Abstract
Introduction
The lack of local side effect profiles for vaccines could hinder vaccination uptake. Since all COVID-19 vaccines are brand-new medications, it is crucial to keep track of any safety related concern.
Objective
This study is aimed to investigate post-vaccination side effects of COVID-19 vaccines and associated factors in Bahir Dar city.
Method
An institutional-based cross-sectional study was conducted among vaccinated clients. A simple random and a systematic random sampling method were used to select the health facilities and the participants, respectively. Bi-variable and multivariable binary logistic regression analyses were executed with odds ratio at 95% confidence intervals at p < .05.
Results
A total of 72(17.4%) participants reported at least one side effect following vaccination. The proportion prevalence was higher after the first dose than that of the second dose and the difference was also found to be statistically significant. In the multivariable logistic regression analysis female participants (AOR = 3.39, 95% CI = 1.53, 7.52), participants with a history of regular medication use (AOR = 3.34, 95% CI = 1.52, 7.33), participants aged 55 and above (AOR = 2.93, 95% CI = 1.23, 7.01), and participants who had taken only the first dose (AOR = 14.81, 95% CI = 6.40, 34.31) were more likely to develop side effects for COVID 19 vaccination compared to their counterparts.
Conclusion
A significant number (17.4%) of participants reported at least one side effect following vaccination. Sex, medication, occupation, age, and type of vaccination dose were factors statistically associated with the reported side effects.
Introduction
Coronavirus disease (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has affected nearly every stage of human life after being spread throughout the whole world (Nicola et al., 2020). As of the 17th of August 2022, it had spread to nearly 600 million people, and caused roughly 6.5 million deaths (Worldometer, 2022). The pandemic also leads to significant public health and economic crisis (Begović, 2022; Kominfo, 2021). The world urgently needed safe and effective vaccines to safeguard vulnerable populations and restore people's lives to their original state given the disease's alarming rate of spread and the massive expense of relying entirely on non-pharmaceutical measures (Flaxman et al., 2020; Haidere et al., 2021). Vaccines are a secure and reliable method of lowering the number of diseases (Sultana et al., 2020). It is commonly agreed that vaccination has had the greatest positive impact on world health (Greenwood, 2014). Several vaccines have been created and tested in an astonishingly short period of time. Public immunization initiatives are now having success in numerous nations by lowering the frequency of new diseases. However, a sizeable segment of the population (estimated at over 20% globally) plans to resist immunization (Khubchandani et al., 2021; Razai et al., 2021; Robinson et al., 2021). The most promising approach to manage this pandemic crisis is the development and implementation of a vaccine in addition to preventive measures like mask use, hand washing, and social isolation as available options to control transmission (Lurie Nicole, Melanie Saville & Richard Hatchett, 2020). The mass immunization effort started in early December 2020 and as many distinct vaccinations across various platforms had been implemented in countries (WHO, 2021).
Literature Review
Over the past years, researchers have been challenged with various limitations to find a holistic and an effective therapeutic approach for the illness. Repurposing already-approved medications and developing novel medications and vaccinations were both part of the research for COVID-19 treatment. Currently, there are some COVID-19 vaccines that have received emergency authorization in a number of nations (Craven, 2021). However, there is a shortage of concrete database comprising compiled information related to the reporting and analysis of adverse effects following the COVID-19 vaccine, leaving knowledge about the adverse events to be dispersed (European Medicines Agency, 2021; Lee et al., 2021; Ramasamy et al., 2020). The United States Vaccine Adverse Event Reporting System (VAERS) received a report of adverse COVID-19 occurrences from the Centers for Disease Control and Prevention (CDC). This revealed that the majority of the adverse reactions to the COVID-19 vaccinations reported in the United States were minor and had relatively few severe reactions. Still, only VAERS cannot deliver the complete data on vaccine adverse effects worldwide as it is a national vaccine safety surveillance program; relies on people's reports of their experiences, and is not designed to determine if a vaccine caused or contributed to an adverse effects on such reports (CDC, 2021a).
