Abstract
Late-onset Pompe disease (LOPD) is a rare neuromuscular disorder, where delays in diagnosis can result in inconsistent patient management and suboptimal use of effective treatments, thereby increasing patient burden. We conducted an exploratory, cross-sectional survey of 20 adults with LOPD in the UK and USA, recruited via patient advocacy groups and vetted databases. Our results showed that patients with LOPD experienced substantial delays from symptom onset to diagnosis. Interactions with healthcare professionals (HCPs) were inconsistent, and LOPD awareness among HCPs was insufficient. Patients were generally satisfied with their treatment, with 90% of patients reporting mild to no discomfort with enzyme replacement therapy or other treatments. However, 60% of patients implemented various nonpharmacological therapies to manage pain or other aspects of LOPD. These exploratory findings highlight persistent gaps in diagnosis and care, emphasizing the need for improved HCP education and integrated support services.
Plain Language Title
Plain Language Summary
Late-onset Pompe disease (LOPD) is a rare condition that affects muscles and nerves. This leads to symptoms such as muscle weakness, trouble breathing, and pain, and patients with this disease often have shorter life expectancies. While treatments exist, there are often significant delays in diagnosis and challenges in daily life. In our survey of 20 adults living with LOPD in the UK and USA, most participants waited years for a correct diagnosis and frequently experienced misdiagnosis and limited awareness of LOPD among healthcare professionals (HCPs). While most patients were satisfied with their enzyme replacement therapy, many reported ongoing pain and a need for better psychological support. Many highlighted the importance of improved communication with HCPs, easier access to information and support groups, and more opportunities to participate in research. These findings show that earlier recognition, coordinated care, and patient-focused support services are essential for improving the lives of people with LOPD.
Keywords
Introduction
Late-onset Pompe disease (LOPD) is a rare, progressive neuromuscular disorder that affects daily life and healthcare interactions. While clinical features and treatments are well documented, patient experiences with diagnosis and long-term care are less explored. These perspectives can inform communication strategies, streamline referral pathways, and shape support services.
Despite available therapies and guidelines, patients often face prolonged diagnostic journeys and inconsistent care.1–11 Previous studies focus on clinical outcomes—little is known about patient experiences with diagnosis, treatment, and healthcare systems.2–5 Misdiagnosis is common before LOPD is recognized, compounding morbidity and reducing quality of life (QoL).6–10 Enzyme replacement therapy (ERT) improves mobility and respiratory function, but QoL gains vary, highlighting the need to minimize delay and optimize treatment.11–13
Our study provides preliminary insights into diagnostic delays, treatment satisfaction, and unmet needs to inform patient-centered care.
Methods
We performed a cross-sectional online survey of 20 adults (≥18 years) with self-reported LOPD residing in the UK or USA (UK, n = 10; USA, n = 10). Sample size was determined by budgetary constraints and the exploratory nature of the study; future research will target larger cohorts.
Selection Process and Sample Size
Participants were recruited through patient advocacy groups in the UK and vetted patient databases in the USA. Eligibility was confirmed via phone screening. All participants provided electronic informed consent.
Survey Instrument
The survey instrument was developed through a literature review and expert input to ensure content validity. It included structured and open-ended questions covering demographics, diagnostic history, treatment experiences, pain management, healthcare interactions, and informational needs. Complete details of the survey instrument are in the Online Appendix.
Data Collection and Analysis
Responses were collected over 4 weeks via a secure platform managed by Global Perspectives and its approved partners. Participation was voluntary with electronic informed consent; data were anonymized and handled in accordance with the General Data Protection Regulation (for UK participants) and applicable US privacy standards. Descriptive statistics summarized categorical variables as counts and percentages and continuous variables as mean ± standard deviation (SD) (with medians where relevant). No inferential comparisons were planned, given the small sample size and exploratory aim.
Results
Respondent Demographics
The survey was completed by 20 participants, balanced between location and sex (50% UK, 50% USA; 50% male, 50% female). Average age was 47.7 ± 12.7 years (median, 46 years; range, 31–71 years). Involvement in patient advocacy was reported by 60% of participants. Educational attainment was distributed across high school (35%), university (30%), and postgraduate/higher (35%). Income and further demographics are summarized in Table 1.
Demographics and Responses of Patients Surveyed (N = 20) for This Study.

Abbreviations: ERT, enzyme replacement therapy; GP, general practitioner; HCP, healthcare professional; LOPD, late-onset Pompe disease.
*Questions for which it was possible to select multiple options.
