience suboptimal functional recovery (Goyal et al., 2016).
Functional independence, defined using a modified Rankin scale (mRS) score of 0–2, is a key goal in stroke recovery. While EVT successfully recanalizes 40%–50% of eligible patients (Zhou, 2022), nearly half still experience poor functional outcomes (Goyal et al., 2016). A major safety concern is symptomatic intracranial hemorrhage (sICH), occurring in 4%–7% of cases. sICH frequently causes early neurological deterioration, typically defined as a ≥4-point increase on the National Institutes of Health Stroke Scale (NIHSS), and is strongly linked to increased mortality (Nogueira et al., 2009). Neurofunctional deterioration affects 10%–15% of patients, serving as both a marker of acute brain injury progression and a predictor of long-term disability and death. Mortality rates remain high at 15%–30% within 90 days after EVT (Nogueira et al., 2009). Evidence suggests perioperative blood pressure (BP) variability is a modifiable outcome predictor after EVT (Nogueira et al., 2009). Balancing cerebral perfusion and bleeding risk through BP management is therefore a central clinical challenge, though optimal thresholds remain unclear.
Current guidelines provide conflicting recommendations: The 2019 American Heart Association and American Stroke Association (AHA/ASA) guidelines permit systolic blood pressure (SBP) <220 mmHg post-EVT (Powers et al., 2019), whereas neurointerventional consensus advocates stricter control (<140 mmHg; Mistry et al., 2018). This discrepancy is compounded by recent randomized trials showing intensive BP lowering failed to improve outcomes (Yang et al., 2022), contrasting with Zhou's findings of reduced 90-day disability with aggressive control (Zhou, 2022). Such clinical equipoise underscores the urgent need for evidence synthesis to inform practice.
As frontline stewards of hemodynamic monitoring, nurses play a pivotal role in detecting critical BP fluctuations that may herald hemorrhagic transformation or cerebral hypoperfusion post-thrombectomy (Maïer, Gory, Chabanne, et al., 2022; Maïer, Gory, Lapergue, et al., 2022). However, the absence of consensus on optimal BP thresholds creates ambiguity in nursing protocols for escalation alerts and antihypertensive administration (Zhang et al., 2024). This uncertainty delays time-sensitive interventions, potentially exacerbating secondary neuronal injury (Hawkes et al., 2022).
Objectives
This study intends to use meta-analysis to integrate data and explore the relationship between BP and functional recovery, sICH, mortality, and neurological deterioration in patients after EVT.
Methods
This meta-analysis adheres to PRISMA guidelines (Page et al., 2021) and includes multinational clinical trials conducted across tertiary medical centers. The study period spans from the initial literature search in February 2024 through the final database update in April 2025, comprising three distinct phases: (1) primary systematic search (February–March 2024), (2) protocol-driven verification (April 2024), and (3) supplementary evidence update (April 2025). As a secondary research synthesis, we incorporated multicenter randomized controlled trials (RCTs) and prospective cohort studies investigating BP management in post-thrombectomy stroke care, excluding case reports and non-peer-reviewed preprints. The PROSPERO registration number is 1248443.
Eligibility Criteria
Inclusion/exclusion criteria were developed per Cochrane Handbook for Systematic Reviews of Interventions 6.5 (2024; Higgins et al., 2024). Inclusion criteria: (1) Population: adults (≥18 years) with acute ischemic stroke undergoing EVT; (2) Intervention: protocolized postoperative BP management (explicit target ranges/duration); (3) Comparator: usual care without standardized BP control; (4) Outcomes: functional independence (mRS ≤ 2) at 90 days, intracranial hemorrhage (ICH), mortality, and neurological deterioration; (5) Design: prospective RCTs published in peer-reviewed journals. Exclusion criteria: (1) insufficient data for meta-analysis (e.g., missing standard deviations, unreported outcome measures); (2) duplicate publications (identical data across multiple journals) or overlapping cohorts; and (3) nonhuman studies, case reports, or observational designs.