All of the information currently available on the side effects of the COVID-19 vaccination has been published through company-funded studies and were overseen by outside third parties (CDC, 2021b). Based on an interim review of pooled data from four clinical trials conducted in the United Kingdom, Brazil, and South Africa, AstraZeneca's COVID-19 vaccine (ChAdOx1nCoV-19 Corona Virus Vaccine [Recombinant]) was shown to be generally safe. Most vaccine recipients (almost 2/3rd) experienced only minor side effects (Voysey et al., 2021). Early vaccination of the community still requires the necessary collection of a post-vaccination safety profile. The lack of local side effect profiles for vaccines could hinder vaccination uptake (Jarrett et al., 2015). Since all COVID-19 vaccines are brand-new medications, it is crucial to keep track of any negative side effects after vaccination. Additionally, it is crucial to produce a safety signal for COVID-19 vaccines at an early stage that can serve as a foundation for further research. Therefore, this study is aimed to assess any adverse effects following the COVID-19 vaccination among individuals who took the vaccine from health facilities in Bahir Dar city.
Methods
Study Design and Period
An institution-based cross-sectional quantitative study was conducted to determine the prevalence of side effects after COVID-19 vaccination among clients in Bahir Dar city from July 2 to July 30, 2022.
Research Questions
The study was conducted to unveil the following questions:
What is the prevalence of COVID-19 vaccine side effects among immunized clients of Bahir Dar city? What factors are associated with the occurrence of the side effects in those vaccinated clients?
Study Setting
The study was conducted among health facilities in Bahir Dar city, which is the capital city of Amhara region. Bahir Dar is located 560 km away from Addis Ababa, the capital city of Ethiopia. The city has seven hospitals and nine health centers with an estimated total population of 390,429 populations.
Sampling and Study Populations
All clients who have received COVID-19 vaccination in Bahir Dar city were considered as the source population. All clients who have received COVID-19 vaccination in the selected health facility in Bahir Dar city and available during the data collection period were taken as a study population.
Sample Size Determinations and Sampling Procedures
The study population of this study was all clients who have received COVID-19 vaccination in the selected health facility in Bahir Dar city, Amhara region. The sample size was determined based on the assumption of the single population proportion formula. Since there is no prior study conducted on side effects of COVID-19 vaccination, a single population proportion formula was used to determine the final sample size:
Inclusion/Exclusion Criteria
Inclusion criteria: all vaccinated clients at Bahir Dar city who are voluntary to participate.
Exclusion criteria: those who refuse to give informed consent, vaccinated out of Bahir Dar city.
Study Variables
Post-vaccination side effect of COVID-19 vaccines is the outcome variable. Independent variables are thematized as socio-demographic factors: age, gender, work experience, profession, educational level, comorbidity condition, known medical condition, regular medication use, known disease conditions, regimen and type of vaccine received by participants, type of first dose of vaccination, type of second dose of vaccination, feeding status during vaccination, reason for vaccination, source of information for COVID-19 vaccination, history of COVID-19 infection, and COVID-19 adverse reaction information were variables included as predictors of COVID-19 adverse reaction.
Data Collection Tools and Procedures
Data was collected from study participants using a pre-tested and structured self-administered questionnaire. The structured questionnaire used for this study was first prepared and translated (English to Amharic then English) for analysis to control the quality of data. The study consisted of 42 questions encompassing the following domains: Socio-demographic characteristics, comorbidity condition, vaccine dose status, participants’ feat of life during the pandemic after being vaccinated and post-immunization side effect prevalence. Data was collected by three pharmacists and one public health officer using telephone interview. Supervision was made by three pharmacy professionals.
Tool Validity and Reliability
The data collection tool was a pre-tested and structured questionnaire which was constructed through the participation of experts from various health specialties (public health, medical laboratory, pharmaceutics, pharmacognosy, social pharmacy, and health informatics). In order to assure the quality, validity, and reliability of the data collection process and the collected data, two days of training were given to the data collectors and supervisors on the objective of the study, data collection procedures, and data collecting tools. The completeness of the questionnaires was checked every day by the supervisors before the actual data collection. Pretest was conducted in Abay Health Center with a sample size of 43. The tool was developed by reviewing all the available literature, and the developed tool was tested for consistency, validity, and reliability using Cronbach’s alpha test. The test result was 0.801 which declares that the variables included in the analysis could effectively measure the outcome variable. Accordingly, the tool developed could predict the outcome variable.