Diagnosis and Treatment
Delays from symptom onset to diagnosis were substantial. Using age-cohort bins for symptom onset (childhood, adolescence, and early/middle/late adulthood), the best-case average delay exceeded 9 years (median, 8 years). Nearly 25% of respondents reported 10–20 years from first symptoms to diagnosis, and 15% reported >20 years. Average age at diagnosis was 36.3 ± 13.8 years (median, 30 years; range, 19–63 years). Delays were similar across age groups: the younger half (<46 years) averaged 8.7 years from symptom onset to diagnosis versus 9.7 years in the older half (≥46 years). Figure 1 depicts age at symptom onset and reported age at diagnosis. One participant described an “11-year diagnosis journey full of misdiagnosis,” adding, “Having to educate every single GP that I see about Pompe disease.” These narratives underscore the systemic gaps in awareness and referral pathways.
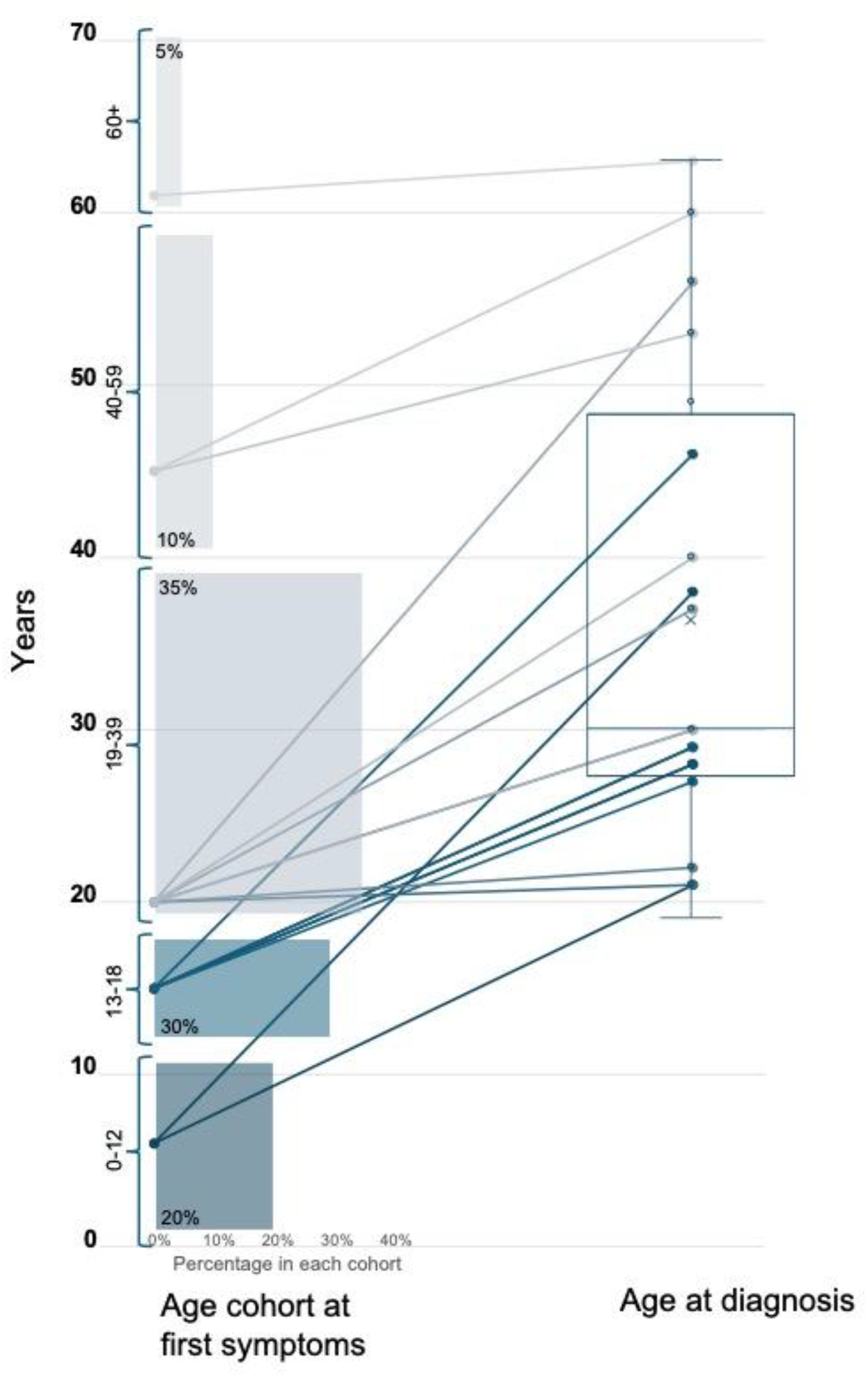
Age cohort of symptom onset with percentages of respondents in each (childhood, 0–12 years; adolescence, 13–18 years; young adulthood, 19–29 years; middle adulthood, 30–59 years; and late adulthood, 60+ years) and reported age of LOPD diagnosis, with per-patient delays to diagnosis (straight lines) and box and whisker plot indicating range, quartiles, median, and mean of reported age at diagnosis data.