Information Sources
We conducted comprehensive searches across major Chinese databases (China National Knowledge Infrastructure [CNKI], Wanfang Data) and international platforms (PubMed, Embase, Web of Sciences) from database inception through April 2024. To ensure data completeness, citation tracking of seminal references was performed through Web of Science's “Cited Reference Search” module. For studies with incomplete datasets, corresponding authors were contacted via email and ResearchGate with structured data-sharing requests, allowing a 14-day response window before final exclusion.
Search Strategy
Developed under the PICOS framework and Cochrane Handbook guidelines (Higgins et al., 2024), our search strategy employed the following Boolean syntax across databases: (“endovascular therapy” OR embolectomy OR thrombectomy) AND (“acute ischemic stroke” OR “vascular occlusion” OR stroke) AND (“modified rankin scale” OR mRS) AND “intracranial hemorrhage” AND (“mortality” OR “death”) AND “neurological deterioration” AND “blood pressure.” This protocol was implemented in English databases, and Chinese version adapted for Chinese platforms. All searches were restricted to publications prior to April 2025.
Selection Process
A dual-independent screening protocol was implemented across four sequential phases. Initially, two reviewers concurrently assessed titles/abstracts against PICOS criteria using standardized screening forms (Appendix) to exclude manifestly ineligible studies. Following deduplication through automated tools complemented by manual cross-verification, full-text retrieval was pursued via institutional access and direct author correspondence, mandating a minimum of three contact attempts. Full-text eligibility discrepancies were resolved through consensus meetings, with any remaining conflicts arbitrated by a cerebrovascular specialist.
Data Collection Process
Following screening, paired reviewers independently extracted data with prepiloted forms. Discrepancies triggered a verification process involving source re-examination, material cross-checking, and expert adjudication, ensuring fidelity to study intent and minimizing errors.
Data Items
Standardized extraction fields encompassed bibliographic identifiers (first author, publication year), sample demographics (intervention/control group sizes), intervention specifics (target BP ranges, monitoring frequency), control group management strategies, 90-day mRS outcomes (functional independence: 0–2 vs. poor prognosis: ≥3), ICH, mortality, and 24 hr neurological deterioration.
Study Risk of Bias Assessment
The Cochrane Handbook (Higgins et al., 2024) evaluated seven methodological domains: random sequence generation, allocation concealment, participant/personnel blinding, outcome assessment blinding, incomplete outcome data, selective reporting, and other biases. Dual independent reviewers assigned low/high/unclear risk judgments, with interrater disagreements resolved through discussion.
Effect Measures
Functional independence, defined as mRS 0–2 at 90 days post-thrombectomy, served as the primary endpoint. Secondary endpoint includes sICH, mortality, and 24 hr neurological deterioration. sICH refers to a disease caused by the rupture of blood vessels in the brain, resulting in significant neurological deficits such as headaches, consciousness disorders, limb weakness, and so on. Mortality rate statistics show the number of all-cause deaths within 90 days. The 24 hr neurological deterioration declines NIHSS ≥4 points within 24 hr. Risk ratios (RR) with 95% confidence intervals (CIs) quantified intervention efficacy for this dichotomized outcome.
Synthesis Methods
Analyses were conducted in RevMan 5.3, employing chi-squared tests (α = .10) and I2 statistics to quantify heterogeneity. Fixed-effects models were prioritized when I2 ≤ 25%, transitioning to random-effects models for substantial heterogeneity (I2 > 25%). Pooled estimates were visualized through forest plots, supplemented by sensitivity analyses excluding high-risk-of-bias studies. All basic research information and outcome data will be presented in tables.
Result
Literature Screening Results
The initial search identified 767 relevant articles, comprising 118 from Chinese databases (CNKI: n = 67; Wanfang data: n = 51) and 649 from English databases (PubMed: n = 258; Embase: n = 35; Web of Science: n = 356).
After screening titles and abstracts against the predefined inclusion/exclusion criteria, 21 articles were retained. Citation tracking of these 21 articles yielded 9 additional eligible studies, resulting in a final selection of 30 articles (Chinese-language: n = 7; English-language: n = 23) during the initial screening phase. Two researchers read the full text of the included literature respectively and finally included seven articles for analysis (Deng et al., 2020; Goyal et al., 2017; Minhas et al., 2021; Mistry et al., 2023; Nam et al., 2023; Yang et al., 2022; Zhou, 2022), including one Chinese article and six English articles.