Data Processing and Analysis
Data was checked for completeness, entered into Epi-data version 4.6, and exported to STATA version.14 for analysis. Descriptive statistics, frequencies, and percentages were computed and presented using graphs and tables. A bi-variable and multi-variable logistic regression model was used to assess factors associated with adverse reaction of COVID-19 vaccination. Variables with a p-value less than or equal to 0.2 was included to multi-variable logistic regression. Multicollinearity was checked among independent variables using a variance inflation factor and model fitness was checked using Hosmer-Lemeshow goodness of fit test. Finally, the results were reported as adjusted odds ratio (AOR) with their 95% CIs.
Result
Demographic Characteristics
A total of 413 participants completed the survey. About 70.94% of the respondents were females and 367(88.86%) were Christians by religion. The majority of the study participants (62.95%) were single by marital status and about one-third (33.41%) were diploma holders. About 3.87% of the participants had reported a known history of medication allergy. Of 413 participants, 334 (80.87%) had at least one co-morbid condition where diabetes mellitus 66(15.98%) followed by, respiratory diseases 40(11.98%), and HIV/AIDS 5(1.58%) were the most common comorbid conditions. Two-thirds (66.83%) of the respondents reported regularly taking medications to treat their underlying medical conditions (Table 1).
Sociodemographic Characteristics of Participants (n = 413).

More than half (53.75%) of the participants received only the first dose of COVID-19 vaccine while 46.25% had completed a double dose vaccine. Three-fourth of the total participants (76.3%) took AstraZeneca as the first dose of vaccination. Two-third (66.83%) of the participants believed that COVID-19 vaccines are safe and effective. About 82.43% of clients that took the first dose responded as they are willing to take additional/booster dose. Majority (90.56%) of the participants had no history of COVID-19 infection prior to taking the vaccination/s (Table 2).
Regimen and Type of Vaccine Received by Participants (n = 413).

Reported Side Effects After COVID-19 Vaccination
A total of 72(17.4%) participants reported at least one side effect following vaccination (Figure 1). Among the 72 participants that reported the side effects after vaccination, 76.4% of them experienced the side effects during the first dose, 18% encountered at the second dose, and 5.6% experienced both during the first and second vaccination (Figure 2). The proportion of the side effects reported by the participants was higher after the first dose than that of the second dose and the difference was also found to be statistically significant (p = .000). The most commonly reported side effect by the participants was injection site pain (26.4%) followed by headache (20.8%) and, high grade fever (11.1%) as presented in Figure 3. No severe cases or death were reported following vaccination among the study participants.

Prevalence (in percentage) of side effects after COVID-19 vaccination among study participants.

Percentage of side effect reports at dose levels of COVID-19 vaccination among study participants.

Percentage of reported side effects after COVID-19 vaccination among study participants.
Timing of the Side Effects and Interventions Taken
Majority of the participants experienced the side effects within 2–6 h of post-vaccination and in 62.5% of the cases the reaction lasted for 2 days (Table 3). Majority of the participants (86.11%) took pain killers to alleviate the symptoms. Only four participants got admitted to a hospital for a close follow-up and all of them recovered without any major consequence. Prescribed tramadol (50 mg) followed by paracetamol (500 mg) and diclofenac (50 mg) were taken by the majority of the participants to alleviate the side effects.
Time to Develop Reported Side Effects and Measurements Taken to Alleviate (n = 72).