Impact on Daily Life, Breathing, and Pain
Participants reported pronounced functional burden despite access to treatment, with 55% describing a moderate impact of LOPD on daily life, and 45% severe impact. Breathing difficulties were common: 95% reported moderate to severe respiratory impact. Pain was also frequent, with 65% reporting moderate to severe pain levels.
Healthcare Provider Awareness and Misdiagnosis
All respondents highlighted limited awareness of LOPD among healthcare professionals (HCPs). Before diagnosis, participants consulted an average of 4.6 ± 2.8 clinicians (median, 3.5; range, 1–11). Misdiagnosis was reported by 75% of respondents. Constraints to specialist access (65%) and treatment delays (35%) were additional barriers. Respondents reported frustration with specialists, noting: Neurologists that should be aware of new treatment options having no idea what they are. Doctors not having any clue what LOPD is. Doctors that have horrible people skills. Doctors that may be burnt out or not up to date on appropriate medical support (pulmonary, primarily).
Treatment Patterns, Discomfort, and Satisfaction
ERT was the most common therapy (90%), except for 2 participants who were not using it. Both were in the lowest income bands and reported severe daily life impact, but other outcomes were similar. Despite widespread ERT use, challenges with access and insurance compliance were noted: Needing to educate each care provider about the disease is tiring and frustrating. Because of the cost of treatment, compliance of insurance companies can be difficult.
This reflects systemic barriers to continuity of care.
Combination therapy was reported by 15%, painkillers by 55%, and physical therapy by 25%; other supports included hydrotherapy and ventilation. Before the current regimen, participants had been prescribed an average of 2.6 ± 2.4 medications (median, 2; range, 1–11). Treatment-related discomfort was generally low: 50% reported none, 40% mild, and 10% moderate. Overall, 70% were satisfied with the treatment.
Pain management remained an unmet need: 25% rated it very effective; 55% somewhat effective; and 20% ineffective. Management approaches included pharmacological treatments (60%), lifestyle changes (70%), physical therapy (40%), and devices (20%); hydrotherapy was also cited.
The most common treatment-related challenges were low HCP awareness (100%), misdiagnosis (75%), limited specialist availability (65%), and treatment delays (35%). Regarding living circumstances, 80% lived with a partner and 20% alone. Caregiver assistance averaged 14.0 ± 16.9 h per week, provided by partners (55%), relatives (35%), friends (15%), adult children (15%), nurses (25%), paid caregivers (20%), and others (10%).
HCP Communication
HCP communication experiences were mixed: 40% rated communication as very effective and 40% as somewhat effective; 10% judged it mostly ineffective, and 10% completely ineffective. Confidence in provider understanding of LOPD varied: 30% felt confident, 20% felt HCPs understood most of the time, 25% half the time, 20% less than half, and 5% reported no HCP understanding.
Psychological Support and Information Needs
Uncertainty about disease progression emerged as a major psychological burden. One participant shared: The hardest part is not knowing the future, the “unknowns,” which is the hardest psychologically. Sometimes I avoid doing activities due to anxiety of dealing with Pompe symptoms, or the inability to do things/keep up with “normal” people.
Psychological support was often insufficient: 20% rated support as very adequate, 40% somewhat adequate, 25% not very satisfactory, and 15% not acceptable. These findings emphasize the need for counseling and mental health support as part of comprehensive care.
To stay informed, respondents used healthcare specialists (75%), search engines (70%), medical websites (70%), social media (65%), general practitioners/primary physicians (10%), and other sources such as support groups/charity events (20%). While 20% felt they knew enough, 75% wanted to learn more, and 1 respondent lacked confidence in their knowledge. Explaining LOPD to others felt easy for 50%, mostly easy for 40%, and difficult for 10%. Patients expressed a strong desire for better access to research opportunities and clinical trials: I would like to be more informed on future trials and how to access them.
This suggests a need for structured communication between clinicians, researchers, and patients to support informed decision-making.