A third-round database update in April 2025 identified one additional eligible RCT (Anadani et al., 2022). To enhance the precision of BP threshold analysis, four supplementary studies (Anadani et al., 2019; Lu, 2022; Maier et al., 2018; Xu, 2020) with high-resolution hemodynamic monitoring records were incorporated during the final search iteration. These studies provided granular temporal data on systolic/diastolic fluctuations, enabling correlative analyses between BP trajectories and three clinical endpoints: functional recovery, hemorrhagic transformation, and mortality. Finally, a total of 12 studies (Anadani et al., 2019, 2022; Deng et al., 2020; Goyal et al., 2017; Lu, 2022; Maier et al., 2018; Minhas et al., 2021; Mistry et al., 2023; Nam et al., 2023; Xu, 2020; Yang et al., 2022; Zhou, 2022) were included in this analysis.
Results of Literature Quality and Bias Risk Assessment
The methodological quality and bias risk of the 12 included studies’ evaluated results are summarized in Figure 1.

The result of literature quality and bias risk assessment.
Data and Extraction Results
The analysis included 4,794 patients with acute ischemic stroke undergoing thrombectomy across all 12 studies. Primary outcomes included 90-day functional recovery assessed using the mRS, ICH incidence, 90-day mortality, and 24 hr neurological deterioration. Baseline characteristics and outcome data are presented in Table 1.
Baseline characteristics and outcome data.

Note. mRS = modified Rankin Scale, SBP = systolic blood pressure, ICH = intracranial hemorrhage.
mRS score ≤2.
24 hr neurological deterioration: ≥4 points decline in National Institutes of Health Stroke Scale (NIHSS) within 24 hr.
Meta-Analysis Results
The Main Result of Meta-Analysis
Eight studies (Anadani et al., 2022; Deng et al., 2020; Goyal et al., 2017; Minhas et al., 2021; Mistry et al., 2023; Nam et al., 2023; Yang et al., 2022; Zhou, 2022) assessed intensive versus standard postoperative BP management in thrombectomy-treated acute ischemic stroke patients. No statistically significant difference was found for 90-day functional independence (mRS ≤ 2: RR = 0.95, 95% CI [0.83, 1.10]; P = 0.53; I2 = 66%), ICH risk (RR = 0.93, 95% CI [0.80, 1.08]; P = .33; I2 = 0%), or 90-day mortality (RR = 1.07, 95% CI [0.83, 1.38]; P = .62; I2 = 26%). Point estimates indicated nonsignificant relative risk reductions of 5% for functional independence failure and 7% for hemorrhage, and a 7% relative increase in mortality. Only two studies (Anadani et al., 2022; Minhas et al., 2021) evaluated 24-hr neurological deterioration, showing no significant difference (RR = 0.80, 95% CI [0.34, 1.86]; P = .60; I2 = 78%). CIs crossed unity for all outcomes, indicating inconclusive evidence regarding benefit or harm. The forest plot is shown in Figure 2.