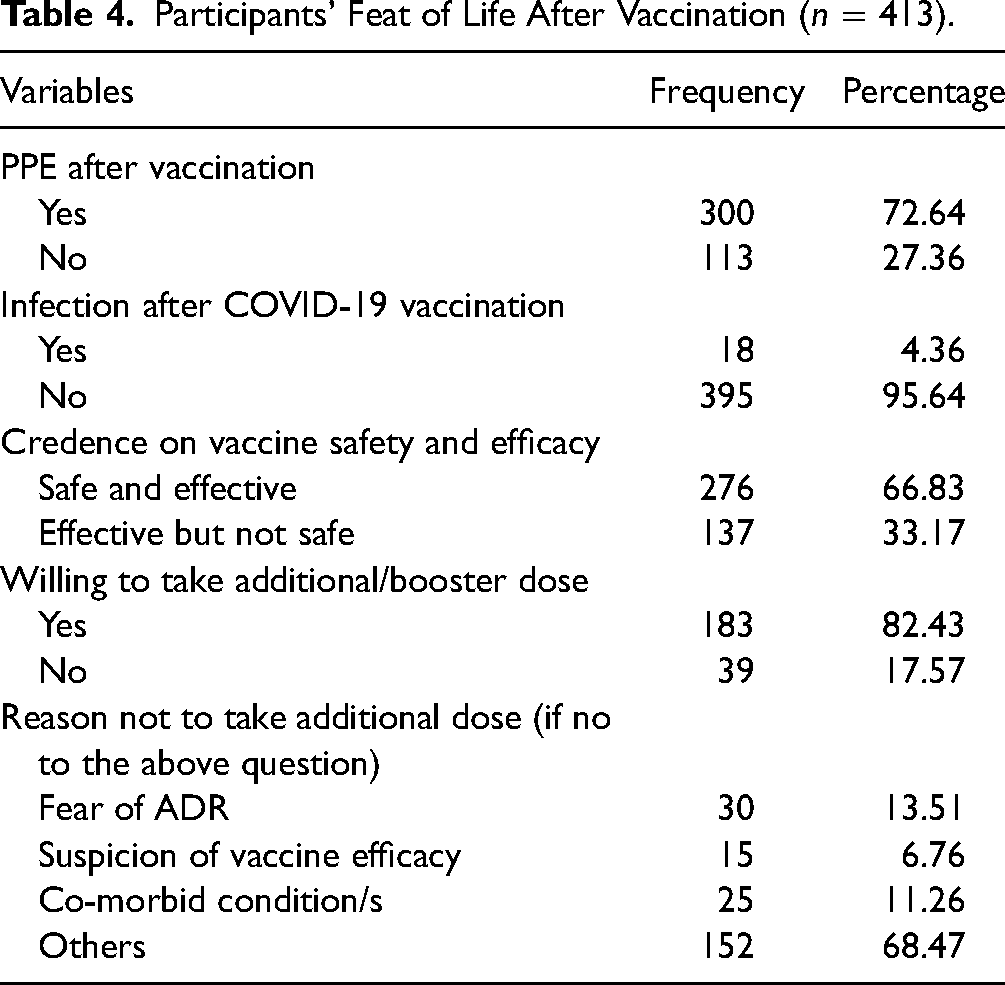
Almost three-fourth (72.64%) of the respondents continued to use personal protective equipment's after being vaccinated. But, only 4.36% of them were diagnosed for COVID-19 post-vaccination. Around 17.57% were reluctant to continue/take additional/booster dose of the vaccine. Fear of post-vaccine side effects and associated complications were the primary reasons for the reported hesitancy (Table 4).
Participants’ Feat of Life After Vaccination (n = 413).
Factors Associated With Incidence of Post-Vaccination Side Effects
The bivariate logistic regression was used to determine the association between the dependent variable, post-vaccination side effects, with different independent variable found in the table (Table 5). Sex, medication, occupation, age, and type of vaccination dose were the statistically associated factors. Accordingly, female participants were 3.39 (AOR = 3.39, 95% CI = 1.53, 7.52) times more likely to develop side effects for COVID-19 vaccination compared to male participants. Likewise, participants who had a history of regular medication use were 3.34 (AOR = 3.34, 95% CI = 1.52, 7.33) times more likely to develop side effects for COVID-19 vaccination compared to those who have no medication use history. Participants who were aged 55 and above were 2.93 (AOR = 2.93, 95% CI = 1.23, 7.01) times more likely to develop side effects for COVID-19 vaccination compared to those aged less than 35 years old. On the other hand, participants who had taken only the first dose of COVID-19 vaccination were 14.81 (AOR = 14.81, 95% CI = 6.40, 34.31) times more likely to develop side effects for COVID-19 vaccination compared to participants who had taken both doses of the vaccine.
Factors Associated With Incidence of COVID-19 Vaccine Side Effects.