Discussion
Our survey highlights persistent diagnostic delays, frequent misdiagnoses, and gaps in provider awareness for adults with LOPD. Diagnostic delays averaging over 9 years and frequent misdiagnoses emphasize the need for clearer referral pathways and improved awareness among primary care and neurology providers. 14 Findings underscore the importance of multidisciplinary care, HCP education, and structured communication to support timely diagnosis and holistic management. Limited understanding of LOPD often left patients feeling isolated and uncertain, reinforcing the need for proactive communication and educational resources. Most patients were satisfied with ERT; however, pain and psychological support remain unmet needs. Anxiety around ERT—including concerns about efficacy and long-term outcomes—suggests opportunities for counseling and peer support programs.11,13 These insights should guide system-level improvements for timely diagnosis, transparent communication, and support services that prioritize patient experience alongside clinical outcomes. Overall, findings align with prior reports, emphasizing the need for earlier recognition, coordinated multidisciplinary care, and targeted psychosocial support to reduce disease burden and improve QoL.9–13
Limitations
This study is limited by its small sample size (n = 20) and restriction to 2 English-speaking countries with distinct healthcare systems, reducing generalizability. Experiences in other regions or demographic groups may differ. Some survey items were subjective, relying on participants’ perceptions of healthcare interactions and HCP knowledge. Additionally, 60% of respondents reported involvement in patient advocacy, which may bias responses toward individuals with greater disease awareness and engagement in care. Despite constraints, findings are consistent with prior literature and underscore persistent challenges, including diagnostic delay, disease burden, and variable effectiveness of current therapies to improve QoL.
Conclusion
Despite treatment advances, adults with LOPD continue to face diagnostic delays and care gaps. Efforts to enhance HCP education, streamline referral pathways, and provide integrated support services are needed to improve patient outcomes.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735261433788 - Supplemental material for Patient Perspectives on Late-Onset Pompe Disease: Insights From a 2025 Patient Snapshot Survey on Diagnosis, Treatment, and Quality of Life
Supplemental material, sj-docx-1-jpx-10.1177_23743735261433788 for Patient Perspectives on Late-Onset Pompe Disease: Insights From a 2025 Patient Snapshot Survey on Diagnosis, Treatment, and Quality of Life by Benjamin W. Gallarda, Ross A. Leslie, Felicia Oyedepo, Claire Almeida, Kevin Ampeh, Guiomar Gonzalez and Benedikt Schoser in Journal of Patient Experience
Footnotes
Acknowledgments
The authors thank Sydney Hooley, Joe Acres, Claire Aukim-Hastie, Eleanor McDermid, Laura Boyd, and Litha Abigail Mfiki, members of the teams of Global Perspectives, an IQVIA business, and EPG Health, an IQVIA business, who participated in managing this research and preparing this manuscript. Parts of these original data were presented by Professor Benedikt Schoser, LMU Munich, Germany, Dr Priya Sunil Kishnani, Duke University Medical Center, USA, and Dr Tahseen Mozaffar, University of California, Irvine, USA, in a satellite symposium on Wednesday, February 5, 2025, titled LOPD: Elevating Care Through Patient Insights at WORLDSymposium 2025 in San Diego, CA, USA. Initial literature review, data analysis, and manuscript drafting were aided by Scite.ai and Microsoft Copilot based on the survey responses, other raw data, summary statistics, and symposium presentations, but final figures, statistics, and manuscript wording were rewritten, reviewed, revised, completed, and approved by the listed authors.
Author Contributions
RAL, FO, and BWG drafted, reviewed, and approved the patient survey; CA, KA, and GG recruited survey participants, conducted the survey, and created the dataset of responses; BWG analyzed the data and drafted the manuscript; BS reviewed the analyzed data; all authors reviewed and commented on the manuscript.
Data Sharing
Survey response data available upon request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The listed authors from EPG Health and Global Perspectives are all employees of IQVIA but have no further conflicts of interest to declare. Benedikt Schoser has received fees and/or honoraria from Alexion, AMDA Foundation USA, Amicus Therapeutics, Argenx, Arthex, Astellas Pharma, Avidity, DFG Caesar, Denali, EU Horizon 2022 DM-Entry, Dyne, EU Horizon 2022 COMPASS, Johnson & Johnson, Kedrion Biopharma, Marigold Foundation CA, Maze Therapeutics, EU Horizon 2022 PaLaDin, Sanofi, Shionogi, and UCB.
Ethics Approval and Patient Consent Statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was conducted independently by EPG Health as part of an unrestricted educational grant from Amicus Therapeutics. The sponsor was not involved in the study design, survey instrument development, data collection, analysis, interpretation of results, or manuscript preparation. All authors had full access to the anonymized data and maintained complete independence in the interpretation and reporting of the study findings.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