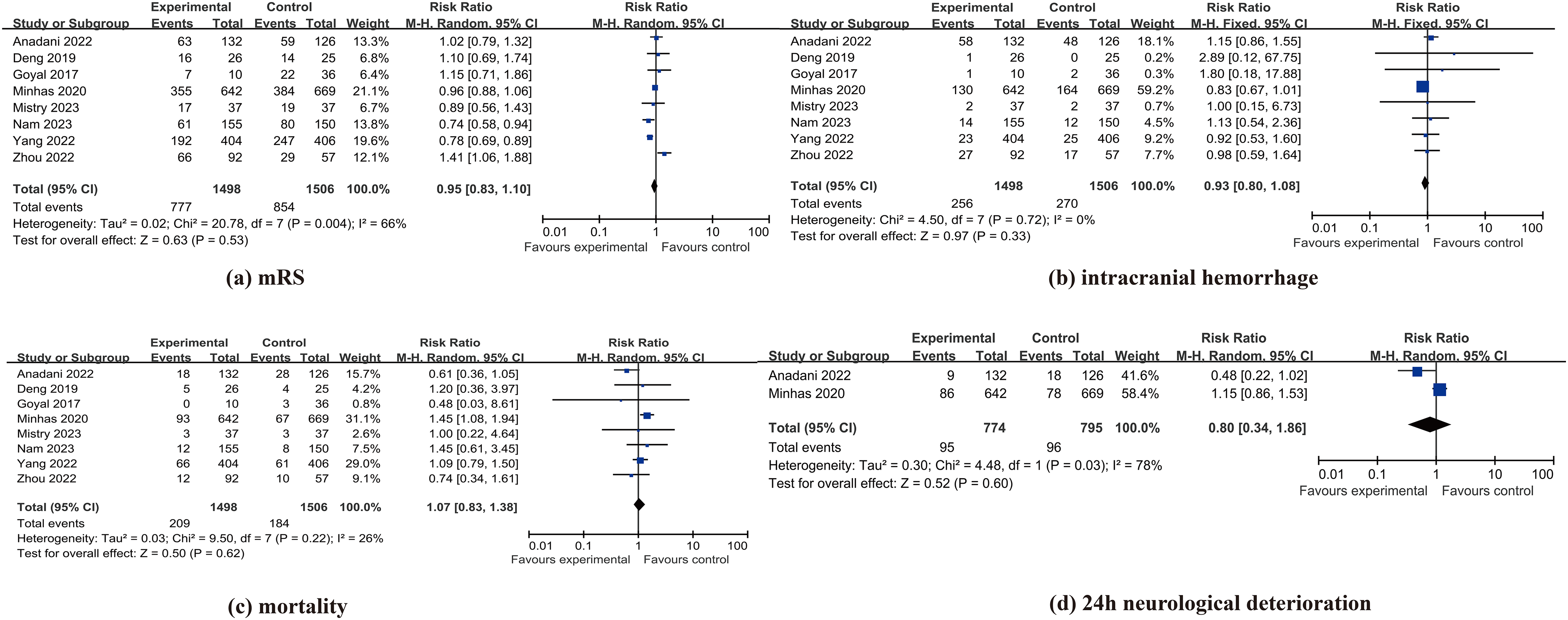
Forest plot.
Effects of BP Control on Clinical Outcomes
Another meta-analysis demonstrated significant associations between post-thrombectomy SBP and clinical outcomes. Five studies (Anadani et al., 2019; Goyal et al., 2017; Lu, 2022; Maier et al., 2018; Xu, 2020) showed patients achieving functional independence (mRS ≤ 2) had significantly lower SBP than those with poorer outcomes (mean difference [MD] = −4.46 mmHg, 95% CI [−5.74, −3.18]; P < .00001; I2 = 4%). Three studies (Anadani et al., 2019; Goyal et al., 2017; Maier et al., 2018) found patients with ICH had significantly higher SBP than those without ICH (MD = +6.55 mmHg, 95% CI [3.22, 9.89]; P = .0001; I2 = 0%). Two studies (Anadani et al., 2019; Goyal et al., 2017) indicated deceased patients had significantly higher SBP than survivors within 90 days (MD = +3.12 mmHg, 95% CI [0.82, 5.42]; P = .008; I2 = 24%). Fixed-effects models were used due to low heterogeneity. The forest plot is shown in Figure 3.

Forest plot for blood pressure.
Heterogeneity Analysis and Sensitivity Analysis
Given the substantial heterogeneity observed in certain outcome measures, we performed stratified subgroup analyses and conducted focused re-evaluations of studies that implemented SBP maintenance protocols targeting thresholds below 140 mmHg.
A subgroup analysis of studies targeting intensive BP control ≤140 mmHg (Goyal et al., 2017; Minhas et al., 2021; Mistry et al., 2023; Nam et al., 2023; Zhou, 2022) revealed no significant difference in functional recovery (mRS ≤ 2) between intensive and standard management (RR = 0.99, 95% CI [0.81, 1.22]; P = .94; I2 = 67%), no significant reduction in ICH risk (RR = 0.87, 95% CI [0.72, 1.04]; P = .13; I2 = 0%), and significantly increased 90-day mortality risk with intensive therapy (RR = 1.32, 95% CI [1.02, 1.70]; P = .03; I2 = 0%). The mortality finding indicates a 32% relative risk increase, with CI excluding the null value (RR = 1). Heterogeneity was substantial only for functional outcomes. The forest plot is shown in Figure 4.