Discussion
Enormous adverse effects occur in health care institutions that induce negative impact on a patient's quality of life, frequently leading to significant morbidity and mortality. Such adverse effects may cause the patients to develop mistrust or negative emotion toward their physicians and seek self-treatment options (O’Donnell, 1994). In the present study, the side effects and associated factors following COVID-19 vaccination among individuals who took the vaccine from health facilities in Bahir Dar city have been investigated. The most reported side effects were injection site pain followed by headache and high grade fever. This report is similar with the symptoms reported by Solomon et al. (2021) and Alemayehu et al. (2022). Adverse effects may occur at any time period after administration. Local intense injection site reactions occur in some vaccines immediately after injection. The severity of the reported side effects was mild to moderate, short-lived, and self-limiting in the majority of cases which is consistent to results from other studies in Kabul (Tequare et al., 2021) and Korea (Azimi et al., 2021; Jeon et al., 2021). Majority of the symptoms lasted for 24−48 h and of those clients, majority of them took pain killers to alleviate the occurred adverse events (Solomon et al., 2021). Life threatening or disabling adverse effects were not experienced in this study. To alleviate the symptoms most of the participants took tramadol. For mildest events, home interventions such as wet cloth compression, simple exercises such as arm moving, drinking home-made fluids and some traditional plants may be taken.
In this study, age group, gender, type of dose, and medication history were found to be the associated factors for the reported effects. Side effects were more observed in female participants than males. This was congruent with a study conducted in Turkey (Riad et al., 2021), Saudi Arabia (Alghamdi et al., 2021), Bangladesh (Sultana et al., 2021), and India (Chakraborty et al., 2022). On the other hand, this finding was discordant with a study conducted in China (Xiong et al., 2021) and Ethiopia (Alemayehu et al., 2022). This gender difference may be explained by major sex hormones exhibiting opposing effects on the adaptive and innate immune system with elevated estradiol led to more robust antibody responses, and testosterone decreasing the vaccine-induced antibody responses (Giefing-Kröll et al., 2015; Kleina et al., 2014).
Similarly, the occurrence of the COVID-19 vaccination side effects was significantly higher in age groups greater than 55 years which was consistent with other findings (Xiong et al., 2021). However, similar studies revealed that COVID-19 vaccine adverse effect is more observed in younger ages (Ramasamy et al., 2020; Tequare et al., 2021). These discrepancies may be due to higher reactogenicity among young people than their older counterparts (Hervé et al., 2019). Hence, COVID-19 vaccination adverse effects might occur with different category of age group whether in younger or older individuals.
Beyond gender and age disparity, other significant predictors of post-vaccination effects in our study were doses of vaccine and history of a regular medication use. A higher incidence of reactogenicity was reported in participants with a history of regular medication use. Among the side effects reported under this study, the prevalence was 76.4% at the first dose and 18% at the second dose. This result was concordant to a study done by Parida et al. (2022); they have reported 38.1% and 26.6% of the adverse drug event among first and second-dose beneficiaries, respectively. In the present study, 17.57% of the respondents hesitate to take the booster dose. Fear of post-vaccine adverse events and developing severe complications were the primary reasons for COVID-19 vaccine hesitancy. This finding was in line with a study conducted in Ghana where anxiety about vaccine side effects was the primary reason for vaccine refusal (Yigit et al., 2021).
Conclusion
About 17.4% of the participants reported at least one side effect following COVID-19 vaccination. Injection site pain is among the most commonly encountered side effects. Sex, medication, occupation, age, and type of vaccination dose were the statistically associated factors.
Strengths and Limitations of the Study
The study tried to uncover the currently sensitive health issue by directly contacting the vaccinated clients with a timely, cost effective, generalizable, and reliability approach. As a limitation, the survey faced shortage of previous research studies on the same topic and lack of potential depth as it is conducted only in one city due to resources scarcity.
Implications for Practice
Spontaneous reporting of adverse event experiences from newer vaccines is particularly useful in identifying rare or delayed reactions that are not recognized in clinical trials. The study report can act as an early warning to help the regulatory body monitor vaccine safety. Community health workers may use the report to provide better medical care in local areas. Further safety data from a larger sample size and of longer duration are warranted to establish safety to the general population.
Footnotes
Acknowledgment
The authors cordially acknowledge the health facilities participated in this study.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Clearance
Ethical clearance was obtained from the ethics committee of Department of Pharmacy, College of Medicine and Health Sciences, Bahir Dar University (reference letter no. 01/28/10/2013). Oral consent was obtained from each study participant after telling the objective of the study. Participants were informed about the benefits and risks of the study. The data collection procedure was anonymous and their privacy was also kept.