Forest plot for subgroup analysis.
Discussion
Summary of Evidence
This meta-analysis included 12 studies (4,794 patients) to explore the impact of intensified hypotension (vs. standard treatment) on prognosis after thrombectomy for acute ischemic stroke. Random-effects model analysis showed that intensified BP lowering did not significantly improve 90-day functional independence (mRS ≤ 2; RR = 0.95, 95% CI [0.83, 1.10]; P = .53), and did not reduce postoperative sICH (RR = 0.93, P = .33), 90-day mortality (RR = 1.07, P = .62), or 24-hr risk of neurological deterioration (RR = 0.80, P = .60). It is worth noting that stratified subgroup analysis found that strict control of SBP within ≤140 mmHg significantly increased the risk of mortality by 32% (RR = 1.32, 95% CI [1.02, 1.70]; P = .03), and there was no significant benefit in functional recovery (RR = 0.87, P = .13) or ICH risk (RR = 0.87, P = .13).
Patients with favorable functional recovery (mRS ≤ 2) exhibited significantly lower postoperative SBP compared to those with poor recovery, with an MD of −4.46 mmHg (95% CI [−5.74, −3.18]; P < .00001), indicating a strong inverse association between BP reduction and functional improvement. Patients who developed ICH had significantly higher postoperative SBP than those without ICH, with an MD of +6.55 mmHg (95% CI [3.22, 9.89]; P = .0001), underscoring elevated BP as a critical risk factor for hemorrhagic complications. Nonsurvivors demonstrated higher postoperative SBP compared to survivors, with an MD of +3.12 mmHg (95% CI [0.82, 5.42]; P = .008). However, the clinical interpretation requires caution, as the CIs partially overlapped the null value.
Mechanism Insights and Clinical Implications
The guideline-recommended permissive BP threshold (systolic ≤180 mmHg) is more consistent with the pathophysiology following cerebral ischemia, as it helps maintain perfusion in vulnerable brain regions (Powers et al., 2019). By contrast, the intervention involving aggressive BP reduction may have failed to achieve the intended outcomes due to several mechanistic and implementation factors. Excessively intensive antihypertensive treatment can exacerbate hypoperfusion in the ischemic penumbra, counteract compensatory arteriolar dilation, and promote oxidative stress or blood–brain barrier disruption (Ghuman et al., 2019; Li et al., 2021). These effects may have been amplified if the dose or intensity of the intervention was too high, or if the duration of BP-lowering extended beyond the therapeutic window, thereby compromising cerebral collateral flow. In comparison, moderate hypertension—within a controlled range—supports collateral circulation, attenuates excessive free radical generation, and balances the benefits of reperfusion against potential injury (Maïer et al., 2021). Therefore, the lack of expected benefits in our study may be attributed to the regimen's excessive intensity or suboptimal timing relative to ischemic evolution.
Limitations
The inherent impracticality of blinding in BP intervention trials introduced performance bias risks, though this reflects real-world clinical practice limitations. While primary analyses demonstrated moderate consistency, subgroup analyses revealed substantial heterogeneity in functional recovery and neurological deterioration, potentially reflecting unmeasured confounding variables. Exclusion of non-Chinese/English studies may underrepresent regional practice variations, despite inclusion of multinational cohorts. Overreliance on mRS scores neglects subjective recovery aspects like cognitive function and quality of life, though we partially mitigated this through complementary safety endpoint analyses. The systematic integration of multiple clinical parameters and rigorous sensitivity testing strengthens the validity of our conclusions, while acknowledged limitations provide clear directives for future research prioritization.
Future Research Directions
The results of this study found that for patients undergoing thrombectomy surgery, the effects of postoperative strong and weak BP reduction on patient recovery were not significant. From the perspective of other complications, perhaps according to guideline standards, not controlling BP would be a better choice. However, the specific range of BP control and effective control methods still need to be explored, and more complete and detailed clinical trials are needed to verify. In the future, personalized and comprehensive BP control plans can be formulated to intervene in patients’ postoperative BP from multiple aspects such as medication, lifestyle, and nursing routines, thereby improving patients’ postoperative clinical prognosis. At the same time, the evaluation method for patient prognosis can also be further improved, supplemented with other evaluation criteria and evaluation methods other than mRS, and provide more inspiration for perioperative care, prognosis, and treatment of patients with acute ischemic stroke.
Implications for Practice
Intensive BP management increases nursing burden through frequent dose adjustments, prolonged intravenous access, and enhanced monitoring of adverse effects (Bress et al., 2024). From a pharmacodynamic perspective, potent agents such as nicardipine or urapidil may impair endothelial function, whereas adopting moderate BP thresholds helps reduce cumulative drug exposure and related complications (Qureshi et al., 2016). In clinical practice, nurses should avoid aggressive intravenous antihypertensive regimens to minimize risks of phlebitis and vascular injury (Kawada et al., 2016; Ye et al., 2022). It is also advisable to favor short-acting agents such as labetalol with appropriate hepatic and renal function monitoring, rather than chronic use of nephrotoxic or hepatotoxic oral antihypertensives (Chun et al., 2023; Sanusi et al., 2024). During continuous intravenous therapy, central venous access or regular rotation of peripheral sites should be prioritized to reduce local complications (Poletti et al., 2018). Beyond technical interventions, nurses should focus on overall clinical context rather than rigid numerical targets, avoiding overtreatment of transient BP fluctuations and placing greater emphasis on neurological assessments such as changes in consciousness or new focal deficits (Maïer, Brauner, et al., 2022; Parati et al., 2018).
Moving away from the “lower is better” paradigm toward individualized BP thresholds can effectively reduce iatrogenic harm and nursing workload while maintaining patient safety (Bress et al., 2024). This approach underscores the evolving role of nursing in evidence-based care—transitioning from passive order implementation to active, collaborative clinical decision-making that enhances both patient outcomes and healthcare efficiency (Friesen-Storms et al., 2015).
Conclusion
In conclusion, this meta-analysis indicates that intensive BP control does not significantly improve functional independence after acute ischemic stroke. The available evidence suggests that adopting individualized BP thresholds, rather than a universal intensive target, better balances the risks of hypoperfusion and hypertension-related injury, thereby optimizing overall neurological outcomes.
Footnotes
Acknowledgments
This study was completed in the Department of Neurosurgery at Beijing Chaoyang Hospital. We are particularly grateful to the staff in Neurosurgery Ward for their time and resources. We would like to thank the corresponding author, Wei Zhang, for her guidance during the research process. We would also like to thank Dr Liu (Yunpeng Liu) for his comments and suggestions on the writing of the paper. Special thanks go to Wei Zhang and Yunpeng Liu for their review of the manuscript and critical technical contribution to system development. In addition, we also appreciate Pengcheng Zuo’s guidance on the statistics section of the article.
Ethical Considerations
This article is a meta-analysis and no ethical review has been performed.
Author Contributions
Jingnan Chen and Ying Gao jointly completed the literature retrieval, quality assessment, and information extraction of the meta-analysis. Jingnan Chen is mainly responsible for the statistical analysis and article writing. Ying Gao reviewed the expertise within the article. Wei Zhang led the whole process of the research and provided expert guidance when necessary, acting as a third party to make decisions when disputes arose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Appendix

| Number | Title | First Author | Year of Publication | Abstract Link | Study Design | Population | Intervention | Comparison | Outcomes | Inclusion/Exclusion | Exclusion Reason |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Intensive blood pressure control after endovascular thrombectomy for acute ischemic stroke (ENCHANTED2/MT): A multicenter, open-label, blinded-endpoint, randomized controlled trial | Pengfei Yang | 2022 | https://pubmed.ncbi.nlm.nih.gov/36341753/ | RCT | Patients after endovascular thrombectomy for acute ischemic stroke | Postop SBP: <120 mmHg | Postop SBP: 140–180 mmHg | (1) 90-day functional recovery (2) ICH (3) 90-day mortality |
Inclusion | - |
Note. RCT = randomized controlled trial, SBP = systolic blood pressure, ICH = intracranial hemorrhage.
